Abstract
Background. Stroke is the leading cause of adult disability, but treatment options remain limited, leaving most patients with incomplete recovery. Patient and animal studies have shown potential of noninvasive brain stimulation (NIBS) strategies to improve function after stroke. However, mechanisms underlying therapeutic effects of NIBS are unclear and there is no consensus on which NIBS protocols are most effective. Objective. Provide a review of articles that assessed effects and mechanisms of repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS) in animal stroke models. Methods. Articles were searched in PubMed, including cross-references. Results. Nineteen eligible studies reporting effects of rTMS or tDCS after stroke in small rodents were identified. Seventeen of those described improved functional recovery or neuroprotection compared with untreated control or sham-stimulated groups. The effects of rTMS could be related to molecular mechanisms associated with ischemic tolerance, neuroprotection, anti-apoptosis, neurogenesis, angiogenesis, or neuroplasticity. Favorable outcome appeared most effectively when using high-frequency (>5 Hz) rTMS or intermittent theta burst stimulation of the ipsilesional hemisphere. tDCS effects were strongly dependent on stimulation polarity and onset time. Although these findings are promising, most studies did not meet Good Laboratory Practice assessment criteria. Conclusions. Despite limited data availability, animal stroke model studies demonstrate potential of NIBS to promote stroke recovery through different working mechanisms. Future studies in animal stroke models should adhere to Good Laboratory Practice guidelines and aim to further develop clinically applicable treatment protocols by identifying most favorable stimulation parameters, treatment onset, adjuvant therapies, and underlying modes of action.
Keywords
Introduction
Globally, stroke is a devastating neurological disorder and a leading cause of death and acquired disability. 1 The majority of stroke patients experience motor impairment, which affects movement of the face, leg, and/or arm on one side of the body. 2 Upper limb motor deficiencies are often persistent and disabling, affecting independent functional activities of daily living. 3 Unfortunately, most stroke patients recover incompletely after stroke, despite intensive rehabilitation strategies.3,4 Although there is a diverse range of interventions (for overview, see review by Pollock and colleagues 4 ) aimed at improving motor outcome after stoke, there is still a pressing need for novel treatment therapies and continued research to reduce disability and improve functional recovery after stroke.
Noninvasive brain stimulation (NIBS) techniques, such as repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS), have shown promising therapeutic potential in stroke patient studies.5,6 The rationale behind rTMS or tDCS therapy is to modulate cortical excitability, increase neural plasticity, and improve functional motor outcome. For many studies, this approach has been based on the interhemispheric competition model. 7 The interhemispheric competition model suggests that functional recovery in stroke patients is hindered due to reduced output from the affected hemisphere and excessive transcallosal inhibition from the unaffected hemisphere. 8 Therefore, improvement in motor deficits may be obtained with NIBS strategies that facilitate excitability in the affected hemisphere or suppress inhibitory activity from the unaffected hemisphere.9,10 Depending on the type and duration of the stimulation protocol, both rTMS and tDCS can be used to increase (>5 Hz rTMS; intermittent theta burst stimulation; anodal tDCS) or decrease (⩽1 Hz rTMS; continuous theta burst stimulation; cathodal tDCS) cortical excitability, with potentially lasting effects beyond the stimulation period, promoting mechanisms of synaptic plasticity. 11 Evidence suggests that rTMS and tDCS techniques are able to induce changes in cortical excitability associated with facilitation or long-term potentiation like plasticity via glutamatergic neurotransmission, or inhibition and long-term depression via GABAergic neurotransmission.12,13 Furthermore, effects of rTMS and tDCS are not restricted to the target region of stimulation, but also affect distantly connected cortical areas, allowing for the modulation of large-scale neural networks. 14
However, despite accumulating evidence of the potential of NIBS, the precise therapeutic mechanisms of action of rTMS and tDCS are largely unidentified and there is no consensus about standardized treatment protocols. Moreover, when deciding on treatment after stroke with either rTMS or tDCS, the poststroke time and lesion status should be considered, and stimulation intensity and duration must be fine-tuned to prevent further tissue damage or the interruption of beneficial plastic changes.15,16 These uncertainties emphasize the critical need for basic understanding of the (patho)physiological processes that are influenced by rTMS and tDCS paradigms after stroke, which may ideally be explored in well-controllable and reproducible experimental animal models.
In animal models of stroke, similar to the human condition, there is a variable degree of spontaneous functional improvement after stroke, associated with a complex cascade of cellular and molecular processes that are activated within minutes after the insult, both in perilesional tissue and remote brain regions.17,18 These events include changes in genetic transcriptional and translational processes, alterations in neurotransmitter interactions, altered secretion of growth factors, gliosis, vascular remodeling, and structural changes in axons, dendrites, and synapses.19,20 Therefore, assessment of the effects of NIBS on endogenous recovery processes in animal stroke models offer excellent opportunities for the exploration of neuroplastic and neuromodulatory mechanisms, which could aid in the optimization of treatment protocols for clinical applications.
