Abstract
Introduction
Bleomycin is an effective chemotherapeutic agent with main side effects including lung fibrosis which limited its clinical use. The aim of this study is to evaluate the protective effect of grape seed and skin extract (GSSE) against bleomycin-induced oxidative damage and inflammation in rat lung, by assessing respiratory index (RI), oxidative and nitrosative stress (SOD and XO activity, NO), fibrotic mediators (hydroxyproline and collagen), apoptosis (cytochrome C and LDH), inflammation (IL-6, TNF-α and TGF-β1), and histological disturbances.
Methods
Rats were pre-treated during three weeks with vehicle [ethanol 10% control] or GSSE (4 g/kg) and then administered with a single dose of bleo (15 mg/kg bw) at the 7th day.
Conclusion
Data suggest that GSSE exerts anti-oxidant, ant-inflammatory, and anti-fibrosis properties that could find potential application in the protection against bleo-induced lung fibrosis.
Introduction
Bleomycin (bleo) is an anthracycline antibiotic largely used as a chemotherapeutic agent in the treatment of a variety of human cancers such as lymphomas, head and neck cancer, and testicular and ovarian cancer. 1 The therapeutic options for bleo-induced lung toxicity are very limited and lung inflammation is a major underlying component of a wide variety of pulmonary fibro-proliferative disorders.
Pulmonary fibrosis constitutes a severe form of lung toxicity, characterized by altered cellular composition of the alveolar region with excessive deposition of collagen. 2 Although the mechanism of bleo-induced lung fibrosis is still unresolved, 3 it is generally admitted that upon binding to DNA and in the presence of catalytic iron, bleo is able to generate ROS and RNS that induce DNA damage, initiating the inflammatory and fibro proliferative change via a concerted action of various cytokines leading to collagen accumulation and finally cell death by both apoptotic and necrotic pathways. There are currently few treatments available for pulmonary fibrosis. Therefore, identification of new therapeutic approaches is of critical importance.
GSSE is a complex polyphenolics mixture containing bioactive components such as proanthocyanidins, flavonoids, and stilbenes 4 exhibiting potent pharmacological properties against various biotic or abiotic stressors, inducing multi-organ dysfunction such as cardiotoxicity, nephrotoxicity, hepatotoxicity, and neurotoxicity. 5 GSSE is also protective against toxicity linked to the use of antineoplastic drugs as doxorubicin 6 or cisplatin 7 and exerted beneficial effects on bleo-induced lung fibrosis. 8 Proanthocyanidins from grape seeds are endowed with anti-inflammatory properties on experimentally induced inflammation in rodent, and their mechanism of action is likely linked to free radical scavenging activity, metal chelation, antioxidative, and mitigation of pro-inflammatory processes.
The aim of our study is to evaluate the preventive role of GSSE (Li et al, 2001)against bleo-induced lung toxicity and fibrosis, thanks to its antioxidant and anti-inflammatory properties.
Material and Methods
Chemicals
Trichloroacetic acid (TCA) CAS 76-03-9, hydrogen peroxide (H2O2) CAS 7722-84-1, 2-methoxyphenol (guaiacol) CAS 90-05-1, bovine catalase CAS 9001-05-2 and 4-(1-Hydroxy-2-methylamino-ethyl)-benzene-1,2-diol (epinephrine) CAS 120003-76-1, and 2,4-dinitrophenyl hydrazine (DNPH) CAS 119-26-6 were purchased from Sigma-Aldrich Co (Germany).
Composition of GSSE
GSSE was processed from a grape cultivar (Carignan) of Vitis vinifera from northern Tunisia. Seeds and skin were dried and grounded separately with an electric mincer (FP3121 Moulinex) until a fine powder was obtained. Powder mixture containing grape seed (50%) and skin (50%) was dissolved in 10% ethanol at dark, vigorously vortexed for 15 min, and centrifuged at 10.000 g for 15 min at 4°C for debris elimination and supernatant containing soluble polyphenols was ready to use. Total phenolic content was determined by the Folin–Ciocalteu colorimetric method, 9 flavonoids and condensed tannins according to Dewanto et al 10 and Sun et al, 11 respectively. GSSE composition was established by HPLC–MS/MS analysis. Briefly, liquid chromatography was performed using a Perkin Elmer system series 200 equipped with a binary micro-pump. The analyses were carried out on a C18 column (Zorbax Eclipse XDB-C18, 4.6 _ 150 mm, particle size 5 mm). The mobile phase A was .1% formic acid in water and the mobile phase B was .1% formic acid in acetonitrile. Elution was performed at a flow rate of 1 mL min−1 and an injection volume of 20 μl with LOD .1 mg.L−1. Tandem mass spectrometry (MS/MS) was carried out using a 3200 QTRAP mass spectrometer (Applied Biosystems/MDS Sciex Forster city USA) equipped with an electrospray ionization (ESI) interface. Data were acquired and processed with Analyst 1.5.1 software. The detector was set in the negative ion mode. The ion trap mass spectrometer was operating in the m/z 50–1700 mass range.
