Abstract
Introduction
Herbal extracts from various parts of the hawthorn plant (Crataegus spp.) are used worldwide for a range of indications. Standardized hydroalcoholic extracts from the leaves and flowers such as WS(R) 1442 and LI 132 are marketed in western countries, mainly as an adjunctive treatment for mild to moderate chronic heart failure and other cardiac conditions.1,2 The active constituents include flavonoids and their glycosylated derivatives such as hyperoside, catechin/epicatechin-derived oligomeric procyanidins (OPC) and many other compounds. WS(R) 1442 is standardized to the OPC content, while LI 132 has a required content of flavonoids.
In traditional Chinese medicine (TCM), both the leaves and the fruits of the Chinese hawthorn (Crataegus pinnatifida Bge. or C. pinnatifida Bge. Var. major N.E. Br.) are used as the herbs shanzhaye and shanzha, respectively. Hawthorn fruit contain high levels of polyphenols, which is used in the concepts of TCM for stimulation to stimulate digestion, promote gastric function, improve blood circulation and remove blood stasis. 3
According to the Chinese Pharmacopoeia, only the fruits and leaves of C. pinnatifida Var. major and C. pinnatifida can be used as Chinese medicine. 3 The main form of herbal medicine used is the dried fruit, which is standardized to contain ≥5.0% citric acid in berries. When the leaves are used, their total flavonoid content should be ≥7.0%, calculated as anhydrous rutin. According to the Chinese Pharmacopoeia, specifically, the content of chrysin should be not less than 0.050%.3-5 Hawthorn fruit is usually prescribed in TCM to treat indigestion with epigastric distension or diarrhoea and for hypertension and hyperlipidaemia. 6 Chinese herbal medicines are also used as a supplementary alternative treatment and in drug development because of the empirical effectiveness and safety.7,8 Besides, some herbal materials used in TCM, such as hawthorn (shanzha), can be taken as part of the normal daily diet. 9
Furthermore, in animal models of atherosclerosis, hawthorn extracts have been found to decrease the serum levels of total cholesterol, low-density lipoprotein (LDL) cholesterol and triglycerides.10-12 When administered to rats fed with an atherogenic diet, the hawthorn fruit extract increased excretion of bile acids, depressed hepatic cholesterol synthesis and enhanced LDL-receptor activity. 13 In a previous study in apolipoprotein E (apoE) knock-out mice, hawthorn leaves flavonoids attenuated the development of atherosclerosis after 16 weeks of treatment, reducing the area of facial aortic lesions by 23.1% (P < .05). 14 Moreover, another study in rats demonstrated that hawthorn extract could prevent the development of atherosclerosis by regulating the serum level of cholesterol. 15 Feng et al found that hawthorn fruit acids were effective in reducing the levels of triglycerides, cholesterol, and glucose, as well as attenuating hepatic steatosis in rats with hyperlipidaemia induced by a high-fat diet. 16
In a clinical study conducted by Walker et al, 79 patients with diabetes taking various diabetic medications took placebo or hawthorn extract 1200 mg daily, which was equivalent to 6 g of dried flowering tops (standardised to 2.2% flavonoids), and a hypotensive effect was observed. 17 The diastolic blood pressure (BP) was reduced (P = .035) with hawthorn extract compared to placebo, but there was no significant effect on systolic BP. 17 Chen et al showed that serum lipids in patients with hyperlipidaemia were reduced by hawthorn, 18 but the study was not controlled. Thus, the potential clinical effects require further confirmation.
As a medicinal food, hawthorn fruit extract may be useful for lowering blood pressure and hyperlipidaemia. The purpose of this study was to evaluate the efficacy and safety of hawthorn fruit extract taken as a “beverage” for 2 months in patients with mildly elevated cholesterol and/or blood pressure. It is hypothesized that the hawthorn berry “drink” may have a hypolipidemic or antihypertensive effect and may be beneficial for long-term administration in patients with mild hyperlipidemia or hypertension. More importantly, it might provide a new strategy for simple control of blood pressure/lipid levels in patients as a nutraceutical taken on a daily basis.
Materials and Methods
Drug and Placebo Preparation
For the 5-month randomized, double-blind, placebo controlled, crossover intervention, the subjects were randomized into 2 groups, each with 36 subjects, who were assigned to receive hawthorn fruit drink (Group A) or placebo drink (Group B) in the first treatment phase of 8 weeks followed by a washout period of 4 weeks when no treatment was given. Then, they entered a crossover to the alternative treatment period of 8 weeks. The drink was prepared from the fruit of Crataegus pinnatifida Bge. Var major N.E. Br through a commercial fruit juice production operation by a contracted manufacturer who was able to source this specific hawthorn fruit.
