Abstract
Background
Walking abnormalities in people with Parkinson’s disease (PD) are characterized by a shift in locomotor control from healthy automaticity to compensatory, executive control, mainly located in the prefrontal cortex (PFC). Although PFC activity during walking increases in people with PD, the time course of PFC activity during walking and its relationship to clinical or gait characteristics is unknown.
Objective
To identify the time course of PFC activity during walking in people with PD. To investigate whether clinical or gait variables would explain the PFC activity changes.
Methods
Thirty-eight people with PD tested OFF medication wore a portable, functional near-infrared spectroscopy (fNIRS) system to record relative PFC activity while walking. Wearable inertial sensors recorded spatiotemporal gait characteristics. Based on the PFC activity (fNIRS) in the late phase of the walking task (final 40 seconds), compared to the early phase (initial 40 seconds), participants were separated into 2 groups: reduced or sustained PFC activity.
Results
People with PD who reduced PFC activity during walking had less impaired gait (eg, faster gait speed) than those who had a sustained increase in PFC activity (P < .05). Cognitive set-shifting ability explained 18% of the PFC activation in the group with a sustained increase in PFC activity (P = .033).
Conclusions
The time course of reduction in PFC activity corresponds to less impaired gait performance in people with PD, while a sustained increase in PFC activity is related to worse cognitive flexibility. Reduction in PFC activity while walking may indicate a less impaired, automatic control of walking.
Keywords
Introduction
Gait automaticity refers to the ability of the central nervous system to control walking with minimal use of executive control that demands attention. 1 Walking difficulties are common in people with Parkinson’s disease (PD) and are associated with loss of independence, and concerns about falling 2 and falls. 3 People with PD have difficulty controlling automatic movements and thus shift to more voluntary control of gait. 4 This compensatory shift from automatic to voluntary gait is achieved by increased attentional demand concurrent with poor locomotor patterns.5-8 Automatic tasks recruit more subcortical regions (eg, basal ganglia and brainstem), whereas voluntary tasks depend more on cortical regions, including the prefrontal cortex (PFC).1,4 Specifically, the PFC represents conscious (voluntary, top-down) attention.1,4 In people with PD, studies5-9 have shown that gait leads to overactive conscious attentional activation mainly on the PFC to compensate for basal ganglia impairment that affects gait automaticity.
Functional near-infrared spectroscopy (fNIRS) studies provide direct evidence of the important role of the PFC during walking. fNIRS systems can be used to record changes in cortical oxygenated (HbO2) and deoxygenated hemoglobin (HHb) concentrations, which are used to infer cortical activity.5-8 Thus, the use of the fNIRS to record PFC activity during walking may provide direct measures of automaticity and executive control. Several studies have found increased PFC activity while walking in people with PD compared to age-matched, healthy control participants.9-11
Increased PFC activity is likely a compensatory mechanism of prefrontal executive control to maintain locomotion in people with PD9-11; However, as cognition is impacted in PD and specifically executive function, it is unknown whether people with PD who have impaired cognition can use this compensatory mechanism to main gait control. Changes in the time course of PFC activity during walking would provide this information since in healthy individuals, an initial, subtle, increase in PFC activity while walking attenuates quickly after a few steps. 12 Specifically, after a brief, initial increase in HbO2 at the beginning of the walking task, a decline in HbO2 will follow. 12 These results suggest a return to automatic control of gait following a quick initial attentional period. This phenomenon has not been investigated in PD, and it would be important to understand whether the initial increase in PFC is sustained for longer in people with PD and whether it is impacted by executive dysfunction in order to develop interventions to restore gait automaticity. Thus, we hypothesize that people with PD who have executive dysfunction would require a sustained increase in PFC activity during walking to compensate for worse gait performance (eg, slower gait). Instead, we predict that people with PD with better executive function will show less impaired gait performance (eg, faster gait). Increased PFC activity while walking is associated with worse executive function (Trail Making Test [TMT]) and worse gait automaticity in people with PD. 9 Thus, although PFC activity while walking would represent conscious attention and executive control, 13 a compensatory increase in PFC may also reflect abnormal attentional and executive demands during walking.
Gait variability is used as an indirect measure of gait automaticity in PD. 14 In our previous study, we showed that PFC activity may reveal changes among people with and without freezing of gait (FOG) of PD that are not detectable by gait variability. 7 Specifically, we found that between-group difference for PFC activity was only observed in the late phase of the walking (40 seconds bout of walking), which might relate to a sustained pattern of PFC activation throughout the walking task by people with PD and FOG (freezers). 7 Based on these previous findings, the current study is a secondary analysis to investigate the changes in the time course of the PFC activity during walking and the influence of FOG, cognitive status, and gait performance. To the best of our knowledge, no study has investigated the time course of PFC activity in people with PD. Frontal executive dysfunction has been implicated in FOG pathophysiology. 15 Thus, FOG and impaired executive function would be associated with an inability to decrease PFC activity during walking resulting in impaired speed and variability of walking (ie, impaired automaticity). Sustained PFC activity has been shown in poststroke patients throughout the walking task, 16 which indicates impaired automaticity.7,16
This study aimed to identify the time course of changes in PFC activity during a bout of walking (ie, late phase of the walking, final 40 seconds, compared to the early phase, initial 40 seconds). People with PD were separated into 2 groups, reduced or sustained PFC activity during walking based on the change in their PFC activity (fNIRS). In addition, we investigated whether clinical or gait variables would explain the time course of changes in PFC activity during a bout of walking.
