Abstract
Background
Mobility and cognitive impairment are prevalent and co-occurring in older adults with multiple sclerosis (OAMS), yet there is limited research concerning the role of disability status in the cognitive control of gait among OAMS.
Objective
We investigated the levels of prefrontal cortex (PFC) activation, using oxygenated hemoglobin (HbO2), during cognitively-demanding tasks in OAMS with lower and higher disability using functional near-infrared spectroscopy (fNIRS) to: (1) identify PFC activation differences in single task walk and cognitively-demanding tasks in OAMS with different levels of disability; and (2) evaluate if disability may moderate practice-related changes in neural efficiency in OAMS.
Methods
We gathered data from OAMS with lower (n = 51, age = 65 ± 4 years) or higher disability (n = 48, age = 65 ± 5 years), using a cutoff of 3 or more, in the Patient Determined Disease Steps, for higher disability, under 3 different conditions (single-task walk, Single-Task-Alpha, and Dual-Task-Walk [DTW]) administered over 3 counterbalanced, repeated trials.
Results
OAMS who had a lower disability level exhibited decreased PFC activation levels during Single-Task-Walk (STW) and larger increases in PFC activation levels, when going from STW to a cognitively-demanding task, such as a DTW, than those with higher disability. OAMS with a lower disability level exhibited greater declines in PFC activation levels with additional within session practice than those with a higher disability level.
Conclusions
These findings suggest that disability moderates brain adaptability to cognitively-demanding tasks and demonstrate the potential for fNIRS-derived outcome measures to complement neurorehabilitation outcomes.
Introduction
Multiple sclerosis (MS) is a chronic neurological disease characterized by diffuse axonal damage and brain atrophy that arises from demyelination and neurodegeneration in the central nervous system.1-3 There is a substantial growth in the prevalence of MS among older adults. In addition, mobility and cognitive impairments are prevalent, co-occurring outcomes in older adults with MS (OAMS)4-6 that typically worsen with MS disease progression expressed through disability status.4,7 Dual-task methodology has provided a valuable testbed for evaluating the cognitive control of mobility in middle to older-aged adults with and without MS through the use of Dual-Task-Walk (DTW) tasks, which have demonstrated utility in predicting fall risk,8-10 yet we are unaware of research examining the moderating role of disability status within DTW outcomes.
Disability status serves as a rough proxy of the influence of structural and functional brain changes in MS.11-15 A recent review 15 highlights how white matter tracts can be disrupted by lesions in persons with MS and can affect neural network function over time. Indeed, there is evidence of differential associations between structural and functional changes with mobility and cognitive outcomes among OAMS. 16 However, research have not yet examined how MS disability-based disease progression is associated with altered cortical activation in DTW conditions. Such an examination is important for the identification of the neural resources used in the performance of cognitive tasks while standing and walking, and implicit learning in OAMS with lower or higher disability that could be targeted in future neurorehabilitation. To that end, the examination of cognitive control changes in cognitively-demanding tasks in OAMS at different levels of disability can yield a better understanding of functional changes arising from both aging and MS.
The effect of disability on DTW outcomes in OAMS may be rooted in changes in brain activation parameters. Indeed, recent reviews and systematic-analyses of studies using functional near-infrared spectroscopy (fNIRS)17,18 have demonstrated increased activity in the prefrontal cortex (PFC) in DTW compared to Single-Task-Walk (STW) conditions in healthy older adults and adults with neurological conditions. This study focused on a wide coverage of the PFC due to its role in coordinating attentional resources in DTW,19,20 and distinct function of different PFC subregions (eg, orbitofrontal, frontopolar, and dorsolateral PFC) for walking and changes with increased age.21,22 Increases in PFC activation are expected in cognitively-demanding tasks, such as standing or walking while talking, relative to quiet standing, in OAMS, particularly in those with lower disability, based on prior work. 23 Given decreases in automaticity observed in neurological populations, 24 including MS,23,25,26 we expect increased PFC activity during STW in OAMS with higher disability, compared to those with lower disability. In particular, neural dysfunction, rather than neural compensation,27,28 would be expected in OAMS with higher disability, given previously observed worsening of cognitive and mobility function.4,7 Additionally, increased utilization of attentional resources to perform cognitively-demanding tasks, up to a task-difficulty-based ceiling, is expected in older adults with and without MS. 29 Thus, we expect OAMS with higher disability to be limited in their ability to further increase their PFC activity during DTW due to a decreased neural reserve, 30 compared to those with lower disability.
Changes in brain integrity that impact disability status may further moderate practice-related changes in neural efficiency in OAMS, and this can be studied through the use of fNIRS to examine PFC activation changes from STW to cognitively-demanding tasks across repeated trials within a single session. Cognitively-demanding tasks include tasks such as reciting alternate letters of the alphabet while standing or walking, which require both cognitive inhibition and working memory and have consistently demonstrated increased neural activation relative to normal walking.18,23,25,26,29 Prior work has demonstrated how aging and MS influence learning and performance.31,32 In particular, recent work has demonstrated the influence of MS on the practice-effects and on the improvements in the efficiency of brain activation during DTW in older adults. 29 Greater decreases in PFC activation over repeated trials are expected in older adults diagnosed with MS with lower disability, compared to those with higher disability, across repeated trials, given the neural efficiency model, 33 where persons with higher brain activation demonstrate comparable or decreased performance.
The present study investigated the levels of PFC activation during gait under single and dual-task conditions in older adults diagnosed with MS with lower and higher disability using fNIRS. Using a standing baseline, we examined relative changes in PFC activation during STW and cognitively-demanding tasks, to further our understanding of functional changes arising from both aging and MS as cognitive demands increase. We hypothesized that in comparison to OAMS with a lower disability level, those with a higher disability level would exhibit increased PFC activity during STW. Further, we hypothesized that OAMS with a higher disability level, in comparison to those with a lower disability level, would exhibit smaller increases in PFC activation levels from STW to DTW in response to the increased cognitive demands. Secondarily, we hypothesized that in comparison to OAMS with a higher disability level, those with a lower disability level would exhibit greater declines in PFC activation levels with additional within session practice. We further assessed the effect of disability and within session practice in OAMS on the cognitive and physical performance via changes in gait stride velocity and correct letter generation in single task alpha or DTW.