Fresh hawthorn fruits were sorted, cleaned and crushed mechanically in a grinder. Water was added to extract the water-soluble content of the fruits, and the pH adjusted to around 3.05. The extract was clarified by digestion with pectinase and pressure filtered to remove insoluble materials. Sucrose was added to a final total content of approximately 19% including the sugar from the fruit. The juice prepared was then filtered and heat sterilized before filling in cans and cooling down. Using this method, 480 kg of water-soluble material was extracted from 7000 kg of fresh hawthorn fruit. Heavy metal and bacterial contents were tested and were within international acceptable limits. The nutritional contents of the 250 mL drink in each can were: 19% (w/w) carbohydrate (mostly sucrose), 0.2% (w/w) protein with a total of 78 kcal in each can. The drink contained the extract from 8 g fresh hawthorn fruit and 15 g sucrose per 100 mL of water. Concentrations of the major components of hawthorn were determined using a high performance liquid chromatography (HPLC) system, with conditions as described by Chang et al. 19 The main components were separated using a C18 column with a gradient elution of 5% and 25% acetonitrile in 25 mM phosphate buffer with the flow rate set to 1 mL/min. The placebo drink used in the study was formulated by the Department of Biochemistry at the Chinese University of Hong Kong. It contained the same calorie content from sucrose along with colourings and flavourings to closely mimic the appearance and taste of the fruit drink.
Subject Selection and Study Procedures
A total of 72 subjects were recruited from the general medical out-patient clinics at the Prince of Wales Hospital, Hong Kong. The inclusion criteria were adult patients aged 18 years to 75 years who were able to provide written informed consent and with either total cholesterol 5.7 to 7.5 mmol/L with triglycerides ≤4.5 mmol/L, or slight elevation of diastolic BP (95-100 mmHg) or systolic BP (130-150 mmHg), taken from the mean of 3 readings seated at rest, as described below. Patients taking one or more antihypertensive medication were allowed to enter the study if their BP was in the defined range. Exclusion criteria included subjects with additional cardiovascular risk factors such as diabetes mellitus or a history of smoking and subjects with any medical history or clinical evidence of established atherosclerotic cardiovascular disease or other conditions requiring long term medication.
Content and Daily Consumption of Major Phenolics in the Hawthorn Drink Compared With the Reported Content of an Ethanolic Extract of Dried Hawthorn Fruit.

aRecommended dose of dried hawthorn fruit is 9-12 g. Total daily consumption of major phenolics is calculated from 3 g taken 3 times daily. 24
bTotal daily intake of major phenolics of the current study (3 cans of hawthorn drink per day).
Randomization, Blindness and Power Calculation
Participants were randomized to Group A or Group B with matching cans containing hawthorn extract or placebo drink according to a randomization code. Participants were assigned blindly in order of their enrolment into the trial. Hawthorn fruit drink and the matching placebos were supplied to the participants by the Department of Biochemistry, the Chinese University of Hong Kong. The drinks were transported to patients’ home directly from the central stock. Other team members, who were responsible for the clinical assessment, were blinded to the randomization code. Participants were instructed to consume one can at breakfast, another before the evening meal, and a third at any time during the day. The clinical study was conducted from June 2000 to August 2001 to include recruitment and completion of the clinical study.
A power calculation based on detecting a decrease in total cholesterol of .5 mmol/L with a standard deviation of .8 mmol/L with 90% power and P < .05 comparing hawthorn drink with placebo, predicted the cross-over study required at least 23 patients in the 2 randomised groups to complete the study.20,21
Ethical Issues
The study was performed in accordance with Good Clinical Practice (GCP) and the Principles of the Declaration of Helsinki. All procedures involving human subjects were approved by the Joint Chinese University of Hong Kong-New Territories East Cluster Clinical Research Ethics Committee with approval number CRE2000.051 dated 2 May 2000. Written informed consent was obtained at the time of enrolment from all subjects. This study was reported with respect to the recommendation for reporting randomized clinical trials as the statement of Consolidated Standards of Reporting Randomized Clinical Trials (CONSORT) in this article. The trial has been registered at https://www.clinicaltrials.gov/ as NCT04672278.
Outcome Assessment
The primary objective of this study was to assess the hypolipidaemic and hypotensive effects, as well as the safety, of the hawthorn fruit drink in patients with mild hyperlipidaemia and/or hypertension. This was performed by monitoring changes in total cholesterol, LDL cholesterol, and BP from baseline to the end of the treatment period.
