Abstract
In this study, we aim to investigate the effect of industrial Olea europaea L. leaf extract (OLE) against bleomycin (BLM)-induced pulmonary fibrosis (PF) in rats. Male Wistar rats were treated with a single intratracheal injection of BLM (4 mg/kg) and a daily intraperitoneal injection of OLE (10, 20, and 40 mg/kg) for 4 weeks. Results of HPLC and LC-MS analysis revealed a large amount of oleuropein (15.43%/DW) in OLE. BLM induced apparent damage of lung architecture with condensed collagen bundles, increased lipid peroxidation which has been deduced from malondialdehyde (MDA) levels: (.9 ± .13 vs .25 ± .12 nmol/mg protein) and hydroxyproline content (.601 ± .22 vs .154 ± .139 mg/g of lung tissue) and decreased catalase (CAT) (5.93.10−5 ± 4.23.10−5 vs 6.41.10−4 ± 2.33.10−4 μmol/min/mg protein) and superoxide dismutase (SOD) (28.73 ± 3.34 vs 50.13 ± 2.1 USOD/min/mg protein) levels compared to the control. OLE treatment (40 mg/kg) stabilized MDA content (.32 ± .15 and .27 ± .13 vs .9 ± .13 nmol/mg protein), normalized SOD (61.27 ± 13.37 vs 28.73 ± 3.34 USOD/min/mg protein), and CAT (5.2.10−4 ±1.8.10−4 vs 5.93.10−5 ± 4.23.10−5 μmol/min/mg protein) activities and counteracted collagen accumulation and hydroxyproline content (.222 ± .07 vs .601 ± .22 mg/g of lung tissue) in the lung parenchyma. Finally, OLE might have a potent protective effect against PF by regulating oxidative parameters and attenuating collagen deposition, due to the existence of large amount of bioactive phenolic molecules.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic lung disease associated with limited effective treatments.1,2 IPF is characterized by excessive fibroblasts proliferation affecting lung parenchyma, leading to fibrotic process and architectural disorganization.3,4 These manifestations are accompanied by severe oxidative stress, inflammation, and angiogenesis among others. 5 Given the limited efficacy and side effects of current treatments, the search for effective bioactive molecules in the treatment of this disease is growing. In consequence, new treatment approaches are inevitable to ameliorate the quality of life and the survival of IPF patients.
The last decade imported a lot of experimental research’s worldwide by exploiting certain medicinal plants due to their large content of bioactive compounds and their relative safety. Several experiments have been carried out with bioactive substances and plant extracts, that seems to be promising by offering a novel therapeutic insight for IPF treatment.6,7
Recently, many studies investigated the protective effect of Olea europaea L. leaf extract (OLE) and its phenolic substances, such as oleuropein, on several experimental health injuries, including the capacity to protect against Busulfan-induced toxicity in rat testes, 8 cyclophosphamide-induced nephrotoxicity in rats, 9 autosomal dominant polycystic kidney disease, 10 isoproterenol-induced myocardial infarction in rats, 11 cartilage degeneration in mice, 12 and against cadmium-induced nephrotoxicity. 13
To our knowledge, no experimental investigations have been performed to study the antifibrotic and antioxidant effect of OLE especially in the lung tissue, which encouraged us to test the effect of an industrial extract of Olea europaea L. on a rat model of pulmonary fibrosis, since it has been described for its efficacy in the treatment of several experimental injuries where oxidative stress, angiogenesis, and inflammation exert a key role. Therefore, this work aimed to study the in vivo effect of this extract on a murine experimental model of bleomycin (BLM)-induced lung fibrosis.