Materials and Methods
Participants
Older adults diagnosed with MS with lower (n = 51, mean age = 65.1 years, female = 36) or higher disability (n = 48, mean age = 64.5 years, female = 30) enrolled in an ongoing study titled “Brain Predictors of Mobility and Falls in Older Adults with Multiple Sclerosis”. 34 Disability groups were defined with the Patient Determined Disease Steps (PDDS) using a cutoff of 3 or more for higher disability. 35 The PDDS is a validated, self-report, ordinal scale (scores range from 0: no disability to 8: bedridden) that has been used to assess MS-related disability. 36 Participants in the current study were tested between September 2019 and August 2023, consisting of a partial use of the OAMS cohort previously reported, 29 and had complete cognitive, mobility, psycho-social, and fNIRS assessments. MS diagnoses, determined using the revised McDonald criteria, 37 were physician-confirmed. Extensive review of medical records was used to evaluate relevant health history and confirm participants were on stable disease-modifying-therapy for at least 6 months prior to study visits. MS participants are recruited from regional treatment centers and patient registry lists. Both MS cohorts were screened using a structured telephone interview to obtain verbal consent, assess medical and psychological history, and screen for dementia, mobility, and functional abilities to determine study eligibility. Following completion of the telephone interview, participants were scheduled for 2 in-person study visits in the medical center. The first visit consisted of a battery of neuropsychological tests, mobility protocols including the combined burst measurement, fNIRS, dual-task walking paradigm, and questionnaires. The second visit included an magnetic resonance imaging (MRI) of the brain and additional questionnaires. Cognitive status was determined via established clinical case conference procedures. 38 Diagnosis of any major neurological, psychiatric, or medical disease (excluding MS), inability to ambulate independently, contraindication to MRI, impairment of vision or hearing that would negatively impact testing served as exclusion criteria. All participants reviewed and signed a written informed consent form in the first study visit. The work described in this manuscript has been executed in adherence with The Code of Ethics of the World Medical Association (Declaration of Helsinki). The study was approved by the Institutional Review Board of Albert Einstein College of Medicine (Protocol #2019-10049). Prior papers have been published from this ongoing study, 29 but the associations among MS-disability, brain activation, and behavior have not been reported, not even partially, in any previous publication.
Protocol
There were 2 single task conditions: (1) STW and (2) Single-Task-Alpha (STA—Cognitive interference task). In STW participants were asked to walk on the electronic walkway, 20 feet long and 4 feet wide, at their “normal pace” for 3 consecutive loops, without any time constraints. In STA participants were asked to stand still while reciting alternate letters of the alphabet out load for 30-second. In DTW, participants were asked to walk around the walkway for 3 consecutive loops at their normal pace while reciting alternate letters of the alphabet. Participants started reciting alternate letters with the letter A or B, which was randomized across STA and DTW conditions. Participants were instructed to pay equal attention to both tasks. In each trial, the 3 test conditions were presented in a counterbalanced order using a Latin-square design. Reliability and validity for this walking paradigm have been established. 39 Three repeated trials, consisting of each task condition, were administered in 1 experimental session to determine the influence of short-term practice effects on PFC activation as well as gait and cognitive performances. 40 Participants received a 5-minute break between each of the repeated trials.
Measures
Gait Assessment
A 4 × 20-foot instrumented walkway using ProtoKinetics Movement Analysis Software was utilized to measure quantitative gait parameters during STW and DTW (Zenometrics, LLC; Peekskill, NY, USA). Stride velocity, cm/second, was used as the gait outcome in the current study. Split-half intra-class correlations for stride velocity in STW and DTW were greater than .95 revealing excellent internal consistency. 39 Stride velocity dual task cost was calculated using the following equation: ((stride velocity in STW—stride velocity in DTW)/ stride velocity in STW) × 100%.
Cognitive Assessment
The number of correct letters was computed for each participant under the STA and DTW conditions. To allow for across-task comparisons, this was measured as the total number of correct letters generated per minute, or letter generation rate. The letter generation rate dual task cost was calculated using the following equation: ([letter generation rate in STA − letter generation rate in DTW]/letter generation rate in STA) × 100%.
fNIRS System
The methods, reliability, and validity of the fNIRS data processing and analyses have been detailed previously. 41 Briefly, fNIRS measures changes in cortical HbO2 levels using light–tissue interaction properties of light within the near infrared range. fNIRS has been validated against traditional neuroimaging methods and is better able to handle motion artifacts. 42 Changes in hemodynamic activity in the PFC were assessed using fNIRS Imager 1100 (fNIRS Devices, LLC, Potomac, MD, USA). The system collects data at a sampling rate of 2Hz. The fNIRS sensor consists of 4 LED light sources and 10 photodetectors, which cover the forehead using 16 voxels, with a source-detector separation of 2.5 cm. The light sources on the sensor (Epitex Inc. type L4X730/4X805/4X850-40Q96-I) contain 3 built-in LEDs having peak wavelengths at 730, 805, and 850 nm, with an overall outer diameter of 9.2 ± 0.2 mm. The photodetectors (Bur Brown, type OPT101) are monolithic photodiodes with a single supply transimpedance amplifier. We implemented a standard sensor placement procedure. The fNIRS sensor pad was placed on the forehead so that the horizontal symmetry axis central (y-axis) coincided with the symmetry axis of the head (ie, in between the eyes). On the vertical axis, the sensor was positioned right above the eyebrows in relation to the international 10 to 20 system so that FP1 and FP2 marker locations were approximately positioned on the bottom voxel row level.
