Abstract
Neurorehabilitation relies on core principles of neuroplasticity to activate and engage latent neural connections, promote detour circuits, and reverse impairments. Clinical interventions incorporating these principles have been shown to promote recovery and demote compensation. However, many clinicians struggle to find interventions centered on these principles in our nascent, rapidly growing body of literature. Not to mention the immense pressure from regulatory bodies and organizational balance sheets that further discourage time-intensive recovery-promoting interventions, incentivizing clinicians to prioritize practical constraints over sound clinical decision making. Modern neurorehabilitation practices that result from these pressures favor strategies that encourage compensation over those that promote recovery. To narrow the gap between the busy clinician and the cutting-edge motor recovery literature, we distilled 5 features found in early-phase clinical intervention studies—ones that value the more enduring biological recovery processes over the more immediate compensatory remedies. Filtering emerging literature through this lens and routinely integrating promising research into daily practice can break down practical barriers for effective clinical translation and ultimately promote durable long-term outcomes. This perspective is meant to serve a new generation of mechanistically minded and caring clinicians, students, activists, and research trainees, who are poised to not only advance rehabilitation science, but also erect evidence-based policy changes to accelerate recovery-based stroke care.
Introduction
The central tenet of neurorehabilitation is to promote better function through gradual biological and psychological recovery, returning impaired effectors and neural synapses to their premorbid levels of motor control and connectivity.1-3 However, the unreal demands of our health care regulatory bodies and balance sheets implicitly incentivize a speedier “compensatory” approach, one that focuses on the repurposing of seemingly unaffected effectors and neural synapses to achieve a minimum level of function.4-9,* Although the lure of rapid and obvious functional gains through a compensatory strategy may outweigh the gradual and subtle changes achieved through a recovery process, succumbing to a compensatory approach early in the rehabilitation progression may adversely hinder recovery later and moreover result in irreversible and negative functional outcomes (eg, learned nonuse, paretic limb contractures).10,11 Even though a compensatory approach is certainly essential when recovery is less likely and may serve as an intermediate step toward recovery in the early stages of rehabilitation, 12 it should neither be the gold standard nor the first line of defense, especially in the acute periods after stroke.
Clinical research provides guidance on how neurorehabilitation could harness the biological and psychological processes of recovery. Systematic reviews, clinical practice guidelines, and large-scale multisite phase III randomized controlled trials (RCTs) enable well-powered statistical inferences, readily digestible even for the novice clinician. These larger-scale intervention studies are nonetheless difficult to come by, demanding years of toil from conceptualization to publication. Moreover, many of these multisite trials have yielded null results, proving no better than standard care.13,† Given the slow and often disappointing results from phase III RCTs, where should the clinician look for insights to continuously improve their neurorehabilitation practice?
Answer: early-phase clinical intervention studies. Robust clinical insights are not only present in systematic reviews, clinical practice guidelines, and large-scale multisite RCTs, but also found in early-phase clinical studies preceding large-scale RCTs.14,15,‡ Even though this literature may be relatively underpowered, early-phase clinical research that survives rigorous peer review gives rise to some of the most novel, insightful, and impactful ideas, holding the most promise to improve the lives of our patients today.
The dialogue on these frontiers between researchers and clinicians is nonetheless fraught with confusion. One primary source of confusion originates linguistically, with early-phase clinical studies riddled with (necessary) lingo, embedded in a scientific writing format demanding experience to efficiently parse.16,17 To encourage more effective dialogue, we distilled 5 features found in rigorous, early-phase, recovery-promoting neurorehabilitation intervention studies to provide busy clinicians with roadmaps to navigate the lingo and efficiently extract simple insights from dense articles (Table 1). A keen eye for these 5 features may not only sharpen the busy clinicians’ ability to efficiently uncover findings worthy of translation, but also inspire the researchers’ ability to design better interventions for motor recovery.
Summary of the 5 Features and Guiding Questions.
