Abstract
Background
Transcranial direct current stimulation (tDCS) can be used to improve post-stroke aphasia. However, given the mixed evidence for its efficacy, individual differences may moderate the relative benefit of this strategy. In planned exploratory subgroup analyses, we examined whether age, education, sex, brain-derived neurotrophic factor status, and baseline performance individually impacted improvement in picture naming between baseline and 1 week after the end of the therapy, then whether the combination of factors that predicted recovery of naming and discourse differed for those who received concurrent tDCS.
Objective
Examine whether individual differences influenced the effect of tDCS on language recovery.
Methods
In this randomized, double-blind, sham-controlled, efficacy study of tDCS combined with language therapy for subacute post-stroke aphasia, patients completed an evaluation including the Philadelphia Naming Test and Cookie Theft picture description, which was analyzed for Content Units (CU) and Syllables/CU. Individual factors were examined using linear models including the interaction between treatment group and subgroup.
Results
Significant interactions were observed between tDCS group and both age and education. The predictors of a positive response to tDCS differed from the predictors of a positive response to language treatment alone. While baseline performance was an important predictor of future performance regardless of treatment group, responses to treatment without tDCS were influenced by age whereas responses to treatment with tDCS were not.
Conclusions
Age and education influence the efficacy of different treatment strategies. Refinement of treatment selection is important to the overall individualization and optimization of post-stroke patient care.
Trial Registration:
ClinicalTrials.gov NCT02674490.
Introduction
There is considerable interest in improving the efficiency and precision of language services provided when a person experiences aphasia after left-hemisphere stroke. Transcranial direct current stimulation (tDCS) refers to the safe, non-invasive, non-painful electrical stimulation of the brain, which modulates cortical excitability by applying weak electrical currents in the form of direct current brain polarization. It is a promising adjunct to traditional therapy1 -5 when applied to left hemisphere language areas to increase cortical excitability (reduce activation threshold) in patients with post-stroke aphasia.6 -8 However, given the mixed landscape of evidence for the efficacy of tDCS 2 and the desire to maximize the precision of care received by all patients, it is important to determine the extent to which individual differences may moderate the relative benefit of this treatment strategy.
Recently, the Stimulating Language in Subacute StrokE (SLISSE) trial was conducted to determine if tDCS augmented computer-delivered lexical treatment (CoDeLT) for improving language in subacute stroke. On the whole, tDCS did not significantly augment CoDeLT in the treatment of the primary outcome measure, naming untrained nouns on the Philadelphia Naming Test (PNT). Though trends were present, there was considerable inter-patient variability. However, tDCS did augment the more ecologically valid secondary measure of how much information patients provided and how efficiently they did so when asked to describe a picture. 9 A pre-specified secondary analysis of these data was to examine whether individual differences in age, sex, education, time since stroke onset, and genotype influenced the effect of tDCS on language recovery. Age-related decline in cortical thickness is thought to widen the distance between the scalp and cortex, thus reducing the focal specificity of current when tDCS is applied.10 -13 We also wanted to examine whether sex differences reported previously, in which younger men received less benefit of tDCS than older women (due to differences in current),10,14,15 were observed in our sample. Prior work has demonstrated that individuals with the val/val genotype of the brain-derived neurotrophic factor (BDNF) gene are more likely to benefit from A-tDCS during aphasia treatment in the chronic phase, 16 but it was important to determine whether this difference was observed in a subacute sample as well.
First, we examined whether each factor (age, sex, education, time post onset, and BDNF status) impacted the primary outcome, change in picture naming on the PNT (untrained items) between baseline and 1 week after the end of the naming therapy, by treatment group (tDCS vs sham). Next, we examined whether the combination of factors that were identified as having a significant impact on change in PNT in the first analysis was similar for the 2 treatment groups when used to predict improvements in naming and discourse performance, once controlling for differences in performance at baseline. Regression equations were calculated in order to examine the value of each individual factor when predicting improvement across each outcome short- and long-term.
Methods
Study Design
SLISSE was a randomized, single-center, double-blind, sham-controlled, efficacy study of A-tDCS combined with a computer-delivered speech and language therapy (SALT) for subacute stroke-induced aphasia completed at Johns Hopkins Hospital and Johns Hopkins Bayview Medical Center in Baltimore, Maryland, previously reported on in Stockbridge et al. 9 All procedures were approved by the Johns Hopkins Medicine Institutional Review Board (IRB00089018). The study was registered with ClinicalTrials.gov (NCT02674490).
Patients
Screened patients were right-handed, adult English speakers within 3 months of acute ischemic left hemisphere stroke who were diagnosed with aphasia using the Western Aphasia Battery-Revised (WAB-R).
17
Individuals with previous neurological or psychiatric disease, seizures, brain surgery, metal in the head, uncorrected visual or hearing loss, scalp sensitivity, medications that lower the seizure threshold, or N-methyl-
Evaluation
After providing informed consent, patients completed a baseline evaluation.
Baseline evaluation included the NIH Stroke Scale 19 (a measure of overall stroke severity), including the description of the Cookie Theft picture, which was analyzed for Content Units (CU) and Syllables/CU.20,21 CU are conceptual targets mentioned by healthy controls when describing the picture and provide a sensitive measure of the content of communication.21,22 Syllables/CU is measured by the total number of syllables (including nonwords such as “uh” and perseverations) in the description, divided by CU, such that lower syllable/CU indicates more efficient discourse. Where no CU were present in the sample, syllables were divided by 0.1.