Data Processing
Preprocessing and Hemodynamic Signal Extraction
Initially, data from each of the 16 fNIRS channels for all participants were visually inspected to identify and eliminate saturation, dark current conditions or extreme noise, which could happen due to incorrect sensor placement. Next, wavelet denoising with Daubechies 5 (db5) wavelet was applied to the raw intensity measurements at 730 and 850 nm wavelengths for spiky noise suppression. 43 Changes in HbO2 were calculated from those artifact-removed raw intensity measurements using modified Beer–Lambert law (MBLL) as previously described. 41 In MBLL, we used the previously published wavelength and chromophore dependent molar extinction coefficients (ε) by Prahl, and age and wavelength adjusted differential pathlength factor (DPF). 41 To remove possible baseline shifts and to suppress physiological artifacts such as respiration and Mayer waves, we first applied Spline filtering 44 followed by a finite impulse response low-pass filter with cut-off frequency at 0.08 Hz. 41 In our analysis, HbO2 was used as our primary proxy for PFC activation as HbO2 is a more reliable and sensitive measure than other fNIRS-derived measures (eg, deoxygenated hemoglobin [Hb]) to locomotion-related changes in cerebral oxygenation. 45 To determine the relative task-related changes in HbO2 concentrations, data epochs in STW, STA, and DTW were corrected relative to proximal 10-second baselines, where participants are asked to count silently by 1, administered prior to each experimental condition as previously described. 41
Individual mean HbO2 and Hb data were extracted separately for STW, STA, and DTW. E-prime was used to synchronize gait and fNIRS events to the millisecond. Internal consistency of HbO2, determined by split-half intra-class correlations within each task, was excellent for STW (0.830), STA (0.864), and DTW (0.849). 39
Covariates
Age, sex, years of education, and global health status (GHS) served as covariates to account for possible effects of confounders and overall cognition. The GHS, a comorbidity measure, computes a total score based on the presence/absence of the following clinical conditions: diabetes, chronic heart failure, arthritis, hypertension, depression, stroke, Parkinson’s disease, chronic obstructive lung disease, angina, and myocardial infarction score (range 0-10). 38
Statistical Analysis
Descriptive statistics were used to summarize all study measures (mean ± SD for continuous measures, count and percent for categorical variables) and tabulated per group. We used linear mixed effects models (LMMs), which account for correlations among measurements within the same participant to compare stride velocity, cognitive performance, and PFC HbO2 between participants with lower and higher disability by task during STW, STA, and DTW, and across trials. Task and trial conditions served as the repeated measures, and disability status (lower vs. higher disability) was the 2-level between group variable, while controlling for voxel. Separate LMMs were conducted for stride velocity (STW and DTW), cognitive performance (STA and DTW) and PFC HbO2 levels (STW, STW, and DTW). In all models, STW served as the reference task condition; trial 1 served as a reference for trials 2 and 3 to evaluate practice effects. Analyses were first run unadjusted and then adjusted for covariates. To examine moderation effects, 2-way interactions of task by trial, group by task, and group by trial were conducted as well as 3-way interactions of group by task by trial. In all LMMs, random intercept and unstructured covariance type were used, fixed effects of all variables of interest and their interactions were tabulated. In all LMMs, post-hoc
Results
Participants
Participant characteristics were tabulated per group (Table 1). There were more females in lower disability (71%) compared to higher disability (63%) group. Mean PDDS was lower in lower (Median [IQR] = 1[1]) compared to higher disability (4[1.25]) group. There were more relapsing remitting MS in lower (71%) compared to higher disability (46%) group. Mean age, GHS, and years of education were comparable in lower (65.1 ± 4.2 years, 1.14 ± 0.89, 15.1 ± 2.3 years of education) and higher (64.5 ± 5.1 years, 1.50 ± 1.38, 15.3 ± 2.5 years of education) disability group. Unadjusted and adjusted analyses were not materially different for all LMMs. Therefore, analyses that fully adjusted for all covariates (age, sex, education, and GHS) were presented to account for confounding factors in analyses.
Sample Characteristics Stratified by Group.
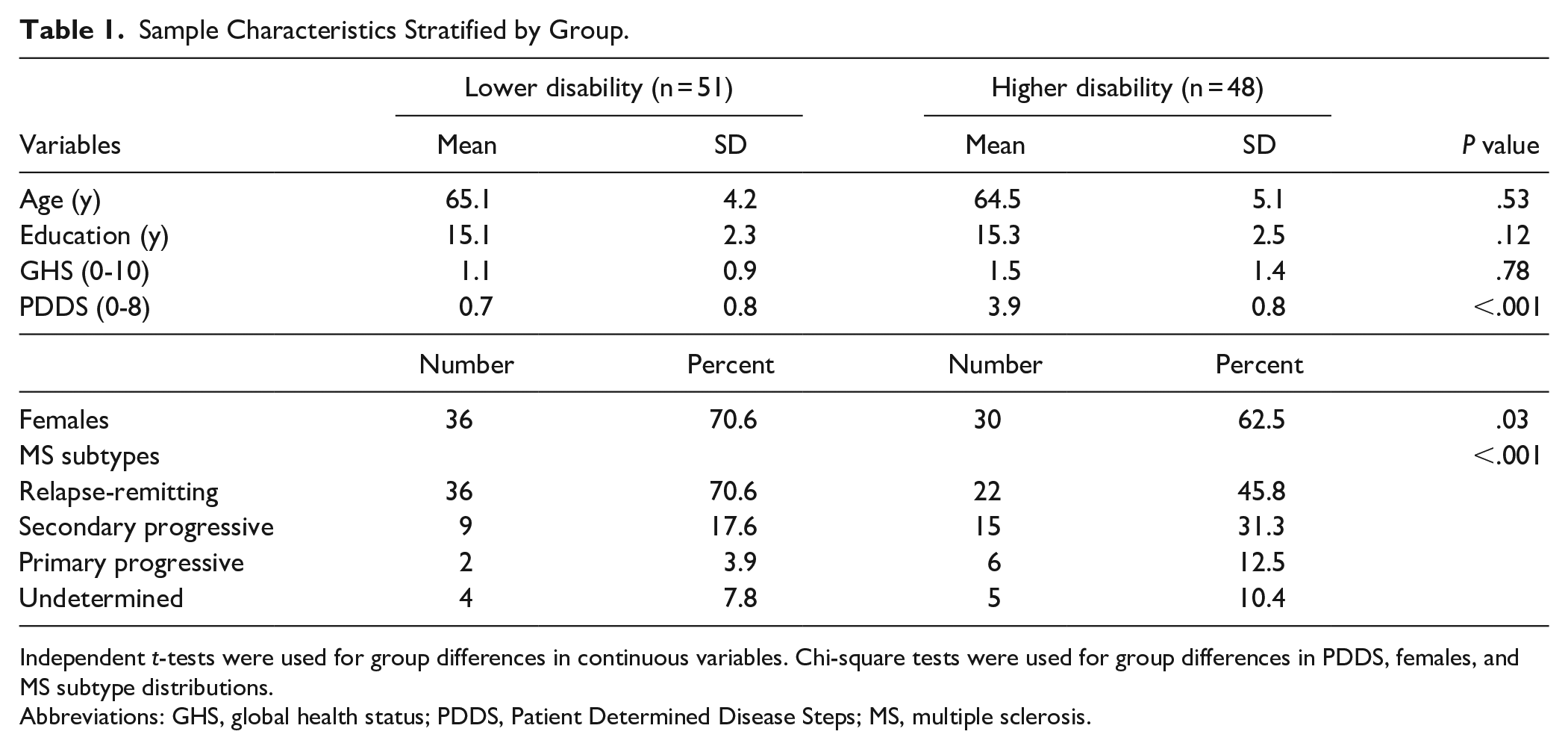
Independent
Abbreviations: GHS, global health status; PDDS, Patient Determined Disease Steps; MS, multiple sclerosis.
