Abstract
Objective
In the current work, we investigated and assessed the ability of the bioactive flavone linarin (LIN) to prevent asthma in the ovalbumin (OVA)-induced rat model.
Methods
The experimental rats’ body weight and relative lung weight were measured after each treatment. Indices of oxidative stress in lung tissue and erythrocytes were assessed. Analyses were conducted on hematological assays, measurement of histamine release in BALF fluid, and inflammatory markers found in blood and BALF, including IL-4, IL-13, TNF-α, IFN-γ, and IgE levels. For histological examination, tissue samples were collected.
Results
Administration of LIN significantly increased the rats’ body weight; however, their relative lung weight decreased. According to our findings, LIN significantly raised SOD, GSH, and CAT levels while suppressing MDA content. The administration of LIN resulted in a considerable reduction in IgE levels, cytokines, and hematological assay influx in the corresponding samples. Histological assessment showed that LIN treatment significantly maintained the anatomy of the lungs and decreased the infiltration of inflammatory cells.
Conclusion
The regulation of oxidative stress and the avoidance of pulmonary airway inflammation are two proposed methods by which LIN may reduce asthma symptoms.

Introduction
The disease known as asthma is widely acknowledged to be characterized by varying degrees of persistent inflammation and structural changes to the airways. 1 This disease affects a sizable portion of the global population in numerous nations. The increasing severity of environmental variables and physiological factors has led to a rise in the frequency of this heteronomous disease, particularly in youngsters, in recent years. 2 Several factors, such as dust, cold air, chemicals, allergens, and respiratory illnesses, can trigger bronchial asthma. These stimuli cause inflammatory cells to become activated, which in turn release cell-derived mediators into the lumen of the bronchi, including lymphocytes, eosinophils, interleukins, neutrophils, IgE, and histamine. Reactive nitrogen and oxygen species are also produced in greater amounts as a result of this process. 3 Asthma symptoms such as frequent coughing, wheezing, dyspnea, and chest tightness cause lung airway impairment and decreased oxygen delivery to the alveoli. An infected person’s regular routines are disrupted, which may exacerbate their illness and increase their risk of death. Men are more prone to get asthma as children, whereas women develop it later in life and throughout adolescence. Moreover, women of childbearing age frequently suffer from allergic illnesses. 4
Inflammatory cells generate mediators that play an important role in asthma and pulmonary inflammation. The lining fluid of the lung is protected against oxidants by high amounts of antioxidants. An imbalance in the antioxidant/oxidant ratio in asthma indicates ongoing inflammation. Reports have highlighted a notable increase in activity associated with asthma. Furthermore, the release of IL-4 and IL-13 by cells aids in the development of allergic inflammatory disorders.5,6 The concept of oxidative stress is derived from the type I hypersensitivity reaction, in which ROS production and secretion by inflammatory cells promote airway inflammation. These ROS have been implicated in the usual bronchial blockage associated with asthma, as well as lung damage and spasms of the bronchial muscles. Thus, lesions at the cellular level of the organism result from the disparity between the mechanisms for generating free radicals and those for defense. Uncertainty exists regarding the cellular and molecular processes underlying the remodeling of the airways in asthmatic patients. It is well-recognized that the lung inflammation associated with long-term asthma episodes is caused by inflammatory cytokines. One potential strategy to stop airway remodeling and the onset of asthma is to focus on modulating cytokines and preventing associated inflammatory cells from penetrating the lung airway.
Despite being widely used for managing asthma, conventional anti-asthmatic medications only provide symptomatic relief and have a higher frequency of systemic and local adverse effects. 3 Numerous economical and easily accessible plant resources have come to light due to the increased interest in natural antioxidants as a substitute for manufactured products and their unfavorable side effects. It is important to study their biological and phytochemical properties, which have led to the discovery of compounds responsible for their bioactivities. Phytoconstituents have drawn the attention of researchers for their health advantages. Numerous phytochemicals are being effectively employed as medications to treat various ailments, including cancer.
Flavonoids, the most abundant natural phenolic compounds, have diverse biological properties and are divided into six subclasses. Despite ongoing studies, they remain intriguing molecules for exploration.
7
This study investigates the anti-inflammatory and antioxidant characteristics of one such flavonoid, linarin, for the treatment of asthma. The Asteraceae and Lamiaceae groups comprise the majority of plant species from which linarin, a glycosylated flavone, has been found. In general, LIN is currently a somewhat understudied medication source. With strong biocompatibility, luteolin, 3,5-dicaffeoylquinic acid, and LIN may be major factors in
This research offers scientific evidence for the traditional usage of LIN as an anti-asthmatic. It investigated how the conventional extract affected an experimental asthma model in rats. We can better comprehend this condition, assess the impact of oxidative stress, and investigate the potential therapeutic benefit of this compound by using the rat model of OVA-stimulated asthma. Utilizing animal models to study asthma is in practice due to ethical limitations in clinical studies. The availability of transgenic animals and a well-defined immune system are two benefits associated with rats. Ovalbumin-sensitized mice and rats are frequently employed as asthma models, showing similar features to human asthma. 9 A common allergen that causes laboratory rodents to experience severe allergic lung inflammation is ovalbumin (OVA), which is generated from chicken eggs. Some similarities between this animal model and the human form of allergic asthma include airway hyperresponsiveness in response to antigen exposure, the production of inflammatory mediators and cytokines, and the presence of eosinophilic lung inflammation.5-11
To assess the preventive impact of LIN and guide future research, we employed the ovalbumin (OVA)-induced asthmatic rat model in this work. In serum and BAL fluid, total and distinct counts of inflammatory cells were carried out to monitor the development of inflammation and evaluate the effects of LIN. The study also focuses on linarin’s antioxidant properties, which may lessen oxidative stress and may even exacerbate bronchial inflammation.