Methods
Participants
Participants were recruited from local neurology clinics via referrals from movement disorder specialist neurologists. Thirty-eight people with idiopathic PD were included in the study. Participants were included if they were aged 55 to 90 years, had a diagnosis of idiopathic PD according to the UK Brain Bank Criteria, were taking anti-parkinsonian medication, gave informed consent to participate, and could cooperate with the testing. Exclusion criteria included inability to stand or walk independently for 2 minutes, nonparkinsonian factors affecting gait (eg, musculoskeletal disorders and vestibular problems), and inability to follow instructions. Freezers were identified by the New Freezing of Gait Questionnaire (NFoGQ ≥ 1: freezers and NFoGQ = 0: nonfreezers). 17 Study procedures were approved by the Oregon Health & Science University (OHSU) Institutional Review Board (eIRB #9903 and #17805), with written informed consent obtained before participation.
Experimental Procedures and Equipment
All participants were tested in their “Off” medication state, at least 12 hours after the last administration of their usual antiparkinsonian medications. Participants were assessed in 1 visit. We assessed the following variables: PD motor severity (Movement Disorders Society Unified Parkinson’s Disease Rating Scale [MDS-UPDRS, Part III]), 18 disease stage (Hoehn and Yahr Rating Scale), 19 cognitive function with the Montreal Cognitive Assessment (MoCA), 20 the Frontal Assessment Battery, 21 the Royall Clock Drawing Tasks (CLOX1 and CLOX2), 22 and the TMT part A and B. 23
Participants walked, at a self-selected comfortable pace, back and forth over a 9-m straight path, with a 180° turn at each end marked by lines on the floor. The walking test included an initial 20 seconds of quiet standing (baseline period) followed by 80 seconds of walking with turning (task period), and again 20 seconds of quiet standing at the end (final period). Participants performed a single trial for the walking test condition. No FOG episode occurred during the protocol. A portable 8-channel fNIRS system (OctaMon, Artinis Medical Systems) recorded changes in HbO2 and HHb bilaterally in the PFC at a sampling rate of 50 Hz. The fNIRS device consisted of 2 light detectors and 8 light emitters (continuous wave diodes with wavelengths of 760 and 850 nm). Three regular channels (interoptode distance of 35 mm) and 1 short-separation channel (interoptode distance of 15 mm) were used for each hemisphere. Optodes were placed on participants’ forehead using a headband with predetermined locations, according to the international 10 to 20 EEG system. This system is the most prevalent system for positioning electrodes 24 that describes scalp locations using relative distances between cranial landmarks with primary landmarks being the nasion, inion, and right and left preauricular points. A digitizer (Polhemus Patriot 3D digitizer) was used to provide 3-dimensional coordinates of anatomical references (nasion, inion, and right and left preauricular points) and positions of optodes.
Processing of fNIRS
Data from the digitizer were entered into the software package NIRS-SPM (http://www.nitrc.org/projects/nirs_spm). 25 The spatial registration routine was used to find the correspondence between the scalp location where the fNIRS measurement was performed and its underlying cortical surface where the source signal was located. 26 Cortical regions assessed included the frontal cortex Brodmann areas 9 (dorsolateral PFC) and 10 (antero-medial frontal cortex; Figure 1). There are a multitude of methods for processing fNIRS data, and the methods tend to be applied heterogeneously across different studies. 27 While there are efforts to standardize signal processing techniques, 28 differences in fNIRS equipment used and experimental paradigms make it difficult to establish a 1-size-fits-all approach. In our case, we chose processing techniques that are suited to our specific task and do not rely on subject-specific parameter selection, or ad hoc tuning. Optical density was automatically converted to hemoglobin concentration as data is exported from OxySoft (Artinis Medical Systems, Elst, The Netherlands). This is accomplished by applying the modified Beer-Lambert law; a DPF of 6 was used for all subjects. The exported hemoglobin concentration values were then processed with custom Python scripts. 29 First, a short-separation channel correction was applied to remove noise due to scalp blood-flow and interfering anatomy (hair, skull, and skin). 30 Briefly, this technique uses the signal from a short-separation channel to remove the noise from the superficial layer of the head. Next the data was corrected for motion artifacts, particularly important given the greater opportunity for such artifacts to arise during a gait task, when compared to a baseline condition (eg, standing still). We opted to use the Temporal Derivative Distribution Repair method 31 as it does not require user-supplied parameters and shows improve artifact removal, when compared to other popular methods. The fNIRS signals were then filtered to remove physiological and equipment noise. We used a bandpass filter (FIR, 1000th order, Fc,low = 0.01, Fc,high = 0.1), 27 which attenuates the heart beat (>1 Hz), respiration (0.2-0.5 Hz), and Mayer waves (0.1 Hz). Finally, we subtracted the mean of the fNIRS signal during the quiet stance period at beginning of each trial from the rest of the signal, as continuous-wave fNIRS measurements are always relative and need to be adjusted by some baseline value.