fNIRS-Derived PFC Activations
The overall LMM found significant effects of task (
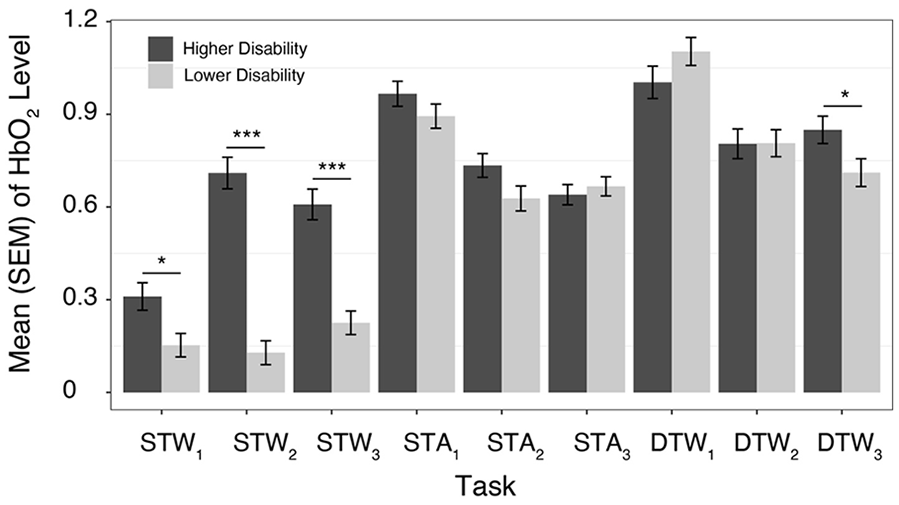
Mean (standard error of the mean [SEM]) of HbO2 levels across tasks and trials (1-3) in older adults diagnosed with MS with lower or higher disability.
Linear Mixed Effects Model Examining Main Effects and Interactions of Group, Trial, Task, as Well as Covariates on fNIRS-Derived HbO2.
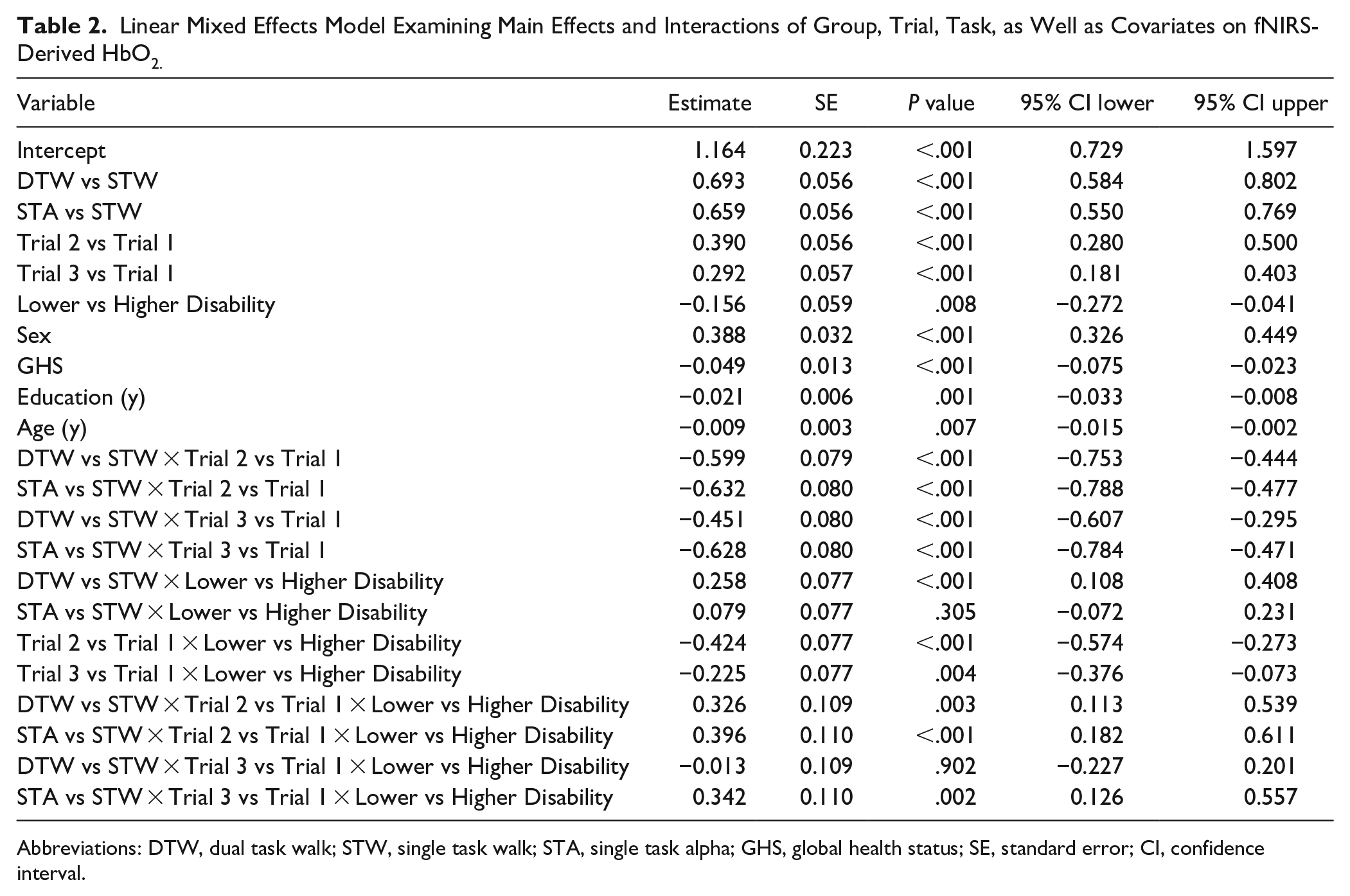
Abbreviations: DTW, dual task walk; STW, single task walk; STA, single task alpha; GHS, global health status; SE, standard error; CI, confidence interval.