Data and Methods
Materials
Sigma-Aldrich (USA) supplied linarin and dexamethasone. TNFα, IL-4, IFN-γ, and IL-13 ELISA kits were bought from R&D Systems (Minneapolis, MN). Before the study began, all chemicals, reagents, kits, and equipment were acquired commercially and made available.
Experimental Animals
All applicable national regulations and institutional policies for animal use and care were followed. The rats were acquired from an approved animal center with appropriate ethical approval for the animal experimentation study. Male Wistar rats were used for the investigation. Animals were kept in polycarbonate cages under 12-hour light-dark cycles at a controlled ambient temperature of 23°C–25°C with 55-60% relative humidity. They had free access to drinking water and regular rat meal pellets. The experiment (in vivo study) lasted 29 days (2024/01) at the First People’s Hospital in Xianyang in Shaanxi Province, China. The animals were separated into four groups of six rats each (the sample size was calculated using the G*Power tool version 3.1.9.6, with a power of 95% and an alpha error probability of 0.05).
OVA Sensitization and Drug Administration
The first group served as the saline-sensitized normal control. The second group acted as the asthma control (exposed to OVA). The third group consisted of OVA-stimulated rats administered with 50 mg/kg of LIN. The fourth group, a positive control, included OVA-exposed rats treated with 3 mg/kg body weight (bw) of dexamethasone (DEX).
On days 0 and 8, all animals except those in the normal control group received an intraperitoneal (i.p.) injection of OVA (1 mg/kg) and aluminum hydroxide (100 mg in 1 mL saline). From day 15, these animals were exposed to 1% OVA aerosol in saline for 20 minutes each day for 14 days via an ultrasonic nebulizer linked to the cages. Group III animals were given LIN orally at 50 mg/kg b.w. one hour after the OVA challenge, once daily for 14 days. Group IV animals were given 3 mg/kg b.w. of dexamethasone orally one hour after the OVA challenge, once daily for 14 days.
On day 29, all animals were slaughtered to remove blood and lung tissue for biochemical and histological studies. The culture was centrifuged, and the supernatant was kept at −80°C for further study. A portion of the lung tissue was preserved in 10% neutral buffered formalin before histological evaluation. 2
Linarin on Body Weight and Relative Lung Weight Changes Induced by OVA
Rats’ body weight was monitored following their initial OVA sensitization. The rats were then sacrificed, and the weight of their left lung was recorded. 9
Hematological Assay
After evaluating lung function measurements, the lungs were lavaged three times with cannulated tracheal tubes filled with 5.0 mL aliquots of standard saline solution. The BALF was centrifuged at 1500 rpm for 10 minutes at 4°C. The cell pellet was resuspended in 1.0 mL of normal saline to count total and differential leukocytes. Following BALF collection, blood was obtained via heart puncture. An aliquot of heparinized blood was stored at 4°C to determine the leukocyte count. Within 30 minutes following blood collection, total and differential leukocyte counts were determined using a hemocytometer using a previously established method. Leishman’s stain was used to stain cytospin smears for calculating the differential cell count. 3
Measurement of Histamine Release in Bronchoalveolar Lavage Fluid
Ovalbumin was infused throughout the day for the histamine assay, and BAL fluid was collected 30 minutes after the final instillation. The colorimetric method was used to determine the histamine concentration in the BAL fluid. Briefly, 0.5 mL of the sample was combined with 0.1 mL of 1% sulfanilic acid and 5% sodium nitrite aqueous solution and incubated for 10 minutes. After incubation, 1.3 mL of 5% aqueous sodium carbonate and 1 mL of 75% ethanol were added, two minutes apart. Absorbance was measured at 530 nm within 20 minutes. 1
Biochemical Assays in Lung Tissues and Erythrocytes
Lung tissue was homogenized in phosphate-buffered saline and centrifuged at 10 000 g for 15 minutes at 4°C. The antioxidant enzymes SOD, CAT, MDA, and GSH were evaluated in the supernatants. Aliquots were kept at −20°C. Erythrocytes obtained from blood samples were used for the same analysis. All assays were performed on both erythrocytes and lung tissues. Each assay was carried out colorimetrically using commercially available kits and following the manufacturer’s instructions. 12
Effect of LIN on Inflammatory Markers
Serum levels of inflammatory cytokines (IFN-γ, TNF-α, IL-4, and IL-13) were evaluated using commercially available ELISA kits, following manufacturer recommendations.