Patients also received the WAB-R, color PNT 23 (untrained items), and a set of 80 trained items. In order to arrive at stable measures of naming that demonstrated high fidelity, the PNT and 80 trained items were administered twice on consecutive days for each timepoint. The task was recorded and scored remotely by individuals blinded to the patient’s clinical profile, and then scores on each administration were averaged to reduce variability. Reliability of PNT scoring was examined using intraclass correlation coefficients with a 2-way mixed model for absolute agreement. 24 Inter-rater agreement (0.79-0.94) and intra-rater agreement (0.89-0.99) were good-excellent. We also administered the Stroke Impact Scale 25 (SIS), a disease-specific quality of life questionnaire. 9
All participants with no contraindication were asked to complete structural and functional magnetic resonance imaging (fMRI) to support localization of the lesion and identification of a stimulation site of healthy tissue. If they consented to research MRI, they were randomized to (1) have the site of stimulation based on greatest area of activation on fMRI during a naming task (as in Fridriksson et al 26 ) or (2) have the site of stimulation based on the area of lesion (from clinical imaging or research MRI). If randomized to lesion-based localization (or declined research MRI), a site of undamaged cortex was stimulated: the left inferior frontal gyrus (L IFG) if no lesion in L IFG, left superior temporal gyrus (L STG) if no lesion in STG, or left prefrontal cortex (L PFC) if lesions in both L IFG and L STG), based on the clinical or research MRI. Most participants declined the research MRI, so analysis of fMRI-determined stimulation site versus lesion-based stimulation site was grossly under-powered and will not be further discussed. Likewise, there was insufficient power to compare the effects of tDCS at different stimulation sites (left STG, IFG, or prefrontal).
Stimulation location amperage were chosen to mirror a previously published positive randomized, sham-controlled trial of A-tDCS (vs sham) plus the same CoDeLT, in chronic post-stroke aphasia. To evaluate the effect of time post stroke (subacute vs chronic) on tDCS effects, we retained the same primary outcome measure, hemisphere of stimulation, and type of stimulation (1 mA A-tDCS). 26 These selections also are consistent with existing literature in healthy individuals and people with aphasia. In healthy individuals, excitatory A-tDCS administered to left perisylvian brain areas improves language processing27 -29 Prior studies have focused more on stimulating language processing centers than on ensuring the underlying tissue being stimulated is, in fact, preserved.6,7 In the only prior sham-controlled trial of language therapy augmented by tDCS in acute-subacute aphasia, 7 patients received 2 mA tDCS over STG with a modest result. However, 4 of 7 patients had lesions of the left STG. This motivated the decision to ensure all stimulation targets were in preserved tissue, even though this necessitated having different stimulation sites from patient to patient. Among designs, 1 mA was the lowest amperage associated with positive effects and was chosen to minimize safety concerns associated with the acute-subacute period.
About 1, 5, and 20 weeks after the final CoDeLT session, patients were re-evaluated on the PNT, trained items, Cookie Theft picture description, and SIS, as in the baseline evaluation.
Treatment
Patients completed 15 45-minute sessions of CoDeLT, identical to the treatment used in Fridriksson et al 26 with a speech-language pathologist over 3 to 5 weeks (ie, 3-5 times a week depending on the individual’s schedule availability). Patients were not prohibited from receiving standard of care rehabilitation treatment for stroke-related deficits, nor were they limited in the prescription of any standard of care pharmacological interventions identified as appropriate by their primary care doctor, except as dictated by the exclusion criteria. During CoDeLT, the anode was placed at the stimulation site under a tight-fitting wrap. The reference cathode electrode was placed on the participant’s right orbito-frontal scalp. For individuals in the active A-tDCS condition, 1 mA of A-tDCS stimulation was induced between 2 5 cm × 5 cm saline-soaked sponges over the targeted cortical region. Ramping up of the current to 1 mA occurred over 10 to 15 seconds to allow participants to habituate to the tingling sensation. A-tDCS stimulation was active only in the first 20 minutes of the 45-minute treatment session. In order to promote blindness to the treatment condition for both the clinician and the patient, the sham tDCS consisted of 30 seconds of 1 mA stimulation which is faded to 0, mimicking the sensation of continuous (eg, 20 minutes) 1 mA stimulation, which is generally perceived for only the initial 20 to 30 seconds.
During CoDeLT, patients were shown an on-screen picture of a target (eg, mop) for 2 seconds then viewed a video of a female speaker saying a word that was either the name of the target (eg, “mop”) or a semantic foil (“broom”), phonological foil (“top”), or unrelated foil (“bridge”). Patients were asked to identify whether the picture and the audio/video presentation of the word matched, using large red “no match” and green “match” response buttons. Half of the picture/word pairs matched. The computer provided immediate visual feedback following a response in the form of a “smiley face” for correct answers and a “frowny face” for incorrect answers, and patients received a final accuracy score for the session.
Statistical Analysis
Two planned exploratory subgroup analyses were conducted on the sample of study completers to follow up with the primary outcome findings. 9 The first analysis examined the influence of age (<55 vs ≥55 years), sex, education (<11 years completed vs ≥12 years), and time since stroke onset to initiation of CoDeLT (<1 month vs ≥1 month), and BDNF status on response to CoDeLT by treatment group (collapsing across stimulation sites that targeted healthy tissue). The software package SAS was used. Given that these were exploratory analyses, it was planned that variables would be binned rather than treated as continuous to improve the predictive model by reducing noise. A linear regression of change in naming accuracy, which included main effects and interaction terms with treatment group for these characteristics was used, adjusted for baseline WAB-R AQ, aphasia type (determined by WAB-R performance), and age (except for when examining age). Given the small sample size and since this is a secondary analysis, a significance level of .10 was used to retain main effects or interaction with treatment in the final reported model.