Materials and Methods
Industrial Olive Leaf Aqueous Extract
Olive leaves (Olea europaea L.) were collected from EL Mansourah, Siliana, Tunisia (Northwestern Tunisia; latitude 35° 48′ 47″ (N), longitude 9° 21′ 52″ (E), altitude 1017 m), during winter 2022. The plant material was identified by Meddeb Marouan, Professor in the High Institute of Biotechnology of Beja, University of Jendouba, Tunisia. Olive leaves were dried and crushed at room temperature. Aqueous extraction (85%) was realized and the extract was then filtered and evaporated under a vacuum at 65°C and stored at 4°C for subsequent assays. Olive leaf extract was prepared in the laboratory of quality control, HERBES DE TUNISIE, AYACHI-Group Company, Mansoura, Siliana-Tunisia.
LC-MS Assay
LC-MS analysis was achieved on a reversed-phase column (Pursuit XRs ULTRA 2.8, C18, 100 × 2 mm, Agilent Technologies, UK). 20 μl of our extract was injected in the column at 30°C. Mobile phases are composed of .1% formic acid in water and .1% formic acid in MeOH as described in our previous study. 14 High resolution mass spectral data were created on a Thermo Instruments ESI-MS system (LTQ XL/LTQ Orbitrap Discovery, UK) attached to a Thermo Instruments HPLC system (Accela PDA detector, Accela PDA autosampler and Accela Pump).
HPLC Analysis for the Determination of Oleuropein Content
Oleuropein content in Olea europaea leaf extract was determined by high-performance liquid chromatography system (HPLC) as described by Bahri et al. 14 Analyses were carried out on a C18 reverse phase (BDS 5 μm, Labio, Czech Republic) packed stainless-steel column (4 250 mm, i.d). HPLC peaks were recognized by congruent retention times and UV spectra and compared with those of the standards.
Phenolic Compound Contents
Total polyphenol and flavonoids content were performed following the methods of Zhishen et al 15 and Singleton et al, 16 respectively. OLE was assessed in triplicate.
Radical Scavenging Power Evaluation by DPPH Assay
Radical scavenging activity of Olea europaea leaf extract was performed according to modified methods of Ba et al. 17 Results were compared to the antioxidant activity of Trolox used as a standard and are expressed as Trolox Equivalent/g of our extract.
BLM-Induced Pulmonary Fibrosis Model
Forty healthy male Wistar rats, aged 3 months and weighting between 180 and 220 g, were obtained from Pasteur institute, Tunis, Tunisia, and maintained in the experimental medicine unit (Faculty of Medicine of Tunis, Tunisia) at 22 ± 2°C with 12 h light/dark cycle. Rats were supplied with food/water ad libitum. The composition of the standard diet is as follows: 3% lipids, 40% carbohydrates, 14.5% proteins, 50 mg/kg iron, and 1.45 mg/kg calcium; it was provided by El-Badr Society (Utique, Tunisia). All experimental procedures followed the guidelines set forth in the Care and Use of Animals and were approved by the Ethics Committee of the National School of Veterinary Medicine of Sidi Thabet (Ref: 162020/FMT) according to the International Council for Laboratory Animal Science (ICLAS) recommendations.
BLM model of pulmonary fibrosis was carried out as described in our previous studies.5,18 Animals underwent anesthesia with pentobarbital sodium solution (Sandoz laboratory, France) and then treated with an intra-tracheal instillation of BLM at 4 mg/kg (Bleomycin®, Laboratories Aventis, France).
In vivo Experimental Study
Rats were divided into 5 groups of 8 animals each: Group I treated with normal saline (the control group). Group II treated with an intratracheal injection of BLM (4 mg/kg bw) (the BLM group). Group III treated with an intratracheal injection of BLM (4 mg/kg bw) and a daily intraperitoneal injection of OLE (10 mg/kg bw). Group IV received a single intra-tracheal instillation of BLM (4 mg/kg bw) and a daily intraperitoneal injection of OLE (20 mg/kg bw). Group V received a single intra-tracheal instillation of BLM (4 mg/kg bw) and a daily intraperitoneal injection of OLE (40 mg/kg bw).
