Abstract
Background. Learning processes of writing skills involve the re-engagement of previously established motor programs affected by Parkinson disease (PD). To counteract the known problems with consolidation in PD, transcranial direct current stimulation (tDCS) could be imperative to achieve a lasting regeneration of habitual motor skills. Objective. To examine tDCS-enhanced learning of writing and explore alterations in cortical excitability after stimulation in PD compared with healthy controls (HCs). Methods. Ten patients and 10 HCs received 2 training sessions combined with 20 minutes of 1-mA anodal tDCS or sham on the left primary motor cortex in a randomized crossover design. Writing skills on a tablet and paper were assessed at baseline, after training, and after 1 week of follow-up. Before and immediately after the intervention, cortical excitability and inhibition were measured during rest and activity. Results. Writing amplitude and velocity improved when practice was tDCS supplemented compared with sham in PD. Benefits were sustained at retention for trained and untrained tasks on the tablet as well as for writing on paper. No improvements were found for HCs. Reduced resting motor thresholds after tDCS indicated tDCS-enhanced cortical excitability. Additionally, increments in motor-evoked potential amplitudes correlated with improved writing in PD, whereas HCs showed the opposite pattern. Conclusion. Our results endorse the usefulness of tDCS-boosted learning in PD, at least when applied to improving writing capacity. Although further confirmatory studies are needed, these novel findings are striking because tDCS-mediated consolidation was found for learning a motor task directly affected by PD.
Keywords
Introduction
Handwriting deficits are a hallmark symptom of Parkinson disease (PD) 1 and include difficulties with amplitude, speed, and fluency. 2 Up to 65% of patients experience writing deficits, often as one of the first symptoms. 1 Dopaminergic medication mainly improves movement speed and to a lesser extent amplitude. 3 Hence, rehabilitation focusing on writing amplitude seems warranted. Recently, it was shown that 6 weeks of intensive amplitude training significantly improved writing amplitude in PD patients when compared with a placebo intervention of stretch and relaxation exercises. 4 These improvements were found during both writing on a touch-sensitive tablet and on paper. However, training resulted in a trade-off between amplitude, writing fluency, and speed.4,5 Moreover, improvements in amplitude were only maintained after the 6-week follow-up on the tablet and not on paper, suggesting limited transfer and retention. 4 These consolidation deficits can be attributed to the typical impact of the neuropathology on striatal function6,7 and reflect one of the largest problems in PD rehabilitation—namely, the maintenance of long-term learning results. 8
In healthy adults, noninvasive brain stimulation with transcranial direct current stimulation (tDCS) enhanced motor learning. 9 To date, effects of tDCS in PD were found to be controversial and notoriously variable.10,11 Studies have shown that stimulation of M1 in rest improved upper-limb task performance in PD.12-14 Online benefits were demonstrated in a small sample of PD patients, which were more pronounced for those with freezing in the upper limb. 15 Long-term tDCS-enhanced training effects were found for the consolidation of both cognitive abilities 16 as well as for gait performance when stimulation was combined with single 17 and multiple 18 training sessions. However, the mechanisms of these effects are still illusive.
The literature also presents neurophysiological investigation of clinical performance. Overall, cortical excitability characteristics in PD, as measured by single and paired-pulse transcranial magnetic stimulation (TMS), have been shown to be variable (ie, increased or decreased compared with healthy controls [HCs]) as well as dependent on medication stage and clinical profiles. 19 Variability in TMS outcomes may also be a result of differences in the TMS stimulation protocols and parameters used. 20 Studies examining the effects of anodal tDCS (atDCS) during rest found increased motor-evoked potential (MEP) amplitudes in PD, 12 specifically when applied to the most-affected hemisphere. 14 No significant changes in MEP size were found when patients received gait training after 13 minutes of tDCS or sham stimulation. 21 Interestingly, in healthy individuals, increased MEP amplitudes were related to improved retention of newly acquired skills after tDCS. 22 No work has been done on this in PD. Work examining cortical silent period (CSP) and short-latency intracortical inhibition (SICI) in early PD, without testing learning conditions, revealed that inhibition was reduced, specifically in the most affected hemisphere. 23 However, the meaning of the observed decrease in SICI remains controversial and difficult to interpret because it was shown that in PD, increased facilitation may be an indirect mechanism that exists on top of the direct influence of decreased intracortical inhibition. 24
So far, no studies exist that address the effects of tDCS combined with writing training in PD on consolidation and TMS outcomes. Hence, the objective of this pilot study was to examine tDCS-enhanced learning effects (ie, online and retention) and explore the associated alterations in cortical excitability in PD patients compared with HCs. Intervention effects were assessed in different tasks using various writing patterns of different sizes to test the transfer of motor learning. Based on previous studies, it was hypothesized that atDCS would enhance compensatory brain activity via immediate increased M1 excitability, resulting in improved writing performance, transfer, and retention compared with sham in both groups.
Methods
Participants
Consecutive samples of 10 PD patients and 10 age-matched HCs were included. A comprehensive description of all inclusion and exclusion criteria can be found in Appendix A (available online). In brief, we included patients from Hoehn and Yahr stage II and III, who were right-handed and right disease–dominant. All PD patients were tested in the on phase of the medication cycle, about 1 hour after the last drug intake, on all occasions. To ensure a stable on state, items 3.4 (finger tapping) and 3.7 (toe tapping) of the Movement Disorders Society sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) III were assessed on each occasion. A deviation of 1 point compared with baseline measurement was allowed, and no greater differences were found (Appendix Table D.1). Part of this study was described in a previous article. 15 The kinematic analysis of writing performance together with the neurophysiological TMS outcomes are reported here for the first time.
The study design and protocol were approved by the local Ethics Committee of the KU Leuven, in accordance with the Code of Ethics of the World Medical Association 2013 (Declaration of Helsinki). The trial was registered as ClinicalTrials.gov Protocol Record NCT02287207.
Experimental Design and General Setup
This pilot study consisted of a sham-controlled randomized crossover design. All participants started with a baseline test session on day 1 (Figure 1). This included assessment of their overall cognitive status (Montreal Cognitive Assessment), 25 mood (Hospital Anxiety and Depression Scale), 26 and a subjective interpretation of their writing deficits (MDS-UPDRS question II.7). 27 Additionally, PD patients completed the MDS-UPDRS part III. 27 Baseline writing performance on a tablet and paper was assessed using the setup indicated in Figure 1 (test 1).
