Abstract
Background. A successful interplay between prefrontal and domain-specific language areas is critical for language processing. Previous studies involving people with aphasia have shown that executive control processes might act on lexical-semantic representations during retrieval. Modulating the prefrontal control network by means of noninvasive brain stimulation might, therefore, improve lexical access in people with aphasia. Objective. The present study investigates the effects of prefrontal transcranial direct current stimulation (tDCS) on lexical access in chronic poststroke aphasia. Methods. We report data of 14 participants with chronic poststroke aphasia. We used a sham-tDCS (S-tDCS) controlled and double-blind within-subjects design. Performances in picture naming, verbal fluency, and word repetition were assessed immediately after stimulation. Results. As compared with S-tDCS, anodal tDCS (A-tDCS) improved verbal fluency as well as the speed of naming high frequency words, but not word repetition. Conclusion. The results of our study suggest that the brain network dedicated to lexical retrieval processing can be facilitated by A-tDCS over the left dorsolateral prefrontal cortex. This finding supports the notion that strengthening executive control functions after stroke could complement speech and language-focused therapy.
Introduction
Recovery from poststroke aphasia is a dynamic process by which undamaged brain networks reorganize adaptively to compensate for the loss of function.1,2 A new approach in aphasic neurorehabilitation is to move from a language-centric understanding of aphasia toward one focusing on nonlinguistic factors that can support and possibly reshape neural networks engaged in language recovery. 1 In the present study, we thus investigated whether enhancing the prefrontal control network by means of noninvasive brain stimulation (NIBS) has a facilitating effect on lexical access in chronic aphasia.
According to the Memory-Unification-Control model, language comprehension and production requires cognitive control and working memory resources located in the left dorsolateral prefrontal cortex (DLPFC).3,4 The left DLPFC has been shown to be intimately associated with many language-related processes, including verbal production, 5 sentence processing,6,7 and discourse performance. 8 In word retrieval, deficits in cognitive control might influence lexical-semantic access via excessive inhibition of lexical representations,9,10 difficulties in selecting among concurrent activated representations, 11 abnormal interference in the retrieval of lexemes, 12 or deficient semantic short-term memory. 13 Modulating the prefrontal cortex by means of NIBS might thus improve lexical access in people with aphasia.
NIBS over nonlinguistic areas with the aim of improving language production has to date rarely been investigated. Some studies showed that stimulating the DLPFC can improve picture naming in Alzheimer disease patients, 14 healthy participants,15-17 and people with aphasia.18,19 Facilitatory effects of excitatory transcranial direct current stimulation (tDCS) might not only depend on stimulation of the DLPFC itself, but also on spreading of activation to anatomically and functionally connected brain areas of the language network. 20 As such, the DLPFC might be a potential target for reorganization of language functions during rehabilitation.
Based on this assumption, the present group study aimed at investigating whether excitatory tDCS administered to the left DLPFC can improve lexical access in people with chronic poststroke aphasia, using a sham-tDCS (S-tDCS)controlled and double blind within-subject design. To date, most studies with aphasic participants investigated the effect of brain stimulation on picture naming but not on other language production tasks (for a review, see Wortman-Jutt and Edwards 21 ). As such, we looked at the effects of prefrontal tDCS on several language measures, including a picture naming task, a word repetition task, and a verbal (phonemic) fluency task.
Phonemic fluency is known as a valid task for assessing both verbal ability, specifically lexical access, as well as executive ability, such as working memory, inhibition, effortful self-initiation, and switching ability.22,23 Adding a phonemic fluency task thus enabled us to compare the impact of DLPFC stimulation on a task measuring mainly lexical access (picture naming) and a task assessing more directly the interplay between executive control and lexical access (phonemic fluency). According to dual-route computational models of auditory repetition,24,25 repeating familiar words involves both a lexical and a nonlexical component, whereas picture naming relies on the lexical route only. In the lexical route, the semantic and lexical representations of the target word are activated, followed by its phonological form for output. On the other hand, the nonlexical route converts auditory input directly to phonological output. It is assumed that this additional nonlexical route in repeating auditory words results in better performance in word repetition as compared with picture naming in people with aphasia, at least in the case of strong impairments of the lexical route. 24 Because there is evidence that executive functions may act on lexical-semantic retrieval, we expected larger effects of prefrontal stimulation on verbal fluency and picture naming, strongly involving lexical access, as compared with word repetition, most probably involving both a lexical and a nonlexical component.