Linarin’s Effect on IgE Levels in Rats with OVA-Induced Asthma
Blood samples (2.5-3.0 mL) were taken in non-heparinized tubes after cardiac puncture. To determine IgE levels, serum was centrifuged at 3000 rpm for 10 minutes and stored at −80°C. Serum IgE levels were measured using commercially available ELISA kits, following the manufacturer’s instructions.
Pathological Evaluation
Lung tissues were sectioned for histological examination. The samples were stored in 10% formalin solution, dehydrated using a graded ethanol series, and embedded in paraffin. The sections were 4 µm thick and stained with hematoxylin and eosin (H&E). The stained sections were viewed and photographed using a light microscope at 400× magnification. 13
Statistical Analysis
Data are presented as mean ± SEM from three independent experiments. Statistical analyses were carried out using GraphPad Prism (version X) and SPSS 20.0 for Windows. To investigate differences between experimental groups, a one-way ANOVA was used. Tukey’s test was used to discover particular group differences while adjusting for Type I errors. Before ANOVA, assumptions were verified using Levene’s test for variance homogeneity and the Shapiro-Wilk test for normality. Effect sizes (η2 for ANOVA and Cohen’s d for pairwise comparisons) were calculated to assess practical significance. Additionally, 95% confidence intervals (CIs) were reported alongside effect sizes to reflect estimate precision. Power analysis was conducted to confirm that the study was sufficiently powered to detect meaningful group differences.
Results
Linarin’s Impact on OVA-Induced Alterations in body Weight and Relative Lung Weight
Figure 1 shows that rats with asthma induced by OVA had a higher relative lung weight compared to rats without the condition. The administration of LIN impacted the body weight and relative lung weight of OVA-challenged rats. The rats’ total weight grew dramatically, but their relative lung weight fell. Rats given DEX as an asthma drug control showed a significant rise in body weight but a decrease in relative lung weight. Effect of Linarin (LIN) Treatment on Ovalbumin (OVA)-Induced Changes in (A) Body Weight and (B) Relative Lung Weight in Rats. Body Weight was Recorded Using a Digital Balance, and Relative Lung Weight was Calculated as the Ratio of Lung Weight to Body Weight at the End of the Experimental Period. Data are Presented as Mean ± SEM From Six Biological Replicates per Group (n = 6). Data are Presented as Mean ± SEM (n = 6) and Analyzed by One-Way ANOVA. #
Impact of LIN on Overall Cell and Differential Leukocyte Counts (×105 Cells/ml) in the Blood of Rats Sensitized to Ovalbumin