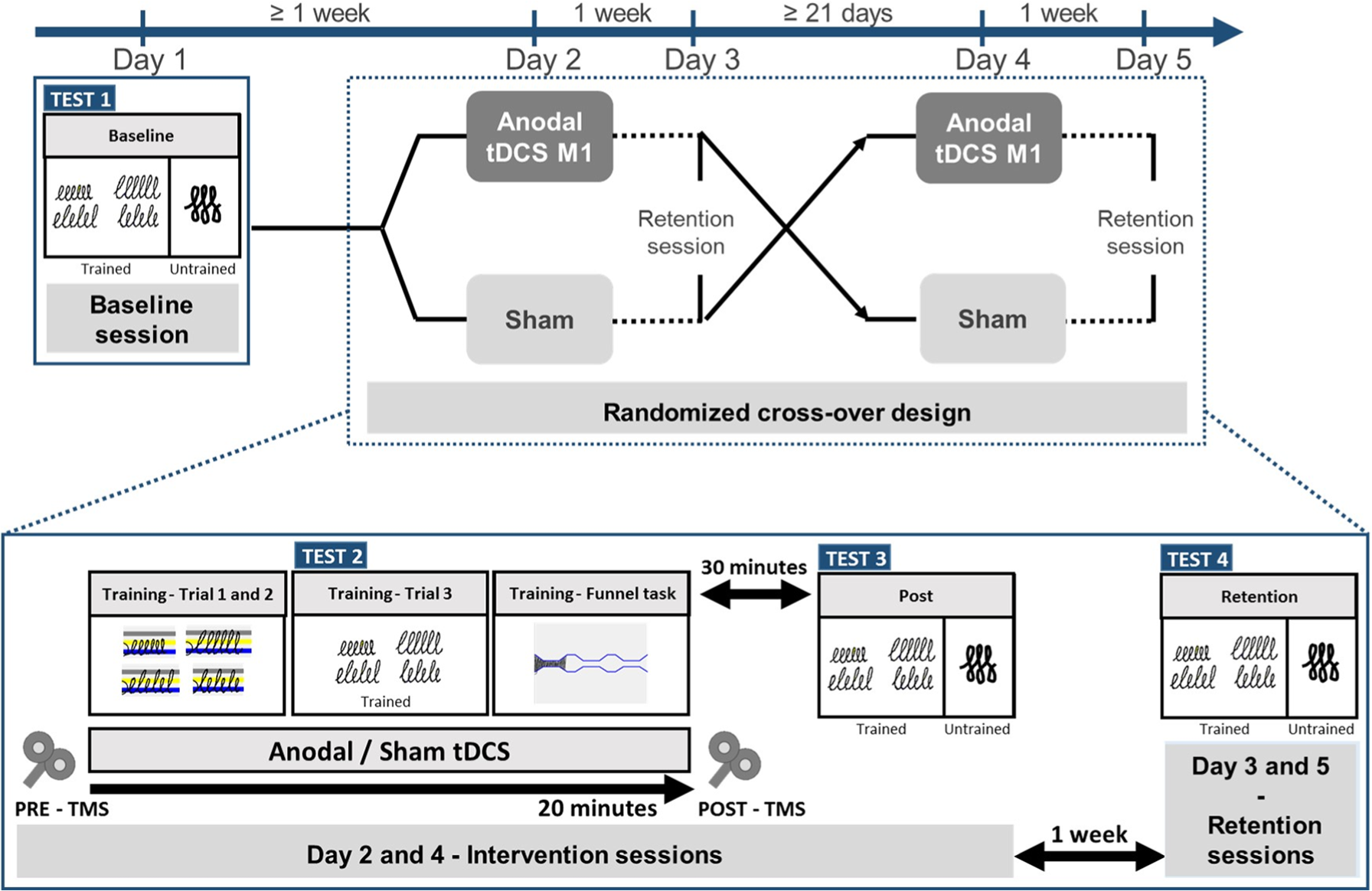
Using a randomized cross-over design each participant was tested in 5 separate sessions, including 1 baseline session, 2 intervention sessions and 2 retention sessions. During the intervention sessions participants received tDCS or sham stimulation in a randomized order. Writing performance during writing without visual cues was assessed during 4 test moments: (i) Test 1 baseline, (ii) Test 2 during the training (i.e. trial 3) combined with stimulation, (iii) Test 3 measured 30 minutes after the intervention and (iv) Test 4 during the retention sessions. The trained sequences were performed during Test 1, 2, 3 and 4 and the untrained sequences during Test 1, 3 and 4. Improvement percentages of all writing parameters were calculated relative to writing during baseline to compare the performances between the tDCS and sham stimulation sessions.
Day 2 consisted of a writing training session with either atDCS or sham stimulation. Immediately before and after the intervention, TMS outcomes were collected. Writing performance was assessed both during and after the intervention session (Figure 1, tests 2 and 3). After a 1-week retention period, writing performance was tested again at a follow-up session (Figure 1, test 4). Following a wash-out period of 3 weeks, 28 all participants underwent the same protocol again with the alternative stimulation, except for the baseline tests.
Writing Tests and Intervention
Writing performance was measured on a 6.4-inch writing tablet. 29 Tests included writing of trained (continuous and alternating) and untrained sequences without cues at a comfortable speed (Figure 1). Outcome parameters were amplitude (percentage of target size), velocity (cm/s), and the coefficients of variation of the within-subject writing amplitude (COVampl) and velocity (COVvel). Additionally, writing on paper with a regular pen (ie, mean writing size [mm], writing velocity, and writing quality) was assessed at baseline, post, and retention using the Systematic Screening of Handwriting Difficulties test (SOS test). 30
The training was performed as depicted in Figure 1, test 2, and consisted of practicing the writing of loops in different patterns (continuous and alternating) and sizes (0.6 and 1.0 cm) in a randomized order during 3 trials (each 2 minutes 24 s). All participants were instructed to write with an amplitude as required at a comfortable speed. The requested writing amplitude was indicated by colored target zones with a bandwidth of 2 mm during the first 2 trials. Because PD patients tend to become dependent on the learning context 31 and have difficulty with retaining a newly formed or adapted internal model of the movement, 8 target zones disappeared after 3 s in the third trial to facilitate this process (Figure 1, test 2). This third trial was used for online assessment. The last part of the training consisted of the funnel task (5 trials of 1 minute each), described in a previous article, 15 to stimulate transfer of training.
