Abstract
Background
Gait disturbances are exacerbated in people with Parkinson’s disease (PD) during dual-task walking (DTW). Transcranial direct current stimulation (tDCS) has been shown to exert beneficial effects on gait performance and cortical excitability in PD; however, its combined effects with treadmill training (TT) remain undetermined.
Objective
To investigate the effects of tDCS followed by TT on DTW performance and cortical excitability in individuals with PD.
Methods
Thirty-four PD participants were randomized to dorsal lateral prefrontal cortex (DLPFC) tDCS and TT group (DLPFC tDCS + TT group) or sham tDCS and TT group (sham tDCS + TT group) for 50 minutes per session (20 minutes tDCS followed by 30 minutes TT), 12 sessions within 5 weeks (2-3 sessions each week). Outcome measures included cognitive dual-task walking (CDTW), motor dual-task walking (MDTW), usual walking performance, cortical excitability, functional mobility, cognitive function, and quality of life.
Results
The DLPFC tDCS + TT group exerted significantly greater improvement in CDTW velocity (P = .046), cadence (P = .043), and stride time (P = .041) compared to sham tDCS + TT group. In addition, DLPFC tDCS + TT group demonstrated a significant increase in resting motor threshold of stimulated hemisphere compared with sham tDCS + TT group (P = .026). However, no significant differences between groups were found in MDTW performance and other outcomes.
Conclusion
Twelve-session DLPFC tDCS followed by TT significantly improved CDTW performance and decreased cortical excitability more than TT alone in individuals with PD. Applying DLPFC tDCS prior to TT could be suggested for gait rehabilitation in individuals with PD.
Clinical trial registration number:
Australian New Zealand Clinical Trials Registry ACTRN12622000101785
Keywords
Introduction
Gait disturbance is one of hallmarks, including decreased gait velocity, shorter stride length, and increased cadence in people with Parkinson’s disease (PD). Performing another task while walking, namely dual-task walking (DTW), is an essential part of daily activities. According to the capacity-sharing model, cognitive resource is shared between tasks when performing dual-tasking, and such sharing depends on task complexity and importance. 1 However, when the available cognitive resource is limited or exceeds the capacity, the task performance is thus decreased. People with PD tend to increase their reliance on cognitive resource during DTW due to gait automaticity deficits. Therefore, performing the secondary task may further exacerbate gait impairments. Regardless of the usual walking performance or the type of concurrent task, a slower velocity during DTW has been reported in individuals with PD. 2 Furthermore, velocity during DTW is associated with executive dysfunction, depression, physical fatigue, and poor balance control in persons with PD, 3 leading to increased fall risks and reduced independence and quality of life.4,5
It has been suggested that abnormal plasticity within M1 reflects a loss of coordination between the basal ganglia and cortical inputs, leading motor impairments in PD. 6 Based on transcranial magnetic stimulation (TMS) data on lower limb motor area, enhanced corticospinal excitability was shown in people with PD. 7 Both corticospinal inhibition and intracortical inhibition have been reported to be decreased in people with PD.8,9 Moreover, previous studies have shown that TMS parameters are related to gait performance in people with PD, suggesting that the corticomotor system may be hyperexcited in order to compensate for the impaired basal ganglia.10-12 Therefore, TMS data is considered to serve as a neurophysiological measure for evaluating motor recovery in individuals with PD, offering insights into cortical excitability and motor impairments. 13
Dorsal lateral prefrontal cortex (DLPFC) plays a critical role in executive functions, including mediating the cognitive process involved in dual-tasking. A previous review highlighted that people with DLPFC lesions exhibit poor motor planning and executive functions and show increased distractibility. 14 Moreover, it has been found that slower gait velocity is associated with DLPFC atrophy in older adults. 15 These findings suggest that DLPFC impairments may contribute to decreased velocity during DTW. In people with PD, greater DLPFC activations were found during DTW compared with usual walking, indicating that they increase reliance on prefrontal cognitive resources.16,17 Liu et al further noted that higher DLPFC activations during DTW correlated with the worse DTW performance, including slower velocity and shorter stride length in individuals with PD. 18 Therefore, enhanced DLPFC efficiency seems to be a feasible approach to improve DTW performance in people with PD. 19 Thus, attempts have been made to stimulate DLPFC regions using neuromodulation techniques to improve DTW in individuals with PD. 20
Transcranial direct current stimulation (tDCS) is a non-invasive brain stimulation technique that alters cortical excitability and is believed as a promising intervention to improve gait performance in people with PD. 21 Previous studies have shown that anodal tDCS over the DLPFC improved functional mobility in individuals with PD and older adults with functional limitations, suggesting that anodal tDCS over the DLPFC may enhance the prefrontal neural efficiency, thereby improving walking ability.22,23 Our recent study revealed that a single session of anodal tDCS on the DLPFC improved DTW and modulated the cortical excitability in people with PD. 24 However, adverse effects of cathodal tDCS on executive function which is important for DTW, was noted in healthy adults. 25 On the other hand, previous studies have shown that anodal tDCS combined with physical training can be more effective in improving walking velocity and balance performance as compared with physical training alone in people with PD.26,27 Among physical training programs, treadmill training (TT) has been suggested as an effective and widely used intervention in clinical intervention for improving gait performance in individuals with PD. 28 However, it is not known the effects of combining DLPFC tDCS and TT on DTW performance. Furthermore, the possible neural changes for such effects should also be explored in people with PD. Therefore, the purpose of this study was to investigate the effects of tDCS followed by TT on DTW performance and cortical excitability in people with PD. We hypothesized that DLPFC tDCS and TT would exert more effects on DTW performance and cortical excitability changes as compared with TT alone in people with PD.
