Abstract
Introduction
Proso millet is rich in phytochemicals like phytic acid, which is believed to lower cholesterol, and phytate, which is associated with reducing cancer risks, along with phenolic acids and benzoic acids. 10 It also contains chlorogenic acid, syringic acid, caffeic acid, R-coumaric acid, ferulic acid, gallic acid, hydroxycinnamic acids, carotenoids, lutein, 9 fatty acids (oleic and linolenic), starch, proteins, 12 and myoinositol hexa-phosphoric acid. 13 Moreover, gluten-free fiber and other important constituents like carbohydrates, and vitamins, zinc, calcium, phenol, magnesium, iron, potassium, phosphorous, and manganese are reported to be present. 14
Previous studies showed that
Methodology
Plant Collection, Extraction, and Fractionation
National Agricultural Research Center (NARC), Bioresource Conservation Institute, Islamabad provided seed sample of
The seeds collected were cleaned, grinded into coarse powder, and macerated with solution (70% methanol+30% distilled water) in amber colored bottle with regular shaking for 4 days. Then filtration was done firstly through cloth then with Whatman filter paper. Same method was repeated for 2nd and 3rd maceration. All collected filtrates were combined; solvent was evaporated under reduced pressure on rotary evaporator (Rotavapor, BUCHI Labrotechnik AG, Model 9230, Switzerland) until a dark reddish brown paste (Pm.Cr) with approximate 16% yield was attained. The obtained extract was stored in dark colored glass bottle. At the time of experiment, Pm.Cr extract was dissolved in DMSO and distilled water. 17
Fractionation was done by dissolving 10 g Pm.Cr in 100 mL distilled water and 100 mL dichloromethane (DCM). Two layers DCM (Pm.Dcm) and aqueous (Pm.Aq) were separated out and dried.
Chemicals and Animals
Almost 99.9% pure chemicals were used for in vivo and in vitro experimental studies. Paracetamol, aspirin, and castor oil were obtained from GSK. Glibenclamide, phenylephrine, doxazocin, losartan, cromakalim, carbachol, and loperamide were obtained from Sigma Chemical Co. (USA). Chemicals to make Krebs and Tyrode’s solution were acquired from Merck (Germany). For experiments white albino rabbits (1.0-2.0 kg) and Sprague Dawley rats (150-250 g) of both genders were taken from Animal House of Faculty of Pharmacy. Animals were housed at 26 ± 1°C. All permitted Ethical rules were followed in the whole research. 18
Phytochemical Screening
Preliminary Phytochemical Examination
The Pm.Cr and Pm.Aq were analyzed qualitatively to detect bioactive compounds.
HPLC Analysis
A simple, speedy and competent binary sloped solvent method developed by our group was used for the identification of phenolic and flavonoids compounds in Pm.Cr. HPLC containing C18 column with an internal diameter of 250*4.7 mm, flow rate of .9 mL/minutes, and ability to separate phenolic constituents was 8–9 and for flavonoids 1–4 in a period of 35–36 minutes was used. Mobile phase used were; A: acetonitrile 70% and methanol 30% and B: H2O and .5% acetic acid. The concentration, retention time, and peak areas were compared with standards. 19
In-Vitro Assays
Preparation of Isolated Jejunum
Experiments were done with Pm.Cr) and its fractions (Pm.Aq and Pm.Dcm) on isolated tissue to observe either it has spasmodic or antispasmodic consequence on GIT. 20
Rabbits were kept on fasting overnight before experimentation but had availability of water. After dissection, jejunum tissue (∼2 cm) was prepared, and kept immediately in Tyrode’s solution having proper oxygen supply. Tissue was hanged in organ bath (oxygenating Tyrode’s solution @ 37°C temperature), 1 g tension was set, and experiment was performed for 45 minutes to equilibrate it. Fresh Tyrode solution was flushed with an interval of 15 minutes before exposure of extract.
