Abstract
Background
Caloric restriction promotes neuroplasticity and recovery after neurological injury. In mice, we tested the hypothesis that caloric restriction can act post-stroke to enhance training-associated motor recovery.
Methods
Mice were trained to perform a skilled prehension task. We then induced a photothrombotic stroke in the caudal forelimb area, after which we retrained animals on the prehension task following an 8-day delay. Mice underwent either ad libitum feeding or alternate day fasting beginning 1-day after stroke and persisting for either 7 days or the entire post-stroke training period until sacrifice.
Results
Prior studies have shown that post-stroke recovery of prehension can occur if animals receive rehabilitative training during an early sensitive period but is incomplete if rehabilitative training is delayed. In contrast, we show complete recovery of prehension, despite a delay in rehabilitative training, when mice underwent alternate day fasting beginning 1-day post-stroke and persisting for either 7 days or the entire post-stroke training period until sacrifice. Recovery was independent of weight loss. Stroke volumes were similar across groups.
Conclusions
Post-stroke caloric restriction led to recovery of motor function independent of a protective effect on stroke volume. Prehension recovery improved even after ad libitum feeding was reinstituted suggesting that the observed motor recovery was not merely a motivational response. These data add to the growing evidence that post-stroke caloric restriction can enhance recovery.
Introduction
Ever since Hippocrates described that fasting may lessen the severity of the “sacred disease,” now known as epilepsy, there has been excitement about caloric restriction’s effects on the brain. 1 Indeed, animal studies since the time of Hippocrates support the evolutionary principle that the brain and body perform at high levels in environments with reduced access to calories. Caloric restriction impacts multiple signaling pathways,2,3 promotes neuroplasticity, 4 and can optimize brain function 5 leading to increased life span, 6 resistance against neurodegenerative diseases, 7 and improved cognitive function.8-10 Here we use the term “caloric restriction” to refer to any paradigm in which an animal’s calories are reduced over a given period.
Motor recovery post-stroke is a complicated process thought to require a change in the output pathways post- as compared to pre-stroke—a process that we refer to here as “neuroplasticity.” Since caloric restriction can impact neuroplasticity, it seems reasonable that caloric restriction post-stroke may impact (perhaps improve) motor recovery. Indeed, recent work has suggested that caloric restriction may improve motor recovery after neurological injury3,11 reduce the effects of acute stroke by reducing infarct volume, increasing neuronal survival, 11 and increasing spatial memory performance. 7
Any intervention aimed at post-stroke motor recovery should consider the “sensitive period”—an early, time-limited period of heightened plasticity characterized by molecular, physiological, and structural changes, which are qualitatively and quantitatively distinct to plasticity mechanisms in the absence of stroke or in the presence of a chronic stroke.12,13 The sensitive period is uniquely sensitive to behavioral and pharmacological interventions and typically lasts 1 to 4 weeks in rodents (likely longer in humans); the most intense period of recovery occurring in the first week post-stroke in mice.13-16
Using a mouse model of focal motor stroke and a skilled prehension task (reach-to-grasp), we tested the hypothesis that imposing caloric restriction early post-stroke would improve motor recovery. We also tested whether these effects were a result of a change in stroke volume related to the introduction of caloric restriction.
Methods
Mice
Adult male C57bl/6 mice (Charles River) 140 to 170 days old were singly housed in custom-made chambers modified for task training and kept on a 12/12-hour light/dark cycle similar to prior experiments. 13 A total of 101 mice were used and 17 mice eventually excluded from the study. a priori, we set criteria to exclude mice that were unable to attain a baseline prehension success rate of >30% (1 mouse excluded), died either pre-stroke (in transfer) or perioperatively (10 mice excluded), or did not show evidence a stroke in the caudal forelimb area (CFA; 6 mice excluded) upon pathological analysis. All animals were handled and cared for according to protocols approved by the Johns Hopkins University Animal Care and Use Committee.
Calorie Restriction
