Abstract
Sodium alginate/cellulose nanocrystal fibers were prepared using a wet spinning method to enhance the mechanical strength of sodium alginate fibers. Cellulose nanocrystals were prepared by sulfuric acid hydrolysis method. The particle diameter size was measured, and the morphology of cellulose nanocrystals was characterized by transmission electron microscopy and scanning electron microscopy. The structure and mechanical properties of sodium alginate/cellulose nanocrystal fibers were characterized by scanning electron microscopy, transmission electron microscopy, X-ray diffraction, and mechanical strength testing. The incorporation of cellulose nanocrystals significantly improved the strength of alginate fibers because of the uniform distribution of cellulose nanocrystals in the alginate matrix. The tensile strength and elongation at break of the alginate fibers increased from 1.54 to 2.05 cN/dtex and from 8.29% to 15.05% with increasing cellulose nanocrystals content from 0 to 2 wt%, respectively.
Introduction
Sodium alginate, which is mainly composed of (1,4)-linked β-D-mannuronic acid units (M) and α-L-gluronic acid units (G) that distribute randomly in different ratios to form M segments, G segments, and MG segments within the macromolecular chains,1–3 is a polysaccharide extracted from natural brown algae. It can be used in biomedical fields and is approved by the Food and Drug Administration (FDA).4–6 Alginate fibers are bio-based fibers produced by wet spinning of sodium alginate aqueous solution into calcium chloride coagulating bath and a special “egg-box” structure could be formed between G segments and Ca2+, and thus, the connection between alginate macromolecules become closer, resulting in the gelation of sodium alginate.7,8 Alginate fibers have been found in many applications in the biomedical fields, such as dressings to treat crush injury because of their good bio-compatibility, bio-degradability, and hygroscopicity.9,10
However, the low mechanical strength greatly restricted the application of alginate fibers. Nanofillers such as carbon black, silica, carbon nanotube, grapheme, and nanosized cellulose have been used to improve the mechanical properties of polymer nanocomposites.11,12 Compared to the pure polymers, the tensile strength and tensile modulus of polymer nanocomposites have been significantly enhanced after nanofillers were added. Among nanofillers, cellulose nanocrystals (CNs) are attractive as they are sustainable and exhibit good mechanical properties. Over the last decade, different polymer/CN composites have been investigated.13–16 Based on the Raman spectroscopy analysis, it was determined that CNs were unoriented in the nanocomposite films and highly aligned in the nanocomposite fibers. Nanocomposite fibers with highly aligned CNs showed higher interfacial shear strength than nanocomposite films with isotropic CNs.17,18 For polymer/CN composite fibers, CNs have been used for reinforcing poly(methyl methacrylate), 19 poly(vinyl alcohol), 20 polyethylene oxide, 21 poly(ε-caprolactone), 22 poly(acrylic acid) (PAA), 23 and poly(lactic acid)24,25 via electrospinning and for reinforcing silk fibers, 26 polyacrylonitrile, 27 and alginate 28 via wet spinning method. The addition of CNs in the polymer fiber has been observed to improve the mechanical properties for many systems. However, one drawback of nanocomposites is that with the increasing concentration of CNs in the polymers, it invariably leads to lower strain to failure, as compared to the control polymer.14,16,19 In this study, the co-solvent approach was used to improve the tensile strength and elongation at break of the alginate fibers by wet spinning. Structure and properties of alginate fibers with various CNs content were studied.
Experimental analysis
Materials
Sodium alginate (food grade, Mn = 3.57 × 105, relative molecular mass distribution value: 1.392, and M/G value: 0.32) was supplied by Haizhilin Biotechnology Develop-ment Co., Ltd. (Qingdao, Shandong, China) Calcium chloride (analytical reagent, Tianjian BASF Chemical Reagent Company, Tianjin, China), microcrystalline cellulose (MCC; analytical reagent, Tianjin Guangfu Fine Chemistry Institute, Tianjin, China), and sulfuric acid (98%, Yantai Sanhe Chemical Reagent Company, Yantai, Shandong, China) were purchased and used as obtained without further purification.
Preparation of CNs
CNs were prepared by sulfuric acid hydrolysis according to the previously reported protocol. 29 The CN suspension was obtained by hydrolyzing MCC samples with 65 wt% H2SO4 at 40°C for 30 min under vigorous stirring. After acid hydrolysis, the suspension was immediately diluted with distilled water, followed by centrifugation at 12,000 r/min for 10 min, which was repeated five times. The suspension was further dialyzed in distilled water at room temperature until neutrality. The CNs dispersion was completed by ultrasonic treatment, and finally, the released CN powder was obtained by freeze drying.