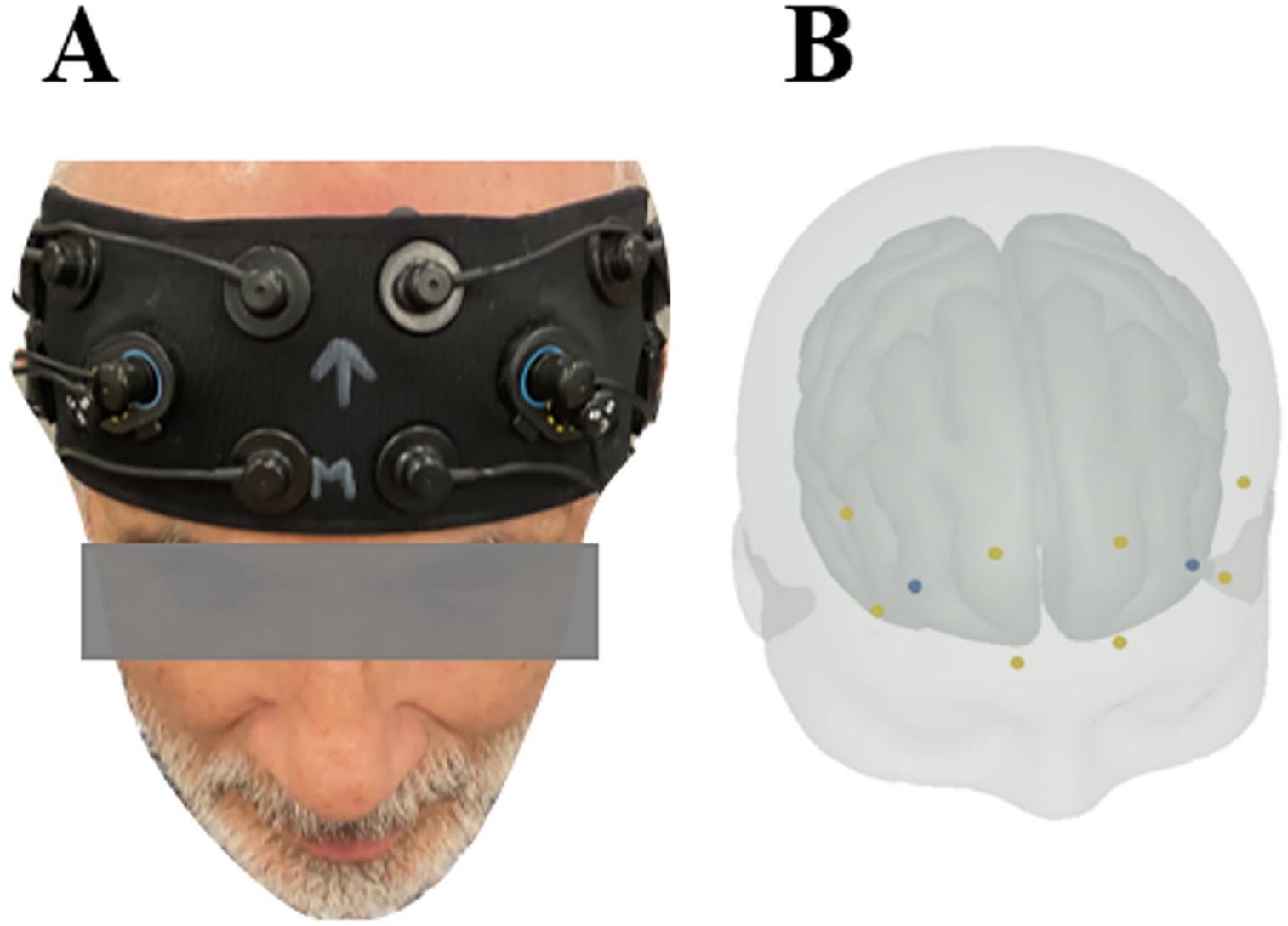
(A) A portable 8-channel fNIRS scalp (OctaMon, Artinis Medical Systems). (B) Optodes on the forehead template with predetermined locations, according to the international 10 to 20 EEG system, where Brodmann areas 9 (dorsolateral prefrontal cortex) and 10 (antero-medial frontal cortex) were assessed.
Definition of Reduced and Sustained PFC Activity While Walking
Baseline correction (relative changes) was performed by subtracting the baseline period (20 s of baseline standing still fNIRS signal) from the early and late phases of walking. Then, relative changes in HbO2 and HHb concentrations were calculated for both the early (median of the first half of the task = initial 40 seconds) and late (median of the second half of the task = final 40 seconds) periods of the walking task, considering straight walk and turns together. Reduced PFC activity while walking was defined as a negative value when subtracting the early phase from the late phase, and confirmed with a visual inspection. Only individuals who had negative values between the phases late and early and presented a PFC activity behavior as demonstrated in Figure 2A, were included in the reduced PFC activity group. Individuals who had positive values when subtracting their PFC activity during the final 40 seconds of walking from the initial 40 seconds and presented a PFC activity as shown in Figure 2B, were included in the sustained PFC activity group.
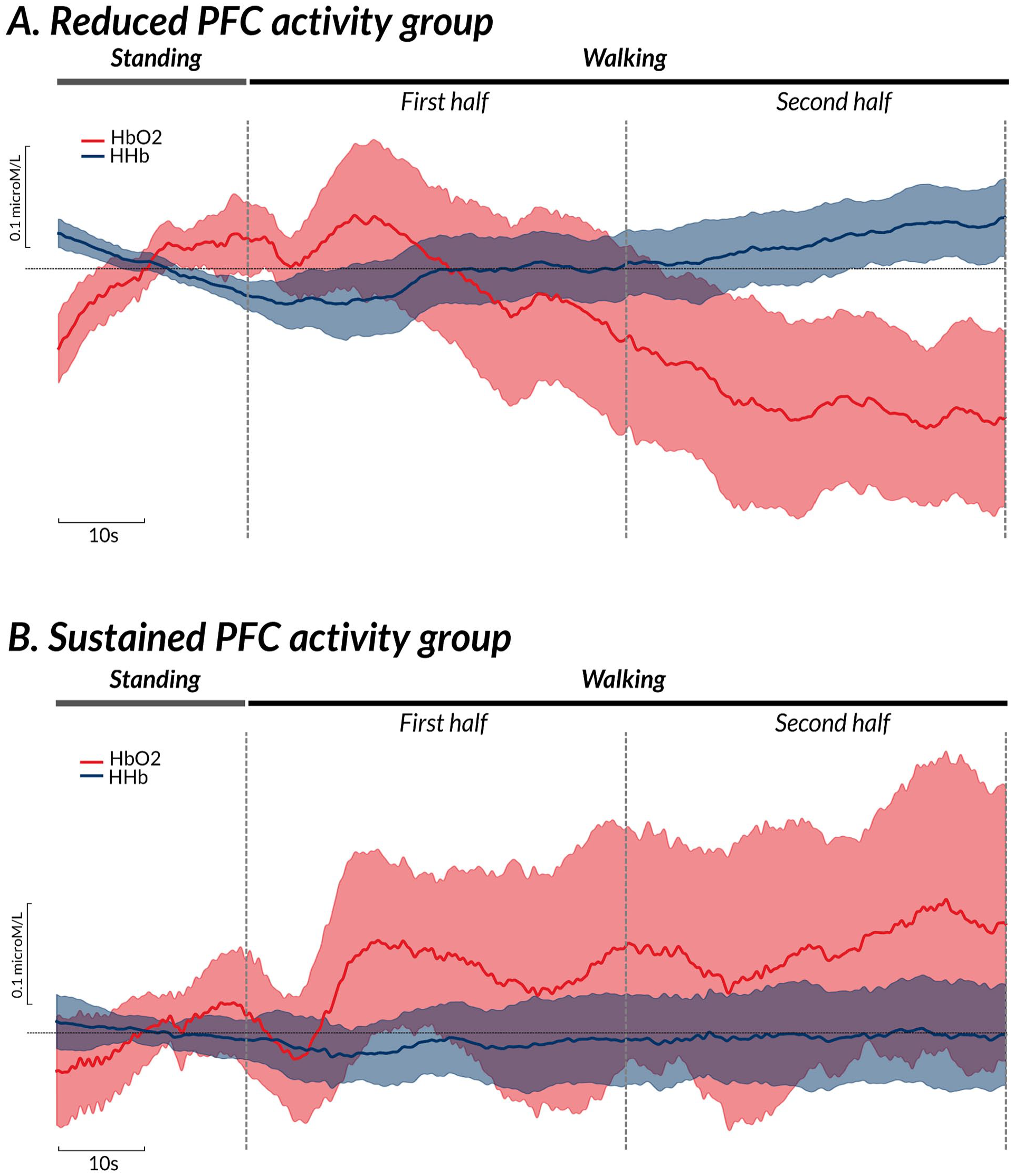
Mean of HbO2 (red bold line) and HHb (blue bold line) signals during 20-seconds standing still and 80-seconds walking task in people with Parkinson’s disease in the reduced (A) and sustained (B) prefrontal cortex activity. The shadow is 95% confidence interval.
Gait Characteristics
Spatiotemporal gait characteristics were calculated from the inertial sensors using the Mobility Lab software, V2 (APDM). 