The second analysis was conducted using IBM SPSS version 29 to determine whether the factors predicting response to treatment that included tDCS differed from the factors predicting response to treatment without tDCS when controlling for baseline performance. Individual factors that demonstrated significant interactions with change in PNT between baseline and 1 week after CoDeLT in the first analysis were retained for the second analysis; those that did not demonstrate significant interactions were abandoned. The retained variables were entered together (not stepwise) into a linear regression to predict change in naming. Each linear regression was repeated using change in naming at 5 and 20 weeks after CoDeLT, and using CU and syllables per CU, at 1-, 5-, and 20-weeks after CoDeLT as the dependent variable (outcome measure). Time points were considered independently to account for potential differences in variability of recovery and treatment effects across the subacute to chronic period. The 9 regressions (3 outcomes × 3 timepoints) were conducted first ignoring treatment condition, then considering each treatment condition separately. A significance level of .05 was used to examine each model. Significant independent predictors also were examined in an exploratory manner.
Results
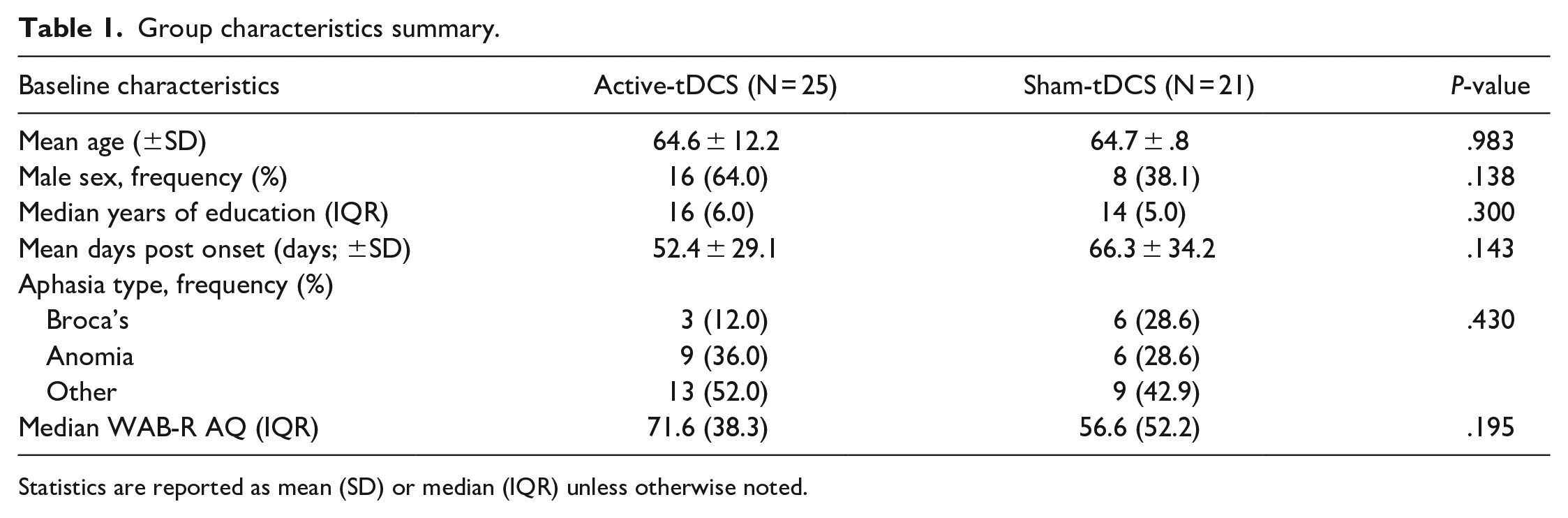
As previously reported, of the 92 patients screened, 58 (63%) were randomized between September 16, 2016, and October 4, 2021 (with the final follow-up evaluation completed March 29, 2022). Thirty patients were randomized to receive active tDCS, and 28 were randomized to receive sham tDCS. Twenty-six patients in the active tDCS group and 25 patients in the sham tDCS group received at least 1 CoDeLT session. Overall, 46 subjects (79%) completed 15 CoDeLT sessions, 4 (7%) were lost to follow-up, 2 (3%) withdrew consent, and 1 (1.7%) subject died. These 46 individuals (25 individuals who received active-tDCS and 21 who received the sham treatment) were included in analysis. Baseline characteristics were similar between the 2 treatment groups (Table 1).
Group characteristics summary.
Statistics are reported as mean (SD) or median (IQR) unless otherwise noted.
Influences of Individual Differences on tDCS Efficacy
Two modifications were made to the a priori statistical approach in response to the sample contents. First, we had intended to bin education with a break point at high school completion (ie, 12 years of education). However, there were only 2 patients with less than 12 years of education, so the subgroup analysis considered education level of 16 years as the cut point (<16 years completed vs. ≥16 years). Second, in the BDNF analysis, only 4 individuals in the sample had the atypical val/met allele polymorphism associated with lower BDNF production; the remainder had the typical val/val polymorphism. The small number with the atypical polymorphism was deemed too small of a sample to examine the effects of BDNF polymorphism. Thus, this variable was dropped.
