Abstract
Background:
People with progressive multiple sclerosis (PMS) present motor (eg, walking) and cognitive impairments, and report fatigue. Fatigue encompasses fatigability which is objectively measured by the capacity to sustain a motor or cognitive task.
Objective:
To investigate the prevalence of walking and cognitive fatigability (CF) and the associated clinical characteristics in a large sample of PMS patients.
Methods:
PMS patients (25-65 years old) were included from 11 sites (Europe and North America), having cognitive impairment (1.28 standard deviation below normative data for the symbol digit modality test [SDMT]). Walking fatigability (WF) was assessed using the distance walk index (DWI) and CF using the SDMT (scores from the last 30 seconds compared to the first 30 seconds). Additional measures were: cognitive assessment—Brief International Cognitive Assessment for multiple sclerosis (MS), cardiorespiratory fitness, 6-minute walk, physical activity, depressive symptoms, perceived fatigue—Modified Fatigue Impact Scale (MFIS), MS impact—MSIS-29, and walking ability.
Results:
Of 298 participants, 153 (51%) presented WF (DWI = −28.9 ± 22.1%) and 196 (66%) presented CF (−29.7 ± 15%). Clinical characteristics (EDSS, disease duration, and use of assistive device) were worse in patients with versus without WF. They also presented worse scores on MSIS-29 physical, MFIS total and physical and reduced physical capacity. CF patients scored better in the MSIS-29 physical and MFIS psychosocial, compared to non-CF group. Magnitude of CF and WF were not related.
Conclusions:
Half of the cognitively-impaired PMS population presented WF which was associated with higher disability, physical functions, and fatigue. There was a high prevalence of CF but without strong associations with clinical, cognitive, and physical functions.
Trial Registration Number:
The “CogEx-study,” www.clinicaltrial.gov identifier number: NCT03679468.
Introduction
Multiple sclerosis (MS) is a chronic demyelinating disease characterized by disability progression of several body functions over-time. 1 Traditionally, MS has been divided into relapsing-remitting and progressive forms. 2 The progressive forms include secondary progressive MS (SPMS) and primary progressive MS (PPMS) and were defined as the accumulation of irreversible clinical disability. While the SPMS has an initial relapsing-remitting phase, this is not the case for PPMS. 2 Of note, progressive forms of MS (PMS) are associated with profound cognitive 3 and walking impairments. 4 In addition, fatigue affects up to 80% of people with MS (pwMS), 5 without overall significant differences in fatigue severity and interference by disease course.6,7
Fatigue in MS presents a broad construct that also includes fatigability, which can be objectively measured by an absolute or relative change in performance over a period of time during or after a given task (eg, motor or cognitive).8,9 Fatigability can be measured at different levels of the International Classification of Functioning, Disability, and Health. At body function level, static and dynamic fatigue indexes related to maximal muscle contractions have been established. 10 Also, muscle fatigue has been related to walking speed. 11 At the activity level, walking fatigability (WF) has been quantified by the decrement (pattern) in the distance walked or gait speed over time during prolonged walking (ie, 6-minute walk test (6MWT) and 12-minute of intermittent walk) sometimes also compared to a fast short walking bout.12-17 In recent years, a distance walked index, DWI, was introduced with a cut-off score of 10% decline in walking distance in the sixth compared to first minute of the 6MWT indicating abnormal WF compared to healthy subjects. 17 While walking is related to some extent to maximal muscle strength, fatigability in MS is considered as related to central impairments in voluntary drive. 18 Cognitive fatigability (CF) can be quantified by a decline in processing speed, reaction time, or accuracy over time after completing demanding cognitive tasks.19,20 The Symbol Digit Modalities Test (SDMT) has been proposed to measure CF, discriminating between pwMS and healthy controls.21,22 As for disabled and non-disabled populations, fatigability is an expected phenomenon while performing physical or cognitive tasks during prolonged time. However, in pwMS, fatigability may appear when performing simple tasks, such as walking for 6-minute, and is considered an important symptom affecting daily life functioning when sustained walking or cognitive functioning is required.
So far, WF and CF, when present, were predominantly reported in studies involving relapsing-remitting MS 9 or mixed samples.15,23 It is expected that more disabled pwMS24,25 and those presenting PMS would suffer of WF. 15 In the study of Leone et al 15 including mixed sample of relapsing-remitting MS and PMS, WF defined by a cut-off of −15% was present in 39% of SPMS and in 50% of the PPMS. On the other hand, the prevalence and magnitude of CF in pwMS have not yet been elucidated. Consequently, it has not been established to which extent PMS is characterized by the presence and the magnitude of WF and CF, whether these are related, and if WF or CF are associated with clinical characteristics. This study investigated the prevalence of WF and CF in a large cohort of cognitively-impaired PMS patients, and documented clinical characteristics related to the presence of WF and CF. We hypothesized that approximately half (40%-50%) of the PMS sample would present WF or CF, and those with WF would be more impaired regarding physical capacity.
Methods
The present study reports a secondary analysis of baseline data from a multicenter randomized controlled trial entitled “Improving Cognition in People with Progressive Multiple Sclerosis (PMS) Using Aerobic Exercise and Cognitive Rehabilitation” (The “CogEx-study,” www.clinicaltrial.gov identifier number: NCT03679468). Approval was received from the local institutional ethical standards committees on human experimentation for any experiments using human subjects. Written informed consent was obtained from all subjects prior to study participation according to the Declaration of Helsinki. A detailed description of methodology and study design has been reported elsewhere 26 and is only briefly summarized below. Of note, although the main reference has been recently published, 27 other papers have already been published based on data from the present study.28-32 All procedures described below were standardized across sites via comprehensive in-person and remote training, a detailed study manual and quality control conducted on a case-by-case basis.