Material and Methods
Declaration of Ethical Approval
All participants gave written informed consent prior to participation. The study was approved by the local Competent Ethics Committee and conducted in accordance with the Declaration of Helsinki and the guidelines of Good Clinical Practice issued by the International Conference on Harmonization (ICH). The trial was preregistered on “clinicaltrials.gov” (NCT02840370) and on the Swiss National Clinical Trials Portal (SNCTP). Participants received a compensation for their participation.
Participants
We report data from 14 participants with chronic aphasia (>6 months poststroke, mean = 34.6 ± 28.6) caused by a stroke (mean age = 57.40 ± 8.84 years). Inclusion criteria were the following: left-hemisphere lesion with intact DLPFC, right-handedness, mother tongue French, normal or corrected-to-normal visual acuity and hearing, no psychiatric comorbidity, no dementia, no intracranial metal or pacemaker. Participants’ demographic and clinical data are summarized in Table 1. From an original group of 19, 3 participants were excluded after the screening because of intracranial metal. One participant was excluded after the first tDCS session because of persisting headaches (9 days), and another participant was excluded from the final analysis because he did not produce a single correct item in any language task. The study was multicentric and took place in the Hôpitaux Universitaires de Genève and the HFR Fribourg—Hôpital Cantonal.
Overview of Demographic and Clinical Data.
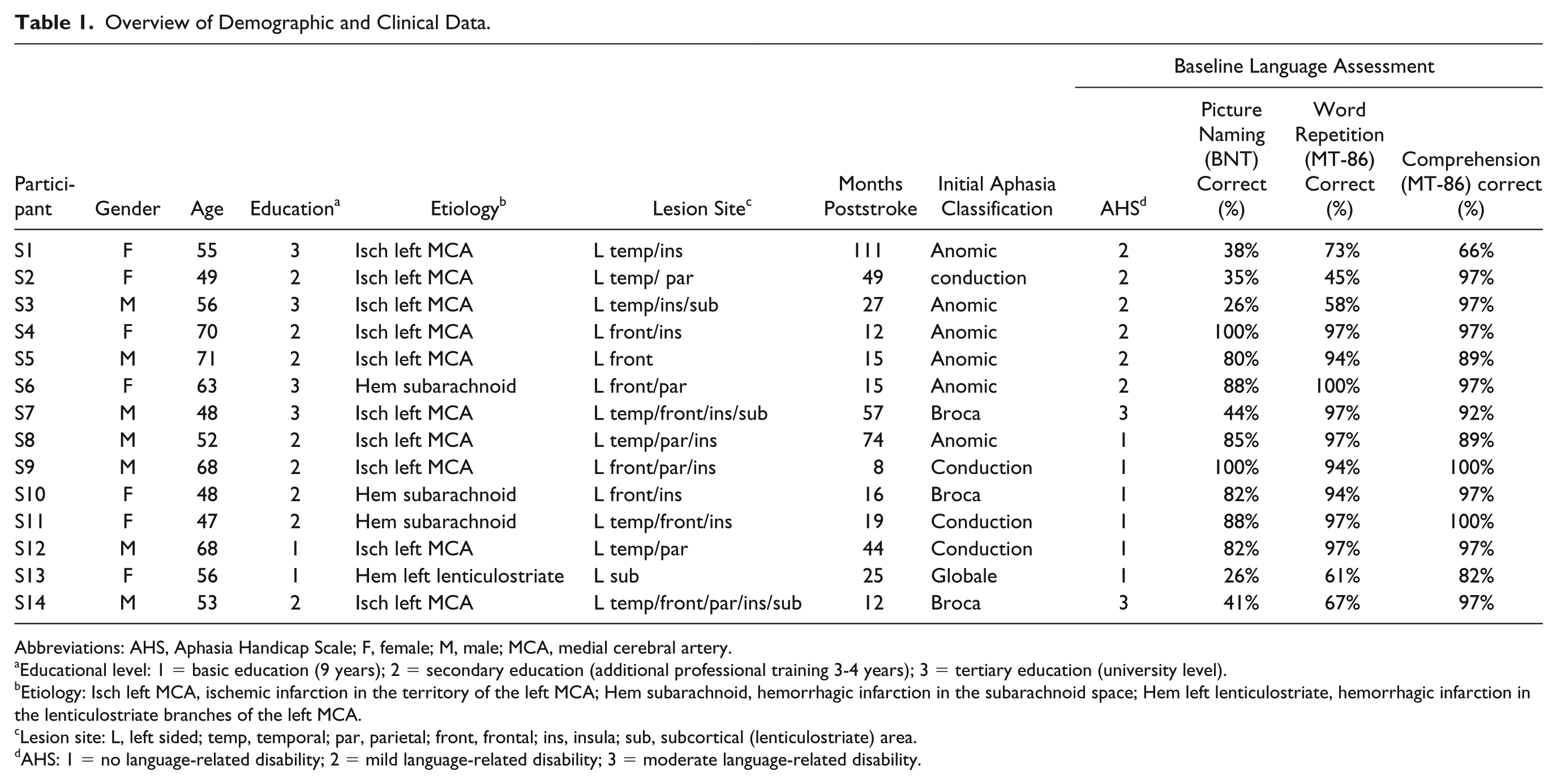
Abbreviations: AHS, Aphasia Handicap Scale; F, female; M, male; MCA, medial cerebral artery.