Our goal was to provide an overview of studies that assessed functional outcomes and potential mechanisms of action of rTMS and tDCS in animal models of stroke, which may guide future studies that aim to improve mechanistic insights and therapeutic utilization of NIBS effects after stroke.
Literature Search Strategy and Study Quality Assessment
A bibliographic search was carried out to identify publications on rTMS or tDCS applications in preclinical stroke studies, using specific keywords that are specified in the rTMS and tDCS sections below. The quality of the methods of each study was assessed based on the Good Laboratory Practice (GLP) guidelines provided by Macleod et al, 21 which have been proposed to prevent the introduction of bias at the bench and the consequent overstatement of neuroprotective efficacy. The GLP guidelines suggest that details of the following 8 points should at least be included in publications: (1) animals (species, strain, source), (2) sample size calculation, (3) inclusion/exclusion criteria, (4) randomization (method), (5) allocation concealment, (6) reporting of animals excluded from analysis, (7) blinded assessment of outcome, and (8) reporting potential conflicts of interest and study funding. The methods of each reviewed article were assessed and scored based on each of the 8 GLP criteria. One point was given for each criterion if all information was present, half a point for partial information, and no point if the information was absent or unclear. The GLP scores, which could range from 0 to 8, for all publications are summarized in Tables 1 and 2.
Repetitive Transcranial Magnetic Stimulation in Stroke Models
In contrast to the majority of scientifically and clinically approved treatments, there is a relative shortage of preclinical nonhuman TMS data. 22 This may be explained by the fundamentally noninvasive character of TMS, resulting in approved use of magnetic stimulators for peripheral nerve stimulation in several countries, including the United States, and Food and Drug Administration approval of rTMS to treat depression without animal safety data. Moreover, there is a lack of appropriately sized coils for studies in small animals. Consequently, there are still many uncertainties about the full therapeutic potential of rTMS protocols, and their precise therapeutic mechanism of action in several neurological and psychiatric disorders.
Since the first publication of a TMS study in rats in 1990, there has been an exponential increase in published animal TMS studies, including preclinical studies in animal models of disease.23,24 Experiments involving repetitive TMS in animal models of Alzheimer’s disease, 25 depression, 26 epilepsy, 27 Huntington’s disease, 28 Parkinson’s disease, 29 and stroke30-41 have already provided substantial insights into the therapeutic potential of TMS.
An in-depth literature search on PubMed, using combinations of keywords (eg, noninvasive brain stimulation, transcranial magnetic stimulation, rTMS, cerebral/stroke/ischemia/infarct, disease/animal model, animal, rodent, rat, mice/mouse, gerbil, large animals/nonhuman primate), for animal models of stroke involving treatment with rTMS, revealed 12 scientific articles published between 2003 and October 2017.30-41 These articles (summarized in Table 1) applied rTMS after experimental stroke to assess (1) effects on ischemic tolerance, 30 (2) underlying therapeutic mechanisms,30-35,39,40 (3) the additive effect of TMS when combined with other therapies,36-38 and (4) the effect of rTMS on gene expression. 41 These studies applied TMS using coils of different shapes and sizes. Either circular or figure-of-eight coils were used, with outer diameter sizes ranging from 12 to 60 mm or 20 to 70 mm, respectively. Figure-of-eight coils generally provide more focal stimulation 42 ; however, the use of smaller circular TMS coils may improve focality in the small rodent brain. 43
TMS Studies in Animal Models of Stroke.
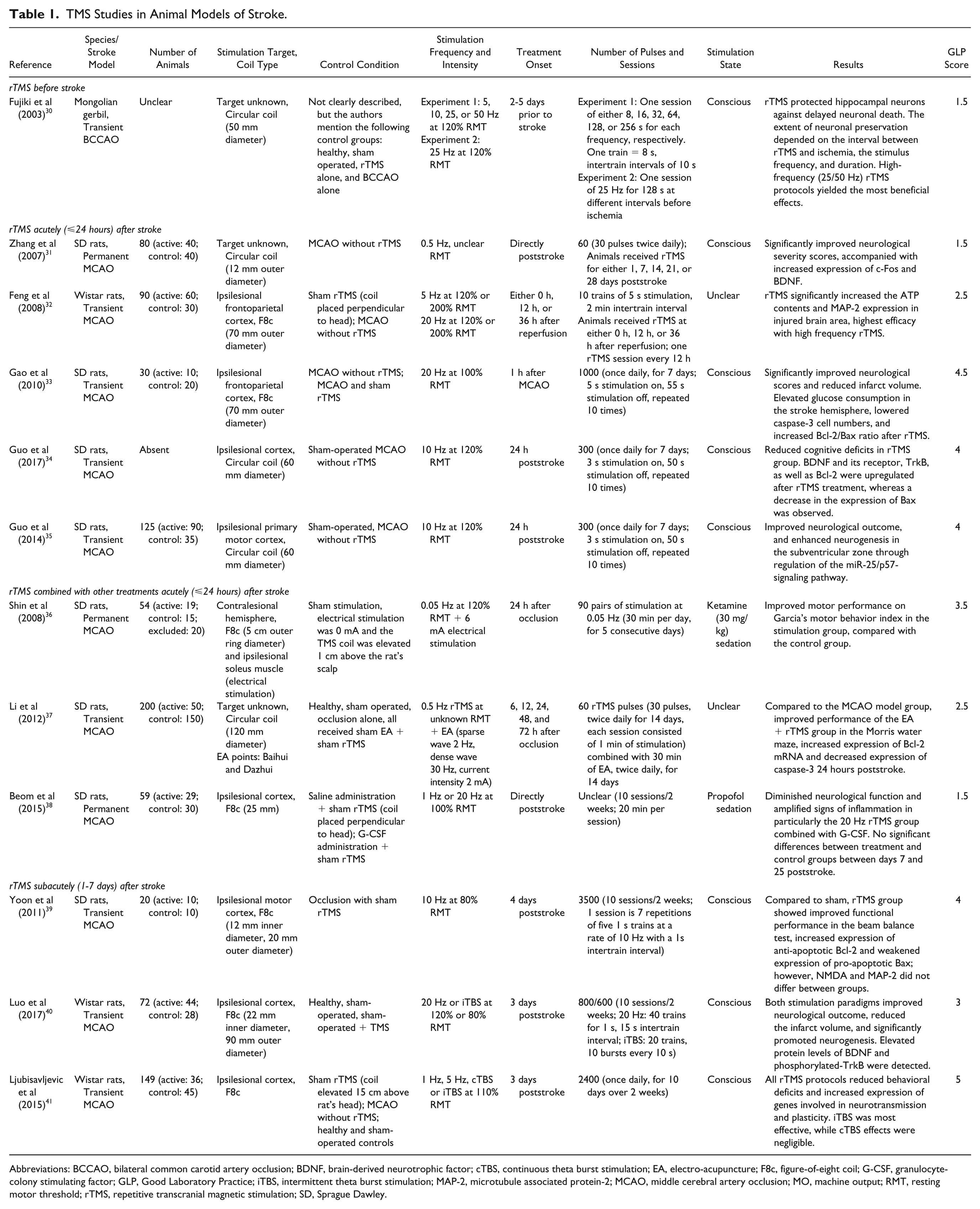