To observe spasmodic/antispasmodic response, extract doses ware added in cumulative way. Further experiments with provoked contractions (High-K+, Low- K+) followed by test substance were done to discover its possible mechanisms. Spasmolytic effect of plant extract may be due to blocking of Ca++ channels or opening of K+ channels. 21 The relaxation effect of plant extract on contractions triggered by Low K+ is usually considered due to possible involvement of K+-channel opening mechanism. 22
To determine its K+ channel activation, low-K+ triggered contraction pretreated with glibenclamide was evaluated with extract doses which inhibited its relaxing effect having similar pattern as cromakalim confirmed its K+-ATP potassium channel opening mechanism. 23
Preparation of Isolated Trachea
Experiments were done on isolated tracheal tissue to observe response on GIT. For tissue preparations Rabbits were kept on fasting overnight before experimentation but had availability of water. After dissection, trachea was prepared, and kept immediately in Kreb’s solution having proper oxygen supply. Tissue was hanged in organ bath (oxygenating Kreb’s solution @ 37°C temperature), 1 g tension was set, and experiment was performed for 45 minutes to equilibrate it. Fresh Kreb’s solution was flushed with an interval of 15 minutes before exposure of extract. 20 Pm.Cr extract doses were applied against baseline and stabilized contractions triggered by CCh and Low-k to find out its cholinergic and potassium channel opening mechanisms.
Preparation of Isolated Aorta
Vasoconstrictive and/or vasodilative effect on isolated aorta preparation was tested. Aorta tissues were prepared from rabbits for which rabbits were kept on fasting overnight before experimentation but had availability of water. After dissection, trachea was removed and kept immediately in Kreb’s solution having proper oxygen supply. Tissue was hanged in organ bath (oxygenating Kreb’s solution @ 37°C temperature), 2 g tension was set, and experiment was performed for 45 minutes to equilibrate it. Fresh Kreb’s solution was flushed with an interval of 15 minutes before exposure of extract. 20 Responses were recorded against stabilized baseline and contractions triggered by PE and low- K+.
In-Vivo Experimentation
Anti-diarrheal Activity
Anti-diarrheal effect of Pm.Cr was evaluated by a previously reported method.
24
Thirty-five rats were taken and divided into 7 groups @ 5 rats per group (100–200 g). Control group was given normal saline (10 mL/kg) and group 2 and 3 were given cromakalim and loperamide, (positive controls). The group 4 and 5 received different doses (100 and 200 mg/kg, p.o.) of Pm.Cr and the group 6 and 7 were pretreated with glibenclamide (GB) After 1 hour, each rat was given (10 mL/kg) castor oil orally and placed in separate box lined with filter paper to observe watery fecal drops for next 1,2,3, and 4 hours.
Statistical Analysis
The results are mean ± SEM. The median effective concentrations (EC50 value) with 95% (CI) were calculated with Graph Pad Prism (GraphPad, San Diego, California, USA: http://www.graphpad.com). The statistics was carried out using one-way (ANOVA) followed by Dunnett’s test in the case of in vivo, a probability (
Results
Preliminary Phytochemical Screening
Phytochemical analysis of Pm.Dcm indicated alkaloids, phenols, and terpenoids, but Pm.Aq contained flavonoids, glycosides, and tannins.
HPLC Examination
Pm.Cr retention time was compared with that of standard. Gallic acid, butylated hydroxytoluene, catechin, and quercetin were detected (Figure 1, Table 1). Comparison of standards and 

Response on Jejunum
Pm.Cr and its fractions were used to assess its response on isolated tissue of jejunum. Pm.Cr exhibited its relaxing effect on naturally contracted tissue at a concentration of 3 mg/mL, stabilized contractions triggered by Low K+, at a concentration range of .01–3 mg/ml of Pm.Cr with EC50 = .358 ± .052, 95% (C.I): .276-.464) (Figures 2 and 5A and 5B), which was inhibited in tissues pretreated with 3 μM glibenclamide (Figure 5C and 5D).
