Abstract
Introduction
Multiple sclerosis (MS) is a chronic neurodegenerative disease affecting the central nervous system (CNS) and consequently leading to physical and cognitive symptoms. 1 One of the most frequent and debilitating symptoms in MS is fatigue,2,3 which has been shown to affect daily activities and reduce quality of life.4-6 Because fatigue is a complex phenomenon, Kluger et al 7 have proposed a unified taxonomy that distinguishes between perceptions of fatigue and performance fatigability. Perceptions of fatigue are described as “subjective sensations of weariness, increasing sense of effort, mismatch between effort expended and actual performance or exhaustion.” (p. 411) 7 Performance fatigability is defined as “the magnitude or rate of change in a performance criterion relative to a reference value over a given time of task performance or measure of mechanical output,” (p. 411) 7 and when assessed in the motor domain, this is referred to as motor fatigability. 8
Studies have assessed fatigability during both functional movements (eg, decrease in walking speed) and isolated muscle contractions with dynamometry. 8 When classified according to the International Classification of Functioning, Disability and Health (ICF) model, fatigability has been assessed at both the body function level and at the activity level. At the activity level of the ICF model, the 6-minute walk test (6MWT) is the most commonly used method to evaluate walking-related fatigability, and it has been suggested as the gold standard.9,10 However recent reviews8,11 have shown that there is no gold standard for the assessment of fatigability on the body function level in MS. Moreover, a review by Severijns et al 8 revealed that protocols assessing fatigability at the body function level (n = 38) are more common than protocols focusing on the activity level (n = 11) of the ICF. This might be explained by the possibility of exploring underlying physiological mechanisms in the former. An interesting observation is that most studies investigating the underlying physiological mechanisms of fatigability are performed in isolated hand models (n = 17/23). 8 However, models involving the lower extremities might be more relevant for physical function and mobility, which is ranked as one of the most important bodily functions by persons with MS.3,12
Despite being an objectively measured phenomenon, fatigability on the body function level is measured by a great variety of dynamometer protocols in terms of type (isometric vs concentric), duration (seconds to minutes), number of contractions (fixed or until exhaustion), involved limb (upper extremities or lower extremities, dominant/nondominant, or weakest/strongest), and intensity (maximal or submaximal contractions).8,11 The most frequently used protocol applies isometric contraction(s) (n = 30/38), and the majority of these use sustained contraction protocols (n = 25/30). However, because peripheral muscular factors are a predominant limiting factor of muscle fatigability in healthy individuals13,14 and a contributor to muscle fatigability in the lower limbs of patients with MS during a sustained isometric contraction, 15 this approach might not be optimal in reflecting fatigability related to a disease in the CNS, such as MS. Furthermore, most functional tasks are dependent on the muscular power output of the lower extremities, which is left out in an isometric protocol because no contraction velocity is involved. 16 Concentric protocols, especially in the lower extremities may, on this basis, be more strongly associated with performance in functional tasks. 8 Seamon and Harris-Love 17 suggest that when considering fatigability in a rehabilitation setting, it is specifically important that fatigability is associated with performance in functional tasks reflecting activities of daily living and perceptions of fatigue. Nonetheless, only a minority of studies have investigated these associations,18-22 and none of these studies compared fatigability induced by both isometric and concentric dynamometer protocols in the lower extremities, making it difficult to conclude which fatigability protocol shows superior association with the patient’s perception of fatigue impact and their ability to perform functional tasks, such as walking.
Therefore, the purpose of the present study was to (1) perform a head-to-head comparison of a sustained isometric and a concentric fatigability protocol in the lower extremities and (2) investigate the association between fatigability (ie, isometric vs concentric force decline) and measures of fatigue and walking. It was hypothesized that the force decline in the concentric protocol would show a stronger association with fatigue and walking than the sustained isometric protocol.
Methods
The present study was a cross-sectional study comparing 2 different fatigability protocols in ambulatory persons with MS. The study was approved by the ethics committee of Region Midtjylland (Journal No. 1-10-72-287-15), and all participants declared written consent prior to inclusion. Participants attended 2 separate test days. The first day of testing included familiarization with the isokinetic dynamometer followed by questionnaires of perceived fatigue impact (Modified Fatigue Impact Scale [MFIS]) and perceived walking ability (12-item Multiple Sclerosis Walking Scale [MSWS-12]). This allowed patients to rest before walking capacity was assessed by the 6MWT. On the second day of testing, participants performed 2 different fatigability protocols in a randomized order (see details below).
Participants
A total of 45 persons with multiple sclerosis participated in the study and were recruited from the MS Clinic of Southern Jutland, Denmark, the MS Clinic at Aarhus University Hospital, Denmark, and through the website of the Danish MS Society. Only ambulatory persons (Expanded Disability Status Scale [EDSS] ≤ 5.5) with a definite MS diagnosis according to the revised McDonald criteria 23 were included. Patients were excluded in case of a relapse within 8 weeks preceding the first test day. Additional exclusion criteria were pregnancy or comorbidities that could possibly affect perception of fatigue impact or performance.