Participants
For this study, 298 PMS patients were enrolled and data were collected across 11 sites in North America and Europe (Canada (1 site), USA (2 sites), United Kingdom (2 sites), Denmark (1 site), Belgium (1 site), and Italy (4 sites)). A table describing the number of patients included per site is shown in the supplementary material (see Supplemental Table 1).To be included in the trial, MS patients had to (a) have a confirmed diagnosis of PMS; (b) be between 25 and 65 years old; (c) have a corrected visual acuity >20/70; (d) demonstrate intact language comprehension based on Token Test scores >28 and to understand instructions; (e) have a physical active score on the Health Contribution Score of the Godin Leisure-Time Exercise Questionnaire <23 units; (f) not be severely depressed based on the Beck Depression Inventory-II scores <29 and16; and (g) demonstrate impaired cognitive processing speed based on SDMT scores ≥1.282 standard deviation-units below the age-, sex-, and education-adjusted normative score (ie, ≤10th percentile). 33 For this study, participants who completed the 6MWT and SDMT were included.
Neuropsychological Evaluation and CF
The neuropsychological assessment was performed in 1 session using the Brief International Cognitive Assessment for MS,
34
which consists of the SDMT (information processing speed), the immediate recall trials of the California Verbal Learning Test, and the Brief Visuospatial Memory Test.
To calculate CF, SDMT correct answers per 30 seconds time intervals were recorded generating 3 scores (ie, first score: total number of correct answers during the first 30 seconds; second score: total number of correct answers from 30 to 60 seconds; third score: total number of correct answers from 60 to 90 seconds). The third and first scores were used to calculate the CF index (CFI) according to the following formula: CFI SDMT = (third score − first score/first score)
Physical Performance and WF
Height and weight were assessed and used to calculate the body mass index. An incremental cardiopulmonary exercise test was conducted to assess peak aerobic capacity (VO2peak) and power using a recumbent stepper. We refer to the protocol paper for a more detailed description. 26 Walking performance was assessed by the 6MWT. Subjects were instructed to walk at their fastest speed, and to cover as much distance as possible, according to the script of Goldman et al. 35 Subjects were notified, without further encouragement each minute. Distances walked per minute and total distance was recorded. Subjects walked back and forth along a 15- or 30-m hallway turning around cones at each end. 36 Free-living moderate-to-vigorous physical activity (MVPA) was measured using waist-worn ActiGraph model GT3x + accelerometers (ActiGraph, Inc., Pensacola, FL, USA) over a 7-day period. Participants wore the accelerometer on an elastic belt around the waist over the non-dominant hip during the waking hours of a 7-day period, and further recorded wear time in a log for compliance. The raw accelerometer data were downloaded and processed using the low-frequency extension into 60-second epochs using ActiLife (ActiGraph Corporation) software. The full procedure and data processing can be found in a previously published paper from the CogEx initiative. 30 In the current study, free-living MVPA is expressed in percent of total wear time (ie,. percent MVPA) across valid days.
WF has previously been defined 15 and was here expressed as the Distance Walk Index (%) which was calculated as follows: DWI = (Distance walked at minute 6 − Distance walked at minute 1/Distance walked at minute 1) × 100. The cut-off for abnormal DWI has previously been reported as slowing down >10% 17 in a sample with predominantly relapsing-remitting MS.
Patient Reported Outcomes
Patient Reported Outcome (PRO)’s included the Hospital Anxiety and Depression Scale, Beck Depression Inventory), Modified Fatigue Impact Scale (MFIS), Multiple Sclerosis Walking Scale (MSWS-12), and the Multiple Sclerosis Impact Scale (MSIS-29) version 2.37,38
Fatigue and Subtypes of Fatigability
Fatigue in MS is a multifaceted concept, characterized by diverse definitions. For example, a list of references for fatigue definitions can be found in the study of Beckerman et al. 39 Frequently cited descriptions9,40-43 (REF) include: a decline in performance following prolonged or unusual exertion, coupled with sensations of sensory, motor, cognitive, or subjective fatigue9,42; a subjective depletion of physical or mental energy, perceived by the individual or their caregiver as an interference to normal activities 41 ; and the perception of reduced mental or physical energy, impacting daily routines.9,43 This fatigue extends to the definition of subtypes of fatigability, including both mental or CF, affecting attentional tasks, and physical fatigue, influencing the initiation and maintenance of motor exercises (eg, reaction time, peak force, and walking speed).9,40,42,43
Although efforts have been made to disentangle fatigability (ie, absolute or relative change in performance over a period of time during or after a given task),9,43 it is unclear how walking and CF root in distinct unidimensional constructs. WF, focuses on the physical aspect of fatigue, particularly in the context of ambulation. The underlying unidimensional construct involves factors such as muscle strength, endurance, and the efficiency of neuromuscular coordination. In MS, the demyelination of nerve fibers disrupts the communication between the central nervous system and muscles, contributing to WF.10,11,44 WF has previously been defined 15 and was here expressed as the DWI (%) which was calculated as follows: DWI = (Distance walked at minute 6 − Distance walked at minute 1/Distance walked at minute 1) × 100.
CF, on the other hand, pertains to the cognitive processes affected by MS-related fatigue. It involves the capacity of the brain to sustain attention, process information, and perform complex tasks over an extended period. The unidimensional construct underlying CF often involves the efficiency of neural networks, neurotransmitter function, and the overall cognitive reserve of the individual. 20 In this study, categorize participants with CF, the number of correct answers on the SDMT was used. During the SDMT, the number of correct answers for each 30 seconds time interval over the total 90 seconds were recorded generating 3 values (ie, first: total number of correct answers during the first 30 seconds; second: total number of correct answers from 30 to 60 seconds; third: total number of correct answers from 60 to 90 seconds). Using these, the third and first values were used to calculate a CF index (CFI) according to the following formula: CFI SDMT = (third total − first total/first total) × 100.
Group Classifications
To investigate the prevalence of WF and descriptive characteristics, participants were allocated into 2 groups: those presenting with WF and those not presenting walking-related motor fatigability (NWF). To allocate the participants into the WF group, a cut-off value of −10% for the DWI was used. 17 To investigate CF and descriptive values, participants were classified in groups by their CF index (CFI SDMT): those presenting with CF and those not presenting with CF (NCF) groups. An arbitrary cut-off value of −10% for the CFI SDMT was used to classify the participants given current lack of established cut-off values.