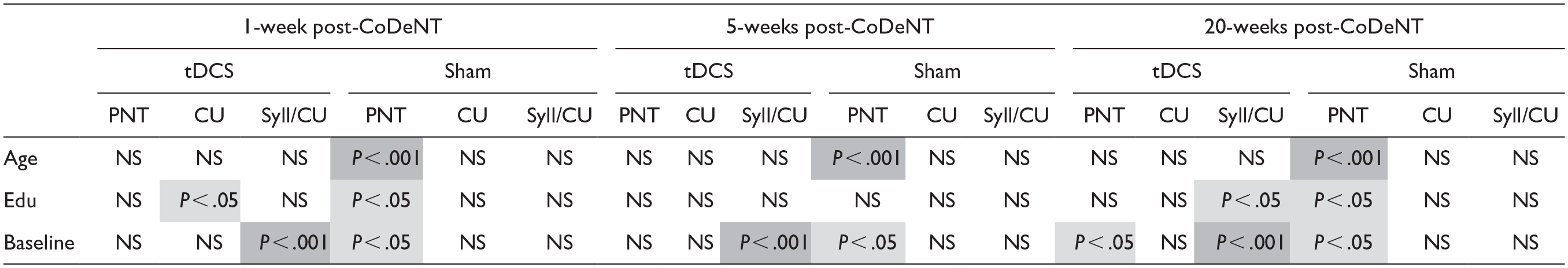
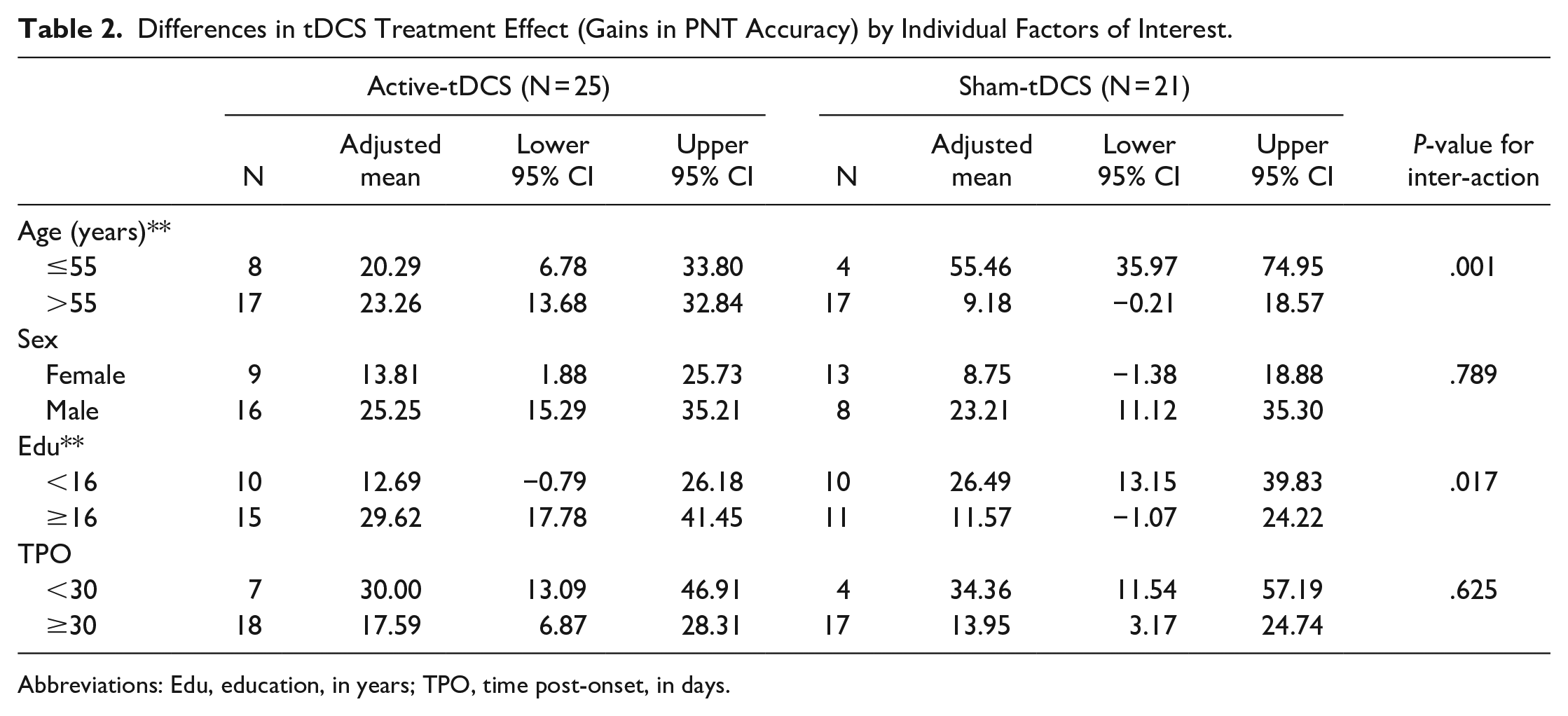
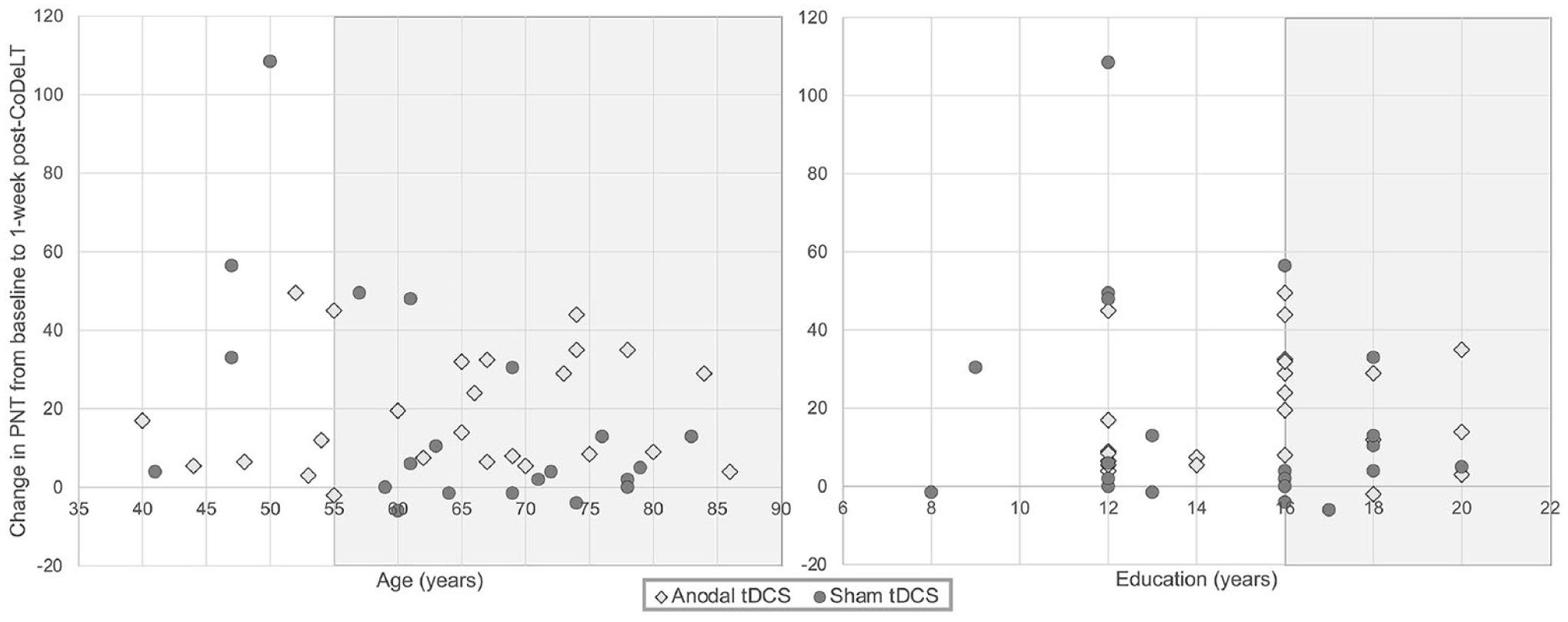
Subgroup analyses of the change in PNT scores between baseline and 1 week after CoDeLT are shown in Table 2. Among these individuals, there was a significant interaction between age group and treatment group. Younger patients (≤55 years old) who received only language therapy showed far more improvement than older individuals in that condition. However, among those who received tDCS in addition to language therapy, the effect of age on naming improvement was negligible. Older patients who received tDCS during CoDeLT showed greater improvement than older patients who had sham during CoDeLT (see Figure 1).
Differences in tDCS Treatment Effect (Gains in PNT Accuracy) by Individual Factors of Interest.
Abbreviations: Edu, education, in years; TPO, time post-onset, in days.
Relationship between change in naming accuracy between baseline and 1-week post-CoDeLT and key variables of interest.
Those who completed college or graduate education who received tDCS showed a greater improvement in PNT scores than those who received sham. In contrast, those with less than a college education who received tDCS showed a smaller mean improvement than those who received sham.
Neither sex nor time since stroke onset within the acute to subacute range had a significant impact on the efficacy of tDCS for the improvement in naming scores, though relatively few patients were recruited within the first month. Thus, as age and education were associated with significant interactions, these were carried forward into the second analysis.
Upon further examination, an individual in the sham condition with a change of over 100 points on the picture naming test appeared to be an influential outlier. When this individual was removed, the significant effects observed remained the same. There remained an interaction effect between age and condition, in which individuals who did not receive tDCS showed a considerable benefit of younger age while those who received tDCS did not (P = .032). There also was a significant interaction between treatment and education (greater response to treatment in those with at least a college education) that remained significant (P = .036). Effects of time since stroke and gender remained non-significant. Given this, the patient was retained in regression analyses.
