Abstract
The composite fibers with conductive and magnetic properties were prepared from the spinning solution consisting of poly(vinyl alcohol), poly(3,4-ethylenedioxythiophene)-poly(sodium-p-styrenesulfonate), and Co-doped Fe3O4 magnetic particles (Co-Fe3O4) via a conventional wet spinning process. The morphology, microstructure, elemental composition, crystallinity, thermal stability, mechanical properties, conductivity, and magnetic properties of the composite fibers were studied. The scanning electron microscopy images showed that the diameters of the composite fibers were in the range of 30–50 μm with a rough surface and reniform cross-section. The energy-dispersive spectra results indicated that the poly(3,4-ethylenedioxythiophene)-poly(sodium-p-styrenesulfonate) particles were mostly distributed on the surface of the composite fiber, while the Co-Fe3O4 magnetic particles mainly existed inside the fiber. The composite fibers were amorphous, but contained a certain crystalline phase, and with the increase of the poly(3,4-ethylenedioxythiophene)-poly(sodium-p-styrenesulfonate) content, the crystallinity of the composite fiber decreased. The conductivity and saturation magnetization of the composite fiber with a Co-Fe3O4 content of 2.68 wt% and poly(3,4-ethylenedioxythiophene)-poly(sodium-p-styrenesulfonate) of 8.6 wt% reached 0.2 S/cm and 1.9 A m2/kg, respectively. Besides, the composite fibers displayed improved thermal stability and good mechanical properties, which are applicable in electromagnetic protection fields.
Keywords
Introduction
Nowadays, the composite polymers with conductive and magnetic properties have received great attention because of their unique properties and technological applications in electrical, optical, and magnetic materials and devices. 1 Especially, polymer fibers with conductive and magnetic properties are of special interest due to their potential applications in electromagnetic interference (EMI) shielding and as microwave-absorbing agents,2,3 healthcare cloth, magnetic filters, and various other applications because of their high forming abilities. 4 Recently, a few articles on the electromagnetic functional fibers have been published. For instance, Qiang et al. 5 reported that in nitrate and dimethylamine borane solution Fe3O4 nanoparticles were plated on the carbon fibers, and the resulting composite fibers had excellent microwave absorption properties. Yang et al. 6 have prepared magnetic carbon composite fibers by means of dispersing magnetite (Fe3O4) nanoparticles in the spinning solution and by an electrospinning process. Since the magnetic properties by single Fe3O4 cannot satisfy the application requirements, new materials with higher magnetic intensity and multi-functions such as magneto-electric, magneto-optic, and magneto-caloric have become the focus of researchers.7–12
Among conducting polymers, poly(3,4-ethylenedio-xythiophene)-poly(sodium-p-styrenesulfonate) (PEDOT-PSS) stands out for its high conductivity, excellent stability in air, and promising applications such as materials in solar cells, organic light-emitting diodes (OLEDs), electrochromic devices, and transparent electrodes.13–16 In particular, the room-temperature conductivity of PEDOT-PSS prepared by a chemical vapor deposition method can reach 1000 S/cm. 17 By combining PEDOT-PSS with magnetic component, the electromagnetic compounds can be obtained; besides, the adjustment of electric and magnetic properties can be realized by adjusting the content and structure of the components; 18 as a result, the aim “thin, light, broad and strong” for a superior electromagnetic material will be realized. 19 Compared with pure Fe3O4, the Co-doped Fe3O4 nanoparticles (Co-Fe3O4) have the specialties of strong magnetic affinity and high magnetic intensity, which have displaced the application of pure Fe3O4 in many fields.
In this work, a common polymer: poly(vinyl alcohol) (PVA) was used as the matrix, and the conductive source of PEDOT-PSS solution and the magnetic source of Co-Fe3O4 nanoparticles were blended with the PVA solution, respectively. After a traditional wet spinning process of PVA, the composite fibers with high conductivity and certain magnetic properties were obtained. It was found that the resulting composite fibers with the Co-Fe3O4 content of 2.68 wt% and the PEDOT-PSS content of 8.6 wt% exhibited high conductivity and ferromagnetic properties, excellent thermal stability, and good mechanical properties, which can be applicable in electromagnetic protection fields.