Statistical Analyses
Descriptive statistics were used to summarize the participant demographic and clinical characteristics using means (standard deviation) for continuous variables, median (25%, 75%) for ordinal variables, and frequencies (%) for categorical variables. Differences between participants with WF only, CF only, both WF and CF, or NWF and NCF were evaluated using chi square test for categorical variables, an analysis of variance (ANOVA) or Kruskal–Wallis test for continuous variables, as appropriate. Pairwise comparisons for the ANOVA utilized Tukey-adjusted comparisons and the Kruskal–Wallis utilized the Dwass, Steel, Critchlow–Fligner method adjustment. The associations between the DWI and CFI, clinical, physical and cognitive measures, and PROs were evaluated using Pearson or Spearman correlation coefficients (
Results
Prevalence of WF and CF and Differences Between Groups
As shown in Figure 1 and Table 1, 51% of our sample was observed to have WF (average DWI = −28.9 ± 22.1%) during the 6MWT, while 66% had CF (average CFI = −29.7 ± 15.0%) for the SDMT. For WF, there was no difference regarding gender prevalence between groups, but the WF group presented a higher percentage of SPMS, while PPMS was more prevalent in the group without WF. Additionally, the proportion of patients using assistive device (bilateral or unilateral) was higher in the WF group compared to the NWF group. For CF, there were no prevalence differences between the CF and NCF groups when comparing gender, SPMS versus PPMS and educational level (ie, college, secondary, and primary).
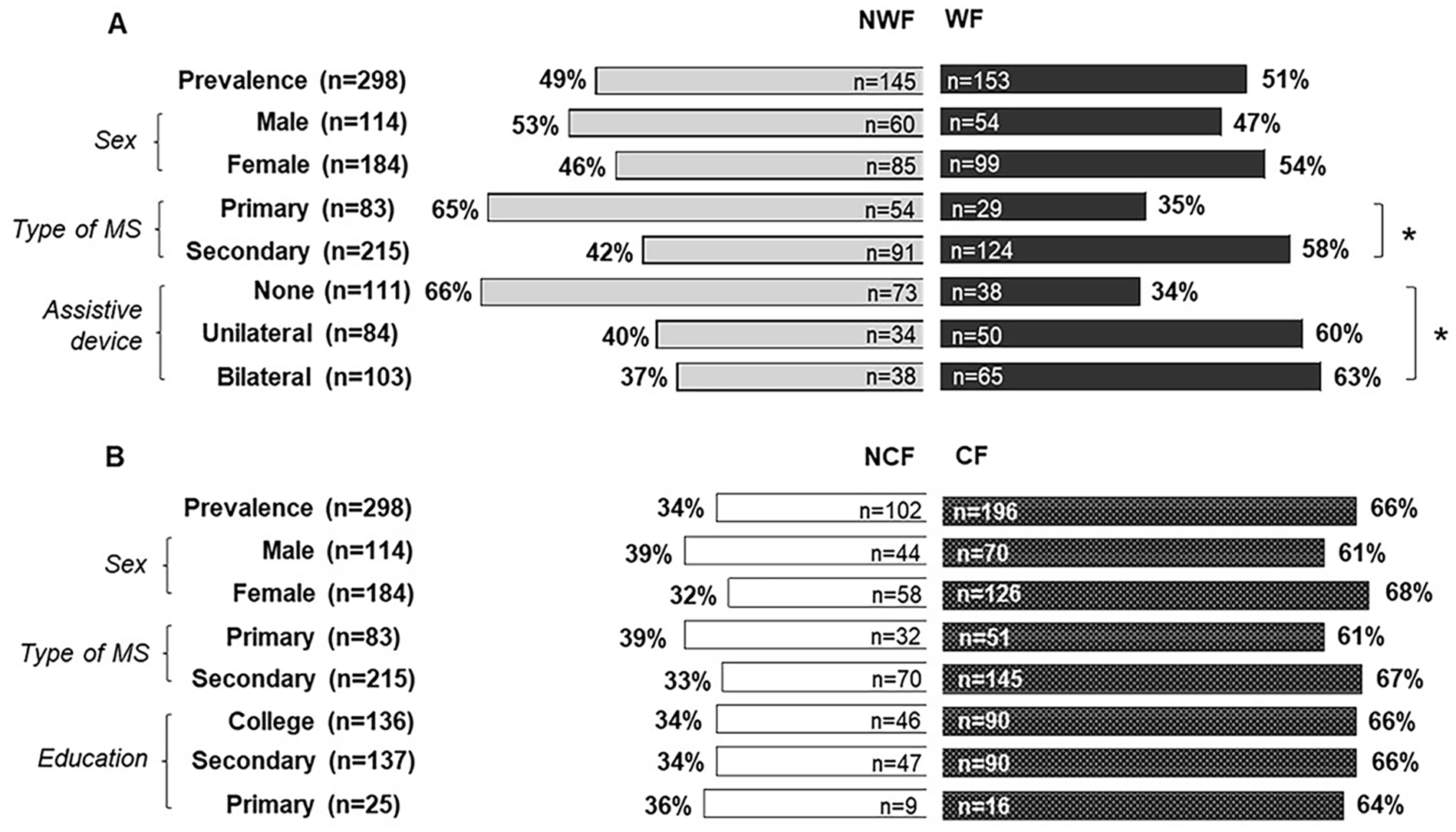
(A) Prevalence of walking fatigability (WF) and (B) cognitive fatigability (CF) in persons with progressive multiple sclerosis and cognitive impairment.
Demographic Characteristics and Clinical Results From the Total Sample and From the WF and CF Groups.