Combined Factor Influences on tDCS Efficacy
Age and education were included in the series of linear regressions predicting outcomes in naming, discourse content, and discourse efficiency when controlling for baseline performance (Tables 3-5). Across treatment groups, models significantly predicted long-term improvement on the PNT (5 and 20 weeks after CoDeNT), and improved informational efficiency at all timepoints, but not changes in content (Table 3). However, the predictive ability of the models differed considerably when groups were divided by treatment group. Among those who received tDCS (Table 4), long-term PNT improvement (20 weeks), short-term improvement in content, and improvement in informational efficiency at all timepoints were significantly predicted by patients’ age, education, and baseline performance (similar to the findings across patients overall). However, among those in the sham group (Table 5), changes in informational efficiency were not significantly predicted by age and education. However, age, education, and baseline performance significantly predicted change in PNT across all timepoints. Examining significant independent predictors provided additional insight into the relationships among the variables. Across predictive regressions, the most common significant predictor of outcome performance was baseline performance. Uniquely, for those who received tDCS, greater educational attainment was associated with recovery toward greater content in the short-term and more syllables per CU (ie, less informationally efficient, more verbose communication) in the longer-term (20 weeks after CoDeLT). In those who received only CoDeLT without tDCS, younger age predicted better naming outcomes short- and long-term. Education also was associated with naming outcomes. Other relationships between the variables were not significant.
Individual Factors That Predicted Outcome Measures Regardless of Treatment.