Seated BP was recorded as the mean of 3 readings taken at 1-minute intervals after 5 min rest sitting, measured by a Dinamap 8100 automated BP monitor (Critikon, Florida, USA). Anthropometric parameters including height and weight were recorded, to calculate the body mass index (BMI) as weight in kg/(height in m) 2 as an index of general obesity.
At the start and end of each treatment period, blood samples were taken after overnight fasting for measurement of lipid profile, including total cholesterol, high-density lipoprotein (HDL) cholesterol, triglyceride and the calculated LDL-cholesterol. Total cholesterol was measured enzymatically (Centrichem Chemistry System, Baker Instruments Co., Allentown, USA). HDL-cholesterol was determined following fractional precipitation with dextran sulphate-MgCl2 and LDL-cholesterol was calculated by the Friedewald formula. 22 Triglycerides were measured by the glyceryl dehydrogenase reaction following the hydrolysis of the triglyceride (Centrichem Chemistry System, Baker Instruments Co., Allentown, USA). The long-term imprecision of the triglyceride assay was 6.9% at 1.02 mmol/L and 4.6% at 2.18 mmol/L.
Blood was taken for the measurement of renal and liver function tests, fasting plasma glucose, urate and complete blood count, which were measured using standard methods as previously described. 23 All assays were performed on the same day as the blood collection.
Statistical Analysis
Only the patients who completed the study were included in the statistical analysis for efficacy (n = 61, 32 in group A, 29 in group B) using the Statistics Package for Social Sciences (SPSS for Windows, version 24, 2016, SPSS Inc, Chicago, USA). Normal probability plots were examined to determine whether any of the variables required transformation to improve linearity. Data from normally distributed parameters are presented as mean ± SD. The only transformed variable in the analysis was LDL-cholesterol. The values of this variable were anti-logged for presentation and expressed as geometric mean (95% confidence interval of the mean).
Treatment-time interactions in the crossover study between different parameters and hawthorn treatment effects were tested by a mixed linear model to evaluate treatment effects and treatment-time interaction in a single analysis. Because of significant treatment-time interactions for diastolic BP, BMI and LDL-cholesterol, only the results for these parameters in the first treatment period were analyzed by independent t test, with 32 subjects in the active treatment arm and 29 subjects in the placebo arm.
Diastolic BP, BMI and LDL-cholesterol changes were then analyzed separately between baseline and after 4 weeks and 8 weeks treatment in the 2 groups in chronological order using paired t-tests. Unpaired t test analyses were used to compare the difference between Group A and Group B in the first study period. The data of the 2 periods for the other variables apart from these 3 parameters were pooled for further analysis. Paired t-tests were used to identify differences of continuous variables between the baseline and the 4-week and 8-week treatment periods, respectively, in Group A and Group B combined. The absolute difference between baseline and 8 weeks for Group A and Group B combined were also calculated to identify any significant effect of hawthorn treatment. Significance was taken as P < .05 (2 tailed for all the statistical tests).
Results
Hawthorn Drink Content
HPLC analysis of the hawthorn drink containing extract from 80 g fresh fruit per L showed concentrations of measured ingredients were procyanidin B2 255 mg/L, epicatechin 218 mg/L, chlorogenic acid 50 mg/L, isoquercitrin 6.2 mg/L and hyperoside 1 mg/L (Table 1). Each 250 mL can of hawthorn drink contained 78 kcal as sugar. The daily intake of these phenolic compounds was calculated to be greater with the hawthorn drink than the recommended dose of 9 g 3 times daily from an ethanolic extract of dried hawthorn fruit, except for the hyperoside (Table 1).
Demographics and Baseline Characteristics
From 72 eligible subjects randomized in the study (Figure 1), there were 39 males and 33 females entered and 11 withdrew before completing the study. Of these subjects, 3 withdrew consent, 2 of them due to abdominal discomfort, and the remaining subjects were withdrawn by the investigator because of intercurrent illnesses including suspected ischaemic heart disease, chest infection and other symptoms, so the data from those subjects were not included in the per-protocol outcome analysis. Consort diagram of patient recruitment in hawthorn study.
Baseline Anthropometric and Clinical Characteristics and Plasma Lipids of the Patients in the Per-Protocol Analysis According to Initial Treatment.

Values are given as mean ± standard deviation or number of patients.
Anti-hypertensive medications include: Angiotensin converting enzyme inhibitor (ACEI): lisinopril; α-adrenergic blockers: prazosin; β-adrenergic blockers: atenolol, metoprolol; calcium channel blocker (CCB): nifedipine, lercanidipine, diltiazem, amlodipine; diuretics: indapamide, hydrochlorothiazide and triamterene.
aLDL-cholesterol was analysed with natural log transformation due to the skewness of data.
bNo subject in this study was taking any lipid lowering agents or antidiabetic medication.
cPatients taking more than one anti-hypertensive medication were allowed to enter the study.