Experimental
Materials
3,4-ethylenedioxythiophene (EDOT; ⩾99.5%) was purchased from Suzhou Yacoo Chemical Reagent Corporation. Poly(sodium-p-styrenesulfonate) (PSS; chemically pure) was provided by Shanghai Jiachen Chemical Co., Ltd. Sodium persulfate (Na2S2O8; analytical reagent) was provided by Tianjin Damao Chemical Reagent Factory. Ferric trichloride (FeCl3; analytical reagent) was purchased from Xilong Chemical Co., Ltd. Ferrous chloride tetra-hydrate (FeCl2·4H2O; analytical reagent) was purchased from Tianjin JinKe Fine Chemical Industry Research Institute. Cobaltous chloride hexahydrate (CoCl2·6H2O; analytical reagent) was purchased from Beijing Chemical Reagents Co. Sodium hydroxide (NaOH; analytical reagent) was purchased from Beijing Huateng Chemical Industry Ltd. Anhydrous sodium sulfate (Na2SO4; ⩾99%) was provided by Nanfeng Group Co., Ltd. PVA (degree of polymerization = 1700, degree of alcoholysis = 99%) was purchased from Shijiazhuang Chemical Fiber Co., Ltd. Nitrogen (N2) was obtained from Beijing Praxair Application Gas Co., Ltd. Dodecyl benzene sulfonic acid sodium salt (DBSAS; analytical reagent) was purchased from Beijing Chemical Reagents Co. All reagents were used as received without further purification, and the water used in the synthesis was distilled water.
Preparation of PEDOT-PSS
In this work, the PEDOT-PSS dispersed solution was prepared by chemical oxidative polymerization. The specific processes are as follows: first, 0.61 g EDOT monomer was added to 40 mL deionized water containing 0.88 g PSS under magnetic stirring. Then 1.28 g Na2S2O8 was added into the mixture and sonicated for 15 min. Next, 2.16 mL FeCl3 solution (1 mol/L) was added drop-wise to the above mixture solution. Finally, the mixture was stirred for 24 h at room temperature. The obtained mixture solution was called PEDOT-PSS and used in the following experiment.
Preparation of magnetic nanoparticles
Co-doped Fe3O4 magnetite nanoparticles were prepared by a chemical co-precipitation process. In this process, all the procedures were completed under nitrogen protection to avoid oxidation. First, 0.56 g FeCl3, 0.34 g FeCl2·4H2O, and 0.24 g CoCl2·6H2O were dissolved in 100 mL deionized water containing 2 g DBSAS under stirring for 30 min. Then, 1.5 mol/L sodium hydroxide solution was added drop-wise to the mixture until the pH of the system is equal to 12 with mechanical stirring at 80°C. After 1 h, the resulting black precipitate was obtained and then washed with distilled water several times until the pH reached 7. The resulting Co-doped Fe3O4 magnetic nanoparticles were called Co-Fe3O4.
Preparation of the spinning solution
The spinning solution was prepared by adding cotton-like PVA to the deionized water with a polymer loading of 17 wt%. First of all, the 1.08 g (or 1.72 g) PEDOT-PSS and 0.58 g Co-Fe3O4 magnetic particles were added into deionized water, respectively, and were mixed by mechanical stirring. Afterward, 20 g PVA was added to the system, respectively, and then swelled for 8 h. The system was then kept at 98°C under mechanical agitation for 3 h to make the PVA dissolved. Finally, the resulting spinning dopes were placed in the vacuum drying oven static at 98°C for 24 h to degas. The dopes were obtained in this way and were called the PEDOT-PSS/Co-Fe3O4/PVA mixture. In the resulting PEDOT-PSS/Co-Fe3O4/PVA mixture, the contents of PEDOT-PSS were 5.4 and 8.6 wt%, respectively, and the content of Co-Fe3O4 magnetic particles was 2.68 wt%. We tried using higher contents of PEDOT-PSS and Co-Fe3O4, but the continuous fibers could not be obtained. When the content of PEDOT-PSS was lower than 5.4 wt%, the resulting fiber was unconductive. The pure PVA spinning solutions were prepared in a parallel procedure without adding PEDOT-PSS and Co-Fe3O4.