Pm.Aq exhibited its relaxing effect on naturally contracted tissue at concentration .01–5 mg/ml while contractions triggered by low K+ relaxed with EC50 = .082 ± .111, 95% (C.I): .040-.167 mg/mL (Figures 3 and 5B).
Pm.Dcm also revealed its spasmolytic response at .01-3 dose concentration with EC50 = .334 ± .147, 95% (C.I): .120–1.063 (Figures 4 and 5C) against Low-K+ as well as on naturally contracted tissue at concentrations .01–1 mg/ml.
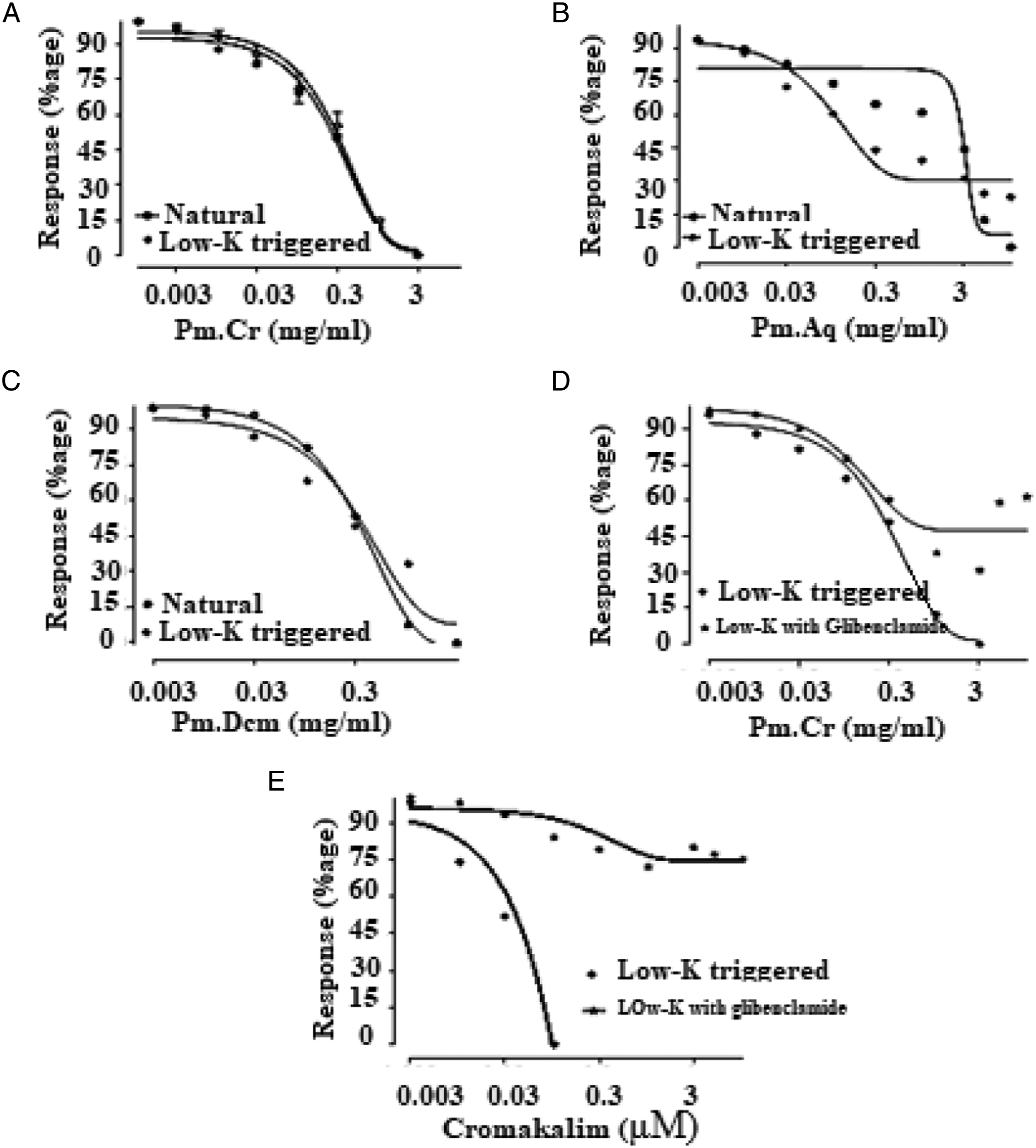
Recorded responses were compared with cromakalim (standard ATP-sensitive K+ channel opening mechanism) having its relaxing effect on contractions triggered by low K+ with EC50 = .184 ± .165, 95% (C.I): .021–.214 mg/mL (Figure 2E). Furthermore, 3 μM glibenclamide blocked the relaxing effect of Pm.Cr on contractions triggered by low K+ with EC50 =.17 ± .021, 95% (C.I): .041–.511 as cromakalim EC50 =.018 ± .100 95% (C.I): .02–.248 mg/mL (Figure 5E).