32 Two inertial measurement units (Opal, APDM) were used to quantify spatiotemporal gait parameters at a sampling rate of 128 Hz. They were located at the both feet of participants. Each inertial sensor consisted of triaxial accelerometers, gyroscopes, and magnetometers, and was securely fixed to the participant’s body with Velcro straps. The inertial sensors and fNIRS system were synchronized through the Artinis PortaSync. All recorded steps corresponding to walking were included in the analysis. Gait measures derived from the Mobility Lab software were processed using manufacturer-provided software, and finally averaged values were exported for further analysis. The following gait measures were calculated: mean gait speed (m/s), mean stride length (m), mean foot-strike angle (°), and mean stride time variability (%).
Statistical Analysis
Since this study is a secondary analysis of our previous research, 7 no power analysis was performed. Independent t-tests were used to investigate differences between the groups (reduced and sustained PFC activity) for demographics characteristics, clinical variables, relative HbO2 and HHb late–early phases, and gait metrics (gait speed, stride length, and foot strike angle). We also performed 2 linear multiple regression (Procedures for Regression of SAS 9.2 software) using the stepwise method for each group (reduced and sustained PFC activity) having the relative HbO2 and HHb late–early as the dependent variable. To explain the variance of the dependent variable in the regression model, we used all variables described in Table 1 as well as gait metrics (gait speed, stride length, foot strike angle, and stride time variability) as independent variables. To avoid collinearity, we included the independent variables in the linear multiple regression analysis if they presented a P ≤ .05 and a correlation lower than 0.7 between them. 33 The significance level for these analyses was set at P ≤ 0.05. Finally, Effect Sizes (ES) and confidence intervals (CI) were calculated for between-group comparisons. ES were classified as small (ES = 0.20-0.49), medium (ES = 0.50-0.79), and large (ES ≥ 0.80). The 95% CI of the ES were calculated using a non-central t distribution, thus, positive and negative CI [ie, not crossing zero (0)] were considered as significant.
Characteristics of the Individuals.