Interaction Analysis
Changes in Blood Pressure, Anthropometric Measurements, Glycaemic Parameters and Plasma Lipid Profile for the 61 Patients Combined With No Treatment-Time Interaction, and for the First 8 Weeks’ Treatment With Placebo or Hawthorn Drinks Separately for Parameters Which Showed a Treatment-Time Interaction (BMI, Diastolic BP, and LDL-Cholesterol).

*P < .05, **P < .01, ***P < .001 compared to respective baseline.
aChanges in lipids in the first 8 weeks’ treatment with placebo (n = 29) or hawthorn (n = 32) for parameters which showed a treatment-time interaction.
bAll data are shown as Mean ± SD except LDL-C.
cArithmetic mean (95% C.I.): LDL-C, low-density lipoprotein cholesterol was analysed with log 10 transformation due to the skewness of data.
dNo significant difference was found between placebo and hawthorn treatments by paired-t test comparison, unless specified.
eNo significant difference was found between the responses to placebo and hawthorn treatments by paired-t test comparison, unless specified.
fMean values were significantly different from those of placebo group (P < .05).
Changes in Renal and Liver Function for the 61 Patients Combined With No Treatment-Time Interaction, and for the First 8 Weeks’ Treatment With Placebo or Hawthorn Drinks Separately for Parameters Which Showed a Treatment-Time Interaction (Alkaline Phosphatase and Total Bilirubin).

*P < .05, **P < .01, ***P < .001 compared to respective baseline.
aChanges in the first 8 weeks’ treatment with placebo (n = 29) or hawthorn (n = 32) for parameters which showed a treatment-time interaction.
bNo significant difference was found between placebo and hawthorn treatments by paired-T test comparison, unless specified.
cNo significant difference was found between the responses to placebo and hawthorn treatments by paired-T test comparison, unless specified.
Effects on Anthropometric Measurements
Body weight and BMI increased significantly over the 8-week periods in Group A and Group B. The mean change in body weight after 8 weeks compared to baseline with Group B (.94 kg; 95% confidence interval [CI]: .52, 1.36 kg) was double that of Group A (.42 kg; 95% CI: −.85, 1.69 kg), but the difference between the 2 groups was not statistically significant (P > .05; Table 3). Figure 2 shows the changes of BMI of the 2 groups separately, Group A (hawthorn) and B (placebo) in chronological order with the end of the washout period indicated as the baseline of period 2. The BMI of Group A increased significantly after either hawthorn or placebo treatment, and the BMI of Group B increased significantly with placebo treatment, but not after hawthorn treatment. This result may be related to individual patient factors. Overall, the change in BMI over the first treatment period was not significantly different between Group B (.5 [.20, .79] kg/m2) and Group A (.45 [.31, .60] kg/m2). Changes in BMI during the study periods for group A (hawthorn/placebo) and group B (placebo/hawthorn).