Response on Trachea
The crude extract and its fractions were assessed to record their response on isolated trachea. Pm.Cr exhibited its relaxing effect on contractions triggered by low K+ and CCh (1 μM) at concentrations 1 and 3 with EC50 = 2.483 ± .05793, 95% (C.I): 1.901–3.364 and EC50 = .611 ± .1045, 95% (C.I): .334–1.154, respectively (Figures 6 and 9A). 3 μM glibenclamide blocked the relaxing effect of Pm.Cr on contractions triggered by low K+ (Figure 9D).
Pm.Aq exhibited its relaxing effect on contractions triggered by low K+ at .3 mg/mL with EC50 = .053 ± .1761, 95% (C.I): .012–.335. Pm.Aq exhibited its relaxing effect on contractions triggered by CCh at .03 mg/mL concentration. (Figures 7 and 9B)
Pm.Dcm exhibited its relaxing effect on contractions triggered by low K+ and CCh (1 μM) at .3 with EC50 = .352 ± .3276. 95% (C.I): .050–.976 and, 95% (C.I): .010–.786, respectively (Figures 8 and 9C).
Recorded responses were compared with cromakalim having its relaxing effect on contractions triggered by low K+ with EC50 = .013 ± .001, 95% (C.I): .01–0. Furthermore, 3 μM glibenclamide blocked the relaxing effect of cromakalim having EC50 = .14, 95% (C.I): .031–.401 mg/mL (Figure 9E).