Wet spinning
A self-made wet spinning apparatus as shown in Figure 1 was used in this experiment. The spinning solutions were maintained at 98°C and pulled into a coagulation bath through a multi-hole spinneret (18 holes, diameter = 0.2 mm) by nitrogen gas which was used to provide the required pressure. A solution of Na2SO4 with a density of 1.31 g/cm3 was used as a coagulation solution and the temperature was controlled at 45°C. A 90°C Na2SO4 solution with a density of 1.29 g/cm3 was used for the draft bath. The temperature of the coagulation bath and draft bath was controlled by a potential transformer. The filament from the spinneret was drawn through a coagulating bath and then drafted on the draft bath. The fibers were preheated on a roller (80°C) and then placed in a 160°C hot box with continuous drawing and winding. All the samples used in this study had the total draw ratio of 4.
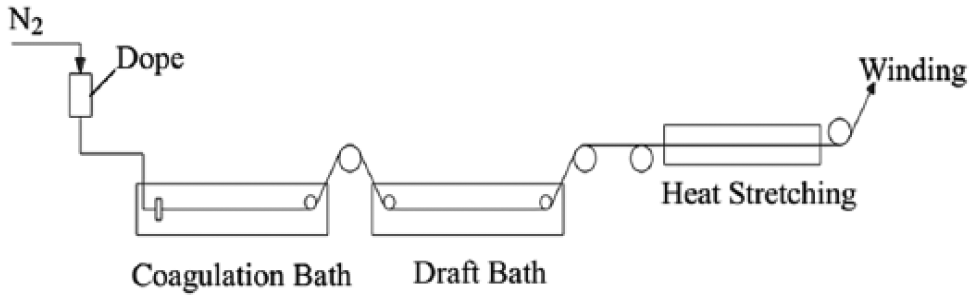
Schematic diagram of the wet spinning process.
Characterization
The chemical structure of the fibers was analyzed by a Nexus 670 Fourier transform infrared (FTIR) spectrometer and the scanning rate is 4 cm−1/s in the range of 4000–400 cm−1. The morphology was studied with a scanning electron microscope (SEM) (JSM-6360LV). SEM/EDS spectra of composite fibers were obtained by an EX-54175JMH energy-dispersive spectrometer (EDS) under liquid nitrogen atmosphere, which adhered to the SEM system. X-ray powder diffraction (XRD) patterns were recorded using Cu Kα (λ = 1.5406Å) radiation at 40 kV and 50 mA with a Rigaku wide-angle goniometer, and the scanning range is 6°–80°. The electrical conductivities of the composite fibers were measured by a four-probe conductivity meter (Keithley 6221+2182A) at room temperature. The diameters of the fibers were obtained via SEM images. The conductivities were calculated by the following formula
where δ is the conductivity (S/cm), I is the constant current through the two outer electrodes, V is the variational potential through the two inner electrodes, L is the distance of the two inner electrodes, and S is the fiber cross-sectional area.
The thermostability was measured by an SII TG 6300 instrument (Japan Seiko Co., Ltd.), operating at a heating rate of 10°C/min under a nitrogen atmosphere from 20°C to 800°C. The tensile strength measurement results of fibers were obtained by a YG004N electronic single fiber tensile strength tester with a constant extension rate method, a jaw gap of 10 mm, and an extension rate of 6 mm/min, each sample was tested 30 times under the same condition, and the results were their average values. Physical property measurement system-vibrating sample magnetometer (PPMS-VSM; PPMS-9T) was used to measure the magnetic property in an applied field of ±10 kOe at room temperature.
