Abstract
Introduction
Ionizing radiation (IR) has numerous beneficial implications in our daily lives but also has negative effects. It has long been recognized that ionizing radiations increase oxidative stress and trigger apoptotic processes and inflammatory factors, causing impairment in the cell function and structure.1,2 The results of extensive studies have demonstrated that direct and indirect mechanisms bring about the damaging effects of ionizing radiations. The direct action disrupts sensitive molecules, whereas the indirect effects (approximately 70%) arise from the interaction of ionizing radiation with molecules of water, the resulting generation of highly reactive free radicals, and the ensuing impact of these free radicals on intracellular substructure.3,4
Melatonin (N-acetyl-5-methoxytryptamine) is produced and emitted by the pineal gland and regulated by the dark/light cycle. 5 Melatonin is superior to traditional antioxidants due to its capacity to capture multiple types of free radicals, stimulate antioxidant enzymes, suppress pro-oxidant enzymes, and reduce inflammatory responses, thereby mitigating the adverse effects of radiation. 6 Unlike conventional antioxidants, melatonin does not undergo redox cycling; as a result, 1 molecule can scavenge up to 10 reactive oxygen species (ROS) as opposed to 1 or fewer reactive oxygen species for conventional antioxidants. 7 Its amphipathic character allows it to traverse cellular membranes and blood-organ barriers, thereby preventing oxidative injury to macromolecules. 8 Melatonin has received much attention because of its positive effects on some human diseases. It has a cardioprotective effect on ischemic heart disease clinical course in elderly patients with insomnia. 9 Also, It is used as an adjuvant treatment for COVID-19. 10 Recently, it was observed that melatonin reduced insulin sensitivity in human adipose tissue. 11 In addition, it was reported that melatonin modulated the female reproductive system by improving follicular morphology and granulosa cell proliferation and reducing apoptotic cells suggesting that melatonin promotes follicular development, maintains primordial follicles, and regulates the female reproductive function. 12 Melatonin has also been reported to regulate testicular development, reduce testicular injury, and maintain sperm function and morphological characteristics. 13
Many women diagnosed with cancer each year are subjected to radiation therapy treatment. The evidence has demonstrated that IR has a gonadotoxic action, including insufficient ovary function and consequent infertility. 14 Keeping ovarian integrity after treatment is a crucial prerequisite for reproductive capacity. Multiple investigations have demonstrated that melatonin, a lipophilic and hydrophilic hormone with potent antioxidant properties, could be utilized as a radioprotectant in wide-range safe doses (10-300 mg/kg),15-17 However, more research is necessary to evaluate the post-irradiation treatment’s mitigating effect. IR has a significant impact on reproductive potential; therefore, this study aims to examine the effect of post-irradiation administration of melatonin on oxidative stress in the ovaries and changes in the level of reproductive hormones and pro-inflammatory cytokines. In addition, this study seeks to evaluate melatonin’s potential role in restoring female reproductive health after gamma radiation exposure.
Materials and Methods
Experimental Animals
Female Wistar rats (3 months old; 120-150 g) were procured from the NCRRT’s animal house in Cairo, Egypt. Animals were supplied with a regular pellet diet and given unlimited access to water. The animals were housed on a 12-hour dark/light cycle, at a uniform temperature (20–25°C) and humidity (50 ± 5%). The entire experiment was executed in accordance with the National Institute of Health’s international guidelines for animal care and handling (NIH publication No. 85 – 23, 1996) and in compliance with the ARRIVE guidelines.
Radiation Facility
At the NCRRT in Cairo, Egypt, whole-body gamma-irradiation was performed with a Cesium (137Cs) source, Gamma Cell-40 biological irradiator. The rats received a single 4 Gy gamma ray dosage. 18 at a rate of 0.38 Gy/min. The irradiation process and dose rate calculation were done according to the Dosimetry and Protection Department at the National Center for Radiation Research and Technology at the time of the experiment.
Melatonin Treatment
According to 19 melatonin (10 mg/Kg body weight) was administered intraperitoneally to female rats for 7, 11, and 15 days consecutively, dispersed in water. Sigma-Aldrich (St. Louis, MO, USA) provided the melatonin.