Animals and Experimental Design
Twenty-four male Wistar rats (195-200 g) from Pasteur Institute (Tunis) were used in agreement with the ethics committee of Tunis University and with NIH guidelines. They were provided with food and water ad libitum and maintained in animal house at fixed temperature of 22 ± 2°C with a 12 h light–dark cycle.
Rats were randomly divided into four groups of six animals each that were daily treated by intraperitoneal (ip)route: Group 1: control: rats receiving 10% ethanol for 21 days; Group 2: GSSE: rats receiving 4 g/kg GSSE for 21 days; Group 3: bleo: rats receiving a single dose of bleo (15 mg/kg) at day 7; and Group 4: bleo + GSSE: rats treated both with GSSE and a single dose of bleo at day 7. At the end of the treatment, rats were anesthetized with urethane [40 mg/mL] and sacrificed and their blood was collected into heparinized tubes and processed for plasma biomarkers determination. Lungs were isolated, homogenized in PBS buffer pH 7.4 with an ultrathurax T25 homogenizator, and centrifuged (15 min at 10.000 g 4 C) and the resulting supernatant was used for lung lipidemia and oxidative stress analyses.
Function Measurements
A volumetric respirometer at constant pressure (Jeulin Everex, France) was used to determine respiratory index measured at 0, 7, and 13 days post-bleo treatment. 12
Oxidative Stress Parameters
The superoxide dismutase (SOD) activity (E.C.1.15.1.1) was determined according to Misra and Fridovich (1972). 13 Reaction mixture consists in 50 mM phosphate buffer Na2CO3/NaHCO3 pH 10, 10 μl lung supernatant, and 10 μl bovine catalase (0.44 U/ml). The reaction was initiated by the addition of epinephrine (20 μl) and the rate of reaction was measured by following changes in absorbance at 480 nm at 30 s interval for 5 min, and activity was expressed as U/min/mg protein.
Xanthine oxidase (XO) activity (EC1.1.3.22) was determined by monitoring uric acid formation from xanthine according to De Souza et al. 14 A .12 mL aliquot of xanthine solution (250 mM) was added to a test tube containing 10 μL lung homogenate and .54 mL potassium oxonate (1 mM) in 50 mM sodium phosphate buffer pH 7.4 that was previously warmed at 35°C for 15 min. The reaction was stopped after 30 min by addition of .1 mL .6 M HCl, and after centrifugation at 3000 g for 5 min, absorbance was measured at 295 nm, and XO activity was expressed as μmol uric acid formed/minute (U)/mg protein.
Lung GSH content was determined according to Ellman 15 using GSH reductase. Briefly, the reaction of GSH with Elman’s reagent 5, 5’-dithiobis-2-nitrobenzoic acid (DTNB) gives rise to a product that can be quantified spectrophotometrically at 405 nm, and results are expressed as nmol GSH/g tissue.
Measurement of NO
NO level was measured by the method of Green et al (1982), 16 using Griess reagent. .5 mL of lung homogenate was precipitated with 200 μl 30% sulphosalicylic acid, vortexed for 30 min, and centrifuged at 3000xg with an equal volume of Griess reagent containing 1% sulfanilamide in 5% phosphoric acid/0.1% naphthalene ethylene-diamine dihydrochloride that was incubated for 10 min in the dark. Absorbance was read at 543 nm using sodium nitrite as standard and NO level was expressed as mol nitrite/mg tissue.