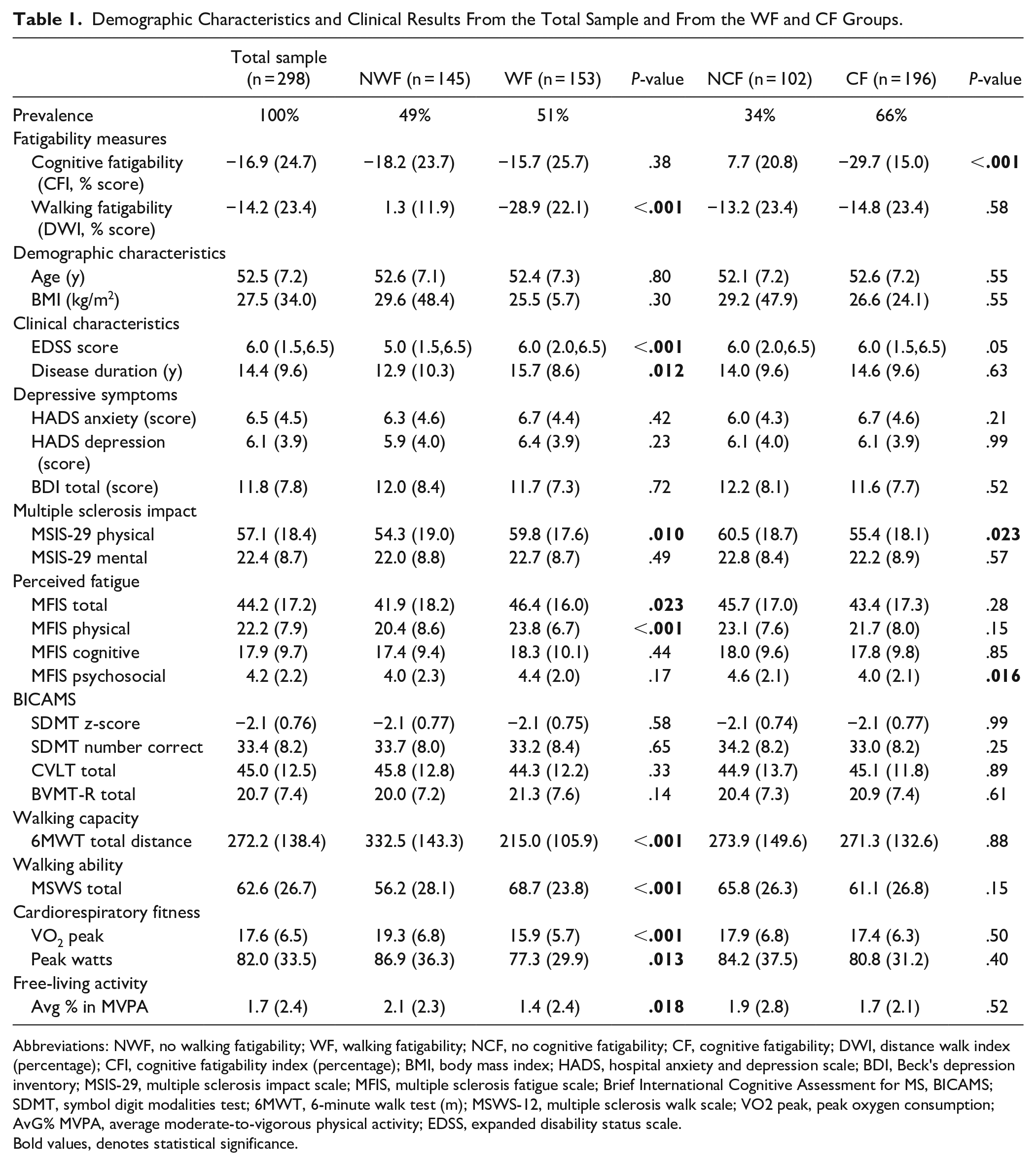
Abbreviations: NWF, no walking fatigability; WF, walking fatigability; NCF, no cognitive fatigability; CF, cognitive fatigability; DWI, distance walk index (percentage); CFI, cognitive fatigability index (percentage); BMI, body mass index; HADS, hospital anxiety and depression scale; BDI, Beck’s depression inventory; MSIS-29, multiple sclerosis impact scale; MFIS, multiple sclerosis fatigue scale; Brief International Cognitive Assessment for MS, BICAMS; SDMT, symbol digit modalities test; 6MWT, 6-minute walk test (m); MSWS-12, multiple sclerosis walk scale; VO2 peak, peak oxygen consumption; AvG% MVPA, average moderate-to-vigorous physical activity; EDSS, expanded disability status scale.
Bold values, denotes statistical significance.
Figure 2 presents the results for the outcomes measured and the comparisons between WF and NWF groups, and CF and NCF groups. PMS patients in the WF group walked shorter distance on the 6MWT, presented a lower VO2 peak, performed less moderate-to-vigorous physical activity (AvG%) and reported higher scores for the impact of MS on walking ability (MSWS-12), physical functions (MSIS-29) and fatigue (MFIS total and physical), compared to the NWF group. Comparisons between CF and NCF groups only showed significant differences for the MSIS-29 physical and MFIS psychosocial, with higher scores for the NCF group.