Experimental Design
The Experiment (in vivo Study) was Continued for Fifteen Days and Done at the NCRRT, Cairo, Egypt
Animals were separated into 8 groups, each containing 6 rats (The sample size was determined using G power program version 3.1.9.6. With a power of 95% and alpha error probability = 0.05). Group 1 (Control) consists of normal healthy rats. Groups 2, 3 & 4 (Radiation): Rats were subjected to 4 Gy whole-body gamma radiation. Group 5 (Melatonin): rats were administered melatonin (10 mg/Kg body weight, i. p.) for 15 days. Groups 6, 7& 8 (Radiation + Melatonin): rats subjected to whole-body irradiation, and then injected with melatonin daily for 7, 11, and 15 days consecutively after irradiation.
Rats were sacrificed at 10 am on the 7th, 11th, and 15th days after irradiation. Blood samples were obtained from the rats by heart puncture. Serum was extracted from blood samples by centrifugation at 3000 r/min and kept at −20°C until biochemical investigations. The ovaries were removed, homogenized in ice-cold 0.1 M phosphate buffer, pH 7.4 (1:10 w/v), and spun for 10 minutes at 3000 r/min. Then, the supernatant was gathered and kept at −20 degrees Celsius until the biochemical examination.
Biochemical Analyses
Measurement of Oxidative Stress Markers, Malondialdehyde (MDA), Total Antioxidant Capacity (TAC), and Protein Carbonyl (PCO), in Ovaries
TAC was evaluated by TAC kit (MyBioSource, Inc. USA), Cat. No: MBS733414_48T. MDA was measured by MDA kit (LifeSpan BioSciences, Inc.), Cat. No. LS-F28018. PCO was measured using assay Kit Cat. No. MAK094 from Sigma Aldrich, St. Louis, Missouri.
Estimation of Apoptotic Marker and Pro-inflammatory Cytokines
Caspase-3 levels were evaluated in serum via a Rat Casp-3 ELISA Kit with Cat. No. CSB-E08857r from CUSABIO. Serum TNF-α was measured utilizing TNF alpha ELISA Kit (ab208348), IL-6 utilizing IL-6 ELISA Kit, and IL-1 beta utilizing IL-1 beta ELISA Kit (ab100705), Sigma Co.
Measurement of Reproductive Hormones
Serum luteinizing hormone (LH) and follicle-stimulating hormone (FSH) were evaluated by Mice/Rat ELISA kit (MyBioSource, Inc. USA), Cat. No: MBS017508 and MBS2509833, respectively. Serum estradiol, progesterone, and testosterone were measured by ELISA kit (Sigma-Aldrich) Cat. No: LS-F5297, CSB-E07282r and MBS282195, respectively.
Statistical Analysis
All of the numbers are given as Mean ± Standard Deviation (n = 6). To determine significant differences between means, data from experiments were examined using a one-way analysis of variance (ANOVA) followed by the LSD test as a Post Hoc. P < 0.05, 0.01, and 0.001 were chosen as the significant values.
Results
Effect of γ-Radiation in Rats
Effect of Melatonin on Oxidative Stress Markers, Malondialdehyde, and Protein Carbonyl and the Total Antioxidant Capacity, in the Ovary of Rats.