Outcomes
Perceived Fatigue
The MFIS 24 was used to assess patients’ perception of fatigue impact. The MFIS is a modified version of the Fatigue Impact Scale, 25 and a higher score indicates a higher perception of fatigue impact.
Walking
Participants performed a maximal 6MWT in a 30-m hallway according to the instructions by Goldman et al. 26 The 6MWT test is a valid, reliable, and responsive outcome considered the gold standard for walking capacity in persons with MS.26,27 The MSWS-12 was used to assess the patient’s own perception of walking. This is a valid and reliable measurement of self-perceived walking ability, where a lower score indicates better walking abilities. 28
Motor Fatigability
While performing isokinetic dynamometry, participants were positioned in the dynamometer (Humac Norm, CSMi, Stoughton, MA) setup for knee extension, as described elsewhere. 29 In a random order, they performed a sustained isometric and a concentric fatigability protocol, with 2 hours rest between protocols. High intraclass correlation coefficients 30 have been shown for dynamometer testing of both concentric 31 and isometric 32 contractions in persons with MS.
Before performing the protocols, familiarization and warm-up exercises were performed. Prior to the isometric protocol, participants performed 5 s × 5 s isometric contractions at approximately 20%, 40%, 60%, 80%, and 100% of maximal voluntary contraction (MVC). Before the concentric protocol, 5 concentric contractions were performed at similar intensities. Just before and just after each protocol, participants were asked to perform an isometric MVC to which an interpolated doublet stimulation of musculus (m.) quadriceps was added by 2 electrodes (Valutrode, Axelgaard, Denmark) followed by a resting doublet stimulation in order to investigate the extent of peripheral and central fatigability. However, these mechanistic investigations are beyond the purpose of this article and will be presented separately. Therefore, the primary focus of this article is the 2 fatigability protocols depicted and highlighted in the methodological presentation (Figure 1). The isometric protocol consisted of a 2-minute sustained maximal isometric knee extension, whereas the concentric protocol consisted of 40 maximal isokinetic knee extensions at 30°/s and at a 90° range of motion (ROM; starting from 90° and ending at 0°). Between each repetition, the leg was passively brought back to the starting position, making the protocol intermittent. The 2 protocols were matched in work time, so that participants had a total muscle contraction time of 120 s in both protocols.
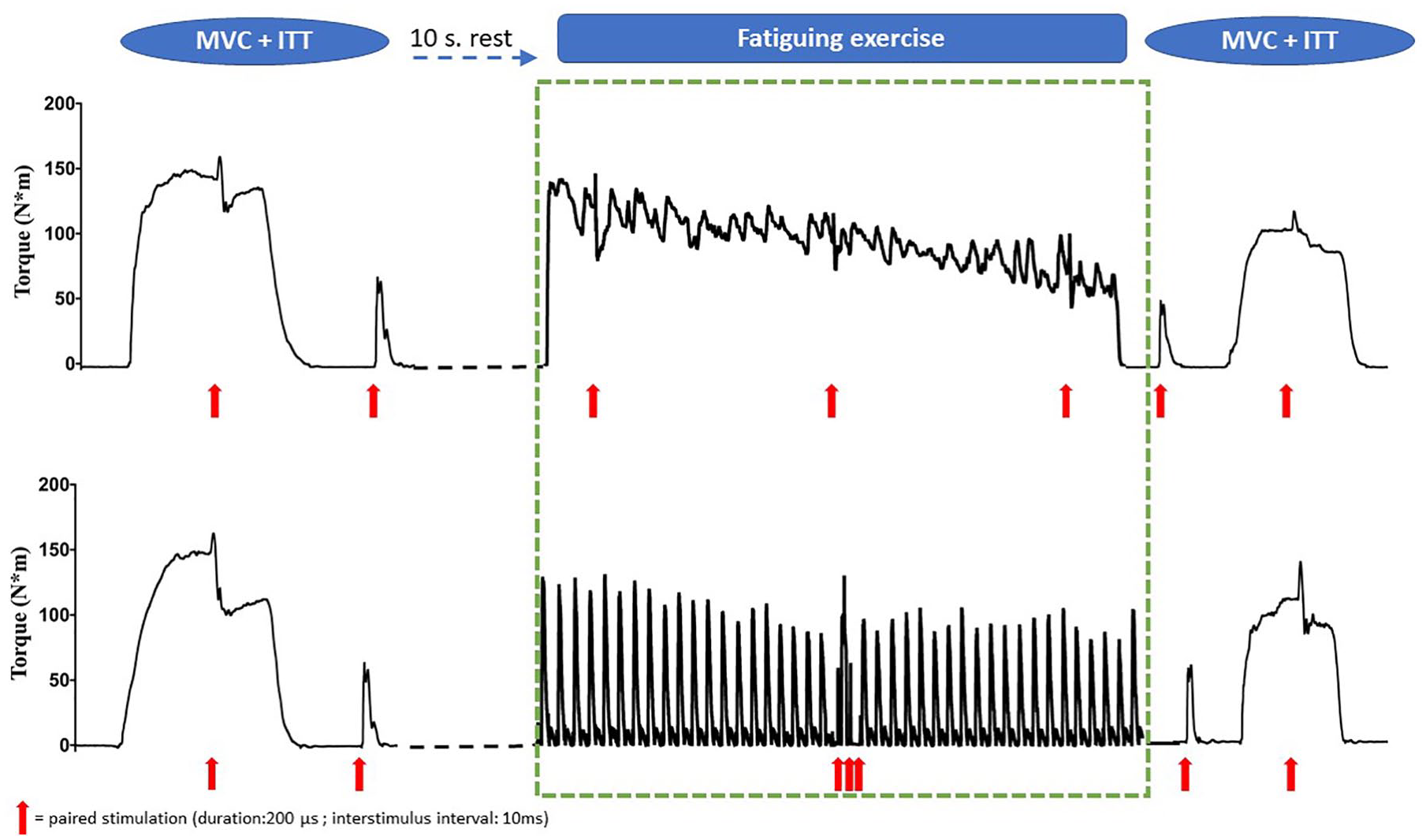
An overview of the 2 protocols, with the green stippled square indicating the part of the protocol that has been used in the present study.