Treatments with OLE started from the third day after the BLM instillation and lasted for 4 weeks (The BLM/OLE groups). The studied rats were fasted overnight (for about 12 hours) on the last day of experimentation before being anesthetized and were then sacrificed and lungs were extracted, cut, and divided into 2 parts for a histopathological evaluation as well as the determination of oxidative stress markers. Total soluble proteins content in rat’s lungs was carried out following Ohnishi et al method. 19
Histopathological Examination
For the histopathological study, rat’s lungs were fixed with 10% neutral-buffered formalin and stained with hematoxylin-eosin (H&E) to identify inflammatory cells and Masson’s trichrome to investigate collagen deposition in lung parenchyma after BLM treatment.
A blinded semi-quantitative scoring/grading system was used to assess the severity of lung fibrosis induced by BLM, as described by Ashcroft et al: 20 Grade zero = “normal lung,” Grade 1 = “minimal fibrous thickening of alveolar or bronchial walls,” Grades 2 to 3 = “moderate thickening of walls without obvious damage to lung architecture,” Grades 4 to 5 = “increased fibrosis with definite damage to lung architecture and formation of fibrous bands or small fibrous mass,” Grades 6 to 7 = “severe distortion of structure and large fibrous areas,” “honeycomb lung”; Grade 8 = “total fibrotic obliteration of the field.”
The extent and severity of inflammation and fibrosis in the lung parenchyma were assessed using an inflammatory index classified according to the following categories: grade 0, absence of inflammation; grade 1, minimal inflammation; grade 2, minimal to moderate inflammation; grade 3, moderate inflammation with thickening of the alveolar walls; grade 4, moderate to severe inflammation; and grade 5, severe inflammation with the presence of follicles. 21
Determination of Antioxidant Enzymes Activities in Lung Tissue
Catalase (CAT) activity was evaluated as described by Bonaventura et al 22 Briefly, samples were mixed with H2O2 and phosphate buffer (pH = 7). The absorbance was recorded at 240 nm, and CAT activity was expressed in μmole of H2O2 consumed/minute/mg of protein.
Superoxide dismutase (SOD) activity determination was performed according to Misra et al. 23 Briefly, 10 μl of bovine catalase (.4 U/μl) was added to our samples and the whole was then mixed with 20 μl epinephrine (5 mg/ml) and 62.5 mM sodium carbonate-sodium bicarbonate buffer. The absorbance was read at 480 nm and expressed by unit of SOD/min/mg protein.
Lipid Peroxidation Levels
Lipid peroxidation was evaluated as described by Ohkawa et al. 24 Briefly, samples were incubated with sodium phosphate buffer at 37°C for 1 hour and then precipitated with trichloroacetic acid. 1% of thiobarbituric acid was added and the mixture was placed in boiling water for 15 minutes. Malondialdehyde (MDA) levels were calculated in terms of nmol/mg protein using molar extinction coefficient of 156000 M−1·cm−1 at 532 nm.
Hydroxyproline Quantification
Hydroxyproline was determined as mentioned by Reddy and Enwemeka (1996). 25 Briefly, the lung tissue (1 g) was mixed with H2O (1 mL) and hydrolyzed in 2 N NaOH at 120°C for 20 min.
The obtained hydrolysate (50 μl) was oxidized for 25 min by adding Chloramine T (450 μl, 56 mM). Then, Ehrlish’s aldehyde reagent (500 μl) was supplemented and the chromophore was developed by incubating the samples at 65°C for 20 min. The absorbance was measured at 550 nm, and hydroxyproline content was expressed as mg/g of lung tissue.
Statistical Analysis
Statistical analysis was performed using SPSS software (version 17.0; SPSS, Inc., Chicago, IL, USA). Statistical significance was evaluated by ANOVA followed by Sidak’s test for all post-hoc multiple comparisons. A P-value of .05 or less was considered as statistically significant.
Results
Quantification and Identification of Compounds by LC-MS and HPLC
Determination of Olive Leaf Extract Bioactive Compounds Using Liquid Chromatography-Mass Spectrometry (LC-MS).