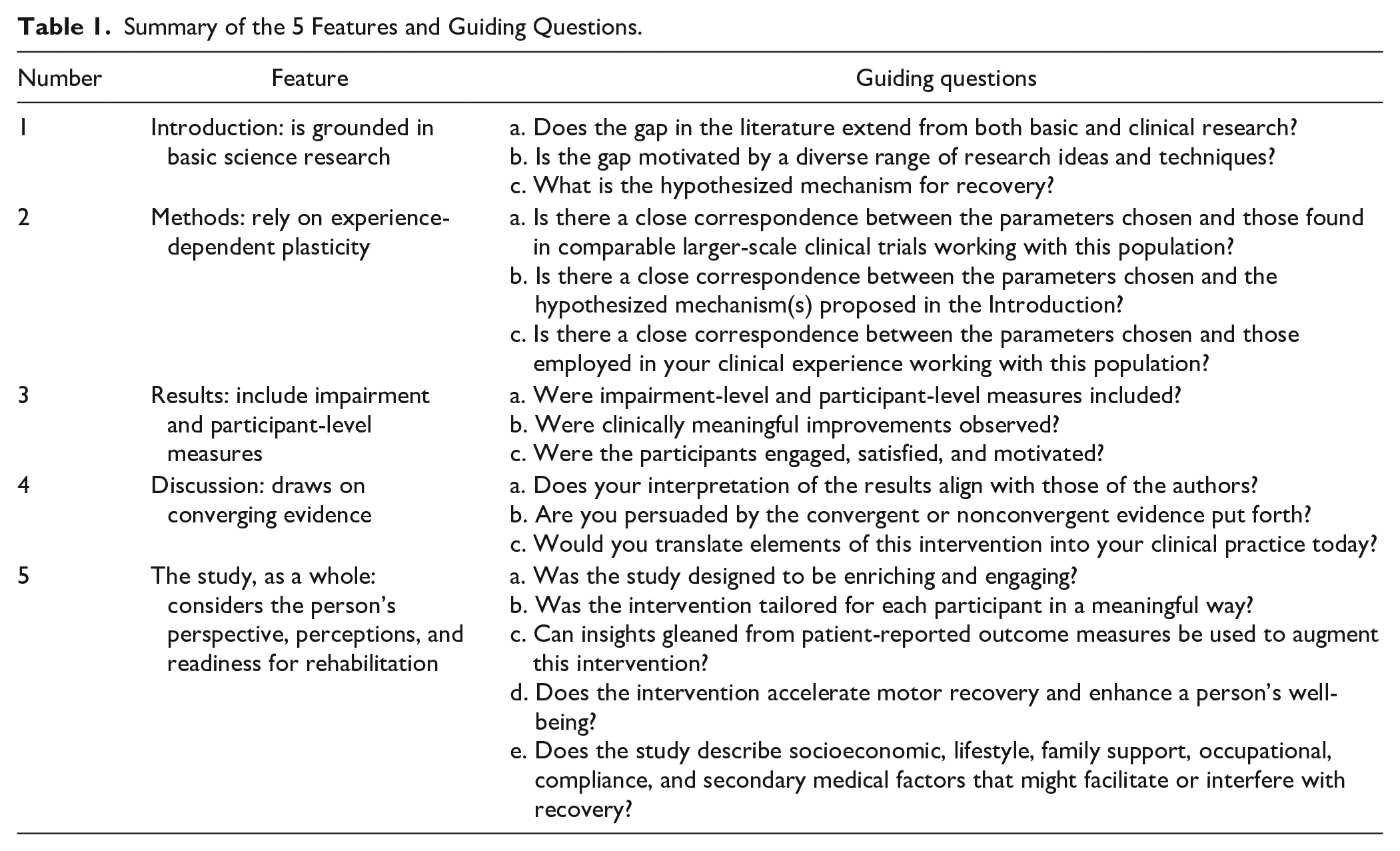
Feature 1: the Introduction Is Grounded in Basic Science Research
Promising clinical research finds its roots in basic science.18-20 With insights generated from behavioral neuroscience with animal models21-26 to cognitive neuroscience with human participants,21-23 basic science researchers can guide the direction of clinical research. Although the clinical application of these studies may not be immediately apparent, the history of science, in particular the fields of motor control and motor learning, have provided countless examples of ideas that have been successfully translated from the bench to the bedside.24-29 For instance, the recovery-promoting effects of rehabilitation in an enriched environment were first characterized in animal models,30,31 which subsequently springboarded the design of human intervention studies. 32
Whether an early-phase clinical intervention is sufficiently motivated by basic science research can be assessed by reading the Introduction and supportive citations. Researchers often use the Introduction to identify the core “knowns” and gaps in the literature, providing a rationale behind why these gaps need to be filled. A mechanism33,§ is often put forth to motivate why an intervention could be effective. While reading these introductory sections, ask yourself whether these gaps naturally extend from both basic and clinical research and, moreover, whether these gaps are motivated by a confluence of research ideas and techniques (eg, systems neuroscience in animals, cognitive neuroscience in humans). Then, ask yourself whether the hypothesized mechanism logically extends previous research. Studies well-positioned at the intersection of basic and clinical research domains are likely those that assert well-reasoned hypotheses relevant to neurorehabilitation.
Feature 2: the Methods Rely on Experience-Dependent Plasticity
It is indisputable that repetition, timing, and challenge are vital ingredients to improve brain reorganization and functional outcomes. 34 Repetition instantiates skill within the neural circuitry by increasing the number of synapses, 35 altering synaptic weights,36-38 and pruning unnecessary synapses. 39 A well-timed rehabilitation during sensitive periods, typically within the first 3 months poststroke, accelerates recovery.18,40,41 The amount of challenge, titrated by the amount of problem-solving required for task success, has also been shown to promote engagement as well as enhance subsequent motor recovery and motor learning.42-47 A study does not need to include all these elements of experience-dependent plasticity to make it impactful, but it should be philosophically grounded in these ideas.
