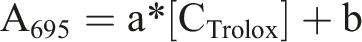Abstract
Pulmonary fibrosis (PF) remains one of the most serious pneumopathies whose diagnosis and physiopathogenesis are still poorly understood and no treatment has been shown to be effective. Recently, many studies have shown a renewed interest in plants thanks to their pharmacological potentials, like horehound, known, for its anti-inflammatory and antioxidant activities. The present study investigated the effects of the aqueous extract of horehound (Mae) on bleomycin (BLM)-induced PF in rats. Thirty rats were divided into three groups. The control group received no treatment, the BLM group received only intratracheally BLM (2 mg/kg), and the Mae group underwent administration of BLM+ Mae (2 mL/kg) daily for 20 days. Obtained results showed that Mae, rich in polyphenols, could significantly improve the damage caused by BLM by reducing the inflammatory index and the fibrosis score, bringing the lung structure of fibrotic rats close to that of control rats. As well, Mae obviously acted on the BLM inflammatory reaction, and the counting of bronchoalveolar lavage fluid (Balf) cells showed an increase in total cell number and a decrease in the infiltration of inflammatory cells in the bronchoalveolar space. In addition, the BLM instillation was accompanied by oxidative stress in the lung, liver, and kidney tissues, proven by an increase in lipid peroxidation, as well as through depletion of superoxide dismutase (SOD) and catalase (CAT). The Mae treatment reversed all disturbances of BLM-induced oxidative stress parameters promoting antioxidant and anti-inflammatory of the latter. These findings point to Mae as a promising candidate for the treatment of pulmonary fibrosis.
Introduction
Idiopathic pulmonary fibrosis (IPF) is a progressive and irreversible lung disease characterized by damage to the alveolar epithelial cells, infiltration of inflammatory cells, proliferation of fibroblasts, and deposition of extracellular matrix. 1
Physiopathologically, this ailment remains unclear and there are many hypothetical factors involved; the interaction of several signaling pathways and cytokines, oxidative stress, and excessive inflammation play an important role in its onset and development. Current treatments are partially effective and mainly based on corticosteroids and immunosuppressants (pirfenidone and nintedanib are the only drugs prescribed to alleviate IPF). 2
Since antiquity, natural products, particularly of plant origin, have made an inexhaustible source of remedy for various diseases (Egyptian, Chinese, and Greek medicines). Given that standard treatment is partially effective and not without deleterious effects, there is a resurgence of interest in natural substances that could have therapeutic effects and prevent pulmonary fibrosis with fewer side effects.3-5
Among these substances, Marrubium vulgare, from family Lamiaceae, is a popular traditionally used herb in many countries, and it is a perennial herb native to Eurasia and North Africa. It is formerly used to treat several illnesses including respiratory infections such as bronchitis, cough, and asthma. 6 Nevertheless, the various extracts of this plant have shown other pharmacological activities, in particular antihypertensive, antispasmodic, analgesic, anti-inflammatory, hypolipidemic, hypoglycemic, vasodilating, antidiabetic, and antioxidant properties.7,8
The therapeutic effects of Marrubium vulgare are widely described and its medical preparations are widespread in several studies. Phytochemically, M. vulgare is characterized by the presence of various compounds, such as alkaloids, steroids, lactones, tannins, flavonoids, and, in particular, terpenes. 9 The most abundant of the terpenes described for this plant is marrubiin, a labdane diterpene furan, known, above all, by its stimulating effects of secretions in the bronchial mucosa, but also, by its anti-arrhythmic, antioxidant, vasorelaxant, antinociceptive, gastroprotective, antispasmodic, immunomodulating, anti-edematogenic, and analgesic effects.6,9-11
In this context, our study was aimed to investigate the curative effects of the aqueous extract of horehound known by their antioxidant and anti-inflammatory effects and especially their richness in polyphenols, on an experimental model of pulmonary fibrosis induced by bleomycin in rats.
Materials and Methods
Plant Material
Aerial parts of Marrubium vulgare were collected during the flowering stage from Djebba Beja (Northwestern Tunisia; latitude 36°48’19” (N), longitude 9°09’55” (E), altitude 700 m), in March 2020. A voucher specimen of the plant representing this collection has been deposited at the Herbarium of the High Institute of Biotechnology of Beja (University of Jendouba, Tunisia).
After cutting into small pieces and drying at a temperature of 25 to 30° C for 7 days, the plant material was pounded using a coffee grinder, to obtain a fine powder which was kept in the dark. Briefly, the aqueous extract was prepared according to the traditional method: 6 g of M. vulgare powder were dissolved in 25 mL of boiling distilled water; the hot infusion was then allowed to stand at room temperature (about 15 min) and was then filtered. The obtained filtrate was stored to be used as it is for biological tests on animals and to undergo the necessary preparations for analytical determinations (UPLC, total polyphenols, and antioxidant activity). 12
Reversed Phase Ultra-Performance Liquid Chromatography Analyses
Reversed phase ultra-performance liquid chromatography (RP-UPLC) analyses were performed on all samples using a 1290 Infinity II UPLC (Agilent Technologies, Santa Clara, CA, USA). The RP-UPLC apparatus was composed of a binary pump (G7120A), a multi sampler (G7167B), an in-line degasser, and a variable wavelength detector (VWD G7114B) adjusted to 214 nm. Then, .22 μm polyvinylidene difluoride (PVDF) filters were used to filter sample into a glass vial. Samples were loaded (50 μL) into EC-C4 column (3 _ 150 mm i.d., 3 micron, Interchim, Santa Clara, CA, USA). A flow rate of 0.3 mL/min at 30°C was used to operate the column. A gradient was applied for the mixture of solvent A (LC-MS grade water with .1% trifluoroacetic acid) and solvent B (LC-MS grade ACN with .1% trifluoroacetic acid) with solvent B increasing from 5% to 30% in 30 min, to 60% until 40 min, and to 95% until 44 min and holding until 47 min, and then back to initial conditions. Each sample analysis was performed in triplicate for ensuring technical reproducibility. 13
Determination of Polyphenols by Folin Reagent
The amount of total phenolic content was quantified according to Folin–Ciocalteu method. 14 Briefly, 50 μL of sample was mixed with 120 μL of Folin–Ciocalteau reagent and 2 mL of distilled water for 5 min, followed by 375 μL of 10% (w/v) sodium carbonate. The mixture was shaken for 2 h at room temperature and the absorbance was measured at 765 nm against a blank using ultraviolet-visible spectrophotometer. The concentration of total polyphenols contained was calculated by referring to a calibration curve obtained using gallic acid as standard. The results obtained are expressed in milligram of gallic acid equivalent per gram of dry matter (mg GAE/g of extract).