Results and discussion
Morphology
The SEM images of pure PVA and PEDOT-PSS/Co-Fe3O4/PVA composite fibers are displayed in Figure 2. From Figure 2, it can be seen that the diameters of all the fibers are in the range of 30–50 μm, and the surface of pure PVA is smoother (Figure 2(a)) than that of the composite fibers (Figure 2(b) and (c)). As the content of PEDOT-PSS increased, the surfaces of the composite conductive fibers became rougher and with more micro-fibers.

SEM images of pure PVA and PEDOT-PSS/Co-Fe3O4/PVA composite fibers: (a) surface image of pure PVA fiber, (b) surface image of the fiber with 2.68 wt% Co-Fe3O4 + 5.4 wt% PEDOT-PSS, (c) surface image of the fiber with 2.68 wt% Co-Fe3O4 + 8.6 wt% PEDOT-PSS, (d) cross-sectional image of pure PVA fiber, (e) cross-sectional image of fiber B, and (f) cross-sectional image of fiber C.
EDS spectral analysis
The cross-section of the fibers is kidney like, and there are many white spots evenly distributed on the surface and cross-section of the composite fibers. It is supposed that these white spots may be the PEDOT-PSS or Co-Fe3O4 particles. To prove this suspection, EDS spectra were tested.
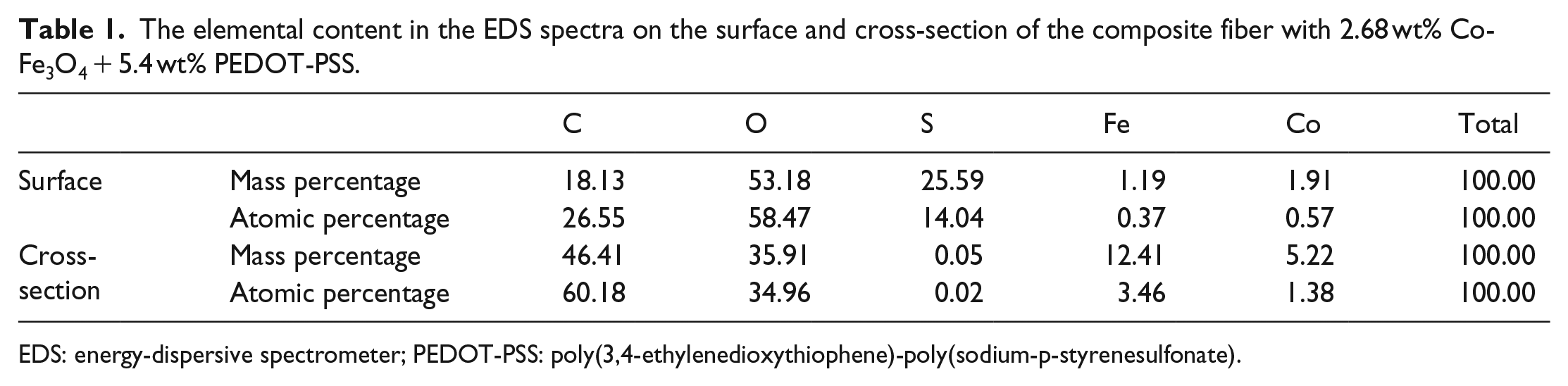
To determine the component of the white spots in the cross-section and surface of the composite fiber, EDS spectra (as shown in Figure 3) were used for further analyzing the elemental composition on the surface and cross-section of the composite fiber with 2.68 wt% Co-Fe3O4 + 5.4 wt% PEDOT-PSS. Table 1 summarizes the elemental content of the EDS spectra. As shown in Figure 3 and Table 1, the bright dots on the surface of composite fiber mainly contain C, O, S, and only a few Fe and Co (0.37% and 0.57% atom amount). However, on the cross-section of the composite fiber, the white dots are mostly composed by C, O, Fe, Co, and a trace amount of S (0.02% atom amount). This indicates that PEDOT-PSS particles largely distribute on the surface of the composite fiber, while the Co-Fe3O4 magnetic particles mainly exist inside the fiber.

SEM images (a, b) and EDS spectra (c, d) of the composite fiber with 2.68 wt% Co-Fe3O4 + 5.4 wt% PEDOT-PSS: (a) surface, (b) cross-section, and (c, d) EDS spectra of the elements found in the square areas in (a) (005) and (b) (004), respectively.