All force data were sampled at 1500 Hz using a TeleMyo Direct Transmission System and MyoResearch Software (Noraxon, Scottsdale, AZ) and analyzed using custom-made software (LabView v 16.0, National Instruments, Austin, TX). Fatigability was expressed as a Fatigue Index (FI) for both the sustained isometric protocol (FIisometric) and the concentric protocol (FIconcentric). The FI is a quantification of the reduction in force output and is calculated as
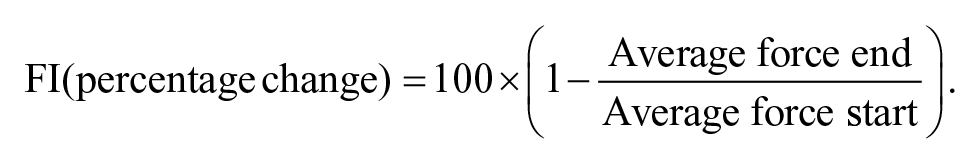
For the sustained isometric protocol, this was the average force of the last 6 s relative to the first 6 s. In the concentric protocol, it was calculated as the average peak force of the last 2 contractions relative to the first 2 contractions.
Statistics
The distribution of the data was assessed by visual inspection of QQ plots and histograms, and all data followed a normal distribution. To compare the 2 different fatigability protocols, the static isometric protocol was divided into 40 bouts of 3 s contraction time, and the average force of these was calculated. Each of these bouts, as well as each of the 40 contractions from the dynamic concentric protocol, was normalized to the “average force start” from the FI (as defined above).The level of maximal force-producing capacity prior to each fatigability protocol as well as fatigability in the 2 different protocols (FIisometric vs FIconcentric) were compared using a paired-samples