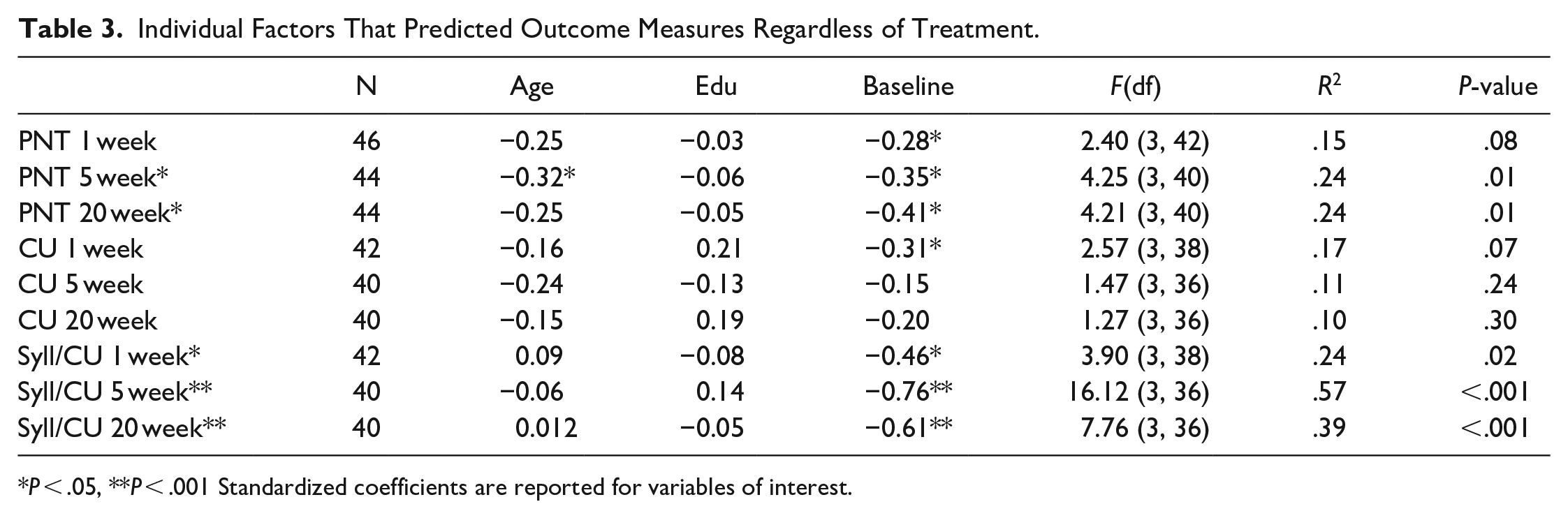
P < .05, **P < .001 Standardized coefficients are reported for variables of interest.
Individual Factors That Predicted Outcomes in Active tDCS Group.
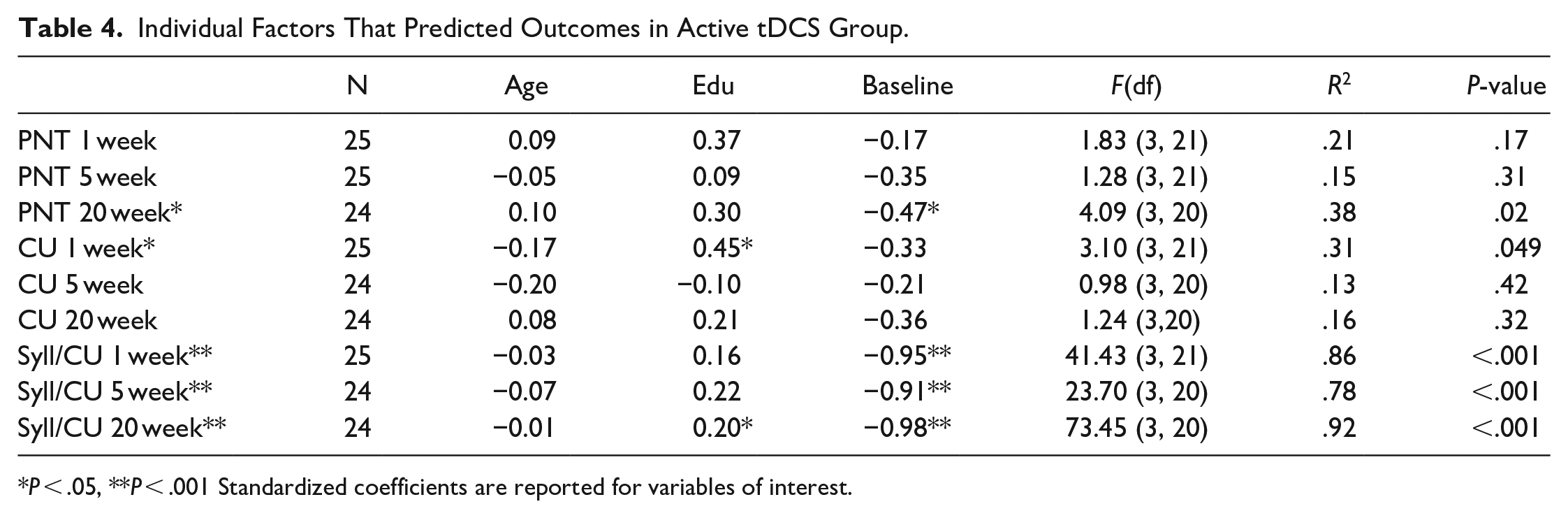
P < .05, **P < .001 Standardized coefficients are reported for variables of interest.
Individual Factors That Predicted Outcomes in the Sham Group (SALT Alone).