Antioxidant Activity
The antioxidant potency of Mae was evaluated in vitro using three tests, the DPPH, the ferrous ion-chelating ability assay, and total antioxidant potency test (total antioxidant capacity (TAC)).
DPPH Free Radical-Scavenging Assay
The DPPH free radical-scavenging activity of different extracts was determined as described by Bersuder et al. 1998. A volume of 500 μL of each sample was mixed with 375 μL of 100% ethanol and 125 μL of .02 mM DPPH in 100% ethanol. The mixtures were then kept for 60 min in dark at room temperature, and the reduction of DPPH radical was measured at 517 nm using a UV–visible spectrophotometer. The control was conducted in the same condition, except that distilled water was used instead of sample. DPPH radical-scavenging activity was calculated as follows
where
Ferrous Ion-Chelating Ability Assay
The ferrous ion-chelating ability was determined according to the method of Decker and Welch.16,17 100 μL of Mae solution were mixed with 50 μL of 2 mM FeCl2 and 450 μL of distilled water. The reaction was initiated by the addition of 200 μL of 5 mM ferrozine. The whole was left to react for 10 min at room temperature, and then the absorbance of the Fe2+-ferrozine complex of red or violet color was measured at 562 nm. The chelating activity of the antioxidant for Fe2+ was thus calculated according to the following formula
where A0 is the white absorbance; Ac is the sample absorbance; and Ah is the absorbance in the presence of the sample. The control was carried out in the same way, except the use of distilled water instead of the sample.
Total Antioxidant Capacity Test
The total antioxidant activity of Marrubium vulgare aqueous extract was evaluated by the phosphomolybdenum method. 18 The assay is based on the reduction of Mo (VI)–Mo(V) by antioxidant compounds and the subsequent formation of a green phosphate/Mo(V) complex at acidic pH. A total of .3 mL of sample extract was combined with 3 mL of reagent solution (.6 M sulfuric acid, 28 mM sodium phosphate, and 4 mM ammonium molybdate). The tubes containing the reaction solution were incubated at 95°C for 90 min. Then, the absorbance of the solution was measured at 695 nm using a spectrophotometer against the blank after cooling to room temperature. Distilled water (.3 mL) instead of extract was used as a blank. A Trolox solution at different concentrations (from .5 to 5 mg Trolox/g of dry extract.) was used for the calibration.
Total antioxidant activity is expressed as equivalent antioxidant using the following equation
where A 695 is the absorbance at 695 nm, [C Trolox ] is the equivalent concentration of Trolox (mg/g), and a and b, respectively, are the origin and the slope of the calibration line.
Animals
Healthy adult male Wistar rats weighting 200–260 g were provided by the animal farmhouse of the High Institute of Biotechnology of Beja (University of Jendouba) and were cared for according to the principles of the local Ethics Committee on Animal Welfare in accordance with the recommendations of the International Council of Laboratory Animal Science. They were provided with standard food (standard pellet diet) and water ad libitum and maintained in animal house under controlled temperature (22±2°C) and on 12/12 h light/dark cycle.
Establishment of BLM-Induced Pulmonary Fibrosis and Marrubium vulgare Treatment
A total of thirty rats were randomly divided into three groups: the normal control group (G1), n = 10: received .9% saline (orally 2 mL/kg body weight “bw”); the BLM model group (G2), n = 10: received an intratracheal instillation of bleomycin solution (2 mg/kg body weight), 3 days later they received by gavage .9% saline (2 mL/kg bw) once daily for 20 days; and the treated group (G3) n=10: received BLM solution intratracheally (2 mg/kg bw), 3 days later they were treated by Marrubium vulgare aqueous extract (Mae) by oral gavage (2 mL/kg bw) once a day for 20 days. For all studied groups, daily gastric gavage was performed each morning at 9 a.m.)
For fibrosis induction, after weighing, G2 and G3 rats were anesthetized by intraperitoneal injection of pentobarbital sodium solution (75 mg/kg bw) (Sandoz laboratory, France). Each anesthetized rat was immediately suspended from a gallows. Induction of fibrosis was done by intratracheal injection of 2 mg/kg b w of bleomycin (Bleomycin®, Laboratories Aventis, France) in 200 μL saline. 19
After the various initial treatment during 20 days, all rats were anesthetized with the same procedure described during the induction of fibrosis and then were euthanized by injection of a lethal dose of sodium pentobarbital (200 mg/kg bw, Nembutal VR Ceva Animal Health). Several samples were taken; blood samples were collected by cardiac puncture and centrifuged at 5000 r/min for 10 min at 4°C. The serum was stored at −80°C for subsequent assays.