Participant Demographics and Measures of Strength, Fatigability, Fatigue, and Walking Ability.
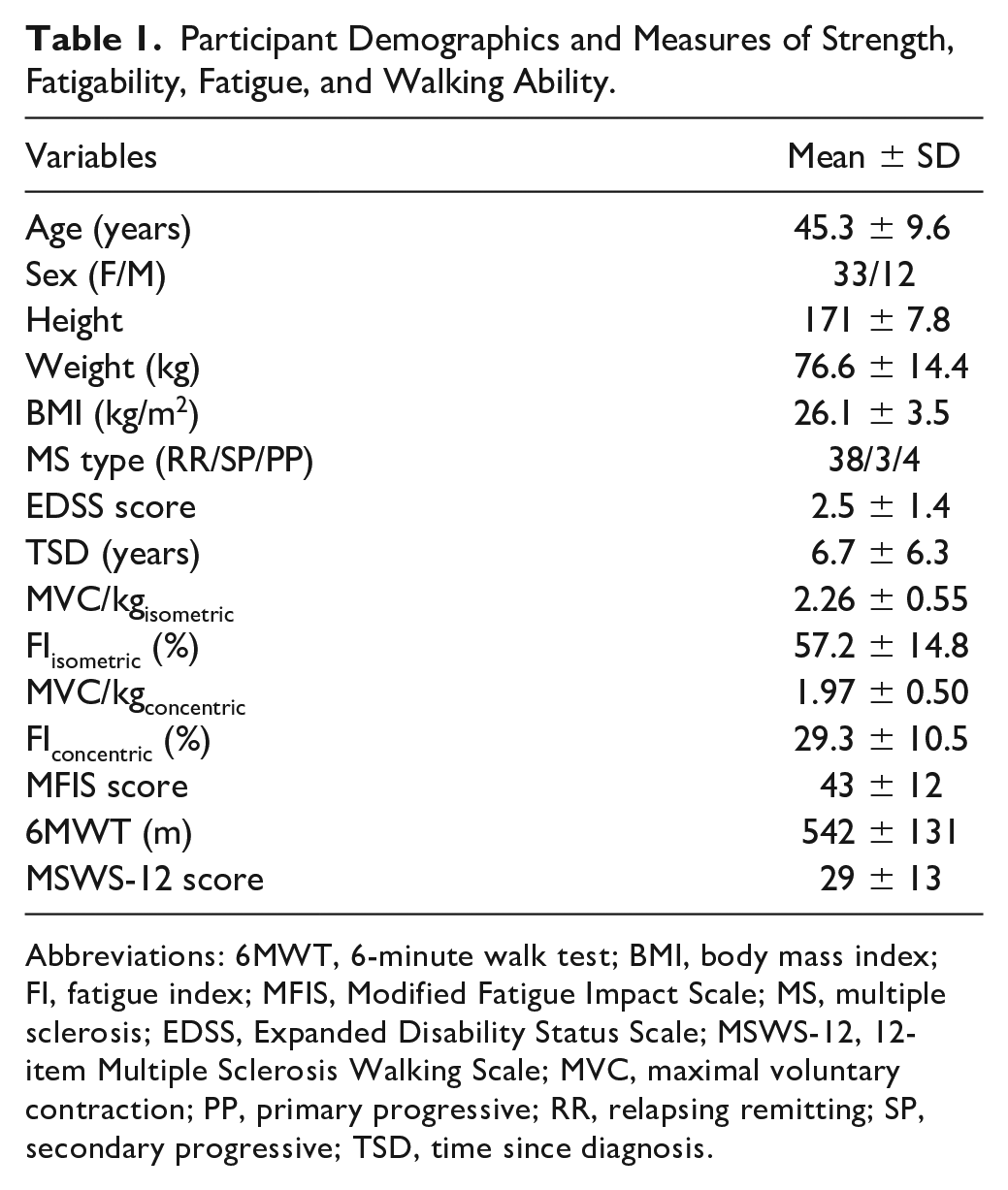
Abbreviations: 6MWT, 6-minute walk test; BMI, body mass index; FI, fatigue index; MFIS, Modified Fatigue Impact Scale; MS, multiple sclerosis; EDSS, Expanded Disability Status Scale; MSWS-12, 12-item Multiple Sclerosis Walking Scale; MVC, maximal voluntary contraction; PP, primary progressive; RR, relapsing remitting; SP, secondary progressive; TSD, time since diagnosis.
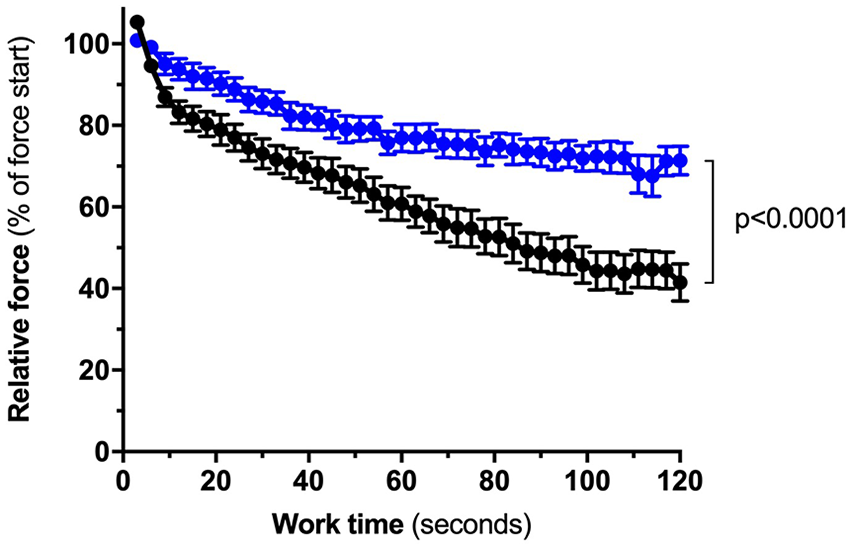
Lower-extremity fatigability induced by the sustained isometric protocol (black) versus fatigability induced by the intermittent concentric protocol (blue); (57.2% ± 14.8% vs 29.3% ± 10.5%, respectively).
Results
A total of 45 participants, 12 men and 33 women, were included in the study. The average age was 45.7 ± 9.3 years, and the average EDSS was 2.5 ± 1.4. The average MFIS score was 43 ± 12, indicating an overall fatigued group of patients according to the cutoff value of 38 suggested by Flachenecker et al. 33 Descriptive data for normalized MVC, FI, MFIS, 6MWT, and MSWS-12 can be found in Table 1.