Our chromatographic analysis (HPLC) also exhibited the appearance of oleuropein peak (15.43%/DW) after 19.23 min at 280 nm (Figure 1). On the other hand, total polyphenols content was evaluated following Folin–Ciocalteu method. Our results (Table 2) showed that the Olea europaea leaf extract contains a high level of total polyphenols in the range of 8524.44 g GAE/100 g dw (g equivalent gallic acid). OLE also contains a high amount of flavonoids in the order of 7128.88 g QU/100 g dw (g equivalent quercetin). Chromatographic peak of oleuropein in OLE recorded at 280 nm, retention time: 19.23 min. Total Polyphenol Contents (Tpc) Expressed in mg Equivalent Gallic Acid (g GA.100g-1 DW), Total Flavonoid Contents (TFC) Expressed in mg Equivalent Quercetin (mg QU/100 g DW), and Antioxidant Activity (Trolox Equivalent μM) of Olive Leaf Extract.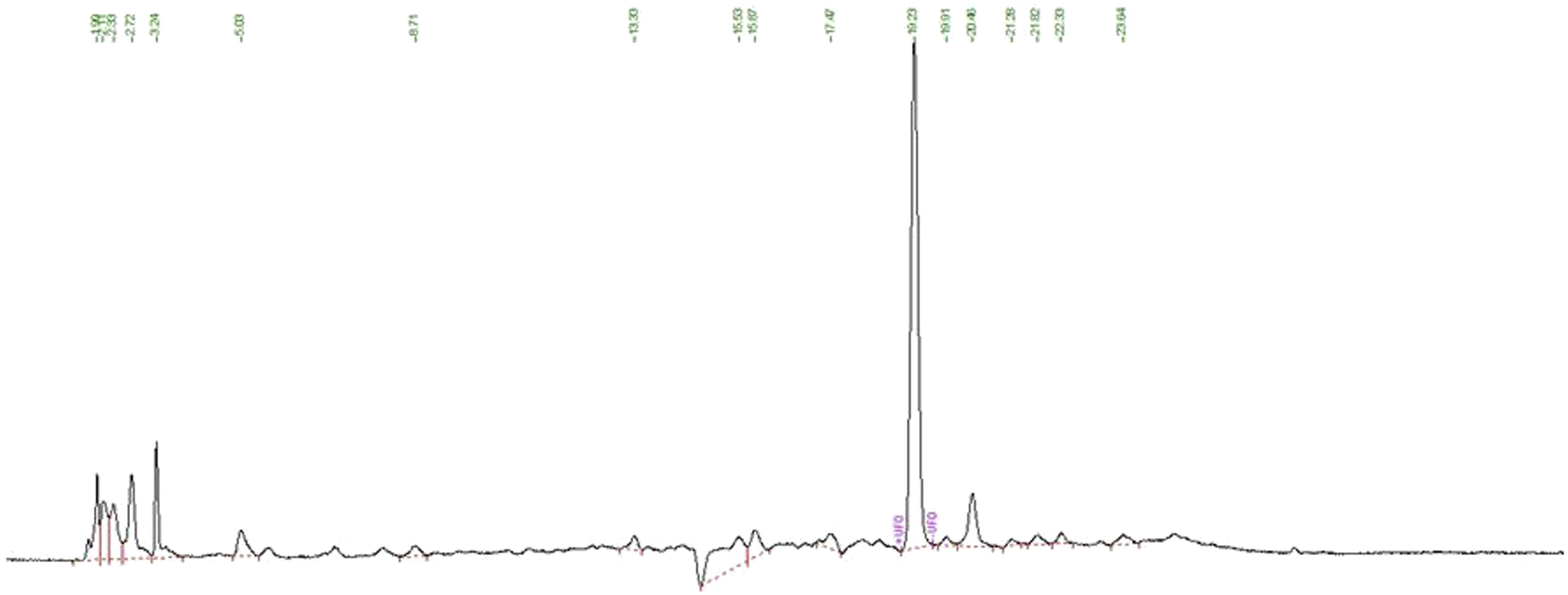

Effect of OLE on Pulmonary Enzymatic Antioxidant Levels: CAT and SOD Activities
Our results showed a significant decrease in CAT activity in the lung tissue after the BLM treatment when compared to the control group (5.93 × 10−5 ± 4.23 × 10−5 vs 6.41 × 10−4 ± 2.33 × 10−4 μmol/min/mg protein, ***P < .001; respectively) (Figure 2). Effect of olive leaf extract (OLE) on lung CAT activity after BLM treatment. Results are expressed as means ± SE (n = 8) ***P < .001 vs C, ##P < .01 vs BLM. Comparisons were performed using Sidak’s Test. C: Control group, BLM: Bleomycin group, BLM
