Abstract
Introduction:
The mechanism by which intestinal mucosal barrier is damaged in severe acute pancreatitis (SAP)-associated impairment is not fully understood.
Methods:
We established an
Results:
We found significant injury of the intestinal mucosal barrier in SAP rats, with overexpression of Beclin-1, LC3, and p65. Pyrrolidine dithiocarbamate showed a bidirectional effect in protecting SAP rats. A high dose of PDTC aggravated disease in rats, while a low or medium dose of PDTC pretreatment, was able to alleviate tissue damage. Pyrrolidine dithiocarbamate changed the expression of Beclin-1, LC3, and p65 in the intestines. The fatty acid-binding protein level was increased in SAP rats with high-dose PDTC or without PDTC pretreatment and was reduced in SAP rats with low- or medium-dose PDTC exposure.
Conclusions:
Autophagy is involved in the impairment of intestinal mucosal barrier during SAP. A suitable dose of PDTC (1 or 10 mg/kg) may decrease the severity of SAP by inhibiting autophagy in intestinal mucosal cells.
Introduction
Pancreatitis is initiated by activation of trypsin within the pancreas, resulting in further activation of various proteases that can damage cells. Acute pancreatitis is commonly induced by gallstones and alcohol, although drugs and certain genetic mutations can also cause this intractable disease. 1 Acute pancreatitis is characterized by edema, hemorrhage, and necrotic inflammation of different severities. Patients suffering from this disease can present with abdominal pain, nausea and vomiting, and fever. An elevated serum level of amylase is the most common sign of acute pancreatitis and is very informative for diagnosis. The prognosis of acute pancreatitis is highly dependent on the severity. Mild acute pancreatitis only displays pancreatic edema and patients had a good prognosis. Severe acute pancreatitis (SAP), however, may have hemorrhage and necrosis in pancreas, leading to secondary abdominal infection, peritonitis, and even shock. The mortality of SAP patients can be high. In clinic, anti-inflammation and supportive care are main strategies for the treatment of SAP. Thus, more detailed mechanisms of SAP are urgently needed to develop a novel treatment.
It has been reported that intestinal mucosa was damaged in case of SAP. The epithelial cells of intestinal mucosa become denatured and necrotic, leading to ischemia and reperfusion of the intestine, dysregulation of intestinal microbiota, and increased permeability of mucosal barrier. These pathological alterations can further induce bacteria and toxin translocation and inflammatory cytokine release, which change SAP from a local disease to a systemic disease. However, the mechanisms of disturbance of intestinal mucosa barrier are far from understood. Previous studies have shown enhanced apoptosis of intestinal epithelial cells in SAP. Autophagy, also known as type II programmed death, is different with but closely connected to apoptosis. 2 In this study, we aimed to investigate whether autophagy plays a role in SAP-related death of intestinal epithelial cells.
Autophagy is a cellular process exerted by lysosomes and is essential for cell survival, differentiation, and homeostasis. 3 The nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling is deeply involved in the regulation of autophagy, and a complicated crosstalk between NF-κB signaling and autophagy has been revealed. 4 Although in many cases, inhibition of NF-κB signaling showed induction of autophagy, 5,6 pyrrolidine dithiocarbamate (PDTC), an NF-κB inhibitor, was reported to be able to suppress autophagy. 7 In addition, using a taurocholate-induced acute necrotizing pancreatitis rat model, Yang and colleagues proved that NF-κB signaling stimulated autophagy in the pancreas. 8 However, in the background of SAP, the interplay between NF-κB signaling and autophagy was less studied, especially in other organs injured by SAP.
Here, we used a rat model to study the autophagy of epithelial cells in terminal ileum. The associations among autophagy, intestinal mucosa permeability, and oxidative stress under PDTC treatment were demonstrated in this study.
Materials and Methods
Animal Model
Sprague-Dawley rats (∼2 months old) weighing 250 ± 30 g were obtained from Animal Center of Hebei Medical University. Rats were cultured for at least 1 week before subjected to experiments. Rats were randomly assigned to 5 groups with 24 rats per group. The normal control group was cultured normally with only necessary vehicle (normal saline) treatment. The SAP group was peritoneally injected with 20%
Hematoxylin and Eosin Staining
The histological analysis was performed as previously described. 8 In brief, formalin-fixed paraffin-embedded tissue was cut into 4-µm thick sections. The sections were then stained with hematoxylin and eosin (H&E) using a standard method. The slides were blindly reviewed by an experienced pathologist.