Whether an early-phase clinical intervention is highly dosed, properly timed, and sufficiently challenging should be evident in the Methods. Ask yourself whether the therapeutic parameters outlined in this section are clearly detailed and show a close correspondence with the hypothesized mechanisms proposed in the Introduction. Furthermore, ask yourself whether the therapeutic parameters chosen are consistent with related research involving a similar demographic cohort (eg, stroke chronicity)48-51,‖ and whether these parameters jive with those proven efficacious in your own clinical practice.
Feature 3: the Results Include Impairment and Participant-Level Measures
Early-phase clinical studies often underscore improvements on activity-level measures (eg, dressing, eating). However, improvements on impairment-level measures (eg, individual joint control, coordination, strength, dexterity, movement quality, visuospatial reasoning, short-term memory, attention) should not be neglected because changes in these measures closely reflect the trajectory of cognitive and motor recovery (especially in the acute stage after stroke). For instance, improvements on the Fugl-Meyer Motor Assessment and Scandinavian Stroke Scale subscores, 2 motor impairment–level measures, 52 are intimately linked to the time frame of neural repair.53,54 Importantly, participant-level measures should not be forgotten; these measures gauge an intervention’s impact on the individual—a critical ingredient for longer-lasting therapeutic and neurophysiological changes (see Feature 5).55-57
Whether a study includes a comprehensive battery of outcome measures should be evident in the Results. While reading this section, ask yourself whether the main findings encompassed impairment-level measures and, if so, whether any clinically meaningful improvements were observed. 57 Furthermore, ask yourself whether the main findings included participant-level outcomes (eg, quality-of-life questionnaires, poststudy satisfaction surveys). If so, did participants find the intervention engaging and meaningful?
