Abstract
Tartaric acid is capable of balancing blood pressure. It is the main constituent of antihypertensive agents (grapes and wine) and has not been scientifically explored as an antihypertensive remedy. This study aimed to investigate the antihypertensive effect of a low-dose tartaric acid in vivo and explore underlying mechanisms in vitro. Intravenous administration of tartaric acid at the dose of 50 µg/kg caused a % fall in mean arterial pressure (MAP) in normotensive and hypertensive rats [51.5 ± 1.7 and 63.5 ± 2.9% mmHg]. This hypotensive effect was partially inhibited by atropine (1 mg/kg) and L-NAME (100 µg/kg) pretreatment. In hypertensive rats, oral administration of tartaric acid (.1, .5, 1, 5, and 10 mg/kg) for 2 weeks resulted in 65 ± 7.3 mmHg MAP at 10 mg/kg. This antihypertensive effect was comparable to the orally administered verapamil (10 mg/kg) for 2 weeks which caused a decrease in MAP 60.4 ± 3.8 mmHg. Tartaric acid relaxed phenylephrine (PE) and High K+-induced contractions with EC50 values of .157 (.043-.2) and 1.93 (.07-2) µg/mL in vitro. This endothelium-dependent relaxation was inhibited with atropine (1 µM) and L-NAME (10 µM) pretreatment. Tartaric acid also suppressed phenylephrine contractions in Ca+2 free/EGTA medium and on voltage-dependent calcium channels, causing the concentration–response curves toward right. Tartaric acid induced negative inotropic and chronotropic effects with EC50 values of .26 (.14-.4) and .60 (.2-.8) in rat atria. It showed its effect by complete blockade against atropine and partially in propranolol pretreatment. These findings provide scientific basis to low-dose tartaric acid as an antihypertensive and vasodilatory remedy through muscarinic receptor-linked nitric oxide (NO) pathway and Ca+2 channel antagonist.
Introduction
Hypertension has been among the most studied topic of the previous century because of its significant contributing comorbidities to heart failure, myocardial infarction, stroke, and renal failure. 1 More than one billion adults worldwide have hypertension with up to 45% of the adult population being affected by the disease. The high prevalence of hypertension is consistent across all socio-economic and income strata. Recent estimates have suggested the number of patients with hypertension could increase to 1.5 billion by 2025. 2 The management of hypertension has side effects that include hypotension, electrolyte imbalances, edema, and renal dysfunction. 3 Therefore, new pharmacological molecules are required with lesser side effects. Tartaric acid (2,3-dihydroxybutanedioic acid, C4H6O6) is an organic white crystalline, odorless dicarboxylic acid present as the main constituent of many plant species. Tartaric acid has a substituted hydroxy group on both the first and second carbons.
It is called fruit acid and is present particularly in grapes, tamarinds, bananas, and citrus fruits. Tartaric acid has been known by the winemaker as a wine diamond as it is a main constituent of wine. Tartaric acid is patented to have a role in balancing of blood pressure. 4 Tartaric acid isolated from the fruit of Japanese apricot was found to be effective against Escherichia coli, Bacillus subtilis, and Streptococcus suis. 5 An in vitro study reported fungistatic and fungicidal activities of tartaric acid against pathogenic fungi such as Trichophyton mentagrophytes var. mentagrophytes, Candida albicans, Aspergillus fumigatus, and Malassezia furfur. 6 Tartaric acid isolated from the citrus fruits was found effective against scurvy. 7 Tartaric acid has been reported as an antioxidant and anti-inflammatory. 8
Acetic acid, citric acid, and docosahexaenoic acid are some of the acids belonging to the class of carboxylic acid that have been reported for their antihypertensive potential.9-11 Similarly, red wine and grapes have been reported for the cardiovascular effects because of their polyphenolic constituents.12-14 However, tartaric acid belonging to carboxylic acid is the main constituent of grapes and red wine and is used as balancing blood pressure has not been investigated for its antihypertensive potential. Based on all the above-mentioned explanations, it may be considered that tartaric acid may have antihypertensive potential and can be used as an effective remedy. Therefore, this research aimed to investigate the antihypertensive effect of tartaric acid in vivo and the underlying mechanism(s) in vitro.