The elemental content in the EDS spectra on the surface and cross-section of the composite fiber with 2.68 wt% Co-Fe3O4 + 5.4 wt% PEDOT-PSS.
EDS: energy-dispersive spectrometer; PEDOT-PSS: poly(3,4-ethylenedioxythiophene)-poly(sodium-p-styrenesulfonate).
FTIR analysis
Figure 4(A) shows the FTIR spectra of the pure PVA fiber and the composite fibers. In all three curves, the distinct broad absorption bands at 3000–3600 cm−1 corresponding to the PVA structural hydroxyl groups are observed; however, the central point of the pure PVA fiber appears at 3405 cm−1 contrasting with that of the composite fiber at 3309 cm−1. The stretching vibration peak of –OH centered at 3309 cm−1 for the composite fibers not only is broad, but also shifts to a lower wavenumber, indicating that a hydrogen bonding interaction existed among PVA, PEDOT-PSS, and the magnetic particles, compared with the pure PVA fiber. 20 The sharp bands at 2906 cm−1 correspond to –CH3, –CH2, and –CH groups. 21 Comparing the spectra of the composite fibers with that of the pure PVA, there appear some new peaks. For example, the C–O–C symmetric stretching and antisymmetric stretching at 1137 and 1087 cm−1, the C–S stretching vibration and bending deformation at 919 cm−1, and the C=C stretching of quinoid structure of the thiophene ring at 1322 cm−1 are all observed, 22 which are all the characteristic peaks of PEDOT, indicating that PEDOT existed in the PVA fibers. The characteristic peaks at 1551, 710, and 511 cm−1 appear in the pure PVA fiber but disappear in the composite fibers, suggesting that there is an interaction among PVA, PEDOT-PSS, and the magnetic particles.

FTIR spectra (A) and Raman spectra (B) of the pure PVA fiber (a) and composite fibers with 2.68 wt% Co-Fe3O4 + 5.4 wt% PEDOT-PSS (b) and 2.68 wt% Co-Fe3O4 + 8.6 wt% PEDOT-PSS (c).
Furthermore, the additional peaks appearing in the composite fibers at 475 and 618 cm−1 correspond to M–O (M = Fe, Co) stretching vibration of magnetic nanoparticles, 23 which is absent in the pure PVA fiber. These results indicate that PEDOT-PSS and magnetic particles were successfully compounded with PVA fibers.
Raman spectroscopy
Figure 4(B) shows the Raman spectra of the pure PVA fiber and the composite fibers. In the Raman spectra, the characteristic absorption peak at 1444 cm−1 corresponding to the in-plane deformation vibration of C–H in the group –CH2 of PVA chains shifted to 1412 cm−1 in the composite fibers, indicating that the addition of PEDOT-PSS and Co-Fe3O4 affected the structure of PVA fiber. The absorption peaks of stretching vibration and bending vibration of the C–C group appear at 1364 and 1088 cm−1, respectively, and the peak at 848 cm−1 is the stretching vibration peak of C–C–O in the PVA main chains. While the characteristic absorption peak at 569 cm−1 is the stretching vibration of C–S on the five-member thiophene ring of PEDOT chains, and the wavenumber 984 cm−1 may be the ring breathing vibration aroused by the hydrogen bonding interaction among PVA skeleton molecular chains.24–26 Comparing the spectra of the composite fibers with those of the pure PVA fibers, some new peaks at 691 and 435 cm−1 appear, which are the Raman spectra of metallic oxide M–O (M = Fe, Co). 27
XRD patterns
The structural changes of the fibers induced by the presence of PEDOT-PSS and Co-Fe3O4 are further attested using wide-angle X-ray diffraction (XRD). The XRD patterns obtained from the pure PVA fibers and composite fibers are shown in Figure 5(A). It is known that the diffraction peaks of the amorphous polymer are broad; however, the polymer containing a crystalline phase will result in sharp X-ray diffraction peaks with high intensity.