Abbreviations: BCCAO, bilateral common carotid artery occlusion; BDNF, brain-derived neurotrophic factor; cTBS, continuous theta burst stimulation; EA, electro-acupuncture; F8c, figure-of-eight coil; G-CSF, granulocyte-colony stimulating factor; GLP, Good Laboratory Practice; iTBS, intermittent theta burst stimulation; MAP-2, microtubule associated protein-2; MCAO, middle cerebral artery occlusion; MO, machine output; RMT, resting motor threshold; rTMS, repetitive transcranial magnetic stimulation; SD, Sprague Dawley.
rTMS Before Stroke
One study has been published in which rTMS was applied prior to experimental stroke. In this study, Fujiki and colleagues found that rTMS before a transient ischemic insult in adult gerbil brain induced ischemic tolerance preventing delayed neuronal death in the hippocampus. 30 The extent of neuronal preservation following rTMS was dependent on the stimulation paradigm, as well as on the interval between stimulation and ischemic stroke. Maximal neuronal preservation and protection was accomplished after the application of 25 Hz rTMS (for at least 128 seconds) at 48 hours before ischemia. This study suggests a potential role for NIBS as a pretreatment in patients undergoing procedures that may induce transient brain ischemia.
rTMS Acutely (⩽24 Hours) After Stroke
The application of rTMS directly after permanent middle cerebral artery occlusion (MCAO) in rats has reportedly led to neuroprotection and improved behavioral outcomes. 31 Following longitudinal treatment with low-frequency (0.5 Hz) rTMS to a nonspecified region of interest for different periods of time (7, 14, 21, and 28 days), Zhang et al found a significant improvement in functional recovery, based on a neurological severity score, as compared with untreated rats. However, these results should be treated with care as statistical testing involved multiple t-testing of differences between 10 experimental groups, without a prior ANOVA (analysis of variance) or correction for multiple comparisons, which could have led to type I (false positive) errors. It was also reported that rTMS-treated animals showed a substantial increase in the expression of c-Fos (at days 7, 14, 21, and 28) and brain-derived neurotrophic factor (at days 7, 14, and 21) in cortical tissue surrounding the infarct area. The authors speculated that rTMS-induced c-Fos expression, indicative of neuronal activation, subsequently led to upregulation of brain-derived neurotrophic factor expression, which could contribute to improved outcome after stroke.23,31
Underlying biological mechanisms of rTMS treatment in the acute phase after ischemic stroke were also explored by Feng and colleagues. 32 Their study focused on the effect of rTMS on ATP content and microtubule associated protein-2 expression. Microtubule associated protein-2, a high-molecular-weight protein, mainly present in mature neurons, plays an important role in mitochondrial axonal transport, maintaining the structural integrity of neurons, and acts in synapse formation and dendrite plasticity.32,44,45 A reduction of microtubule associated protein-2 expression due to an ischemic lesion might decrease mitochondrial axonal transport, leading to reduced ATP availability and eventually neuronal death.46-49 Rats treated with ipsilesional high-frequency (20 Hz) rTMS paradigms, starting 1 hour after transient MCAO, revealed significantly increased ATP content and microtubule associated protein-2 expression in the affected hemisphere, compared with lower frequency rTMS (5 Hz) and untreated sham/control groups. The increased microtubule associated protein-2 expression following high-frequency rTMS might suggest ongoing processes of neuronal repair. 50
Repetitive TMS treatment (for 7 days) initiated 1 hour after transient ischemia has also been reported to reduce apoptosis. In a study by Gao and colleagues, ipsilesional high-frequency rTMS (20 Hz) resulted in significantly reduced numbers of caspase-3 positive cells and an increased ratio of anti-apoptotic Bcl-2 over pro-apoptotic Bax in the affected hemisphere, compared with control groups without rTMS treatment. 33 Ipsilesional high-frequency rTMS treatment also reduced the infarct volume and improved the neurological outcome, which could be explained by the blocking of apoptosis and the maintenance of glucose utilization in the ischemic hemisphere as observed with fluorodeoxyglucose microPET.