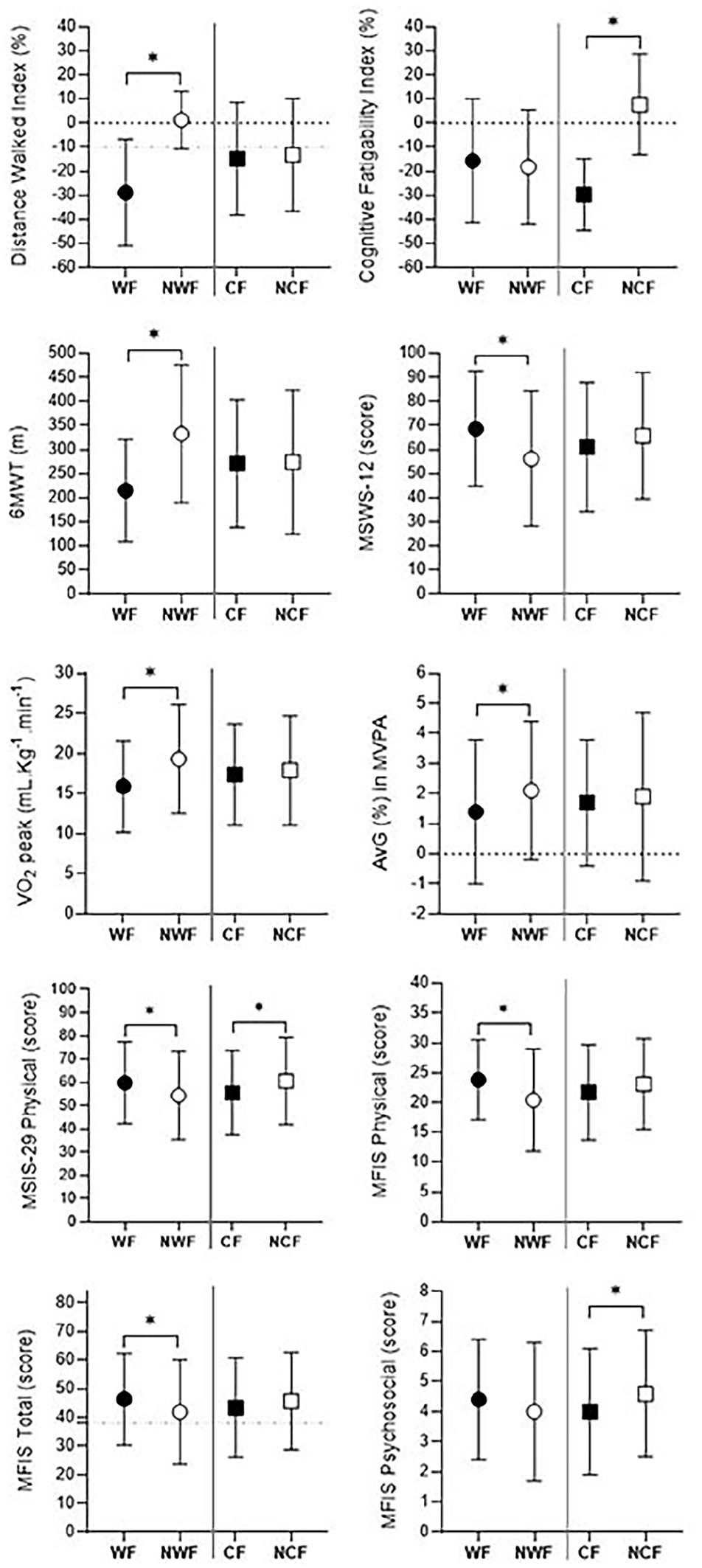
Comparisons between walking fatigability (WF) versus no walking fatigability (NWF), and cognitive fatigability (CF) versus no cognitive fatigability (NCF) groups. Values are presented as mean and standard deviation.
Walking and SDMT Response Patterns During the 6MWT
Figure 3 presents the distance walked minute-by-minute of the 6MWT for the WF and NWF (A), and CF and NCF (B) groups. Distance walked was significantly lower in the last minute of the 6MWT for the WF compared to the NWF group (Figure 3A). No differences in distance were found between CF and NCF groups (Figure 3B). Figure 3C presents the number of right answers on the SDMT in every 30 seconds for the WF and NWF groups. In the Figure 3D the SDMT response shows the distinct patterns occurring in the CF and NCF groups.
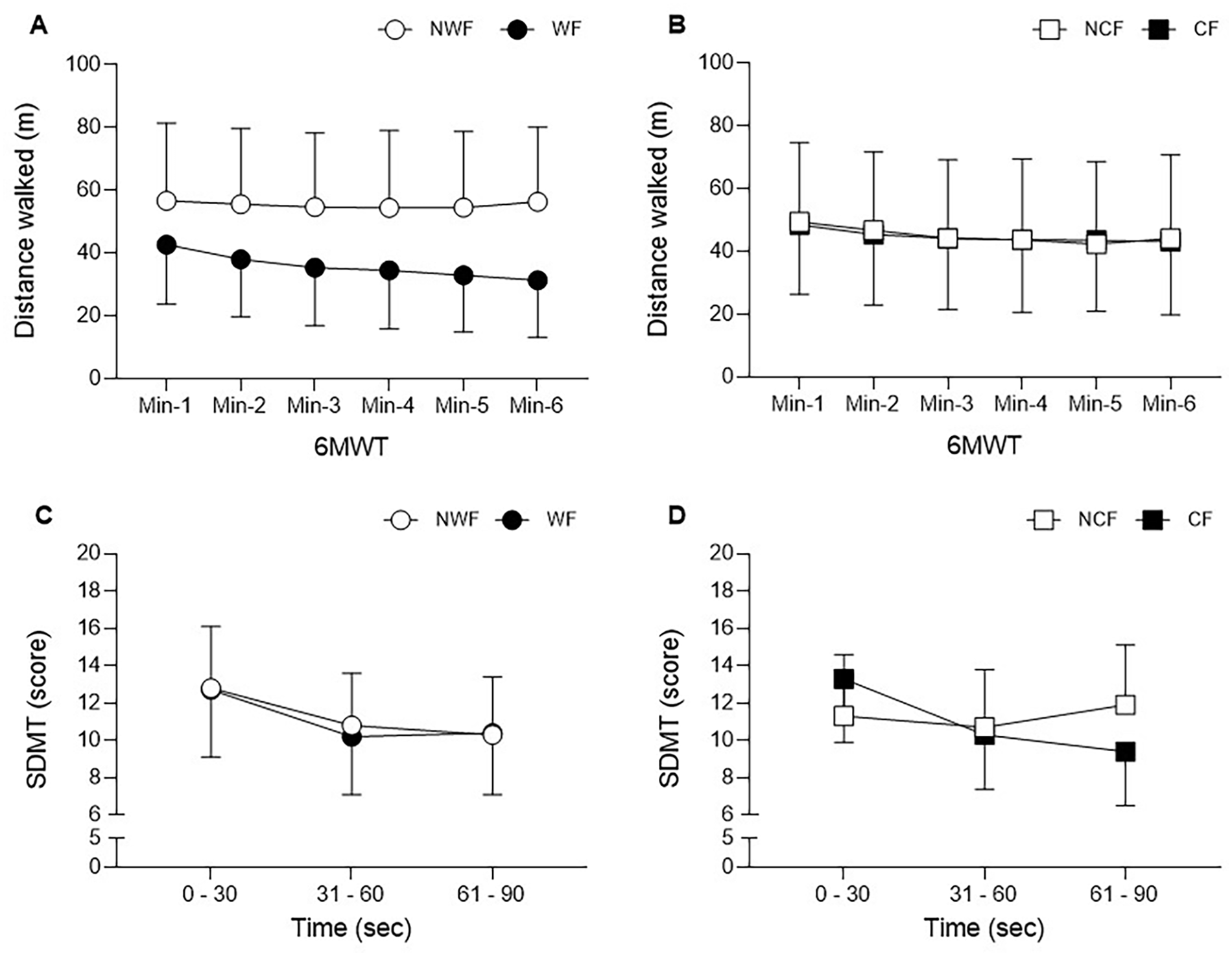
Behavior of walking and cognitive capacity in people with progressive multiple sclerosis. (A and B) Distance walked minute-by-minute of the 6-minute walk test. (A) Walking fatigability (WF) and no walking fatigability (NWF) groups. (B) Cognitive fatigability (CF) and no cognitive fatigability (NCF) groups. (C and D) Symbol Digit Modality Test, scores presented in intervals of 30 seconds. (C) Walking fatigability (WF) and no walking fatigability (NWF) groups. (D) Cognitive fatigability (CF) and no cognitive fatigability (NCF) groups. Values are presented as mean and standard deviation.