Materials and Methods
Drugs and Standards
Drugs and standards were procured from (Sigma Chemical Company, St. Louis, MO) including tartaric acid, acetylcholine chloride (Ach), atropine sulfate, norepinephrine bitartrate, L-NAME (Nω-Nitro L-arginine methyl ester hydrochloride), verapamil hydrochloride, propranolol. Sodium thiopental was a gift from Abbott Laboratories, Pakistan.
Animals
BalbC mice (18-25 g) were used for acute toxicity study and Sprague-Dawley (SD) rats (200-250 g) were used for experimentation. These animals were kept in the animal house had specifications of 12 hr cycle of light/dark, normal room temperature, and free access to food and water in the Department of Pharmacy, COMSATS University Islamabad (CUI), Abbottabad Campus, Pakistan. All the experimentation was conducted following the rules of the Institute of Laboratory Animal Resources, Commission on Life Sciences, National Research Council 15 after the approval of the Ethical Committee of the Department of Pharmacy, CUI, Abbottabad Campus, Pakistan (license no. PHM.Eth/CS-MO2-059-0721).
Pharmacological Investigation
Safety Profile
BalbC mice weighing 18-25 g were grouped into 6 groups of 5 mice each. Mice were fasted overnight and only received water for 24 hrs. Group-I acted as a control group and received normal saline. Other groups were administered different doses of tartaric acid (50, 100, 200, 400, and 450 mg/kg), orally. Normal behavioral parameters including alertness, grooming, convulsions, hyperactivity, lacrimation, salivation, sweating, urination, touch response, pain response, writhing reflex, corneal reflex, gripping strength, righting reflex, and rate of mortality were observed and recorded for 72 hours. 16
In Vivo Measurement of Blood Pressure
Hypertension induction by 8% high salt
Rats were divided into groups having 5 rats each. Group 1 received a normal diet and water and acted as normal control. Group 2 was considered hypertensive control and received 8% sodium chloride in food and water for 2 weeks. After the end of the 2 weeks, the mean arterial pressure (MAP) of all the rats was measured before and after the intravenous administration of tartaric acid, and the % fall in MAP was calculated.
Another protocol was adopted for 2 weeks. SD rats were divided into 8 groups having 5 rats each. Group 1 received a normal diet and water and was the normal control group. Group 2 was hypertensive control and received 8% sodium chloride in food and water. The other 5 groups were given 8% sodium chloride in diet and water for 2 weeks and different doses of tartaric acid (.1, 1, 5, 10, and 15 mg/kg). The 7th group was a standard group and received verapamil 10 mg/kg. At the end of the high salt diet protocol, the effect of tartaric acid on blood pressure was determined and it was calculated as mean arterial pressure (MAP) and observed for 30 min.
17
MAP was calculated by the formula
Experimental Procedure
Sodium thiopental 50-90 mg/kg intraperitoneally was injected to induce anesthesia. Cannulation of the trachea was done with polyethylene tubing PE-20 and PE-50 was used to cannulate the right carotid artery and left jugular vein. PE-50 of the right carotid artery was connected to PowerLab (ML 846) Data Acquisition System via transducer (MLT 0699) and bridge amplifier (N12128). Control responses of standards, acetylcholine (ACh) (1 μg/kg), and norepinephrine (NE) (1 μg/kg) were obtained before testing tartaric acid in both groups. Injection of each dose (1-50 μg/kg) of tartaric acid was made. Normal saline .1 mL was injected after every dose to ensure the availability of every dose to the target site. To investigate the mechanisms of antihypertensive effect in vivo atropine and L-NAME were used in the normotensive group. For this, the normotensive group was again divided into 2 groups having 5 rats each.
After the administration of Ach, rats were stabilized and atropine (1 mg/kg) was injected. After 20 min, again Ach was administered and then different doses of tartaric acid (1, 3, 10, 30, and 50 µg/kg) were given intravenously to check the involvement of muscarinic receptors.
To check the involvement of nitric oxide, nitric oxide synthase blocker L-NAME (100 µg/kg) was given intravenously for 20 min that was followed by different doses of tartaric acid (1, 3, 10, 30, and 50 µg/kg). 18 The difference in the blood pressure relevant to each dose was determined as the steady-state value before and after the administration of a drug. The percent fall in MAP was calculated as control-fall/control×100.