Sustained Isometric Versus Concentric Fatigability
In the concentric protocol, 1 participant did not perform the test as instructed (ie, force increased substantially in the last 10 repetitions), and the data of another participant were not obtainable because of technical issues (leaving n = 43 for analyses). As seen in Figure 2, the mean FI was higher in the sustained isometric protocol than in the concentric protocol (57.2% ± 14.8% vs 29.3% ± 10.5%, respectively,
There were no significant differences in MVC prior to either of the protocols (
Fatigability and Perception of Fatigue Impact
There was no association between sustained isometric fatigability and fatigue impact (
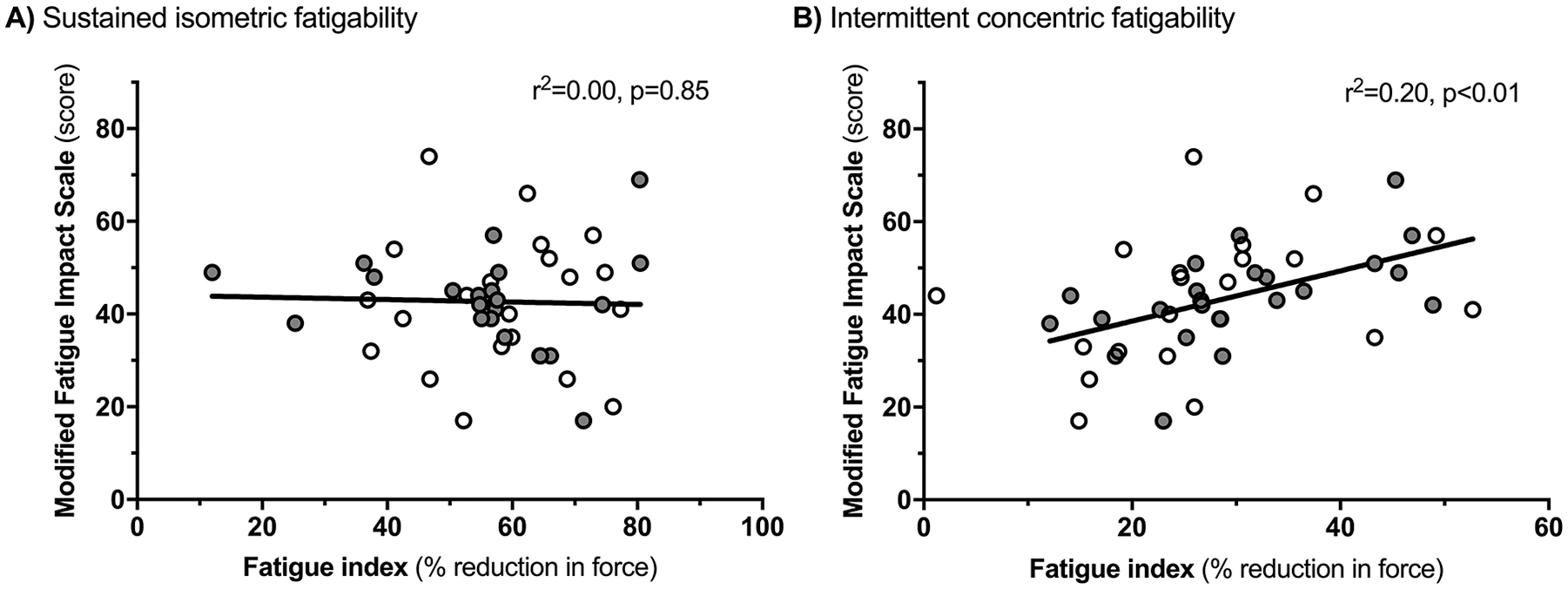
Associations between perception of fatigue impact and lower-extremity fatigability induced by (A) a sustained isometric protocol and (B) an intermittent concentric protocol. The gray circles indicate that this test was done as test 2 on the test day.
Simple Unadjusted (Model 1) and Multivariable Regression Analysis (Models 2 and 3) of the Associations Between Lower-Extremity Sustained Isometric or Concentric Fatigability and Fatigue (MFIS) and Walking (6MWT and MSWS-12). a
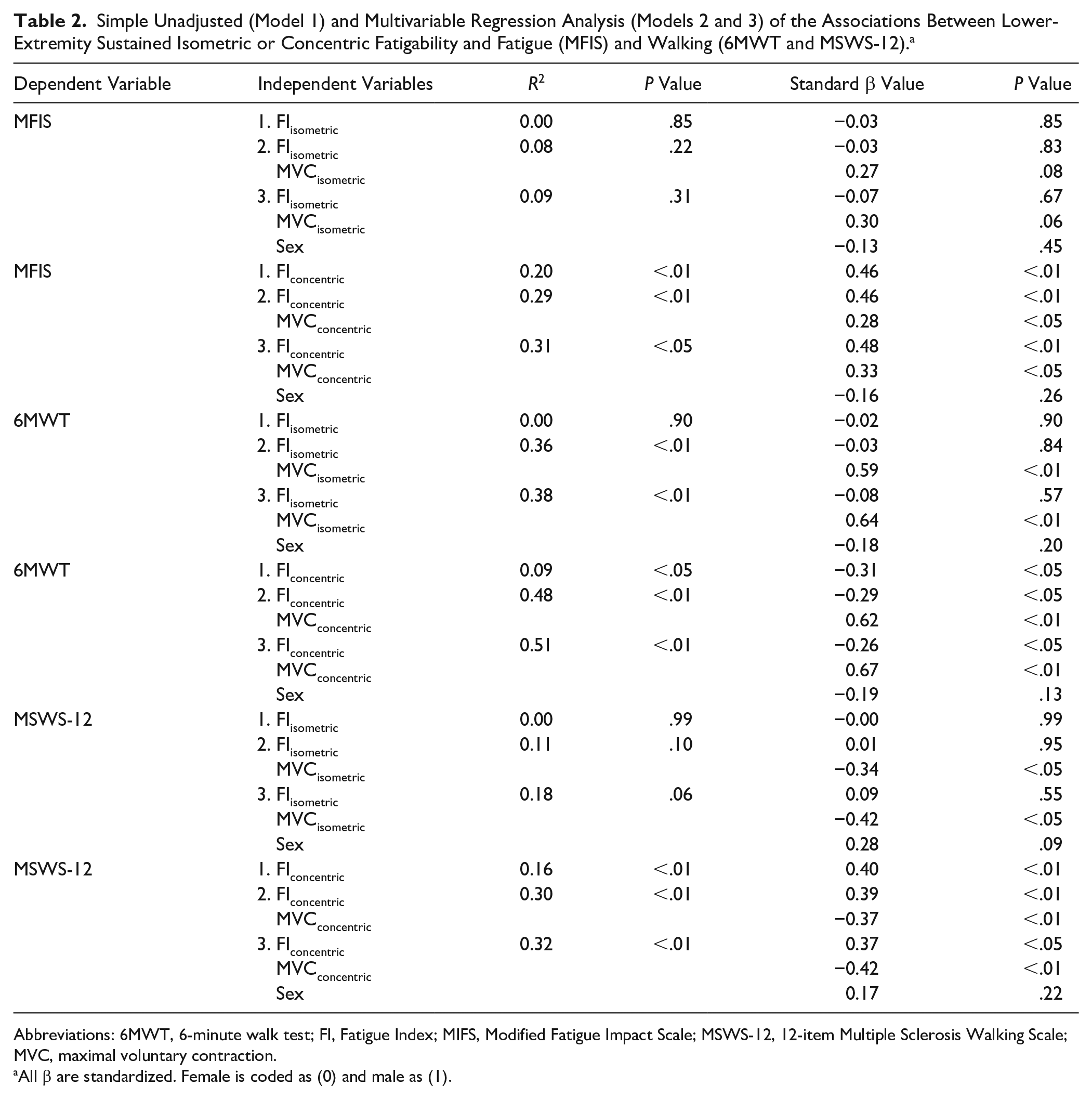
Abbreviations: 6MWT, 6-minute walk test; FI, Fatigue Index; MIFS, Modified Fatigue Impact Scale; MSWS-12, 12-item Multiple Sclerosis Walking Scale; MVC, maximal voluntary contraction.
All β are standardized. Female is coded as (0) and male as (1).
Concentric fatigability showed a significant association with fatigue impact (
When directly comparing the associations between fatigue impact and the 2 independent variables, concentric fatigability was shown to have a stronger association than sustained isometric fatigability (
Fatigability and Walking
The level of sustained isometric fatigability was not associated with any of the measures of walking capacity (6MWT distance:
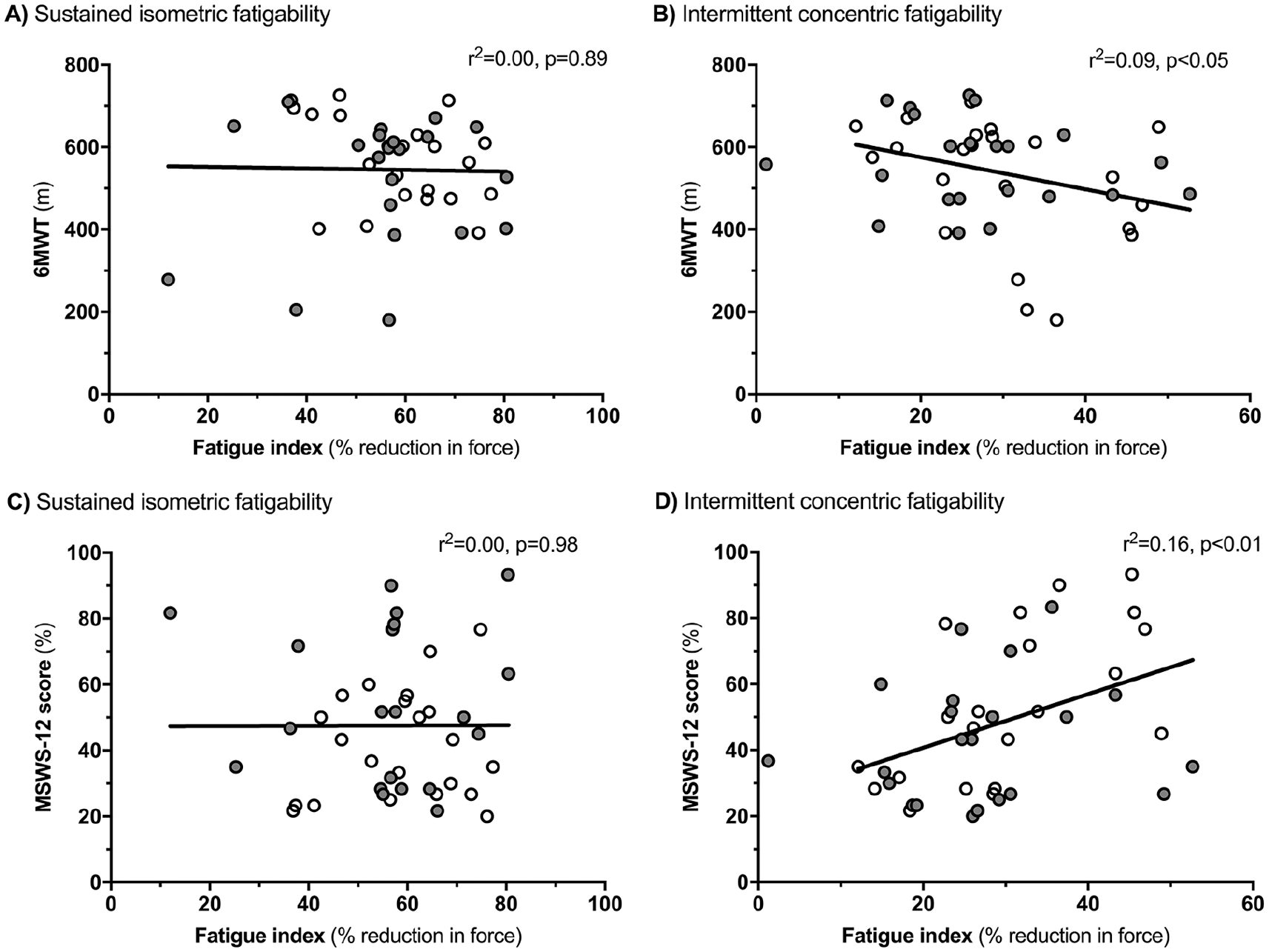
Association between 6MWT distance and (A) lower-extremity fatigability induced by a sustained isometric protocol and (B) fatigability induced by an intermittent concentric protocol. Association between MSWS-12 score and (C) lower-extremity fatigability induced by a sustained isometric protocol and (D) lower-extremity fatigability induced by an intermittent concentric protocol. The gray circles indicate that this test was done as test 2 on the test day.
Concentric fatigability was associated with both measures of walking capacity (6MWT distance:
