Abstract
Introduction
In 2013, the global prevalence of stroke was 25.7 million with a pooled cumulative 10-year risk of 39.2%. 1 Stroke survivors have approximately half the cardiovascular fitness (CRF) 2 and a 40% to 50% greater energy cost of walking compared with nonstroke individuals. 3 These limitations facilitate an inactive lifestyle that results in further deconditioning and an increased risk of future cardiovascular events. 4
Maintaining cardiovascular health is the most important strategy for lowering the risk of recurrent stroke,
5
but historically, stroke rehabilitation programs provide limited opportunity for cardiovascular training,
6
especially for high-intensity cardiovascular training. Physical activity and exercise recommendations poststroke include moderate-intensity continuous cardiovascular exercise (MICE) to improve CRF with a suggested intensity of 40% to 70% maximal oxygen consumption (
High-intensity interval training (HIIT) has emerged as a powerful time-efficient alternative to MICE for individuals without known disability,
13
as well as clinical populations.14-16 HIIT is characterized by high-intensity bursts of cardiovascular exercise interspersed between bouts of recovery and aims to maximize exercise intensity with a lower time commitment and total exercise volume.
13
HIIT has been shown to be more time-efficient
16
and an effective alternative to traditional MICE training, with evidence showing similar or even superior adaptations despite substantially lower exercise volumes in both clinical and non-clinical populations.
13
Current evidence suggests that HIIT is superior to MICE in individuals with heart disease,
14
with greater
HIIT could supplement the low cardiovascular intensity typically achieved in stroke rehabilitation programs by achieving higher intensities required to optimize recovery. 10 Importantly, the exercise parameters in HIIT may be adapted to suit the needs of different populations and training objectives. 21 Despite the perceived benefits, there exist noteworthy barriers to the implementation of HIIT within the stroke population, including cardiovascular deconditioning, 22 stroke-related neurological sequelae such as motor and balance impairments 23 and an even greater cardiovascular stress on individuals with stroke 24 compared with MICE. Safety, therefore, needs to be assured through rigorous trials.
Poststroke HIIT protocols may be categorized into 3 main types depending on high-intensity burst duration, recovery duration, and recovery type.
22
Albeit preliminary, recent evidence suggests effectiveness in HIIT-associated functional,17-19 cardiovascular, 17 and neuroplastic25,26 outcomes in individuals poststroke. HIIT may provide an alternative exercise option with the potential to invoke more pronounced cardiovascular and cerebrovascular benefits.
The aim of this article is to provide an updated review of HIIT in stroke rehabilitation practice by (
Current Evidence Surrounding the Safety and Effectiveness of HIIT in Stroke
Is HIIT Safe in Individuals With Stroke?
Among individuals with coronary artery disease, 14 myocardial infarction14,27 and heart failure, 28 there have been only 2 nonfatal cardiac arrests in more than 46 000 hours of HIIT during cardiac rehabilitation, with occurrences similar to MICE. 29 To reduce the risk of adverse events during HIIT training poststroke, preemptive medical screening and careful monitoring of heart rate, blood pressure and rating of perceived exertion (RPE) during exercise has been implemented. 22 In addition, because of the high occurrence of silent myocardial ischemia rates reported within the stroke population, exercise testing with electrocardiography is recommended prior to initiating cardiovascular exercise. 7 Continuous telemetry may be used but may not be available in many stroke rehabilitation settings. 22 In research studies, the risk of orthopedic injury has been minimized by excluding individuals with primary orthopedic conditions (eg, fractures, active rheumatoid arthritis) or severe motor impairments (eg, inability to walk without assistance18,19,30) and incorporating the use of body weight support, orthotic devices (eg, splints and ankle-foot orthoses) or providing handrail support as needed during training. 22 With such prospective safety monitoring in place, no serious adverse events were observed in the 10 studies that were identified to date,17-19, 23, 25, 26, 30-33 and only 2 occurrences of symptomatic hypotension and near syncope during recovery. 31
Important to consider is the response within the cerebrovasculature, which is different than the periphery. 72 High-intensity exercise has the potential to provoke rapid increases in blood pressure that may be transferred to the cerebrovasculature. 72 This might, theoretically, increase the risk of hyperperfusion injury 72 subsequent to functional impairments in dynamic cerebral autoregulation. While this may be of concern, whether the degree of hypertensive response induced with high-intensity exercise is potentially damaging has not been confirmed. Intense exercise in individuals without disability has the potential to increase blood-brain barrier permeability without causing structural brain damage. 35 From a practical perspective, however, it appears prudent to mitigate this potential risk on the cerebrovasculature by gradually increasing the exercise intensity over the first 10 seconds of every high-intensity burst. 72
There is also the potential for hypotensive responses secondary to venous pooling during recovery periods, 31 which highlights the importance of adequate hydration, and considering the use of seated recovery positions over standing positions to minimize fall risk. While using active, rather than passive, recovery may be applied to mitigate hypotensive responses, clinicians should be aware that active recovery may decrease HIIT tolerance. 22
Collectively, the evidence suggests that HIIT is a safe approach with no reports of serious adverse events poststroke. However, implementing large randomized controlled trials to investigate safety in a broader range of stroke populations, such as subacute and chronic stroke and with and without comorbid conditions, would be beneficial.