According to the second protocol, oral doses of tartaric acid were given for 2 weeks, as described earlier, each group’s MAP was observed for 30 min, and mean values were calculated. 19
In vitro studies in isolated rat aorta
The thoracic aorta was isolated from rats of all groups (normotensive and hypertensive). After that, it was kept in a petri dish having Kreb’s solution. Aortic rings of 2-3 mm in width were made. To check the integrity of endothelium, some aortic rings of normotensive rats were made denuded by intentionally damaging the internal surface of the aortic ring with forceps. These rings failed to induce 80% or more relaxation and were considered denuded rings with damaged endothelium. 20
Experimental Procedure
After aortic ring preparation from all the rats of the above-mentioned groups, each aorta was hanged in a tissue bath. The tissue bath contained Kreb’s solution at 37°C temperature and carbogen (5% CO2 in 95% O2). The composition of Kreb’s solution was (mM): NaCl 118.2, NaHCO3 25.0, CaCl2 2.5, KCl 4.7, KH2PO4 1.3, MgSO4 1.2 and glucose 11.7 (pH 7.4). Response on aortic rings was measured by PowerLab Data Acquisition System via force transducer and bridge amplifier. 2 g resting tension was applied to rings and 60-90 min time was given to equilibrate the tissues with the washing of tissues after 15-20 min. Steady-state contractions were achieved by repetitive administration of phenylephrine PE (1 µM) and high potassium (High K+). All tissues were stabilized and tartaric acid (.01-10 µM) in a cumulative way was added. Tartaric acid on vascular reactivity was evaluated by different mechanisms including the level of calcium influx through voltage-dependent (VDCs) or receptor-operated channels (ROCs) and from an internal store(s). To investigate the mechanisms of action, different protocols were followed in vitro.
Evaluation of muscarinic receptors involvement
To check muscarinic receptors involvement, endothelium-intact rings were preincubated with atropine (1 μM). After that acetylcholine (1 μM) was administered to confirm the blockade of muscarinic receptors. Response of tartaric acid was determined by administering (.01-10 µM) concentration of tartaric acid cumulatively. 21
Evaluation of the nitric oxide-mediated effect
After tissue stabilization, L-NAME (10 μM) was used to preincubate the aortic rings to block nitric oxide synthase and after 15-20 min, different concentrations of tartaric acid (.01-10 µM) were added to the organ bath to check its effect against nitric oxide synthase enzyme. 22
Endothelium-independent effects on vascular smooth muscle
To check the relaxant response of each extract on vascular smooth muscles specifically, aortic tissues were stabilized and high K+ (80 mM) was injected to induce contraction. From (.01-10 µM) concentration of tartaric acid was then added cumulatively, and relaxation was expressed as the percent of the contractions induced by K+ (80 mM). 17
Calcium influx through intracellular stores
Isolated rat aorta was placed in Ca+2-free/EGTA (ethylene glycol tetraacetic acid) solution (15-20 min). The composition of Ca+2-free/EGTA Kreb’s solution was (mM): glucose 11.7, EGTA .05, KCl 4.7, KH2PO4 1.3, MgSO4 1.2, NaCl 118.2, NaHCO3 25.0 (pH 7.4). After that PE (1 μM) was administered to induce contraction. To refill the intracellular stores, Kreb’s solution was used to wash the tissue 3-4 times and incubated for 40 min. The medium was again replaced with the calcium-free solution and incubated. Aortic tissues were preincubated with different concentrations of tartaric acid than phenylephrine (1 μM) was used to induce contractions. The contractions produced by phenylephrine were compared in the presence and absence of tartaric acid. 23
Involvement of calcium through voltage-dependent calcium channels
Rings from the aorta were prepared as described previously. After stabilization, PE (1 µM) was administered and stable contraction was achieved. Aortic tissue was washed with Ca+2-free solution 4-5 times. Concentrations response curves (CRCs) of Ca+2 (CaCl2) was made. Aortic tissue was pretreated with tartaric acid for 30 to 45 min. CRCs of Ca+2 were constructed and compared with control under similar experimental conditions and a possible calcium channel blocking effect was measured.