Educational level: 1 = basic education (9 years); 2 = secondary education (additional professional training 3-4 years); 3 = tertiary education (university level).
Etiology: Isch left MCA, ischemic infarction in the territory of the left MCA; Hem subarachnoid, hemorrhagic infarction in the subarachnoid space; Hem left lenticulostriate, hemorrhagic infarction in the lenticulostriate branches of the left MCA.
Lesion site: L, left sided; temp, temporal; par, parietal; front, frontal; ins, insula; sub, subcortical (lenticulostriate) area.
AHS: 1 = no language-related disability; 2 = mild language-related disability; 3 = moderate language-related disability.
Lesion Characteristics
Lesion mapping was performed with the MRIcron software. 26 Magnetic resonance imaging (MRI) scans were available for 11 participants, and computed tomography (CT) scans were available for 3 participants. The mean time elapsed between stroke and MRI/CT was 4.8 days (SD = 3.5; range = 1-12), not including 1 participant for whom 172 days had elapsed, because for this participant a clearly demarked zone was only identifiable in the later scan. None of the participants had any additional neurological event between the date of the MRI/CT used and the date of the experiment. All lesions were delineated directly on the individual MRI/CT images for every available transversal slice. Each MRI/CT image and lesion was normalized to the T1 MNI templates using the spatial normalization algorithm provided by SPM12 (http://www.fil.ion.ucl.ac.uk/spm). If normalization failed (n = 2), the lesions were drawn directly on the T1-weighted MNI brain template implemented by MRIcron and visually controlled for different slice angles. Figure 1 shows the localization and the degree of overlap of the brain lesions of the 14 participants.
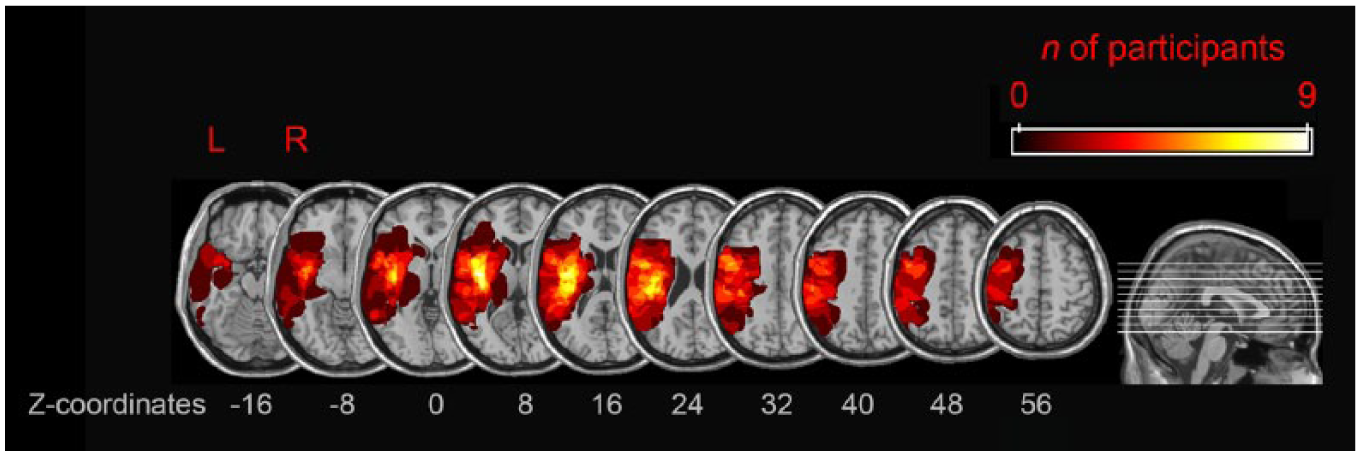
Lesion overlap of the 14 participants who completed the study.