In the blood of rats given OVA, the infiltration of inflammatory cells, including neutrophils, eosinophils, and total cells, was assessed. In comparison to the non-sensitized control group, ovalbumin-sensitized control animals showed significantly greater levels of each differential count and overall leukocyte count in blood samples. Administration of LIN significantly reduced the total cell count, neutrophils, and circulating eosinophils compared to the OVA-sensitized control group (Figure 2). The positive control group had a similar lower concentration of inflammatory cells as the OVA-stimulated group. Effect of LIN on Total Leukocyte Count and Differential Leukocyte Profiles in the Blood (×105 Cells/ml) of Ovalbumin (OVA)-Induced Asthmatic Rats. (A) Total Leukocyte Count was Determined Using a Hemocytometer Following RBC Lysis With Turk’s Solution. (B) Eosinophil and (C) Neutrophil Counts Were Assessed by Differential Staining of Blood Smears Using Leishman’s Stain, Followed by Microscopic Evaluation. Data are Presented as Mean ± SEM (n = 6) and Analyzed by One-Way ANOVA. #
Effect of LIN on Histamine Release in BAL Fluid
When rats were exposed to OVA, the histamine concentration in their BAL fluid increased considerably compared to the control group. OVA-induced rats had histamine concentrations of 30 to 32 μg/mL, while the control group had 15 to 17 μg/mL. LIN significantly reduced histamine levels in BAL fluid to 21 ± 1 μg/mL, which was comparable to the positive control (Figure 3). Effect of LIN on Histamine Release in Bronchoalveolar Lavage Fluid (BALF) of Ovalbumin (OVA)-Induced Asthmatic Rats. Histamine Levels Were Quantified Using an ELISA-Based Assay Specific for Rat Histamine. Treatment Groups Include OVA (OVA-Induced Control), OVA + LIN (OVA-Induced and Treated With 50 mg/kg Linarin), and OVA + DEX (OVA-Induced and Treated With 3 mg/kg Body Weight Dexamethasone). Data are Presented as Mean ± SEM (n = 6) and Analyzed by One-Way ANOVA. #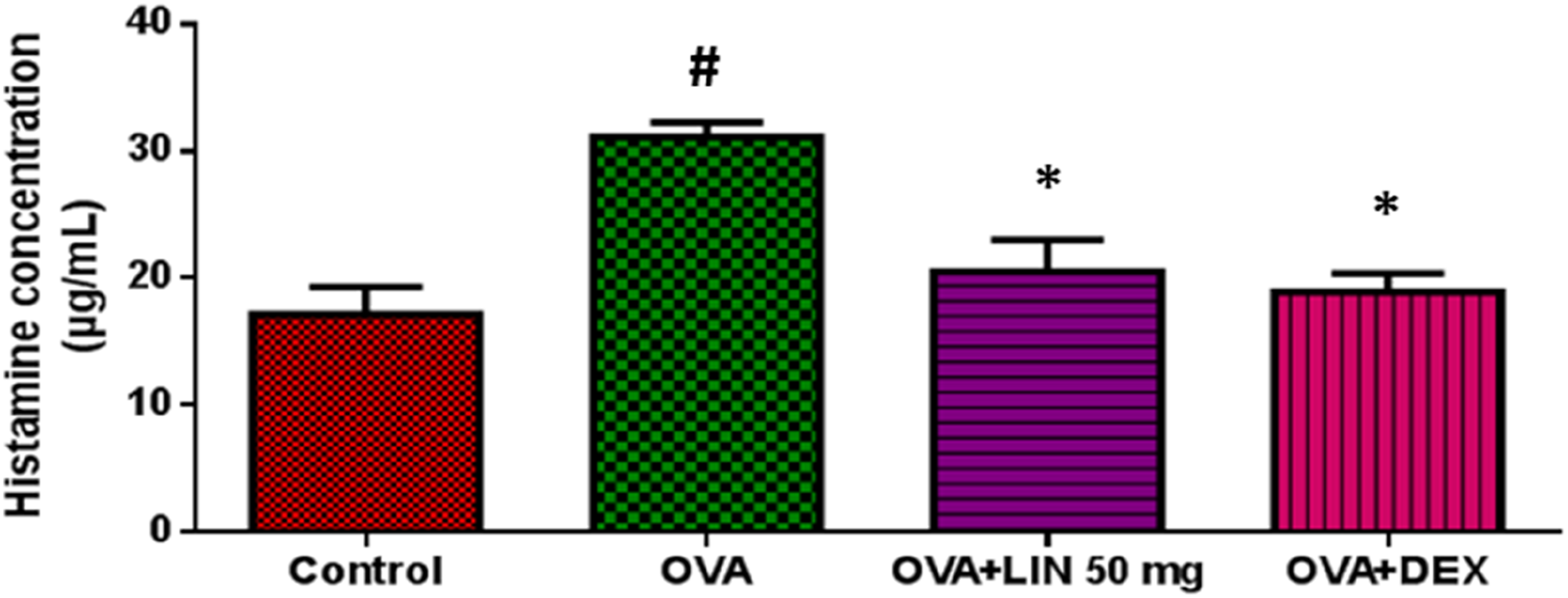
Impact of LIN on Oxidative Stress Indicators in Lung Tissue
Biochemical investigation of the OVA-challenged lungs demonstrated oxidative stress, with significantly lower levels of both enzymatic and non-enzymatic antioxidants than in normal control rats (Figure 4A and C). In contrast to the normal control group, rats infected with OVA exhibited considerably higher MDA levels (Figure 4D). The research revealed that LIN-treated rats had considerably higher levels of SOD, CAT, and GSH. In comparison to DEX, LIN therapy increased SOD, CAT, and GSH activity levels. MDA levels were around two times lower in LIN-treated, OVA-induced rats. Compared to the positive control DEX, the MDA reductions were significantly more effective. Effect of LIN on Oxidative Stress Markers and Antioxidant Enzyme Levels in Lung Tissue of Ovalbumin (OVA)-Induced Asthmatic Rats. (A) Superoxide Dismutase (SOD) Activity was Measured Based on Its Ability to Inhibit Pyrogallol