Methods
Subjects
The study was approved by the ethics committees of Taipei Veterans General Hospital and National Yang Ming Chiao Tung University. This trial was registered at the Australian New Zealand Clinical Trials Registry (ACTRN12622000101785 on 24/01/2022) and conformed to the Consolidated Standards of Reporting Trials checklist. All the data were collected at the Taipei Veterans General Hospital. The inclusion criteria were: idiopathic PD confirmed by the neurologist, aged 50 to 80 years, ability to walk independently for at least 10 m without using walking aids, and a score of ≥24 on the Mini-Mental State Examination (MMSE). The exclusion criteria were: any contraindication to tDCS or TMS (eg, skin lesions on sites for stimulation, metal implants, or pacemakers); neurological or orthopedic diseases that would affect participating the study; past or current history of non-superficial tumors or malignant tumors; or participants who attended other neurorehabilitation programs within 3 months. All participants provided signed informed consent before participation. All procedures were conducted during the “ON” medication state, which was 1 to 2 hours after taking the medication. The pre- and post-outcomes were measured at the same time of day for each participant.
Experimental Design
This study was a double-blinded (assessor and participant blinded), randomized, controlled trial with pre- and post-measurements. After baseline assessment, a researcher who was not involved in this study selected sealed envelope to assign participants to DLPFC tDCS and TT group (DLPFC tDCS + TT group) or sham tDCS and TT group (sham tDCS + TT group). The participants were blinded to their group assignment. Participants in the DLPFC + TT group received 20 minutes of DLPFC tDCS, followed by 30 minutes of TT. Participants in the sham tDCS + TT group received sham tDCS stimulation followed by 30 minutes of TT. The total 50-minute training program was administered 2 to 3 sessions per week in 5 weeks for a total of 12 sessions by the same physical therapist.
Intervention
tDCS
The DLPFC tDCS protocol (electrode size, intensity, duration, and placement) used in present study was according to our previous study, which showed one session of such tDCS protocol improved DTW in individuals with PD. 24 The tDCS was delivered by a current stimulator (Eldith DC Stimulator, NeuroConn, Germany) through a pair of saline-soaked sponge electrodes (7 cm × 5 cm). The stimulation intensity was set at 2 mA with a current density of 0.07 C/cm2, which is well below the threshold for tissue damage. 29 For DLPFC tDCS, the anode electrode and the cathode electrode were respectively placed over the DLPFC (F3) of dominant hemisphere and the contralateral supraorbital area with the current delivered for 20 minutes. 24 In sham tDCS, the electrodes placements were the same as described for DLPFC tDCS above. However, the current was only delivered for 60 seconds (ramp-up and ramp-down of 30 seconds), and 0 mA for the remainder of the period.
Treadmill Training
Immediately following tDCS (DLPFC tDCS or sham tDCS), participants in both groups received 30 minutes of TT (Biodex, Shirley, New York). It was suggested that tDCS should be delivered prior to motor training, rather than during the training, to prime cortical excitability for the subsequent motor training, thereby optimizing motor learning processes. 30 Mishra et al’s study showed that delivering DLPFC tDCS during walking task did not improve functional mobility, supporting that concurrent tDCS with motor training may interfere the modulatory effects of tDCS. 31 Therefore, DLPFC tDCS followed by TT was designed to enhance the possible motor learning in the present study. The treadmill velocity began at individual’s comfortable walking velocity, and was increased by an increment of 0.2 km/h every 5 minutes as tolerated. The criteria for increasing velocity was based on participant’s perceived exertion of “somewhat hard” or lower (ie, a Borg rating of perceived exertion <13). 32 The physical therapist encouraged the participants verbally to walk with a large stride, symmetry, and upright posture as possible.
Outcome Measures
The primary outcome in this study was DTW velocity. The secondary outcomes included temporal–spatial gait parameters during DTW and usual walking, cortical excitability, cognitive function, functional mobility, and quality of life.
To assess the DTW performance, including cognitive dual-task walking (CDTW) and motor dual-task walking (MDTW), the GAITRite system (CIR system, Inc., Havertown, Pennsylvania) was used. The CDTW was walking while performing serial subtracting by 3, starting from a random 3-digit number. The MDTW was walking while holding a tray with a glass of water. Each DTW was repeated 3 times with a 60-second rest between trials. The average of the 3 trials was used for data analysis. Gait parameters of interest were velocity, cadence, stride length, stride time, and dual-task cost (DTC). The DTC was calculated to indicate the dual-task interference. The formula of DTC is (DTW velocity − usual walking velocity)/usual walking velocity × 100%. 33
Cortical excitability of the tibialis anterior (TA) muscle was measured using a TMS (Magstim 200 magnetic stimulator; Magstim Company, Whiteland, Dyfed, UK). The resting motor threshold (RMT) and motor evoked potentials (MEP) indicate the corticospinal excitability of the M1. Additionally, the silent period (SP) duration and short-interval intracortical inhibition (SICI) were measured to indicate cortical inhibition. Each parameter, we employed 10 trials and averaged for the report data. The MEPs of the TA muscle were recorded using a Nicolet Viking EDX EMG system (Natus Neurology Inc./Nicolet Biomedical Inc., USA). The EMG electrodes were placed on the TA muscle belly and the ground was placed over the patella. TMS stimuli were delivered through a double-cone coil (diameter 110 mm) placed on the M1, with the participants lying supine comfortably wearing a fitted cap marked with a coordinate system (distance, 1 cm). To target the motor cortex presentation of the TA muscle, the coil was positioned 1 cm posterior to the vertex (Cz) and 1 to 2 cm laterally to either the left or right side to stimulate the corresponding motor area. 34 TMS coil was moved over the scalp in 1 cm steps to determine the optimal scalp location (hot spot). After identifying the optimal location on the scalp, the contour of the coil was marked to ensure consistent placement throughout the experiment. The assessor constantly monitored the position of coil to maintain consistency. The RMT was determined as the lowest stimulus intensity required to elicit MEPs with peak-to-peak amplitude greater than 0.05-mV in at least 5 of 10 successive stimuli. 35 RMT was expressed as a percentage of the maximum stimulator output. The SP duration was determined during isometric contraction of the TA muscle. Ten pulses were applied at an intensity of 120% RMT as the participant contracted TA muscles at a maximum of 20% voluntary contraction. The contraction level was maintained using visual feedback from EMG signals displayed on a computer monitor. The SP duration was determined from MEP onset to the recurrence of at least 50% of EMG background activity. 36 The SICI was evaluated using paired pulses during the TA muscle is relaxed. The conditioning stimulus and the testing stimulus were set at 80% and 120% RMT respectively with a short inter-stimulus interval (2 ms). SICI was expressed as the percentage of inhibition using the following formula: 100 − (conditioned MEP/unconditioned MEP) × 100. 37
To assess usual walking performance, participants walked with their comfortable speed without additional task for 3 times. The gait parameters of interest including velocity, cadence, stride length, and stride time, were measured by the GAITRite system. The Timed up and go test (TUG) was used for functional mobility. 38 We recorded the time taken to rise from a chair, walk 3 m, turn around, walk back to the chair, and sit down.
