Abstract
Background
Actual and imagined cued gait trainings have not been compared in people with multiple sclerosis (MS).
Objective
To analyze the effects of cued motor imagery (CMI), cued gait training (CGT), and combined CMI and cued gait training (CMI-CGT) on motor, cognitive, and emotional functioning, and health-related quality of life in people with MS.
Methods
In this double-blind randomized parallel-group multicenter trial, people with MS were randomized (1:1:1) to CMI, CMI-CGT, or CGT for 30 minutes, 4×/week for 4 weeks. Patients practiced at home, using recorded instructions, and supported by ≥6 phone calls. Data were collected at weeks 0, 4, and 13. Co-primary outcomes were walking speed and distance, analyzed by intention-to-treat. Secondary outcomes were global cognitive impairment, anxiety, depression, suicidality, fatigue, HRQoL, motor imagery ability, music-induced motivation, pleasure and arousal, self-efficacy, and cognitive function. Adverse events and falls were continuously monitored.
Results
Of 1559 screened patients, 132 were randomized: 44 to CMI, 44 to CMI-CGT, and 44 to CGT. None of the interventions demonstrated superiority in influencing walking speed or distance, with negligible effects on walking speed (η2 = 0.019) and distance (η2 = 0.005) observed in the between-group comparison. Improvements in walking speed and walking distance over time corresponded to large effects for CMI, CMI-CGT, and CGT (η2 = 0.348 and η2 = 0.454 respectively). No severe study-related adverse events were reported.
Conclusions
CMI-GT did not lead to improved walking speed and distance compared with CMI and CGT alone in people with MS. Lack of a true control group represents a study limitation.
Trial registration
German Clinical Trials Register, DRKS00023978.
Introduction
Multiple sclerosis (MS) is a chronic, inflammatory demyelinating disease of the central nervous system. 1 MS markedly impacts gait, particularly accentuated during faster walking speeds. 2 Recent meta-regression findings indicate that people with MS (pwMS) exhibiting higher disability levels tend to adopt a more cautious and stable gait pattern, contrasting those with lower disability levels. This pattern is consistent among pwMS categorized as fallers compared to nonfallers. 3
Motor imagery (MI) refers to the mental simulation of movement without physical execution. 4 This approach involves simulating bodily movements or rehearsing imagined movements, to enhance motor performance. 4 Two modes of MI have been identified: visual mode (visualizing oneself moving) and kinesthetic mode (perceiving one’s body moving). 4 The kinesthetic mode relies on pre-existing motor representations and acquired motor skills, 4 making the ability to walk and prior experience prerequisites for effective MI of walking. Originally developed to enhance athletic performance,4,5 MI has been studied in rehabilitation settings.6,7 It could be an alternative method to stimulate movement in pwMS, as it shares temporal characteristics and activates brain areas involved in actual motor actions. 4 Preliminary evidence suggests that MI practice may benefit MS related fatigue, 6 and tele-MI has shown improvements in physical, cognitive, and psychosocial functioning. 7 However, impairment in MI ability, especially in those with cognitive dysfunction and depression, 8 may reduce or nullify its effectiveness.
The benefits of music- and rhythm-based interventions in people with neurological conditions have been demonstrated by numerous studies.9-11 External rhythmic cues have further been found to enhance MI timing and ability in pwMS and therefore, 12 we combined MI with rhythmic auditory stimulation in our previous work.13,14 Rhythmic auditory stimulation utilizes rhythmic auditory cues to assist with cyclical movements, particularly walking. 15 It involves the use of auditory cues, such as metronome cues, musical beat, rhythmic verbal cues, or combinations of these.13-15 Gait training incorporating rhythmic auditory stimulation has demonstrated effectiveness in enhancing walking function and various gait parameters among individuals with neurological conditions, 15 including stroke 9 , PD, 10 and MS. 16 This approach may facilitate rhythmic entrainment, which synchronizes neural rhythm processes with external rhythmic-auditory cues. 15 Music- and rhythm-based interventions have additional advantages, as they can be self-administered by patients in their homes, necessitating minimal therapist assistance. This approach provides patients with flexibility in terms of when and how to engage with the interventions. 17
Our previous randomized controlled trials (RCT) represent the only available evidence on the effectiveness of combining MI and rhythmic auditory stimulation in pwMS.13,14 These trials included individuals with mild to moderate MS, who did not have cognitive dysfunction or depression. Findings from these studies indicated that music- and verbally cued MI was superior in effects over non-cued MI on walking, fatigue, and quality of life.
To date, no studies have compared the effects of cued MI, cued actual gait training, and a combination thereof in pwMS. Therefore, in line with the conclusions of a systematic review, 18 using a double-blind randomized parallel multicenter trial, we aimed to determine the effects of actual and imagined rhythmic-cued gait training, versus a combination of both on walking, fatigue, cognitive and emotional functioning, and health-related quality of life (HRQoL) in pwMS. The study also explored music-induced pleasure and arousal, music-induced motivation in exercise, and MS-related self-efficacy. We hypothesized, that:
Actual and imagined rhythmic-cued gait training, as well as a combination of both, will significantly improve walking, fatigue, HRQoL, emotional, and cognitive functioning in pwMS.
The effects of cued MI combined with cued gait training are superior to those of cued MI and cued gait training alone.
Methods
Study Design and Participants
The Consolidated Standards of Reporting Trials Statement (CONSORT) was used to guide the reporting of this study (see CONSORT Checklist in Supplemental Material 1). As previously detailed in the published trial protocol,
19
the RIGMUC (
Randomization and Masking
Eligible patients were randomly assigned (1:1:1) to either to receive either cued MI, combined cued MI and cued gait training, or cued gait training by stratified block randomization, performed by an independent researcher using online software-generated random numbers (Sealed Envelope, London, UK), and permuted blocks of 3, 6, and 9 (unknown to study personnel until trial completion). Stratification was by gender, age (<40, ≥40), and disability (EDSS 2.0-3.5, 4.0-5.0), and sequentially numbered sealed opaque envelopes were employed for each stratum. Group allocation was concealed until trial completion, and assessors masked to participants’ group allocation. Different personnel handled intervention delivery and assessment, and they were instructed not to conceal patients’ group allocation. Participants were informed that the study was evaluating different gait training types with musical stimulation, without disclosing the study hypotheses.