Lung Hydroxyproline and Collagen Content
The hydroxyproline content was measured according to the method described in the hydroxyproline assay kit (obtained from Sigma-Aldrich Co (Germany). Briefly, 100 μL lung homogenate were added to 100 μL concentrated hydrochloric acid (12 M) into a microplate well and hydrolyzed at 120°C for 3 h. 50 μL of supernatant from each sample and standard well was evaporated to dryness under vacuum to which was added 100 μL of chloramine T/oxidation buffer mixture, incubated at room temperature for 5 min. After addition of 100 μL DMAB reagent to each sample and standard well and incubation for 90 min at 60°C, absorbance was measured at 560 nm using a microplate reader. Lung collagen content was determined based on hydroxyproline assay and expressed using the following equation:
Lung collagen content = hydroxyproline x13.5 17 and results are expressed as μg collagen per mg wet lung tissue.
Pro-Inflammatory Cytokines Measurement
IL-6 pro-inflammatory cytokine in lung tissue supernatants was determined using quantikine IL-6 R immunoassay (R&D Systems, Minneapolis, MN). TNF-α and TGF-β1 were determined using TNF-α ELISA kit (catalog number DY2279, R&D Systems, USA) and TGF-β1 ELISA kit (catalog number MB100B, R&D Systems, Minneapolis, MN), respectively, according to manufacturer’s protocols.
Cytochrome C and LDH Activity
The determination of cytochrome C, an important mediator of the apoptotic pathway, was carried out according to Margoliash and Schejter. 18 LDH activity (E.C.1.1.1.1.), which is a relevant biomarker of bleo-induced lung fibrosis, was determined according to Bergmeyer 19 using a commercial kit from Biomaghreb Tunisia.
Protein Determination
Total proteins were measured according to Lowry et al (1951) with BSA as standard. 20
Histopathology
Lung samples were collected and fixed in 4% (v/v) formalin, dehydrated with alcohol baths, and embedded into paraffin. Four micrometer sections were colored with hematoxylin–eosin (HE) and examined under light microscope.
Statistical Analysis
STATISTICA program (version 8.0) was used to develop the statistical analysis. All obtained results were described as mean ± standard deviation. The observed variation was examined by one-way ANOVA (P < .05). First of all, we verified the prerequisites for the analysis of variance (normality of distribution (Shapiro–Wilk test) and homogeneity of variances (Levene test). Once relevant modifications were found, Tukey’s test was applied to fix which values diverged significantly. (*) indicated significance vs control and (x) vs bleo.
Results
GSSE Composition
LC-MS/MS data of phenolic compounds found in GSSE.
Determination of Respiratory Index (RI)
Effect of bleo and GSSE on respiratory index.

Lung Oxidative and Nitrosative Stress
Bleo significantly decreased SOD enzyme activity by −27.66% (Figure 1A) and increased XO activity by +79.64% (Figure 1B). Bleo also increased GSH by +58.76% (Figure 1C) and NO level by +277.6% (Figure 1D) and GSSE restored efficiently these oxidative and nitrosative stress biomarkers close to control. Effect of GSSE on bleo-induced lung SOD [A] and XO [B] activities, GSH level [C], and NO metabolite [D]. Rats were daily treated with bleo with or without GSSE [4 g/kg bw]. Results are expressed as mean ± SEM. [n = 6] P < .05 was considered significant: * for bleo vs control, and § for bleo +GSSE vs bleo.
Lung Fibrotic Mediators
We further sought to determine the effect of bleo on lung hydroxyproline (Figure 2A) and collagen (Figure 2B). Bleo highly increased hydroxyproline by +361.01% as well as collagen by 360.63% which were significantly but only partially corrected by GSSE by −28%. Effect of GSSE on bleo-induced lung fibrotic mediators as hydroxyproline [A] and collagen [B].