Associations Between WF, DWI, Clinical Measures, and PROs
Table 2 presents the correlation coefficients and the 95% confidence interval between the DWI% and clinical measures and PRO’s for the total sample of PMS patients. Although there were some significant correlations between DWI and depression, physical fatigue and physical function, in general, correlations were weak.
Pearson and Spearman Correlations of Walking Fatigability, Calculated by the Distance Walk Index (DWI%), and Demographic and Clinical Characteristics, Depression, Perceived Fatigue, Cognitive Functions and Physical Outcomes.
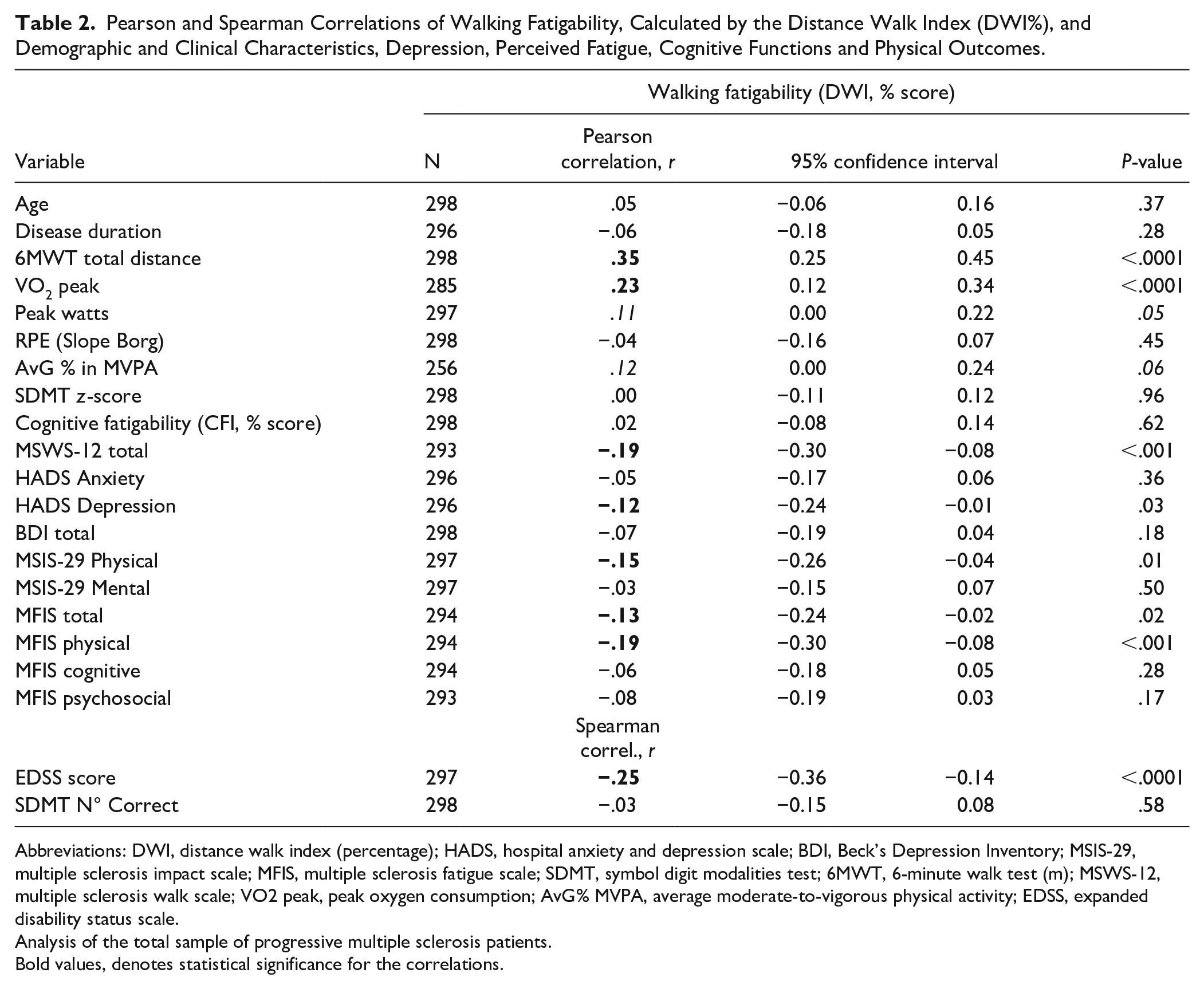
Abbreviations: DWI, distance walk index (percentage); HADS, hospital anxiety and depression scale; BDI, Beck’s Depression Inventory; MSIS-29, multiple sclerosis impact scale; MFIS, multiple sclerosis fatigue scale; SDMT, symbol digit modalities test; 6MWT, 6-minute walk test (m); MSWS-12, multiple sclerosis walk scale; VO2 peak, peak oxygen consumption; AvG% MVPA, average moderate-to-vigorous physical activity; EDSS, expanded disability status scale.
Analysis of the total sample of progressive multiple sclerosis patients.
Bold values, denotes statistical significance for the correlations.