Intervention
In all 3 groups, the home-based RIGMUC intervention was 30 minutes/day including short breaks, 4 days/week for 4 weeks. A template for intervention description and replication (TIDieR) checklist is provided with the published study protocol. 19 Interventions were provided by physiotherapists, occupational therapists, and psychologists who had at least 5 years professional experience. They were trained by the lead investigator during the initial preparatory trial phase on participants’ instructions and interventions. Participants in all groups received written information about their respective study intervention and an audio mix, specifically created for this study using Audacity® Version 3.0.0. They could download the mix onto their electronic devices or access it through study contact discs. To enhance attention and adherence, the audio mix was changed weekly. Participants could choose their preferred days and times for home-based practice. During the 4-week intervention period, participants received weekly support calls (a minimum of 4 calls in total), and additional calls were made at the 4-week follow-up and upon request. These calls aimed to maintain motivation, ensure adherence, and address any queries. Practice frequency and duration were documented in a pre-formatted study diary and recorded during weekly phone call interviews. All participants received their usual care, with any treatment changes closely monitored.
Cued MI (CMI)
Participants in the CMI group practiced cued kinesthetic MI related to walking. Various walking variations were suggested, such as imagining/taking large steps, walking upright, or stomping. They underwent one-on-one familiarization with cued kinesthetic MI. This involved a 20-minute audio mix demonstration and verbal instructions. The familiarization process utilized the PETTLEP (Physical, Environmental, Task, Timing, Learning, Emotional, and Perspective) approach to MI, addressing various aspects like physical position, arousal, imagined environment, imaged task, temporal regulation, learning processes, emotional aspects, and MI perspective. 5
Imagined gait training with rhythmic cuing in 2/4 or 4/4 time signatures with distinct emphasis on ON and OFF beats (pulse groups) was employed. 30 Instrumental music with a regular beat, accentuated on the first or the first and third beat, was selected, and the beat was amplified with metronome cues. A compilation of suitable rhythmic sequences, with a tempo ranging from 80 to 120 beats per minute, slightly increasing from week 1 to 4, were used and blended with verbal instructions. Rhythmic verbal cues intermittently accentuated the cuing, using phrases like “step step” or “raise foot.”
Cued Gait Training (CGT)
Participants in the CGT group practiced cued actual gait training, employing the same principles and procedures as described for CMI. Various walking variations were suggested, such as imagining/taking large steps, walking upright, or stomping. Actual gait training with rhythmic cuing was employed.
Combined cued MI and cued gait training (CMI-CGT)
Participants in the CMI-CGT group engaged in combined actual and imagined gait training with rhythmic cuing, following the same principles and procedures as described for both CMI and CGT. They divided their practice time, spending 15 minutes on each cued kinesthetic MI practice and CGT.
Data Collection
Assessments were conducted by neurologists, physiotherapists, occupational therapists, and psychologists, each possessing a minimum of 5 years of professional experience and receiving training from the lead investigator. Assessors remained masked to participants’ group allocation throughout the study. Participants were assessed at baseline (week 0), 4 weeks post-intervention, and 13 weeks follow-up in their respective study centers. Baseline data included demographic and disease-specific information, such as age, gender, handedness, disability (EDSS), MS phenotype, disease duration, and disease-modifying treatment (DMT). Participants were instructed to report adverse events to their intervention provider directly. Adverse events were also recorded retrospectively during support phone calls and on-site assessments. These events were systematically classified as serious or non-serious, and any potential association with the intervention was assessed.
Outcomes
Primary Outcomes
Co-primary outcomes were the change in walking speed, assessed by the Timed 25-Foot Walk (T25FW) 31 and the change in walking distance, as measured by the 2-Minute Walk Test (2MWT), 32 between baseline and 4 weeks post-intervention. A change of 20% in the T25FW was considered a priori a clinically significant difference in walking speed. 33 The T25FW involves patients walking a designated distance of 25 feet (7.62 meters) as fast as possible, while ensuring safety, and they may use assistive devices if necessary. Two trials were conducted, and the average was taken for scoring. The T25FW has shown excellent psychometric properties in pwMS across disability levels. 33 Results obtained from the T25FW reflecting walking speed have demonstrated practical significance in real-life. 33 The 2MWT adhered to the guidelines established by the American Thoracic Society, initially developed for the 6-Minute Walk Test, and subsequently modified by international experts from the NIH Toolbox. The 2MWT has shown excellent validity 34 and was recommended as an appropriate replacement for the 6-Minute Walk Test. 32 A 20% change in 2MWT was interpreted a priori being a clinically significant difference in walking distance. 34
Secondary Outcomes
Secondary outcomes were global cognitive impairment (Montreal Cognitive Assessment [MoCA]),23,24 anxiety and depression (Hospital Anxiety and Depression Scale [HADS]),25,26 suicidality (narrative screening), 29 fatigue (Neurological Fatigue Scale - Multiple Sclerosis [NFI-MS]),35,36 and HRQoL (Multiple Sclerosis International Quality of Life questionnaire [MusiQoL]). 37 Further secondary outcomes were MI ability (Kinesthetic and Visual Imagery questionnaire-10 [KVIQ-10] 38 ; mental chronometry test comparing the duration of imagined and actual walking over 6 meters on a marked path [mental chronometry ratio = imagined walking time/actual walking time; average from 2 trials]),39,40 music-induced motivation in exercise, using Brunel Music Rating Inventory-2 (BMRI-2), 41 music-induced pleasure and arousal (Self-Assessment Manikin [SAM]), 42 MS related self-efficacy (Unidimensional Self-Efficacy Scale for MS [USE-MS]),43,44 and cognitive function, including attention, visual scanning, working memory and psychomotor speed (Symbol Digit Modalities Test [SDMT]) 45 at 4 weeks post-intervention and at 13 weeks follow-up. Additionally, throughout the study, adverse events, adverse reactions, and falls were monitored (logs, phone calls, at assessments). Using a checklist and weekly phone calls, adherence was recorded. 19 A detailed description of the secondary outcomes is provided in Supplemental Material 2.