Is HIIT Effective in People Poststroke?
To date, 10 studies including 5 training studies17-19,23,30 (3 randomized controlled trials17-19) and 5 single-session studies25,26,31-33 have investigated the potential application of HIIT within the context of stroke. To summarize the evidence around the effectiveness of HIIT after stroke, findings from these 3 randomized trials are presented in Table 1, panel A. Additionally, studies examining outcomes of tolerance and feasibility31-33 (of which one was an abstract 31 ) and markers of neuroplasticity25,26 are presented in Table 1, panel B. Together, preliminary results from randomized trials suggest that HIIT may be effective in improving mobility and gait outcomes and cardiovascular health and shows preliminary evidence of positively influencing neuroplasticity.
Summary of (A) Training Studies and (B) Single-Session Studies Investigating HIIT in Individuals Poststroke.

Abbreviations: HIIT, high-intensity interval training; MICE, moderate-intensity continuous exercise; ES, effect size; 6MWT, 6-minute walk test; HRR, heart rate reserve; 10MWT, 10-meter walk test;
HIIT Is Associated With Mobility and Gait Improvements
Of the 3 randomized controlled trials, Pohl et al
19
reported that
HIIT Improves Cardiovascular Health
Exercise intensity has been shown to influence cardiovascular health in young nonclinical populations, with evidence showing that, with consistent exercise volumes, higher exercise intensities are more effective in improving CRF.
34
CRF plays a critical role in managing cardiovascular risk, with a 1-MET (metabolic equivalent; 3.5 mL/kg/min) increase translating into a 10% to 25% improvement in survival.
36
It is crucial to identify the most effective ways to improve and maintain CRF in the stroke population. Improvements in
Taken together, the aforementioned body of evidence reveals the following early trends: (
HIIT May Promote Neuroplasticity
Establishing strategies to enhance neuroplasticity is critical to promote motor recovery poststroke. 42 HIIT may facilitate neuroplasticity and motor learning after stroke, although to date, studies are limited to either poststroke animal models, 12 single-session exercise sessions poststroke25,26 or healthy populations. 38
In animal models of stroke, a recent review suggests that higher exercise intensities were necessary to optimize the expression of neurotrophins and modify synapses and dendrites to promote neuroplasticity.
12
Within healthy humans, a single 20-minute bout of HIIT at 90% of maximal power output enhanced corticospinal excitability, a marker of neuroplasticity within the motor cortex,
38
whereas another study found no effects following a single 30-minute bout of MICE training at 60%
Within a stroke population, a single bout of HIIT performed immediately after motor practice improved skill retention after 24 hours, demonstrating the potential for HIIT to trigger modest neuroplastic changes and accelerate neurorecovery. 26 Notably, skill level was increased at retention, suggesting that a single bout of HIIT may not only strengthen procedural memories but also foster off-line gains occurring between practice sessions. 26 It is possible that repeated bouts of HIIT combined with motor practice may show a cumulative effect on motor learning gains and thus accelerate functional recovery.26,43
In summary, the exercise intensity and ideal time poststroke to initiate cardiovascular exercise to optimally influence neuroplasticity following a stroke remain unknown. 12 In poststroke animal models, delaying rehabilitation to 30 days postevent have failed to show beneficial effects compared to 5 to 14 days poststroke, implying that there is an early window of opportunity to influence neuroplasticity. 12 There may be a critical period within the first few months poststroke in humans where neuroplasticity is most influenced by exercise. 44 However, exercise very acutely postevent may not be beneficial for recovery. 45 Determining more definitively when the brain is most responsive to change would be an important consideration for future trials.