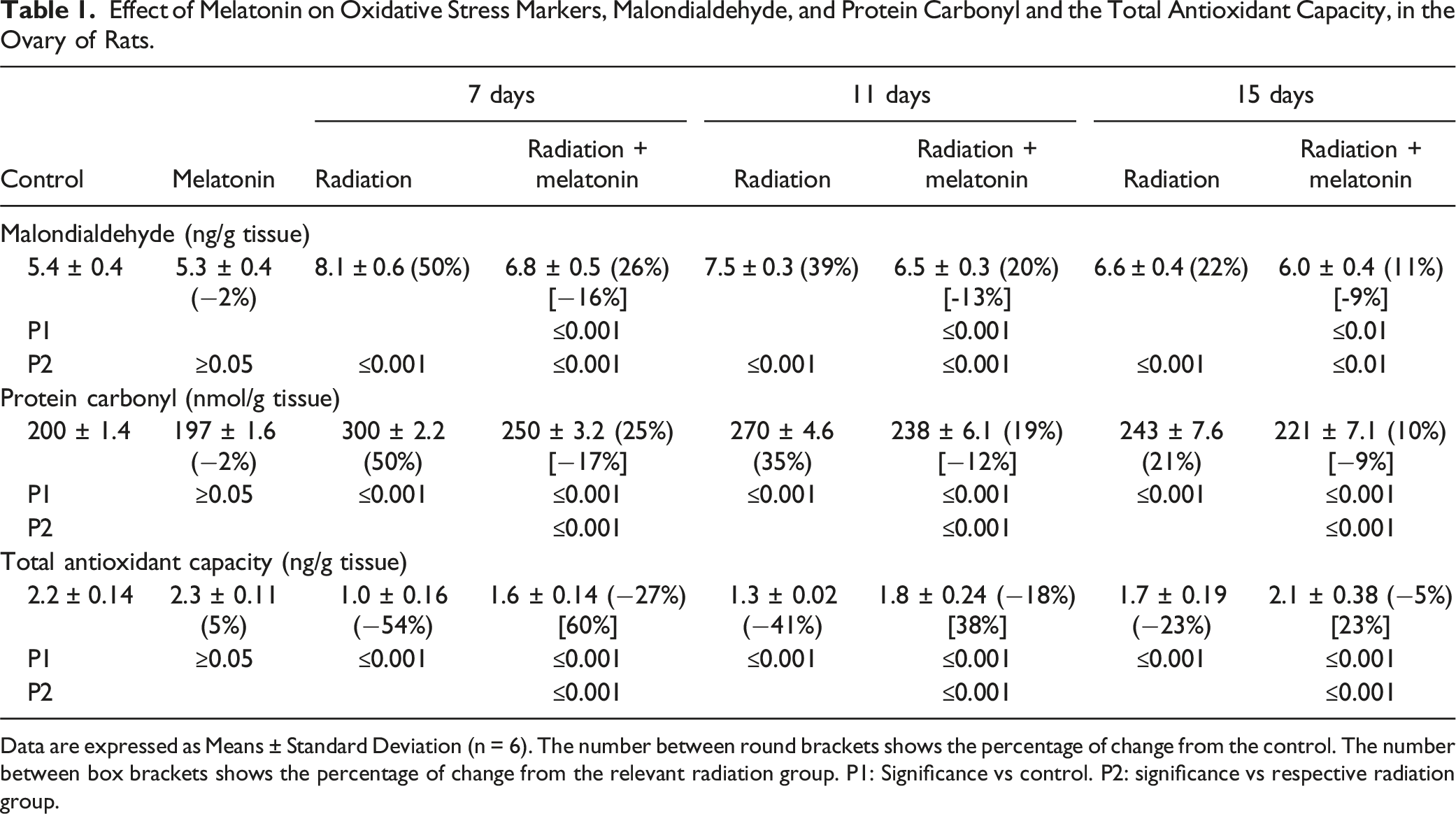
Data are expressed as Means ± Standard Deviation (n = 6). The number between round brackets shows the percentage of change from the control. The number between box brackets shows the percentage of change from the relevant radiation group. P1: Significance vs control. P2: significance vs respective radiation group.
Effect of Melatonin on Pro-inflammatory Cytokines, Tumor Necrosis Factor Alpha (TNF-α), Interleukin-1β, Interleukin-6 and Caspase-3 in the Serum of Rats.

Data are expressed as Means ± Standard Deviation (n = 6). The number between round brackets shows the percentage of change from the control. The number between box brackets shows the percentage of change from the relevant radiation group. P1: Significance vs control. P2: significance vs respective radiation group.
γ-Irradiation resulted in a significant drop (P ≤ 0.001) in E2-estradiol, testosterone, progesterone, LH, and FSH levels after 7, 11, and 15 days of irradiation, compared to control rats. These changes were more pronounced on the 7th day (Figure 1). Effect of melatonin on serum reproductive hormones, E2-estradiol, testosterone, progesterone, Luteinizing Hormone, and Follicle Stimulating Hormone in rats. Data are expressed as Means ± Standard Deviation (n = 6). a: significance vs control, b: significance vs radiation. a: significance at 0.05, a* significance at 0.01, a** significance at 0.001 b: significance at 0.05, b* significance at 0.01, b** significance at 0.001.