MRI was employed to explore brain activation patterns, while a qualitative component was integrated to evaluate the subjective experiences, acceptability, and coping of participants using CMI or/and CGT through phone call interviews. Findings from these data will be reported in future publications.
Sample Size Estimation
The sample size for this study was determined using previous study data 13 and Cohen’s d effect sizes related to walking speed and distance outcomes, utilizing 95% CI and adjusted estimates of the pooled standard deviation (0.27 and 0.32 for changes in T25FW between groups A & C and B & C, respectively; 59.92 and 37.71 for between-group changes in the 6-Minute Walk Test, 6MWT), where group A practiced music- and verbally-cued MI, group B performed metronome- and verbally-cued MI, and group C served as a control group. From this, Cohen’s d effect sizes of 3.20 and 2.71 (T25FW) and 1.32 and 2.07 (6MWT) resulted. As 3 intervention groups were used in the present study, a conservative d of 0.74 was chosen. Based on 80% statistical power, α < .025 (adjusting for multiple comparisons of the primary outcomes), d = .74 and 2-tailed tests, the target sample size per group was estimated to be 37 participants. Considering an anticipated attrition rate of 15%, a total sample size of 132 participants was determined.
Statistical Analysis
Statistical analyses were conducted using IBM SPSS software, release 28.0 (IBM Corporation, Armonk, NY, USA), and GraphPad Prism 9 from San Diego, California. A 2-tailed
As a priori planned, after checking test assumptions, for continuous variables (T25FW, 2MWT, and mental chronometry), a mixed design ANOVA was conducted using time as the within-subject factor and group as between-subject factor, with the 3 DMT categories serving as covariates (no DMT, moderately effective DMT, highly effective DMT). 19 Post-hoc Bonferroni adjustment was applied as deemed appropriate. Effect sizes were computed as partial Eta-squared values (η2). Mean between-group differences (95% CI) were estimated. A Chi-squared test was employed to determine the number of participants displaying a clinically significant difference in the co-primary outcomes, indicated by a ≥20% change in walking speed (T25FW) 33 and walking distance (2MTW) 34 across the 3 groups.
For categorical data, the calculation of differences between post-intervention and baseline values was followed by Kruskal-Wallis and Dunn’s multiple comparisons tests. Within-group changes between baseline and post-intervention or follow-up, and between post-intervention and follow-up were estimated using Wilcoxon signed rank test on split file data, followed by a Bonferroni correction for multiple (3) comparisons.
Results
We screened 1559 potential participants between February 9, 2021, and November 15, 2022. Following screening, 132 participants were randomly assigned to either CMI (n = 44), CMI-CGT (n = 44), or CGT (n = 44). Five patients (3.8%) withdrew, did not complete the intervention, and therefore were not included in the intention-to-treat population. Contact was lost with 2, 1 had a MS relapse, 1 had severe side effects from their DMT, and 1 reported lack of time (Figure 1). Five further participants were lost to follow-up, resulting in a total attrition rate of 7.6%: 9.1% in the CMI group, 2.3% in the CMI-CGT group, and 11.4% in the CGT group (non-significant difference).
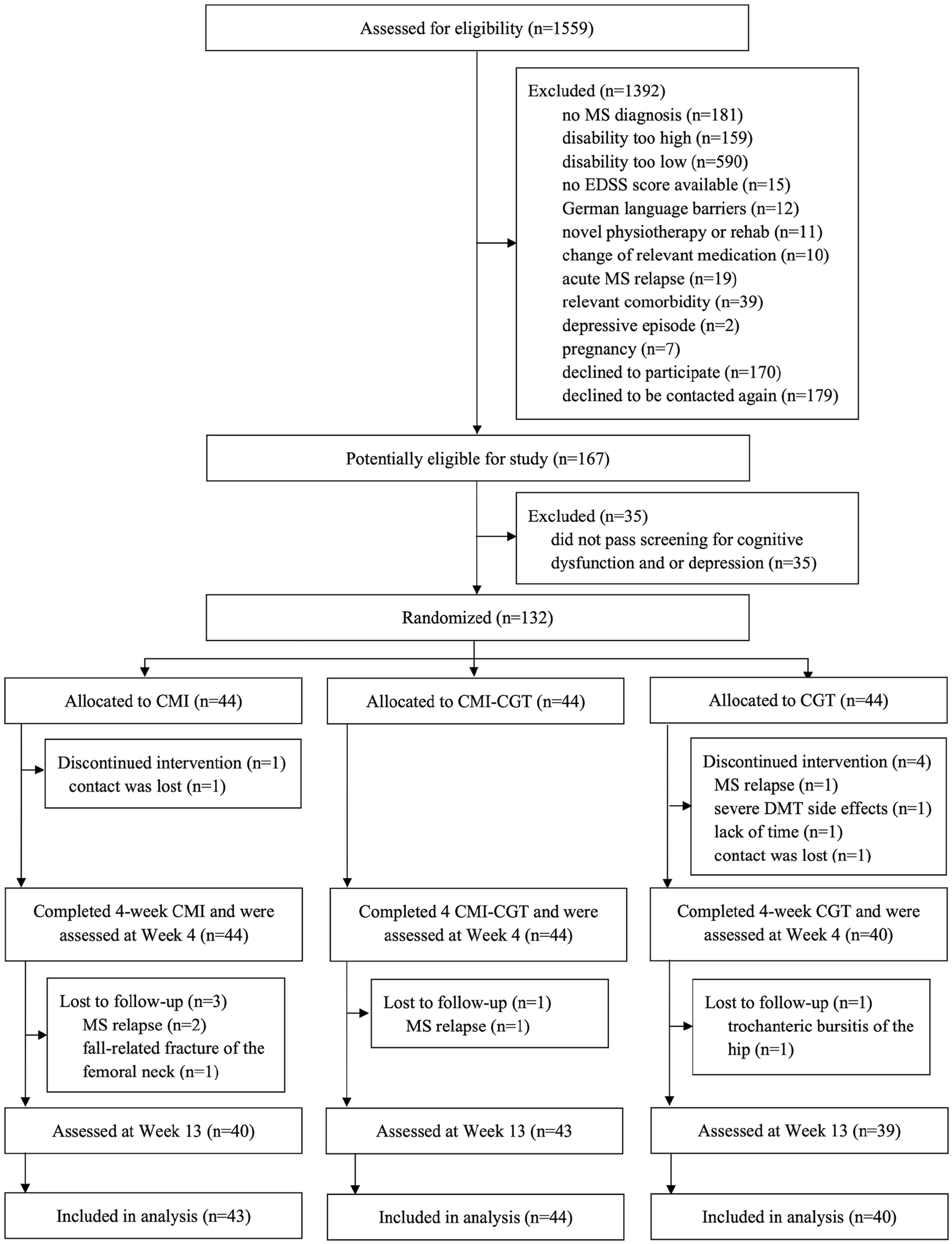
CONSORT flow diagram depicting participant recruitment, randomization, and attrition.