Experimental Procedure
Participants took part in 3 sessions: a screening, 1 anodal (A-tDCS) session, and 1 S-tDCS session. In the first session, the inclusion/exclusion criteria were evaluated, and a speech-language pathologist/neuropsychologist conducted the aphasia screening. To measure baseline language abilities, repetition and oral comprehension were assessed using subtests of the MT-86, 27 and naming abilities were evaluated with a French version of the Boston Naming Test. 28 Global aphasia-related handicap/disability was quantified with the Aphasia Handicap Scale (AHS), a 5-point scale in which participants rated their perceived disability and autonomy in everyday life.29,30 The 2 tDCS sessions were separated by a 1-week interval. Between the 2 sessions, participants followed their daily life routine and did not undergo any speech therapy. The order of stimulation (A-tDCS/S-tDCS) was randomly assigned to the participants and not communicated to the investigators/therapists who gave the task instructions to the participants. All primary outcome measures were assessed immediately after (3.7 ± 1.2 minutes) tDCS stimulation. Based on studies showing that tDCS-induced changes in synaptic activity seem to outlast the duration of stimulation only when stimulation is paired with ongoing synaptic activation,31,32 participants already trained the same processes during stimulation with parallel versions of the same tasks (picture naming, repetition, phonemic fluency). In addition to the picture naming, fluency, and repetition task, participants also conducted a nonverbal Flanker task, 33 both during as well as after stimulation. Because the focus of the present article is on the effect of prefrontal tDCS on lexical access, in the following, we only describe the tasks related to this research question. Results of the Flanker task can be found in Supplementary Material A. The order of tasks was counterbalanced between participants. Parallel lists were used for the language tasks and counterbalanced across sessions (1, 2) and stimulation (A-tDCS, S-tDCS) by the lab engineer.
Transcranial Direct Current Stimulation
tDCS was administered using a battery driven stimulator (DC-Stimulator Plus, NeuroConn). The electrodes were slid into saline-soaked sponges (0.17 mL/cm2) before being positioned. With this amount (4.3 mL saline for each side of the active electrode and 6 mL saline for each side of the reference electrode), we were sure not to oversoak the sponges but keep good electrode contact. The anode was placed over the left DLPFC, individually located with the Beam F3 system. 34 The cathode was positioned over the right supraorbital area. According to computational modeling, this electrode montage exhibits high current density in the frontal lobes of the nonlesioned brain. 35 A smaller anode (5 × 5 cm2) was used to increase focality, whereas a larger reference electrode (5 × 7 cm2) was used to render the stimulation over the supraorbital area functionally less effective. 36 In the A-tDCS condition, the current was set at 1 mA with a duration of 20 minutes, which results in a current density of 0.04 mA/cm2 (1/25 cm2). The current was ramped up for 30 s at the beginning and ramped down for 15 s at the end of the stimulation. In the S-tDCS condition, the current was ramped up for 30 s followed by 40 s of direct current, 15 s fade out, and 19 minutes without any stimulation.
Primary Outcome Measures and Analyses
The tasks were programmed in E-Prime 2.0 (Psychology Tools, Inc, Pittsburgh, PA). Word productions were recorded, and response latencies were systematically checked with a speech analysis software (Audacity). Importantly, the person checking accuracy and response times (RTs) was blinded toward the stimulation condition (A-tDCS, S-tDCS).