Two different types of caloric restriction were employed in these studies (Figure 1). Animals were fed either 500 mg (Bio-serv Product# F0171) or 45 mg chocolate-flavored pellets (Bio-serv Product# F0299). Chocolate flavored pellets were used to encourage feeding even when the animals were not weight deprived. Additionally, the chocolate-flavored pellets contained 9% more carbohydrate, 45% more fat, 14% less protein, and 8% more caloric intake per pellet to help offset possible nutritional deficiencies. All animals received water ad libitum. None of the animals, regardless of diet, (ad libitum, limited daily feeding, or alternate daily fasting), exhibited signs of undernutrition, such as exsiccation or fur abnormalities, stool paleness, or increased stooling, which are symptoms indicative of intestinal malabsorption. 17
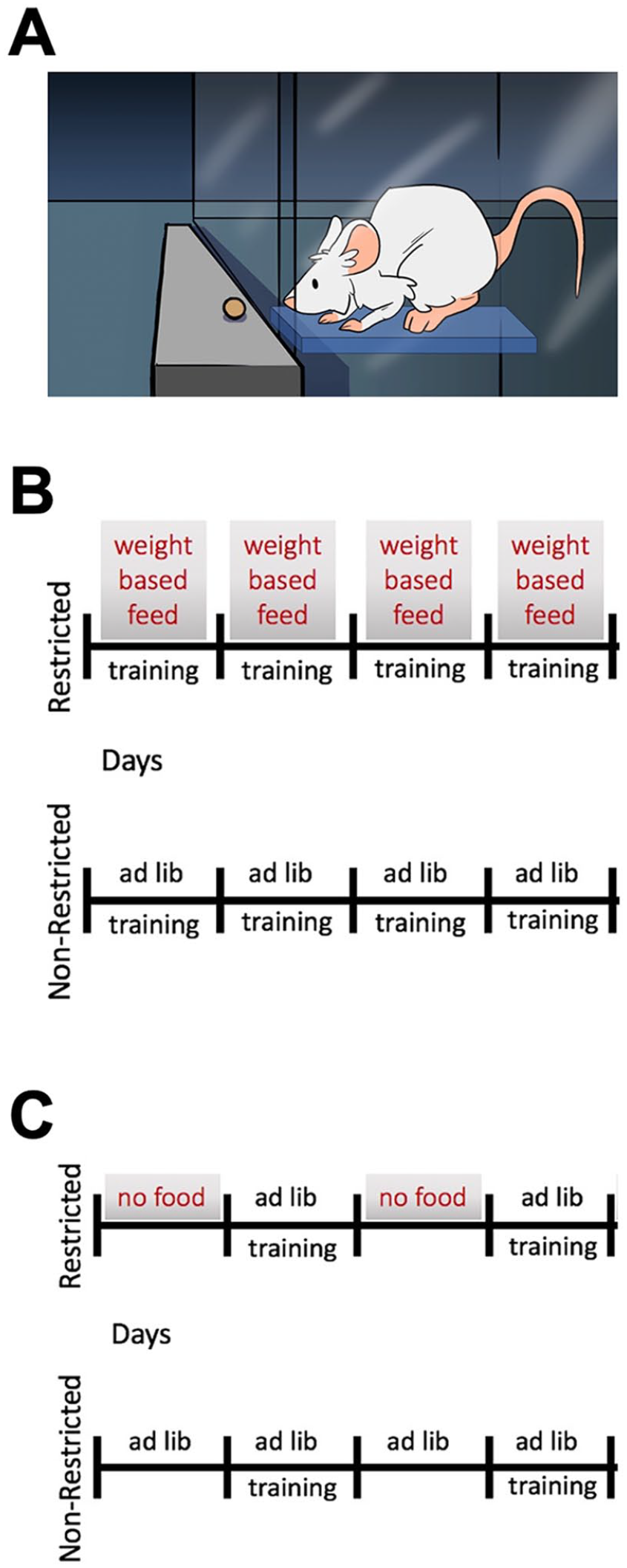
Caloric restriction protocols. (A) Limited daily feeding protocol. Animals in the restricted group received daily feeding (modified to maintain weight of approximately 85% of starting weight) + daily prehension training. Animals in the ad libitum group received ad libitum daily feeding + daily prehension training. (B) Alternate day fasting protocol. Animals in the restricted group received every-other-day ad libitum feeding (without any weight-based modifications) + every-other-day prehension training, which occurred on the same day as feeding. Animals in the ad libitum group received ad libitum daily feeding + every-other-day prehension training.
Limited Daily Feeding
Control group mice were fed ad libitum. Experimental group mice were fed approximately 85% to 90% of the food by weight that control mice received. Both control group mice and experimental group mice were weighed daily. Control group mice had no changes to their food amount; however, experimental group mice food amount was modified to keep their weight at 85% (±5%) of their starting weight (Figure 1A).
Alternate Daily Fasting
Control group mice were fed ad libitum daily while experimental group mice received alternate day, ad libitum feeding with no feeding on subsequent days (Figure 1B). This was accomplished by removing food at a precise time of day, and then providing food at the same time the next day and repeating this cycle of fasting and feeding days. Feeding days began with prehension training; since training requires food, there was no prehension training on non-feeding days. Thus, alternate day fasting animals have prehension scores reported every-other-day.
Skilled Prehension Task Training
Training was conducted as described previously.13,15,16 Briefly, standard cages were modified by cutting an approximately 1 cm wide slit in the front and a platform was added (Lincane Aluminum Sheet, Home Depot, SKU # 1001195127) inside of the cage and near the slit. This platform allowed animals to efficiently approach the prehension platform (Figure 1A).
Mice were familiarized with the 45 mg chocolate pellets (45 mg Bio-Serv precision pellets, F0299). Each mouse then underwent paw-preferencing, which consisted of the trainer encouraging the animal to approach the cage slit by showing the food pellet to the animal. Two food pellets were presented simultaneously, one on either side of slit and resting on a steel bar the same height as the platform in the cage. The trainer ascertained which paw the animal preferred to use during an average of 10 trials. Paw preferencing grasps were not scored as part of an animal’s prehension success. Paw preferencing occurred only once per animal.
After paw preference was determined, animals were trained and scored. During training, 45 mg pellets were placed on the steel bar that is the same height as the platform found in the cage (Figure 1A). Pellets were placed on the side of the steel bar forcing preferred paw use. A double-sided tape was used on the steel bar (Scotch Double-Sided Tape, 3M) so that the 45 mg pellets could be placed on the double-sided tape about 0.5 cm away from the cage slit. Special care was taken to ensure that the pellets were not placed closer to the cage. Successful grasps occurred when the animal reached its forelimb through the slit, grabbed the pellet, and ate the pellet without knocking the pellet from its resting space. Alternatively, if the animal dropped the pellet, lost control of the pellet in any way, or knocked the pellet before the animal consumed it, then the attempt was recorded as a miss. If the animal did not touch the pellet, then the reach did not count as an attempt; thus, the percent of successful grasps was based upon the total number of pellets provided. A training block consisted of 30 pellets with each pellet presented 1 at a time; the percentage of successful grasps was chosen as the behavioral measure of motor function. A training block continued until the animal reached for 30 pellets and each reach was scored as either “correct” or “not correct.” One training block was performed once per training day.