Mean age of the intention-to-treat population was 49.1 years (SD 11.0), ranging from 28.4 to 79.3 years, and 93 (73.2%) were women, 33 (26.0%) were men, and 1 (.8%) reported diverse gender. Baseline characteristics and walking performance at baseline (2MTW, T25FW) were comparable across groups, although the number of patients with progressive disease was slightly greater in the CGT group (
Baseline Characteristics of Intention-to-Treat Population.
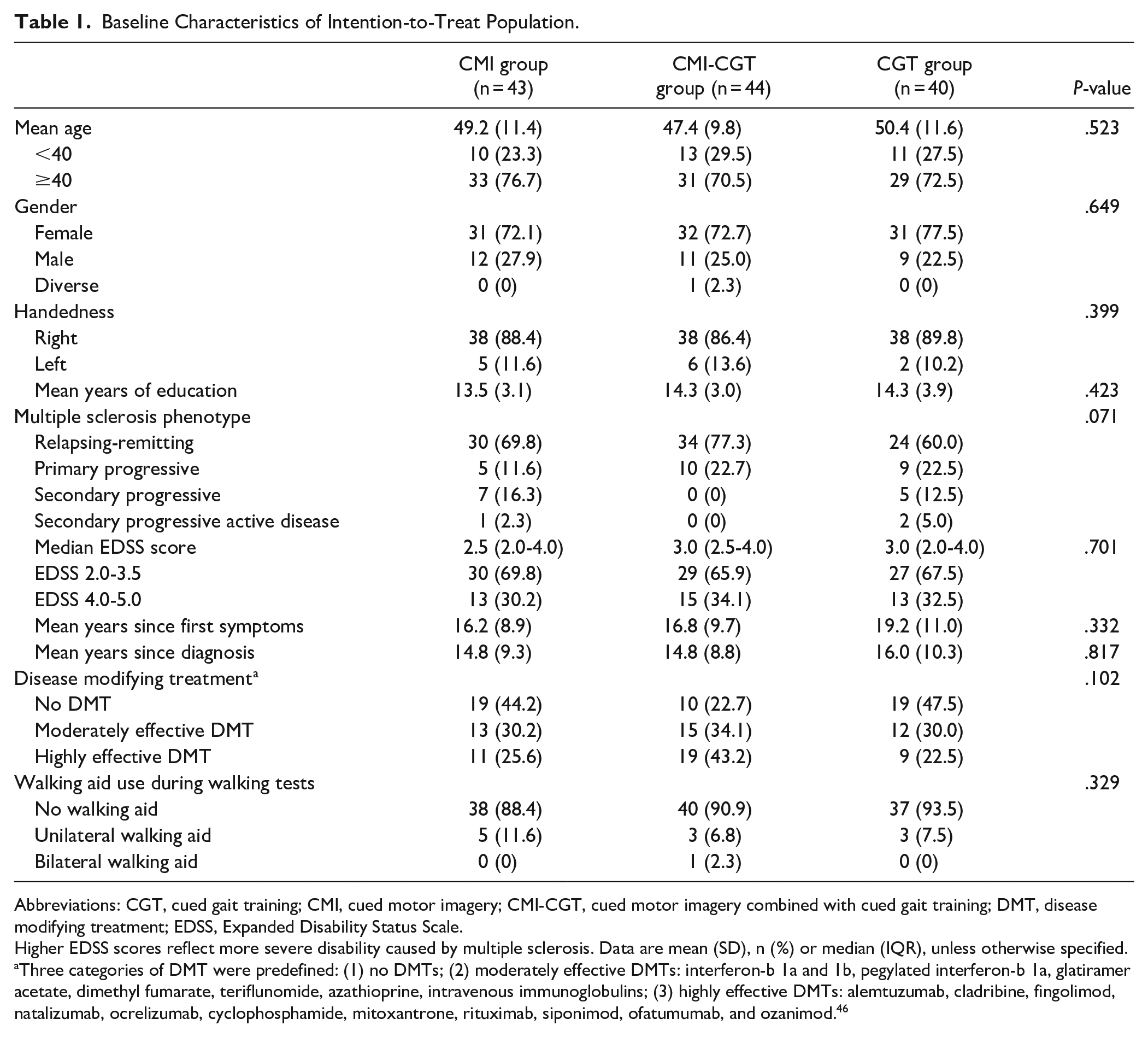
Abbreviations: CGT, cued gait training; CMI, cued motor imagery; CMI-CGT, cued motor imagery combined with cued gait training; DMT, disease modifying treatment; EDSS, Expanded Disability Status Scale.
Higher EDSS scores reflect more severe disability caused by multiple sclerosis. Data are mean (SD), n (%) or median (IQR), unless otherwise specified.