Effect of Melatonin in Control and Irradiated Rats
In normal healthy rats, melatonin treatment did not cause any significant changes in oxidative stress indicators (MDA, PCO, and TAC) and pro-inflammatory cytokines (TNF-α, IL-1β, and IL-6) compared to control rats (Tables 1 and 2). Additionally, no significant changes were observed in caspase-3 and reproductive hormones (E2-estradiol, testosterone, progesterone, LH, and FSH) in rat serum after melatonin treatment, compared to control rats (Table 2 and Figure 1).
However, administration of melatonin to irradiated rats resulted in a significant improvement in ovary redox status indicators; MDA, PCO, and TAC, compared to the corresponding irradiated group (Table 1). Specifically, the levels of MDA decreased significantly by 16%, 13%, and 9% after 7, 11, and 15 days of irradiation and treatment, respectively. Similarly, the levels of PCO decreased by 17%, 12%, and 9% after 7, 11, and 15 days of irradiation and treatment, respectively. Additionally, the levels of TAC increased by 60%, 38%, and 23% after 7, 11, and 15 days of irradiation and treatment, respectively, compared to the corresponding irradiated group.
The results also showed that melatonin treatment of irradiated rats resulted in a decrease in pro-inflammatory cytokines (tumor necrotic factor alpha, interleukin-1 beta, interleukin-6) and caspase-3 Levels (Table 2) at all time intervals, compared to irradiated rats. Specifically, the levels of tumor necrotic factor alpha were decreased significantly by 15%, 12%, and 9% after 7, 11, and 15 days of irradiation and treatment, respectively. Similarly, the levels of interleukin-1 beta decreased by 16%, 11%, and 9% after 7, 11, and 15 days of irradiation and treatment. The levels of interleukin-6 decreased by 11%, 12%, and 9% after 7, 11, and 15 days of irradiation and treatment, respectively. Additionally, the levels of caspase-3 decreased by 13%, 11%, and 8% after 7, 11, and 15 days of irradiation and treatment, respectively, compared to the corresponding irradiated group.
A noticeable increase in reproductive hormones (E2-estradiol, testosterone, progesterone, LH, and FSH) at all time intervals was observed in irradiated-treated rats, relative to the irradiated group (Figure 1). Specifically, the levels of E2-estradiol were increased significantly by 56%, 19%, and 16% after 7, 11, and 15 days of irradiation and treatment, respectively. Also, the levels of testosterone increased by 37%, 26%, and 25% after 7, 11, and 15 days of irradiation and treatment. The levels of progesterone increased by 50%, 33%, and 21% after 7, 11, and 15 days of irradiation and treatment, respectively. Additionally, the levels of LH and FSH increased by 44%, 25%, & 14% and 33%, 16%, and 14% after 7, 11, and 15 days of irradiation and treatment, respectively, compared to the corresponding irradiated group.
Discussion
Several methods have been devised in recent years with the goal of minimizing the severity of tissue harm caused by IR exposure. Increasing evidence has demonstrated that melatonin has no toxic effects, no addictive properties,20,21 and effectively scavenges the extremely hazardous hydroxyl radical (.OH), peroxynitrite anion (ONOO-), nitric oxide (NO.), superoxide anion (O2-.), hydrogen peroxide (H2O2), singlet oxygen (1O2), and guanosine radical (G.). 22
The ovaries consist of several cell types which are all susceptible to radiation damage.23-26 γ-Irradiation of rats in this study induced oxidative stress in the ovary manifested by a marked elevation in MDA and PCO associated with a significant drop in TAC, relative to control rats. The significant increase of lipid peroxidation product MDA and protein oxidation product (PCO) might be the result of the interaction of •OH produced by water radiolysis after radiation exposure with lipids and proteins, respectively. The procedure resulted in a disturbance of cell homeostasis due to a decrease in cell membrane integrity, altering the structure and activities of the cells. 27 The rise in oxidative byproducts was coupled with a considerable decrease in TAC, which could be attributed to the increased use of the antioxidant system to eliminate free radicals produced by irradiation.