HIIT and the Potential to Promote Exercise Enjoyment and Influence Exercise Adherence
A unique feature of HIIT is its potential to improve exercise enjoyment, and ultimately adherence. 46 HIIT interventions may have limited reach, effectiveness, adoption, and maintenance secondary to its potentially unpleasant stimulus. 46 However, adherence to HIIT and exercise enjoyment has been shown to be similar or superior to that of MICE in healthy individuals 47 and those with cardiometabolic disease. 16 The ability to vary the exercise parameters of HIIT makes it an adaptable exercise option for many ages and populations. 22 In addition, because of its high time-efficiency, HIIT may appeal to both clinicians and patients who are often under time constraints. However, whether HIIT may be an effective strategy to promote long-term exercise adherence and effectiveness, greater physical activity enjoyment and improved psychosocial outcomes poststroke remains unknown.
Considerations for Optimizing Exercise Parameters for HIIT Poststroke
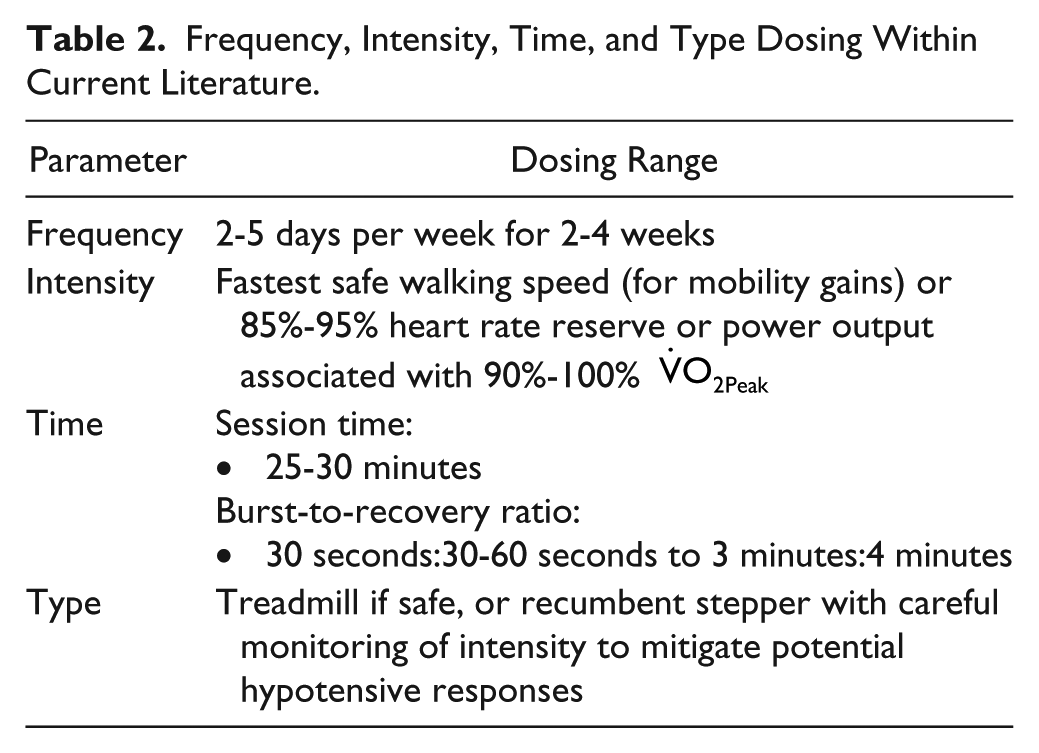
Frequency
Although limited, current evidence from 3 randomized controlled trials17 -19 suggests that the health benefits of HIIT after stroke can be achieved by exercising 2-3 times per week for 4 weeks. Delivering HIIT 5 times per week for as little as 2 weeks has led to greater improvements in gait speed and stride length, 18 while training 3 times per week for 4 weeks resulted in greater improvements compared with MICE protocols in gait (gait speed,17,19 gait economy, 17 cadence, 19 stride length 19 ), function (Functional Ambulation Category 19 ), and CRF (ventilatory threshold 17 ).