Co-Existence of Motor and CF
Correlation analyses revealed that CF and WF were not related (
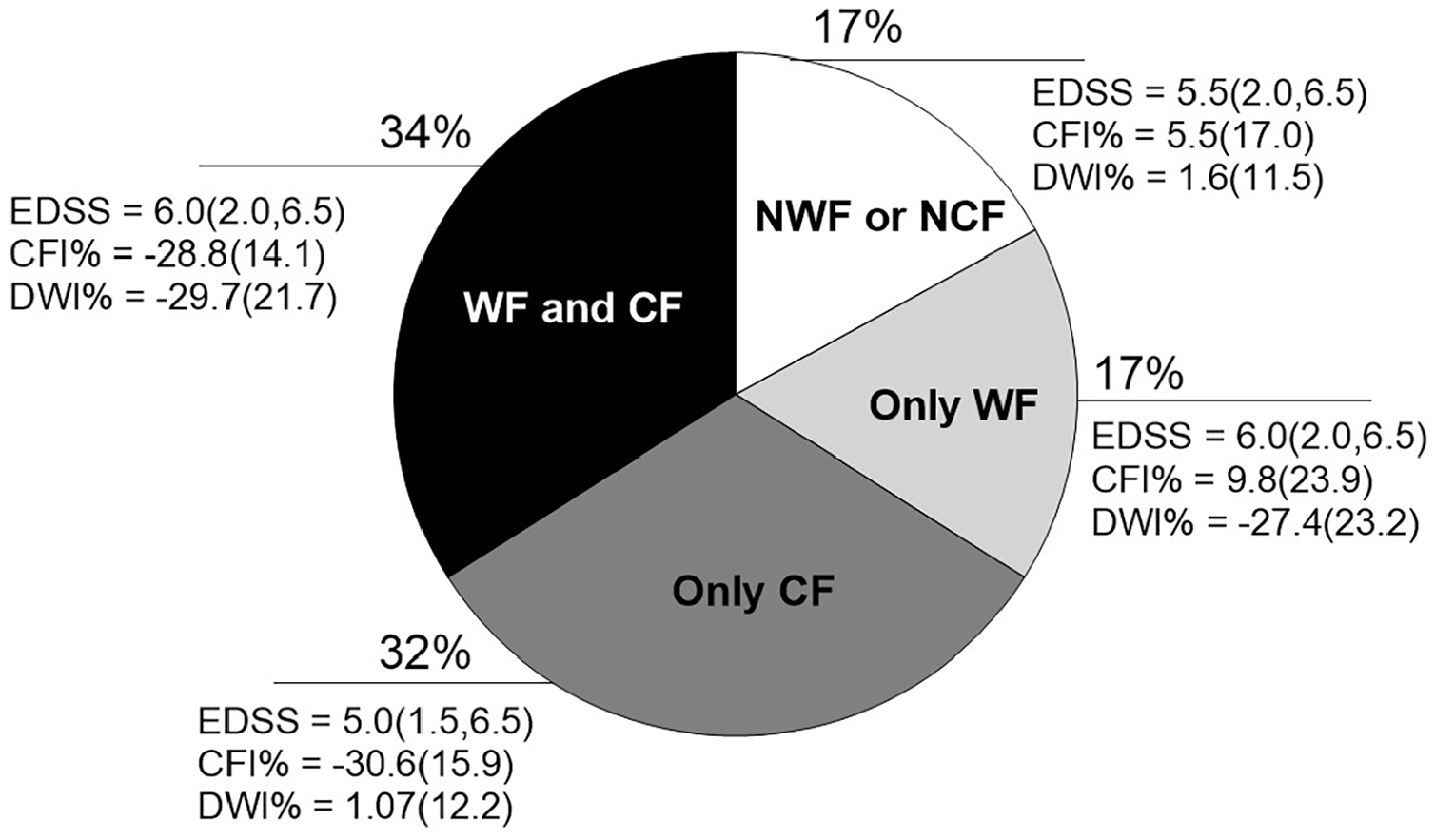
Distribution of progressive multiple sclerosis patients regarding the isolated or simultaneous presence of motor and cognitive fatigability.
As for comparisons between subgroups, patients classified with only CF presented better physical functions (walking and aerobic capacity), higher level of moderate-to-vigorous physical activity, lower score in the MFIS total and significantly less in the physical and psychosocial subcategories, when compared to the only WF and Both WF and CF subgroups. Table 1 of the Supplemental Data.
Discussion
The present study reported on the prevalence, magnitude, and association of clinical characteristics with walking and CF in a large sample of cognitively-impaired progressive MS patients. Overall, the study showed WF in half of the PMS sample, which was weakly associated with more severe disability. Two-thirds of the PMS participants showed abnormal CF but this was unrelated to disability level or to WF.
WF was present in half of the 298 cognitively impaired PMS patients, while presenting a high magnitude of deceleration over 6 minutes time (ie, DWI = −28.9%). It is hypothesized that the substantial magnitude of slowing down in the WF group can have impact on daily life activities. However, only small correlations were present between the DWI and free-living MVPA reflecting physical activity. The results on the prevalence of WF in our sample with predominantly EDSS 5 to 6, is comparable to the previous report of Leone et al. 15 The WF group included a higher proportion of PMS patients using an assistive device. Other studies showed that the prevalence of WF is lower in mildly disabled relapsing-remitting MS patients, as well as the magnitude of the DWI.16,17 An unexpected finding was the higher proportion of SPMS in the WF group. One could suggest that PPMS patients would be more disabled and thus more likely to have WF. In order to elucidate this result, we verified potential differences between pwMS with primary and secondary progressive phenotypes (results not shown, see Supplemental Results). SPMS patients had however longer disease duration and showed higher scores for the physical category of the MFIS and MSIS-29, lower number of correct answers in the SDMT and lower physical fitness (VO2 peak and peak watts). Although the current study uses the phenomenological classification of patients using secondary and primary progressive descriptors, it seems that WF is a symptom that is most frequent observed in more disabled patients. This fits the new framework including clinically and biological based definitions of MS progression, 1 potentially including WF as a clinical marker of disease progression.