The full duration of writing training was combined with stimulation (DC-Stimulator, NeuroConn GmbH, Germany) consisting of 1 mA atDCS or sham delivered for 20 minutes via a pair of saline-soaked electrodes (surface 35 cm2, current density ±0.03 mA/cm2). Because upper-limb deficits, including handwriting, represent a typical dysfunction of the basal ganglia motor circuits for which M1 is considered an important output node,32,33 stimulation was targeted for this area with the anode placed over the left M1. The cathode was placed over the right supraorbital area. During stimulation, the mean contact impedance was 5.8 ± 1.6 kΩ for all participants. 15 The estimation of tDCS-induced electric fields was not included in the current study. To check the blinding procedure, a Visual Analogue Scale (VAS) was administered, evaluating participants’ perception of how strongly they felt the stimulation on a 10-point scale. Details of data processing of the writing parameters can be found in Appendix B.
Cortical Excitability Assessment
A detailed description of TMS procedures, including hotspot replication and data processing, can be found in Appendix C. Cortical excitability was assessed by (1) resting motor threshold (RMT; ie, lowest stimulation intensity eliciting MEPs with an amplitude > 50 µV in at least 5 of 10 consecutive TMS pulses) and (2) single-pulse MEP amplitude at rest (MEPrest) measured with 10 TMS pulses, whereby the stimulation intensity (percentage maximum stimulator output [%MSO]) was set to evoke MEPs with a 1 mV peak-to-peak amplitude when the muscle was at rest prior to the measurement. This stimulation intensity (%MSO) was kept constant during the experimental session. 12 Additional outcomes were MEP size during contraction (MEPactive), CSP, and SICI. For the MEPactive and CSP, participants maintained a constant 20% of the maximum voluntary APB contraction measured with a load cell (LLB350, Futek) while 10 unconditioned TMS pulses were applied with an intensity of 120% of the RMT. SICI was measured at rest using a paired-pulse paradigm consisting of a block of 10 unconditioned pulses and 10 conditioned pulses. During the paired-pulse measurements, the test stimulus, with an intensity adjusted to 1 mV MEP, was preceded by a subthreshold conditioning stimulus with an intensity of 90% of the RMT and an interstimulus interval (ISI) of 2 ms.23,34
Statistical Analysis
All participants were included in the statistical analysis that was conducted using SPSS software (version 24 SPSS, Inc, Chicago, IL) with significance levels of P <.05. Clinical characteristics between PD patients and HCs were compared using appropriate parametric or nonparametric tests. A Wilcoxon signed-rank test was used for investigating blinding of the participants as assessed by VAS scores. Differences in writing performance between PD patients and HCs at baseline were compared with the independent-samples t-tests.
Our primary analysis looked into the intervention effects on writing performance using linear mixed-effect models with baseline writing ability (MDS-UPDRS question II.7) as a covariate. Significant interactions were explored by Bonferroni’s multiple comparison post hoc tests. More specifically, analysis of the trained tasks included Group (PD or HC), Condition (atDCS or sham), Time (during stimulation; ie, trial 3 of writing loops, post or retention), and Size (small or large) as fixed factors. For the untrained writing task and SOS test, fixed factors were Group, Condition, and Time (post or retention). At a secondary level, the effects of the intervention on TMS outcomes were investigated. The post/pre ratios of MEP sizes (ie, MEPrest and MEPactive) were analyzed using linear mixed-effect models with Group and Condition as fixed factors. Linear mixed-effect models for all other TMS outcomes (ie, RMT, CSP, and SICI) included as fixed factors Group, Condition, and Time (pre or post).
To assess associations between improved writing performance (relative to baseline) and increased cortical excitability (ie, post/pre ratios of RMT and MEPrest and MEPactive), point biserial correlations were used for both groups separately. For this analysis, binary scores were calculated for the RMT and MEP values (yes, if the ratio was >1, and no if the ratio was ≤1). For the correlations that included outcomes of the trained task, the average performance of the 2 sizes (ie, small and large) for each pattern (ie, continuous and alternating) was used.
Results
Clinical Outcomes
There were no dropouts, and no adverse events occurred. Comparing VAS scores after tDCS and sham revealed no significant difference between conditions in either group (z = 1.332; P = .183). When analyzing the baseline clinical profiles of PD patients and HCs, PD patients reported more writing deficits as measured with question II.7 of the MDS-UPDRS (U = 1.0; P < .001). Groups were otherwise comparable for age, gender, cognition, and mood (Table 1).