Treatment with OLE at 40 mg/kg revealed a significant increase in CAT activity in comparison with the BLM group (5.2 × 10−4 ± 1.8 × 10−4 vs 5.93 × 10−5± 4.23.10−5 μmol/min/mg protein, ##P < .01; respectively).
On the other hand, our results demonstrated a significant diminution in SOD activity in the lung tissue following BLM treatment compared to the control group (28.73 ± 3.34 vs 50.13 ± 2.1 USOD/min/mg protein; *P < .05; respectively) (Figure 3). Effect of olive leaf extract (OLE) on lung SOD activity in rats after BLM treatment. Results are expressed as means ± SE (n = 8) *P < .05 vs C, ###P < .001 vs BLM. Comparisons were performed using Sidak’s Test. C: Control group, BLM: Bleomycin group, BLM/10 mg/kg: Bleomycin + OLE at 10 mg/kg, BLM/20 mg/kg: Bleomycin + OLE at 20 mg/kg, BLM/40 mg/kg: Bleomycin + OLE at 40 mg/kg.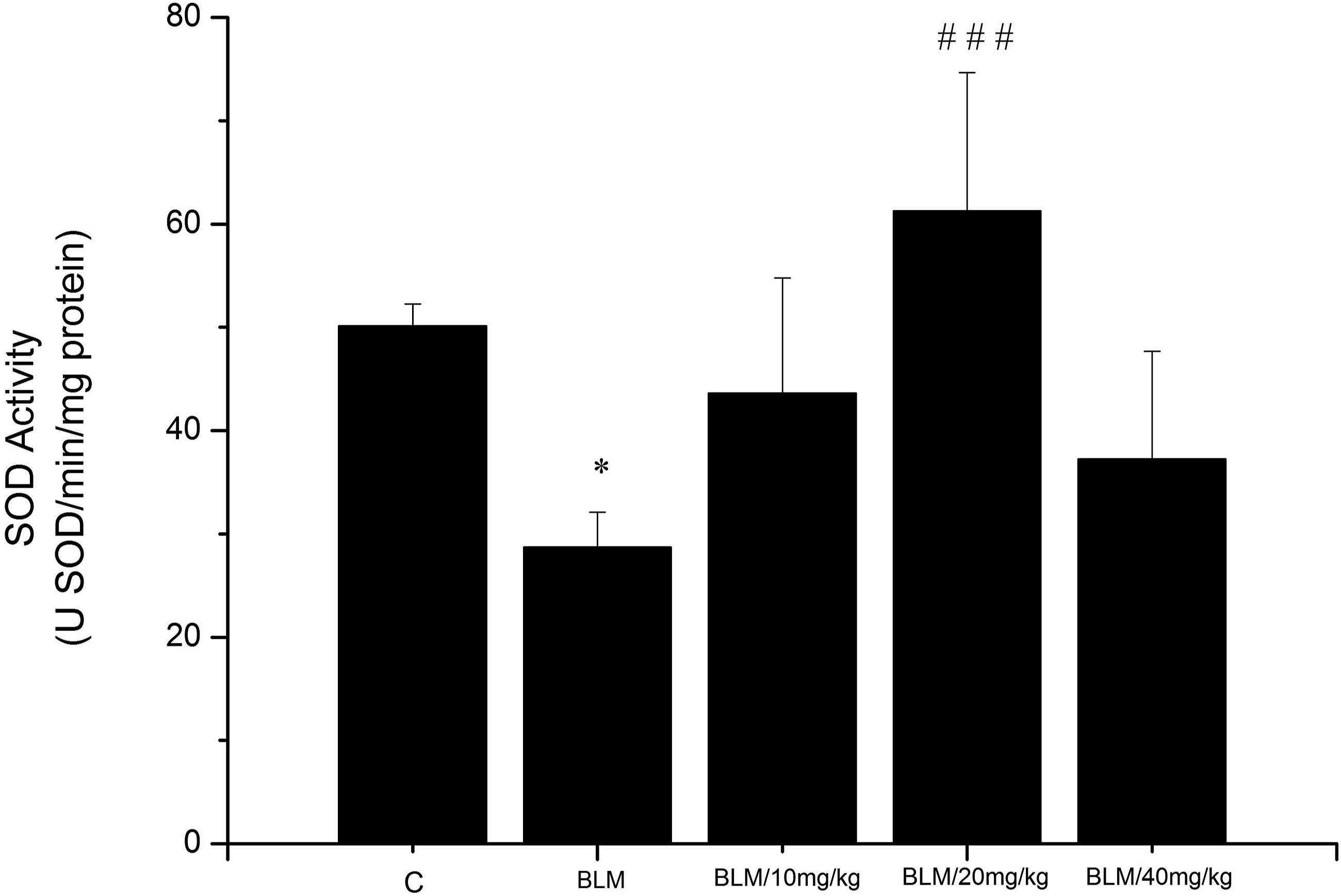
OLE at 20 mg/kg induced a significant restoration of SOD levels in comparison with the BLM group (61.27 ± 13.37 vs 28.73 ± 3.34USOD/min/mg protein, ###P < .00; respectively).
Effect of OLE on Lipid: Peroxidation
Our results showed a significant increase in MDA levels in the BLM group in comparison with the control group (.9 ± .13 Effect of olive leaf extract (OLE) on BLM-induced lipid peroxidation in the rats lung. Results are expressed as means ± SE (n = 8) ***P < .001 vs C, #P < .05 vs BLM,