Autoxidation. (B) Reduced Glutathione (GSH) Levels Were Estimated Using Ellman’s Reagent (5,5′-Dithiobis-2-nitrobenzoic Acid). (C) Catalase (CAT) Activity was Determined by Monitoring the Decomposition of Hydrogen Peroxide at 240 nm. (D) Malondialdehyde (MDA), an Indicator of Lipid Peroxidation, was Measured Using the Thiobarbituric Acid Reactive Substances (TBARS) Assay. Data are Presented as Mean ± SEM (n = 6) and Analyzed by One-Way ANOVA. #
Effect of LIN on Markers of Oxidative Stress in Erythrocytes
To test whether the animals’ erythrocytes were engaged in antioxidant reactions, we measured how LIN affected the levels of SOD, GSH, CAT, and MDA. The model control group showed significantly greater levels of MDA and significantly lower levels of the antioxidant enzymes SOD, GSH, and CAT than the normal control group (Figure 5A–D). The findings demonstrated that LIN is a promising antioxidant, as it significantly increased levels of SOD, GSH, and CAT relative to the positive control and substantially decreased MDA levels. Effect of LIN on Oxidative Stress Markers and Antioxidant Enzyme Activities in Erythrocytes of Ovalbumin (OVA)-Induced Asthmatic Rats. (A) Superoxide Dismutase (SOD) Activity was Assessed by Its Ability to Inhibit Pyrogallol Autoxidation. (B) Reduced Glutathione (GSH) Levels Were Determined Using Ellman’s Reagent (5,5′-dithiobis(2-nitrobenzoic Acid), DTNB). (C) Catalase (CAT) Activity was Measured by Monitoring the Decomposition of Hydrogen Peroxide at 240 nm. (D) Malondialdehyde (MDA) Levels, an Index of Lipid Peroxidation, Were Quantified Using the Thiobarbituric Acid Reactive Substances (TBARS) Assay. Data are Presented as Mean ± SEM (n = 6) and Analyzed by One-Way ANOVA. #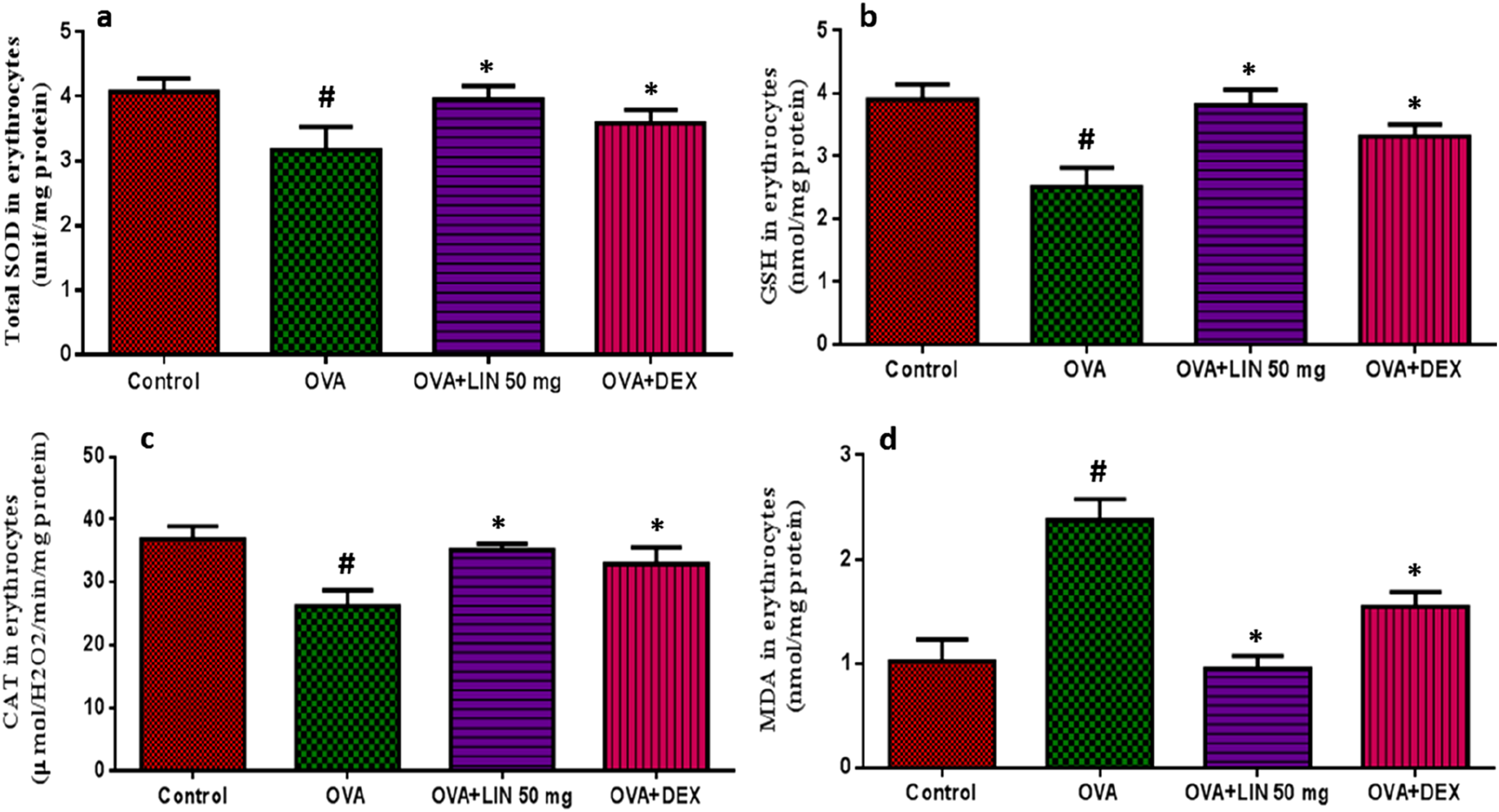
Impact of LIN on Inflammatory Cytokine Levels in Rats with OVA-Stimulated Asthma
Figure 6 demonstrates that TNF-α, IFN-γ, IL-4, and IL-13 were the inflammatory cytokines assessed in OVA-stimulated rats. OVA-challenged rats showed significantly higher levels of TNF-α, IL-4, and IL-13, whereas IFN-γ levels decreased compared to normal control rats. LIN therapy substantially inhibited the expression of inflammatory cytokines. Pro-inflammatory cytokine production was reduced by LIN therapy. Specifically, we observed a 40% decrease in IL-4, a 16% decrease in IL-13, and a 30% decrease in TNF-α levels. We also examined IFN-γ production, and the findings indicated that LIN slightly increased IFN-γ levels in OVA-stimulated rats. Effect of LIN on the Levels of Inflammatory Cytokines in Ovalbumin (OVA)-Induced Asthmatic Rats. The Concentrations of (A) Interleukin-4 (IL-4), (B) Interleukin-13 (IL-13), (C) Tumor Necrosis Factor-Alpha (TNF-α), and (D) Interferon-Gamma (IFN-γ) Were Measured in Lung Homogenates Using Enzyme-Linked Immunosorbent Assay (ELISA) Kits Specifically for Rat Cytokines. Data are Presented as Mean ± SEM (n = 6) and Analyzed by One-Way ANOVA. #