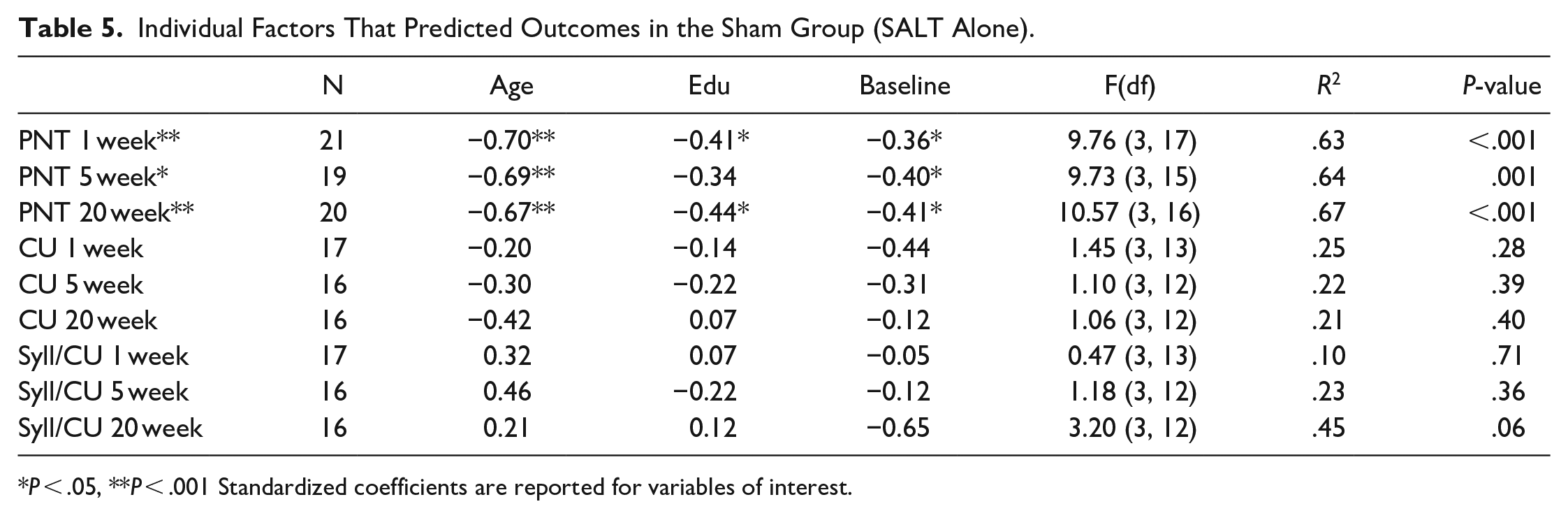
P < .05, **P < .001 Standardized coefficients are reported for variables of interest.
Discussion
This study examined whether a patient’s age, sex, education, and time-post stroke within the acute to subacute window of recovery were associated with differences in the observed benefit from anodal-tDCS coupled with CoDeLT. We examined 3 outcome variables: picture naming on the PNT, and both the quantity of content and efficiency with which content was conveyed in a sample of discourse describing a picture. First, we examined the relationship between the factors individually and the primary outcome measure, change in naming accuracy on the PNT. Then we determined whether combinations of factors were contributing differently to predict the extent of recovery both in naming and discourse in the 2 treatment groups (summarized in Table 6).
Summary of Significant Predictors From Treatment Group Regressions.
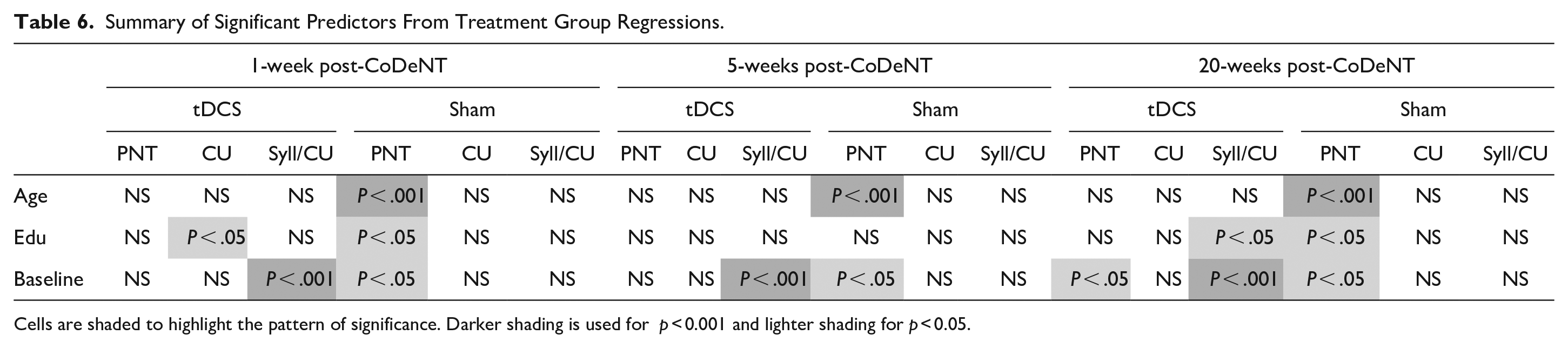
Cells are shaded to highlight the pattern of significance. Darker shading is used for p < 0.001 and lighter shading for p < 0.05.
Age was associated with significant differences in response to tDCS as measured on the PNT. Younger people experienced less benefit from tDCS relative to CoDeLT alone. However, older people (>55 years) who received tDCS experienced much greater average improvement in naming scores on the PNT than older people in the sham group (gains of 23.3 vs 9.2 items). In other domains, such as face-name association in healthy adults, the opposite differential effect of age has been reported; that is, younger people but not older people showed a response to tDCS. 30
Education also was an important factor mediating the relative benefit of tDCS on picture naming. Post-baccalaureate education was associated with more than double the improvement among those who received tDCS. However, among those who received CoDeLT alone (sham-tDCS), a trend toward the reverse was noted; those with fewer years of formal education appeared to recover better. Although it was not anticipated, it is not a wholly unique finding. Recent work has found that anodal tDCS selectively improved working memory in healthy aging adults 31 and episodic memory in cognitively impaired adults (with Alzheimer’s dementia) 32 who had completed a greater number of years of education. Conversely, less educated groups saw no added benefit of its application. To our knowledge, this is the first study to demonstrate this interaction effect of tDCS and education on picture naming in adults with post-stroke aphasia. While there is no clear mechanistic explanation for this phenomenon, it is possible that differences in tDCS efficacy related to education may reflect differing use of compensatory strategies and the extent to which those strategies activate perilesional structures relevant to the task. 31
Other factors were not associated with consistent differences in response to tDCS. Individuals in both groups improved more if they were within the first month (acute to subacute period) than if they began the study at a later point in their recovery, but there was no interaction between this and treatment condition. Likewise, males appeared to recover better, but there was no interaction between this and treatment condition.