Stroke Induction
The surgical setup was as described in prior work, 13 which we have used with success During stroke, animal anesthesia was induced with 4% isoflurane and maintained under 2% to 2.5% with 2 L of O2. The location of motor areas was identified based upon prior anatomic 18 and functional 19 data. These data also indicate that these areas are geographically consistent within a given strain. Briefly, under isoflurane anesthesia, the skull was exposed via skin incision, and a point 2 mm lateral and 0.5 mm anterior from bregma was located, corresponding to the CFA contralateral to the mouse’s preferred paw. This point on the skull was thinned using a Dremel power drill. A fiber optic bundle of a cold light source (Zeiss 1500 electronic, Jena, Germany) with a 20-G aperture was centered at 2 mm lateral and 0.5 mm anterior from bregma for CFA infarction. The brains were then illuminated through the intact skull for 15 minutes, starting 5 minutes after the intraperitoneal injection of 150 μl of a 10 mg/mL rose Bengal solution in sterile normal saline. Animals were monitored on a warming blanket during recovery.
Tissue Preparation and Histology
On the day of sacrifice, the mice were placed under deep anesthesia with 2.5% avertin and transcardially perfused with 4% paraformaldehyde (PFA) in 0.1 M sodium phosphate, pH 7.4. The brains were dissected out and placed in 4% PFA for 24 hours. Brains were coronally sliced at 50 µm on a vibrating microtome. Free-floating sections were washed 3 minute × 5 minute in PBS and placed for 4 hours 10% normal goat serum and 0.04% triton X-100, in tris-buffered saline. Sections were subsequently washed 3 minute × 5 minute in tris-buffered saline with 0.04% triton ×100, 1 minute × 5 minute tris buffered saline, and mounted in ProLong Gold reagent (Invitrogen).
Quantification of Stroke Volume
From brain slices prepared as above, histology detected by the endogenous fluorescence in the green fluorescent protein (GFP) emission channel (525 nm) was collected. The entire anterior-posterior extent of the CFA contralateral to the preferred paw was imaged and reconstructed in 3 dimensions using Zen Blue Imaging software (Zeiss). An investigator blinded to conditions demarcated the stroke pathology and volumes were calculated using Zen Black imaging software to quantify 3-dimensional stroke volume as previously described. 20
Statistics