Following the sacrifice, the abdominal cavity and the chest were opened. Some lobes of the right lung were used for analysis of the cells of the bronchoalveolar lavage fluid; Balf samples were obtained by intratracheal injections of saline (4 _ 5 mL) via a catheter and re-aspiration of the liquid between two fractions of Balf. The lobes of left lungs were fixed by intratracheal injection of a 10% formalin solution (6 to 8 mL) and immersed in formalin for 48 h before histological examination. In addition, the rest of the lung, liver, and kidney tissues were removed and used for a biochemical determination of protein, malondialdehyde (MDA) levels, as well as antioxidant enzyme activities (SOD and CAT).
During the experiment, the body weight of rats was measured every 7 days, and the weight gain of rats in each group was compared. The ratio of net lung weight (mg) to the bw (g) for each rat was used as the lung index. 4
Pathological Examination
Tissue samples from left lungs were placed in formalin, dehydrated in a graded series of ethanol, embedded in paraffin, cut into 4 mm thick serial sections, and stained with hematoxylin and eosin to identify inflammatory cells and Masson’s trichrome to identify collagen deposition. Histological grading of fibrous lesions was performed using a blind semi-quantitative scoring system adopted by Aschroft et al.,20,21 for the extent and severity of inflammation and fibrosis in lung parenchyma. The severity of inflammation was categorized as one of the following: Grade 0, absence of inflammation; grade 1, minimal inflammation; grade 2, minimal to moderate inflammation; grade 3, moderate inflammation with thickening of the alveolar walls; grade 4, moderate to severe inflammation; and grade 5, severe inflammation with the presence of follicles. The severity of interstitial fibrosis was determined using the grading system, described by Ashcroft et al.20,21 The entire lung section was observed at _100 magnification and a score ranging from between 0 (normal) and 8 (total fibrosis) was assigned. The categories of pulmonary fibrosis were as follows: Grade 0, normal lung; grade 1, minimal fibrous thickening of alveolar or bronchial walls; grades 2 to 3, moderate thickening of walls without obvious damage to lung architecture; grades 4 to 5, increased fibrosis with definite damage to lung architecture and formation of fibrous bands or small fibrous masses; grades 6 to 7, severe distortion of structure and large fibrous areas, “honeycomb lung” was placed in this category; and grade 8, total fibrotic obliteration of the field. The mean score of all sections was taken as the fibrosis score of that lung section. Micrographics were captured using a Yujie microscope Y-2016T-LCD.
Bronchoalveolar Lavage
The Balf samples were centrifuged at 3000 r/min for 5 min at 4°C, the supernatant was discarded, and the cell pellet was resuspended with 50 μL of saline solution. 10 μL of the cell suspension was pipetted and the total cells were counted with a hemocytometer. Then, 30 μL of cell suspension was pipetted, and cell smears were prepared and stained with Wright’s-Giemsa to distinguish different types of cells under the light microscope. 22
Lipid Peroxidation and Antioxidant Enzymes Activities Measurements
Tissue (lung, liver, and kidney) peroxidation and serum lipid peroxidation were determined by malondialdehyde (MDA) measurement, as an indicator of oxidative stress using the 2-thiobarbituric acid (TBA) method. 23 This method involves the reaction of TBA with MDA, which can then be detected spectrophotometrically at 532–535 nm. The MDA levels were determined by using an extinction coefficient for MDA–TBA complex of 1.56 105 M/cm and expressed as nmol/mg protein. The activity of superoxide dismutase (SOD) was determined using modified epinephrine assays. 24 At alkaline pH, superoxide anion O2− causes the autoxidation of epinephrine to adenochrome, while competing with this reaction, SOD decreased the adenochrome formation. One unit of SOD is defined as the amount of the extract that inhibits the rate of adenochrome formation by 50%. Enzyme extract was added in 2 mL reaction mixture containing 10 mL bovine catalase (CAT) (.4 U/mL), 20 mL epinephrine (5 mg/mL) and 62.5 mM sodium carbonate/bicarbonate buffer, pH .2. Changes in absorbance were recorded at 480 nm. The activity of CAT was assessed by measuring the initial rate of H2O2 disappearance at 240 nm. 25 The reaction mix contained 33 mM H2O2 in 50 mM phosphate buffer, pH 7.0, and the activity of CAT was calculated using the extinction coefficient of 40 mM/cm for H2O2.
Protein Determination
Protein content was assayed as described by Lowry et al. 26 using bovine serum albumin as standard. The Lowry Assay “Protein by Folin Reaction” has been the most widely used method to estimate the amount of proteins (already in solution or easily soluble in dilute alkali) in biological samples. First, proteins are pre-treated with copper ion in alkali solution, and then aromatic amino acids in the treated sample reduce the phosphomolybdic acid and phosphotungstic acid present in the Folin reagent. The end product of this reaction has a blue color. The amount of proteins in the sample can be estimated via reading the absorbance (at 750 nm) of the end product of the Folin reaction against a standard curve of a selected standard protein solution (in our case; Bovine Serum Albumin-BSA solution).
Statistical Analysis
Experimental data was performed with IBM SPSS 25.0 software for statistical analysis. Data were presented as mean ± standard error of the mean (SEM). Statistical differences between the groups were analyzed with one-way analysis of variance (one-way ANOVA) followed by the post hoc LSD test. P < .05 was considered statically significant.