Response on Aorta
Pm.Cr tested on aorta did not produce any effect on baseline. Contractions triggered by low K+ and PE (1 μM) relaxed with Pm.Cr at a concentration of .01–3 and .01–5, respectively, with EC50 = .383 ± .063, (95% (C.I): .271–.548 mg/mL and EC50 = 05.29 ± .315, (95% (C.I): 3.993–11.56 (Figures 10 and 12A). Glibenclamide @ 3 µM blocked the relaxing effect of Pm.Cr on contractions triggered by low K+ (Figure 12B).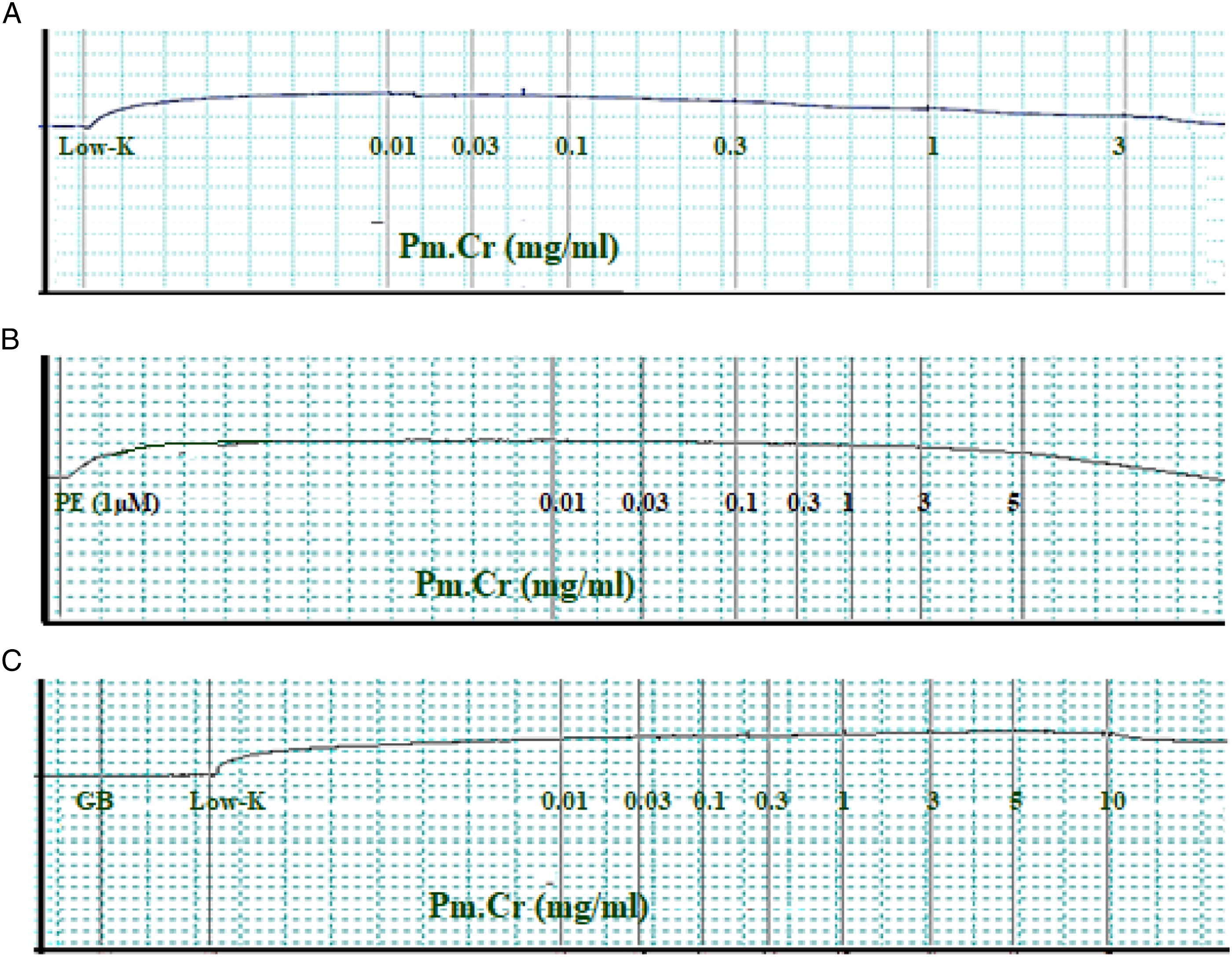
Pm.Aq completely relaxed contractions triggered by PE (1 μM) at the concentration of .01–3 with EC50 = .092 ± .010, (95% (C.I): .044–.1915 mg/mL but had not against low-K+. Pm.Dcm fraction revealed its vasorelaxant effect on contractions triggered by low K+ at concentration of .01–3 with EC50 = .864 ± .0011, 95% (C.I): .658–2.433 mg/mL. It also relaxed stabilized contraction of PE (1 μM) at .01–5 with EC50 = .02931 ± .213, 95% (C.I): .004–.280 mg/mL (Figure 11).