28
From Figure 5(A), it can be seen that the PVA fiber shows a monoclinic crystal structure with the crystalline peaks appearing at 2θ = 11.3°, 16.1°, 20.3°, 22.7°, 32.5°, and 34.7° corresponding to the crystal indices of (100), (001), (101,

XRD patterns (A) and thermogravimetric curves (B) of the pure PVA fiber (a) and composite fibers with 2.68 wt% Co-Fe3O4 + 5.4 wt% PEDOT-PSS (b) and 2.68 wt% Co-Fe3O4 + 8.6 wt% PEDOT-PSS (c).
Besides, it can be observed that the addition of PEDOT-PSS and Co-Fe3O4 particles also affects the crystallization of PVA fiber. With the increasing content of PEDOT-PSS, the diffraction peaks become weak and broadened, indicating a change from the crystalline phase to the amorphous phase, gradually. The diffraction peak at 2θ = 20.3° is the (101) diffraction of PVA crystal, which is attributed to the intermolecular interference between PVA chains in the direction of the intermolecular hydrogen bonding. 31 After adding the PEDOT-PSS and Co-Fe3O4 particles, the intensity of the (101) diffraction peak decreases, and it may be due to the formation of hydrogen bonds among PVA, PEDOT-PSS, and magnetic particles, which will decrease the number of hydrogen bonds between PVA chains and result in a smaller crystallite size of PVA. However, the diffraction peaks of the magnetic particles are hardly observed, which may result from the little content of magnetic particles (only 2.68 wt%).
Thermal performance analysis
The thermal stability of the pure PVA fibers and the composite fibers were studied by TG, which is an important method to detect the degradation behavior of the fibers. As shown in Figure 5(B), the thermograms are similar to each other for both the pure PVA fiber and the PEDOT-PSS/Co-Fe3O4/PVA composite fibers. The first weight loss of about 5–6 wt% occurring at 20°C–100°C was attributed to the elimination of absorbed moisture, and the second ones started at 220°C and 260°C representing the degradation of the PVA molecular chains. 32 The weight losses above 320°C were due to further decomposition of the polymer backbone, while for the composite fibers it contained the decomposition of the PVA backbone and the PEDOT-PSS conductive particles. The final amount of residual carbon ranged from 13.7% to 26.2%. Moreover, it can be seen that the decomposition point (260°C) of the composite fibers is higher than that of the pure PVA fiber (220°C), which may be caused by the hydrogen bond between PVA chains and PEDOT-PSS and Co-Fe3O4 (proved by FTIR spectra). Furthermore, their char residuals are also more than those of the pure PVA indicating an improved thermal stability. In brief, the PEDOT-PSS/Co-Fe3O4/PVA composite fibers have higher thermal stability than the pure PVA fiber.
Mechanical performance
The drawing strength is the most important index of fiber’s spinnability, and it has a close relationship with the process performance. 33 Through testing the stress–strain curve of the pure PVA fibers and the PEDOT-PSS/Co-Fe3O4/PVA composite fibers, we found that, for all of the samples, there existed yield points, which belonged to strong and tough materials. 34 The tensile test results are tabulated in Table 2. As shown in Table 2, the breaking strengths of the composite fibers are all lower than that of the pure PVA fiber and decrease with the increase of the PEDOT-PSS content. However, the elongation at break follows a trend of first decreasing and then increasing, and the initial modulus first increases and then decreases, with the increase of the PEDOT-PSS content. For the pure PVA fiber, the breaking strength, elongation at break, and initial modulus are 5.57 cN/dtex, 20.66%, and 159.32 cN/dtex, respectively. As to the composite fiber with the PEDOT-PSS content of 5.4 wt%, the breaking strength, elongation at break, and initial modulus are 5.33 cN/dtex, 12.55%, and 262.05 cN/dtex, respectively. The mechanical properties of the two composite fibers are better than those of pure PEDOT/PSS fiber, 35 which are benefited from the process of high-ratio drawing and high-temperature heat setting. The 2.68 wt% Co-Fe3O4 + 8.6 wt% PEDOT-PSS sample has a lower initial modulus of 59.05 cN/dtex, which is attributed to the higher PEDOT-PSS content. If the PEDOT-PSS content continues to increase, the mechanical properties of the composite will quickly decrease, so that no continuous fibers can be formed.