Results
Reversed Phase Ultra-Performance Liquid Chromatography Analyses of Mae Composition
Reversed phase ultra-performance liquid chromatography is an advanced analytical procedure used to identify the mean polyphenols present in Mae. As shown in Figure 1 and Table 1 results of RP-UPLC analysis showed a phenolic abundance whose main nine peaks correspond to the following polyphenols: Gallic acid, protocatechuic acid, catechin, chlorogenic acid, caffeic acid, epicatechin, vitexin, rutin, and quercetin with a retention time of 12.213 min, 14.196 min, 21.371 min, 21.909 min, 22.999 min, 28.710 min, 30.554 min, 36.730 min, and 42.038 min, respectively. Reversed phase ultra-performance liquid chromatography profile of polyphenols (λ = 280 nm) from M. vulgare aqueous extract. Peaks: 1. Gallic acid, 2. protocatechuic acid, 3. catechin, 4. chlorogenic acid, 5. caffeic acid, 6. epicatechin, 7. vitexin, 8. rutin, and 9. quercetin. List of Reversed Phase Ultra-Performance Liquid Chromatography Identified Metabolites in M. vulgare Aqueous Extract.

Total Polyphenol Content
Total Polyphenols Content of the Aqueous Extract of M. vulgare L.

DPPH Radical-Scavenging Activity
Scavenging of DPPH is accomplished by transferring hydrogen/electron from a given antioxidant to DPPH. The principle is based on the conversion of DPPH from the radical form to the non-radical form DPPH-H by feeding hydrogen atoms from antioxidant molecules to reduce an alcoholic solution to a stable free radical DPPH. When compared to Trolox, which has a constant percentage of free radical inhibition (ranging from 93.57 at .5% mg/mL to 99.80% at 5 mg/mL), Mae’s percentages of free radical inhibition increased with concentration (from 67.46% at a concentration of .5 mg/mL to 89.62% at a concentration of 5 mg/mL). Under the influence of Mae, the absorbance of the DPPH radical was significantly reduced, showing Mae’s significant antioxidant potential (Figure 2). Antioxidant activities: (A) Scavenging activity of M. vulgare extract (at final concentration ranging from .5 to 5 mg/mL) on the DPPH radical. (B) Effect of Mae on chelating ability for Fe2+ ions. Negative control, without antioxidant or. extract; values presented as mean ± SD. (C) The total antioxidant capacity of the M. vulgare aqueous extract.
Ferrous Ion-Chelating Ability Assay
The absorbance of the Fe2+-ferrozine complex increased linearly by the extract in a dose-dependent manner. From a concentration of 3 mg/mL, 100% metal-chelating activity is obtained, which indicates that the extract had significant chelating activity and captured ferrous ions. The metal scavenging effect of BHA under these conditions was used as a positive control and had a high chelating activity approaching that of Mae (Figure 2).
Total Antioxidant Capacity
The standard used is ascorbic acid at different concentrations and the expression of total antioxidant capacity is in milligrams of Trolox equivalent per gram of sample (Mg Trolox/g Extract). The antioxidant power rate of the Mae was obtained from Trolox calibration curve. The obtained results showed an important antioxidant activity of this Mae (3.5 ± .09 mg in Trolox equivalent/mg DW at a concentration of 5 mg/mL) (Figure 2).
Mortality and Morbidity
Throughout the experiment, we did not record any deaths in all the studied rats. The morbidity of the rats decreased in the days following the induction of fibrosis (BLM). In addition, no toxicity or adverse side effects were observed from gastric gavage treatment with Mae.
Effects of Mae Treatment on Rat Body Weight Variations and the Lung Index
The follow-ups of the changes in body weight were calculated as follows: [(final weight _ initial weight)/final weight] × 100. The findings revealed a slight but substantial variation in body weight changes between the investigated groups, particularly within the first 5 days after fibrosis induction. Weight gained steadily in all groups, with a higher gain beginning in week two and then stabilizing. The lung index was calculated using the ratio of net lung weight (mg) to body weight (g) for each rat and was compared between the study groups. The latter was significantly higher in the BLM group rats than in the control group, according to the findings. The group given Mae, on the other hand, saw a considerable fall in this indicator, bringing it back to the level of the control group (Figure 3). Effect of Mae and/or BLM treatment on rat body weight (A) and lung index (B). Number of rats, n = 10/group. Results are presented as the mean ± SD. *P < .05 vs C group. Mae, Marrubium vulgare aqueous extract; BLM, bleomycin; ANOVA, analysis of the variance.
Histological Analysis
To verify the effects of Mae on lung tissue damaged by fibrosis induced by BLM, two stains: H&E and trichrome were performed to evaluate the pulmonary parenchyma changes caused by this pathology.
The results obtained showed that the control group is characterized by an intact pulmonary tissue structure without any alveolar thickening or dilation of the bronchi.