###
P < .001 vs BLM. Comparisons were performed using Sidak’s Test. C: Control group, BLM: Bleomycin group, BLM/10 mg/kg: Bleomycin + OLE at 10 mg/kg, BLM/20 mg/kg: Bleomycin + OLE at 20 mg/kg, BLM/40 mg/kg: Bleomycin + OLE at 40 mg/kg.
Treatment of rats with OLE at 20 and 40 mg/kg induced a significant decrease of MDA levels in comparison with the BLM group (.32 ± .15 and .27 ± .13 vs .9 ± .13 nmol/mg protein, respectively; P < .001).
Histological Analysis
Figure 5 displays histopathological findings in pulmonary tissue after H&E staining. The control group showed normal lungs (Figure 5A). The BLM treatment showed histological disorganization of lung tissue characterized by interstitial thickening surrounding large follicles (Figure 5B). Treatment with OLE at 40 mg/kg revealed an apparent enhancement of the lung architecture and a decrease in the lung damage caused by the BLM treatment (Figure 5E). Rats lung histoarchitecture (H&E Staining) × 100. Sections are marked by intense inflammation (stars) and a disruption of alveolar architecture with the presence of lymphoid follicles and interstitial thickening (arrows). (A) Control group; (B) BLM group, (C) BLM/OLE at 10 mg/kg group, (D) BLM/OLE at 20 mg/kg group, (E) BLM/OLE at 40 mg/kg group, one representative example is presented for each group.