Cognitive function was measured using Trail Making Test (TMT) A and Chinese version of TMT-B which is a reliable and valid instrument with good test-retest reliability. 39 TMT-A evaluates attention and processing speed, and TMT-B mainly assesses cognitive flexibility. The TMT-A is composed of 25 numbers arranged randomly and the participants were instructed to draw a line connecting in ordered sequence as quickly as possible. In the Chinese version of TMT-B, the English letters were replaced by 12 Chinese zodiac sign (rat, ox, . . . dog, and pig). 39 The participants drew a line to connect numbers and 12 Chinese zodiac sign in alternating sequence as quickly as possible. The scores are recorded as the time required to complete the test. The quality of life was assessed using 39 item Parkinson’s Disease Questionnaire (PDQ-39), comprising 39 self-report question on a 5-point Likert scale. 40 The PDQ-39 has 8 domains, including mobility, daily activities, emotional well-being, social support, stigmatization, knowledge, communication, and bodily discomfort. Total score was transformed to a 0 to 100 scale. A higher score reflects lower quality of life.
Sample Size
The sample size was calculated by using G*Power v3.1.9.7. with type I error of 0.05, power of 80%, and effect size of 0.536. 27 The effect size was referenced from a previous study exploring the effect of gait training combined with tDCS to improve gait performance in people with PD. The minimum sample size of each group was 14. However, considering a 20% attrition rate of data processing and drop-out, we determined that at least 17 participants in each group were required to detect a significant difference in DTW velocity.
Statistical Analyses
All analyses were performed using SPSS 29.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were determined for participants’ characteristics and baseline outcomes. The Shapiro-Wilk test was used to assess the normality. The baseline differences between 2 groups were assessed by chi-square test or independent t-test. Linear mixed models were used to analyze the effect of time, group, and the interactions between group and time, and all models were controlled for baseline values. Models were estimated using restricted maximum likelihood estimation to account for missing data and without any data imputation. 41 Compound symmetry covariance structure was selected based on Schwarz’s Bayesian Criterion. 42 A paired t-test was conducted to compare within group data before and after intervention. Bonferroni post hoc tests were used to adjust for multiple comparisons. Due to not being normally distributed for the TMS data, we used the Wilconxon sign ranks test for within-group comparisons, followed by Bonferroni correction. Statistical significance was set at P < .05.
Results
A total of 39 participants were screened for the eligibility of participating this study. As a result, we recruited 34 participants and randomly assigned them to either the DLPFC tDCS + TT group (n = 17) or the sham tDCS + TT group (n = 17). None of participants reported any adverse events during the intervention, and 3 of them withdrew from the study: 1 (in the DLPFC tDCS + TT group) was injured in a fall at home and 2 (1 for each group) were unable to participate due to personal reasons. In addition, 2 participants did not complete the post-intervention TMS measurements due to their 120% RMT exceeding the maximum of stimulator output (Figure 1). No significant differences were observed in demographic characteristics and other baseline outcomes between the 2 groups (Table 1).