Gait Performance: Stride Velocity
The overall LMM revealed significant effects of trial (
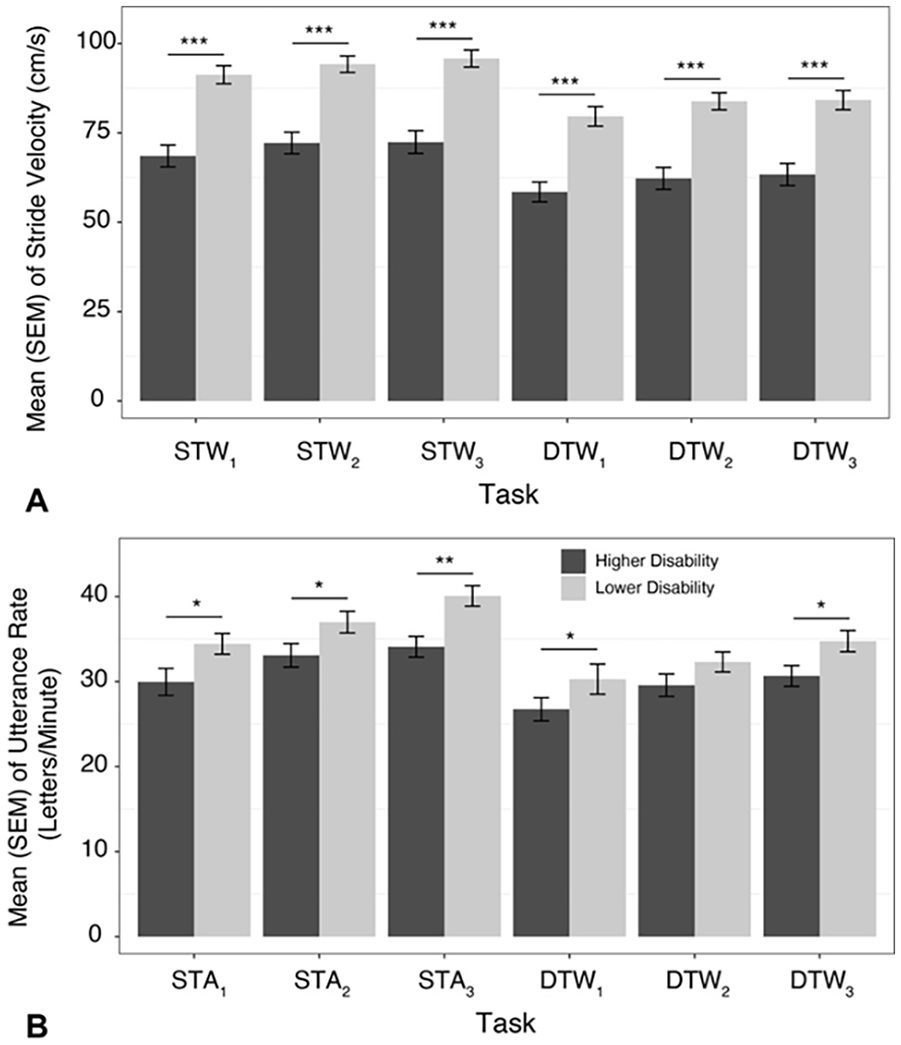
Mean (SEM) of (A) stride velocity and (B) utterance rate across tasks and trials (1-3) in older adults diagnosed with MS with lower or higher disability.
Linear Mixed Effects Model Examining Main Effects and Interactions of Group, Trial, Task, as Well as Covariates on Stride Velocity.
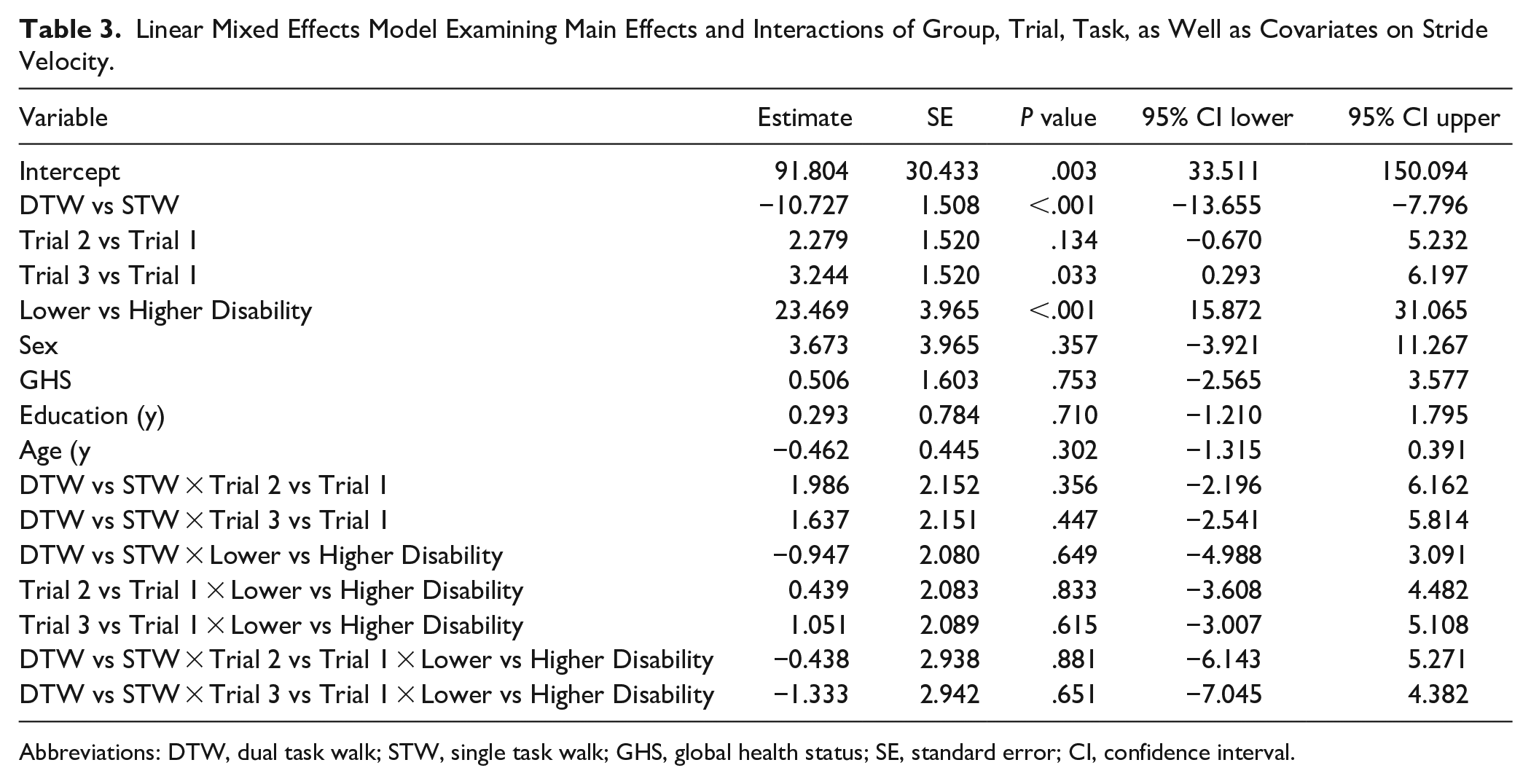
Abbreviations: DTW, dual task walk; STW, single task walk; GHS, global health status; SE, standard error; CI, confidence interval.