Lung Inflammation
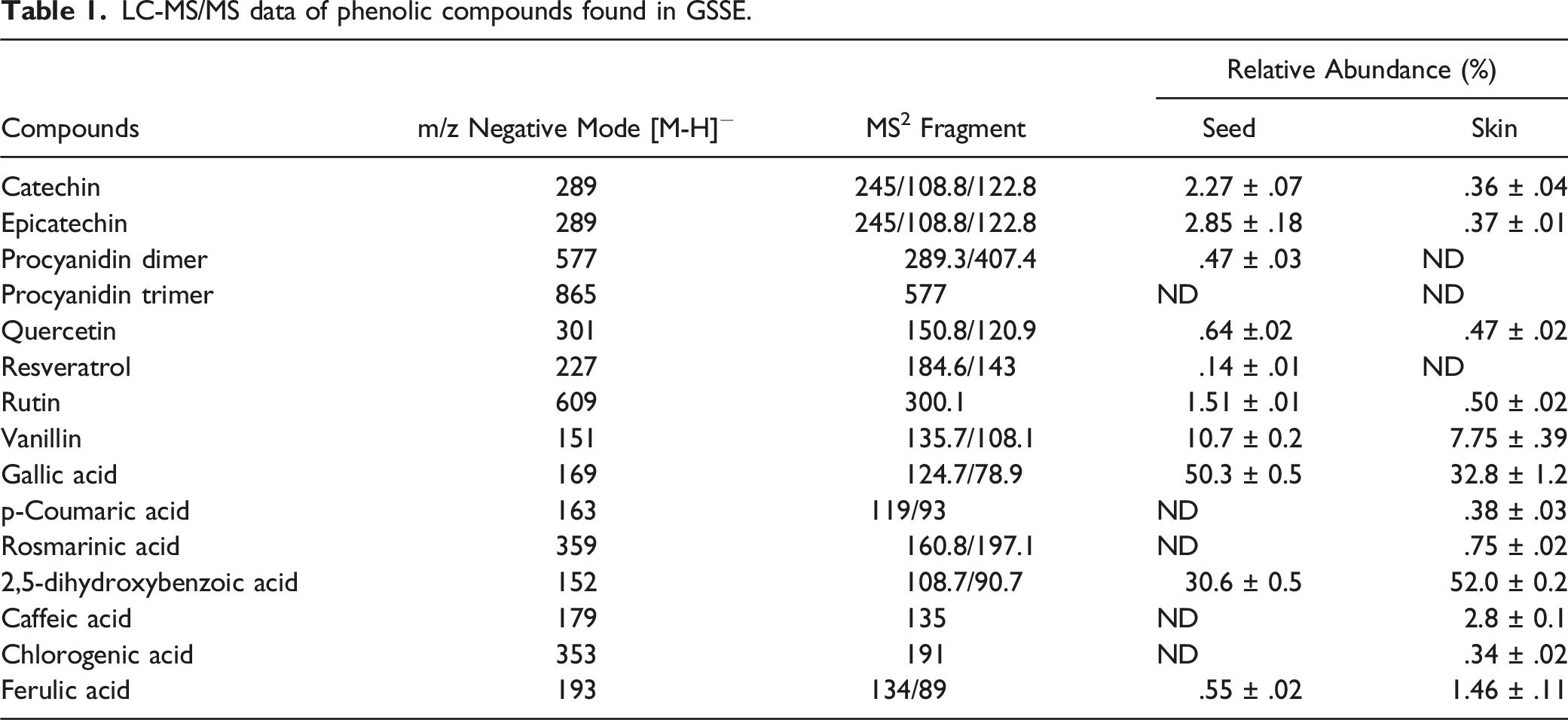
We also assessed the bleo-induced lung inflammation and found that bleo increased the pro-inflammatory IL-6 cytokine reaching +322% (Figure 3A), TNF-α by +280% (Figure 3B), and TGF-β1 by +313% (Figure 3C). GSSE efficiently corrected IL-6 till lower level than control, whereas GSSE corrected TNF-α and TGF-β1 close to control level. Effect of GSSE on bleo-induced lung inflammation: IL-6 [A], TNF-α [B], and TGF-β1 [C].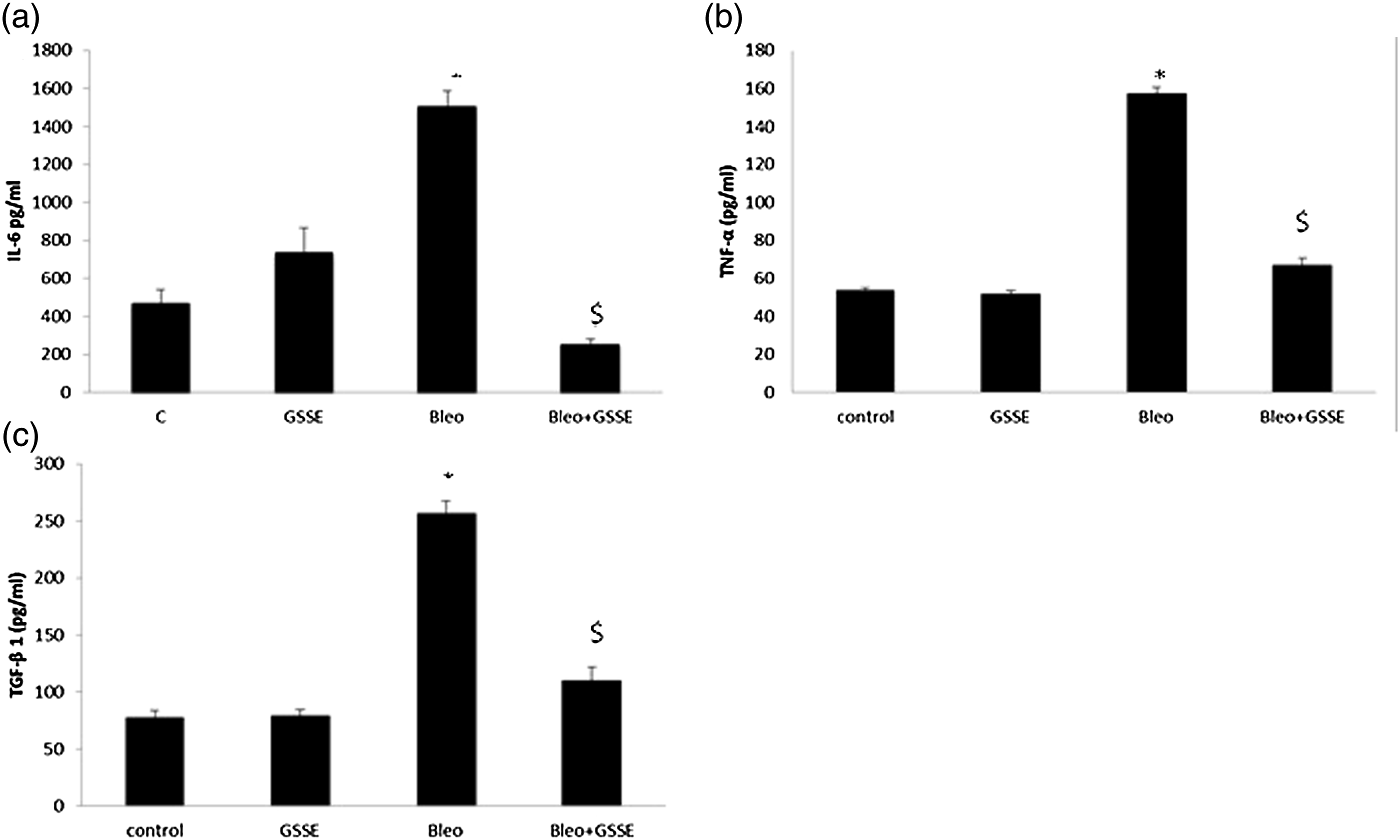
Lung Pro-Apoptotic Mediators
LDH level was significantly higher in bleo-exposed lung when compared to control (Figure 4A) which was abrogated till near control upon GSSE. Furthermore, bleo slightly increased cytochrome C activity (Figure 4B), and unexpectedly, GSSE which acted in concert with bleo, further increased the cytochrome C pro-apoptotic mediator activity. Effect of GSSE on bleo-induced lung pro-apoptotic mediators as LDH [A] and cytochrome C activity [B].
Histopathology
We also investigated the effect of bleo on lung architecture and found that bleo induced the thickening of alveolar septa as well as an interstitial infiltration by inflammatory cells, and GSSE reduced the severity of bleo-induced lung damages (Figure 5). Rat lung histology: control [A], bleo [B], and bleo + GSSE [C].
Discussion
Our study mainly showed that bleo altered overall lung architecture and respiratory index and provoked an oxidative/nitrosative stress and chronic inflammation, and an increase in hydroxyproline and collagen deposition finally leding to fibrosis.21,22
Oxidative stress is a prominent mechanism associated with the pathogenesis of lung fibrosis, 23 and in our case, bleo induced an oxidative stress status within the lung as assessed by depressed SOD activity and GSH level and increased XO activity.
Superoxide dismutase (SOD) catalyzes the conversion of superoxide free radical anions (O2−) to H2O2 24 and glutathione is an intracellular antioxidant that protected the lung from endogenous or exogenous lung toxicants. 25 Moreover, XO is a cytosolic metalloflavoprotein implicated in the pathogenesis of a wide spectrum of diseases, which is an important source of oxygen-free radicals and cell damage.