In contrast, the pulmonary tissue structure of the rats in the BLM group was severely damaged and characterized by significant peribronchic, intra-alveolar and interalveolar inflammation, dilated and cystized bronchi, very dense inflammatory infiltrate, complete disappearance of the structure of alveoli, and alteration of the epithelial lining. Thus, an abundance of inflammatory cells (inter- and intra-alveolar plasma cells, lymphocytes, polynuclear neutrophils, macrophages, etc.), coupled with a proliferation of fibroblasts compared to the control group (Figure 4). Evolution of histological alterations in rat lungs after BLM instillation and/or Mae treatment (A) Hematoxylin and eosin–stained lung tissue (magnification, ×100; scale bar, 10 μm). (B) Masson’s trichrome–stained lung tissue (magnification, ×100; scale bar, 10 μm). (C) Effect of Mae and/or BLM treatment on the fibrosis score in rat lungs. (d) Effect of Mae and/or BLM treatment on the inflammation score in rat lungs. A. Control group; B. bleomycin group; and C. Mae group. (n = 10, X ± S); *: P < .05 vs control, #: P <.05 vs bleomycin and control groups.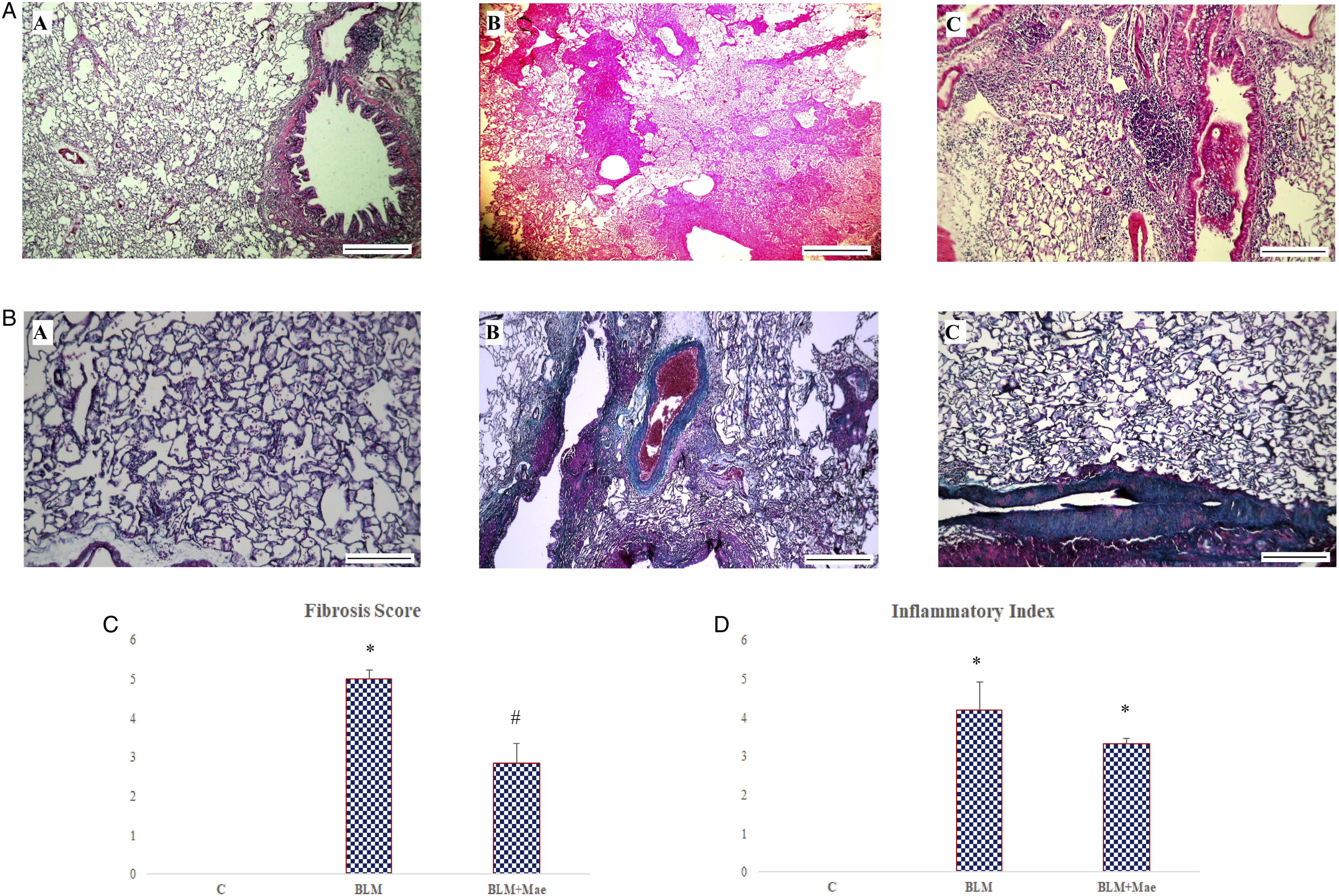
The Mae-treated group showed a little improvement in lung tissue characterized by focal interalveolar fibrosis with an inflammatory infiltrate intra-alveolar, peribronchial, plasma cells, rare lymphocytes, and very widespread pigmented macrophages.
For Masson’s trichrome stain, the sections confirm the H&E results with a conservation of the tissue structure of the control group showing a thin layer of collagen on the bronchial wall. Also, the BLM group confirms the installation of fibrosis in the pulmonary tissue justified by a deposit of collagen in the bronchial wall and interstitial lung and at the level of the intra- and interalveolar septum (Figure 4).
The results of the Mae-treated group showed a weak improvement in the distribution of collagen which was shown to be local with the good conservation of tissue of few pulmonary regions especially in the inflammatory infiltrate.
Estimation of Bronchoalveolar Lavage Cells
Bronchoalveolar lavage fluid was collected and the number of total cells as well as the number of inflammatory cells (lymphocytes and monocytes) were counted.
The results obtained showed that the total number of cells in Balf is higher in the Mae group (43.66 ± 7.28) compared to the BLM (35.91 ± 2.81) and C (12.08 ± 2.39) groups.
Regarding BALF’s monocytes and lymphocytes, their numbers were increased under the influence of BLM (respectively, 11±1.87; 22.5±10.25) compared to the control group (3.5 ± 1.04; 8 ± .89). In contrast, treatment with Mae dramatically reduced the number of lymphocytes (11.66±4.63) and reset the number of monocytes (3.66±1.032) to values similar to the control group.