Recovery time following a HIIT session is important to consider, as recent evidence suggests a minimum of 3 days between training sessions in older adults (mean age 63 ± 3.4 years) to recover optimally and reduce the risks associated with accumulated fatigue, both of which may hinder exercise adherence. 48 It may be prudent, therefore, to initially prescribe 2 days per week of training particularly in older adults with stroke, and increasing as tolerated.
Intensity
Prescribing exercise intensity based on %
Intensity appears critical to modulate the effects of cardiovascular training on neuroplasticity and motor learning.38,39 In individuals without disability, pairing an acute bout of high-intensity exercise at workloads associated with 90% of peak power output with skilled motor practice was effective in improving motor learning,
38
while moderate-intensity exercise at a power output associated with 60%
Modalities that allow clinicians to directly monitor and easily adjust power output, such as cycle ergometers or steppers, may be preferred particularly in the early stages of HIIT training, as power output can be easily and immediately changed. Moreover, these types of seated modalities are safer than a treadmill and allow for individuals with a broad range of function to participate. In principle, if the goal is to increase cardiovascular intensity, steppers are more appropriate, especially for patients with biomechanical limitations or balance impairments affecting gait. Treadmills are beneficial if balance or biomechanical limitations are not present at high intensities of walking. Also, due to the type of movement, treadmills could possibly lead to a larger transfer of locomotor gains. It is important to note, however, that changes in gait efficiency commonly observed after stroke 7 may confound intensity prescription during walking exercise. Thus, it may be more appropriate to prescribe treadmill HIIT based on maximum safe walking speed.17-19,31,33
RPE may also be used in conjunction with measures such as HR, blood pressure, or power output, to ensure that exercise intensity is maintained within tolerable limits.17,25,26 The RPE method allows for a simple and versatile way of measuring an individual’s subjective level of exertion. An RPE of 14 to 16 on the BORG 6 to 20 scale is considered to be “high-intensity.” 10 Given that 62% of individuals with stroke have been found to have silent coronary artery disease and may be prescribed beta-blockers, 51 the RPE allows individuals the option to self-regulate their exercise intensity when HR is not a viable option and when a high-quality symptom-limited exercise test measuring peak HR on the beta-blocker is not available. The RPE can be a universal “exercise regulator” that can be used regardless of environment, 21 with special consideration given to individuals with communication impairment (eg, aphasia) following stroke where the scale may need to be represented in pictorial form. 52 Alternatively, another method to prescribe exercise HR with beta-blocker use is a modified Karvonen equation equal to 0.8 × (maximum heart rate − resting heart rate) + resting heart rate. 53
Time
Within the literature on HIIT in stroke, session durations ranged from 20 to 30 minutes17-19,23,30 but burst to recovery ratios were highly variable amongst studies, ranging from 30 seconds:30 seconds 17 to 4 minutes:3 minutes. 23 Arguably, these studies examining HIIT poststroke enrolled higher functioning participants who had the ability to walk independently with or without assistive devices17-19,25,26,33 and could walk safely on a treadmill.17,18,32,33 For individuals who are not able to walk independently or who may not be able to withstand longer intervals and longer session durations, protocols may require adaptation, particularly at the commencement of the exercise training. 10
Type
Most studies to date of HIIT in stroke have focused on the use of treadmill walking,17-19,23,30 either with19,23 or without 17 body weight support while one study (abstract) of single-session HIIT used a recumbent stepper. 31 While the task-specificity nature of treadmill training has led to improvements in gait and functional outcomes after stroke,17-19,23,30 its application in the literature to date has been limited to individuals who are able to walk without physical assistance from another individual or when the necessary resources, such as body weight support apparatus and trained staff, are available. Supported modalities, such as the recumbent stepper, have been studied less but offer the opportunity to include individuals with a broader range of functional abilities and have been shown to increase CRF. 54 Thus, given the task-specificity of treadmill walking, this modality would be recommended for functional gains in walking ability if safety permits, whereas a seated modality such as recumbent stepper may be more feasible for increasing CRF in people with lower functional abilities that are not able to safely walk on a treadmill.