Picture Naming Task
The 4 picture lists used for A-tDCS/S-tDCS online (during stimulation) and offline (directly after stimulation) all consisted of 24 black and white line drawings selected from the picture database of Cycowicz et al, 37 which was normed in French by Alario and Ferrand. 38 The lists were matched for several factors, which could have an influence on naming RT—that is, name agreement, image agreement, visual complexity, age of acquisition, log-transformed word frequency, and number of syllables. 39 Each of the 4 lists consisted of 6 very-high-frequency (frequency of >50/million), 6 high-frequency (frequency of 20-50/million), 6 low-frequency (frequency of 5-20/million), and 6 very-low-frequency words (frequency of <5/million). Frequency values are based on the film frequencies reported in the LEXIQUE database. 40 In addition, the lists were matched for log-transformed phonological neighborhood density and syllabic structure 40 because these factors can have an effect on language production errors in people with aphasia.41,42 Moreover, each of the 4 lists contained the same amount of living and nonliving objects.
In each trial, the image was presented centrally on the screen for 14 s after a fixation cross of 1 s. Participants were instructed to name the pictures as fast and accurately as possible.
Phonemic Fluency Task
In the phonemic fluency task, participants were instructed to name as many words as possible within 1 minute, starting with a given letter. Two letters with comparable initial letter frequency in French were used each during A-tDCS/S-tDCS (“R,” 0.095%, and “P,” 0.093%) and after A-tDCS/S-tDCS (“B,” 0.053%, and “T,” 0.050%). 40 The comparable letters were counterbalanced across sessions (1, 2) and stimulation (A-tDCS, S-tDCS).
Repetition Task
In the repetition task, participants were instructed to repeat 24 spoken words as fast and accurately as possible. The participants were given 6 s to repeat each word. After 6 s, the next spoken word was presented via speakers.
Each of the 4 lists of 24 stimuli equally consisted of 6 very-high-frequency, 6 high-frequency, 6 low-frequency, and 6 very-low-frequency words, with the same frequency categories as used in the picture naming task. 40 Each of these 4 word frequency categories included 2 monosyllabic words, 2 bisyllabic words, and 2 trisyllabic words, of which always 1 word had a complex syllabic structure (with syllabic cluster) and 1 a simple syllabic structure (without syllabic cluster). The lists were, therefore, matched for word frequency, number of syllables, syllabic structure, and additionally also for word length and log-transformed phonological neighborhood density based on the LEXIQUE database. 40
Statistical Analyses of Primary Outcomes
We analyzed the differences in reaction time (RT) and accuracy between S-tDCS and A-tDCS in the language tasks with paired sample t-tests (1-tailed). Wilcoxon signed-rank tests were performed when the assumption of normality was violated. To control for multiple comparisons for the main effects of stimulation on language production (fluency, picture naming RT, picture naming accuracy, repetition RT, repetition accuracy), a Bonferroni adjusted α threshold was used (0.05/5 = 0.01).
For the picture naming and repetition task (RT and accuracy), we further performed exploratory post hoc 2 × 4 repeated-measure ANOVAs with the 2 factors Stimulation (anodal, sham) and Word frequency (very low, low, high, very high) to investigate if accessing high-/low-frequency words is differentially modulated by prefrontal tDCS.
Secondary Outcome Measures and Analyses
Modulatory Effects of Participants’ Characteristics on the Effect of Stimulation
To evaluate whether specific participant characteristics (age, education level, months poststroke, aphasia type, AHS or baseline measures of naming, repetition, and comprehension) modulated the effects of stimulation, we performed additional mixed-design ANOVAs and ANCOVAs.
Lesion Analysis
We performed an exploratory voxel-based lesion-symptom mapping (VLSM) to identify the location of brain lesions that could influence the effects of stimulation. The lesions and the scores of the outcome measures, which showed an effect of stimulation, were submitted to statistical mapping analyses using the VLSM algorithms implemented in the NPM software by Rorden et al. 26 t-Tests on the scores of the outcome measures were run separately on each voxel to compare performance in participants with versus without lesion at this voxel. The statistical tests were conducted only on voxels damaged in at least 3 participants. The results of the t-tests were then color coded and projected on the MNI template brain using MRICro 43 (for similar approaches, see Manuel et al 44 and Spierer et al 45 ).
Assessment of tDCS-Induced Discomfort
Within the first 3 minutes of stimulation, the participants evaluated discomfort 3 times (30 s, 1.5 minutes, and 2.5 minutes after starting the stimulation) with a 6-point Likert scale showing happy to pained faces. Moreover, each participant was asked in a structured interview whether he/she experienced any of the following side effects after stimulation: headache, neck pain, burning sensation on the skin, itching, redness of the skin, difficulties in concentrating, and/or a change in mood. Besides measuring discomfort, a comparison between anodal and sham stimulation of these ratings (Wilcoxon signed-rank tests) also gave us an indication on the successful blinding of the participants.