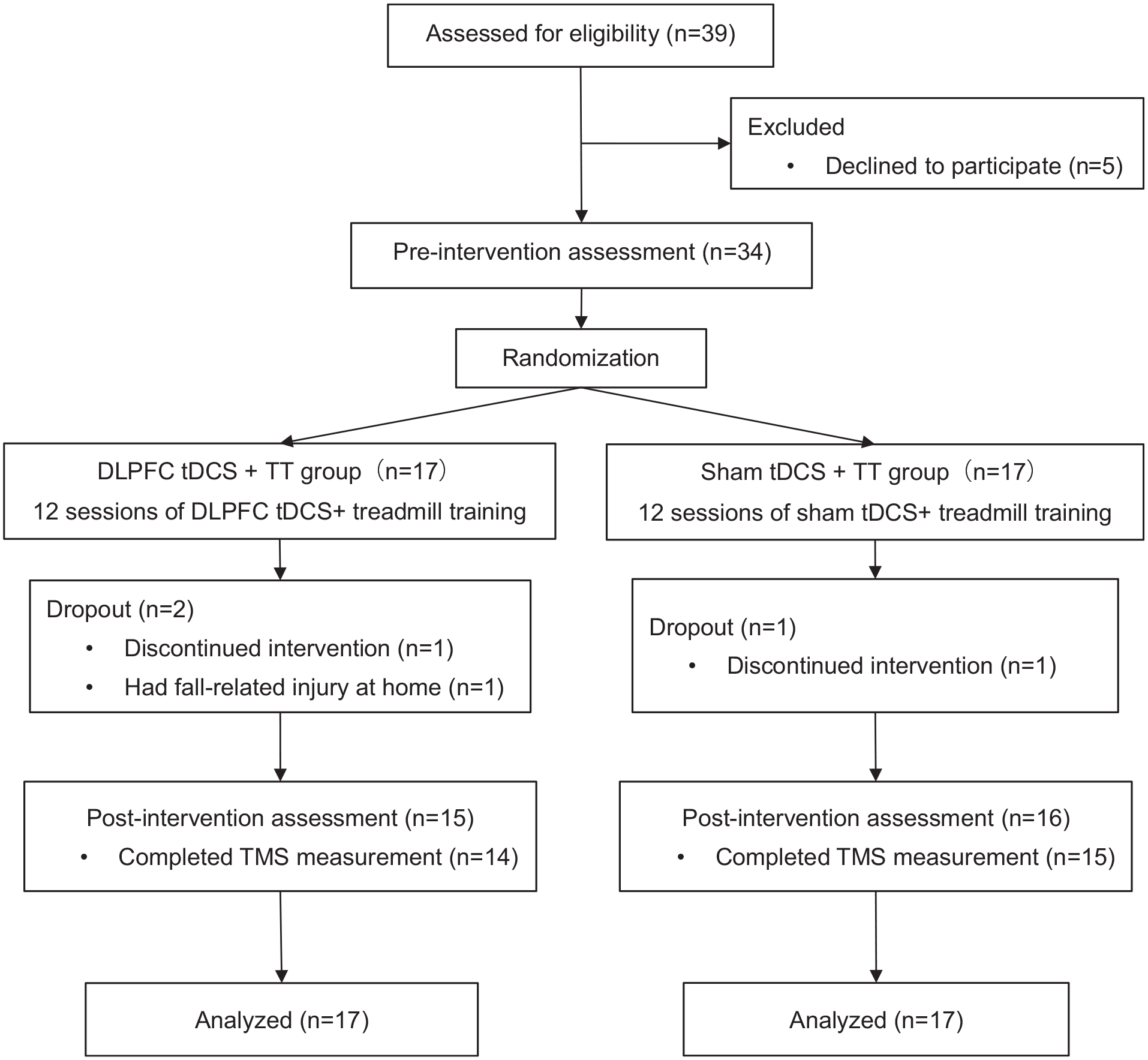
Flowchart of the participant inclusion and study procedures.
Demographic Characteristics of Participants (n = 34).
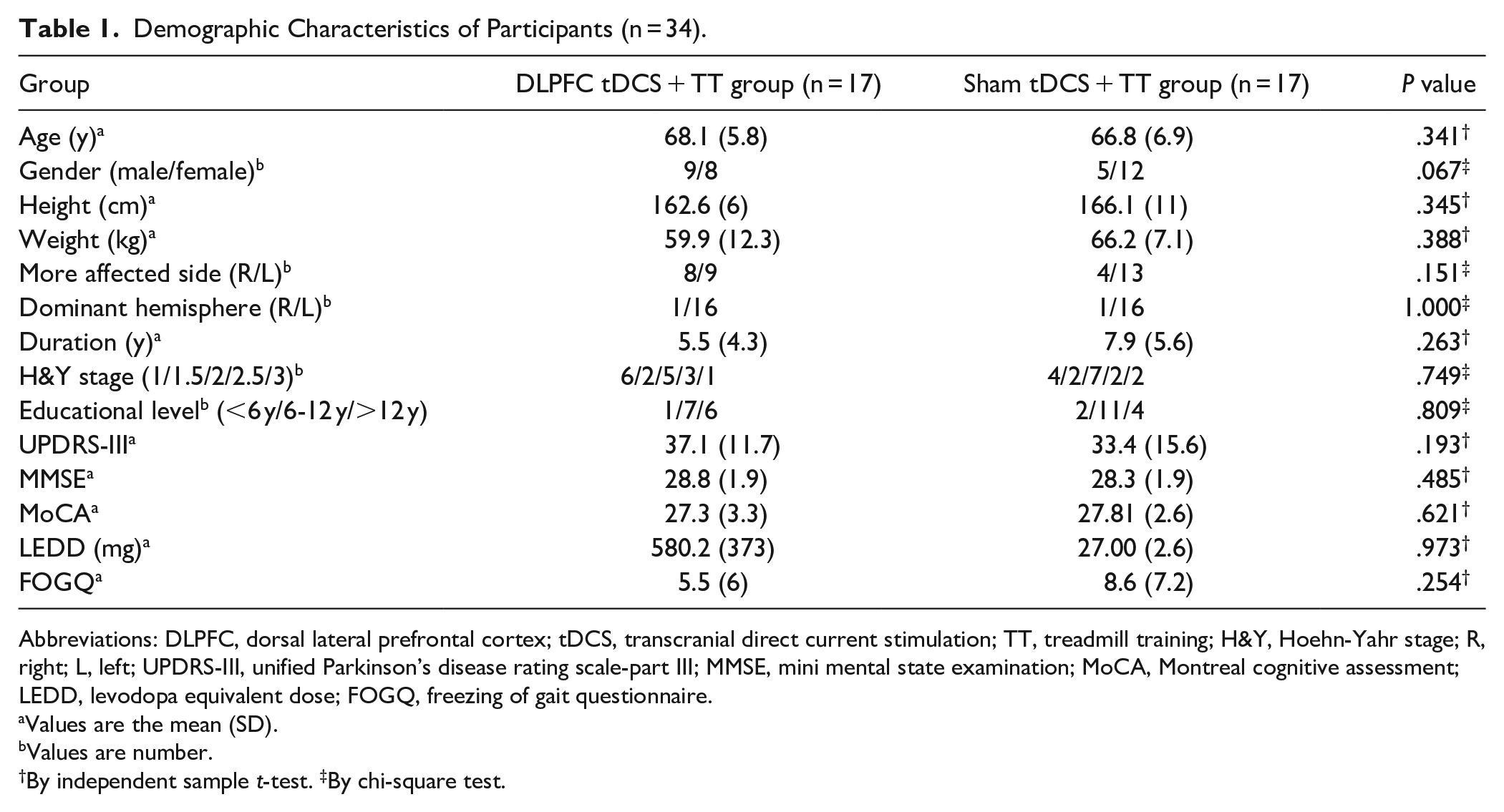
Abbreviations: DLPFC, dorsal lateral prefrontal cortex; tDCS, transcranial direct current stimulation; TT, treadmill training; H&Y, Hoehn-Yahr stage; R, right; L, left; UPDRS-III, unified Parkinson’s disease rating scale-part III; MMSE, mini mental state examination; MoCA, Montreal cognitive assessment; LEDD, levodopa equivalent dose; FOGQ, freezing of gait questionnaire.