Enhancing Effect of LIN on OVA-Induced Alterations in Serum IgE Levels
Next, we examined whether LIN administration had any effect on IgE-mediated allergic asthma induced by the OVA challenge. The OVA-induced group had higher serum total IgE levels than the control group (Figure 7). However, compared to OVA-induced rats, LIN treatment significantly alleviated the effects of OVA-induced asthma by lowering serum IgE levels. Animals treated with DEX as a positive control showed outcomes similar to those of LIN-treated animals. Effect of LIN on Serum IgE Levels in Ovalbumin (OVA)-Induced Asthmatic Rats. Total IgE Concentration in Serum was Measured Using a Rat-specific Enzyme-Linked Immunosorbent Assay (ELISA) Kit. Data are Expressed as Mean ± SEM (n = 6) and Analyzed by One-Way ANOVA. #
Histopathological Modifications in the Lungs of OVA-Induced Rats
We conducted a histological investigation to assess the pathological aspects of OVA-induced allergic lung inflammation and the effect of LIN on it (Figure 8). Lung slices from normal control rats exhibited a healthy structure, with thin bronchial walls and well-organized alveolar gaps. In contrast, lung sections subjected to OVA exposure displayed edema, marked inflammation, thickening of intrabronchial walls, and distorted alveolar spaces. In rats with OVA-induced asthma, LIN treatment preserved lung anatomy, including regular alveolar spacing, thin intrabronchial walls, and reduced infiltration of inflammatory cells. Rats in the positive control group recovered equally in terms of lung morphology. Representative Photomicrographs of Lung Tissue Sections From (A) Normal Control Rats (Group I), Showing Normal Alveolar Architecture; (b) OVA-Induced Asthma Control Rats (Group II), Exhibiting Marked Peribronchial and Perivascular Inflammatory Cell Infiltration, Airway Wall Thickening, and Epithelial Disruption; (C) LIN-Treated Rats (50 mg/Kg; Group III), Showing Reduced Inflammatory Infiltration and Partial Restoration of Airway Structure; and (D) Dexamethasone-Treated Rats (Positive Control; Group IV), Displaying Near-normal Histoarchitecture With Minimal Inflammation. Lung Sections Were Stained With Hematoxylin and Eosin (H&E) and Observed Under Microscope.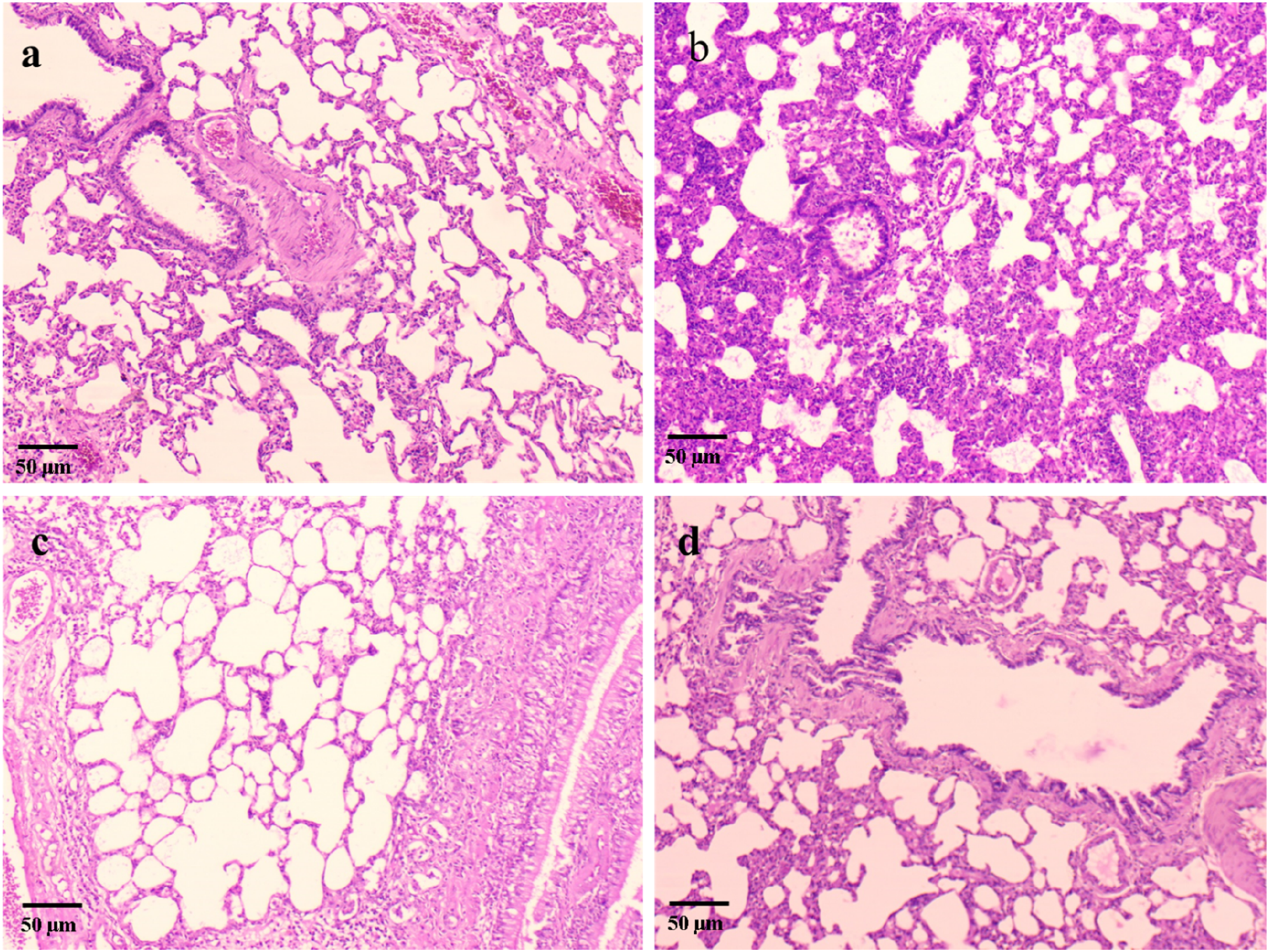
Discussion
Asthma is a long-term inflammatory respiratory condition involving numerous intricate pathways. It is well known that asthma triggered by allergens results in significant lung airway remodeling and inflammation. Because the illness is chronic, therapy necessitates ongoing care. Long-term control therapies for asthma now include persistent beta-agonists, dietary medicines, and inhalation of corticosteroids. 