Anti-apoptotic effects of ipsilesional high frequency (10 Hz) rTMS (for 7 days) were also observed when introduced 24 hours after stroke. 34 Compared with an untreated stroke group, rTMS significantly reduced apoptosis in the CA1 region of the hippocampus after poststroke stimulation. Along with reduced neuronal apoptosis, neurogenesis was enhanced in the hippocampi of the rTMS group, which was accompanied by improved cognitive function in the Morris water maze task. The validity of these positive treatment effects is difficult to judge as the total number of included animals and group sample sizes were vaguely reported for this study.
Repetitive TMS-induced enhancement of poststroke neurogenesis has also been demonstrated by an earlier study from the same research group. In this study, Guo and colleagues applied 10 Hz rTMS treatment (for 7 days) to the lesioned hemisphere starting 24 hours after transient MCAO in rats. 35 This significantly increased the proliferation of adult neural stem cells in the ipsilateral subventricular zone and upregulated micro RNA-25 in the ischemic cortex, as compared with sham-operated and untreated model groups. 35 Unfortunately, this study lacked a sham rTMS group. Other researchers have demonstrated the potential of rTMS to promote the proliferation of adult neural stem cells in healthy rat brain, 51 which corroborates the hypothesis that rTMS may enhance poststroke neurogenesis.
rTMS Combined With Other Treatments Acutely (⩽24 Hours) After Stroke
Some studies have combined TMS with other therapies, such as peripheral nerve stimulation, 36 electro-acupuncture, 37 and granulocyte-colony stimulating factor (G-CSF) administration, 38 for the treatment of stroke. Conjoint electrical stimulation of a peripheral nerve (ipsilesional to the lesion) and contralateral motor cortex stimulation using TMS is also referred to as paired associative stimulation, a method that can induce long-lasting changes in cortical excitability. 52 Following daily paired associative stimulation for 5 consecutive days, starting 24 hours after permanent MCAO in rats, Shin et al found that the motor behavioral index (7 days poststroke) was significantly higher in the stimulation group than in the sham-stimulated group. 36
The combination of rTMS and electro-acupuncture may improve learning and memory abilities in rats, as shown by Li et al. 37 In this study, electro-acupuncture electrodes were inserted at Baihui (right midpoint of the parietal bone) and Dazhui (posterior midline) points; however, the rTMS target region/hemisphere was not defined. The combination of 0.5 Hz rTMS with electro-acupuncture treatment (for 14 days) appeared to have an anti-apoptosis effect, by altering the expression of caspase-3 (reduced expression) and Bcl-2 (increased expression) in peri-infarct tissue. Additionally, improved learning and memory abilities in the treatment groups were demonstrated by shorter escape latency times in the Morris water maze task, compared with control groups. Treatment started either at 6, 12, 24, 48, or 72 hours after transient MCAO and was found to be most effective when started 24 hours poststroke.