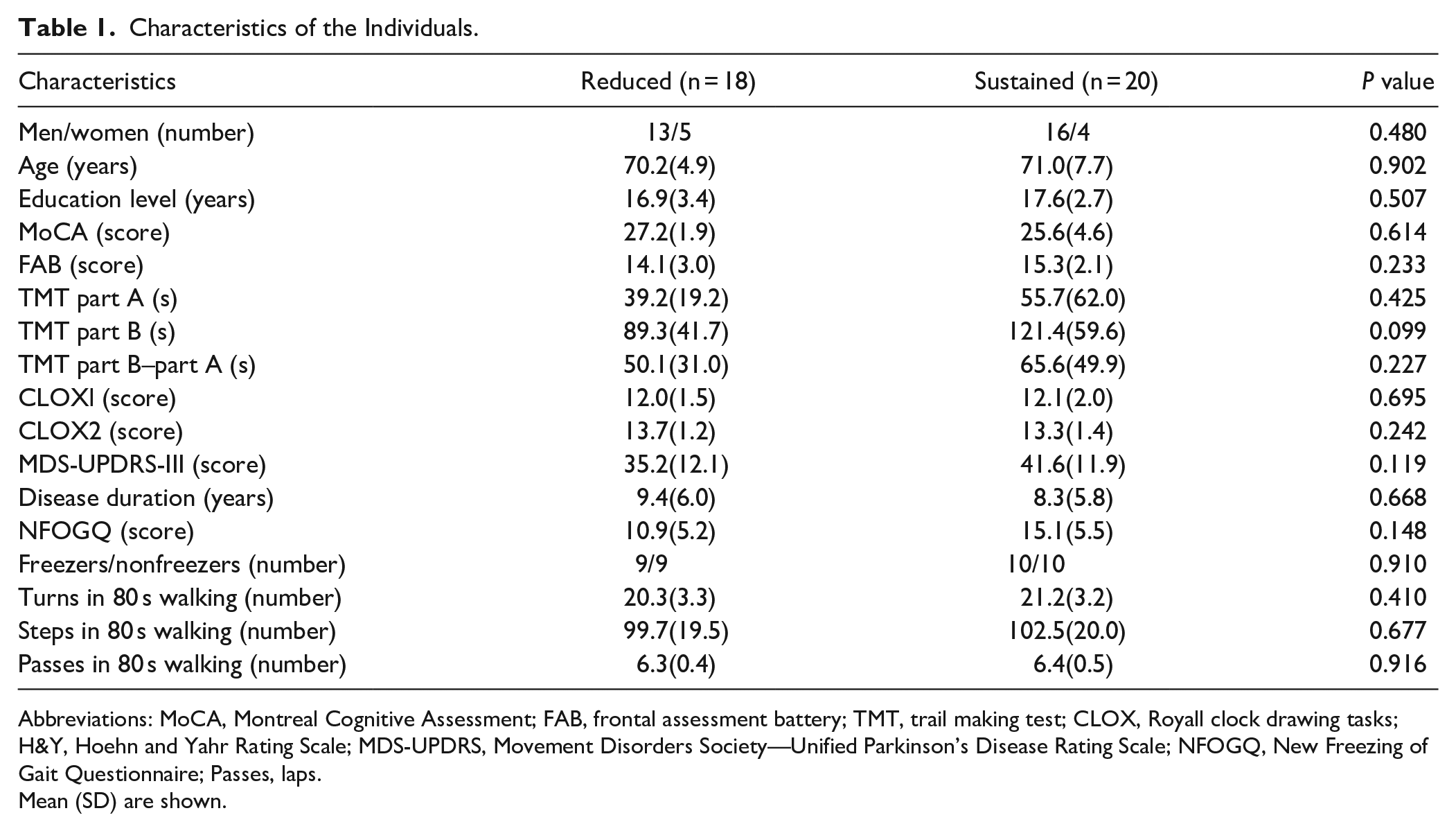
Abbreviations: MoCA, Montreal Cognitive Assessment; FAB, frontal assessment battery; TMT, trail making test; CLOX, Royall clock drawing tasks; H&Y, Hoehn and Yahr Rating Scale; MDS-UPDRS, Movement Disorders Society—Unified Parkinson’s Disease Rating Scale; NFOGQ, New Freezing of Gait Questionnaire; Passes, laps.
Mean (SD) are shown.
Results
Participants
All demographic characteristics and clinical scale scores are reported in Table 1, which shows no difference between the groups (reduced and sustained PFC activity).
People With PD Who Have Reduced PFC activity While Walking Have Faster Gait Than Those Who Have a Sustained Increase in PFC Activity
Reduced PFC activity in the late phase during a walking bout was smaller than the early phase (relative HbO2 late–early = −0.12 µmol/L and relative HHb late–early = 0.04 µmol/L) in 18 people with PD regardless of FOG status. In contrast, a sustained increase in PFC activity in the late phase, compared to the early phase (relative HbO2 late–early = 0.10 µmol/L and relative HHb late–early = 0.02 µmol/L) was observed in 20 people with PD. Figure 3 shows the between-groups difference for relative HbO2 late–early (P < .0001, ES = −1.81, 95% CI = −2.56 to −1.06) and relative HHb late–early (P = .0355, ES = 0.52, 95% CI = 0.01 to 1.05).
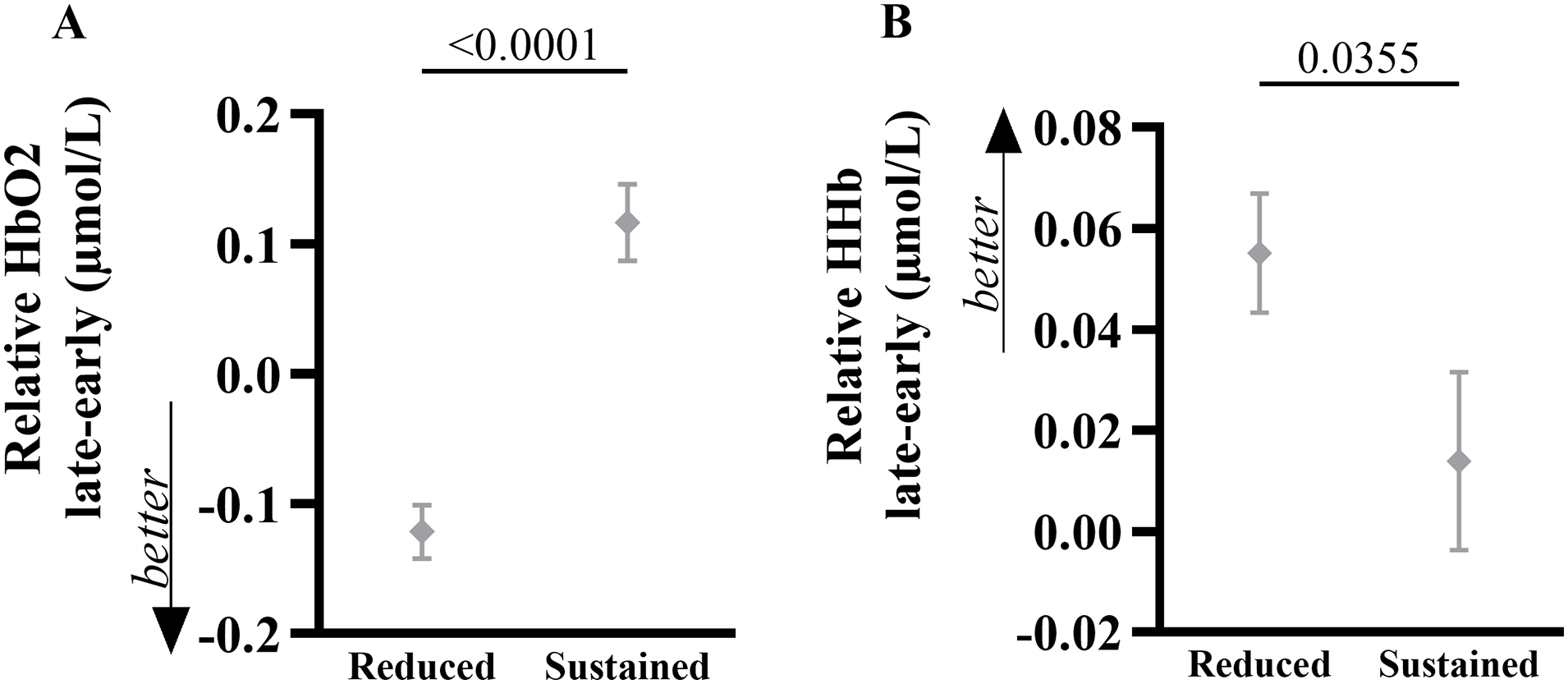
Mean (SD) of the relative HbO2 (A) and HHb (B) late–early for both groups (reduced and sustained prefrontal cortex activity). P value is shown.