In summary, while various combinations of the FITT parameters may lead to a range of benefits, individualized protocols are warranted. While the above considerations appear to yield improvements in gait, function, and cardiovascular health, they are based on limited evidence regarding HIIT in the stroke population and a wide range of dosing. Based on the current body of evidence, Table 2 offers considerations for FITT parameters for clinicians to consider when prescribing HIIT poststroke.
Frequency, Intensity, Time, and Type Dosing Within Current Literature.
Potential Mechanisms Underlying Improvements in Functional, Cardiovascular, and Neuroplastic Outcomes With HIIT
While there exists little research to date examining specific mechanisms underlying the benefits of HIIT poststroke, evidence of potential mechanisms from other populations may be extrapolated.
Potential Mechanisms Underlying Improved Functional Outcomes
An increase in neuromuscular recruitment during treadmill training in nondisabled individuals may lead to improved oxidative capacity and efficiency within skeletal muscle. 55 This may also be a potential mechanism by which treadmill HIIT aids in improved functional outcomes following stroke where decreased oxidative capacity in skeletal 56 can be a result of decreased neuromuscular recruitment. 57 Indeed, it has been shown that, after controlling for training frequency and duration, higher treadmill speeds yield more positive functional outcomes after stroke than general rehabilitation exercises alone.18,19 Moreover, the rest periods provided in HIIT allow opportunities for feedback from therapists and mental practice during the training session, which involves a high level of cognitive processing and may lead to enhanced motor practice. 58
Potential Mechanisms Underlying Improved Cardiovascular Health
Studies investigating HIIT versus MICE in individuals with cardiometabolic disease have shown that amelioration of peripheral mitochondrial dysfunction (typically present in many chronic diseases) may play a role in improving CRF. 16 Increased mitochondrial biogenesis, essential to the structural integrity of skeletal muscle, 59 occurs even after a single bout of low-volume HIIT in nondisabled individuals through increased PGC-1α, which is correlated with improved CRF. 13 Additionally, increases of 50% to 60% in maximal rate of Ca2+ uptake into the sarcoplasmic reticulum have been observed in individuals with cardiometabolic disease following HIIT. 60 This may decrease muscle fatigue and improve skeletal muscle function, ultimately leading to improvements in CRF. Repeated bouts of deoxygenation that occur during HIIT compared with MICE may also contribute to adaptations in skeletal muscle oxidative capacity. 16
Centrally, HIIT has been shown to improve cardiac ejection fraction in individuals with heart failure, showing the potential for HIIT to influence left ventricular remodeling. 61 Compared with MICE, HIIT has also lead to greater improvements in stroke volume, mitral annular excursion, ejection velocity and systolic mitral annular velocity in individuals with metabolic syndrome. 16 Arguably, whether these mechanisms apply to the poststroke population is not known. Given that individuals poststroke typically yield a lower absolute power output and a dose-response with intensity and improvements in cardiovascular health in stroke is unknown, we are proposing that similar possible mechanisms may be seen after stroke, but further research is needed.