Recorded responses were compared with cromakalim which showed relaxing effect on contractions triggered by low K+ with EC50 = .01, 95% (C.I): .02–.277 mg/mL. Furthermore, 3 μM glibenclamide blocked the relaxing effect of cromakalim with EC50 = .15 (95% (C.I): .032–.437 mg/mL (Figure 12).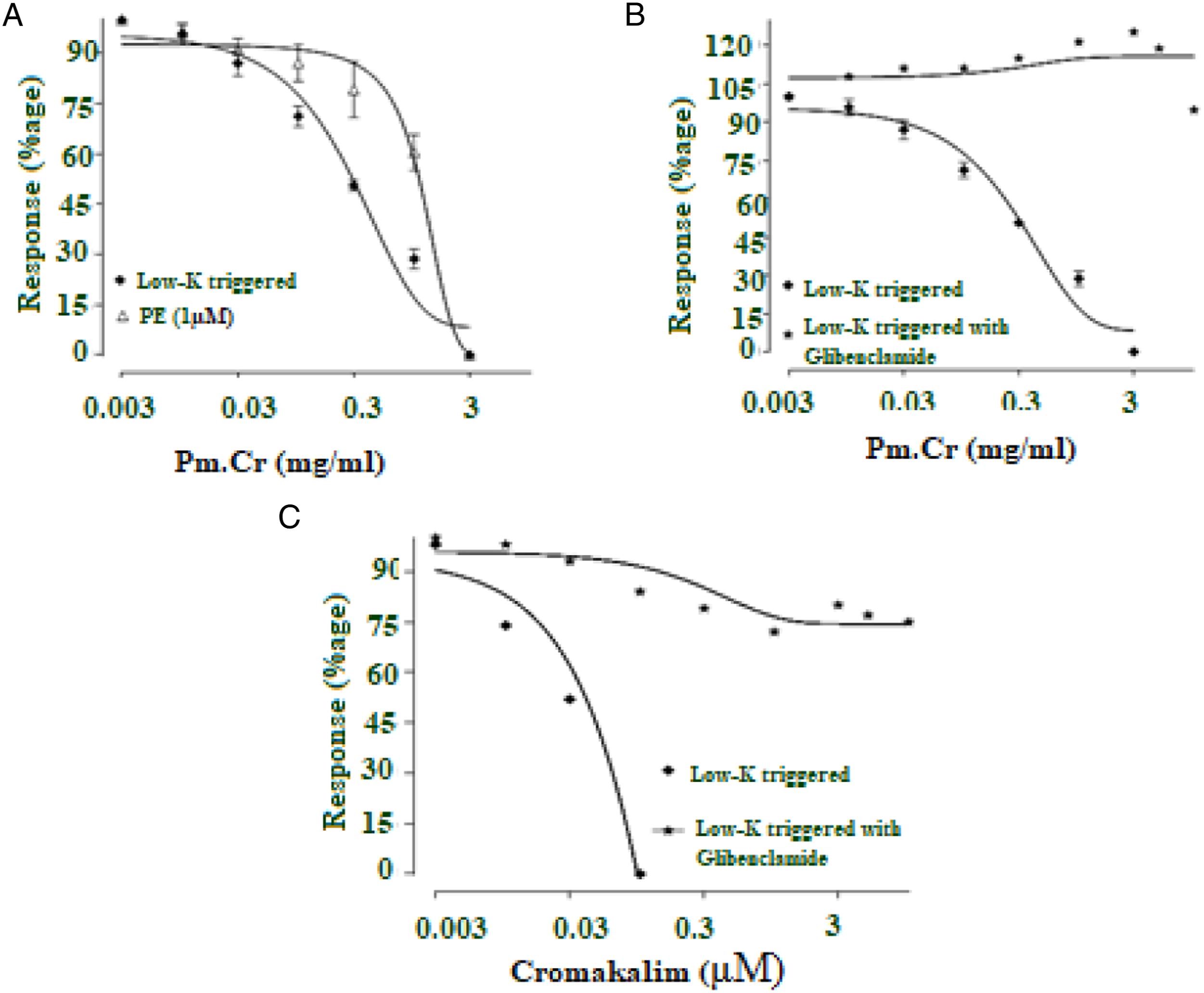
In-vivo Experimentation
Anti-diarrheal Effect of Pm.Cr
Pm.Cr exhibited potent anti-diarrheal effect, with comparison to control group, the tests groups had percentage protection of 64.65% (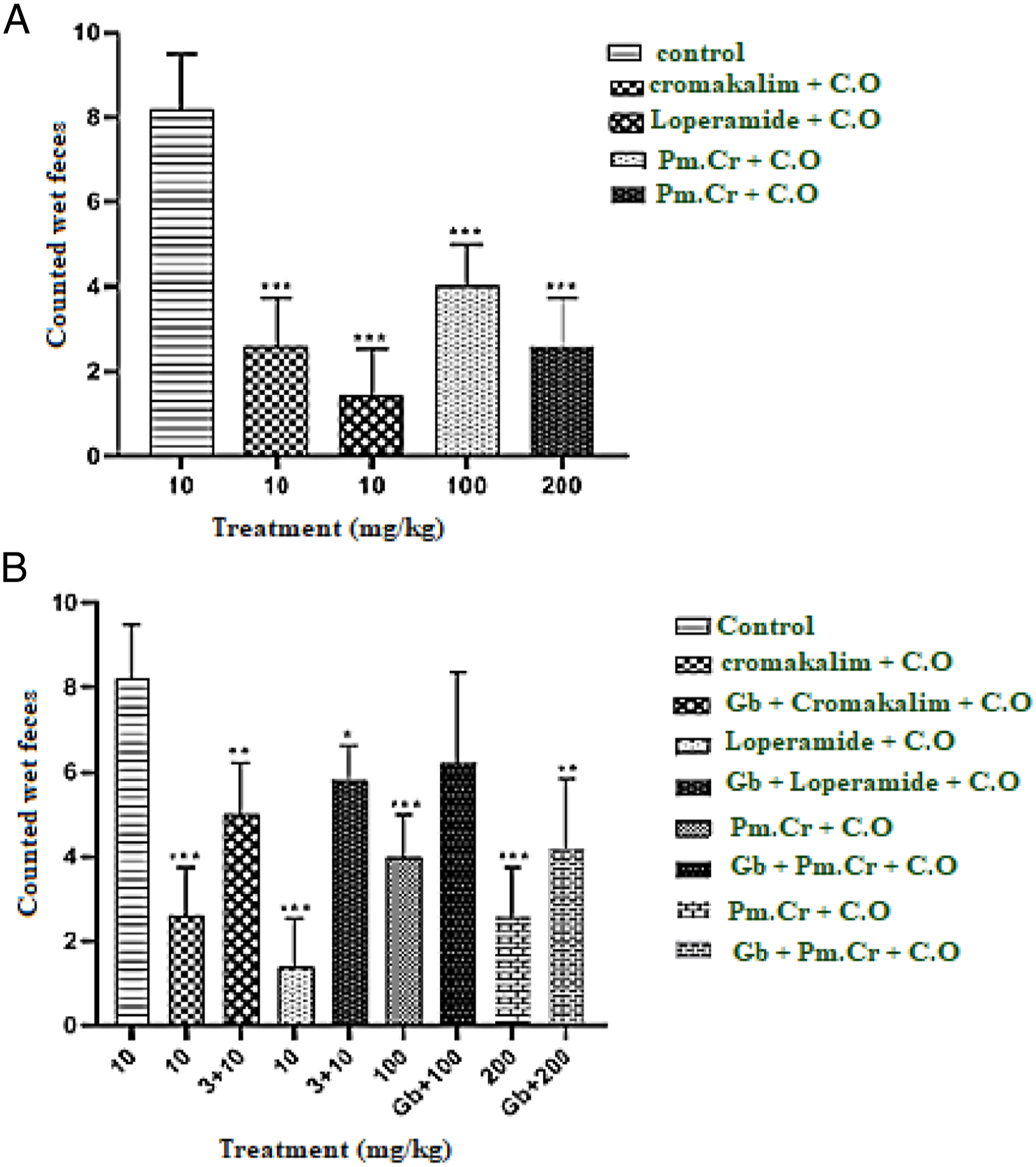
Discussion
Medicinal plants are gaining importance worldwide because of their potential therapeutic use and lack of side effects. Based on the folk medicinal use of
It has been perceived that plants exhibit inhibitory effects via activation of K+ channel. 20 The use of low-K+ induced depolarization causes opening of K+ channels. Due to presence of K+ channels in intestinal and epithelial cells, K+ channel openers produce relaxation of smooth muscle by decreasing intracellular calcium via hyperpolarization of membrane. 26
To evaluate whether the inhibitory effect of Pm.Cr is due to involvement of K+-channels, it was tested on stabilized contractions of jejunum triggered by low-K+, where it caused complete relaxation. The observed spasmolytic response may be due to the opening of potassium channels. 22 Moreover, this antispasmodic activity may be attributed to presence of flavonoid (catechin, quercetin),27,28 alkaloids, 29 tannins, 30 and quercetin 31 which are detected by preliminary phytochemical screening and HPLC studies. This K+ channel opening activity was found to be more dominant in organic fraction as compared to aqueous fraction. Potassium channel opener drugs have widespread therapeutic use in gastrointestinal disorders like diarrhea, 32 in CVS as antihypertensive, 33 and as bronchodilator in hyperactive airways diseases like asthma and cough. 34