To evaluate the effect of tartaric acid through ROCs, aortic rings were washed four to five times with normal Kreb’s solution, and the effect of tartaric acid was determined on phenylephrine (PE) (1 µM)-induced sustained contractions. 24
Isolated Right Atrial Preparations
The right atrial flap was prepared into strips by dissecting and cleaning fatty tissues and hung in the organ bath. The organ bath was already filled with Kreb’s solution aerated with carbogen and a set temperature of 32°C. The atrial flap was allowed to stabilize at 1 g tension and waited till the spontaneous force of contraction and heart rate were achieved. 25
Atropine (1 μM) was used to block muscarinic receptors and after 15-20 min effect of tartaric acid (.01-10 µM) was evaluated against cardiac effects mediated through muscarinic receptors.
Propranolol (1 μM) was used to pretreat atrial strips and after 15-20 min, different concentrations of tartaric acid (.01-10 µM) were administered to check the involvement of adrenergic receptors, and the effect was compared before and after treatment with propranolol.
Statistics
Data were articulated in the mean, SEM (standard error of the mean), and EC50 values were defined with a 95% confidence interval (CI). The % fall in MAP was shown by one-way ANOVA followed by post hoc Tukey HSD test and in vitro the percent vasorelaxation was expressed by two-way ANOVA followed by post hoc Bonferroni test. Differences were measured significant at *P < .05, **P < .01 and ***P < .001.
Results
Safety Study
Tartaric acid was investigated up to the dose of 450 mg/kg in mice and no behavioral changes or death was observed for 72 hrs after administration of tartaric acid.
Effect of Tartaric Acid on MAP
Before testing tartaric acid, the effect of norepinephrine and acetylcholine was evaluated to confirm the normal response of animals toward standard drugs. Animals showed a rise and fall in MAP in response to standards, respectively. For evaluation of the effect of tartaric acid, normotensive (n = 18) and 8% NaCl-induced hypertensive (n = 6) rats were used. Before starting protocols, blood pressure, calculated as MAP, was measured in both normotensive (118 ± 2.25 mmHg) and hypertensive (185 ± 4.18 mmHg) rats (Figure 3). Acetylcholine was used as the standard drug in normotensive rats and it caused dose-dependent % fall in MAP 26.6 ± .74, 39.3 ± .66, 48.3 ± .66, 65.2 ± .9 and 65.4 ± 1.3 at the particular doses of 1, 3, 10, 30 and 50 µg/kg (Figure 1A). After intravenous administration of tartaric acid at the same doses as acetylcholine, the % fall in MAP observed was 9.7 ± 1.1, 22.8 ± .6, 30.5 ± .9, 39.2 ± 1.2, and 51.5 ± 1.7 (Figure 1B). The observed % fall in MAP was statistically significant (P < .01) at 50 µg/kg. Presents % fall in mean arterial pressure induced by the 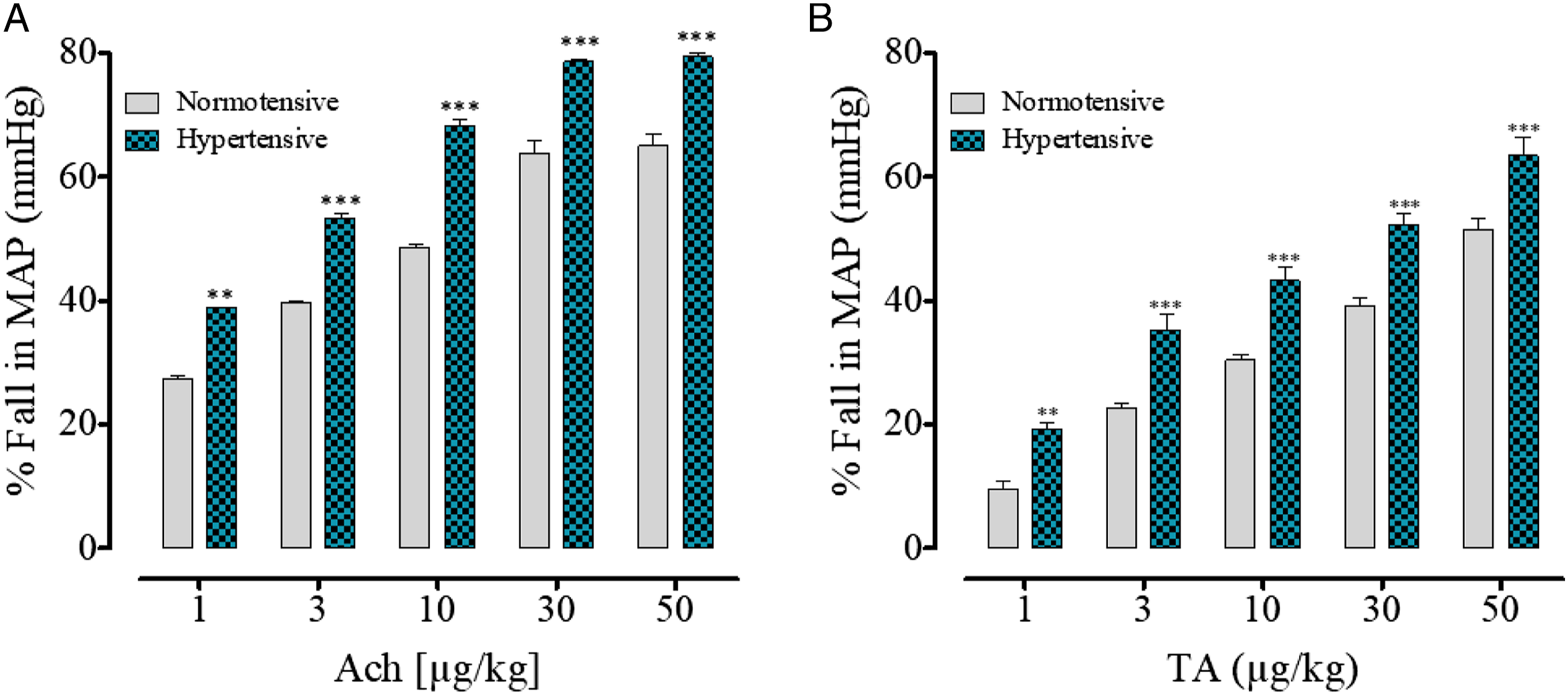
The effect of tartaric acid was also checked in 8% NaCl-induced hypertensive rats under similar conditions. The % fall in MAP was 38.9 ± .32, 52.7 ± 1.0, 68.7 ± .72, 78.4 ± .8 and 79.4 ± .24 observed after intravenous administration of acetylcholine to hypertensive rats at 1, 3, 10, 30 and 50 µg/kg doses (Figure 1A). At the same doses, tartaric acid induced a fall in the blood pressure, calculated as % fall in MAP, which was 19.2 ± 1.2, 35.35 ± 2.4, 43.3 ± 2.2, 52.3 ± 1.9, and 63.5 ± 2.9 (Figure 1B). The overall results showed that intravenous administration of tartaric acid caused higher % fall in MAP in hypertensive rats as compared to the normotensive rats (Figure 1B).
For the evaluation of possible mechanisms, atropine (1 mg/kg) and L-NAME (100 µg/kg) were used to pretreat normotensive rats. A standard cholinergic agent, acetylcholine’s hypertensive effect was reversed in the presence of atropine (Figure 2A), thus, confirming the validity of the protocol. Amazingly, after intravenous administration of tartaric acid, the reversal in the antihypertensive effect was observed in the atropine and L-NAME pretreated groups (Figure 2B). Graphs show a % fall in mean arterial pressure by 
Effect of Tartaric Acid on Blood Pressure After Oral Administration
Tartaric acid (.1, .5, 1, 5, and 10 mg/kg) with 8% high salt diet resulted in the reversal of the development of hypertension in these groups. The dose-dependent observed MAP was 163.3 ± 10.6, 145.4 ± 2.5, 98.8 ± 7.2, 77.4 ± 5.3, and 65 ± 7.3, respectively. It was compared with the MAP (185 ± 4.18 mmHg) in the hypertensive control group (Figure 3). Shows mean arterial pressure in normal control (NC), high salt-induced hypertensive control (HC) group, and after oral administration of tartaric acid (T.A) and verapamil in 8% NaCl hypertensive rats for 14 days (n=6).