Participant Characteristics. a
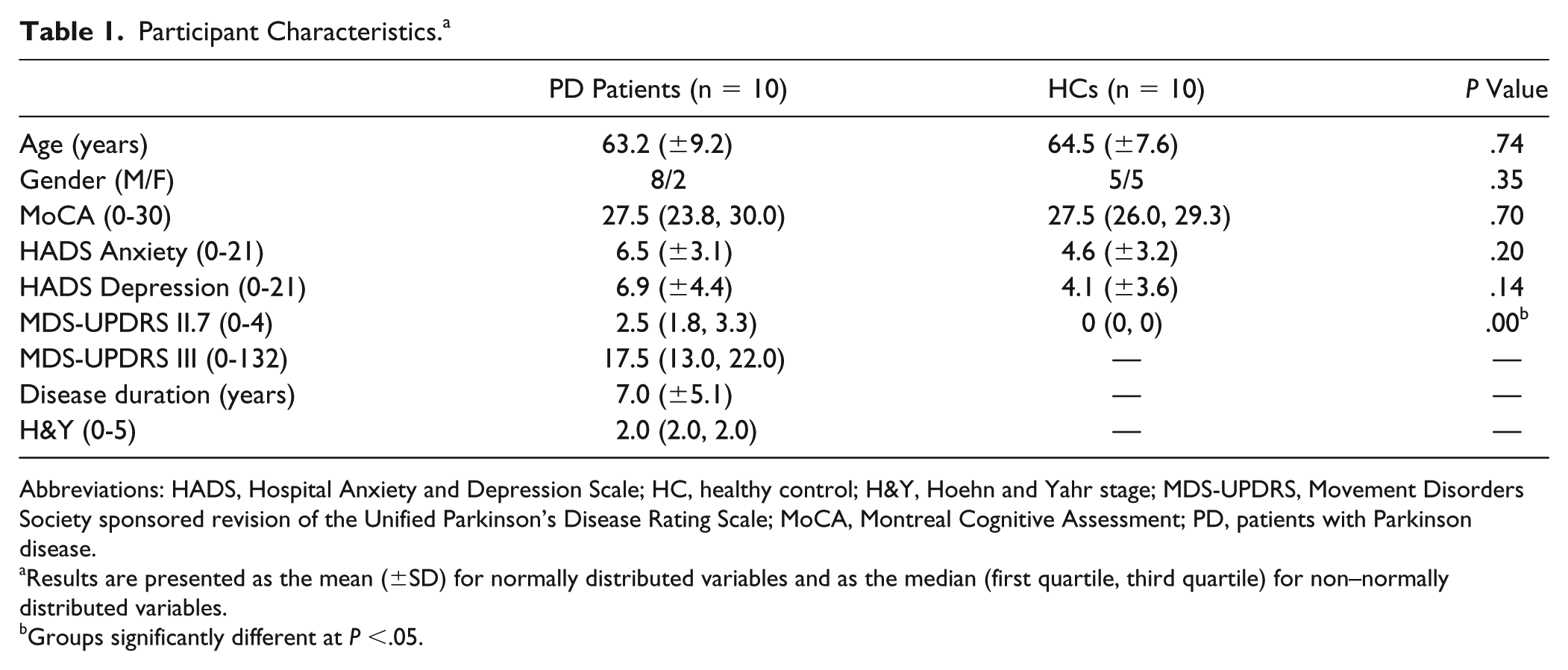
Abbreviations: HADS, Hospital Anxiety and Depression Scale; HC, healthy control; H&Y, Hoehn and Yahr stage; MDS-UPDRS, Movement Disorders Society sponsored revision of the Unified Parkinson’s Disease Rating Scale; MoCA, Montreal Cognitive Assessment; PD, patients with Parkinson disease.
Results are presented as the mean (±SD) for normally distributed variables and as the median (first quartile, third quartile) for non–normally distributed variables.
Groups significantly different at P <.05.
Writing Performance on the Tablet
An overview of all variables extracted from the writing tablet is provided in Appendix Tables D.2 to D.4.
Writing Amplitude
At baseline, PD patients wrote smaller compared with HCs for all tasks on the tablet, albeit not significantly. Comparing intervention effects between patients and HCs for the continuous and alternating pattern of the trained task showed significant Group × Condition × Time interactions [continuous: F(2, 176) = 3.347, P = .037; alternating: F(2, 176) = 3.356, P = .037]. For both patterns, a significant Condition × Time interaction was found for PD patients [continuous: F(2, 88) = 3.551, P = .033; alternating: F(2, 88) = 3.850, P = .025] but not for HCs (Figures 2A and 2B). Post hoc analysis revealed that patients wrote significantly larger during tDCS (continuous: P = .014; alternating: P = .010) and at retention measurements after tDCS (continuous: P = .005; alternating: P = .001) compared with sham. Also, writing amplitudes of PD patients after tDCS were significantly larger at retention compared with posttesting (continuous: P = .049; alternating: P = .017; Figures 2D and 2E). The writing amplitude of the untrained task also showed a significant Group × Condition × Time interaction [F(1, 48) = 4.363; P = .042]. There was a significant Condition × Time interaction for PD patients only [F(1, 24) = 11.616; P = .002]. Post hoc tests comparing amplitudes after tDCS and sham revealed that when atDCS was applied, patients wrote significantly larger at both post and retention measurements (respectively, P = .029 and P = .0005; Figure 2F).
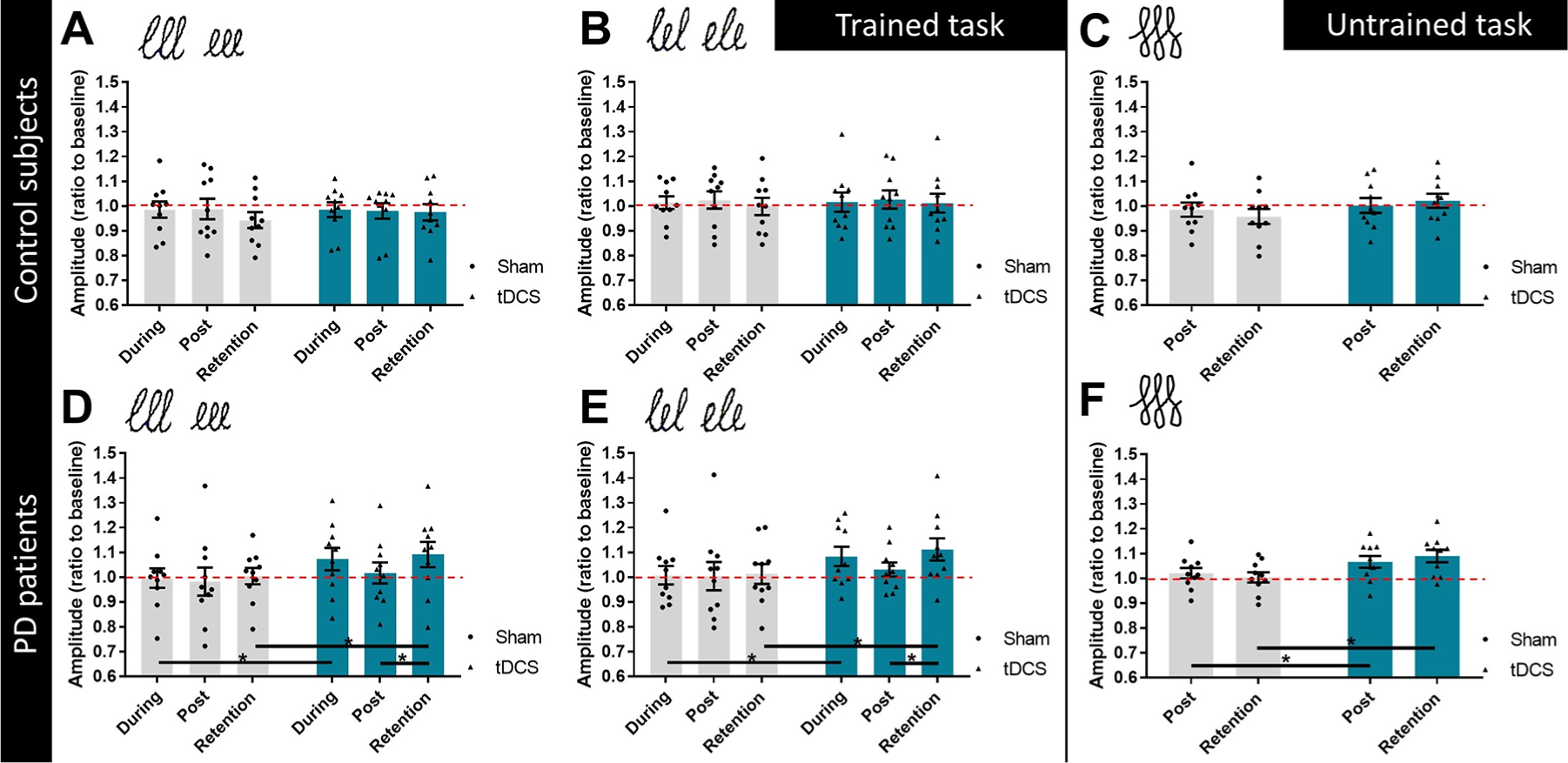
Writing amplitude (ratio to baseline) for healthy controls (A-C) and PD patients (D-F) during sham stimulation and anodal tDCS (black dots and triangles, respectively). Data are presented as group means (± standard error of the mean) as well as individual data points. Values above 1 (dotted line) demonstrate a larger writing amplitude compared to baseline performance. (A) Writing amplitude of control subjects during