When directly comparing the associations between both measures of walking capacity and the 2 independent variables, no statistical difference was observed between protocols for 6MWT, whereas concentric fatigability was shown to have a greater association with MSWS-12 than sustained isometric fatigability (
Discussion
The majority of studies investigating fatigability in persons with MS have used sustained isometric protocols on the body function level of the ICF model. However, daily and functional tasks rely on lower-extremity concentric muscle contractions, and this is the first study to show that a lower-extremity concentric fatigability protocol is more strongly associated with fatigue impact and some measures of walking than a sustained isometric protocol, in a head-to-head comparison.
Sustained Isometric Versus Concentric Protocol
Several studies have applied sustained isometric protocols to induce fatigability in the knee extensors.15,18,34 Some studies reported a lower level of fatigability than the present study,18,34 which is likely explained by the duration of protocols (30 s of sustained contractions as compared with the 2 minutes in the present study). In support of the present findings (sustained isometric protocol: mean FI ≈ 57%), Skurvydas et al 15 also applied a protocol with a 2-minute sustained contraction and found a comparable level of fatigability (mean FI ≈ 65%). In the concentric protocol, the mean FI was 29% in the present study, in line with previous reported values for the knee extensors (mean FI ≈ 26%-32%).19,31,35 However, a direct comparison should be performed cautiously because previous protocols differ with respect to either the velocity of contractions 35 or the applied definition of fatigability.19,31
The concentric protocol that was applied in the present study induced a relatively low level of fatigability using low-velocity muscle contractions. Because it has previously been shown that muscle strength impairments in persons with MS are more pronounced at faster concentric contractions, 36 a higher angular velocity might have resulted in more pronounced fatigability and, therefore, potentially an even better association with the 6MWT. Future studies should address this aspect.
Fatigability and Perception of Fatigue Impact