Shear stress–mediated improvements in endothelial function within the systemic vasculature may also provide insight into improved cerebrovascular function. 62 Animal models show strong evidence of shear stress–mediated improvements in endothelial function within the cerebrovasculature 62 brought on through chronic exercise. In healthy adults, HIIT has been shown to induce acute improvements in systemic flow–mediated vasodilation. 63 As well, nitric oxide bioavailability is greater following HIIT compared with MICE, 64 suggesting that there may be other mechanisms contributing to improved vascular function beyond increases in shear stress stimulus. Arguably, an acute bout of exercise at high-intensity stimulates oxidative stress and transient endothelial dysfunction, but the overall improvements to endothelial function prevail as greater nitric oxide bioavailability is observed a few hours later. 65 It is unclear, however, whether such systemic effects of arterial function can be extrapolated to the cerebrovasculature, and the role of exercise intensity in mediating these effects remains unknown. 66
Potential Mechanisms Underlying Changes in Neuroplasticity
Evidence surrounding whether cardiovascular exercise can have positive effects on cognition shows mixed results. Cardiovascular exercise has been shown to improve cognition in healthy individuals 64 through positive changes in the structure and function of different areas of the brain, including gray matter and hippocampal neurogenesis. 11 However, a recent Cochrane review suggests no evidence that aerobic physical activities that improve cardiorespiratory fitness, have any cognitive benefit in healthy older adults. 67 Cardiovascular exercise also promotes changes within the primary motor cortex (M1), a key target area to improve motor recovery following a stroke. 68 Even a single bout of cardiovascular exercise activates neuroplastic mechanisms responsible for motor learning. 69
Intensity has been shown to be critical in modulating the neuroplastic and motor learning effects of cardiovascular training in healthy individuals. Higher intensities of cardiovascular exercise are required to increase the expression of brain-derived neurotrophic factor and insulin-like growth factor I, neurotrophins that modulate neural repair processes in animal models, 12 leading to neuroplastic and motor recovery benefits.12,70 HIIT-associated increases in corticospinal excitability 38 and brain-derived neurotrophic factor 40 may explain the improvements in motor learning in healthy individuals 38 and retention of motor skills after stroke. 26 An additional key characteristic of HIIT is the sinusoidal high-low flow through the brain and across the cerebrovascular endothelium, which may be a candidate stimulus that optimizes release of vasoactive/growth factors. 71
Overall, while we can extrapolate potential mechanisms underlying HIIT-associated improvements from other populations, further research is warranted to establish mechanisms that may be responsible for the effects of HIIT within the stroke population and to align mechanistic changes with meaningful improvement in cognitive or motor recovery.
HIIT: Clinical Implications and Considerations for Future Direction of HIIT Poststroke
HIIT has shown to be effective at enhancing functional recovery,17,23,30 CRF,17,30 and gait parameters17-19,23,30 within stroke survivors. HIIT has the potential to be integrated into clinical practice to remediate functional impairments following stroke, however, further pragmatic study is warranted to establish the feasibility, safety, and efficacy to guide implementation. HIIT has the potential to contribute to enhanced neuroplasticity and motor recovery poststroke, as higher exercise intensities are required to enhance the expression of neurotrophins that augment neural repair processes.36-38 Training parameters to optimize functional and health benefits still need to be established. Moreover, future research should address the effectiveness of HIIT among individuals with a broader range of functional abilities, along with larger trials to establish the effectiveness of HIIT across all phases of stroke recovery.
The application of HIIT in individuals with stroke has been shown to improve cardiovascular function and motor performance.17-19,23,30 Optimal HIIT parameters, as well as HIIT-associated benefits to neuroplasticity and cerebrovascular function, and for those with lower functional abilities who are unable to walk, remain largely unknown. Larger randomized controlled trials are necessary to establish (
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Canadian Stroke Trials for Optimized Results (CaSTOR), a joint initiative of the Canadian Stroke Consortium and HSF Canadian Partnership for Stroke Recovery. MR was supported with funds from the Reseau Provincial de Recherche en Adaptation-Readaptation (Recherche Clinique), the Canada Foundation for Innovation (John R. Evans Leaders Fund) and the Montreal Centre for Interdisciplinary Research in Rehabilitation (New Investigator Fund). AT was supported by a personnel award from the Heart and Stroke Foundation, Ontario Provincial Office (CS I 7468). DMB is supported by a Royal Society Wolfson Research Merit Award (No. WM170007) and has also received support from the Higher Education Funding Council of Wales. MP is funded by the Canada Research Chairs program. We acknowledge support from the Canada Research Chair Program (to JJE).