the continuous pattern of the non-cued trained task, (B) Writing amplitude of control subjects during the alternating pattern of the non-cued trained task, (C) Writing amplitude of control subjects during the non-cued untrained task, (D) Writing amplitude of PD patients during the continuous pattern of the non-cued trained task, (E) Writing amplitude of PD patients during the alternating pattern of the non-cued trained task, (F) Writing amplitude of PD patients during the non-cued untrained task. * Indicates p-value < 0.05 (post hoc tests).
Writing Velocity
Compared with HCs, PD patients wrote slower for all writing tasks on the tablet at baseline, though not significantly. Analysis of intervention effects of the trained task revealed significant Group × Condition interactions [continuous: F(1, 176) = 12.399; P = .001; alternating: F(1, 176) = 5.819; P = .017]. Post hoc analysis demonstrated that for the continuous pattern, patients wrote significantly faster in the tDCS condition compared with sham (P = .007). For the alternating pattern, post hoc results were not significant. No significant interactions were found for the writing velocity of the untrained task.
Coefficients of Variation of the Within-Subject Amplitude and Velocity
At baseline, PD patients showed generally higher COV values for both the amplitude and velocity compared with HCs, although differences between groups were only significant for the COVampl of the small and large letters of the alternating pattern of the trained task (respectively, t(18) = 0.776, P = .024, and t(18) = 2.201, P = .001). Results of the linear mixed-model analysis revealed significant Group × Size interactions for the COVampl and COVvel of the continuous pattern of the trained task (respectively, F(1, 176) = 4.360, P = .038, and F(1, 176) = 7.010, P = .009). A lower variability for the large loops compared with the small loops was found post hoc for the COVampl of PD patients (P = .029). No significant interactions were found for the COVampl and COVvel of the alternating pattern of the trained task.
For the untrained task, a significant Group × Condition × Time interaction [F(1, 48) = 5.368; P = .025] was found for the COVampl. There was a significant Condition × Time interaction for PD patients only [F(1, 24) = 4.392; P = .047]. Post hoc testing showed a significantly lower variability of amplitude after tDCS at retention measurements compared with sham (P = .025; Figures 3A and 3B). There was also a significant Condition × Time interaction [F(1, 48) = 6.513; P = .014] for the COVvel of the untrained task, though without significant differences post hoc.