The reduced PFC activity group showed faster gait speed (P = .015, ES = 0.68, 95% CI = 0.05 to 1.32), bigger stride length (P = .002, ES = 0.76, 95% CI = 0.12 to 1.41), bigger foot-strike angle (P = .002, ES = 0.82, 95% CI = 0.21 to 1.43), and a strong trend toward lower stride time variability (P = .073, ES = −0.42, 95% CI = −1.02 to 0.18) than those of the sustained PFC activity group (Figure 4).
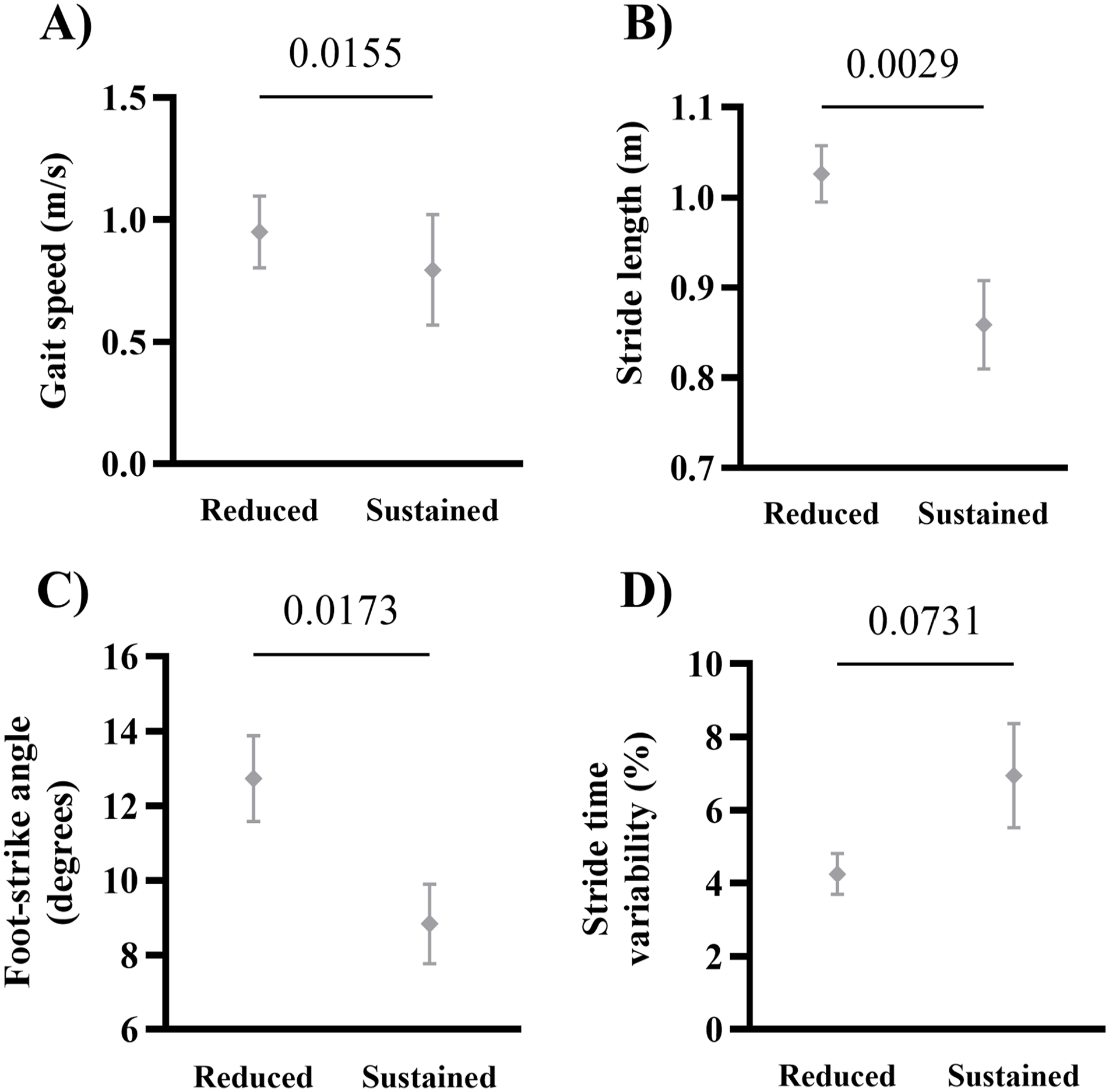
Mean (SD) of the gait speed (A), stride length (B), foot-strike angle (C), and stride time variability (D) for both groups (reduced and sustained prefrontal cortex activity). P values are shown.