Effect of Tartaric Acid on Vascular Reactivity
When the contractions were achieved after the administration of phenylephrine PE (1 μM), acetylcholine was administered to induce relaxation. For evaluation of the possible mechanism of relaxation, atropine (1 μM) and (10 μM) L-NAME pretreatment was done in normotensive rat aortic rings. As from in vivo results, the relaxation was reversed with the antagonists, and also with the removal of endothelium as shown in Figure 4A. Tartaric acid also relaxed PE precontractions in rat aortic preparations with a respective EC50 value of .157 (.043-.2) µg/mL (Figure 4B). In the denuded aortic rings with damaged endothelium, comparatively less relaxation was induced by tartaric acid (Figure 4B). In endothelium-intact rat aortic rings, relaxation induced by tartaric acid was significantly (P < .01) blocked in presence of antagonists i.e., atropine which caused 35% relaxation and L-NAME with a respective EC50 value of 2.76 (1-3.76) µg/mL, shifted the concentration–response curves to the right. Vasorelaxant effects depend on endothelium 
The effect of tartaric acid was also determined in hypertensive aortic rings for comparative studies. This comparison was conducted to counter-check the endothelium damage as this damage is due to high salt. Thus, this outcome was confirmed by administering acetylcholine to hypertensive rats and it failed to cause relaxation in hypertensive aortic rings (Figure 4A). Tartaric acid caused the relaxation of the phenylephrine precontraction and shifted the concentration–response curves to the right (Figure 4B).
In another study, aortic rings were incubated with high K+ in normal Kreb’s solution, where administration of tartaric acid in ascending concentrations also induced relaxation against high K+ with an EC50 value of 1.93 (.07-2) µg/mL, like verapamil (Figures 5A and B). Endothelium-independent vasorelaxant effects of 
Effect on Intracellular Ca+2 Stores
Vasodilation was caused by tartaric acid involving intracellular Ca++ stores; Ca++ free/EGTA medium was used to incubate aortic rings. Tartaric acid and verapamil were added and suppression of phenylephrine PE peak formation was studied (Figures 6A and B). The tartaric acid was comparable to verapamil. (A) The effect of tartaric acid and 
Effect on Voltage-dependent Calcium Channels
Tartaric acid was added cumulatively in precontracted aortic rings with PE and high K+. This addition of tartaric acid inhibited the contractions of PE and high K+ with EC50 values of .086 (.014-.1) and 1.12 µg/mL (.88-2), respectively, like verapamil (Figures 7A and B). In rat aortic rings preparation and Ca2+ free/EGTA medium, preincubation of tartaric acid at different concentrations (.01-10 µg/mL) caused the non-competitively shift of Ca+2 CRCs to the right (Figure 7C), similar to that resulted by verapamil (Figure 7D). Concentration based vasodilator effect of 
Rat Atrial Rhythmic Contractions
From the normotensive group, right atria flaps were made, stabilized, and rhythmic contraction and relaxation pattern was achieved. Suppression of both rate and force of contractions were observed after the addition of ascending concentrations of tartaric acid to atria. Propranolol pretreatment partially suppressed heart rate (negative chronotropic) and force of contraction (negative inotropic). However, atropine fully obliterated both rate and force of contraction (Figure 8). Concentration–response curves show tartaric acid on spontaneous rate and force of contraction in isolated rat atrial flaps in the presence and absence of atropine (1 μM) and propranolol (1 μM) (n = 6).
Discussion
Tartaric acid nutritious honey is patented in China in 2001 as it is capable of balancing blood pressure. 4 Tartaric acid belongs to the class of carboxlic acids and some of the carboxylic acids including acetic acid, citric acid, and docosahexaenoic acid have been known for their antihypertensive potential.9-11 Similarly, red wine and grapes containing tartaric acid have been reported as the cardiovascular effects because of their polyphenolic constituents.12-14 However, tartaric acid patented as the balancing of blood pressure, belonging to the class of carboxylic acid having the same molecular moieties and is the main constituent of grapes and red wine has not been explored as an antihypertensive remedy and underlying mechanisms were also not investigated. In the current study, tartaric acid was explored in normotensive and 8% sodium chloride–induced hypertensive rats in vivo followed by mechanistic investigation in in vitro.