In the second analysis, age and education were examined as potentially useful predictors of improvement from treatment when controlling for baseline performance both in the primary outcome measure, which directly related to the CoDeLT therapy task, and when considering generalized improvements to discourse. Age, education, and baseline performance predicted long-term outcomes in naming performance (5 and 20 weeks) and long-term outcome in discourse efficiency across groups (Table 3). When those who received tDCS were considered independently (Table 4), the pattern of outcomes for which predictions from age, education, and baseline ability were significant was very similar to the group as a whole (only PNT change measured at 5 weeks was no longer significant, while content measured 1 week after therapy was). Even where significant, the model predicting long-term improvement in PNT explained a modest 38% of the variance (non-significant models performed even more poorly). While prediction of improvement in informational efficiency was very good in those who received tDCS (78%-92% of the variance explained), this was driven nearly exclusively by the predictive influence of baseline performance. However, in the sham group (Table 5), age, education, and baseline performance together only significantly predicted improvements in naming, the task most closely capturing the skill targeted in CoDeLT. These factors did not predict improvement in discourse measures (ie, models predicting discourse outcomes were not significant).
Also informative was the pattern of significant independent predictors. Age was not a significant predictor of outcomes in any model among those who received tDCS but was a strong independent predictor of improvement in naming among those who did not receive tDCS. These findings suggest the introduction of neurostimulation may have compensated for the otherwise considerable negative effect of age in the context of plasticity-dependent naming recovery. In the standard behavioral therapy context, youth and, to a lesser extent, education influence the relationship between a patient’s baseline and their outcomes (together explaining between 63% and 67% of the variance). However, when tDCS is utilized, this is no longer the case.
The evidence for independent effects of education were sparse relative to age, perhaps because in some ways education is a more imprecise measurement of what may be a more important ground truth influencing therapy outcomes, such as baseline cognitive capacity, complex reasoning, or adult language use. Although there were some models in which education was a significant independent predictor, there was no clear pattern to these instances. This result is consistent with previous studies and meta-analyses, reporting that education is not a robust predictor of recovery or treatment response.33 -36
Recent systematic reviews have found that tDCS is a beneficial adjunct to language therapy only in certain instances.2,4,13,37 Prior investigations have focused on the differential effects of stimulation location, type of language therapy, and dosage. The effectiveness of this adjunct also appears impacted by neuroanatomical morphology, which varies by age and sex. Since tDCS modulates membrane potentials leading to changes in the neuronal firing rate, 38 factors like cortical folding, skull thickness and composition, distance between the scalp and cortex, cortical thickness, and brain volume interact to moderate the conduction current density measurable in a given targeted region. 13 Individuals who receive a greater degree of current density in the targeted regions appear to have greater benefits of tDCS. 39 Thus, for example, areas with lesser volume 40 and thinner skulls,11,41 are more susceptible to functional changes. Meanwhile, greater white matter integrity in language pathways and lesser integrity in dementia-related pathways also have been associated with greater effects of tDCS on cognitive linguistic change. These changes were associated with decreases in long-distance functional connectivity between the targeted site and the areas associated with language retrieval. 42
Across analyses, when considering the treatment groups separately, an important limitation was the small sample size. Results seen here should be interpreted conservatively and would benefit from replication in a larger sample. A second limitation is the potential variability of the stimulation montage. Although 1 mA was applied to all participants who were in the active tDCS condition, it is possible that not all participants experienced this equally due to the anatomical factors discussed above. However, there is evidence from simultaneous fMRI and tDCS that stimulation of any component of the network activated by a task (in this case, the lexical task) will affect the activation of the entire network or its undamaged parts. 43 Given that all 3 of our stimulation sites are in areas typically activated in lexical processing, we propose that stimulation of the 3 sites had essentially the same effect on the language network, to the extent allowable by the participant’s lesion. As with any trial, there is a possibility that unmeasured external influences, such as differences in standard of care treatment patients received or differences in spontaneous recovery, could have unexpectedly influenced the results, though there is not an a priori reason to anticipate that such influences would have differentially affected the tDCS or sham treatment group. Future work may wish to consider not only the broader context in which tDCS is administered (eg, patient access to standard of care treatment), but also the potentially differential effects of treatment schedule and intensity in the acute-subacute phase.
The ongoing refinement of identifying which individuals are the most likely to benefit from tDCS is an important contribution to the overall individualization and optimization of post-stroke care. While precision medicine necessitates comprehensive and multidimensional assessment of a person’s personal and medical traits, based on the present findings, neurostimulation with tDCS may hold unique benefit for older patients. We hope future work will replicate and expand upon these findings to further identify which features lead to the best responses to treatment when behavioral therapy is augmented with tDCS.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Hillis receives compensation from the American Heart Association as Editor-in-Chief of Stroke and from Elsevier as Associate Editor of Practice Update Neurology. All authors receive salary support from NIH (NIDCD) through grants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by National Institutes of Health/National Institute on Deafness and Other Communication Disorders (NIH/NIDCD): P50 DC014664 and R01 DC05375. The imaging resources for this study were funded by NIH grant 1S10OD021648.
Data Availability
Deidentified participant data will be made available upon publication from the Inter-university Consortium for Political and Social Research (ICPSR). Study protocol and related documents are available on via ClinicalTrials.gov.