Effects on Blood Pressure, Lipid Profile and Other Metabolic Parameters
The readings for systolic BP in Group A and Group B were the highest at week 8, at 139 ± 10 and 140 ± 10 mmHg, respectively (Table 3). However, there was no significant increase in diastolic BP after the first 8 weeks treatment in Group B (1.7 ± 6.9 mmHg), whilst there was a significant increase in Group A (3.1 ± 7.6 mmHg, P < .01). Figure 3 shows the changes in diastolic BP of Groups A and B separately, indicating Group A had higher baseline values and levels peaked after 8 weeks of both the 2 treatments returning to baseline levels after the wash out. The diastolic BP of Group A increased significantly after 8 weeks of treatment with hawthorn or placebo, and for Group B it was not significantly changed. Figure 4 shows that total cholesterol increased with hawthorn or placebo treatment in Group A, but not in Group B, which was largely due to increases in both LDL-cholesterol and in the cholesterol carried in very low-density lipoprotein (VLDL) particles, as the triglyceride levels tended to increase over the 8-week periods with both treatments. The LDL-cholesterol levels in the 2 groups were not significantly different at baseline, but values in subjects receiving hawthorn first were slightly higher and increased more during treatment with both drinks (Table 3, Figure 4). Changes in diastolic BP during the study periods for group A (hawthorn/placebo) and group B (placebo/hawthorn). Changes in LDL-cholesterol and total cholesterol during the study periods for group A (hawthorn/placebo) and group B (placebo/hawthorn).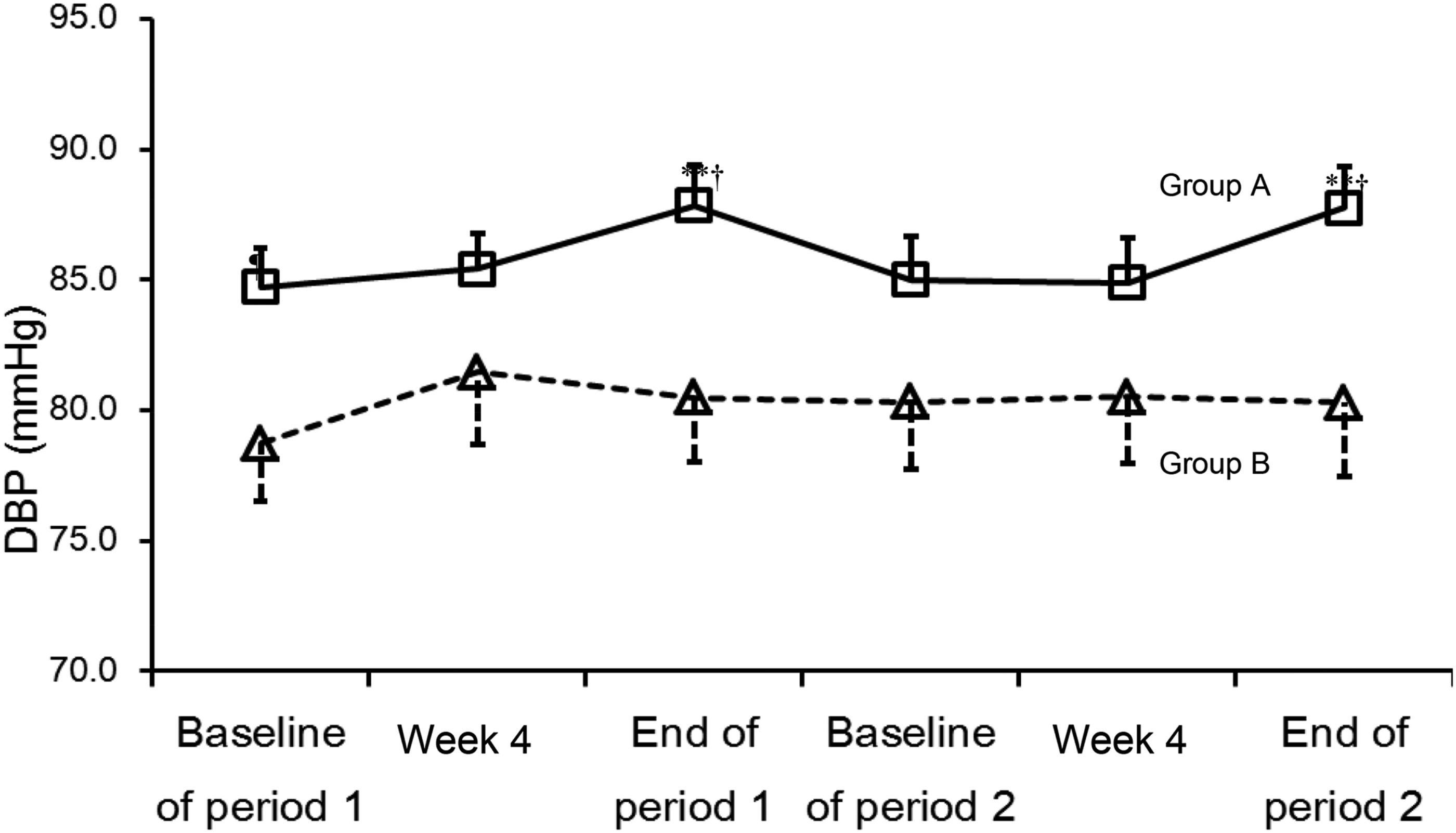

HDL-cholesterol was lowest at week 4 with the hawthorn drink (1.26 ± 0.29 vs 1.32 ± 0.27 mmol/L at baseline, P < .05) but there was no difference from baseline at the end of the study periods with either the hawthorn or the placebo drink.
Both Groups A and B had the highest blood glucose levels after 4 weeks and levels remained elevated compared to baseline at week 8 (placebo 5.56 ± 1.06, hawthorn 5.49 ± 1.04 mmol/L), but only the value with placebo was significantly higher (P < .01) than baseline (Table 3).
Tolerability, Safety and Adverse Effect Evaluation
After placebo treatment, there was a significant increase in alkaline phosphatase and alanine transaminase and decrease in albumin at week 8 (Table 4), while there was no significant change among these 3 parameters at the end of the hawthorn treatment period. No significant changes were seen in plasma creatinine, potassium and sodium in both groups.