Behavioral data were analyzed based on prior work with a linear mixed-effect model.15,16 Briefly, row means were analyzed with multiple a mixed-effect analysis (assuming non-Gaussian distributions) and corrected for multiple comparisons using the Yekutieli method with a 1% false discovery rate approach. Comparisons were made between each group’s mean with the other group’s mean for the given time-period. We have previously shown that using such an approach account for any mouse- and group-specific differences at either baseline or after training. 15
Weight data were analyzed using 2-way analysis of variance (ANOVA) with comparison to 100% of pre-stroke baseline as well as to the opposing condition for each animal; multiple comparisons were corrected for using the Tukey method as each group’s mean was compared with the other groups’ means for the given time period.
Stroke volume data were analyzed using 2-way ANOVA between the conditions (ad libitum, alternate day fasting for the entire poststroke period, versus alternate day fasting for the first 7 days poststroke). No significant interaction was found between the conditions.
Results
Caloric Restriction Had No Significant Effect on Prehension Prior to Stroke
We have previously shown that wild-type mice trained to perform a skilled prehension task reach maximum accuracy by approximately 10 days.13,15 Mice trained to reach with or without caloric restriction performed ultimately similarly (Figure 2). First, we compared animals that were food deprived to 15% of their starting weight to animals fed ad libitum. Animals performed similarly (Figure 2A), despite a decrement in weight (Figure 2B). Second, we compared animals fed using alternate day fasting to animals fed ad libitum. Animals fed using alternate day fasting performed ultimately similar to animals fed ad libitum (Figure 2C). The weights of animals are shown (Figure 2D).
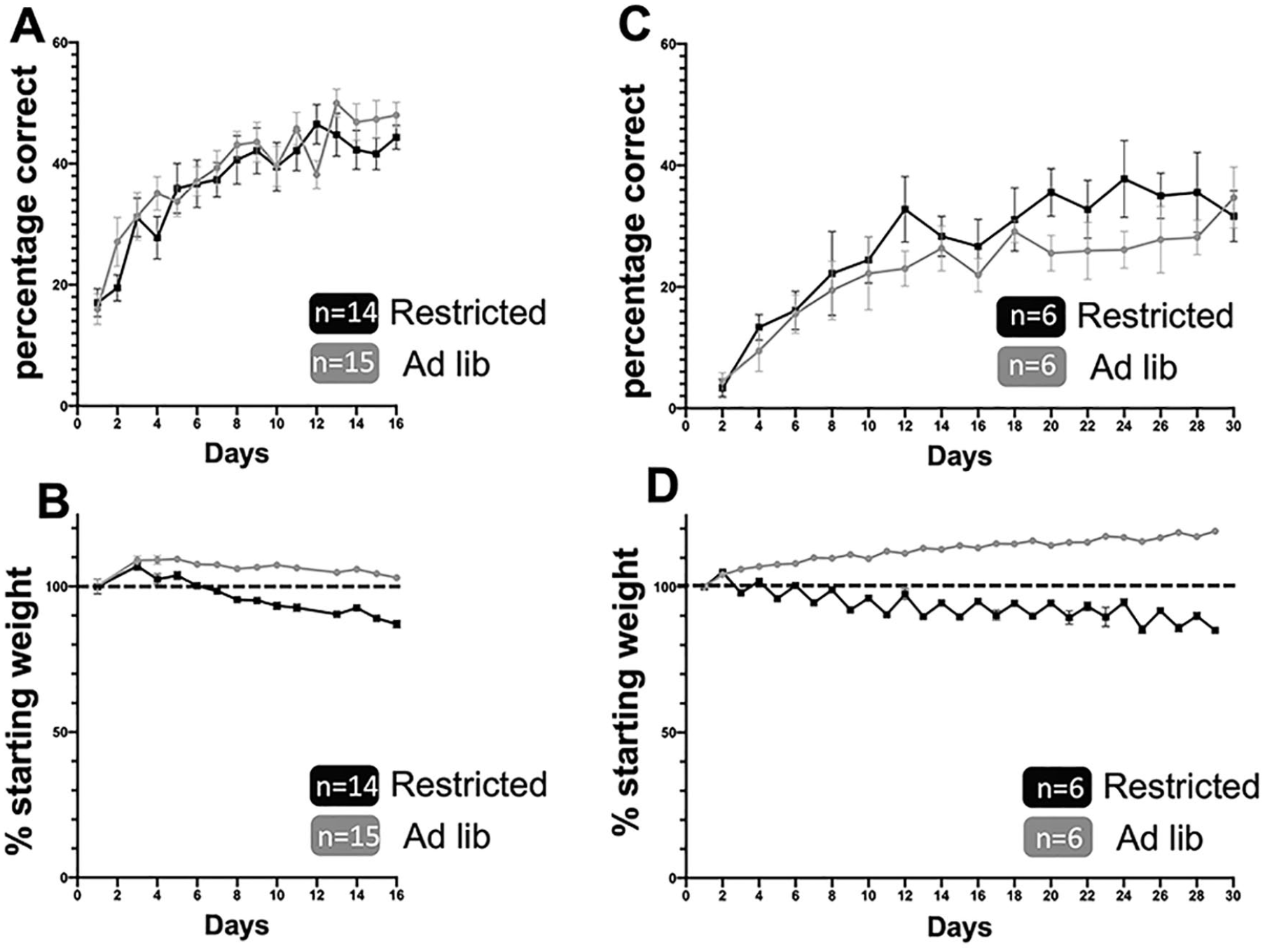
Caloric restriction does not alter prehension training success. (A and B) Animals treated with the limited daily feeding protocol (A) prehension success (mean ± scanning electron microscope [SEM]) and (B) weights as a percentage of beginning weight. (C and D) Animals treated with the alternate day fasting protocol (C) prehension success (mean ± SEM) and (D) weights as a percentage of beginning weight.