Values are the mean (SD).
Values are number.
By independent sample t-test. ‡By chi-square test.
DTW Performance
The mean, standard deviation, and the results of linear mixed models for CDTW and MDTW are presented in Table 2. There were group and time interactions on CDTW velocity (F1,32 = 4.30, P = .046), cadence (F1,32 = 4.43, P = .043), and stride time (F1,32 = 4.516, P = .007). However, no statistically significant differences on gait parameters during MDTW were observed between groups (Figure 2). In the within-group analysis, participants in DLPFC tDCS + TT group showed a significant increase in CDTW velocity (t = −3.38, P = .010), cadence (t = −3.15, P = .030), and stride length (t = −3.48, P = .015) after 12 sessions of intervention. However, a significant increase in cadence (t = −5.00, P < .001) and a significant reduction in CDTW stride time (t = 3.44, P = .015) were noted in sham tDCS + TT group. Regarding the MDTW performance, participants in DLPFC tDCS + TT group showed significant improvements in velocity (t = −4.84, P < .001), cadence (t = −3.67, P = .010), stride length (t = −3.99, P = .005), and stride time (t = 3.325, P = .020). However, the MDTW performance did not improve in sham tDCS + TT group.
CDTW and MDTW Performance in Both Groups.
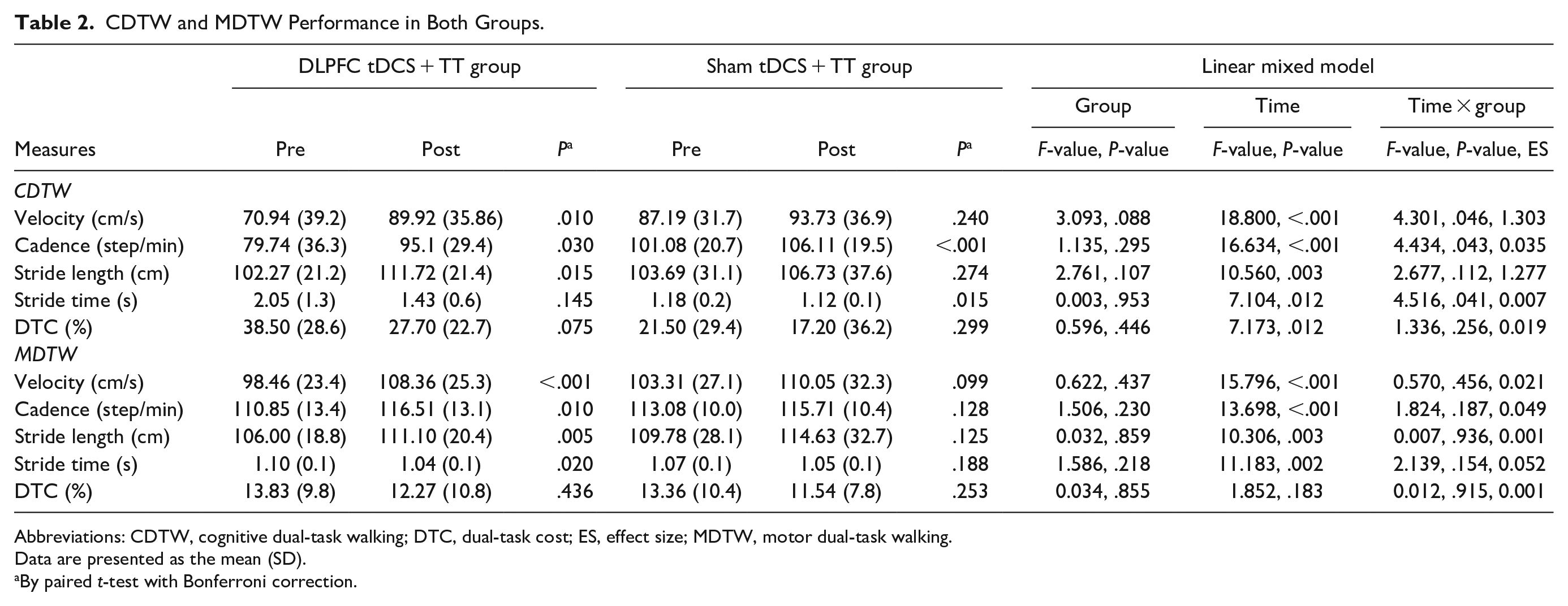
Abbreviations: CDTW, cognitive dual-task walking; DTC, dual-task cost; ES, effect size; MDTW, motor dual-task walking.
Data are presented as the mean (SD).
By paired t-test with Bonferroni correction.