Subjective Symptoms and Adverse Events Reported by all 72 Patients That Entered the Study.
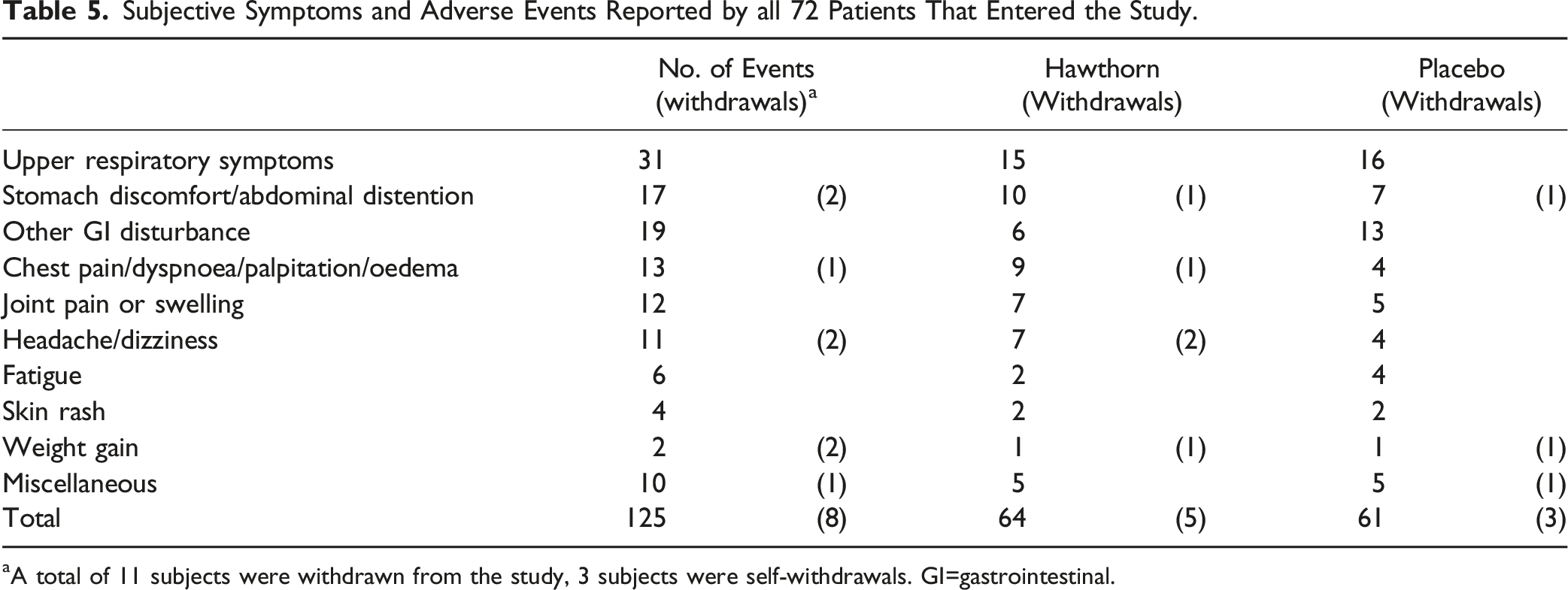
aA total of 11 subjects were withdrawn from the study, 3 subjects were self-withdrawals. GI=gastrointestinal.
Discussion
In this study, there were increases in body weight, and therefore BMI, and in systolic BP, total cholesterol and blood glucose with both treatments, which were not statistically different between treatments. Elevations of body weight and glucose were observed after 4 weeks with both treatments and subsequently there was a tendency for reduction in body weight and plasma glucose towards baseline levels between weeks 4 and 8 in the group taking hawthorn drink. Furthermore, a significant increase in diastolic BP and a decrease in HDL-cholesterol were found with the hawthorn treatment. LDL-cholesterol, diastolic BP and BMI were analysed without combining the 2 groups due to the carry-over effects. After the 1-month washout period, the lipid profile had almost returned to the original baseline level. The pattern of change was repeated in the second period of the study. However, it appeared that the patients in Group A (hawthorn/placebo) were more sensitive to the interventions resulting in greater changes in LDL-cholesterol and diastolic BP than those in Group B (placebo/hawthorn), possibly because of a more adverse baseline lipid profile with higher total cholesterol and tendency to higher LDL-cholesterol. The increase in LDL-cholesterol and BP appears to be related to the increase in calorie intake and body weight in this more hyperlipidaemic patient group, but we did not make an assessment of total dietary intake to confirm that.