An attempt to use rTMS to enhance the neuroprotective effects of cytokine G-CSF, by administering treatment directly following permanent MCAO in rats, has been unsuccessful. 38 G-CSF and its receptors are widely expressed in the central nervous system and involved in various processes that can contribute to neuroprotection and neurorepair, such as anti-apoptosis, neurogenesis, anti-inflammation, cellular growth, arteriogenesis, anti-oxidation, and stem cell recruitment. 53 In animal models, administration of G-CSF has reportedly reduced ischemic infarct volume and facilitated functional recovery, particularly after transient cerebral ischemia. 54 Beom et al hypothesized that the combination of G-CSF treatment with ipsilesional rTMS would enhance the effects of G-CSF and reduce its adverse effects (splenomegaly, headache, bone pain, and emergence of bone marrow disease). 38 However, the combination therapy of G-CSF (for 5 days) and ipsilesional high-frequency rTMS (for 2 weeks) exerted a deleterious effect on functional recovery. Beom et al speculated this may be related to the reduced expression of angiogenic mechanisms, enhanced inflammatory responses, or inappropriate timing. 38 These findings are contradictory to results from other experimental studies that reported improvements in motor function, increased neurogenesis, and reduced apoptosis after single treatment with G-CSF 54 or rTMS.33-35
rTMS Subacutely (1-7 Days) After Stroke
Neuroprotective and neurotrophic effects of rTMS have also been reported when treatment was initiated in the subacute phase, that is, between 1 and 7 days, after experimental stroke. Yoon and colleagues performed daily ipsilesional high-frequency (10 Hz) rTMS between 4 and 18 days after transient MCAO. 39 This therapeutic paradigm resulted in enhanced functional improvement in the beam balance test, and reduced neuronal apoptosis, as compared with a sham stimulation group. 39
Three days after transient MCAO in rats, Luo and colleagues started ipsilesional 20 Hz rTMS or intermittent theta burst stimulation for 10 days. 40 Both stimulation protocols promoted neurogenesis in the ipsilateral subventricular zone, and increased neural progenitor cell migration in the peri-infarct striatum, as compared with a control group, a sham-operated group, and a TMS-stimulated sham-operated group. Assessment of neurological function revealed significant main effects of group and time at 14 days after stroke, but there was no significant interaction between the 2 factors.
Ljubisavljevic and colleagues investigated the effects of 4 different ipsilesional rTMS protocols (1 Hz, 5 Hz, continuous and intermittent theta burst stimulation—starting 3 days poststroke) on gene expression after transient MCAO in rats. 41 Compared with the untreated stroke group, the theta burst stimulation protocols induced significant improvement in behavioral deficit scores, without affecting infarct size, after 2 weeks of treatment. Changes in gene expression were largely dependent on stimulation frequency and pattern. Nonetheless, rTMS generally upregulated a large range of genes involved in neuroprotection, neurotransmission, angiogenesis, neural repair, and neuronal plasticity.
Transcranial Direct Current Stimulation in Stroke Models
Over the past couple of years, the use of tDCS as a therapy for psychiatric and neurological disorders has been increasingly investigated in clinical as well as preclinical studies.55,56 For application of tDCS in animal models, the majority of studies have employed an electrode montage similar to a setup that has been originally described by Liebetanz et al. 57 In this approach, a small, plastic jacket (3.5 mm2 contact area) is fixed onto the cranium using nontoxic cement. Saline and a wire electrode are inserted into the plastic jacket before stimulation. In addition to the unilateral epicranial electrode, a large rubber-plate electrode (counter electrode) is placed onto the thorax of the animal. A weak, constant, electrical current (0.1 µA to 10 mA) can then be applied transcranially. 58 Safety guidelines for the application of cathodal 58 and anodal 59 tDCS protocols in animals have been defined. Unlike TMS, tDCS currents do not evoke action potentials, but rather modify the transmembrane neuronal potential and modulate the firing rate of individual neurons in response to supplementary inputs. 60
Transcranial DCS treatment has shown therapeutic potential in animal models of Alzheimer’s disease, 61 epilepsy, 62 neuropathic pain, 63 Parkinson’s disease, 64 and stroke.65-71 An in-depth literature search, with various combinations of keywords (eg, noninvasive brain stimulation, transcranial direct current stimulation, tDCS, cerebral/stroke/ischemia/infarct, disease/animal model, animal, rodent, rat, mice/mouse, gerbil, large animals/nonhuman primate), for animal models of stroke involving treatment with tDCS, revealed 7 articles published between 2010 and October 2017 (summarized in Table 2). These articles aimed to assess the safety and efficacy of tDCS in acute to subacute stroke,65-68 and to identify which functional 69 and cellular70,71 changes are associated with tDCS-induced recovery after stroke.
tDCS Studies in Animal Models of Stroke.