14 Although many conventional medications are available on the market, their effectiveness is limited; they exhibit a variety of negative effects, desensitize receptors, and pose compliance problems. Additionally, they are unable to block all the mechanisms that cause asthma. In rat models of OVA-induced asthma, airway remodeling, and inflammation were significant outcomes. In this work, we used OVA induction to create animal models of allergic asthma. Due to differences in lung tissue and health status, asthmatic rats are known to have different body weights and relative lung weights than normal rats. Dexamethasone is one of the few commercially available anti-asthmatic medications. In OVA-induced asthmatic rats, the outcomes of LIN treatment demonstrated significant preventive effects on airway inflammation, which were very similar to those of the positive control medication.
The ovalbumin-induced elevation in blood levels of inflammatory cell infiltration into the rat airways was considerably reversed by LIN therapy in the current investigation. This shows that LIN’s anti-inflammatory effect may be mediated by suppressing the leukocyte subpopulation. Elevated eosinophil counts in the blood and lungs are directly linked to airway hyperresponsiveness in allergic pathways. LIN’s ability to inhibit blood levels of eosinophils suggests that it is a valuable tool for treating allergen-induced bronchial hyperresponsiveness. According to earlier research, LIN reduces neutrophils, lymphocytes, and macrophages to mitigate acute lung injury. 15
Histamine is a powerful modulator of many biological processes. 16 It can stimulate both homeostatic activities, such as digestive regulation, and pathological mechanisms, such as allergy stimulation, by inducing both inflammatory and regulatory responses. 4 The impact of LIN on histamine levels in BALF has been demonstrated in this study. Following IgE cross-linking by an allergen, mast cells produce histamine, a primary amine that causes vasodilatation, mucus hypersecretion, edema, and contraction of smooth muscle cells. The reduced recruitment of histamine into BAL fluid in rats demonstrated LIN’s suppressive effects on airway inflammation.
Asthma is associated with high levels of oxidative stress, caused by both increased oxidant pressures and reduced antioxidant capacity. 17 Preventing oxidative stress could help decrease the progression of damage and inflammation. SOD, GSH, CAT, and other antioxidants all play important roles in controlling ROS generation. Reduced levels of antioxidant enzymes and GSH in OVA-challenged rats showed the existence of oxidative stress. However, rats sensitized in the lung and erythrocytes showed considerably higher MDA levels. Because LIN is an antioxidant, it could scavenge the ROS produced, inhibit lipid peroxidation, and preserve GSH and antioxidant enzyme levels.