Figure 6 exhibits histopathological findings in the pulmonary tissue following Masson’s trichrome staining. Our results showed a large area of collagen stained in green following the BLM treatment (Figure 6B). Treatment with OLE at 40 mg/kg showed a significant reduction in collagen deposition and fibrotic areas in comparison with the BLM group (Figure 6E). Rats lung histoarchitecture (Masson’s trichrome staining) × 100. Sections are marked by inter-alveolar edema (stars) and the presence of inflammatory infiltrates in the alveolar septa (arrows). (A) Control group; (B) BLM group, (C) BLM/ OLE at 10 mg/kg group, (D) BLM/OLE at 20 mg/kg group, (E) BLM/OLE at 40 mg/kg group. One representative example is presented for each group.
Fibrosis Score and Inflammatory Index
Fibrosis Score and Inflammatory Index.

Results are expressed as means ± S.E. (n = 8), *** P < .001 vs. Control; ##P < .01 vs. BLM, ###P < .001 vs. BLM.
Monitoring of the variation in the inflammatory index between the different histological sections of various studied groups did not show any significant difference between the groups treated with OLE and the BLM group (Table 3).
Effect of OLE on Hydroxyproline Content in the Lung
Hydroxyproline content in the lung tissue, as shown in Figure 7, was significantly increased in the BLM group when compared with the control group (.601 ± .22 vs .154 ± .139 mg/g of lung tissue, respectively; P < .001). Our results showed a significant decrease in hydroxyproline content following OLE treatment at 40 mg/kg in comparison to the BLM group (.222 ± .07 vs .601 ± .22 mg/g of lung tissue, respectively, P < .01). Effect of OLE on BLM-induced changes in hydroxyproline content in the lung. Results are expressed as means ± SE (n = 8) ***P < .001 vs C, ##P < .01 vs BLM. Comparisons were performed using Sidak’s Test. C: Control group, BLM: Bleomycin group, BLM/10 mg/kg: Bleomycin + OLE at 10 mg/kg, BLM/20 mg/kg: Bleomycin + OLE at 20 mg/kg, BLM/40 mg/kg: Bleomycin + OLE at 40 mg/kg.
Discussion
In this experimental study, we aimed to investigate the effects of Olea europaea leaf aqueous extract rich with oleuropein on murine pulmonary fibrosis induced by BLM.
The toxic effect of the BLM implicates reactive oxygen species (ROS) formation that involves Fe (II) oxidation to Fe (III) inducing oxygen reduction to free radicals. 7 The BLM was also described to induce ferroptosis of the lung epithelial cells 26 and to activate apoptosis signal-regulating kinase-1 (ASK1) which is a member of the mitogen-activated protein (MAP) kinase family and are normally activated by oxidative stress. 27
The results of this study revealed clearly significant oxidative disturbance after the BLM treatment as indicated in MDA, SOD, and CAT levels evaluation which corroborates with those of Liu et al. 28 Interestedly, OLE induced significant restoration of these activities. Indeed, treatment with OLE induced a significant increase in CAT activity in lung parenchyma which was dropped after the BLM treatment, and these results corroborate with those of Rouibah et al 29 and Elgebaly et al 30 OLE has been described to protect rats against oxidative stress by inducing a reduction/inhibition of the ROS formation which can improve CAT production. 31 Moreover, this extract has shown the presence of 2 main bioactive components (oleuropein and hydroxytyrosol) 29 which enhance the pharmacological efficacy of this extract including antioxidant activity. 32 Furthermore, the SOD activity was also significantly restored after OLE treatment compared to the BLM group. This may be caused by the large presence of phenolic compounds which have the capacity to reduce ROS formation and/or stimulate SOD production and therefore oxidative balance regulation. Our results also substantiate those of Elgebaly et al 30 which find an amelioration in some antioxidant enzymes levels such as CAT and SOD in the rats liver. Coban et al 33 also indicated that OLE can decrease oxidative damage by acting as an antioxidant without affecting the antioxidant system in some rat tissues such as the liver. OLE was also described to ameliorate the antioxidant capacity by scavenging free radicals in cultured human blood cells treated with permethrin. 34 All these findings reinforce and confirm our results and led us to conclude that OLE has a potent oxidative inhibitory effect.