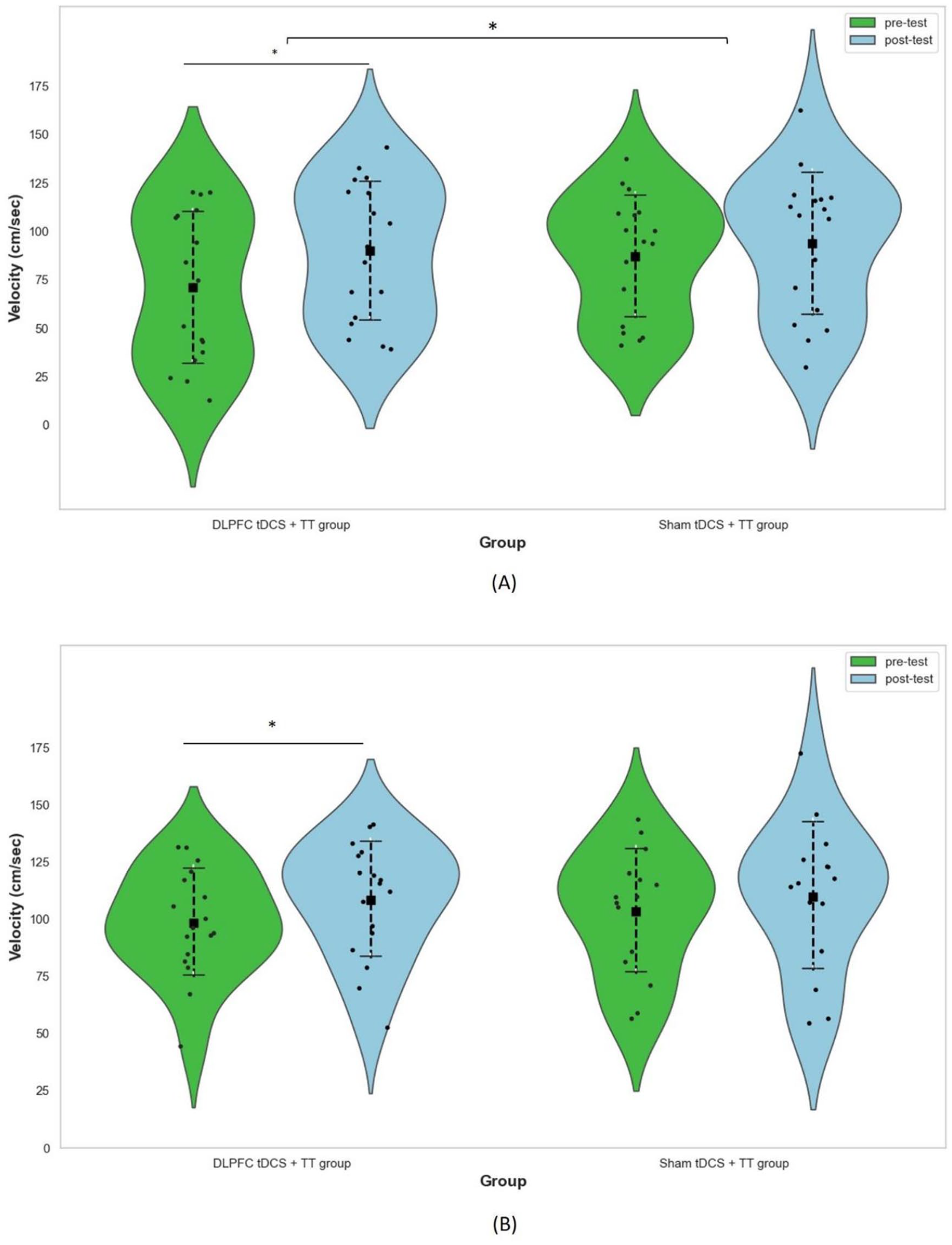
Effect of tDCS followed by TT on velocity during (A) CDTW and (B) MDTW.
Cortical Excitability
Table 3 shows the results of cortical excitability in the DLPFC tDCS + TT group and sham tDCS + TT group. Linear mixed models revealed that the RMT of stimulating hemisphere increased significantly more in DLPFC tDCS + TT group than sham tDCS + TT group (F1,32 = 5.50, P = .026). Moreover, RMT of stimulating hemisphere (t = 3.22, P = .024) and MEP of stimulating hemisphere (t = 3.21, P = .024) decreased significantly after DLPFC tDCS followed by TT. However, there were no significant changes after sham tDCS followed by TT.
Corticospinal Excitability After Different Interventions.