Results are presented on the histogram (Figure 5) prove that Mae could remarkably inhibit the formation of inflammatory cells in Balf which suggests an attenuation of the damage caused by BLM-induced pulmonary fibrosis, in relation to the anti-inflammatory effects of this remedy. Counts of the total and the individual number of immune cells in Balf. (n = 10, X ± S); *: P < .05 vs control, #: P < .05 vs bleomycin and Mae groups.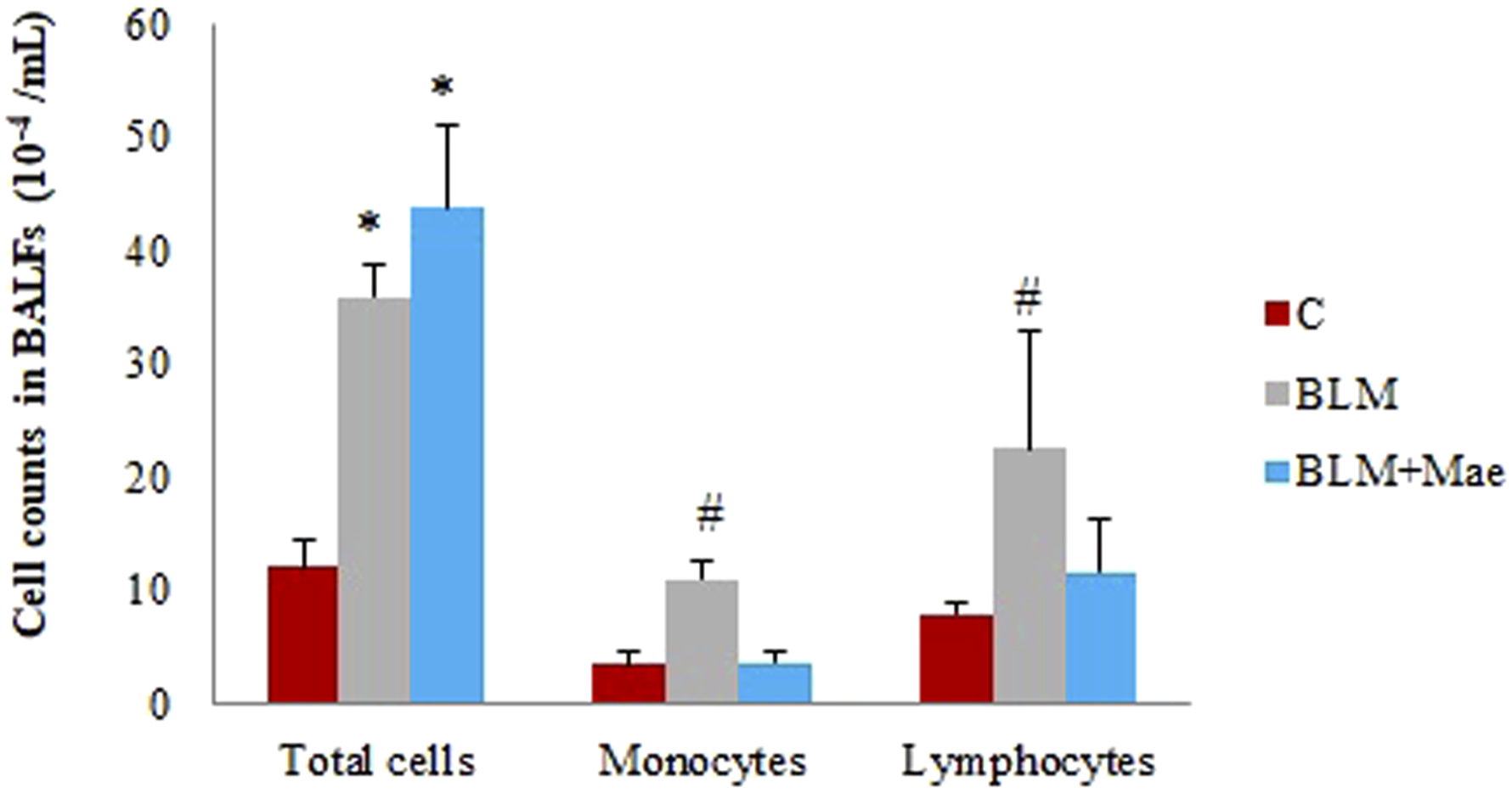
Lipid Peroxidation and Antioxidant Enzymes Activities Measurements
Effects of Mae and/or Bleomycin Treatment on Lipoperoxidation and Protein Level in Rat Lungs, Kidney, and Liver.