Three categories of DMT were predefined: (1) no DMTs; (2) moderately effective DMTs: interferon-b 1a and 1b, pegylated interferon-b 1a, glatiramer acetate, dimethyl fumarate, teriflunomide, azathioprine, intravenous immunoglobulins; (3) highly effective DMTs: alemtuzumab, cladribine, fingolimod, natalizumab, ocrelizumab, cyclophosphamide, mitoxantrone, rituximab, siponimod, ofatumumab, and ozanimod. 46
Participant-reported adherence with the intervention was median 16 sessions (IQR 15-16) across all groups: CMI, CMI-CGT, and CGT. There were 3 hospital admissions, 1 due to a fall in the CMI group, 1 because of a trochanteric bursitis of the hip, and 1 due to severe side effects of DMT treatment, the latter in the CGT group. Four participants experienced a MS relapse (CMI group, n = 2; CMI-CGT group, n = 1, CGT group, n = 1, corresponding with 2/1/1), and 1 had a COVID-19 infection (CGT group). During the 4-week intervention period, 17 (13.4%) participants experienced one or more falls, of those 6 were in the CMI group, 4 in the CMI-CGT group and 7 in the CGT group. During the 13-week follow-up period, 23 (18.1%) participants had ≥1 fall, 11 in the CMI group, 3 in the CMI-CGT group and 9 in the CGT group. Patients reported symptoms as expected, comprising fatigue (n = 5; 3/0/2), musculoskeletal pain (n = 12; 5/2/5), dizziness (n = 3; 1/0/2), weakness (n = 2; 0/2/0), and spasticity (n = 4; 1/2/1). None of the serious adverse events or falls were deemed related to the study intervention or procedures.
For the first primary outcome of walking speed, no significant group by time interaction was found, F(4, 237) = 0.344,
Co-primary Outcomes (By Intention to Treat).
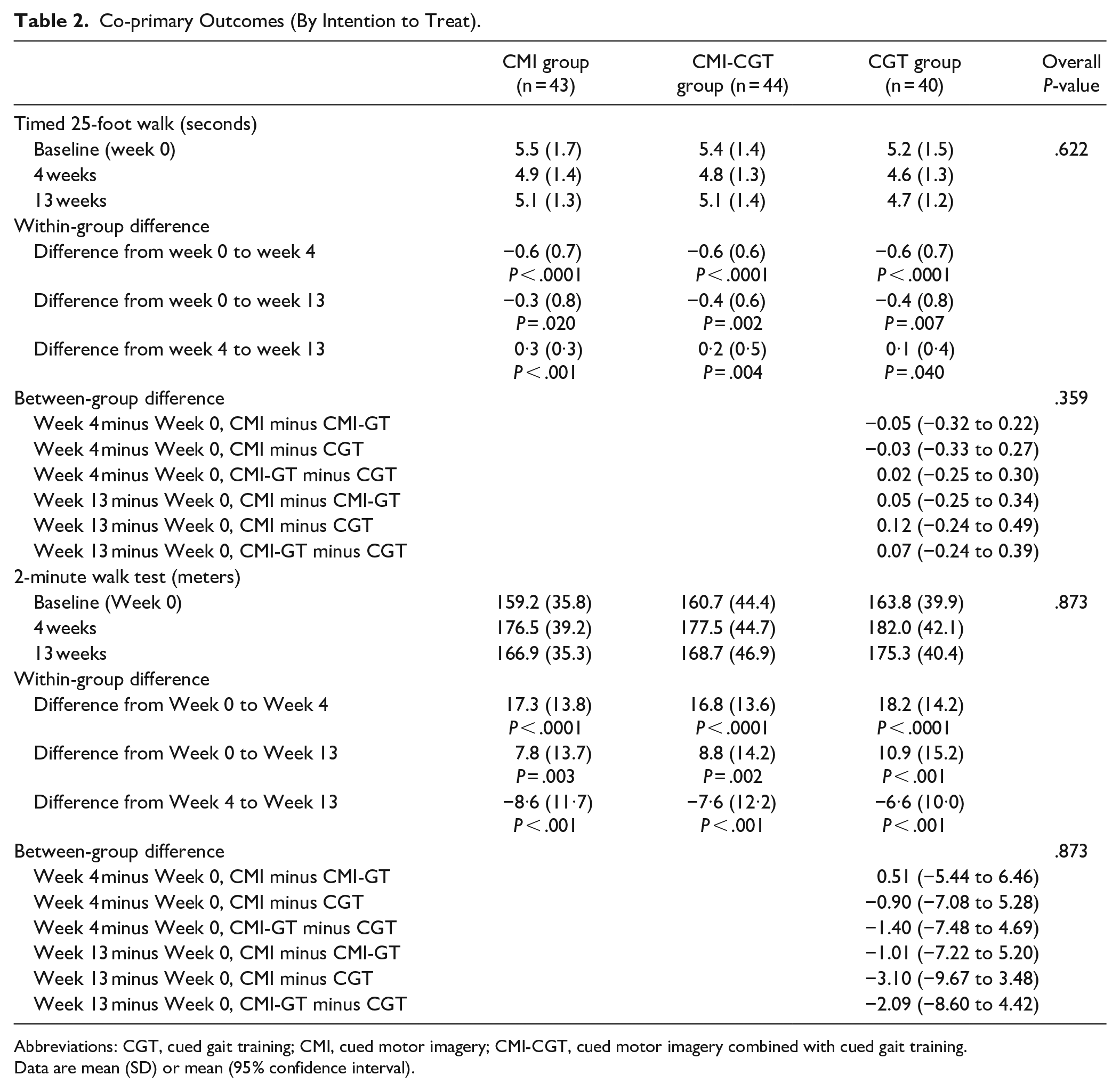
Abbreviations: CGT, cued gait training; CMI, cued motor imagery; CMI-CGT, cued motor imagery combined with cued gait training.
Data are mean (SD) or mean (95% confidence interval).