Cognitive Performance: Letter Generation Rate
The overall LMM revealed significant effects of trial (
Linear Mixed Effects Model Examining Main Effects and Interactions of Group, Trial, Task, as Well as Covariates on Letter Generation Rate.
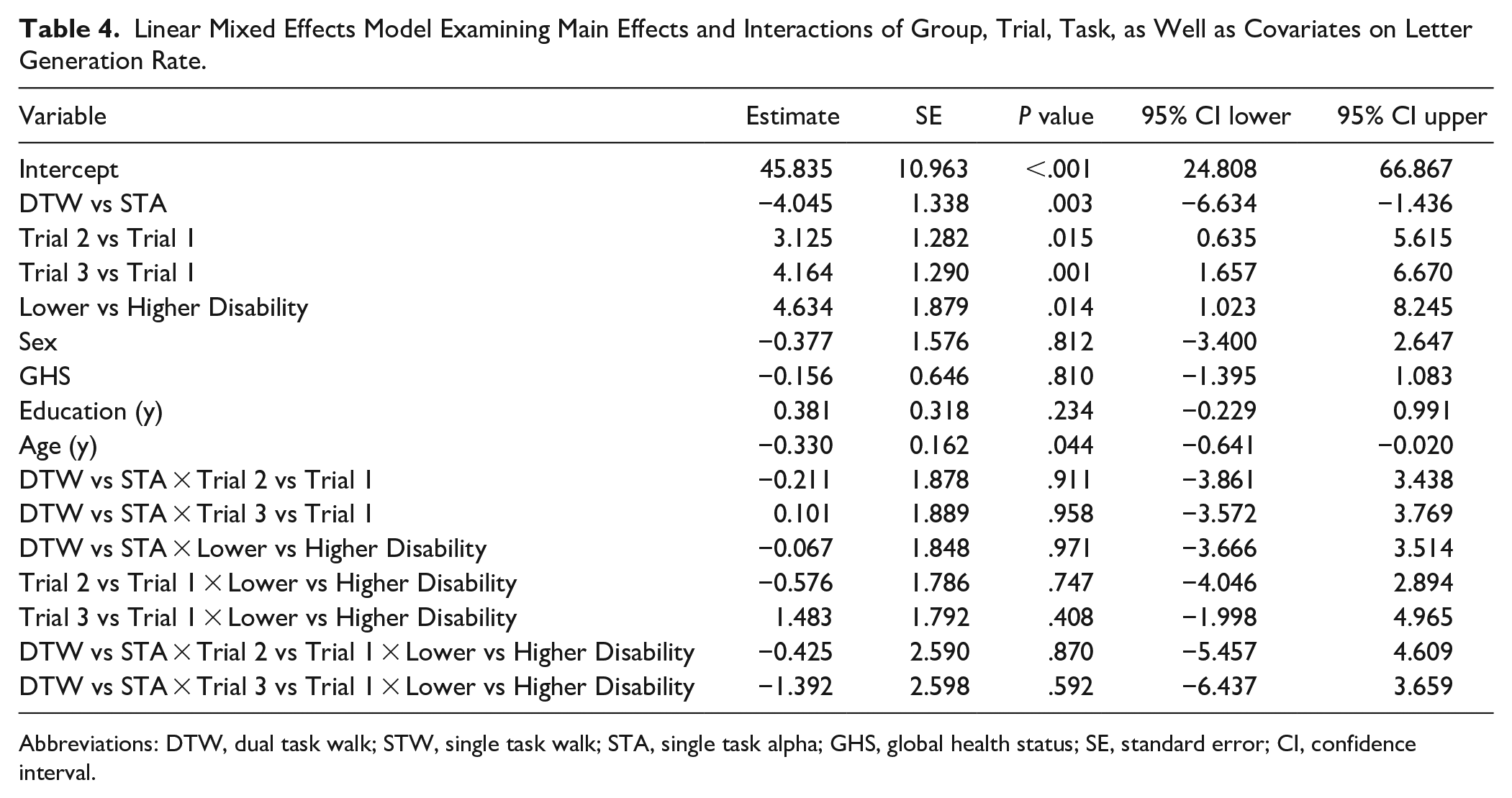
Abbreviations: DTW, dual task walk; STW, single task walk; STA, single task alpha; GHS, global health status; SE, standard error; CI, confidence interval.
Within-Session Practice Effects Stratified by Task and Group
To further clarify within-session practice effects, on all 3 outcome measures (ie, HbO2 levels, stride velocity, and letter generation rate), effect sizes, and their corresponding
Effect Size Estimates Using Beta Coefficients From Linear Mixed Effects Models for Between Trial Comparisons Stratified by Task and Group.