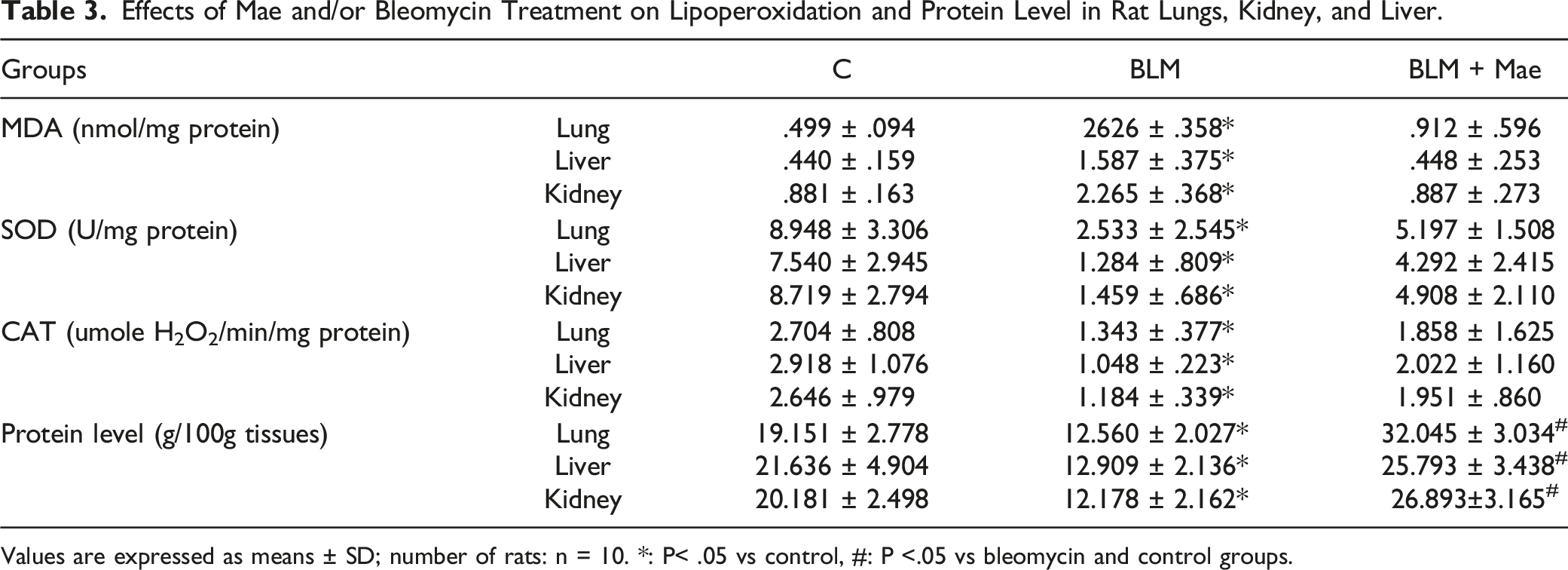
Values are expressed as means ± SD; number of rats: n = 10. *: P< .05 vs control, #: P <.05 vs bleomycin and control groups.
On the other hand, tissue samples from the lungs, liver, and kidneys showed significantly lower levels related to superoxide dismutase (SOD) activities as well as catalase in the BLM group compared to other groups (P < .05). The Mae-treated group showed a correction of the oxidation–reduction processes caused by BLM; Mae restored the antioxidant status of SOD and CAT as that of the control group in all studied tissues.
Protein Determination
The measurement of proteins in the different tissues according to the method of Lowry et al. demonstrated a clear decrease in protein levels in the samples of the BLM group caused by bleomycin instillation. In contrast, treatment with Mae increased these rates which were even higher than those in the control group (Table 3).
Discussion
Pulmonary fibrosis is the most serious and common pulmonary disease, characterized by progressive perforation of the alveoli, causing inflammation and scarring of the lungs. As a result, the lungs become rigid and lose elasticity, and the alveolar wall becomes thick and gradually prevents alveolar–capillary exchange. 27
One of the major consequences caused by pulmonary fibrosis is the abnormal and excessive production of collagen coupled with that of fibroblasts in the extracellular matrix. 28
This pneumopathy remains one of the most serious poorly understood lung diseases and still without any effective treatment until today.
In this way, our study aims to test the effectiveness of the aqueous extract of Marrubium vulgare (Mae) on an experimental model of pulmonary bleomycin-induced fibrosis in rats.
Moreover, the choice of Mae is based above all on its anti-inflammatory and antioxidant effects played by the high content of total polyphenols (73.619 mg EGA/g extract compared to other horehound extracts of different origins: Serbia; 54.65 ± .87; 29 Morocco; 5,45 ± 0,08, 30 and Algeria; 48.62 ± 1.73 31 ).
The identification of the main polyphenols present in horehound infusion was carried out by adapted RP-UPLC technique, the latter showed high levels mainly of catechin, chlorogenic acid, and caffeic acid, known for their characteristics of being extremely well absorbed at the intestinal level and especially for their antioxidant, 32 anti-inflammatory, antimutagenic, antihypertensive, 33 antidiabetic effects, as well as their implications in the reduction of oxidative stress and the stimulation of blood platelet reactions. 34
The benefits of this natural remedy have been confirmed by an in vitro evaluation of the antioxidant activity, based on the tests of DPPH radical-scavenging activity, ferrous ion-chelating ability, and total antioxidant capacity, having proven the indisputable antioxidant contribution of extract of horehound consistent with the results of Ettaya et al. 35
Studies in the literature on pulmonary fibrosis have linked it with significant variations in lung index and body weight; the calculation of the lung index showed excessive increases in the BLM group when compared with the control group; thus, a remarkable temporary decrease in body weight just after bleomycin instillation was identified, reflecting the damage following inflammation and general state of stress caused by this anticancer agent (BLM). Following Mae treatment, the lung index and the variations in body weight were returned to values close to those of the control rats (group C).