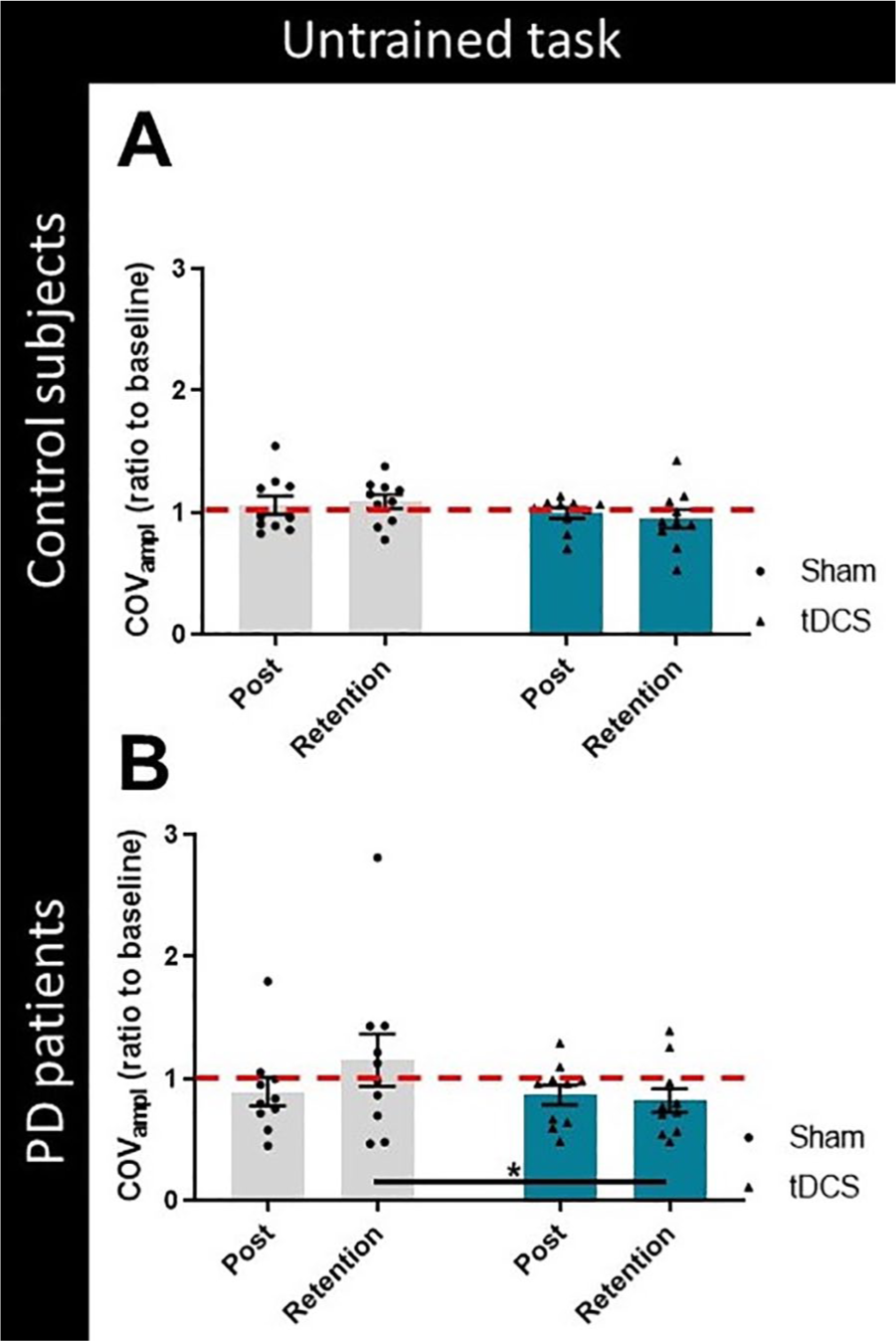
Coefficient of variation of the within-subject stroke amplitude (ratio to baseline) during the non-cued untrained task for healthy controls (A) and PD patients (B) during sham stimulation and anodal tDCS (black dots and triangles, respectively). Data are presented as group means (± standard error of the mean) as well as individual data points. Values below 1 (dotted line) demonstrate improved writing variability compared to baseline performance. * Indicates p-value < 0.05 (post hoc tests).Abbreviations: COVampl, coefficients of variation of the within-subject writing amplitude; PD, Parkinson disease; tDCS, transcranial direct current stimulation.
Writing Performance on Paper
At baseline, PD patients wrote smaller and slower and with worse writing quality (ie, SOS score) compared with HCs, although it was not statistically significant. Comparing the intervention effects, a nonsignificant Group × Condition interaction [F(1, 48) = 3.363; P = .073] was found for writing scores. Exploring this result in more detail because of its clinical significance, a post hoc analysis showed improved quality (ie, lower scores indicate improved writing quality) for PD patients after tDCS compared with sham stimulation (P = .020; Figures 4A and 4B). No significant interactions were found for the writing size and velocity of the SOS test. An overview of the results is provided in Appendix Tables D.2 to D.4.
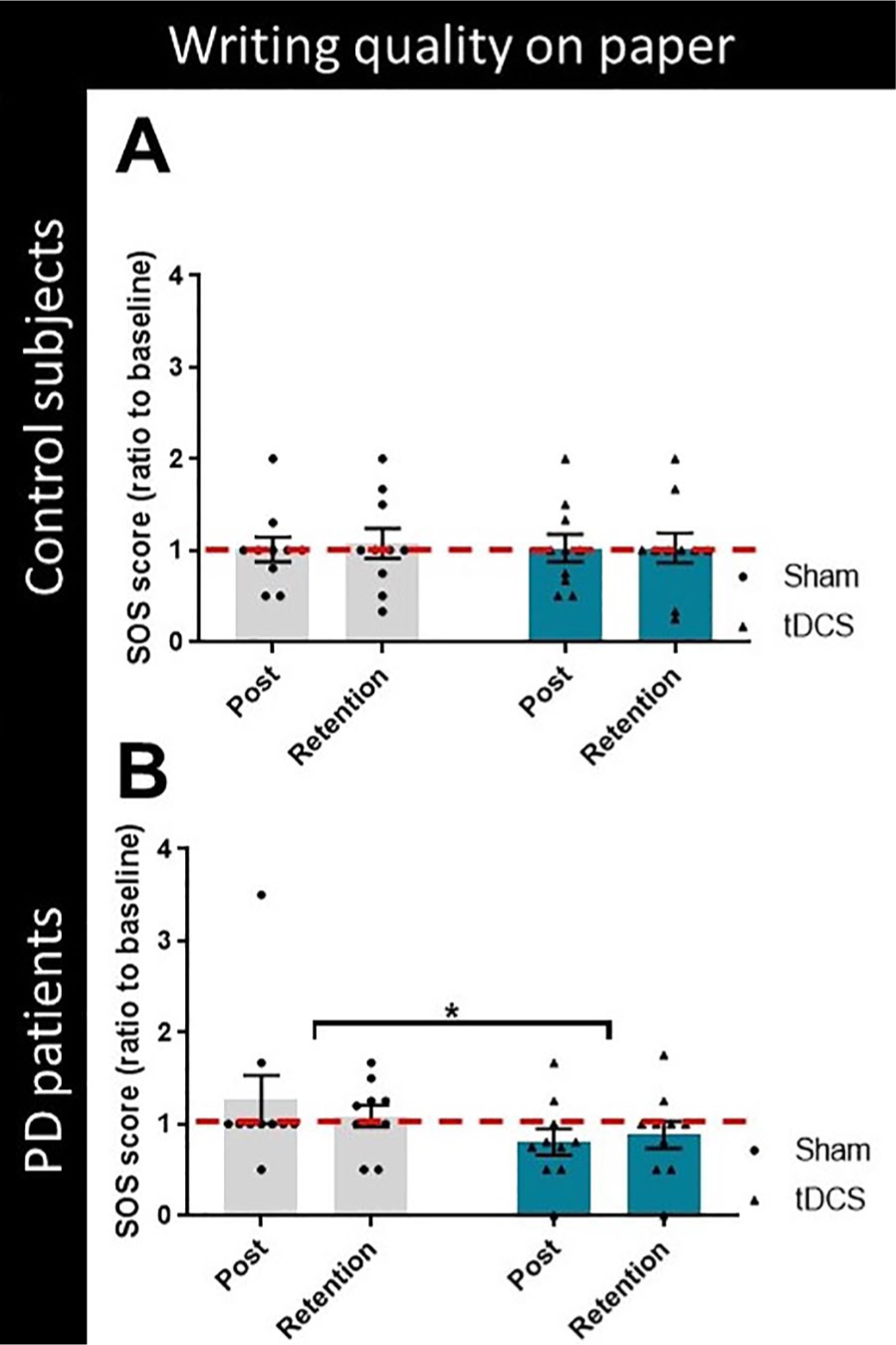
Writing scores on paper (ratio to baseline) for healthy controls (A) and PD patients (B) during sham stimulation and anodal tDCS (black dots and triangles, respectively). Lower writing scores indicate improved writing quality. Data are presented as group means (± standard error of the mean) as well as individual data points. Values below 1 (dotted line) demonstrate improved writing quality on paper compared to baseline performance. * Indicates p-value < 0.05 (post hoc tests). Abbreviations: PD, Parkinson disease; SOS, Systematic Screening of Handwriting Difficulties; tDCS, transcranial direct current stimulation.