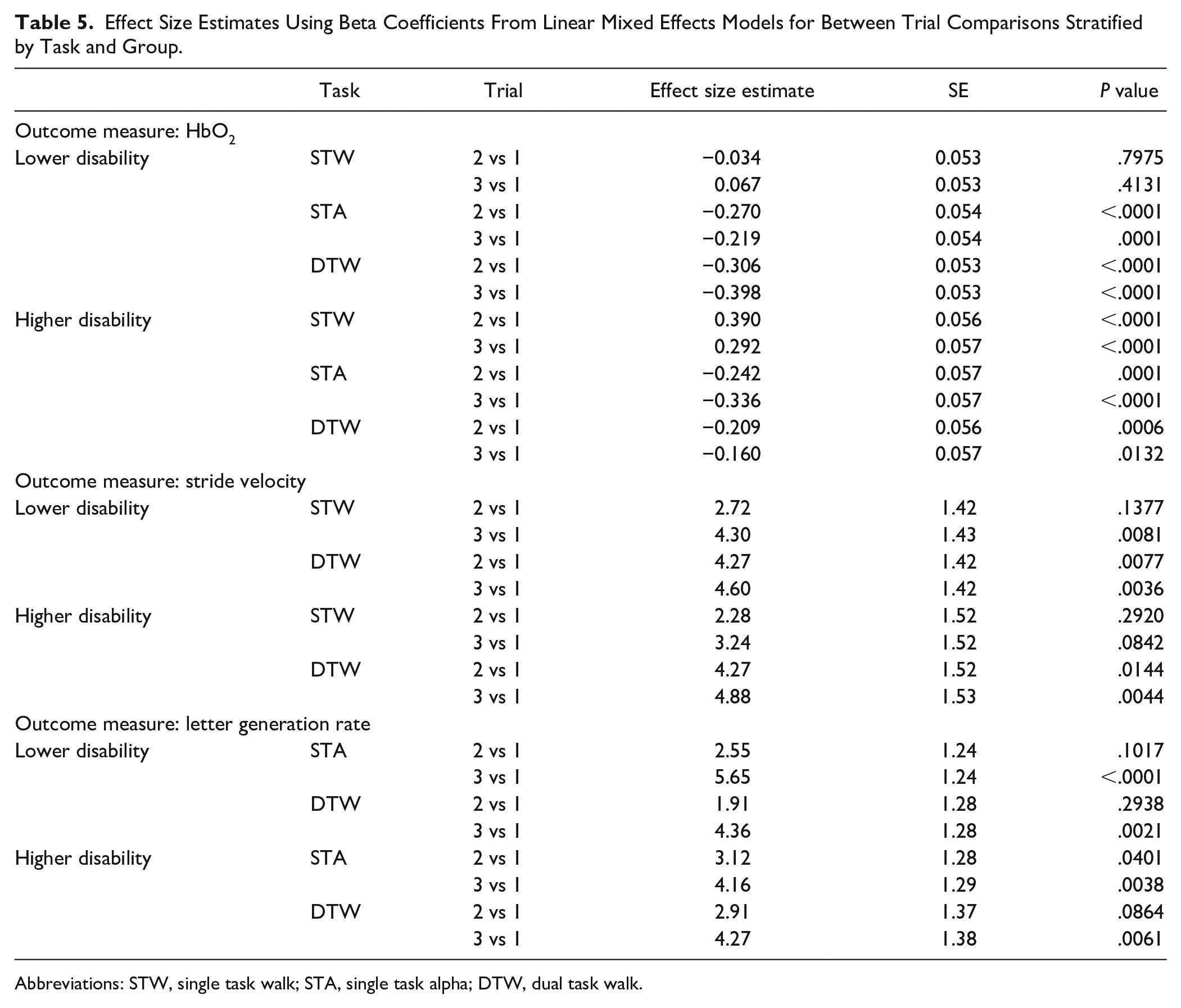
Abbreviations: STW, single task walk; STA, single task alpha; DTW, dual task walk.
Motor and Cognitive Dual Task Costs
Overall, LMMs revealed a significant trial and cohort effect for stride velocity dual task costs (
Discussion
The current study investigated PFC activation pattern differences in OAMS with lower versus higher disability and changes in neural efficiency across repeated trials within a single session. Consistent with our primary hypothesis, we found that in comparison to OAMS with a lower disability level, those with a higher disability level exhibited increased PFC activity during STW, consistent with lower automaticity. 24 Further, consistent with our second hypothesis, OAMS with a higher disability level, in comparison to those with a lower disability level, exhibited a smaller increase PFC activation level from STW to a cognitively-demanding task (ie, STA or DTW). This finding can be interpreted as a decreased capacity to utilize available resources for task activation. In addition, consistent with our third hypothesis, we found that in comparison to OAMS with a higher disability level, those with a lower disability level exhibited greater declines in PFC activation levels with additional within session practice. While both physical and cognitive performance was worse in the higher, compared to the lower, disability group, significant practice effects were observed in both groups in all tasks.
Effect of Higher Disability on Older Adults Diagnosed With MS
Consistent with prior work in OAMS, motor and cognitive dual task performance were worse in the higher, compared to the lower, disability group. 29 A recent review by Kirkland et al 47 of the dual task literature in persons with MS has demonstrated some inconsistencies in findings.48,49 Thus, the use of a reliable and valid 39 walking paradigm in OAMS presents an opportunity to examine the effect of the coupling of age-related and MS-related changes on mobility and cognitive function, although the effects of age per se were not examined in the current paper. In particular, increased PFC activation was observed in the higher, compared to the lower, disability group, consistent with the concept of neural inefficiency, 33 given the decreases observed in gait and cognitive performance in the higher disability group (ie, increased activation without a corresponding increase in task performance). Additional between-trial comparisons, stratified by task and group, supported significant practice effects on both gait and cognitive performances across tasks in OAMS. However, improvements in stride velocity across trials were more notable in STW, given the larger effect sizes, and increased stride velocity dual task cost observed. Lastly, no significant effects on letter generation rate dual task costs were observed, even though significant improvements in cognitive performance were seen in both STA and DTW from trial 1 to 3, which merit further exploration.
Consistent with prior evidence 23 that disability levels provide a moderating effect on PFC HbO2 levels in walking tasks in persons with MS, we found a significant 2-way interaction of task × group. Compared to those with a lower disability level, OAMS with a higher disability level demonstrated a decreased capacity to increase PFC activation levels when going from STW to STA or DTW, consistent with a decrease in neural reserve 30 in OAMS with higher disability. However, the choice of reference task must be considered in the interpretation of findings, as both OAMS with a lower and higher level of disability demonstrate a similar behavior in STA.
Effect of Practice and Moderation Due to Disability Levels in Older Adults Diagnosed With MS
While overall comparisons of PFC HbO2 levels in the first to the second and third trials revealed a significant increase in PFC activation levels, these changes were primarily driven by increased activation in OAMS with higher disability across repeated trials of STW. OAMS with a higher disability level, compared to those with a lower disability level, exhibited smaller declines in PFC activation levels with additional within session practice, as demonstrated by a significant 2-way interaction of trial × group. Consistent with principles of neural efficiency 33 and evidence of intact implicit motor learning in MS, 50 we found lower neural efficiency in OAMS with higher disability. Further, during DTW and STA, compared to STW, greater declines in PFC activation levels were seen with additional within-session practice, as demonstrated by a significant 2-way interaction of task × trial. This effect is due to the declines in PFC HbO2 levels seen across trials during DTW and STA, in contrast to the increases seen across trials in STW in both groups. We find that practice-related improvements in both gait, in trial 3 relative to trial 1, and cognitive performance, in trials 2 and 3 relative to trial 1, together with the improved efficiency in PFC HbO2 levels seen in DTW and STA in older adults diagnosed with MS, suggest improved utilization of neural resources in the performance of cognitive tasks while standing and walking, and implicit learning that could be targeted in future neurorehabilitation.