Feature 4: the Discussion Draws on Converging Evidence
Well-established clinical knowledge is supported by convergent lines of evidence. Different methods (eg, quantitative, qualitative), models (eg, animal, human), and levels of evidence (eg, case studies, systematic reviews, meta-analyses) should coalesce to support the study’s conclusions. For instance, constraint-induced movement therapy (CIMT), an intervention that involves functionally oriented task practice of the paretic upper extremity along with restraint of the less-impaired upper extremity, has efficaciously minimized many deleterious effects of learned nonuse in humans58,59 and in deafferented monkeys. 43 In addition, the signature CIMT protocol or a modified version has been applied during the acute, 49 subacute, 60 and chronic 58 periods after stroke. Evidence in favor of CIMT ranges from a very early case study 61 to phase II 49 and phase III RCTs, 58 altogether forming an enriching but nonetheless controversial scientific discourse.20,62-66,¶ Although it is not a perfect form of therapy, the convergence of evidence to date suggests that some but not all of the components of CIMT are ripe for clinical translation.64,67
Whether a study’s findings are supported by converging lines of research should be evident in the Discussion, a place where the authors put forth their interpretation of the results. Ask yourself whether your interpretation jives with that of the authors? If there was a discrepancy, was it resolved with reference to supporting evidence from a diverse range of research domains (eg, brain imaging, animal work, observational studies) and multiple levels of verification (eg, case studies, systematic reviews)? Ultimately, a well-written Discussion should help you situate the study’s findings into a broader scientific context and with strong converging evidence (or a strong rationale for nonconverging evidence) may inspire you to translate these insights into your clinical practice.
Feature 5: the Study, as a Whole, Considers the Person’s Perspective, Perceptions, and Readiness for Rehabilitation
An intervention centered on principles of experience-dependent plasticity may promote recovery; however, its effectiveness may rely on the participant’s level of engagement and motivation for self-practice.68,69 Meaningful and individually tailored interventions, via goal setting sessions and simulations of real-life scenarios, 70 may promote longer-lasting therapeutic improvements and sustained neurophysiological changes. 50 Moreover, interventions that account for the person’s preferences, lifestyle, occupation, family support system, socioeconomic milieu, and personal well-being may further augment motor performance and learning.50,51,71-74 Although interventions that are meaningful and engaging have achieved similar, 71 if not better, transfer of motor ability from the clinic to real-life settings, 75 how these interventions can be used to reverse motor impairments beyond the outcomes achieved by conventional therapy remains to be seen.76,#
Although recovery in neurorehabilitation is complex and multifaceted, this process may be perfected through genuine collaboration between the person and the clinician-scientist.77,78 This collaborative spirit may be overtly hardwired into the study’s design, like for instance, with each participant receiving an individually tailored intervention plan in the participant’s home setting. This collaborative spirit may also be subtle, for instance, with the inclusion of satisfaction surveys that periodically solicit the participant’s opinion, level of interest, effort, and engagement in the intervention. Whether an intervention is designed to be collaborative may permeate through various sections of the article: As you read the Introduction, ask yourself whether the study’s intervention was designed to engage the participant in an enriching environment (Feature 1). As you read the Methods, ask yourself whether the study is designed to challenge the participant in a meaningful and individually tailored manner (Features 2 and 5)? When you read the Results, ask yourself whether important insights can be gleaned from patient-reported outcome measures or person-specific information (eg, socioeconomic, lifestyle, family support, occupational, compliance, secondary medical factors) that might facilitate or interfere with the intervention’s effectiveness for future participants (Feature 3). Finally, as you read the Discussion, ask yourself whether the converging evidence indicates that the intervention can be applied to not only accelerate motor recovery, but also enhance a person’s well-being (Feature 4).79,80
Conclusion
To narrow the gap between the busy clinician’s working knowledge and the cutting-edge motor recovery literature, we have distilled 5 features of early-phase neurorehabilitation research centered on recovery-based interventions. We submit these aspirational features to a new generation of mechanistically minded, caring clinicians who strive to advance their clinical practice as soon as the most promising evidence becomes available.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