The results of the present study differ from some of the animal studies with hawthorn extracts. Zeng et al found that the active ingredient of hawthorn berry powder significantly reduced serum lipid levels (P < .05) and cholesterol/triglycerides, which also included very low-density lipoprotein (VLDL) and low-density lipoprotein (LDL) levels in hyperlipidaemic mice. 11 Moreover, Zhang et al. also found that an ethanol extract of hawthorn berry significantly reduced body weight and serum total cholesterol, triglyceride, LDL-cholesterol, and liver cholesterol levels in hyperlipidaemic mice as compared to the control group. In addition, Total Antioxidant Capacity (T-AOC), Superoxide Dismutase (SOD) and Glutathione Peroxidase (GSH-PX) activities were significantly elevated in mice in the hawthorn berry group (P < .05). 12 Lipid metabolism in animal models differs from humans so it may not be appropriate to compare the animal results with this clinical study directly and different types of hawthorn fruit extracts have different constituents.10,18 These animal study results indicated that hawthorn berry extract may have an effect to reduce hyperlipidaemia.
The present study demonstrated a small decrease in HDL-cholesterol with the hawthorn treatment, which was different from a small randomized controlled clinical trial in Australia. That study showed a significant (P = .026) increase in HDL-cholesterol from 1.47 ± .37 at baseline to 1.54 ± .44 after 4 weeks treatment with a hawthorn fruit and Chinese kiwifruit-extract compound, but no change with a placebo. 25 In the current study, some of the ingredients in the hawthorn drink were identified and quantified. Among the chromatographic peaks tested, 5 major peaks were identified as polyphenolic compounds (procyanidin B2, epicatechin, chlorogenic acid, isoquercitrin and hyperoside). Some of the previous studies have not quantified the potential active ingredients of hawthorn containing formulations,6,26 but the main difference in the present study is probably related to the excess energy gain of the sugar-sweetened hawthorn fruit drink.27,28
Hawthorn fruit is thought to be beneficial to the cardiovascular system, partially due to its effect on serum cholesterol and BP and a range of other effects including antiplatelet aggregation effects, vasodilating effects, endothelial protective effects, and antiarrhythmic effects as described in detailed reviews.6,29 It has been suggested that hawthorn fruit can reduce serum cholesterol through several mechanisms including inhibition of 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, activation of LDL receptors, inhibition of absorption of dietary cholesterol by down-regulating the activity of intestine acyl-coenzyme A: cholesterol acyltransferase (ACAT), conversion of cholesterol to bile acids, and activation of peroxisome proliferator-activated receptor α (PPAR-α) in adipose tissue.10,13,26,30 Moreover, a randomized placebo controlled clinical trial demonstrated that the mean systolic BP of prehypertensive patients treated with a commercially available combined capsule containing Allium sativum, Crataegus, Ortosiphon and Hibiscus was lower than the placebo treated group (124 ± 9 vs 132 ± 7 mmHg, P < .0001) after 12 weeks treatment. 31 However, a previous study in prehypertensive and hypertensive patients showed no evidence that short term treatment with capsules containing standardized hawthorn extract had a significant BP lowering effect or influenced nitric oxide-mediated vasodilation. 32 In the current study, the hawthorn drink was used instead of capsules or pills to assess whether hawthorn could be used as a nutraceutical.
Many previous studies have shown that prolonged intake of excess energy from sucrose or fructose is associated with adverse effects including increased BP, 33 childhood obesity 34 and development of type 2 diabetes. 35 The present study subjects had mean BMI >25 kg/m2 so at least half would be considered overweight by Asian standards, and they were mildly hypertensive and hyperlipidaemic and already had a high risk of developing diabetes. A 5-year follow up cohort study indicated the increased risk in weight gain and developing diabetes (relative risk: 1.42 and 1.70 respectively) of Chinese who had frequent (≥2 soft drinks) per week compared to those that rarely consumed sweetened drinks. Hence, any beneficial effect of hawthorn extract may have been negated by the adverse effects of the increased intake of sucrose in the hawthorn extract drink used in our study. It is generally accepted that sugar-sweetened beverage consumption is associated with increased cardiometabolic risk and a recent analysis in women from the California Teachers Study found that consumption of ≥1 serving per day of sugar-sweetened beverage was associated with increased risk of cardiovascular disease, coronary revascularization and stroke. 36