For the remainder of the testing, alternate day fasting was chosen because it has previously been shown to maintain any positive effects of caloric restriction without decreasing average daily food intake or nutritional intake.5,6,21,22 Further, the food pellets used for the alternate day fasting had enhanced nutritional components (Materials and Methods section). Animals in both the ad lib and alternate day fasting groups received the same food pellets. Prior work has shown no reduction in nutrition with these food pellets.
Caloric Restriction Improved Post-Stroke Motor Recovery
We have previously shown that post-stroke rehabilitative training on the prehension task beginning after a 8-day delay led to incomplete motor recovery; however, if training began after a 1-day delay, recovery was complete.13,15,16 The main hypothesis of this study was that caloric restriction during the sensitive period would improve poststroke motor recovery even when rehabilitative training was delayed by 8 days (i.e. caloric restriction would encourage recovery during a time period where prior work has shown that motor recovery is incomplete—even with rehabilitative training). To test this hypothesis, we trained mice on the prehension task to an asymptotic level of performance, induced a focal stroke in the CFA contralateral to the preferred paw, and calorically restricted animals as described. Alternate day fasting beginning 24 hours post-stroke and continuing through training days led to a significant improvement in prehension skill (Figure 3A) (results summarized in Table 1). The deprived animals showed a decrement in weight that fluctuated with feeding (Figure 3B) and was lower than their starting point throughout the poststroke training period.
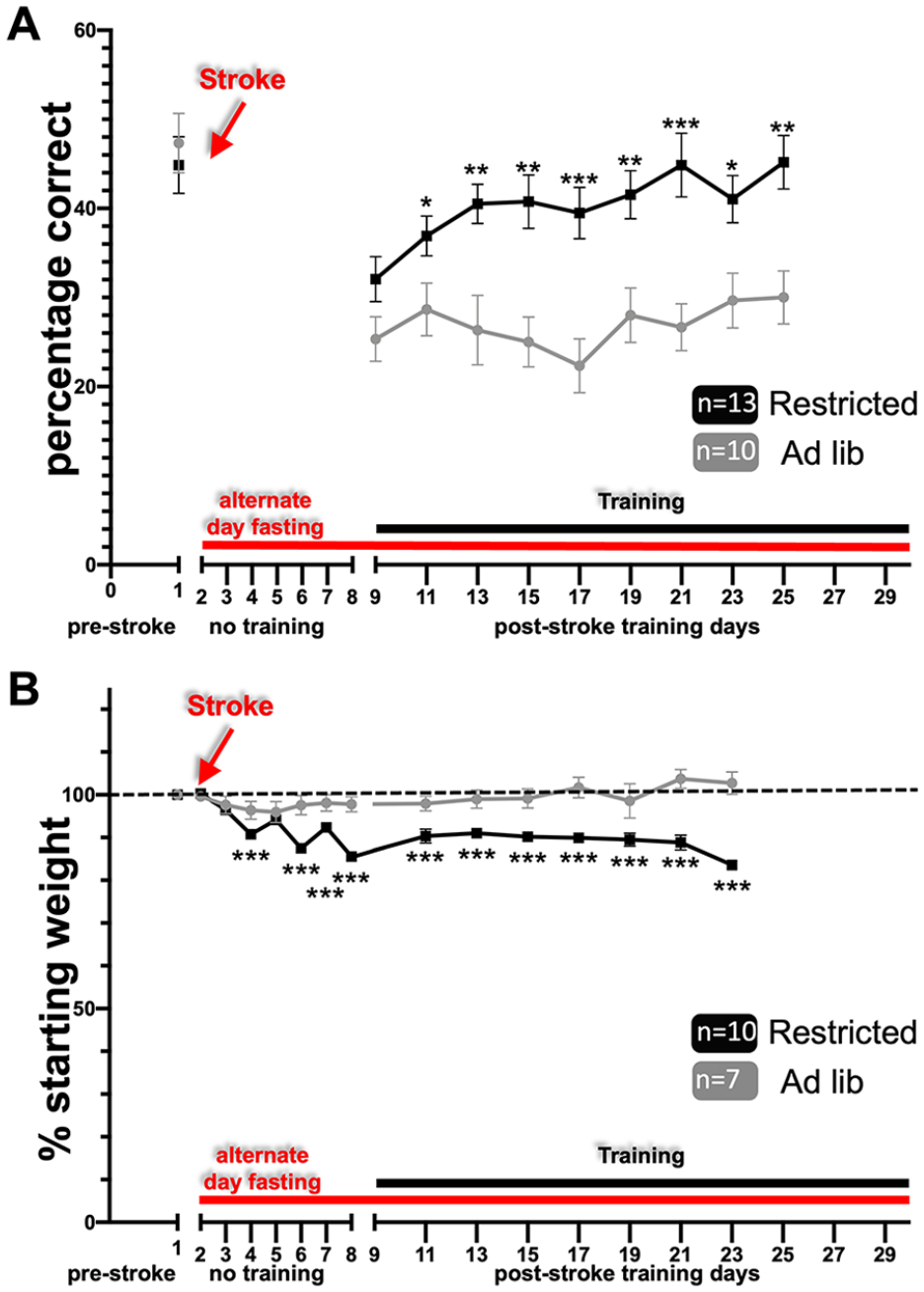
Alternate day fasting post-stroke leads to improved recovery. (A) Animals treated with the alternate day fasting protocol (A) prehension success (mean ± SEM) and (B) weights graphed as a percentage of pre-stroke weight (mean ± SEM). Animals experienced alternate day fasting (red line) beginning 24 hours post-stroke and continued during the course of their post-stroke rehabilitation. Training (black line) began after an 8-day post-stroke delay. SEM, standard error of the mean.
Summary of the Effects of Alternate day Fasting on Post-Stroke Motor Recovery.
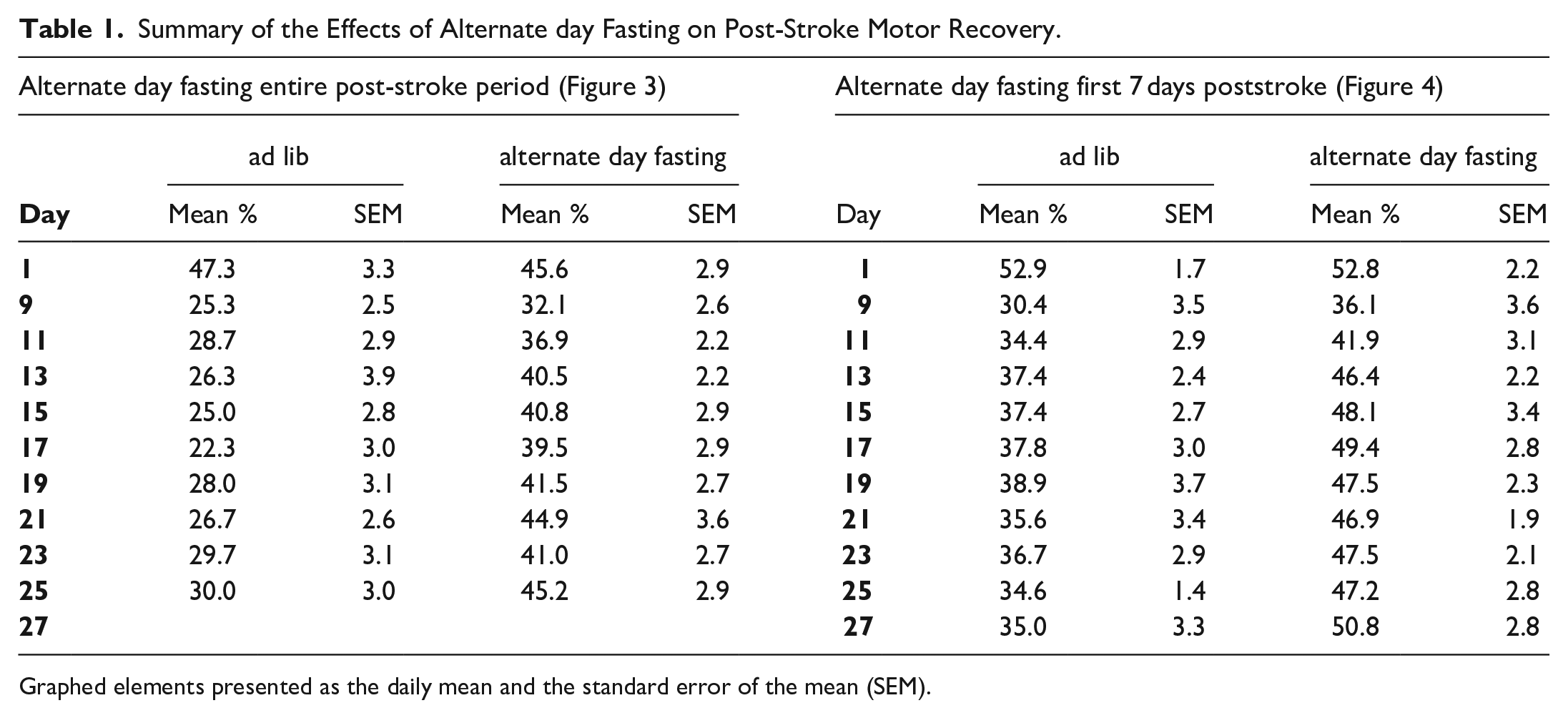
Graphed elements presented as the daily mean and the standard error of the mean (SEM).