Cytokines are proteins that govern immune responses and inflammation. The cytokine class includes interleukins, interferons, and tumor necrosis factors. Cytokines influence the expression of adhesion molecules, cell division, proliferation, and death, as well as immunoglobulin manufacture and chemotaxis in target cells. 18 Research has mostly focused on type 2-high asthma, which is linked to elevated levels of type 2 inflammatory biomarkers, including immunoglobulin E (IgE), IL-4, IL-5, and IL-13. 19
Pro-inflammatory cytokines cause damage to nearby cells at the site of injury, hastening disease progression. Previous research has shown a link between ROS production and the allergic response that causes inflammation. Our findings demonstrated that LIN treatment reduced the levels of Th2 cytokines, such as IL-4 and IL-13. Numerous investigations, both in vivo and in vitro, have linked cytokines such as TNF-α to the inflammation of asthmatic airways. In patients primarily dependent on corticosteroid medication, TNF-α blocking action may represent a potential therapeutic option for asthma. Asthmatic airways exhibit high levels of TNF-α, a cytokine that promotes inflammation. Compared to the normal control group, we observed notably higher TNF-α expression levels in the OVA-control group. The present investigation also demonstrated that, compared to the positive control, LIN administration markedly reduced TNF-α expression.
One known factor preventing airway smooth muscle contraction in asthmatic airway inflammation is IFN-γ, an anti-inflammatory cytokine.1,2,20 Clinically, it has been demonstrated that asthmatic patients triggered by allergens exhibit reduced BALF IFN-γ levels. The OVA-stimulated asthmatic rats showed considerable modulation in the levels of these inflammatory mediators. Because of its anti-inflammatory effects, LIN administration dramatically lowered inflammatory mediator concentrations in rats with OVA-induced asthma. Cytokines linked with inflammation can enhance the inflammatory response by increasing the release of chemoattractant factors by alveolar macrophages and airway epithelial cells, as well as the creation of adhesion molecules by leukocytes and epithelial cells. Previous studies have shown that LIN has anti-inflammatory characteristics, resulting in lower levels of TNF-α, IL-1β, and IL-6.15,21
Linarin, a flavonoid glycoside found in medicinal plants, has shown promise in treating asthma by modifying immunological responses. It regulates important signaling pathways in asthma pathogenesis, including the NF-κB pathway that reduces pro-inflammatory cytokine production. Linarin also regulates the MAPK signaling cascade, which is critical for cytokine synthesis and immune cell activation. It exerts immunomodulatory effects on various immune cells, contributing to asthma pathogenesis. Linarin exhibits antioxidant properties, reducing reactive oxygen species (ROS), regulating apoptosis pathways, and enhancing Nrf2 activation. It also influences histone modification, affecting gene expression in immune responses and reducing the transcriptional activity of inflammatory gene promoters. Understanding these mechanisms could help develop LIN-based therapeutic strategies for asthma management.
Rat models are limited in accurately representing the complex and heterogeneous asthma subtypes found in humans due to significant differences in respiratory system structure, immune response mechanisms, drug metabolism, and pharmacokinetics between rodents and humans. The pathophysiology of ovalbumin-induced asthma in rats does not fully mimic the heterogeneity of human asthma subtypes. Do translation issues arise due to differences in drug metabolism and pharmacokinetics between rodents and humans, and rats cannot report symptoms such as breathlessness or chest tightness, limiting the assessment of therapeutic effectiveness beyond biomarker analysis. Preclinical-to-clinical translation challenges exist, as reductions in inflammatory markers in rats may indicate potential anti-asthmatic effects but do not guarantee clinical efficacy in human trials. Safety and toxicity considerations are crucial, as adverse effects may arise due to differences in drug metabolism, off-target effects, or immunogenicity in humans. Regulatory hurdles and ethical considerations must be addressed before human trials, and the translation of experimental compounds from animal models to human subjects requires rigorous scrutiny.
Furthermore, one of LIN’s key effects is the inhibition of IgE, which mediates allergic responses associated with asthma and further reduces the subsequent response of inflammatory allergies in the airways. Histology results supported the findings, revealing a thin bronchial wall with fewer inflammatory cell infiltrations and preserved alveolar space. This discovery reinforces LIN’s ability to reduce airway inflammation and thus demonstrates its anti-asthmatic properties. Consequently, further research is required to synthesize the medication, assess its safety and effectiveness, and elucidate the molecular basis of this observation.
Conclusion
As a result, our findings may indicate complex interactions between LIN’s antioxidant, anti-inflammatory, and immune-regulatory properties in treating asthma. Ovalbumin-stimulated rats were treated with LIN, which is commonly used to treat inflammation and asthma. This treatment was shown to be effective in reducing pulmonary inflammation by significantly lowering cytokine levels and the infiltration of inflammatory cells in the lungs. Ultimately, this research supports the conventional medicinal use of linarin for the treatment of inflammation in asthmatic airways. It is possible that LIN’s antioxidant properties reduced oxidative stress by enhancing enzymatic antioxidant activity and decreasing membrane lipid peroxidation. However, more research is needed to confirm its safety before LIN may be produced as a pharmaceutical medication for human use.
Footnotes
Ethical Statement
The study was approved by the Ethics Committee of IEC of The First People’s Hospital of Xianyang (Approval Number: LG-IIT-2023052).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be made available on request.