Allopurinol inhibition of XO to patients with chronic obstructive pulmonary disease was able to alleviate oxidative stress through lowering glutathione and 26 lipid oxidation. 27 Another study carried out on reactive nitrogen species (RNS) confirmed that anticancer drugs increased NO production and lung inflammation. 28 Furthermore, NO reacts with O2− to form peroxynitrite leading to acute lung injury.29,30
Bleo highly induced cytokines such as TNF-α, TGF-β1, and IL-6 which promote the progression to lung fibrosis as previously reported. 31 Interestingly, IL-6 eliciting both pro-inflammatory and profibrotic effect was shown to be elevated in mice suffering from pulmonary fibrosis.32,33
Besides inflammation, bleo induced cytotoxicity and apoptosis as indicated by increased LDH activity and release of cytochrome C to the cytosol. 34 Moreover, LDH is enhanced in rat lung upon intratracheal administration of bleo. 35
Lung neutrophils may also release pro-inflammatory and pro-apoptotic cytokines to injure adjacent cells, and our current study demonstrates high level of cytochrome C, a relevant marker for apoptosis that reflected the deleterious effect of bleo into the lung. Unexpectedly, our study showed that pretreatment with GSSE promoted cytochrome C activity, which is in disagreement with Song et al (2014) who demonstrated that astaxanthin, a powerful antioxidant, inhibits apoptosis of alveolar epithelial type II cells by suppressing the release of cytochrome C into the cytosol. 36 However, our data are in full agreement with the recently described synergistic effect of quercetin and apigenin with cisplatin leading to increased cytochrome C and apoptotic leukemia cell death. 37
In this context, recent studies have shown that the inhibition of Rho/Rho-kinase signaling decreases fibroblast expression of anti-apoptotic BCL-2 proteins and promotes IPF lung fibroblast apoptosis while decreasing lung fibrosis in vivo. 38 In our present case, bleo-treated animals were characterized by thickening of lung alveolar septa, inflammatory cell infiltration, and loss of alveolar–capillary membrane integrity, leading to breathing disturbance.
Undoubtedly, the most relevant result drawn from the current study is the protection afforded by high dosage GSSE against bleo-induced lung fibrosis. GSSE protection was demonstrated to occur through anti-inflammatory and anti-oxidative pathways that occurred in various experimental settings including prevention against lithium neurotoxicity 39 or arsenic cardiotoxicity. 40
Pretreatment with GSSE significantly reduced the level of hydroxyproline and collagen deposition as well as the severity of the histological signs, which are consistent with previous studies in the field.41,42 Other studies have shown that bleo increased lung hydroxyproline content which was reduced using various antioxidants as ellagic acid, 23 pyrrolidine dithiocarbamate, 24 and GSE. 43 Bleo-induced oxidative stress into the lung is associated with a higher risk of lung damage, and GSSE prevented lung depletion of endogenous antioxidant by acting either as ROS scavenger or enhancer of endogenous antioxidant.
Our results are in accordance with a previous study suggesting that ellagic acid pretreatment affected NO production into the lung treated with bleo or cisplatin. 23 Administration of curcumin to nicotine-treated rats decreased NO level into the lung, 44 as did GSSE to lithium-treated rats. 33 GSSE preventing bleo-induced lung IL-6, TNF-α, and TGF-β1 highlighted its anti-inflammatory ability. As bleo-induced ROS and pro-inflammatory cytokines, GSSE might also decrease ROS production in lung macrophages. Moreover, as GSSE significantly decreased lung LDH, this result fully corroborated previous data obtained with gossypol, an effective polyphenol at both preventing and treating bleo-induced pulmonary fibrosis, emphasizing LDH as a potential therapeutic target for pulmonary fibrosis. 45
In conclusion, due to its multitargeted effect and safety, high dosage GSSE should be tested in large scale trials either as nutritional supplement into various food recipes in a preventive and holistic approach of lifestyle-related diseases or alternatively packaged into nanoformulation to be used as a more suitable galenic form into chemotherapeutic protocol or tested as an adjuvant in clinical trials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Ministry of Higher Education, Carthage University, Faculty of Sciences of Bizerte, Laboratory of Bioactive Substances [LSBA], and the Centre de Biotechnologie de Borj Cedria, BP 901, 2050 Hammam-Lif, Tunis.