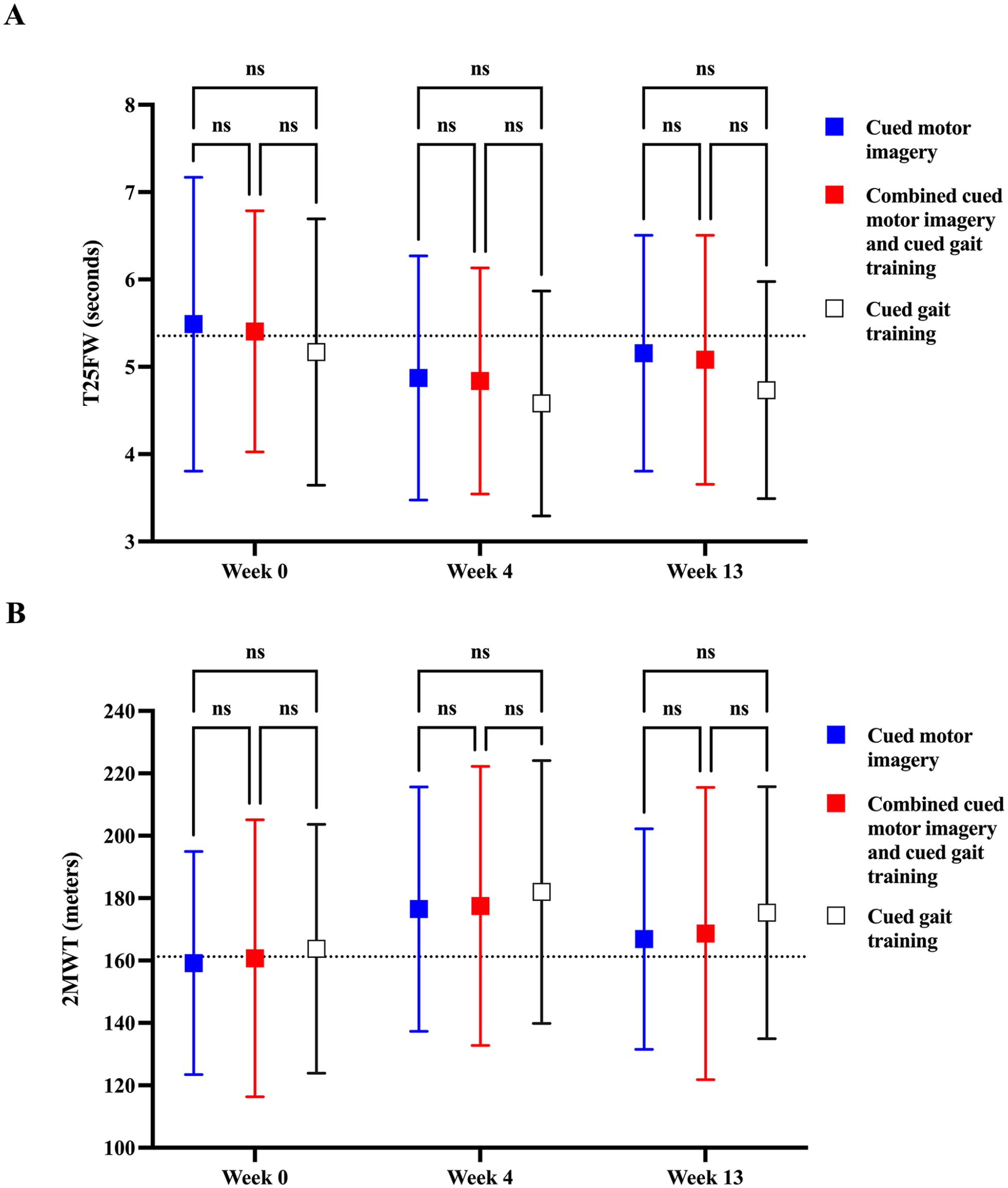
Change in primary outcomes from baseline: Change in walking speed (A) and walking distance (B) from baseline to 4-weeks post-intervention and 13-week follow-up as assessed by the Timed 25-Foot Walk (T25FW) and 2-Minute Walk Test (2MWT), respectively. Squares represent means, while error bars denote standard deviations. Dotted lines illustrate the grand mean from all groups at baseline. Lower values on the T25FW and higher values on the 2MWT indicate improvement.
With respect to the second co-primary outcome of walking distance, no significant group by time interaction was observed, F(4, 237) = 0.310,
For the secondary outcomes, between baseline and post-intervention, and baseline and follow-up, a statistically significant interaction effect was only seen for MI ability (KVIQ-10),
With respect to HRQoL, following all 3 interventions, patients reported improvements in several domains of HRQoL, but there was no statistically significant interaction effect or between-group difference (for
Relevant Secondary Outcomes (By Intention to Treat).
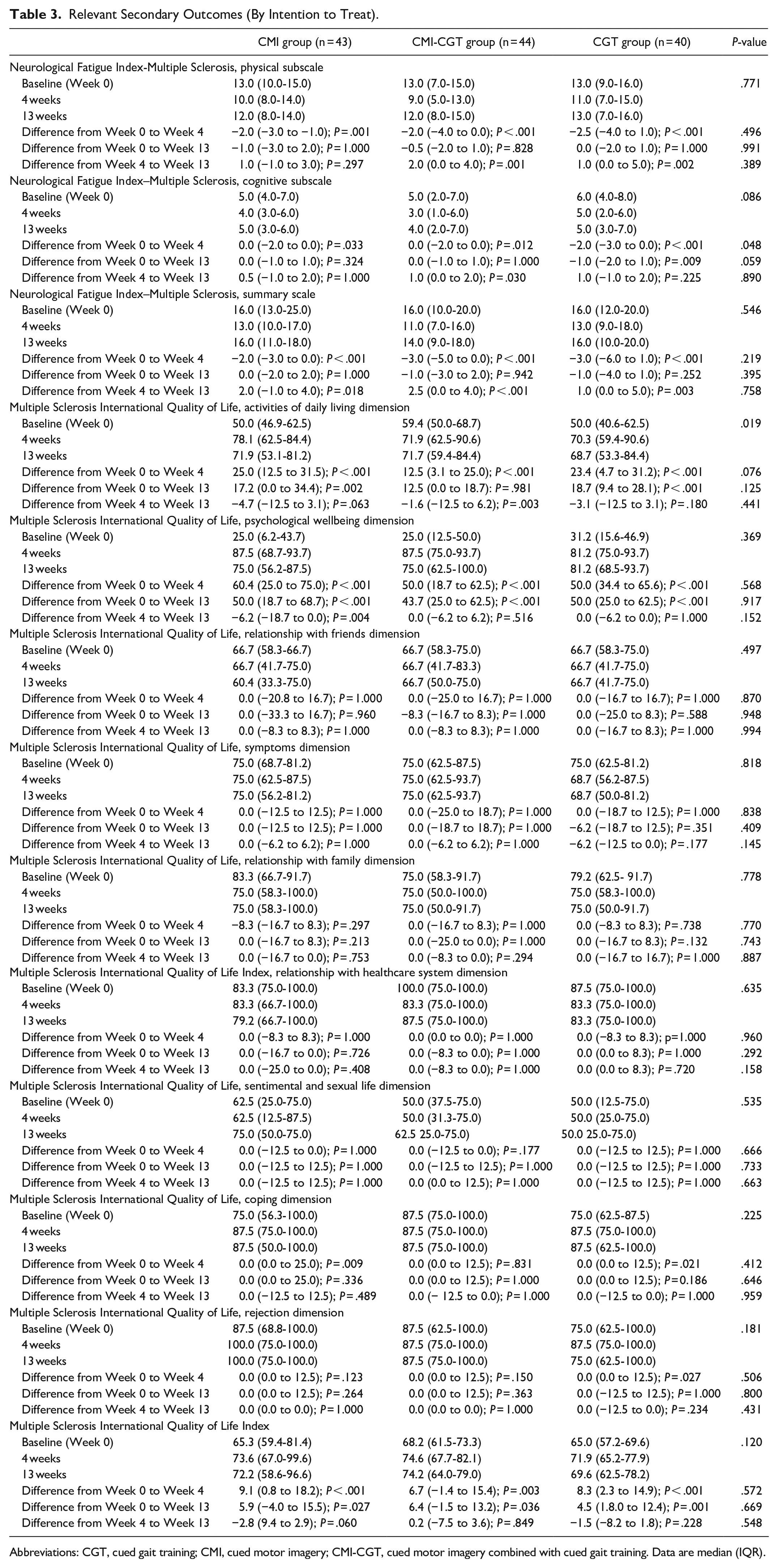
Abbreviations: CGT, cued gait training; CMI, cued motor imagery; CMI-CGT, cued motor imagery combined with cued gait training. Data are median (IQR).
Additional findings from the secondary outcomes of global cognitive impairment, anxiety and depression, suicidality (screening variables), MI abilities, music-induced pleasure and arousal, music-induced motivation in exercise, MS related self-efficacy, and cognitive function are reported in Supplemental Material 3.
Discussion
In this multicenter double-blind randomized parallel trial involving people with mild to moderate MS, 4 weeks of home-based CMI, CMI-GT, and CGT resulted in similar improvements in walking speed and walking distance, which were partly sustained at 13-week follow-up. Contrary to our initial hypothesis, the combined CMI-GT intervention did not yield greater effects, and there were no differences between the groups. Although a proportion of participants achieved clinically meaningful improvements in walking speed and walking distance, there were no differences between the groups. Similarly, for the secondary outcomes of fatigue, HRQoL domains of activities of daily living, psychological wellbeing, coping, rejection, and total HRQoL, as well as cognitive function, no interaction effect and no between-group differences were seen. We found evidence for improvements in fatigue and HRQoL from baseline to post-intervention, while after 13 weeks of follow-up, fatigue levels returned almost to baseline values, while most gains in HRQoL and cognitive function were sustained. While prior studies have shown effectiveness of tele-MI on cognitive function in pwMS, 7 we acknowledge the potential influence of a practice effect on the observed cognitive improvements. 47
Our findings align with the results of reviews reporting improvements in walking and gait parameters after gait training with rhythmic auditory cues in people with MS, 16 PD10,15 and ABI including stroke.9,15 Included studies utilized different strategies for movement cuing such as music, metronome cues, step sounds, internal signaling techniques like (mental) chanting, playing musical instruments, and music therapy interventions combined with dance. In contrast, we employed a combination of music, metronome, and verbal cueing. Results from the cited studies suggest that the type of cueing may significantly contribute to the effects on motor and cognitive function achieved. 