Results
Primary Results: Effects of Stimulation on Language Production
In the phonemic fluency task, we found an effect of Stimulation [t(13) = 2.65, P = .010, dav = 0.28; Figure 2], showing that participants produced more words after A-tDCS (Mean = 6.5; SD = 3.53) than after S-tDCS (Mean = 5.5; SD = 3.50). No effects of Stimulation (A-tDCS vs S-tDCS) were found in the repetition and picture naming task (all P ⩾ .233; Table 2).
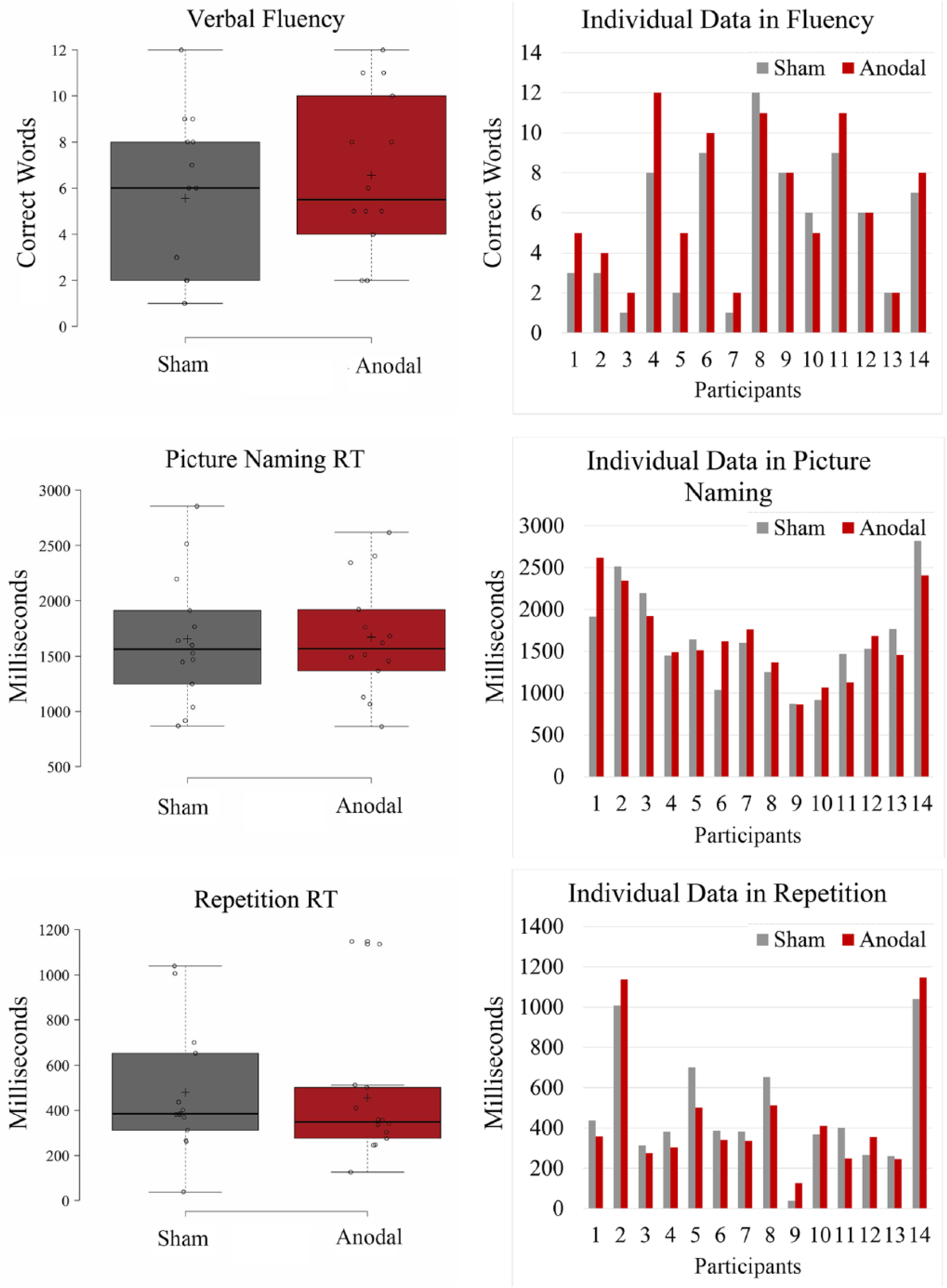
Boxplots of the main results after anodal and sham stimulation in the fluency, picture naming, and repetition tasks generated with the BoxplotR web tool42. Center lines show the medians. Box limits indicate the 25th and 75th percentiles as determined by R software. Whiskers extend 1.5 times the interquartile range from the 25th and 75th percentiles.