A previous review of the chemistry of hawthorn, detailed at least 27 different Crataegus species and 2 hybrids native to Europe, Asia and North America. In the chemical constituents studied, there are 49 distinct flavonoid compounds, 5 hydroxycinnamic acids, 6 sugars, 10 organic or phenolic acids, 26 terpenes and 56 essential oil constituents reported to date. 37 In the current study, the hawthorn drink was made from an aqueous extract from fresh hawthorn berries. The major form of usage in TCM is the dried fruit. The common dosage in TCM is 9-12 g of the dried fruit, 2 to 3 times daily, as herbal tea or as one of the herbs in a prescription. 30 The chemical profile of the fruits is similar to those of other flowering top parts, but the amount of the individual flavonoids and OPCs are different, with the fruits containing more hyperoside and the leaves having higher contents of vitexinrhamnoside. 38
The daily consumption of 5 major phenolics (epicatechin, procyanidin B2, chlorogenic acid, hyperoside, isoquercitrin) was assessed in the current study. As hawthorn fruits contain substantial amounts of organic acid, they exist and are stable in acidic conditions and the pH of the study drink was 3.05. 19 Moreover, temperature may also be an important factor in the maintenance of the stability of the active ingredients of the study fruit drink. 24 The hawthorn drinks were stored at 4°C and delivered to the subject’s home after each monthly visit. It was recommended to the study subjects that the drinks should be stored at room temperature and consumed within one month. It has been previously estimated that under these conditions the loss of active ingredients should be less than 10%, but this would not greatly affect hawthorn performance. 24
In this study, alanine aminotransferase and alkaline phosphatase were slightly elevated in the placebo group after 8 weeks, which may be associated with weight gain and increased liver fat content in some susceptible subjects. This should not have a great effect on the subjects. In addition, the rationale for adding sugar to hawthorn beverages is to make them more palatable. We considered that the subjects may have been reluctant to consume unsweetened hawthorn beverages because they are too acidic and may cause stomach upset. However, overall, apart from weight gain and related changes, there were no significant adverse effects of hawthorn fruit drinks observed. A meta-analysis of monotherapy hawthorn trials reported that mostly mild to moderate adverse events were found in 5577 patients in the analysis, including dizziness/vertigo and gastrointestinal complaints, which were similar to those in our study.39,40
There are a number of limitations in this study, such as not controlling all of the relevant variables, especially controlling the amount of calorie intake, which may have influenced the results. Besides, 15% (w/w) sucrose was added to the hawthorn beverage to improve the palatability and to avoid gastrointestinal reactions, which may not only increase the risk of weight gain and cardiovascular risk factors, but also mask the benefits of hawthorn itself. Therefore, when designing similar trials in the future, the addition of sugar should be minimized, and it may be possible to formulate the extract into capsules or tablets for better patient compliance. In addition, it may be more effective to use questionnaires to collect adverse reactions from the subjects and to assess dietary intake in general and specifically of phenolics. Hawthorn extracts have been shown to have therapeutic effects on heart failure, lowering serum levels of total cholesterol, LDL-cholesterol, and triglycerides, and antihypertensive effects in some studies, but the evidence from clinical studies is not conclusive. Therefore, studying the therapeutic effects of hawthorn extracts in clinical settings is an important research direction in the future.
Conclusion
In this study, the hawthorn extract beverage tended to be associated with less increase in body weight and fasting plasma glucose than the placebo drink with matching calorie intake, but the differences were not statistically significant. During the active and placebo interventions with increased caloric intake there were reversible adverse effects on body weight, blood pressure, blood glucose, and blood lipids, suggesting potential harm if sugar-sweetened beverages are consumed over a long period of time. It may be useful to test an alternative formulation of hawthorn berry extract without additional calories to determine if this is beneficial to patients with mild hypertension and/or hyperlipidaemia on a long-term basis.
Footnotes
Acknowledgments
We would like to thank Ms. Karen Wong for excellent assistance in patient recruitment, Ms. Kathy YC Tsang and Ms. Emily WM Poon for technical support, Ms. Hermia WF Kwong, Ms. Eliza CY Choy, Ms. Winnie MN Yeung, Ms. Maybo SL Lin and Ms. Swen YW Ip for nursing support and all the patients who participated in the study.
Author Contributions
Conceptualization, B.T. and W.K.H; Methodology, B.T. and W.K.H; Software, W.Z., T.T.C. and B.S.F.; Validation, B.T.; Formal Analysis, W.Z., T.T.C. and B.S.F.; Investigation, W.Z., T.T.C. and B.S.F.; Resources, B.T.; Data Curation, W.Z., T.T.C., J.C.C. and B.S.F.; Writing-Original Draft Preparation, W.Z., T.T.C. and B.T.; Writing-Review & Editing, W.Z., T.T.C., W.K.H., B.T. and J.C.C.; Visualization, W.Z. and B.T.; Supervision, B.T. and W.K.H; Project Administration, B.T.; Funding Acquisition, B.T.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Hawthorn and placebo drinks were funded by the Department of Biochemistry, the Chinese University of Hong Kong, Hong Kong.