9,10 Similar to our previous results in pwMS, 14 their findings suggest that greater improvement in walking occurs when rhythmic auditory cues are embedded in music. 9 Study findings also indicated improvements in walking10,48 and speech 11 after active singing interventions in people with PD. In pwMS, research consistently demonstrates that music-based therapy outperforms conventional therapy or no intervention across diverse areas, such as walking, gait parameters, fatigue, coordination, dexterity, balance, endurance, lower extremity strength, emotional well-being, and pain. 16
Regarding our results showing similar improvements after CMI as following CGT or their combination, research has reported the effectiveness of Therapeutic Instrumental Music Performance training with MI on post-stroke cognitive and emotional functioning. 49 Furthermore, one study in people with PD reported that mental singing led to greater improvements in walking than actual singing or external cueing strategies such as music cuing. 48 Another, albeit small study (n = 15) in people after stroke utilized locomotor MI with auditory step rhythm and found significant improvements in functional mobility after cued visual MI compared to cued kinesthetic MI. 50
Not all improvements in walking, fatigue, and HRQoL were sustained at 13-week follow-up. Consistent with our findings, review results showed that active music-based intervention effects tended to diminish after the intervention was discontinued in people with PD, suggesting that music-based therapy should be continued for longer periods of time. 10 This may pose a specific challenge for individuals with disabilities, as they need to maintain continuous commitment to the training for optimal outcomes. 51
Ensuring intervention tolerability is crucial for adherence. To achieve this, we used daily self-reported study diaries and conducted weekly individual telephone interviews. This allowed us to assess symptom severity and identify any new symptoms. Participants experienced various symptoms such as fatigue, musculoskeletal pain, dizziness, weakness, and spasticity. Importantly, no serious adverse events or falls were associated with the intervention or procedures.
Limitations and Outlook
We acknowledge the absence of a true control condition in our study, which may have compromised internal validity, causality attribution, and the ability to assess placebo effects. The lack of a control group hinders baseline comparisons and understanding natural progression, potentially leading to confounding practice effects and regression toward the mean, thus limiting the study’s robustness and interpretability. 52 Despite recognizing the importance of a control group, we opted to focus on comparing 3 distinct rhythmic-cued gait training interventions due to existing literature highlighting their efficacy. Although previous studies have indicated the effectiveness of rhythmic-cued gait training in people with MS, these studies were likely underpowered.53,54 Our decision not to include a control group was influenced by our prior RCT where both music-cued and metronome-cued motor imagery interventions demonstrated significant improvements in walking outcomes compared to controls.53,55 In retrospect, including a control group would have been appropriate.