This is the first study reporting on prevalence of CF and its magnitude in a large sample of PMS with impaired information processing speed. CF was present in two-thirds of the PMS sample with an average performance decline (“last 30” compared to the “first 30”) of −29.7% on the SDMT. Previously, in a mixed sample (ie, RRMS and PMS) values of approximately 25% was reported for the CFI using the PASAT 45 supporting the results of this study. CF has been recently studied in the MS population with various cognitive tests including PASAT and SDMT, but mostly often absolute scores per timespan (eg, 30”) were presented with comparison between groups instead of a percentual decline.23,45-48 Also, there is still a need to define discriminative validity by means of a validated cut-off value to discriminate normal versus abnormal CF. In the present study, the −10% cut-off was arbitrarily chosen to classify patients presenting CF, similar to the cut-off point of WF. There weren’t any differences between the CF and NCF in disease characteristics, or in any objective outcome measure. The only significant difference in PRO were found in the MSIS-physical and MFIS-psychosocial, with CF patients presenting lower scores, less MSIS-29 physical and MFIS psychosocial impact. One explanation could be that, for the MSIS-physical, despite the difference being small (ie, 5.1 points), 49 this may have been driven by the particularly better physical capacity for those with CF only (and not WF). However, we also have to point out that MFIS-psychosocial is based on items as doing things away from home and motivation to participate in social events. Considering that doing things away from home require some independence or would be easier if a person has less disability, as well as to participate in social events, the lower level of disability in those presenting only CF may have influenced this finding. There was no relationship between CF and cognitive impairment which supports the existing literature. 50 Future studies can also investigate the relationship in other cognitive domains than information processing speed only.
Finally, the present study examined the associations between WF and CF. No associations were found between the 2 domains of fatigability measured during a walking and an information processing speed task. In fact, 34% of the sample had a combination of both CF and WF, 17% showed WF only, 17% did not present any fatigability, and 32% was classified with CF only. As expected, those classified with both CF and WF were more disabled showing a moderate correlation (
This is the first study investigating fatigability in a sample including only people with PMS. Methodological considerations apply such as the representativeness of the sample, given that the population had to be cognitively impaired and physically activity, and the use of only 1 task per domain to quantify fatigability with particular methodological modalities. It is also acknowledged that the applied outcome measures for fatigability are not yet established as “golden standard,” and thus results need to be interpreted with some caution. Further investigations in the field are warranted on better understand the constructs of the measures. For example, the DWI is only documenting changes in gait speed while it is known that also changes can occur in the gait pattern or perceived effort. 51 Crucially, the findings regarding symptom associations within subgroups of fatigability (ie, only WF only, only CF, and both WF and CF) may be influenced by the classifications applied to distinct constructs and should be considered as preliminary evidence, given our current focus on presenting prevalence results specifically related to cognitive and WF.
Conclusions
Half of the PMS patients with cognitively impaired processing speed were identified with WF, and it was accompanied by significant reduced physical capacity captured by objective outcome measures (ie, cardiorespiratory and walking capacity) and also confirmed in the self-reported walking ability and self-reported physical fatigue. There was a high prevalence of CF but without any differences in clinical, cognitive, and physical functions between CF and NCF groups. However, established cut-off values for CF are needed. Cognitive and motor fatigability were not related.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241236161 – Supplemental material for Prevalence and Associated Clinical Characteristics of Walking-Related Motor, Cognitive, and Fatigability in Progressive Multiple Sclerosis: Baseline Results From the CogEx Study
Supplemental material, sj-docx-1-nnr-10.1177_15459683241236161 for Prevalence and Associated Clinical Characteristics of Walking-Related Motor, Cognitive, and Fatigability in Progressive Multiple Sclerosis: Baseline Results From the CogEx Study by Cintia Ramari, Mieke D’hooge, Ulrik Dalgas, Anthony Feinstein, Maria Pia Amato, Giampaolo Brichetto, Jeremy Chataway, Nancy D. Chiaravalloti, Gary R Cutter, John DeLuca, Rachel Farrell, Massimo Filippi, Jennifer Freeman, Matilde Inglese, Cecilia Meza, Robert W. Motl, Maria A Rocca, Brian M. Sandroff, Amber Salter, Daphne Kos and and Peter Feys in Neurorehabilitation and Neural Repair
Footnotes
Author Contributions
Cintia Ramari: Conceptualization; Data curation; Formal analysis; Visualization; Writing—original draft; Writing—review & editing.Mieke D’Hooge: Investigation; Project administration; Writing—review & editing. Ulrik Dalgas: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Visualization; Writing—review & editing. Anthony Feinstein: Conceptualization; Data curation; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Writing—review & editing. Maria Pia Amato: Funding acquisition; Investigation; Methodology; Project administration; Resources; Writing—review & editing. Giampaolo Brichetto: Funding acquisition; Investigation; Methodology; Project administration; Resources; Writing—review & editing. Jeremy Chataway: Data curation; Funding acquisition; Investigation; Methodology; Project administration; Resources; Writing—review & editing. Nancy D. Chiaravalloti: Funding acquisition; Investigation; Methodology; Project administration; Resources; Writing—review & editing. Gary R Cutter: Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Writing—review & editing. John DeLuca: Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Validation; Writing—review & editing. Rachel Farrell: Funding acquisition; Investigation; Methodology; Project administration; Resources; Writing—review & editing. Massimo Filippi: Funding acquisition; Investigation; Methodology; Project administration; Resources; Writing—review & editing. Jennifer Freeman: Funding acquisition; Investigation; Methodology; Project administration; Resources; Validation; Writing—review & editing. Matilde Inglese: Funding acquisition; Investigation; Methodology; Project administration; Resources; Writing—review & editing. Cecilia Meza: Funding acquisition; Investigation; Methodology; Project administration; Resources; Writing—review & editing. Robert Motl: Data curation; Funding acquisition; Investigation; Methodology; Project administration; Resources; Validation; Writing—review & editing. Maria A Rocca: Funding acquisition; Investigation; Methodology; Project administration; Resources; Validation; Writing—review & editing. Brian M. Sandroff: Data curation; Funding acquisition; Investigation; Methodology; Project administration; Resources; Validation; Visualization; Writing—review & editing. Amber Salter: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Software; Supervision; Validation; Visualization; Writing—review & editing. Daphne Kos: Investigation; Methodology; Project administration; Resources; Supervision; Validation; Writing—review & editing. Peter Feys: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant from the Multiple Sclerosis Society of Canada (grant number #EGID3185).
ORCID iDs
Supplementary material for this article is available on the
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