Set-Shifting Partially Explains the PFC Activity Behavior in the Sustained PFC Activity Group
We performed linear multiple regression analysis for both groups using all variables described in Table 1, as well as gait metrics into regression model. We used these analyses for each group separately as we wanted to know which variables could explain reduced and sustained PFC activity. Finally, when we did a multiple regression with the whole group, no variable entered into the model to explain reduced PFC activity, then, we did not include this analysis. Only the sustained PFC activity group presented an association between dependent and independent variables. We observed that the MoCA score (r = .48, P = .016) and TMT part B (r = −.52, P = .009) were associated with the relative HbO2 late–early. These variables entered the linear multiple regression model; however, only the TMT part B significantly (P = .033) explained 18% (adjusted R2 = .18) of the relative HbO2 late–early in the sustained PFC activity group. No association was observed between relative HHb late–early and any variable.
Figure 5 illustrates the relationship of the difference between the relative HbO2 late–early with the TMT part B in the sustained PFC activity group. Visually, 2 data points on the left side of the correlation line in Figure 5 seem to affect the slope of the regression. However, even when we removed those 2 data points, a significant correlation still was observed between the relative HbO2 late–early with the TMT part B (r = −.49, P = .019).
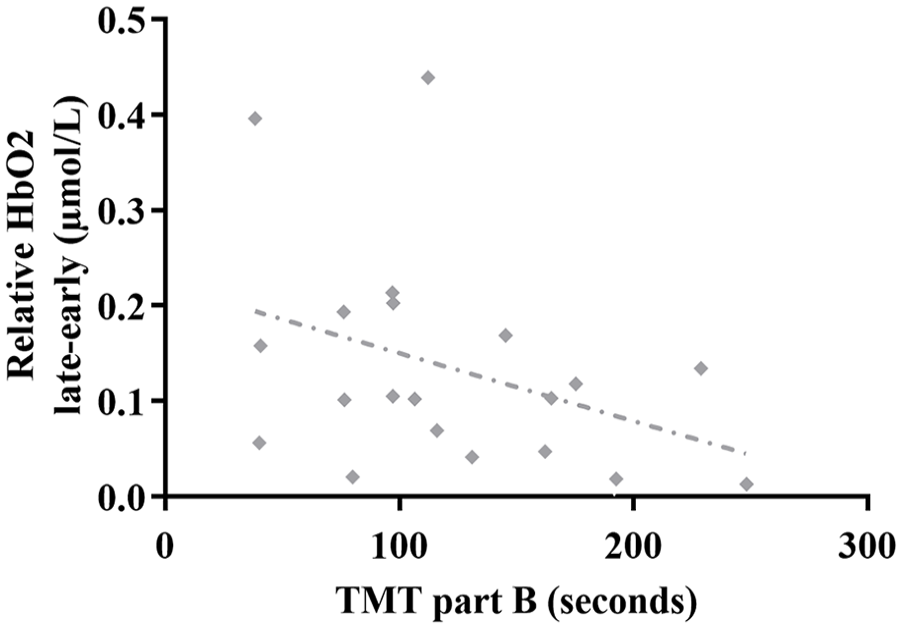
Correlation of the relative HbO2 late–early phases during a walking bout with the TMT part B in the sustained PFC activity group.
Discussion
This is the first study to show the time course of changes in PFC activity while walking in people with PD. The time course of reduced PFC activity during a bout of walking corresponded to less impaired gait performance (eg, faster gait speed and bigger stride length and foot-strike angle) in people with PD, while sustained PFC activity corresponded to more impaired gait performance (eg, slower gait speed and reduced stride length and foot-strike angle). In addition, set-shifting was the only variable that explained 18% of sustained PFC activity (P = .033).
Increased PFC activity is likely a compensatory mechanism of prefrontal executive control to maintain locomotion in people with PD.9-11 Previous studies have demonstrated that people with PD have overall larger PFC activity and gait impairments during single-task walking compared to healthy controls.9-11 In our previous study, 7 the higher PFC activity reported may reveal changes between freezers and nonfreezers that are not detectable by gait variability (a surrogate measure for gait automaticity); the PFC difference might be related to a more voluntary control of gait in the freezer group. The current study looked at the time course of changes in PFC activity during a walking task. It showed that those individuals with PD who decreased PFC activity during a bout of walking had faster gait speed and larger stride length and foot strike angle, regardless of FOG status. These results indicate that gait automaticity is impaired in people with PD who have sustained PFC activity in the late phase of walking. On the other hand, reduced PFC activity in the late phase of walking may suggest a gradual transition to more automatic control of walking. Thus, we hypothesize that a more preserved ability to walk using less attentional demand would be due to automatic resources available in basal ganglia and brainstem.
Locomotion is controlled by multiple neural systems hierarchically distributed throughout the central nervous system including the spinal cord, brainstem, cerebellum, basal ganglia, and motor cortex. 34 A previous study demonstrated that older adults with a faster gait speed have a greater basal ganglia functional connectivity than those with a worse gait speed who have lower basal ganglia functional connectivity. 35 Another study showed that walking capacity was associated with greater functional connectivity between the mesencephalic locomotor region with the anterior cingulate cortex and the paravermal cerebellum in older adults. 36 On the other hand, people with PD who have sustained PFC activity in the late phase of walking may have gait impairments associated with decreased automatic resources that require basal ganglia and brainstem control, thus, compensating with sustained PFC activity during walking. Increased PFC activity during poor walking would indicate less available resources in subcortical and brainstem areas. Since we can distinguish individuals who have gait impairments associated with sustained PFC activity during walking, we may be able to more effectively personalize treatments to restore gait automaticity in people with PD, regardless of FOG status.