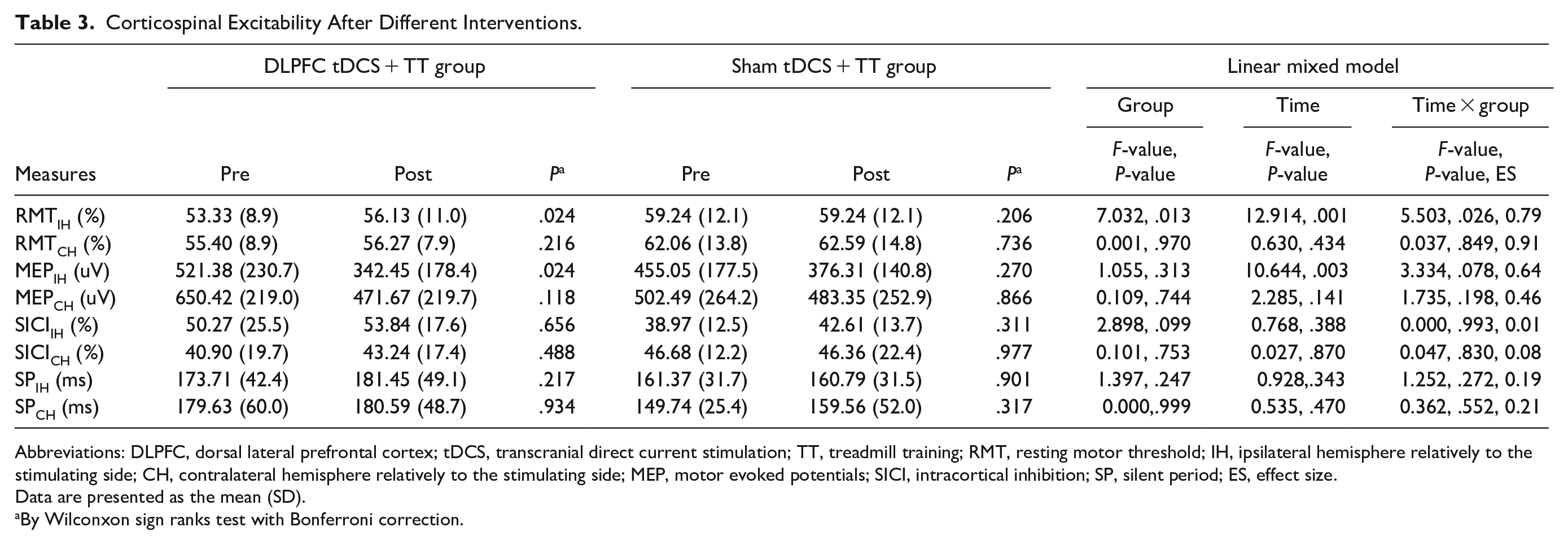
Abbreviations: DLPFC, dorsal lateral prefrontal cortex; tDCS, transcranial direct current stimulation; TT, treadmill training; RMT, resting motor threshold; IH, ipsilateral hemisphere relatively to the stimulating side; CH, contralateral hemisphere relatively to the stimulating side; MEP, motor evoked potentials; SICI, intracortical inhibition; SP, silent period; ES, effect size.
Data are presented as the mean (SD).
By Wilconxon sign ranks test with Bonferroni correction.
Secondary Outcomes
There were no significant differences in usual walking performance, TUG, PDQ-39, and other cognitive outcomes between groups. However, participants in both DLPFC tDCS + TT group and sham + TT group showed significant increases in usual walking velocity (DLPFC tDCS + TT group: t = −3.80, P = .008; sham + TT group: t = −3.02, P = .032) and stride length (DLPFC tDCS + TT group: t = −3.21, P = .020; sham + TT group: t = −3.19, P = .024) after training. The DLPFC + TT group, but not tDCS + TT group, showed a significant improvement in the TUG performance (t = 3.326, P = .004) after training (Table 4).
Usual Walking and TUG Performance After Different Interventions.
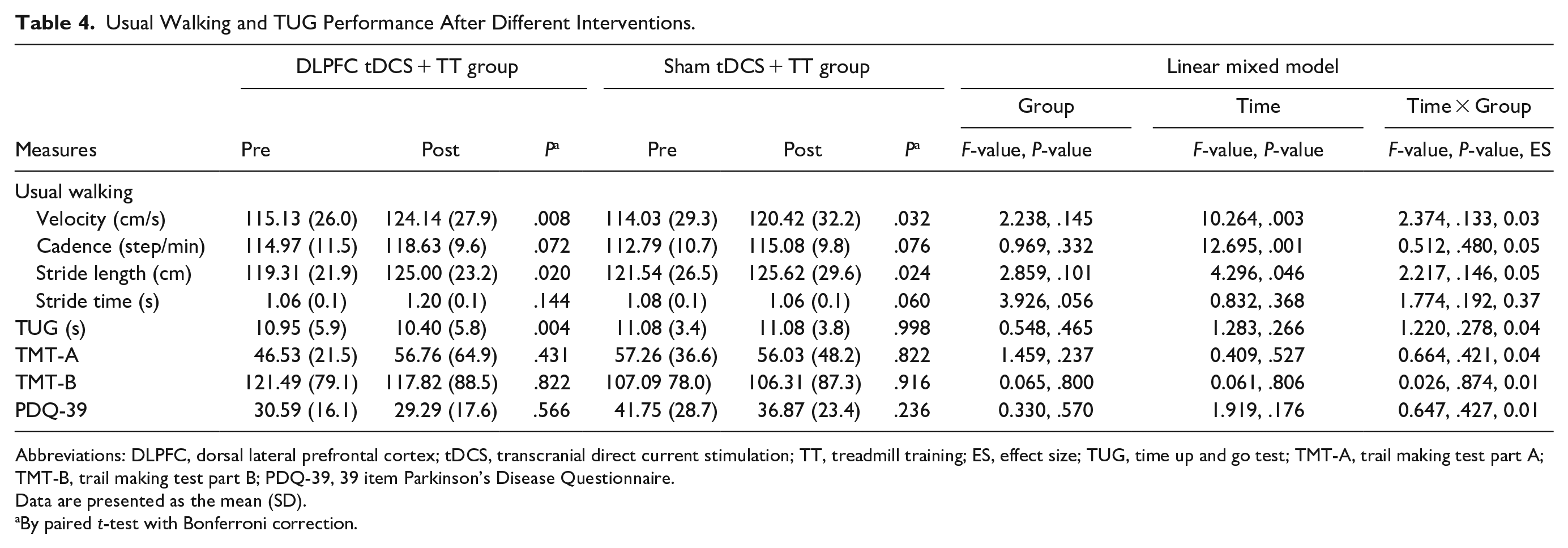
Abbreviations: DLPFC, dorsal lateral prefrontal cortex; tDCS, transcranial direct current stimulation; TT, treadmill training; ES, effect size; TUG, time up and go test; TMT-A, trail making test part A; TMT-B, trail making test part B; PDQ-39, 39 item Parkinson’s Disease Questionnaire.
Data are presented as the mean (SD).
By paired t-test with Bonferroni correction.
Discussion
This randomized double-blind controlled trial is the first study to explore the effects of DLPFC tDCS followed by TT on dual-task gait performance and cortical excitability in individuals with PD. The results showed that DLPFC tDCS followed by TT can result in better effects on CDTW performance but not on MDTW as compared with sham tDCS and TT. Regarding cortical excitability, DLPFC tDCS and TT significantly decreased the excitability of the M1 more than sham tDCS and TT.