Western Blotting
Intestine tissues (100 mg) were collected after 12 or 24 hours of
Fatty Acid-Binding Protein Assay
Peripheral blood was taken from rats, and serum was isolated. The fatty acid-binding protein (FABP) level was detected using Quantikine ELISA Kit (R&D Systems, Minneapolis, Minnesota) according to the manufacturer’s instructions.
Superoxide Dismutase and Malondialdehyde Detection
Intestine tissue homogenate was acquired after 12 or 24 hours of
Statistical Analysis
All data are presented as the mean (standard deviation). Multiple group comparison was performed using one-way analysis of variance. Comparison of different time points in mice with the same treatment was conducted using unpaired Student’s t-test. Statistical analysis was performed using SPSS version 16.0 (SPSS Inc., Chicago, Illinois). P < .05 was considered as statistically significant.
Results
Pyrrolidine dithiocarbamate Ameliorates l -Arginine-Induced Pancreatic Damages in Rats
Intraperitoneal administration of

Histology of pancreas. Rats were treated as indicated. After 12 or 24 hours of
Pyrrolidine dithiocarbamate Improves Intestinal Damage in SAP Rats
To investigate whether PDTC can influence intestines in case of SAP, we first examined the pathological alterations of intestines in the rats. In contrast to the healthy manifestations of intestines in control rats, SAP rats displayed intestinal pneumatosis. Hematoxylin and eosin staining of rat intestines showed damage of intestinal villi and loss of epithelial cells (Figure 2). Furthermore, congestion, edema, and inflammation were observed in the intestinal lamina propria. All these pathological changes were severe in rats with long-term

Histology of intestine. Rats were treated as indicated. After 12 or 24 hours of
Fatty acid-binding protein was reported to be correlated with gut dysfunction and could be used for evaluating the severity of SAP in patients.
11,12
We then tested serum level of FABP in rats. As expected, FABP level of the SAP group was much higher than that of the control group (Figure 3). Consistently, the P100 group also had an elevated FABP level, which was comparable to that of the SAP group. Of note, the FABP level in both P1 and P10 groups was reduced compared to that of the SAP group, although it was still significantly higher than that of the control group. At 12 hours after

The FABP level in different groups. Rats were treated as indicated. After 12 or 24 hours of
Pyrrolidine dithiocarbamate Reduces p65 Expression in the Intestine of SAP Rats