We observed that set-shifting (TMT part B scores) explained 18% of the increased PFC activity in people with PD. Although 18% is a small percentage, it may indicate that impairments in cognitive flexibility is somewhat associated with the sustained or increased PFC activity during walking in people with PD who have a more impaired gait pattern. Cognitive flexibility is part of the executive subdomain of set-shifting, 37 which is controlled by the frontal lobe and associated areas. 38 Cognitive flexibility is required for adapting behavior to external influences,39,40 such as walking that requires accelerations and decelerations, turns, coordination of upper and lower limbs, and attention. A previous study showed that faster gait speed is strongly correlated with better cognitive flexibility (TMT part B minus A) in older adults. 41 Similarly, our data showed that people with PD who have a less impaired gait patterns showed somewhat better cognitive flexibility (Table 1), and consequently decreased PFC activity during the late phase of the walking task since they can adapt their locomotion patterns during walking phases (eg, acceleration and deceleration, turns and attention). Table 1 shows a trend (P = .09) toward higher TMT part B scores (worse) for the sustained PFC activity group, which suggests this group may have worse cognitive flexibility compared to the reduced PFC activity group. However, a negative association between higher PFC activity and faster TMT part B scores (Figure 5) for the sustained PFC activity was found. Although this seems counterintuitive, it may indicate that, among those people with PD who have poor cognitive flexibility, the ones who have better performance in the TMT part B (better cognitive flexibility) may be able to increase PFC to compensate for poor automaticity but resulting in worse gait pattern (Figure 4). On the other hand, those with worse cognitive flexibility may be unable to increase PFC activity as a compensatory mechanism. PFC has dense connections to the subcortical nuclei and the cerebellum,42,43 thus, people with PD may rely upon frontal areas while walking when OFF medication, but levodopa could facilitate recruiting of the cerebellum and ameliorate gait automaticity in fMRI studies. 14 Based on these studies and our findings (OFF medication), a lower PFC activity while walking could reflect decreased compensatory PFC activity related to better gait and executive function (a strong trend for the TMT part B scores) in the reduced PFC activity group compared to the sustained PFC activity group. In people with PD, better executive function may require a lower demand for PFC activity during walking, but future studies should test this hypothesis.
We previously demonstrated that freezers have higher PFC activity while walking and worse gait patterns compared to nonfreezers. 7 Interestingly, in the present study, we had a similar number of freezers within each group (Table 1). In a sub-analysis where we compared freezers and nonfreezers within each group, we found no difference between them, which suggests that the freezers in the reduced PFC activity group who have a less impaired gait pattern use less PFC activity during the late phase of the walking bout. Thus, future studies should investigate if improving gait patterns in freezers would prevent them to overload the compensatory executive control (PFC activity) during walking.
Key Study Limitation and Strength
This study has limitations. First, we do not have a healthy control group to verify if the time course of sustained PFC activity in the late phase of walking is specific for PD. Second, since we have observed acceptable reproducibility in walking and turning trials with fNIRS, 44 we have used only 1 walking trial in the current study. As the literature shows, gait is less variable when walking continuously compared to performing multiple short intermittent walks 45 and based on that literature, we believe this protocol better addresses gait spatiotemporal measures and PFC while walking than short, intermittent walks, although we know it could be a limitation. The strength of our study is the robust data analysis methods for fNIRS processing, especially the use of short-separation channels to remove the superficial hemodynamic response from the fNIRS signal. This approach reduces the likelihood of false positive results. 46 Another strength of our study is that the people with PD were assessed in OFF medication that demonstrates the true state of disease and reduces the possible effects of confounders associated with levodopa intake, as previous studies showed that levodopa improves spatiotemporal gait patterns, 47 increases the connectivity of the basal ganglia to the cortex, 48 while increases 8 or reduces PFC activity during walking. 6
Clinical Implications
A previous study showed that individuals with PD, including those who have FOG, can recruit the right cerebellum area which is associated with gait automaticity (step time variability) in the dopaminergic ON state when performing a fMRI protocol that simulates walking (virtual reality gait), whereas in the OFF state during worse gait automaticity they depend on cognitive control (eg, orbitofrontal cortex). 14 In the current study, the individuals were assessed in OFF medication during mobile imaging (fNIRS) and showed, for the first time, that even in the OFF medication state, some people with PD who have a less impaired gait have also reduced PFC activity during walking. Our data suggest that it may be possible to identify individuals who have gait impairments and executive dysfunction associated with sustained PFC activity during walking and then provide more effectively personalized treatments. Individuals with impairments in both gait and cognition and sustained PFC activity during walking may benefit from a different treatment approach than those without substantial impairments in both gait and cognition and reduced PFC activity during walking. For example, although levodopa decreases PFC activity during walking compared to OFF medication, 6 optimizing the treatment with levodopa and donepezil for people with PD who have impairments in gait and cognition would be an alternative, as people with PD stand to benefit most from this treatment than levodopa alone. 6 Moreover, sustained PFC activity may distinguish future fallers from non-fallers of PD since higher levels of PFC activity during walking predict falls in older adults, even after controlling for multiple confounders (eg, cognitive status, slow gait, and previous falls). 49 Finally, sustained PFC activity may distinguish people with PD with impairments in set-shifting and gait patterns from people without substantial impairments in set-shifting and gait patterns.
Conclusions
People with PD who have reduced PFC activity in the late phase of walking have a less impaired gait pattern than those who have sustained PFC activity. The sustained PFC activity during walking is partly related to poor cognitive flexibility. Thus, reduced PFC activity while walking may indicate a less impaired, automatic control of walking in people with PD.
Footnotes
Acknowledgements
We thank our participants for generously donating their time to participate.
Author Contributions
Carla Silva-Batista: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing. William Liu: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing. Rodrigo Vitório: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing. Samuel Stuart: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing. Joseph F. Quinn: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing. Martina Mancini: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by grants from the National Institutes of Health Career Development Award R00 HD078492-04 (PI Mancini) and R01 HD110389 (PI Mancini), an OHSU Center Pilot Grant Award (PI Mancini).