Cortical Excitability
Cortical excitability did not significantly differ between patients and HCs at premeasurements. Results for RMT revealed a significant Condition × Time interaction [F(1, 54) = 8.346; P = .006]. Post hoc analysis showed a reduction in RMT immediately after tDCS compared with sham for all participants (P = .007). For all other TMS outcomes, no significant interactions were found. A complete overview of cortical excitability results is provided in Appendix Table D.5.
Correlations
Point-biserial correlation analysis showed significant correlations for PD patients between increased excitability as measured with MEPrest and improved writing amplitudes of the untrained task after sham stimulation (ie, postintervention; rpb = 0.755, P = .024). In PD, similar correlations were found between increased MEPactive values and improved writing velocity of the trained alternating pattern after sham at post and retention measurements (both rpb = 0.802; P = .005). In contrast, for HCs, improved writing amplitudes of the trained task were negatively correlated with increased cortical excitability at rest (ie, MEPrest) during tDCS stimulation (continuous and alternating pattern: rpb = −0.816; P = .004) as well as after tDCS at postmeasurement (alternating pattern: rpb = −0.667; P = .035). No significant correlations between writing performance and RMT were found for both groups.
Discussion
The present sham-controlled study was designed to determine the possible effects of tDCS-enhanced writing training on consolidation of learning and its association with alterations in cortical excitability in PD patients compared with HCs to inform a future larger study. We found significant evidence that tDCS improved online writing performance, retention, and transfer in PD patients. This finding is striking given the striatal deficiencies that impair the robustness of motor learning in this patient group. 8 In addition, tDCS was associated with reduced RMT, though this did not result in significant alterations of other cortical excitability outcomes. Intriguingly, increased excitability after sham correlated with improved writing performance in PD patients, whereas in HCs, the same increased excitability correlated with worse writing performance. Together, these novel findings hint at different underlying processes through which tDCS-supplemented relearning of writing is achieved in PD versus age-matched HCs.
Affected Handwriting in PD and tDCS-Enhanced Writing Skill Learning
Handwriting is a habitual but complex skill that requires precise coordination, adequate visuomotor control, and an automatic generation of sequences. Although micrographia is a well-known problem in PD, handwriting difficulties extend beyond amplitude alone, frequently referred to as dysgraphia. 35 In this study, we found that writing parameters tended to be worse at baseline compared with HCs, illustrating mild dysgraphia consistent with mild disease. We examined to what extent 1 session of training combined with tDCS affected the different phases of motor learning. Stimulation was always applied to the most-affected (left) M1, a primary region of interest for both writing and motor learning.9,14,15
In line with our hypothesis, we found that M1-tDCS resulted in improved writing performance during stimulation as well as enhanced retention and transfer of writing skills compared with sham. More specifically, during tDCS, patients showed increased writing amplitude and speed. Few studies examined online tDCS effects in PD, and they showed variable results. Whereas enhanced working memory 36 and gait 17 were reported previously, sequence learning during the serial reaction time task did not improve during tDCS compared with sham. 37 This disagreement may be explained by paradigm differences, because the former studies as well as the present one required focusing on either a motor or a cognitive component, whereas the serial reaction time task involves attending to both. In healthy individuals, beneficial effects of anodal M1-tDCS were found for manual dexterity, also suggesting a task-dependent effect. 38 In the present pilot study, patients showed increments in writing speed during tDCS, but not immediately after, possibly explained by differences in on- and offline learning mechanisms. The effects of tDCS during stimulation were earlier suggested to depend on changes in membrane potentials, 39 whereas the modification of NMDA-receptor sensitivity, cortical neurotransmitter concentration changes, and alterations in transmembrane proteins were thought to be responsible for aftereffects.40-42 However, at 1-week retention, sustained tDCS-enhanced performance was found for writing amplitudes and variability for both the trained and untrained patterns as well as for writing on paper in PD. This suggests that processing of consolidation was ongoing and that learning gains were stored more efficiently in motor memory under the impetus of tDCS. The mechanisms underlying improved retrieval of motor encoding even during untrained tasks are presently illusive. Recently, it was shown that bilateral atDCS increased brain-derived neurotrophic factor (BDNF) serum levels together with improved motor function in PD, 43 a result that was also found after intensive rehabilitation. 44 BDNF may play a role in processes that are involved in synaptic efficacy linked to neuroplasticity (i.e. long-term potentiation (LTP) and long-term depression) 45 and possibly explain the observed retention and transfer effects after tDCS in the current study. However, this assumption needs to be examined in future work. Remarkably, the aftereffects in this study were found after only 1 training session. Previous research in PD has shown that improved motor performance is maintained for 3 months following multiple stimulation sessions.13,18 This suggests that repeated application of tDCS may induce even greater efficacy. 46
Finally, the finding that stimulation was only significantly beneficial for PD patients is somewhat remarkable in the light of earlier findings of abnormal motor cortex excitability and reduced capacity for LTP compared with HCs, implying less potential for motor learning. 23 This could have 2 explanations. First, the effects of tDCS could have had an extra beneficial effect and reversed these abnormalities. Second, because writing is often a well-consolidated skill, the writing paradigm may have been too easy for HCs, resulting in ceiling effects or preventing incremental learning effects by a single session of training combined with tDCS in this group.