Since PDTC is a known NF-κB inhibitor
13
and we showed that PDTC reduced intestinal damage in SAP rats, we thus hypothesized that NF-κB might play a role in SAP-associated intestinal damage. We detected overexpression of p65 in the intestine tissue from SAP and P100 rats compared to that from the control rats (Figure 4A and B). Pyrrolidine dithiocarbamate significantly decreased p65 expression in P1 and P10 groups, and p65 expression in P10 group was even lower than that in P1 group. Interestingly, while p65 expression was higher in long-term
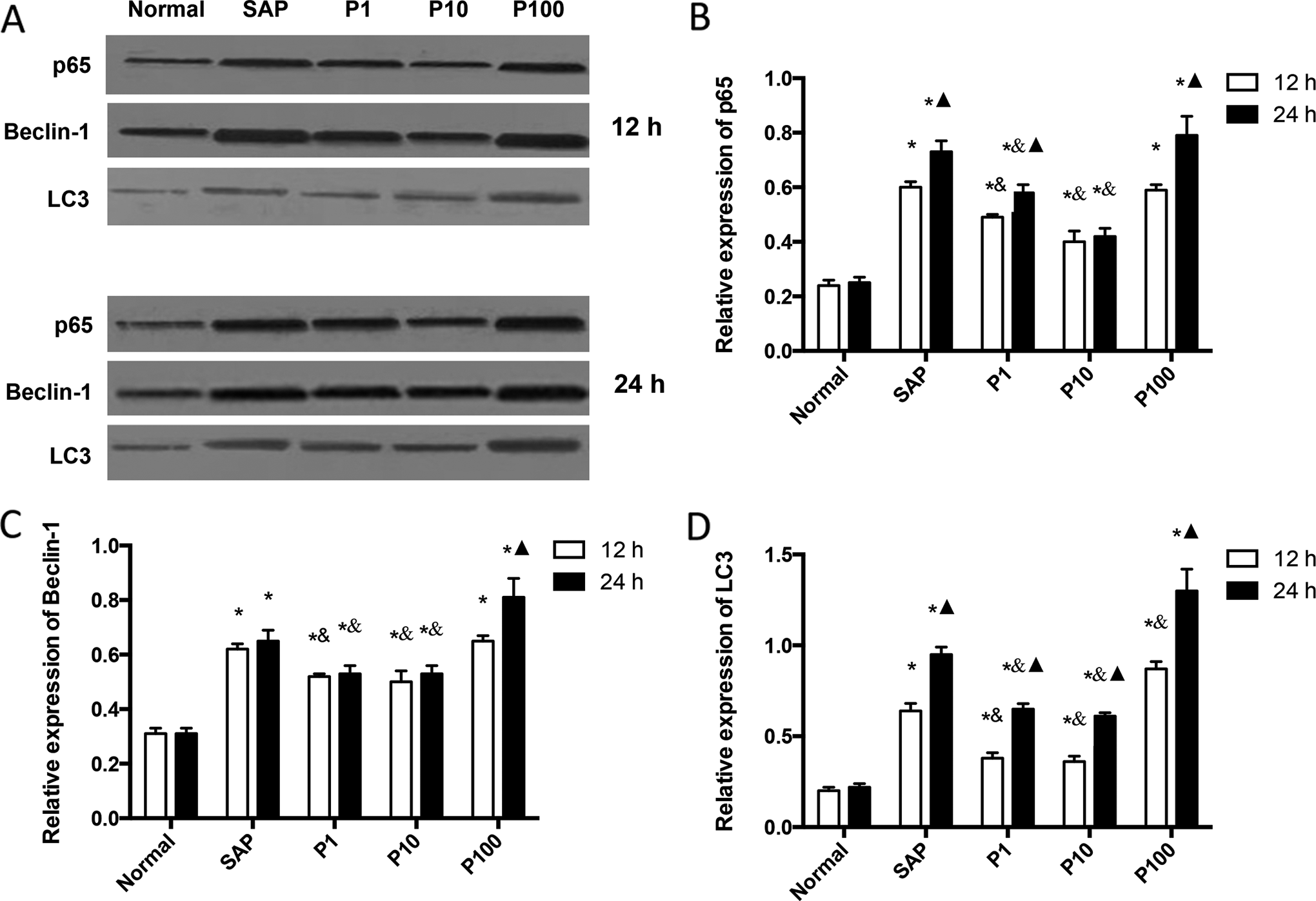
Changes of p65 and autophagy-associated proteins in different groups. A, Representative bands of western blotting. B-D, Statistical analysis of western blotting bands for each protein. *, P < .05 as compared to the normal control group at the same time point; &, P < .05 as compared with the SAP group at the same time point; ▴, P < .05 as compared with 12 hours in the same group. N = 12. SAP indicates severe acute pancreatitis
Pyrrolidine dithiocarbamate Inhibits SAP-Associated Autophagy in Rat Intestines
To study the roles of autophagy in SAP-associated intestinal injury, we investigated the expression of 2 autophagic markers Beclin-1 and LC3.
14
In general, both proteins were induced by
Pyrrolidine dithiocarbamate Enhances Anti-Oxidative Activity in the Intestines of SAP Rats
Oxidative stress has crosstalk with both autophagy and the NF-κB signaling.
15,16
We found that

Oxidative stress was evaluated in the guts of rats. The levels of (A) MDA and (B) SOD were determined in the indicated groups. *, P < .05 as compared to the normal control group at the same time point; &, P < .05 as compared with the SAP group at the same time point; ▴, P < .05 as compared to 12 hours in the same group. N = 12. MDA indicates malondialdehyde; SOD, superoxide dismutase; SAP, severe acute pancreatitis.
Discussion
The close relationship between intestine and pancreas in pancreatitis can be explained by both anatomic and functional reasons. Translocation of bacteria within the intestine is considered a second hit following SAP, leading to multiorgan dysfunction. However, how pancreatitis induces bacterial translocation has not been fully understood. It was well-known that increased permeability of intestinal mucosal barrier can greatly increase the possibility of bacterial translocation. Thus, understanding the mechanism by which SAP causes impairment of intestinal mucosal barrier is important to develop novel strategy to treat SAP.