The administration of melatonin after irradiation induced a marked improvement in oxidative stress biomarker levels (MDA, PCO, and TAC) in the ovaries, relative to the irradiated group’s corresponding values. In line with these findings, previous studies have shown that melatonin mitigated the ionizing radiation-induced oxidative stress by decreasing H2O2 level, and enhancing the activities of GPX and GR enzymes, as well as increasing GSH content. 16 Konturek et al., observed that melatonin given prior to ischemia induction restored oxidative markers to near-control levels. 28 In the same line, NaveenKumar et al., reported that the injection of 20 mg/kg melatonin for three days helped to minimize the amount of lipid peroxidation and ROS in Swiss albino mice. 29 Dal Bosco et al., demonstrated that intraperitoneal melatonin administration (20 mg/kg) for 14 days in rats reduced oxidative stress and lipid peroxidation. 30
Melatonin exerts its effects through both receptor-independent and receptor-dependent antioxidant mechanisms. Its free radical scavenging and antioxidant characteristics are mostly owing to its electron-rich aromatic indole ring, resulting in it being a powerful electron donor capable of greatly reducing oxidative stress. 31 Melatonin becomes a free radical, the indolyl radical cation, following the transfer of an electron to the hydroxyl radical. However, its reactivity is relatively inadequate, hence it is not hazardous to cells. 32 Furthermore, Melatonin combines with H2O2 to generate N1-acetyl-N2-formyl-5-methoxykynuramine, which may have substantial scavenging activity. 33 Melatonin therefore scavenges the alkoxyl radical directly, preventing the spread of lipid peroxidation.34-36 The increase in TAC may result from the activation of melatonin receptors which stimulates antioxidant enzyme expression, including glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), and catalase.37,38 Also, melatonin enhances intracellular glutathione levels 39 and inhibits the pro-oxidative enzymes lipoxygenase and nitric oxide synthase. 40 Indeed, nuclear factor erythroid 2-related factor 2 (Nrf2) plays a key role in regulating the antioxidant capacity of cells by controlling the transcription and translation of different antioxidant enzymes. Studies have suggested that melatonin’s antioxidant effects could be mediated by the Keap1-Nrf2-ARE pathway. 41
In this work, oxidative stress in the ovarian tissues of irradiated rats was combined with a significant rise in pro-inflammatory cytokine levels (TNF- α, IL-1β, and IL-6) and caspase 3, compared to the control rats. In agreement with these results, a previous study demonstrated that 4 Gy γ- irradiation caused an elevation in caspase3, cytochrome C, and P53 in ovarian and testicular tissues16,18 The rise in pro-inflammatory cytokines concomitant with the increase in oxidative stress biomarkers might be based on the correlation between inflammation and oxidative stress, 1 of which can be easily induced by another. 42 Following irradiation, RNS, and ROS may trigger intracellular signaling cascades that increase the expression of pro-inflammatory genes. On the other hand, inflammatory cells produce many reactive molecules, which causes severe oxidative stress. 43 The marked increase in caspase-3 may be due to oxidative stress, which induces the discharge of mitochondrial cytochrome c into the cytosol, activating caspases and leading to apoptosis. 44
The administration of melatonin to female rats after irradiation has reduced pro-inflammatory cytokine, and caspase-3 levels, compared to the irradiated group’s corresponding values. The results are consistent with a previous study showing that melatonin reduced inflammatory cytokines (CRP and IL-6) & caspase-3 and increased the anti-inflammatory cytokine IL-10 levels in the serum of irradiated rats (4 Gy) 3 and 14 days post-irradiation. 16 The results substantiate that melatonin can modulate pro-inflammatory cytokine expressions in various pathophysiological situations.37,45 Melatonin supplementation may be useful in lowering IL-1 release,36,46 TNF-α, and IL-6.29,38 Melatonin’s demonstrated anti-inflammatory benefits could be due to an association of its characteristics. Melatonin eliminates and destroys O2-., resulting in less ONOO- production. This, consequently, hinders the stimulation of poly (ADP-ribose) synthase, which plays a role in the control of nuclear factor kappa B (NF-κB), a transcription factor implicated in the activation of pro-inflammatory cytokine production. Therefore, melatonin modulates the NF-κB signaling pathway during inflammation. 