Paired Sample t-Tests and Wilcoxon Signed-Rank Tests for the Effects of Stimulation on Language Production.
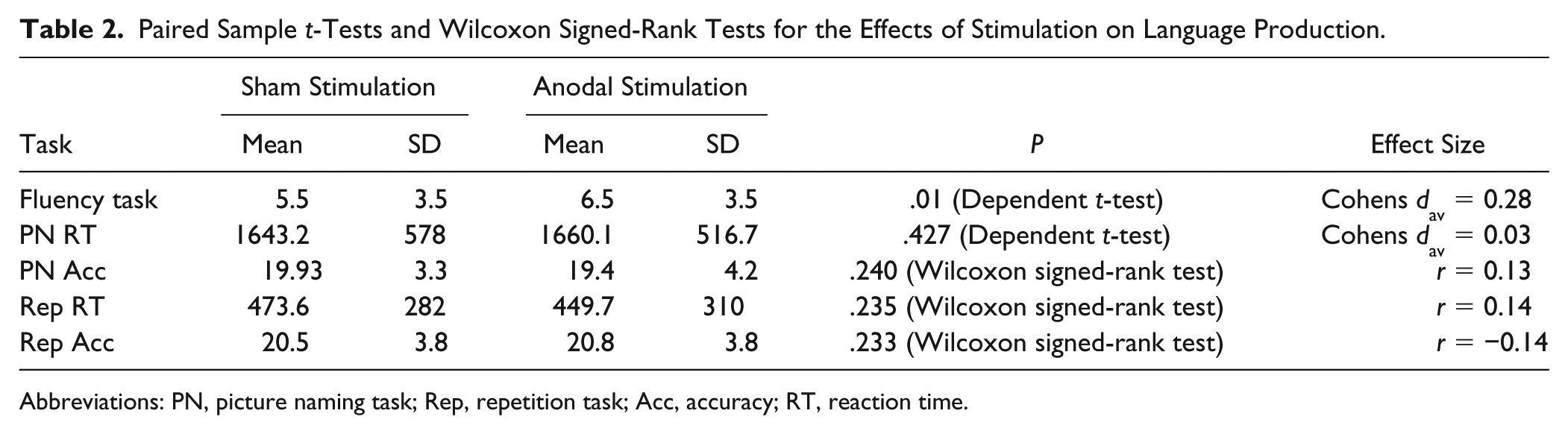
Abbreviations: PN, picture naming task; Rep, repetition task; Acc, accuracy; RT, reaction time.
For the picture naming task, the exploratory 2 × 4 ANOVA with the within-subject factors Stimulation (anodal/sham) and Word Frequency (very high, high, low, very low) revealed a main effect of Word Frequency [F(3, 36) = 2.885; P = .049; η p 2 = 0.19], showing that the fastest responses were given in very-high-frequency words and the slowest responses in very-low-frequency words. Moreover, a significant interaction between Word Frequency and Stimulation [F(3, 36) = 3.213; P = .034; η p 2 = 0.21] revealed faster responses after anodal stimulation (mean RT = 1264.9 ms) than after sham stimulation (mean RT = 1913 ms) in very-high-frequency words only. For the repetition task, the 2 × 4 ANOVA showed no main effects and no interaction.
As can be seen in Figure 2, the effects of stimulation within individual participants were not consistent across the different language tasks.
Modulatory Effects of Participant Characteristics on Verbal Fluency
A mixed-design ANOVA with the within factor Stimulation (anodal, sham) and the between factor self-reported handicap in the AHS (no disability, mild disability, moderate disability) revealed a main effect of Stimulation [F(1, 11) = 8.49; P = .014; η p 2 = 0.44] and an interaction between stimulation and the self-reported handicap in the AHS [F(2, 11) = 4.71; P = .03; η p 2 = 0.46]. As can be seen in Table 3, all participants who reported language-related disability (n = 8) showed higher scores in the verbal fluency task after anodal stimulation as compared with sham stimulation, whereas only 1 out of 6 participants without language-related disability showed a positive stimulation effect. No other participant characteristic modulated the effect of stimulation (P > .05 for all ANOVAs and ANCOVAs).