The intravenous administration of tartaric acid in normotensive rats resulted in reduced blood pressure, calculated as MAP, suggesting its antihypertensive potential. To determine its efficacy in hypertensive rats, rats were kept on a diet of high salt (8% sodium chloride) for 14 days. It is interesting to note that intravenous shots of various doses of tartaric acid in hypertensive rats proved to be more effective as antihypertensive, compared to normotensive rats. These outcomes were further assessed whether tartaric acid has the potential to reverse hypertension development in rats; some of the groups were orally fed tartaric acid and high salt for 14 days. At the end of the 2 weeks of treatment, the blood pressure of all the groups was measured and in orally treated groups, a 5 mg/kg dose showed the required response. In the hypertensive group, tartaric acid remarkably lowered blood pressure at the maximum doses of 30 and 50 µg/kg. In other protocol of oral treatment, initially, different doses of tartaric acid were given orally to check the optimal response. The doses were .1, .5, 1, 5, 10, and 15 mg/kg. However, no therapeutic significant difference was observed at the dose of .1 and .5 mg/kg, and no significant results were observed between 10, 15, and 20 mg/kg. Based on outcomes, optimal doses were selected and compared. Optimal response-producing doses (.1, 1, 5, 10, and 15 mg/kg) of tartaric acid were selected. These were interesting results and showed that tartaric acid is a potent antihypertensive remedy at a low dose that was comparable to the orally treated verapamil (10 mg/kg). It is reported in the literature that high salt intake led to the development of hypertension 26 which is the same as human hypertension with dietary sodium. 27 Different mechanisms are involved in the development of hypertension due to high salt. Some are the alteration of homeostasis of the renin-angiotensin system, provoking water retention. High salt is also involved in the reduction of sympathetic activity and increase in plasma volume. It is also responsible for the lowering of arterial vasodilator capacity by causing alterations of endothelial Ca+2 signaling and abnormal high production of 20-hydroxyeicosatetraenoic acid. Overall upregulated the system and finally resulted in vascular dysfunction by increasing oxidative stress and a reduced nitric oxide bioavailability. 27 Thus, high salt induces hypertension due to an increase in blood volume and endothelium dysfunction. Nitric oxide (NO) is one of the important vasoactive substances, released from vascular endothelium. It has a major role in a fall in blood pressure by dilating blood vessels. To study if tartaric acid has prevented the development of hypertension due to improving endothelium function in vivo, we used normotensive rats to have a clear insight into its effect mediated through muscarinic receptors and normal endothelium-derived NO.
Various intravenous doses of tartaric acid to normotensive pretreated rats with nitric oxide synthase (L-NAME), 28 reversed its antihypertensive effect, indicating NO as its endogenous mediator. In vascular endothelium, there is a link between muscarinic (M3) receptor activation and nitric oxide synthase. 29 From these findings, it was considered that tartaric acid may also affect vascular muscarinic receptors. To confirm this speculation, atropine (a muscarinic receptors antagonist) 30 was used as an antagonist in normotensive rats. The antihypertensive effect of acetylcholine (a muscarinic receptor agonist) 30 was blocked with atropine pretreatment in vivo. Like acetylcholine, the hypotensive effect of tartaric acid was abolished in the presence of atropine in vivo. This effect was of greater extent as of L-NAME pretreated rats. This indicates that tartaric acid showed its effect through NO in vivo via the activation of vascular muscarinic receptors.
The in vivo findings were proceeded to confirm the involvement of vascular and/or cardiac muscarinic receptors. To confirm this, a series of in vitro experiments were conducted on isolated rat aorta and atrial strips. Interestingly, the vasorelaxant effect was observed upon the cumulative addition of tartaric acid to isolated aortic rings, precontracted with phenylephrine. The maximum response was achieved at the dose of 1 µg/mL. It was of great interest when this relaxation was obliterated in denuded rings (damaged endothelium) and aortic rings from the hypertensive rats. These findings strongly suggest the role of endothelium-derived relaxing factor-like NO. The vascular endothelium is of special importance as it controls the vascular tone and blood pressure. The overall vascular effects are controlled by the mediators released from endothelium. One of the major mediators is nitric oxide, coupled with the muscarinic receptors, present in the vascular endothelial cells. The activation of muscarinic receptors activates the Gq-PLC-IP3 pathway that causes the stimulation of endothelial nitric oxide synthase enzyme and production of nitric oxide and vasodilation. 31 To confirm the hypothesis from in vivo findings, normotensive rat aortic rings were pretreated with L-NAME on the cumulative addition of tartaric acid; the effect was reversed, suggesting NO as the mediator of its effect. Moreover, in atropinized aortic rings, the vasorelaxant effect was also blocked which indicates that the effect is due to the muscarinic receptor-linked nitric oxide release. It was observed that atropine, L-NAME, or denudation failed to inhibit relaxation to tartaric acid at higher concentrations, suggesting its effect on vascular smooth muscles. Thus, to have insight into their effect on vascular smooth muscles, further experiments were conducted.