46 Simultaneously, melatonin reduces NO production, hence lowering ONOO- generation. Furthermore, melatonin suppresses the development of P-selectin and intercellular adhesion molecule (ICAM-1), which may lead to a reduction in the recruitment of polymorphonucleates into the inflammatory region. Melatonin inhibits inflammation by decreasing nitrotyrosine, a molecule that indicates the nitration of tyrosine by ONOO-. 47 Caspase 3 Level improvement verifies that melatonin exerts its anti-apoptotic activities mainly by inhibiting caspase 3 activity, 48 elevating the anti-apoptotic activity of Bcl-2 protein expression, and reducing Bax proapoptotic activity, and cytochrome c release. This may occur via the JAK2/STAT3 pathway. 41 The anti-apoptotic effect of melatonin may be related to its ability to easily cross biological membranes and reach mitochondria. Once there, it reduces mitochondrial ROS generation and calcium release. Melatonin also helps maintain mitochondrial integrity by preserving its membrane potential and increasing the efficiency of the mitochondrial electron transport chain, leading to increased ATP production. 6
Female rats exposed to irradiation had significantly lower levels of the reproductive hormones E2-estradiol, testosterone, progesterone, LH, and FSH in comparison with the control group. The results align with a previous study by Rezk et al., who found a significant decrease in FSH and LH in female rats exposed to 4 Gy gamma radiation 7 and 15 days after irradiation. 49 Additionally, Ebrahim, observed that exposure of female rats to 5 Gy significantly decreased serum levels of FSH, LH, E2, and progesterone 3 days after irradiation. 50 The decrease in male sex hormones was also noted by Ezz El Arab et al., who found that whole-body irradiation at a dose level of 6 Gy reduced serum levels of testosterone, FSH, and LH at 2 weeks and 2 months after irradiation in rats. 51 This reduction was more pronounced after 2 weeks. The insufficiency of reproductive hormones is probably because of an aberration in the hypothalamic-pituitary-gonadal axis, resulting in deregulation of the release of hormones, 52 besides depletion of ovarian follicles. 53
Melatonin treatment for rats following irradiation resulted in a substantial rise in reproductive hormone levels, which may be attributable to the impact of melatonin on pituitary gland gonadotrophs that release LH and FSH. 54 This could be further attributable to its anti-apoptotic effect in decreasing follicular cell death. 55 It was reported that the antioxidant and antiapoptotic activities of melatonin have been shown to promote oocyte maturation and ovulation, 56 and regulate progesterone production. These effects are mediated by MT1 and MT2 melatonin receptors and are dependent in part on activation of the PI3K/AKT signaling pathway. 57 Additionally, studies have reported that melatonin may have the potential to reduce the hypertrophy of the ovaries associated with polycystic ovary syndrome (PCOS), restore ovarian function, and increase serum levels of progesterone, estradiol, LH, and FSH in PCOS rats. 58
Conclusion
The results of the current study demonstrate that melatonin reduces the harmful impacts of radiation on the ovaries and reproductive hormones through direct and indirect antioxidative mechanisms, which in turn reduces the levels of pro-inflammatory cytokines (TNF- α, IL-1β, and IL-6) by inhibiting the nuclear factor kappa B (NF-κB) signaling pathway, and inhibits apoptosis via a caspase-dependent pathway. Therefore, melatonin may be employed as an effective mitigating factor to reduce the detrimental effects of radiation on the reproductive system due to the synergistic interdependence of its antioxidant, anti-inflammatory, and anti-apoptotic, properties.
Footnotes
Acknowledgments
We would like to express our special gratitude to our colleagues and members of NCRRT, EAEA for providing the necessary irradiation facilities.
Author contributions
Shereen M. Shedid, Nahed Abdel-Aziz, Fatma R. Algeda and Helen N. Saada contributed to the achievement of the research, analysis of the results, and writing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