Cromakalim (ATP dependent K+ channel opener) inhibited low K+ induced contractions. To confirm the K+ channel activation mechanism, low-K+ triggered contractions pretreated with glibenclamide was tested with extract doses which inhibited its relaxing effect having similar pattern as cromakalim confirmed opening of K+-ATP channel. 35
To check it bronchodilator effect, Pm.Cr was applied on contractions triggered by low K+ and CCh. Pm.Cr started a reduction of low- K+ and CCh-triggered contractions, being more potent against low K+. The results proposed that bronchodilator activity is due to K+ channel opening mechanism. Besides, flavonoids have potent bronchodilator effects and presence of flavonoids is confirmed by preliminary phytochemical and HPLC screening.36,37
Potassium channel opener drugs have widespread therapeutic usage in hypertension. 32 Pm.Cr was further investigated for possible effects on cardiovascular systems. Pm.Cr was applied on low K+, and PE-triggered contractions in the isolated aorta. Pm.Cr resulted in relaxation of low K+ and PE-triggered contractions. The relaxation of low K+ contractions showed momentous inhibition in GB pretreated tissues, which confirms the influence of K+-ATP channels as a mode of vasodilation that is used for hypertension treatment. 38 Catechin is used in the treatment of hypertension 28 while gallic acid acts as a cardio protective in cardiovascular problems. 39