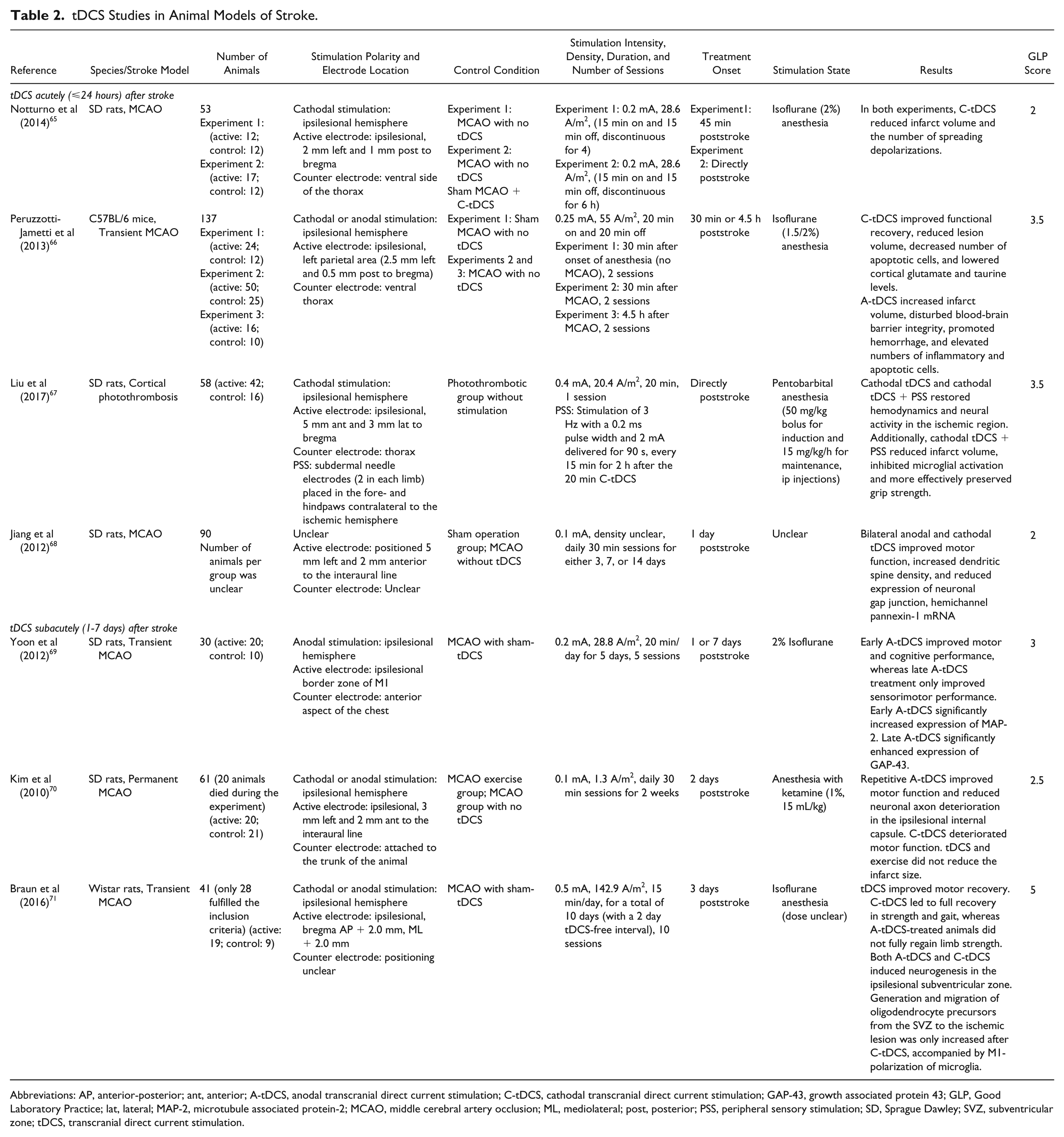
Abbreviations: AP, anterior-posterior; ant, anterior; A-tDCS, anodal transcranial direct current stimulation; C-tDCS, cathodal transcranial direct current stimulation; GAP-43, growth associated protein 43; GLP, Good Laboratory Practice; lat, lateral; MAP-2, microtubule associated protein-2; MCAO, middle cerebral artery occlusion; ML, mediolateral; post, posterior; PSS, peripheral sensory stimulation; SD, Sprague Dawley; SVZ, subventricular zone; tDCS, transcranial direct current stimulation.
tDCS Acutely (⩽24 Hours) After Stroke
In the first hours following experimental stroke, cathodal tDCS has been shown to significantly reduce the number of peri-infarct depolarizations, 65 which are believed to contribute to infarct growth. 72 Following discontinuous ipsilesional cathodal stimulation for either 4 hours (starting 45 minutes after MCAO) or 6 hours (starting directly after MCAO) the infarct volume was significantly reduced by 20% or 30%, respectively, compared with nonstimulated MCAO control groups. 65 Accordingly, the degree of infarct reduction correlated with the extent of cathodal tDCS-induced decrease in peri-infarct depolarizations. In correspondence with these results, Peruzzotti-Jametti and colleagues found that ipsilesional cathodal tDCS for 30 minutes, starting 4.5 hours after transient MCAO in mice, had a significantly favorable treatment effect compared with sham-stimulation and anodal tDCS. 66 Cathodal tDCS led to reduced edema and inflammation, decreased the number of apoptotic cells, and lowered cortical glutamate, creatine, and taurine levels. Consequently, the cytoarchitecture of the cerebral cortex was relatively preserved after cathodal tDCS, resulting in smaller infarct volume and better functional recovery.
The combination of ipsilesional cathodal tDCS and peripheral sensory stimulation of the contralateral forelimb has also been shown to be therapeutically beneficial. Mice treated with this therapeutic strategy immediately after photothrombotic stroke in the sensorimotor cortex showed improved perilesional hemodynamics and enhanced recovery of neural activity in the first hours after stroke as compared with untreated animals and tDCS-treated animals without combined peripheral sensory stimulation. 67 Subsequently, this combination therapy resulted in reduced microglial activation, smaller infarct volumes, and better preserved grip strength 2 days after stroke.