The results of the present study are in line with the results of Steens et al 20 and Wolkorte et al, 37 who similarly did not find an association between muscle fatigability induced by a sustained isometric protocol and perceived fatigue impact. However, when adjusting for muscle strength, these studies did find an association between the level of fatigability in isolated hand models and perception of fatigue. Likewise, a study by Severijns et al 38 did find an association between their 30-s static FI in 4 different upper-limb muscles and perception of fatigue. These findings are in contrast with the findings of the present study but may be caused by differences in blood flow restrictions during different types of sustained isometric contractions (ie, intensity of contraction and muscles involved) 39 ; also, the involvement of the large lower-extremity musculature in the present study leads to more pronounced peripheral fatigability. If so, this might result in motivational issues and a subsequent overestimation of fatigability. 40
Only 2 previous studies have investigated the association between concentric fatigability and perceptions of fatigue in persons with MS, and they failed to find such an association.19,21 However, these studies involved either a fatigability protocol, including voluntary breaks, 21 or had a markedly lower sample size and, thereby, a lower statistical power. 19 Therefore, the observed association between the level of concentric fatigability and perception of fatigue impact is of great clinical interest. Specifically, this novel observation suggests a shift toward concentric fatigability protocols when assessing fatigability on the body function level of the ICF model. Moreover, it can also serve as a rationale for resistance training as a strategy to manage fatigue because resistance training has been shown to improve muscle endurance. 41
Surprisingly, our results showed a positive association between muscle strength and fatigue impact, suggesting a higher fatigue impact in stronger individuals. This finding is somehow unexpected and needs to be further investigated in future studies. One possible explanation could be that higher muscle strength is associated with greater muscle fatigability, 42 and greater muscle fatigability has further been found to be associated with higher fatigue impact. However, our data did not support an association between muscle strength and muscle fatigability (data not shown). Therefore, another speculative explanation could be that stronger patients engage in more fatiguing tasks during daily activities than weaker individuals, and this could influence the perception of fatigue impact.