Preparation of SA/CN fibers
The dope solution for preparing the pure alginate fibers was 5 wt% sodium alginate aqueous solution, whereas the spinning dope suspension for preparing of the alginate/CN composite fibers was prepared by mixing homogenized CN suspensions (diluted from the as-prepared CN suspension by distilled water, followed by sonication for 20 min) with the as-prepared alginate solution. The weight ratio of CNs to sodium alginate was varied in the range of 0.5–16 wt%. Both the pure alginate solution and the alginate/CN suspensions were left at room temperature for degassing before being extruded through a spinneret into the coagulation bath containing 5 wt% CaCl2. The obtained fibers were drawn at a ratio of ~1.2 between two sets of rollers. Finally, they were collected on bobbins and washed with distilled water and then soaked in alcohol for 24 h, followed by drying in the air.
Characterization
Transmission electron microscopy analysis of CNs
Morphology of CN particles was performed on a high-resolution transmission electron microscope (JEM-2010, JEOL, Tokyo, Japan). The objective lens raster was added to enhance the contrast, and the images were recorded using a charge-coupled device (CCD) digital camera. The sample was prepared by dispersing 10 mg of CNs in 5 mL of distilled water by sonication for 30 min. A droplet of the sample dispersion was deposited on a carbon film. A thin layer was suspended over the holes of the grid. The specimen was finally dried in air at ambient temperature, and the transmission electron microscopy (TEM) images were taken at an accelerating voltage of 200 kV.
Particle diameter distribution of CNs
CNs (1 g) were dispersed into 30 mL of distilled water and then treated by ultrasonic treatment for 30 min with the power level of 120 W. The particle diameter distribution of CNs at 25°C was measured by a laser particle size analyzer (ZEN3600, Malvern, England).
Scanning electron microscopy analysis of alginate/CN fibers
The micromorphological structure of fibers was studied with Phenom-World BV, Holland, scanning electron microscopy (SEM). Prior to examination, the fiber samples were fractured in liquid nitrogen and the section of the fibers was coated with gold, and then, they were observed and photographed.
X-ray diffraction analysis of alginate/CN fibers
X-ray diffraction (XRD) of the samples was measured on a X-ray diffractometer (DMAXRB-II, Japan) using Cu-Kα radiation. The XRD patterns were recorded with a step size 0.05° and scanning speed of 1°/min for 2θ = 5°–55°.
Mechanical properties of alginate/CN fibers
Tensile strength of the samples was measured on a fiber electron tensile tester (YG06, Laizhou Electronic Machine Co., Ltd., China). The samples were kept at relative air humidity of 60% at 20°C for 24 h for the mechanical test. The gauge length was 100 mm, and the crosshead speed was 100 mm/min. A total of 30 individual samples were tested from each group.
Results and discussion
Particle size and morphology of CNs
Figure 1 shows the particle size distribution of CNs. It was apparent that the particle size of CNs was between 10 and 1000 nm. The average particle size of CNs was calculated as 116 nm, and the dispersion degree index (PDI) was 0.428, indicating that the molecular weight of CNs is evenly distributed. 30 The morphologies of CNs were probed using SEM and TEM. The representative images of CNs are presented in Figure 2. CNs exist as irregular circular nanoparticles and agglomerates because of their high specific surface area.
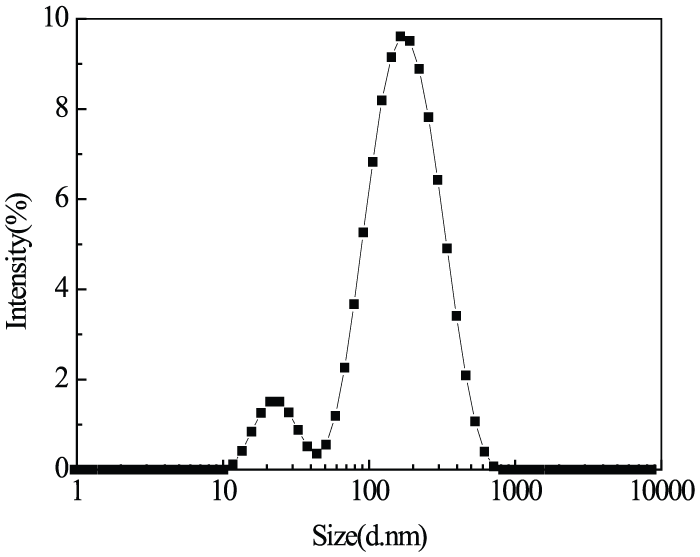
Average particle size distribution of CN powder.

SEM and TEM images of CNs.