The Effects of tDCS on Cortical Excitability
Contrary to previous research,19,23,47 we found no significant differences in motor cortical excitability and inhibition between PD patients and age-matched HCs at baseline. The fact that patients were tested on medication could explain these results. Earlier studies showed that TMS outcomes were normalized by dopaminergic medication as well as by the expectancy of receiving treatment (ie, placebo effect).24,48,49 The current study did highlight a significantly lower RMT after learning with atDCS compared with sham in all participants. This lower RMT is partially consistent with our hypothesis that atDCS would enhance M1 excitability. However, other excitability outcomes were not significantly modified, and no differences in tDCS aftereffects were found between PD patients and HCs. Most tDCS studies that investigated motor cortical excitability did not include an assessment of RMT, although 1 study found RMT changes after 1 session of repetitive TMS over the supplementary motor area together with improved writing performance in PD 50 and another after atDCS in stroke patients. 51
For other TMS outcomes, several studies found increased MEP amplitudes and decreased inhibition (reduced SICI and CSP) together with beneficial effects on behavior after anodal M1-tDCS in healthy individuals52-54 and even more so in older adults. 55 Studies examining the effects of atDCS on cortical excitability in PD also showed increased MEP sizes,12,14 though not consistently.21,56 Similar to our results, no tDCS aftereffects on SICI were found when combining tDCS with treadmill walking. 57 This may underscore that tDCS has no impact on inhibitory mechanisms. Another possible explanation for the negative outcome on inhibition is the large variability in response to tDCS and TMS protocols, as described for healthy individuals 58 and patients with PD. 59 We were able to recruit a relatively homogeneous group of patients, and tDCS was always applied to the most-affected hemisphere. 14 However, using the current TMS protocol, this study was probably underpowered to reveal true facilitatory or inhibitory effects on motor cortex excitability.
Interestingly, correlation analysis showed different associations between increased cortical excitability and writing performance for patients and HCs, suggesting a distinct response to training. In PD patients, increased cortical excitability after sham was correlated to improved writing performance, whereas the opposite was found for HCs. Although the associations found are preliminary, we speculate that increased M1 excitability may signify a compensatory mechanism 23 for the loss of striatal function in PD patients, not seen in HCs.
Implications for Neurorehabilitation
The present results point favorably to the ability for tDCS to boost motor learning. Overall, tDCS is relatively easy to apply and has good acceptance rates in clinical populations.10,28 Therefore, implementation is theoretically feasible both in a clinical setting and during home practice. In line, telemonitoring of a home tDCS application is currently being tested in a clinical trial in PD. 60 However, before clinical embedding is considered on a wide scale, more information is needed on the safety and risks of tDCS, especially across application of multiple sessions. Furthermore, larger studies are needed to corroborate the present findings, thereby minimizing the risk for type I or II errors.
Study Limitations
The results obtained with our TMS paradigm need to be interpreted with caution because the number of MEPs used to measure cortical excitability and inhibition was below the required 20 or 26 for single- and paired-pulse TMS, respectively. 61 In addition, we used a fixed intensity of 90% of the RMT for SICI, whereas SICI responses were found to be dependent on the intensity of the subthreshold conditioning stimulus in both PD patients and HCs. 47 Future studies should measure the individual response to different intensities to estimate the SICI curve. 52 We used a stimulation intensity of 1 mA, which may not have been high enough to induce cortical excitability modifications. However, a recent meta-analysis of multiple tDCS studies did not show differences in cortical excitability after comparing 1-mA or 2-mA intensities. 62 Combining tDCS-enhanced training with dopaminergic medication is probably beneficial for sequence learning and consolidation as the ability to modify cortical connections is increased.63,64 Alternatively, medication effects may have masked the effects of tDCS on learning.
Using a crossover design is advantageous for small sample sizes with considerable individual variability. However, the lack of prewriting tests conducted during the intervention sessions constituted a limitation of our design. Based on previous studies, the incorporation of a wash-out period of a minimum of 21 days should have been sufficient to negate learning and tDCS effects.9,10 Also, test-retest reliability of tDCS is not known for either PD patients or HCs (elderly). In young HCs, a fair intraindividual reliability has been reported for the effects of atDCS on TMS measures. 65 For future studies, we recommend inclusion of parallel designs with larger samples to control for the high variability of tDCS results, prebehavioral tests on the same day of training interventions, and distinguishing between on and off medication learning.9,20 Finally, our small sample size, including people with mild disease only, limits the ability to generalize our findings on the effect of M1-tDCS on motor learning consolidation in PD.
Conclusion
We found preliminary evidence for tDCS-enhanced writing performance in PD together with robust retention and transfer of learning even after 1 session. Alterations in cortical excitability were more equivocal but pointed to increased excitability after atDCS, specifically beneficial for PD patients rather than for HCs. These novel findings make a strong case for further study based on a more definitive study design into tDCS-mediated motor learning in PD and encourages clinicians to consider its implementation in future rehabilitation programs.
Supplemental Material
2019.08_-_Appendix-Pilot_tDCS_in_PD_Effects_on_Writing_Revision2_Clean – Supplemental material for tDCS-Enhanced Consolidation of Writing Skills and Its Associations With Cortical Excitability in Parkinson Disease: A Pilot Study
Supplemental material, 2019.08_-_Appendix-Pilot_tDCS_in_PD_Effects_on_Writing_Revision2_Clean for tDCS-Enhanced Consolidation of Writing Skills and Its Associations With Cortical Excitability in Parkinson Disease: A Pilot Study by Sanne Broeder, Evelien Nackaerts, Koen Cuypers, Raf Meesen, Geert Verheyden and Alice Nieuwboer in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank all participants for their commitment. We thank Ir Marc Beirinckx for the development of the writing tablet and for providing technical support.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