Mechanical performance of the pure PVA fiber and the PEDOT-PSS/Co-Fe3O4/PVA composite fibers.

PVA: poly(vinyl alcohol); PEDOT-PSS: poly(3,4-ethylenedioxythiophene)-poly(sodium-p-styrenesulfonate).
Conductivity and magnetic properties
The conductivity of the composite fibers was measured by the four-point method at room temperature and calculated according to formula (1). It was found that the electrical properties of the composite fiber were strongly affected by the PEDOT-PSS content. When the PEDOT-PSS content was 5.4 wt%, the conductivity was 0.04 S/cm, but for a PEDOT-PSS content of 8.6 wt% the conductivity reached 0.2 S/cm. Compared with the pure PEDOT-PSS fiber prepared by Okuzaki and Ishihara, 35 the conductivity of which was only 0.1 S/cm, the higher conductivity of PEDOT-PSS/Co-Fe3O4/PVA fibers might result from the formation of the consecutive phase of PEDOT-PSS in the solid fibers. Although its content was low, it is an effective conductive path of the electron conduction.
Figure 6 shows the room-temperature magnetic hysteresis loops of the PEDOT-PSS/Co-Fe3O4/PVA composite fibers, and the inset shows the magnetic hysteresis loop of pure Co-Fe3O4. It can be seen that the saturation magnetization (Ms) of pure Co-Fe3O4 is 64.54 A m2/kg, and for the fibers with the PEDOT-PSS contents of 5.4 and 8.6 wt%, the Ms values are 1.91 and 1.71 A m2/kg, respectively. It is known that the magnetic property of the fiber mainly comes from the added magnetic particles, so the maximum Ms values of composite fibers are only slightly different. The coercivity (Hc) strengthens with the increasing loading of PEDOT-PSS, which are 1.6 × 104 A/m (5.4 wt%) and 4.1 × 104 A/m (8.6 wt%), respectively. This means that the magnetic particles after being dispersed in the fibers are hard magnetic with the increasing content of PEDOT-PSS particles. For pure magnetic particles, it has small single-domain nanoparticles; after adding the non-magnetic PEDOT-PSS particles, the single-domain nanoparticles of magnetic particles are enlarged, resulting in the decrease of dipolar interaction between inter-particles, therefore causing an increased coercivity.36–38 The remnant magnetization (Mr) of the composite fibers is 0.39 and 0.54 A m2/kg for the PEDOT-PSS contents of 5.4 and 8.6 wt%, respectively.

Magnetic hysteresis loops of the composite fibers at room temperature: (a) 2.68 wt% Co-Fe3O4 + 5.4 wt% PEDOT-PSS and (b) 2.68 wt% Co-Fe3O4 + 8.6 wt% PEDOT-PSS. The inset shows the magnetic hysteresis loop of pure Co-Fe3O4.
Conclusions
Multifunctional composite fibers with conductive and magnetic properties were prepared by blends of PEDOT-PSS, Co-Fe3O4 magnetic particles, and common PVA via a conventional wet spinning process. Compared with the pure PVA fiber, the resulting composite fibers present lower crystallinity, higher thermal stability, and slightly decreased mechanical properties. For a composite fiber of 2.68 wt% Co-Fe3O4 + 8.6 wt% PEDOT-PSS, it has the conductivity of 0.2 S/cm and an Ms of 1.9 A m2/kg. The composite fibers showing high conductivity and good mechanical properties can be used in many fields requiring electromagnetic protection.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (51873003), Scientific and Technology Key Project of Beijing Educational Committee (KZ201910012015), The Beijing Great Wall Scholars Incubator Program (CTT&TCD20180321), Collaborative Innovation Project in Chaoyang District, Beijing (CYXC1718), and Special funds for the construction of high level teachers of Beijing Institute of Fashion Technology (BIFTXZ201802).