On the other hand, previous studies have often attached pulmonary fibrosis to an imbalance of the metalloproteinase balance caused by the abnormal production of collagen and the problems of its degradation. The resultant state is oxidative stress, reflecting a deregulation of the oxidation/antioxidant balance, caused by oxygen-reactive species (ROS) in the whole organism, especially in lung, liver, and kidney tissue. 36 In our study, we followed the variations of antioxidant and oxidant enzymes; superoxide dismutase (SOD) and catalase, and the latter underwent a significant decrease under the effect of BLM against an increase in MDA activity in all tissues studied. 37
In fact, these enzymes (SOD and CAT) have a complementary action on the radical cascade at the level of O2•− and H2O2, ultimately leading to the formation of water and molecular oxygen, but the disproportionation of O2•− is quite long, which allows the latter to either oxidize components of biological macromolecules (nucleic acids, proteins, etc.), or generate other reactive oxygen species that are much more toxic than the superoxide itself. Indeed, the liver is the central organ of this production and the hepatic microsomes generate more than 80% of H2O2 produced, which in turn could be considered as a potentially toxic reactive derivative of O2 and involved in the generation of the state of stress. 38
The attenuated levels of these enzymes in the studied tissues were confirmed by the increase in the levels of MDA resulting mainly from the action of ROS on polyunsaturated fatty acids; it is a powerful marker of lipid peroxidation in cells. In addition, the oxidation of LDL is directly linked to immune cells such as monocytes, macrophages, and lymphocytes, by transforming them into foam cells, an initial step in the inflammatory process. 39
As well, the treatment with Mae reversed this state of stress by bringing the activities of these enzymes close to that measured in the tissues of the control rats. This proves a possible anti-inflammatory and antioxidant potential of this extract exhibiting a power against the damage caused by BLM-induced fibrosis. These deductions can be explained, in part, by the high levels of protein in the different studied tissues (lung, kidney, and liver), highlighting the protein power in maintaining the cellular structures of the cited tissues. In addition, proteins are enormously involved in immune reactions as oxygen carriers (hemoglobin), immunoglobulin (antibodies), receptors, and hormones. 40
According to previous results in the literature, the bronchoalveolar lavage fluid (Balf) collected from the lungs of rats in the BLM group showed an increased number of inflammatory cells after BLM-intratracheal administration. 22 The resulting lung inflammation is a cellular process, characterized by the infiltration of inflammatory cells (such as lymphocytes, macrophages, and monocytes) into the alveolar space and the interstitial tissue of the lung. These inflammatory cells confirm the onset of pulmonary fibrosis by stimulating the production of soluble mediators capable of initiating and amplifying local inflammation, followed by the secondary activation of resident fibroblasts. This change (increase in number of lymphocytes and monocytes) was attenuated in the samples of the Mae-treated group. Also, the number of total cells counted in the Balf was considerably increased in the Mae group, suggesting the implication of this remedy in the inhibition or suppression of inflammatory factors and by preserving almost all the cells of the alveolar–capillary wall and the alveolar lumen. 31
These results were confirmed by those of the histological study which showed histopathologic changes representing the most specific and characteristic variables accompanying pulmonary fibrosis. In this context, the microscopic observations following the HE and TR staining showed an enormous arrangement of collagen especially at the level of the alveolar septum, inflammatory infiltrate, and histiocytes in the BLM group. This later characterized by the activation of pulmonary fibroblasts and their differentiation into myofibroblasts which is a key step in the development of IPF. 41 These myofibroblasts represent the major factor for the secretion of a large quantity of proteins of the extracellular matrix (fibronectin, laminin, and collagen). Accumulation especially of collagen and its deposit in this extracellular matrix will be major origin of a disorganization of the latter and which accelerates the installation of the IPF. 28 The thickening of the ECM and the inhibition of its degradation causes the hardening of the alveolar–capillary exchange wall which is the source of the difficulties in the exchange of respiratory gases with the blood circulation, as well as a weakening of pulmonary compliance.
This damage was mitigated under the effect of Mae, which could inhibit the expression of inflammatory markers (monocytes, lymphocytes, fibrocytes, macrophages, etc.) in the lung tissue of Mae-treated rats. This moderating effect of horehound extract on FP is probably due to its high content of polyphenols and flavonoids, whose antioxidant, anti-inflammatory, anti-atherogenic, antihypertensive, anti-diabetes, and anti-depressant effects have been confirmed in many studies of the literature.42-44
Conclusion
Results of this study were encouraging. This natural remedy (Mae) significantly reduced the damage caused by pulmonary fibrosis in rats. Moreover, this potential action related, above all, to the inhibition of the inflammatory response (Balf cells, fibrosis score, and inflammatory index) and the improvement of the antioxidant properties is possibly due to the richness of this extract in polyphenols. Ultimately, this work may shed light on the mechanism of action of horehound in the prevention and treatment of pulmonary fibrosis and further studies are needed to identify the active compounds responsible for the anti-fibrotic effects.
Acknowledgments
The authors are grateful to all persons who helped to conduct this study.
Footnotes
Author’s Contributions
Abidi A ensured the preparation of the experimental protocol and its realization, was involved in all the analyses carried out, wrote the manuscript, and interpreted the results obtained.
Abidi A is identified as the guarantor of the paper, taking responsibility for the integrity of the work as a whole, from inception to published article.
Dhaouefi J ensured the chemical analysis of the natural extracts by the UPLC technique, antioxidant activities, and the protocol realization.
Brinsi J ensured the bronchoalveolar fluid analysis and the protocol realization.
Tounsi H read the HE and trichrome histological sections and is involved in the interpretation of its results.
Sebai H participated in revision and correction of manuscripts.
All authors have reviewed and approved the submission of this manuscript version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This scientific work was funded by the Tunisian Ministry of High Education and Scientific Research.
Ethics Approval and Consent to Participate
Animals were cared for according to the principles of the local Ethics Committee on Animal Welfare (University of Jendouba: UJ2021-03-4022) in accordance with the recommendations of the International Council of Laboratory Animal Science.