Additionally, there is a limited generalizability of results to patients with more severe disability in MS. We included participants with mild to moderate disability, based on previous evidence of declining MI ability with increasing disability levels.8,56 However, it is uncertain if those with higher disability levels can effectively imagine themselves moving with music-cuing. To address this gap, further research is needed to investigate the impact of music-cuing on MI ability in pwMS with more severe disability.
Due to the standardized nature of the intervention, individual musical preferences were not accounted for, which could have potentially influenced the results. However, this effect would have affected all groups equally. Gathering data on adherence was based on diary recordings and weekly telephone interviews, since the intervention took place at participants’ homes. While this approach may be considered a limitation, the interviews were designed to closely monitor participants’ progress and how they managed the intervention, ensuring data reliability and minimizing the impact of this limitation.
In conclusion, in people with mild to moderate MS, none of the interventions demonstrated superiority in influencing walking, with negligible effects on walking speed and distance observed in the between-group comparison. Improvements in walking speed and walking distance over time corresponded to large effects for CMI, CMI-CGT, and CGT. Furthermore, none of the interventions demonstrated superiority in improving fatigue, HRQoL, emotional and cognitive functioning in pwMS. No severe study-related adverse events were reported.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241260724 – Supplemental material for Actual and Imagined Music-Cued Gait Training in People with Multiple Sclerosis: A Double-Blind Randomized Parallel Multicenter Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683241260724 for Actual and Imagined Music-Cued Gait Training in People with Multiple Sclerosis: A Double-Blind Randomized Parallel Multicenter Trial by Barbara Seebacher, Birgit Helmlinger, Daniela Pinter, Bettina Heschl, Rainer Ehling, Stefanie Hechenberger, Markus Reindl, Michael Khalil, Christian Enzinger, Florian Deisenhammer and Christian Brenneis MD in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-2-nnr-10.1177_15459683241260724 – Supplemental material for Actual and Imagined Music-Cued Gait Training in People with Multiple Sclerosis: A Double-Blind Randomized Parallel Multicenter Trial
Supplemental material, sj-docx-2-nnr-10.1177_15459683241260724 for Actual and Imagined Music-Cued Gait Training in People with Multiple Sclerosis: A Double-Blind Randomized Parallel Multicenter Trial by Barbara Seebacher, Birgit Helmlinger, Daniela Pinter, Bettina Heschl, Rainer Ehling, Stefanie Hechenberger, Markus Reindl, Michael Khalil, Christian Enzinger, Florian Deisenhammer and Christian Brenneis MD in Neurorehabilitation and Neural Repair
Supplemental Material
sj-docx-3-nnr-10.1177_15459683241260724 – Supplemental material for Actual and Imagined Music-Cued Gait Training in People with Multiple Sclerosis: A Double-Blind Randomized Parallel Multicenter Trial
Supplemental material, sj-docx-3-nnr-10.1177_15459683241260724 for Actual and Imagined Music-Cued Gait Training in People with Multiple Sclerosis: A Double-Blind Randomized Parallel Multicenter Trial by Barbara Seebacher, Birgit Helmlinger, Daniela Pinter, Bettina Heschl, Rainer Ehling, Stefanie Hechenberger, Markus Reindl, Michael Khalil, Christian Enzinger, Florian Deisenhammer and Christian Brenneis MD in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-4-nnr-10.1177_15459683241260724 – Supplemental material for Actual and Imagined Music-Cued Gait Training in People with Multiple Sclerosis: A Double-Blind Randomized Parallel Multicenter Trial
Supplemental material, sj-pdf-4-nnr-10.1177_15459683241260724 for Actual and Imagined Music-Cued Gait Training in People with Multiple Sclerosis: A Double-Blind Randomized Parallel Multicenter Trial by Barbara Seebacher, Birgit Helmlinger, Daniela Pinter, Bettina Heschl, Rainer Ehling, Stefanie Hechenberger, Markus Reindl, Michael Khalil, Christian Enzinger, Florian Deisenhammer and Christian Brenneis MD in Neurorehabilitation and Neural Repair
Supplemental Material
sj-pdf-5-nnr-10.1177_15459683241260724 – Supplemental material for Actual and Imagined Music-Cued Gait Training in People with Multiple Sclerosis: A Double-Blind Randomized Parallel Multicenter Trial
Supplemental material, sj-pdf-5-nnr-10.1177_15459683241260724 for Actual and Imagined Music-Cued Gait Training in People with Multiple Sclerosis: A Double-Blind Randomized Parallel Multicenter Trial by Barbara Seebacher, Birgit Helmlinger, Daniela Pinter, Bettina Heschl, Rainer Ehling, Stefanie Hechenberger, Markus Reindl, Michael Khalil, Christian Enzinger, Florian Deisenhammer and Christian Brenneis MD in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We warmly thank all patients with MS for their participation in our study and Tabitha Jade-Emmanuel for proofreading this manuscript. Furthermore, thank the psychologists, physiotherapists and occupational therapists who supported the study organization, Andreas Mühlbacher, MSc, and Barbara Linert, provided the study intervention, Katrin Schüler, Christina Willard, BSc, Magdalena Dippner, BSc, and Bettina Rahm. Additionally, or performed the blinded assessments, Mag. Johanna Wenter, Mag. Helga Gruters, Mag. Christine Schuster, Ursula Miller, Alissa Mali, BSc, Johanna Wüstner, BA, BSc, Simone Kircher, MSPhT, Sabrina Kienast, BSc, Nina Zenleser, BSc, Lisa Reitmeir, BSc, and Gudrun Martinz, MSc.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Bristol-Myers Squibb (NA-CL-MS-PI-13909).
Data Sharing
Requests will be reviewed by the corresponding author and study sponsor, who will consider the feasibility and appropriateness of the request and the credentials of the requester. If the request is deemed reasonable and in line with scientific purposes, de-identified participant data will be shared by the corresponding author with colleagues who made the request. To ensure data protection and responsible usage, requesters will be required to sign a data sharing agreement before obtaining access to the requested data.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