Fatigability and Walking
The present study found an association between the level of concentric fatigability and MSWS-12, which no previous study has reported. This observation is of great clinical relevance because the MSWS-12 has been shown to be the most sensitive tool when assessing walking impairment in mild MS, where walking impairments often cannot be detected by objective walking tests.
43
Moreover, a weak but significant association between concentric fatigability and 6MWT distance (
The multivariable analyses, including the respective fatigability protocols, muscle strength, and sex, revealed that both models were associated with 6MWT distance and MSWS-12 score. It is interesting to note that for this head-to-head comparison of fatigability protocols, the level of sustained isometric fatigability was not a predictor of either of the walking measures, whereas the concentric fatigability was a significant predictor of both measures. However, in relation to walking, the muscle strength seems to be a stronger predictor indicating a hierarchical order, with muscle strength being the primary prerequisite for walking and muscle endurance (fatigability) being secondary. This way of thinking is in line with results from the study by Severijns et al 38 showing that upper-limb function is reliant on maximal finger strength (eg, ability to lift an object) and, second, on elbow flexor endurance (eg, ability to hold on to the object).
In summary, and with regard to the head-to-head comparison between fatigability protocols, it seems that when measured on the ICF body level, concentric fatigability is more closely related to some measures of walking (ICF activity level) than sustained isometric fatigability.
Limitations
The present study has some limitations that must be kept in mind when interpreting the results. First, because of the cross-sectional design of the study, no causal relationships can be inferred. Second, we are not able to determine whether it is the nature of the protocol (sustained vs intermittent) or the type of contraction (isometric vs concentric) that is decisive. Therefore, future studies should investigate the difference between an intermittent isometric versus an intermittent concentric protocol, allowing identification of the decisive factor. Third, a minority of participants (n = 5) were not able to perform a full 90° ROM, which potentially could lead to an underestimation of fatigability. However, the participants were not excluded from analysis because they were short of less than 10° degrees of ROM and only on a minority of the 40 repetitions. Finally, the present study only involved ambulatory persons with MS, and the results may therefore not be representative for the whole MS population.
Conclusions and Future Perspectives
A recent review revealed that most fatigability protocols are assessed on the ICF body function level, in protocols applying sustained isometric contractions. This offers better opportunity to investigate the underlying mechanisms of fatigability because this is normally simpler during isometric contractions (eg, performing electromyographic measurements and/or the interpolated twitch technique). However, the present study provides evidence that this way of assessing fatigability is related neither to patients’ perception of fatigue impact nor walking measures, limiting the functional value of this outcome. In contrast and of great clinical relevance, the findings of the present study provide the first evidence that a lower-extremity concentric fatigability protocol is a superior reflection of some measures of walking capacity and perception of fatigue impact as compared with a sustained isometric protocol. On this basis, we suggest that concentric protocols should be the focus of future studies investigating fatigability or use fatigability as an outcome in exercise interventional trials. This, in combination with investigations of underlying mechanisms, may ultimately expand our current knowledge and help improve management of fatigue and functional capacity in persons with MS. In stroke survivors, resistance training has shown the potential to increase muscle endurance (ie, decrease fatigability), 44 and the results of the present study might, therefore, also serve as a rationale for applying resistance training in clinical practice and fatigue management in persons with MS.
Footnotes
Acknowledgements
The authors wish to thank the Danish National Multiple Sclerosis Society and the Augustinus foundation for funding the study. None of the sponsors played a role in the study design; in the collection, analysis, and interpretation of data; in the writing of the article; or in the decision to submit the article for publication.
Authors’ Note
Conception or design of the work: MR, TK, LGH, and UD. Data collection: MR, LT-M, TP, and LGH. Data-analysis: LT-M, MR, LGH, and UD. Interpretation of data: all. Drafting the work: LT-M, MR, and UD. Final approval and revision of the version to be published: all. Agreement to be accountable for all aspects of the work: all.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: UD has received research support, travel grants, and/or teaching honorary from Biogen Idec, Merck Serono, Novartis, Bayer Schering, and Sanofi Aventis as well as honoraria from serving on scientific advisory boards of Biogen Idec and Genzyme. LGH has received research support, travel grants, and/or teaching honoraria from Biogen and Sanofi Genzyme.