Improved motor function has also been observed by Jiang and colleagues, who allegedly applied repetitive tDCS in rats through the implantation of pericranium electrodes. 68 However, from the article’s Methods section it is unclear whether the authors indeed applied tDCS or a 10 Hz transcranial alternating current stimulation protocol. Nonetheless, the authors found that daily stimulation (starting 1 day after permanent MCAO) resulted in a significant increase of dendritic spine density in the cortex, compared with control groups, on several time points (days 3, 7, and 14) poststroke. In addition, the expression of hemichannel pannexin-1 mRNA, which is potentially involved in hypoxic depolarizations, was reduced. 68
In contrast to the reported positive effects of cathodal tDCS applied to the ipsilesional hemisphere in the acute phase after stroke, ipsilesional anodal tDCS has been shown to result in elevated numbers of inflammatory cells, augmented instability of the blood-brain barrier, and increased hemorrhage and infarct volume in the first hours after stroke. 66
tDCS Subacutely (1-7 Days) After Stroke
Several studies have explored the effects of tDCS applied in the subacute phase of stroke. Yoon and colleagues applied repetitive ipsilesional anodal tDCS for 5 days, starting either 1 day (early treatment) or 1 week (late treatment) after transient MCAO in rats. 69 Both treatment groups showed an improvement in cognitive performance and motor function scores; however, motor function improvement was slightly better in the late treatment group. Their immunohistochemical findings revealed significantly increased levels of microtubule associated protein-2 and growth associated protein-43 (a neuronal plasticity marker) in the perilesional and contralesional cortices in both treatment groups, which correlated with the cognitive and motor improvements. Magnetic resonance imaging and spectroscopy data showed that anodal tDCS did not affect infarct volume or metabolite levels.
Improved motor function was also observed in a study by Kim et al, where ipsilesional anodal tDCS treatment was applied over a period of 2 weeks, starting 2 days after permanent MCAO in rats. 70 Histologically, no clear changes were observed in infarct volume; nonetheless, axonal integrity in the ipsilesional internal capsule was better preserved. Repetitive ipsilesional cathodal tDCS, however, was associated with diminished functional recovery at 16 days postinfarct. The authors speculated that cathodal tDCS may have contributed to the already decreased excitability of the infarcted brain, thereby suppressing recovery mechanisms. The latter findings are in contrast with a recent study by Braun et al, in which accelerated functional recovery and neurogenesis after transient MCAO in rats were observed in response to ipsilesional delivery of either anodal or cathodal tDCS when applied 3 days after stroke. 71 In this study, different aspects of motor function were influenced depending on the polarity of stimulation. For example, limb strength and gait were fully restored in animals treated with cathodal tDCS, whereas anodal tDCS-treated animals regained their gait, but not their full limb strength. Furthermore, cathodal tDCS triggered the generation and migration of oligodendrocyte precursors from the subventricular zone toward the ischemic lesion alongside an M1-polarization of microglia. The effect of cathodal tDCS on these cellular processes might have resulted in increased functional recovery as compared with the anodal tDCS-treated group.
Contradictory findings by Kim et al 70 and Braun et al 71 on the effects of cathodal tDCS on motor recovery may have been due to differences in the stroke model (permanent vs transient MCAO, respectively), the stimulation protocol, or the anesthesia during tDCS.
Discussion
We reviewed the main findings of 19 studies that applied either rTMS or tDCS transcranially in small rodents after experimental stroke. In general, most articles reported stimulation-induced tissue preservation or functional improvement after stroke, as compared with either untreated stroke control or sham stimulation groups. Several advantageous effects, including ischemic tolerance, neuroprotection, and neurorepair, mediated by molecular mechanisms involved in anti-apoptosis, neurogenesis, and neuroplasticity, were measured after rTMS and tDCS.
Only a few preclinical studies have directly compared the effects of different stimulation paradigms, and so far the majority of published rTMS and tDCS studies only assessed stimulation of the lesioned hemisphere. Moreover, ipsilesional high-frequency rTMS or intermittent theta burst stimulation appear to be more favorable for the induction of ischemic tolerance and expression of factors involved in preservation or recovery of postischemic tissue as compared with ipsilesional low-frequency rTMS or continuous theta burst stimulation.30,32,33,35,41 The latter inhibitory paradigms may have more significant therapeutic potential when applied to the contralesional hemisphere, as demonstrated in clinical stroke studies.73-75 The combination of rTMS with adjunct therapy in experimental stroke models has yielded both positive (paired associative stimulation, 36 rTMS plus electro-acupuncture 37 ) and negative results (rTMS plus G-CSF 38 ), and clearly requires further investigation.