In castor oil induced diarrhea, Pm.Cr showed antidiarrheal effect like cromakalim, 40 which is spasmolytic and antidiarrheal activity. Castor oil increases intestinal liquid and promotes diarrhea because of its active constituent, ricinoleic acid (hydrolysis of oil),41,42 and produces contractions in colon. 43 The detected antidiarrheal properties of the plant extract may be due to involvement of potassium channels which decreased by pretreatment with glibenclamide (GB; an ATP-dependent K+ channel blocker). 44 The observed antidiarrheal activity which may be due to the presence of flavonoids (catechin and quercetin), tannins, and terpenoids present in extract. 45
Conclusion
The in vitro and in vivo studies of
Footnotes
Acknowledgments
The authors want to acknowledge Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2022R93), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Authors’ Contributions
FS and MA planned the project. MA and AAA performed the experiments as well as worked on the statistical analysis of data and results interpretation. AAA, MH, LC, MI, CG, and FS drafted the manuscript. All authors have read and agreed to the submission of the manuscript.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was attained from Ethical Committee of Baha-Uddin Zakariya University, Multan (EC /12PhL-S/2018) dated 26 March 2018. Researchers agreed using the approved informed consent documented before their enrollment into study. All experiments on animals were performed in accordance with the relevant guidelines and regulations of Commission of Laboratory Animal Resources of Life Sciences (NRC, 2011), and was permitted by the Ethical Committee of BZU, Multan.
Funding
This research was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project Number (PNURSP20222R93) Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