In this study, we found that DLPFC tDCS followed by TT exerted better effects on CDTW velocity and stride length in people with PD. Likewise, the additional effects of DLPFC tDCS on CDTW have been demonstrated in older adults with mild cognitive impairment (MCI). 43 The increased CDTW velocity after the anodal DLPFC tDCS has been observed in individuals with PD,24,44 suggesting that the DLPFC tDCS decreased the impact of dual-task interference during walking. A functional near-infrared spectroscopy study showed that DLPFC tDCS improved CDTW performance and reduced prefrontal activity in older adults, suggesting that DLPFC tDCS facilitated prefrontal neural efficiency during CDTW. 23 Given that DTW performances improved after 12-session training, our findings support the hypothesis that anodal tDCS on DLPFC improved DLPFC neural efficiency during DTW. 23 However, the hypothesis should be evaluated in future research. Baek et al study found that older adults who exhibit low CDTW may be able to improve their performance while walking on a treadmill with fewer attentional resources. 45 Furthermore, Thumm et al showed that compared to over-ground walking, the gait pattern is more stable and the prefrontal activation is lower during treadmill walking in people with PD. 46 Therefore, we attribute the CDTW improvement in our participants may not only to DLPFC tDCS effects, but also to the automaticity induced by the TT. Even though the minimal clinically important difference (MCID) in DTW velocity is unknown, our data showed that the improvement of CDTW velocity in the DLPFC tDCS + TT group reached a moderate (0.14 m/s) MCID in usual walking velocity in PD, 47 which may reflect the importance of DLPFC tDCS followed by TT in clinical rehabilitation. Additionally, we did not observe significant improvements in CDTW after TT alone, although body weight-support TT with additional group therapy has been demonstrated to improve DTW performance. 48
On the other hand, although DLPFC tDCS followed by TT resulted in significant improvement in MDTW after training, such improvement did not reach to a significant level as compared with TT alone. Two possible reasons may explain the insignificant group difference in MDTW. First, it has been showed that CDTW is more challenging than MDTW in people with PD. 33 In our participants, the velocity of CDTW (81.14 ± 36.12 cm/s) was significantly lower than that of MDTW (100.73 ± 24.92 cm/s; P < .001). Second, Liu et al showed that MDTW performance is related with wider cortical regions in addition to prefrontal cortex. 18 Therefore, multiple cortical stimulation sites or longer training period may be needed to enhance MDTW improvement.
There is growing evidence supporting the benefits by applying DLPFC tDCS with exercise to improve motor performance in individuals with PD. 49 However, evidence regarding the brain changes after such intervention is not available. In the present study, we found a decreased M1 excitability after DLPFC stimulation followed by TT. Cao et al have shown that intermittent theta burst stimulation (an excitatory form of rTMS) to the DLPFC decreased cortical excitability in M1 in healthy adults. 50 A recent TMS-electroencephalography (EEG) study revealed the modulated effect of DLPFC tDCS across functional cortical networks, including M1, in healthy adults. 51 Furthermore, previous studies have demonstrated that DLPFC tDCS increased dopamine release in striatum.52,53 An animal study has shown that TT can increase dopamine levels in the striatum. 54 Based on these findings, we suggest that the decrease in M1 excitability may be linked to modulation of the dopamine system in people with PD, thereby improving CDTW performance. On the other hand, a study reported that anodal tDCS should be delivered before motor training to prime cortical excitability, thereby enhancing training effects. 30 Karim et al suggested that anodal tDCS facilitates subsequent learning by reducing the threshold for NMDA-receptor activation.55,56 Taken together, our findings support the priming effects of tDCS in enhancing motor performance. 30
Interestingly, we found that decreased M1 excitability only in the stimulated hemisphere, but not in the non-stimulated hemisphere. Consistent with our results, Conceição et al showed that DLPFC activity did not increase in the non-stimulated hemisphere after the DLPFC tDCS and aerobic exercise combined sham tDCS, suggesting that anodal tDCS primarily modulates neuroplasticity in the stimulated hemisphere. 57
In accordance with the previous studies that strongly support the positive effects of TT on PD gait performance. 28 It is not surprising that both DLPFC tDCS with TT and TT alone resulted in significant improvements in usual walking velocity and stride length in our study. Previous studies showed no change in TUG performance after a single session of DLPFC tDCS in people with PD.24,31 However, we did note a significant improvement in TUG after 12 sessions of DLPFC tDCS and TT, and such improvement was not achieved by TT only. On the other hand, the cognitive function measured by TMT-A and TMT-B did not change significantly after training in both groups. This result could be explained by the reason that we applied tDCS with TT rather than combining cognitive training. A previous study has demonstrated that DLPFC tDCS with cognitive training improved cognitive function in individuals with PD. 58
Limitations
This study has some limitations. First, the study only demonstrated the effects of tDCS followed by TT, and the effects of tDCS prior to, or concurrent with, TT are unknown. Future studies need to investigate the differences in different combination timings. Second, we did not include a group that did not receive any kind of intervention, so that it would limit the interpretation of our findings. Third, the study did not measure follow-up changes; therefore, it could not demonstrate whether the observed positive effects could be maintained.
Conclusions
The present study showed that DLPFC tDCS followed by TT exerted more beneficial effects on CDTW performance and cortical excitability than TT alone in individuals with PD. Applying DLPFC tDCS prior to TT could be suggested for use during gait rehabilitation in people with PD.
Footnotes
Acknowledgements
The authors would like to thank the participants.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Ministry of Science and Technology (MOST-106-2314-B-010- 037-MY3) and National Health Research Institutes (NHRI-EX110-10913PI).