Many efforts have been put to answer this question and tried to design therapeutic strategy. For example, high-mobility group box 1 (HMGB1)-induced toll-like receptor (TLR) 4 and TLR9 in ileum contributed to the dysfunction of intestinal mucosal barrier, and anti-HMGB1 displayed a significant preventive role in an SAP mouse model. 17 Increased NF-κB and decreased occludin, an intestinal tight junction protein, also contributed to impairment of intestinal barrier function during SAP, and inhibition of NF-κB mitigated intestinal damage and inflammation. 18 High-mobility group box 1 is a classic damage-associated molecular pattern (DAMP) and causes sterile inflammation. 19 Therefore, local inflammation caused by DAMPs and bacteria during SAP is important for the impairment of intestinal mucosal barrier. To this end, PDTC can be reasonably applied to protect SAP rats.
Most existing literatures indicated apoptosis as the main type of death for intestinal mucosal cells during SAP. 20,21 However, few studies have been focused on the autophagy of intestinal mucosal cells in case of SAP. A recent report showed that autophagy was activated in the intestinal epithelial cells. 22 The authors demonstrated that a high level of LC3 decreased the risk of bacterial translocation with a healthier intestinal barrier. Although we found a similar overexpression of LC3 in our rat model, we, on the contrary, showed a harmful rather than protective role of autophagy in intestinal mucosal barrier. Our conclusion was supported by the fact that inactivation of autophagy using PDTC alleviated injury of intestinal mucosal barrier. It is well known that autophagy has physiological roles in the maintenance of intestinal homeostasis by promoting cellular survival. 23 Intriguingly, the effects of autophagy inactivation were highly dependent on the dosage of PDTC. A high dose of PDTC (100 mg/kg) did not alleviate and even worse the impairment of intestinal mucosal barrier. This bidirectional effect of PDTC coincided with the distinctive roles of physiological and pathological autophagy. We thus speculate that a suitable level of autophagy is critical for the homeostasis of intestinal mucosal cells, and an increased level of autophagy in these cells is induced by SAP. When a low or medium dose of PDTC is used, overactivated autophagy can be properly weakened to the physiological level; however, in the presence of high-dose PDTC administration, autophagy may be completely inhibited, resulting in decreased survival of intestinal mucosal cells. Our present study showed that 10 mg/kg PDTC displayed the best performance in inhibiting autophagy and protecting intestinal mucosal integrity. The bidirectional efficacy of PDTC also implicates that the therapeutic window of PDTC, or other autophagy inhibitors, needs to be carefully explored in humans. In addition, the underlying effects and mechanisms of enhanced autophagy in intestinal mucosal cells during SAP are still elusive and need further investigation.
In agreement with previous studies, we demonstrated increased oxidative stress in the guts of SAP rats. For example, Deng et al reported that the NADPH oxidase inhibitor apocynin attenuated intestinal barrier dysfunction using a sodium taurocholate-induced SAP rat model. 24 Oxidative stress including reactive oxygen species and reactive nitrogen species is considered the converging point of autophagy-inducing stimuli. 15 However, an improperly high level of oxidative stress harms intestinal mucosal barrier since anti-oxidative materials can alleviate the impairment of gut barrier. 24,25 Since NF-κB induces oxidative stress generation, 16 the NF-κB inhibitor PDTC is able to function as an anti-oxidative stress agent. 26,27 In fact, PDTC was reported to improve the survival of taurocholate-induced SAP rats. 28 Additionally, recently some investigators showed preliminary results of improved intestinal mucosal barrier with treatment of 40 mg/kg PDTC. 18 We used pretreatment of PDTC ranging from 1 to 100 mg/kg and found a bidirectional effect of PDTC. More importantly, we noticed simultaneous alterations of NF-κB expression and MDA and SOD levels. Our results suggested that inhibition of NF-κB by a suitable dose of PDTC was probable to protect SAP-associated intestinal injury by suppressing autophagy and oxidative stress. Nevertheless, gut is not the only organ that impaired during SAP. Interestingly, PDTC also reduced SAP-associated lung injury by inhibition of NF-κB. 29 However, whether autophagy plays a role in such process is unclear.
Conclusions
We used an
Footnotes
Author Contribution
Huan Yang and ShuCan Ma contributed equally.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