A significant trial × task × group interaction indicated that changes over trials in task-related PFC activation were moderated by disability level. Additional between-trial comparisons, stratified by task and group, supported significant practice effects, or declines in HbO2 across trials in STA and DTW in both groups. However, significant increases in HbO2 across trials were observed in OAMS with higher disability during STW. These findings suggest that walking under single task conditions, over repeated trials, may have been more cognitively demanding for OAMS with higher disability compared to lower disability. These findings are again consistent with the concept of neural inefficiency, where increased use of neural resources is expected in individuals with reduced brain integrity, even on relatively simple tasks. As MS progresses and both disability and structural brain damage increases, in addition to compensatory activation, decreases in neural efficiency are expected based on observed associations between processing inefficiency and white matter atrophy in MS. 51 MS negatively impacts the structural integrity of the brain,45,52 which may explain the lower efficiency observed in OAMS with higher disability in STW. Alternatively, differences between groups in STW across trials may be a result of an increased need for attentional resources in OAMS with higher disability, given associations between higher disability and increased muscle co-contraction or spasticity.53,54 In particular, given that white matter pathology leads to changes in cortical metabolic responses, which may be the driver of the fNIRS effects observed in this study, the role of brain network function disturbances on disability-based MS disease progression should be further explored.
To our knowledge, this is the first study demonstrating variable within-session improvements in the use of neural resources while dual task walking in OAMS, which diverge by level of disability. The ability of OAMS with both lower and higher disability to adapt to cognitively demanding tasks within a single session is significant, despite the co-occurrence of age-related and MS-related changes in structural and functional brain function is crucial, as it bolsters the evidence for dual task training, as a promising potential avenue for neurorehabilitation. Partly consistent with prior work examining the effect of a complex walking intervention, without the use of transcranial direct current stimulation, in older adults with lower mobility and executive function, 55 OAMS with both lower and higher disability demonstrated acute improvements in efficiency within a single session, consistent with longitudinal effects. However, differences between acute and longitudinal effects on PFC activation and gait performances may be expected, given prior work, 55 that merit further examination with the use of neuromodulation. Further, PFC activation levels provide a potentially modifiable factor to use when quantifying future interventions, consistent with a recent meta-analysis of fMRI studies of cognitively demanding tasks that found the PFC to be ideal for examining practice-related changes in brain efficiency. 33
Study Strengths, Limitations, and Future Directions
This study furthers prior investigations of dual task walking in persons with MS by providing novel insights into the moderating effect of disability on PFC activity in OAMS. This study suggests than even in short time courses and a single session, fNIRS may provide a useful measure of brain-related and practice-related changes in OAMS. MS participants were well-characterized, but due to study design and eligibility criteria requiring ambulatory capabilities, relatively intact cognition, and MRI, generalizability of the findings to more variable and impaired samples should be evaluated in future work. The use of the PDDS as a measure of MS-related disability provides an easy-to-administer, validated 36 self-report, measure that can identify individuals with distinct practice-related changes in behavioral and neural activation patterns. While the PDDS has been validated against the clinical Expanded Disease Disability Scale, 36 gait impairment, 56 and spinal cord reserve, 57 future work should examine additional associations with neural markers of disease progression. While analysis controlled for key potential confounders, the unweighted summation of 10 diseases used to derive the GHS variable provided limited adjustment for comorbidity in the current study. The examination of only the anterior and dorsolateral PFC and part of the inferior frontal gyrus is a limitation of this study, as the control of locomotion is dependent on numerous brain regions and networks outside of the PFC.58-60 As we are limited by the current fNIRS system to record from only cortical areas, or in a limited part of the PFC, the extent of disability-related or practice-related changes in brain activation levels observed in this study may be limited. Supplementary analyses examined the effects of disability and PFC region on Hb levels, which suggest differences in dorsolateral PFC regions and interactions with hemisphere, which merit further examination in future work. Furthermore, limitations of the current fNIRS device include the non-presence of short channels which preclude direct assessment of skin responses that might influence cortical changes in oxygenation patterns while walking. However, these factors as well as the effect of motion artifacts, while important, were not likely to influence the findings in this study given that experimental conditions were administered in a random order and had the same walking environment and physical requirements. Future work should examine different cognitive tasks, with different levels of difficulty, to further evaluate the ceiling effect in brain activity in OAMS. In addition, the examination of gait variability as a proxy for gait control and associations between changes in brain activity and motor and cognitive outcomes should be examined in future work. Lastly, the acute effects in behavioral and fNIRS outcomes seen in cognitively demanding tasks should be examined in longer time intervals in future work, to evaluate the implications on motor learning and retention of benefits in older adults diagnosed with MS.
Conclusion
In summary, this study provided the first evidence that performance in cognitively-demanding tasks and the efficiency of its associated PFC activation levels can be improved within a single session of repeated trials in OAMS with different levels of disability. We found that OAMS with higher disability, compared to those with lower disability, had increased PFC activation during STW. Further, we found that OAMS with higher disability, compared to those with lower disability, had smaller increases in PFC activation levels, when going from a STW to a more cognitively demanding task. We also found that OAMS with a higher disability level, compared to those with a lower disability level, exhibited smaller declines in PFC activation levels with additional within-session practice. These findings suggest that disability moderates brain adaptability to cognitively-demanding tasks and demonstrate the potential for fNIRS-derived outcome measures to evaluate neurorehabilitation outcomes.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241273411 – Supplemental material for Disability Moderates Dual Task Walking Performance and Neural Efficiency in Older Adults With Multiple Sclerosis
Supplemental material, sj-docx-1-nnr-10.1177_15459683241273411 for Disability Moderates Dual Task Walking Performance and Neural Efficiency in Older Adults With Multiple Sclerosis by Manuel E. Hernandez, Robert W. Motl, Frederick W. Foley, Mary Ann Picone, Meltem Izzetoglu, Michael L. Lipton, Mark Wagshul and Roee Holtzer in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank all participants for their participation in this study.
Author Contributions
Manuel E Hernandez: Conceptualization; Formal analysis; Writing—original draft; Writing—review & editing. Robert Motl: Conceptualization; Writing—review & editing. Frederick W Foley: Conceptualization; Writing—review & editing. Mary Ann Picone: Conceptualization; Writing—review & editing. Meltem Izzetoglu: Conceptualization; Formal analysis; Methodology; Writing—review & editing. Michael Lipton: Conceptualization; Writing—review & editing. Mark Wagshul: Conceptualization; Writing—review & editing. Roee Holtzer: Conceptualization; Data curation; Funding acquisition; Investigation; Project administration; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Izzetoglu has a minor share in fNIRS device. All other authors have no conflicts of interest to report in relation to the current article. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants from the National Institutes of Health (R01NS109023).
Supplementary material for this article is available on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