The cumulative addition of tartaric acid in isolated precontracted aortic rings with phenylephrine and high K+ resulted in relaxation. Verapamil, a Ca+2 channel blocker, 32 also inhibited phenylephrine and high K+ precontractions. These results showed the inhibition of Ca+2 movements. Phenylephrine induces vascular contraction by influxing Ca+2 through receptor-operated Ca+2 channels and releases from the intracellular store. 32 Similarly, high K+ (> 30 mM) induces contractions by allowing Ca+2 entry through voltage-dependent Ca+2 channels. 33 So, the inhibitory effect of tartaric acid against the phenylephrine and high K+ precontractions suggests Ca+2 entry blocking effect.
To explore the involvement of the release of internal Ca+2, aortic rings were incubated in Ca+2-free/EGTA medium. In this medium, phenylephrine shows peaks that result from the release of Ca+2 from the endoplasmic reticulum. In a concentration-dependent addition of tartaric acid, aortic tissues prevented the formation of a spike of phenylephrine, indicating inhibition of Ca+2 release from the internal store, similar to verapamil.
To determine the involvement of Ca+2 through voltage-gated Ca+2 channels (VDCCs), the inhibitory response of tartaric acid on Ca+2 entry was evaluated. Ca+2-free/EGTA medium was used to incubate aortic rings and CaCl2 concentrations response curves (CRCs) were obtained in triplicate. Preincubation of the aortic rings with tartaric acid induced a non-competitive shift in the CaCl2 CRCs with suppression of maximum response, in a concentration-dependent manner. This effect was also similar to verapamil, indicating Ca+2 channel blocking effect. These vascular in vitro findings showed that tartaric acid has muscarinic receptor-linked NO (endothelium-dependent) and inhibitor effect on Ca+2 moment (endothelium-independent) mechanistic properties that result in a decrease in the vascular resistance and fall in blood pressure in both the normotensive and hypertensive rats. However, the effect of tartaric acid on heart rate and possible cardiac output was studied using rat atrial flaps.
Cumulative addition of different concentrations of tartaric acid in isolated rat atrial flaps completely suppressed both force (negative inotropic) and rate of contractions (negative chronotropic). Based on the previous findings of the involvement of muscarinic receptors, atropine (a muscarinic receptor blocker) was used to preincubate atrial strips. This pretreatment reversed the cardiac effects of tartaric acid, indicating activation of cardiac M2 receptors. It was further rectified when propranolol, an adrenoceptor antagonist, 34 pretreatments did not change the cardiac effects of tartaric acid. These findings indicate that tartaric acid possesses atropine-sensitive negative inotropic and chronotropic effects that explain the decrease in cardiac output that resulted in a fall in blood pressure, observed in vivo.
Conclusions
Tartaric acid, a dicarboxylic acid has blood pressure lowering and vasodilatory properties at a very low dose comparable to verapamil at the same dose. Its mechanisms involve muscarinic receptors (M2 and M3), NO synthase enzyme, and Ca+2 channel antagonist and cardiac suppression. Both antihypertensive and vasodilatory results provide a scientific strength to the tartaric acid nutritious honey that is used as an effective agent for balancing of blood pressure. These results give scientific basis to carboxylic acid having antihypertensive potential. The hypotensive effect of grapes and wine may also be due to tartaric acid presence as the main constituent. These results conclude that tartaric acid can be used as an effective agent at a low dose in the management and prevention of hypertension.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Institutional Review Board Statement
Healthy, adult male SD rats with an average weight of 210 ± 10 g were utilized for the in vitro and in vivo antihypertensive analysis after getting approval from the departmental ethical committee (license no. CS-MO2-059-0721) COMSATS University Islamabad, Abbottabad Campus.