The reviewed tDCS studies point toward neuroprotective and neurorestorative effects in animal stroke models, which depend on the polarity and onset of stimulation treatment. Cathodal tDCS of the ipsilesional hemisphere within minutes to hours after stroke reduced progression of ischemic damage.65,66 Additionally, the therapeutic benefits of cathodal tDCS may be enhanced when combined with peripheral sensory stimulation, resulting in preservation of neurovascular function and improved functional recovery. 67 On the other hand, hyperacute ipsilesional anodal tDCS led to progression of degenerative processes. 66 Repetitive cathodal and/or anodal tDCS of the ipsilesional hemisphere during later stages after stroke may promote various recovery-enhancing factors,68-71 although this depends on poststroke timing.69,70
The optimal therapeutic time window, in combination with the preferred stimulation protocol, for poststroke NIBS treatment has yet to be determined. From the current review, it seems that the application of rTMS can have advantageous effects irrespective of treatment onset. The onset of poststroke rTMS treatment will, however, affect the extent to which different neuroprotective and neurorestorative molecular mechanisms are influenced, which depend on various stroke characteristics (eg, type, location, and severity of stroke; age; comorbidities; etc). Similarly, the optimal stimulation parameters and duration may vary.
Similar to NIBS studies in stroke patients, the reviewed studies in animal stroke models employed variable protocols, and treatment efficacy was assessed with different outcome parameters, making it difficult to directly compare interventions and to determine the exact translational value of the applied stimulation protocols. 76 Repetitive TMS was applied with either circular or figure-of-eight coils, with outer diameter sizes ranging from 12 to 60 mm or 20 to 70 mm, respectively. Focal stimulation with relatively large figure-of-eight coils can be achieved in rodents by secure fixation and lateralized coil positioning. 77 However, the majority of the reviewed rTMS studies reported stimulation of the animals while being restraint by hand and conscious, which has most likely negatively affected the focality of the stimulation. Additionally, stimulation frequencies (0.5-50 Hz), intensities (80% to 200% of the resting motor threshold), and number of pulses per session varied extensively between studies (see Table 1). In stroke patients, rTMS above 25 Hz and 130% of the resting motor threshold has been applied infrequently,75,76 as stimulation at high frequencies and intensities is considered unsafe and increases the risk of seizures. 78
Like the rTMS studies, the tDCS studies showed variability in terms of equipment, regions of interest, and stimulation intensity, density, and duration (see Table 2). Notably, unlike the rTMS studies, in all reviewed publications, animals were anesthetized during tDCS. The effect of (different types of) anesthesia on tDCS outcome remains largely unknown and requires further investigation. In the majority of the reported tDCS studies, stimulation parameters were within the safety limits specified by Liebetanz and colleagues. 58 However, recent anodal tDCS studies have reported the detection of lesions at electrode current densities of 47.8 A/m2 79 and 20.0 A/m2, 59 which is significantly below the previously reported safety threshold of 149.9 A/m2 using cathodal tDCS. 58 This suggests that the safety threshold for lesion induction using tDCS could have been underestimated. The 20.0 A/m2 lesion threshold for anodal stimulation is at least more than 10-fold higher than the typical electrode current density of 0.28 to 2.0 A/m2 utilized in human studies.80,81 The application of lower stimulation parameters is therefore recommended in future tDCS studies of animal models, to improve the validity of the data and to facilitate translation to the clinic. 82
We observed that many of the included studies were of relatively low quality based on the GLP assessment criteria 21 (Tables 1 and 2). Therefore, we cannot rule out that some of the reported findings might have been confounded by bias and could overstate the neuroprotective efficacy of rTMS and tDCS, similar to what has been reported for preclinical stroke drug trials. 83 We found that none of the included studies in this review described how the sample size was calculated or whether allocation concealment was implemented. Only 37% of the studies specified inclusion/exclusion criteria based on lesion size, cerebral perfusion status, or behavior poststroke. Exclusion of animals was poorly reported. Only 47% of the studies reported both the strain and source of the animals, and the age of animals was often unclear. The majority of studies reported potential conflicts of interest (79%), blinding of outcome assessment (63%), and random allocation to experimental groups (74%), although the method of randomization was generally not mentioned.
Conclusion
Even though the number of studies that assessed NIBS in animal models of stroke is still limited, the recovery-enhancing effects are encouraging and reflect the translational value of these investigations. Treatments with rTMS and tDCS in animal stroke models have shown that different protocols can have positive influences on functional recovery through ways of neuroprotection or neurorepair. However, the exact therapeutic mechanisms of rTMS and tDCS remain incompletely characterized. Furthermore, it should be mentioned that the limited number of reports of negative or null effects, and the relatively low study quality of several articles, might reflect a publication bias. Preclinical research on modes of action of different NIBS protocols in animal stroke models can provide critical information for the development of NIBS strategies for effective treatment after stroke. Consequently, prospective studies should investigate the effects of multiple stimulation protocols at different time points after stroke, on both the ipsilesional and contralesional hemispheres, to be able to identify optimal treatment protocols that would maximally enhance functional recovery. Prior computer simulations of the induced electrical field can provide essential insights in the location and focality of the stimulation approaches in rodent brain.43,84 Additionally, the rationale and criteria for the selection of the study parameters should be made explicit. Ideally, these studies would follow recent guidelines for preclinical stroke treatment studies, involving randomization and blinded assessments,21,85 and include measures of behavioral outcome and (image-based) markers of neuroprotection and neurorepair that are straightforwardly translatable to the clinic.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Netherlands Organization for Scientific Research (VICI 016.130.662).