To test if improved prehension skill was related directly or indirectly to reduced weight, we next trained mice on the prehension task to an asymptotic level of performance, induced a focal stroke in the CFA contralateral to the preferred paw, and fed animals every-other-day beginning 24 hours post-stroke for only 8 days (Figure 4). Using this scheme, we recorded oscillating weight loss compared to baseline for the first 8 days post-stroke, which subsequently normalized with reinstitution of ad libitum feeding (Figure 4B). Nevertheless, animals that were fed every-other-day only immediately post-stroke still showed improved prehension skill as opposed to animals that were fed ad lib (Figure 4A; results summarized in Table 1).
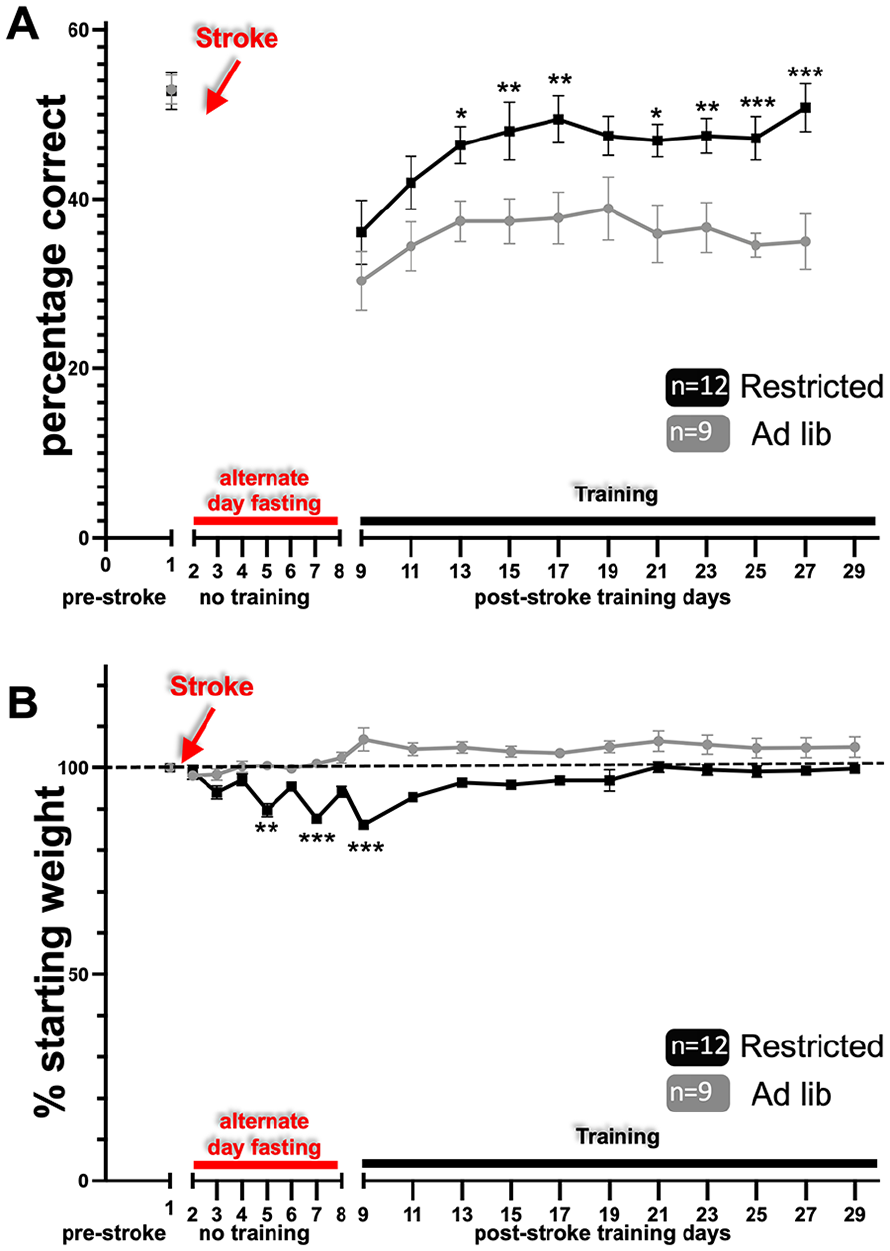
Early alternate day fasting post-stroke leads to improved recovery. (A) Animals treated with the alternate day fasting protocol (A) prehension success (mean ± SEM) and (B) weights graphed as a percentage of pre-stroke weight (mean ± SEM). Animals experienced alternate day fasting (red line) beginning 24 hours post-stroke and continued during the course of their post-stroke rehabilitation. Training (black line) began after an 8-day post-stroke delay. SEM, standard error of the mean.
Caloric Restriction Did Not Alter Stroke Volume
To test if caloric restriction led to changes in stroke volume, mice were euthanized within 2 hours of their last training session, and stroke volumes were measured. Regardless of the condition, there were no significant stroke volume differences between animals receiving an ad lib diet, animals calorically restriction for the first 7 days, versus animals calorically restricted for the entire post-stroke training period (Figure 5).