Histological results showed for the first time a potent antifibrotic effect of OLE in the lung tissue. We recorded a significant reduction in collagen deposition and fibrotic areas and a large decrease in fibrosis score in rats co-treated with OLE at 20 and 40 mg/kg compared to those treated with the BLM alone. Recently, Lupinacci et al 35 indicated that OLE can counteract fibrotic process in peritoneal dialysis patients by inhibiting tumor growth factor β1 (TGFβ1)-induced epithelial-mesenchymal transition, which plays a potent role in the installation and the progression of the lung fibrosis. OLE treatment was also described to play a protective role against carbon tetrachloride-inducing liver fibrosis in rats by exerting an antioxidant effect which is responsible for hepatoprotection. 36 These results support our findings in which OLE induced apparent enhancement of the lung architecture and a decrease in the lung damage and hydroxyproline content induced by BLM.
Recently, OLE effects have been studied in various experimental injuries. Majumder et al 37 highlighted its synergic effect with Bromelain to attenuate lung carcinogenesis associated with inflammation and oxidative stress via regulating the expression of various inflammatory markers and also modulating the activity of pulmonary antioxidant armories. 37 Romero Marquez et al demonstrated that OLE enriched in oleuropein can reduce β-amyloid and tau proteotoxicity which provides a new insight in the treatment of Alzheimer. 38 Hong et al 39 showed also the anti-inflammatory and anti-rheumatic effects of the Tunisian OLE. 39
By examining the phytochemical composition of OLE by HPLC and LC-MS analysis, a large amount of oleuropein (15.43%/DW) and the presence of hydroxytyrosol was found. Both olive phenols were described to play an effective role against coronary heart diseases and different human tumors including colorectal, thyroid, breast, skin, digestive, brain, lung, blood, and cervical by inhibiting tumor cells proliferation, migration, invasion, and angiogenesis.40,41 Hydroxytyrosol was also described to exert an antiangiogenic effect, 42 an anti-inflammatory role on collagen-induced arthritis in rats, 43 and an inhibitory effect on matrix metalloproteinase-2 levels in some cell extracts which are powerful actors in the development of fibrotic process. 44 All these findings reinforce the protective antifibrotic role of OLE, especially for its content in some bioactive compounds such as oleuropein and hydroxytyrosol which play potential biological activities and that can serve to inhibit the deleterious effects of BLM.
Conclusion
In summary, our findings demonstrate clearly and for the first time that the industrial OLE can play an improving role against BLM-induced pulmonary fibrosis and oxidative stress in rats. Indirectly, these results suggest that the constituents of OLE may modulate cellular dynamics during fibrosis progression so that some other parameters determination such as TGFβ and interleukins densities, need to be done to better elucidate the mechanisms of action of this extract. These effects are due to a large amount of some bioactive molecules such as oleuropein and hydroxytyrosol. Further studies are required to detect the antifibrotic effect of these 2 molecules separately in vivo and in vitro, as well as the molecular mechanism involved.
Footnotes
Acknowledgments
This scientific work was funded by the Tunisian National Agency for the Promotion of Scientific Research (ANPR) and Laboratory of quality control, HERBES DE TUNISIE Company AYACHI-Group, 6131, Mansoura, Siliana-Tunisia, within MOBIDOC device, in order to conduct applied research in the socio-economic environment.
Author Contributions
SB and SJ conceived and designed the research. SB, AA, AN, RA, MM, and RBA conducted experiments. SB analyzed data. SB wrote the manuscript. SJ and AA revised the manuscript. All authors read and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Tunisian National Agency.