Rheological properties of the spinning solutions
Effect of shear rate on viscosity
The rheological properties of the spinning solution have great influence on processability, spinning process, and quality index of finished fibers.10,31,32 Figure 3 shows the effect of shear rates on viscosity of blend solutions. It was indicated that the viscosity values of the four samples all decrease with increasing shear rate. The viscosity values of the SA/CN blend solutions tend to be constant at low shear rates (near zero), which is the character of Newtonian fluids. The viscosity values begin to decrease with increasing shear rates, exhibiting shear thinning behavior. There are many entanglement points among macromolecule matrix, including the intramolecular hydrogen bonds in alginate matrix and the intermolecular hydrogen bonds between alginate and CNs, which have transient properties, that are dissembled and reconstructed continually and then reach dynamic equilibrium under a certain condition. 33 Some entanglement points were dissembled with increasing shear rate, leading to the reduction of the viscosity. In addition, the shear stress inside the entanglement points could not relax in time with the increase of shear rate, resulting in decrease in the transferred momentum capability of macromolecules between the flowing layers, and thus, the traction forces between the flowing layers were reduced, which manifested in the decrease of viscosity.

The viscosity of SA/CN solutions at different shear rates.
Effect of temperature on viscosity
Figure 4 displays a decreasing trend of SA/CN blend spinning solution viscosity with an increasing temperature at the same shear rate. Based on piecewise transition mechanism, the flowing of macromolecules is mainly because of the displacement of the molecular chains’ gravity center along with flow direction and the glide over each other between molecular chains. High temperature provides sufficient energy for the glide, and thus, the mobility of molecular chains is strengthened and the friction inside molecules with each other is reduced, leading to the decrease of viscosity. Therefore, the flowing properties of blend solutions can be improved by raising the temperature. However, high temperature causes energy waste and the degradation of macromolecules. So, the temperature of solutions should be strictly controlled according to the different requirements.
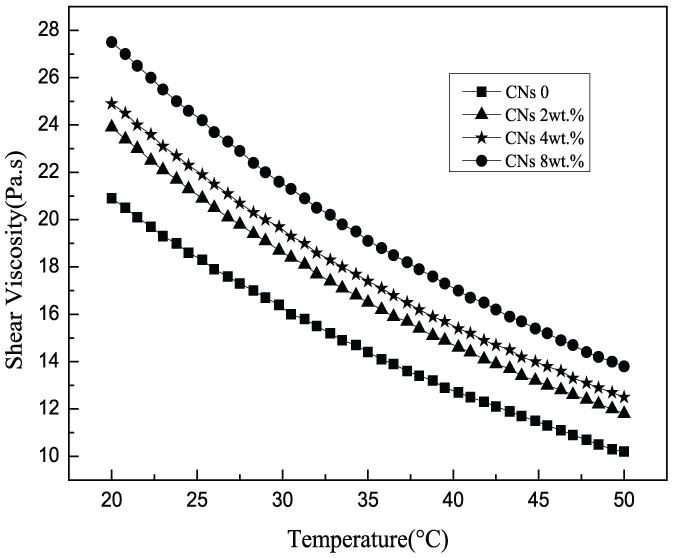
The viscosity of SA/CN solutions at different temperatures.
SEM images of SA/CN fibers
Figure 5 shows the SEM photos of the cross section of the alginate fibers with different CNs content. The tensile fracture surface of the pure alginate fiber was very smooth, just like a mirror, which belongs to the typical brittle fracture. 34 The stripes of the fiber fracture surface increase with increasing CNs content, indicating that the deformability of the matrix was changed with the addition of CNs. The “mirror surface” of the cross section nearly disappears with the addition of 2 wt% CNs, replaced by uneven saw-tooth structures. This trend becomes increasingly apparent with the addition of CNs, indicating that the addition of CNs effectively improves the brittle fracture of alginate fibers.35–37 However, it is noteworthy that some white particles appeared on the cross section of the fiber when CNs content reached 16 wt%, which may be attributed to the aggregation of CNs in the matrix.

SEM images of the alginate fibers’ cross-section with different CN contents: (a1 and a2) 0 wt%, (b1 and b2) 0.5 wt%, (c1 and c2) 2 wt%, (d1 and d2) 8 wt%, and (e1 and e2) 16 wt%.
XRD spectra of SA/CN fibers
Figure 6 presents the XRD curves of alginate fibers with different contents of CNs. There is quite a wide diffraction peak in the curve of pure calcium alginate fiber (a), indicating that there is no crystalline region in the sodium alginate macromolecules. The intense diffraction peak at 2θ = 22.6° and less intense diffraction peak 2θ = 15° were the characteristic peaks of CNs, which did not appear when CNs content was ⩽4 wt%, indicating that CN powder was dispersed evenly in the alginate matrix. 8 The diffraction peak at 2θ = 22.6° appeared when CNs content was 8 wt%, while both of the diffraction peaks at 2θ = 22.6° and 2θ = 15° appeared when CNs content reached 16 wt%, because of the enhancement of the signal with the addition of CNs, indicating that the crystallinity of alginate fibers increased.