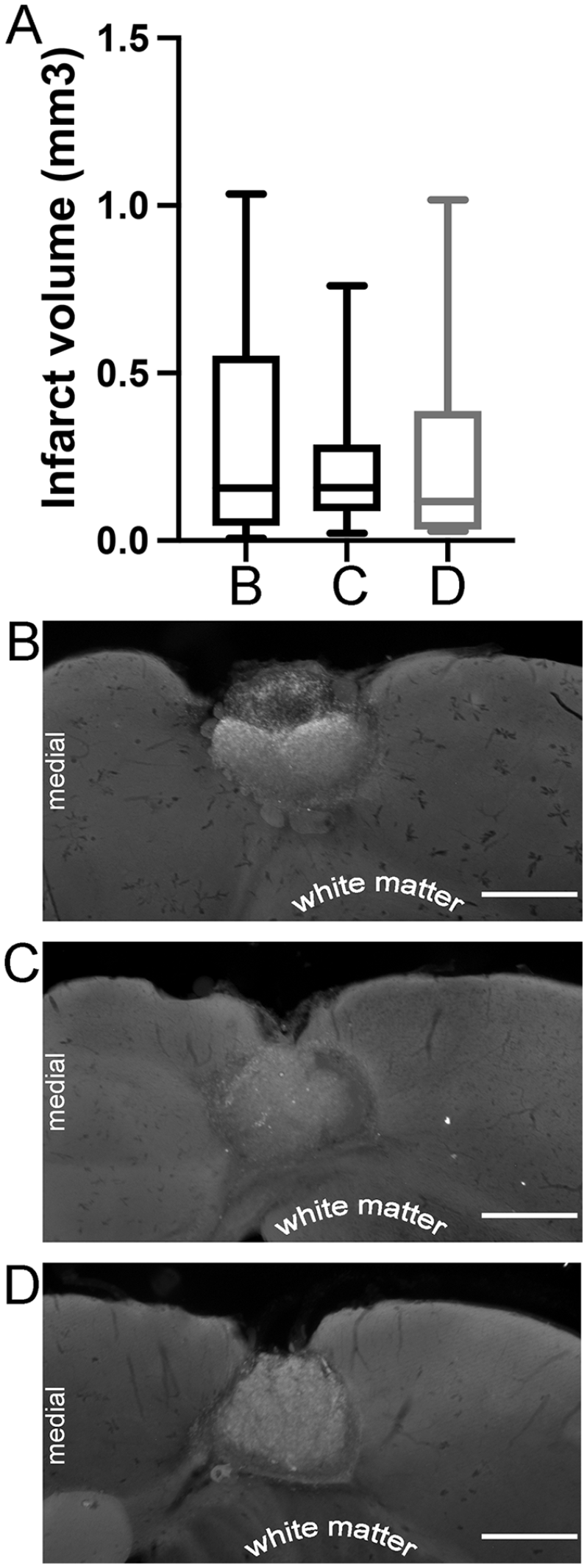
Infarct volumes. (A) There was no significant difference between CFA infarct volumes in mice experiencing (B) ad libitum feeding (n = 23), (C) alternate day fasting for the entire post-stroke time period (n = 13), or (D) alternate day fasting for the first 7-days post-stroke followed by ad libitum feeding (n = 14). (B, C, and D) Representative histology for the indicated conditions. Scale bar = 500 um. Error bars (A) represent standard error of the mean.
Discussion
Using a mouse photothrombotic stroke model, we show here that alternate day fasting during the post-stroke sensitive period leads to improved motor recovery, even if ad libitum feeding was reinstituted and weights normalized. The enhanced motor recovery was independent of stroke volume differences between the conditions.
Caloric Restriction and Stroke Recovery
Physiological stressors tend to impair stroke recovery. For example, sleep deprivation, 23 depressed mood,17,24 pain, 17 and malnutrition 25 have all been associated with reduced post-stroke recovery. Although intense and/or prolonged caloric restriction may act as a physiological stress, 26 alternate day fasting was associated with improved stroke recovery when introduced early after stroke. Since a highly conserved behavioral trait of mammals is to be active when hungry and sedentary when satiated, we tested if hunger-motivation was responsible for the improved recovery. The observation that animals continued to improve even after ad libitum feeding was reinstituted and weight normalized (Figure 4) suggests that the observed motor recovery is not merely a response to reduced weight and any associated motivational response correlating with reduced weight. For example, on post-stroke day 9 (Figure 4), the animals are no longer calorically restricted (there is no feeding difference between the groups marked “restricted” and “ad lib”). Indeed, the upward slope of prehension recovery continues despite weight normalizing to 100% of pre-stroke levels. Nevertheless, there are 2 potential confounds with this interpretation. First, it is possible that weight does not correlate perfectly with motivation. For example, motivational responses to caloric restriction may follow a different timeline than does weight. Second, we did not record no-contact reaches (prehension attempts in which the animal extended a paw but did not contact the pellet), suggesting the possible bias that calorically restricted animals may have been more active during a non-recorded phase of our training. Nevertheless, our results suggest that caloric restriction may act via mechanisms distinct from motivation during the post-stroke sensitive period.
Caloric restriction in general, and alternate day fasting in specific, has been shown to produce remarkably complex and coordinated adaptations in the healthy brain.2,5,21 For example, caloric restriction increases brain-derived neurotrophic factor (BDNF) expression in several regions of the brain, which in part mediates caloric restriction’s enhancement of synaptic plasticity and neurogenesis. 2 There is evidence that alternate day fasting can enhance brain function as indicated by improved motor function and improved memory. 2 There is additional data that caloric restriction impacts the activity of the nicotinamide adenine dinucleotide (NAD)-dependent lysine deacetylase sirtuin 1 (Sirt1),27,28 which is involved in the epigenetic and non-epigenetic adaptive responses of neurons to metabolic challenges. 29
Although the exact pathways implicated in post-stroke recovery remain elusive, recent work has suggest that caloric restriction increases BDNF and Sirt1 signaling in the peri-infarct brain tissue. 11 In addition, alternate day fasting after spinal cord injury increased the ratio of full-length to truncated trkB (the receptor for BDNF) in the spinal cord, which correlated with neuroplasticity and improved recovery. 22 Both the BDNF and Sirt1 signaling pathways have been implicated in stroke recovery.30,31 These results imply that caloric restriction may enhance the ability of surviving and relevant nervous tissue to engage in recovery promoting neuroplasticity. Further, caloric restriction is likely to act on multiple pathways at a cellular, organ, and organismal level.5,10 In contrast to interventions that act on a single signaling pathway, caloric restriction’s plethoric effects are likely to be important when encouraging a recovery response post-stroke. 12
Caloric Restriction and the Sensitive Period
One important aspect of our paradigm is the pairing of rehabilitative training with alternate day fasting. In prior work, we have shown that there is diminishing responsiveness to motor rehabilitative training over the first week post-stroke—a period that we refer to as the sensitive period.15,16 Rehabilitative training during this sensitive period leads to near complete recovery that is not seen if training is delayed for 1 week. Post-stroke, there is a subsequent cascade of degeneration, neurotoxicity, inflammation, and apoptosis in the ischemic core and penumbra, with consequences for neuronal and synaptic survival. Consequently, in the peri-infarct region and connected areas, there are gene expression changes, electrophysiological changes, and structural changes which are thought to mediate recovery and are likely influenced and/or enhanced by rehabilitative input.12,32 Our data suggest that caloric restriction interacts “positively” with these recovery-relevant mechanisms during the sensitive period.
Prior studies have suggested that caloric restriction is neuroprotective and may lead to improved outcomes via reduced injury.11,22. Alternatively, our results indicate that prehension recovery is mediated via mechanisms distinct from reducing stroke volume (since fasting conditions had no effect on stroke size). One reason for the difference between our results and others’ suggesting that caloric restriction is neuroprotective may relate to the different outcome tests, the timing of the caloric restriction, and the type of stroke model employed.33-35 For example, Arumugam et al 35 suggests a possible relationship between caloric restriction and reduced infarct volume; however, the animals in their study were calorically restricted for months prior to transient middle cerebral artery occlusion/reperfusion ischemia. It is difficult to compare our post-stroke alternate day fasting paradigm to Arumugam et al’s 35 pre-injury fasting, especially since pre-injury fasting alters cellular stress responses and likely encourages a protective response. Another difference between our study and the Arumugam et al 35 study is that our photothrombotic strokes likely experience different repair dynamics compared to the larger strokes created by their vessel occlusion models of ischemia—especially since such models are particularly susceptible to neuroprotection.
Nevertheless, others have reported results similar to ours even when using large vessel strokes. For example, Roberge et al 7 reported no evidence of a neuroprotective effect despite demonstrating large vessel, post-stroke, caloric restriction-induced spatial memory recovery. 7 Further, Roberge et al 7 also employed early and repeated rehabilitative training during the sensitive period. Coupled with ours, these results suggest that employing early rehabilitation is important in directing recovery-based neuroplasticity mechanisms.
Neural repair strategies for stroke recovery (perhaps involving BDNF, Sirt1, and other signaling pathways) represent a major opportunity to reduce the global impact of stroke. 37 We presume that caloric restriction operates at least in part through these pathways. Our observed motor recovery was independent of neuroprotection, at least as measured by stroke volume, and is consistent with other studies showing similar outcomes.
Importance to Human Recovery
Concerns regarding the applicability of caloric restriction in humans include feasibility and excessive weight loss. However, the Comprehensive Assessment of the Long-term Effects of Reducing Intake of Energy trial, in which non-obese adults were randomized to a normo-caloric diet or a 25% caloric restriction, demonstrated feasibility of the diet, sustained metabolic adaptation to caloric restriction, re-duced oxidative stress, 38 and minimal if any side effects 39 in human subjects. Nevertheless, and despite repeated trials showing that uninjured human subjects can maintain their weight even while engaging in normal activity, 40 caloric restriction has not been tested on stroke patients. Combined with our data, these studies suggest the possibility that caloric restriction early after stroke could safely improve post-stroke recovery in humans. For example, alternate day fasting in the subacute, post-stroke period may be feasible and effective when combined with multi-disciplinary services that oversee nutrition, rehabilitation, and secondary stroke prevention. Even though analysis of energy expenditure suggests that post-stroke patients are not hypermetabolic, 41 care should be exercised when applying alternate fasting post-stroke. It is conceivable that post-stroke metabolic demands are altered compared to normal, suggesting that any caloric restriction could potentiate muscle atrophy, weight loss, or other side effects. As such, the direct application of caloric restriction to patients post-stroke should be carefully planned and studied.
Limitations and Future Studies
Although our study serves as a benchmark linking alternate day fasting and the enhanced recovery seen during the sensitive period, our study did not investigate specific mechanisms. Future studies might consider casting a wide net that investigates multiple underlying mechanisms. Additionally, future studies might investigate the efficacy of different types of caloric restriction and how they interact with factors important in post-stroke recovery including advanced-aged and/or vascular risk factors (eg, diabetes mellitus, hyperlipidemia, and obesity).
Finally, future studies might investigate the timing of caloric restriction and its effects on recovery. The sensitive period of upper extremity recovery is a “fuzzy” temporal boundary lasting approximately 1 week in mice, perhaps 2 to 4 weeks in rats, 14 and as long as 3 to 6 months in humans.12,32 Other stroke-induced symptoms (eg, aphasia) may follow different recovery timelines.12,42 As such, when human studies are considered, one must consider the interaction between caloric restriction, its timing relative to stroke, the feasibility of caloric restriction, and the outcome measure.
Conclusions
We show here that alternate day fasting enhances post-stroke motor recovery independent of weight and independent of stroke volume. These data support further investigation of the mechanisms of action of caloric restriction in stroke models and support further clinical trials in patients.
Footnotes
Author Contributions
Mahlet D. Mersha: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Writing—original draft; Writing—review & editing. Robert Hubbard: Investigation; Methodology. Steven R. Zeiler: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Software; Supervision; Validation; Visualization; Writing—original draft; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to acknowledge the grateful philanthropic donations, which funded this study in its entirety, made by Jackie Lapidus on behalf of Len Lapidus, a grateful patient interested in stroke recovery.