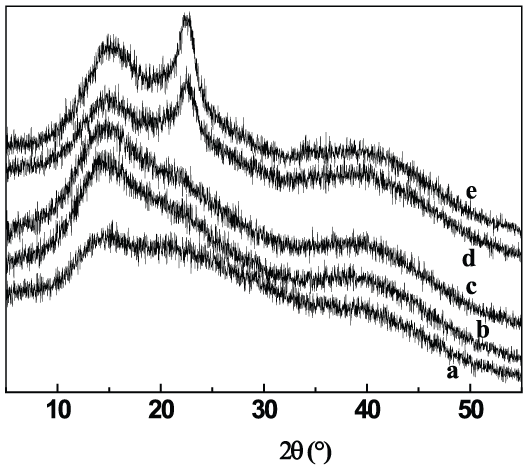
XRD spectra of SA/CN fibers with different CNs contents: (a) 0 wt%, (b) 0.5 wt%, (c) 2 wt%, (d) 8 wt%, and (e) 16 wt%.
Mechanical properties of SA/CN fibers
The mechanical properties of the composite materials could provide important information about their internal structure. The mechanical behavior of the fibers, including the pure alginate fiber and the fibers modified with various contents of CN powder, was investigated by tensile testing at room temperature. Figure 7 shows the stress–strain curves of the fiber materials. The tensile strength and elongation at break were determined from the curves. The content of CN powder has a profound effect on the tensile properties of alginate fiber, and the presence of a small amount of CN powder largely improved the tensile properties. It is noteworthy that the addition of CN powder improves the tensile strength and toughness of alginate fibers comparable to the pure alginate fibers. At the level of 8 wt% CNs, the fibers show the highest tensile strength and toughness. The tensile strength and elongation at break increased from 1.54 to 2.05 cN/dtex and from 8.29% to 15.05% with increasing CNs content from 0 to 2 wt%, respectively. This indicated that CN powder into the alginate matrix resulted in strong interactions between fillers and matrix. The enhancement in strength is directly attributed to the reinforcement provided by the dispersed CN powders. When the content is 16 wt%, the tensile properties of the alginate/CN composite fiber decrease significantly, which is possibly because of the aggregates of CN powder.
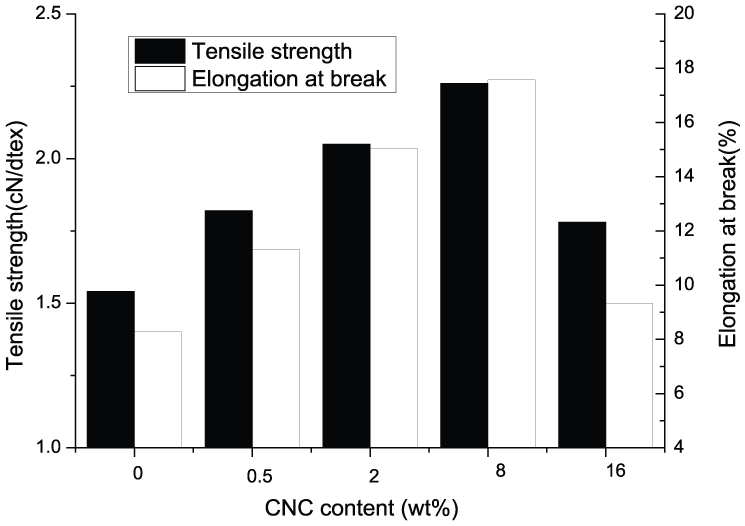
Mechanical properties of SA/CN fibers.
Conclusion
SA/CN fibers were obtained by wet spinning through a CaCl2 coagulation bath. CNs were prepared by sulfuric acid hydrolysis method. The particle size of CNs was between 10 and 1000 nm and the molecular weight of CNs is evenly distributed. The viscosity of the SA/CN spinning solutions decreases with increasing shear rates, exhibiting shear thinning behavior and decrease with an increasing temperature at the same shear rate. SEM images show that the addition of CNs effectively improved the brittle fracture of alginate fibers and enhanced the mechanical strength. The content of CN powder has a profound effect on the tensile properties of alginate fibers, and the presence of a small amount of CN powder largely improved the tensile properties. It is noteworthy that the CN powders strengthen and toughen alginate fibers when the CNs content is less than 8 wt%, resulting in the increase of the tensile strength and the elongation at break. The tensile strength and elongation at break increased by 33% and 82% with increasing CNs content from 0% to 2 wt%, respectively. This indicated that CNs powdered into the alginate matrix resulted in strong interactions between fillers and matrix. Since SA/CN fibers are bio-based, low-cost, high-strength, and easy-to-prepare fibers, they could greatly broaden the application of alginate fibers in biomedical fields, chemical engineering, garments, and other areas.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the National Key Research and Development Program of China (grant no. 2017YFB0309001).