Cross-Tabulation of the Variables AHS × Responders in Verbal Fluency.

Abbreviations: A-tDCS, anodal tDCS; S-tDCS, sham tDCS; tDCS, transcranial direct current stimulation.
Yes, participants with higher scores after A-tDCS than S-tDCS; No, participants with equal or lower scores after A-tDCS as compared with S-tDCS.
Lesion Analysis
The VLSM, including difference scores between anodal and sham stimulation as predictor did not reveal significant differences (P < .05, uncorrected) between the lesions of responders and nonresponders in the verbal fluency task. Visual inspection of the t-map (Figure 3) suggests that there might be a tendency for more frontally located brain lesions in responders as compared with nonresponders.
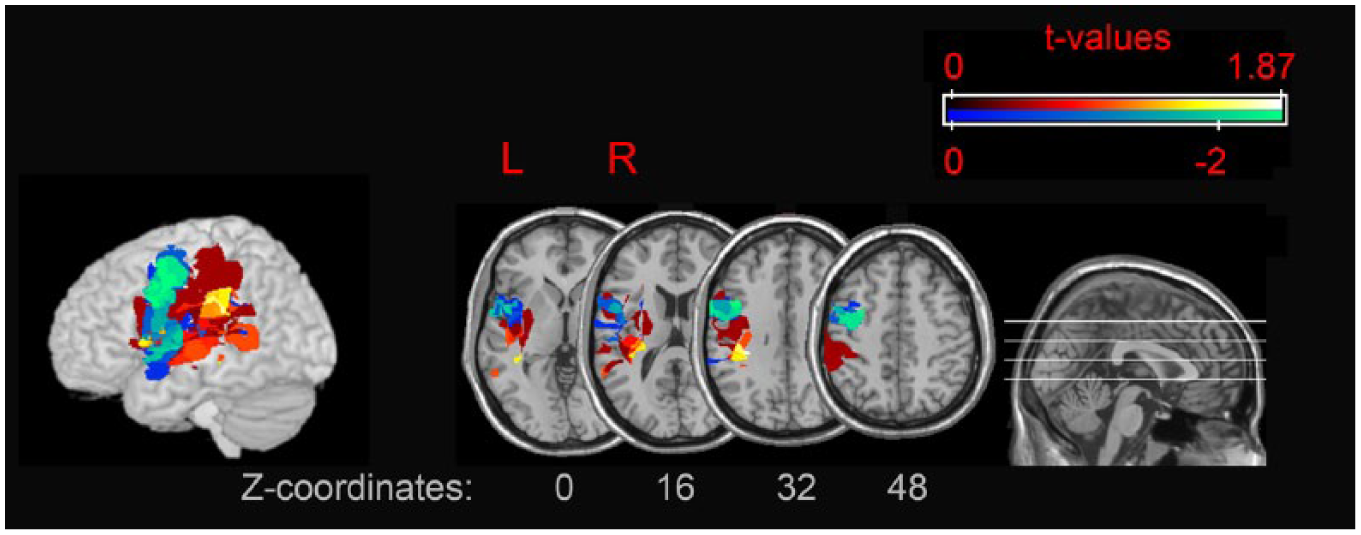
t-Map with difference scores (sham-anodal) in verbal fluency as predictor. Light colors indicate higher t-values. In blue/green: damaged regions associated with improvements in verbal fluency after A-tDCS; in red/yellow: damaged regions associated with no improvement in fluency after A-tDCS.
Discomfort Ratings
Discomfort within the first 3 minutes of stimulation ranged between 0 and 4.5 on a scale of 6. Statistical analyses revealed that the discomfort ratings were comparable between A-tDCS and S-tDCS after 30 s and 1.5 minutes of stimulation (Wilcoxon signed-rank test, P > .05), but participants noted a slightly stronger discomfort in A-tDCS as compared with S-tDCS after 2.5 minutes (Wilcoxon signed-rank test, P = .05). A comparable amount of side effects was reported after S-tDCS versus A-tDCS (see Supplementary Material B).
Exploratory Analysis of Online Effects
To investigate whether the effects of A-tDCS already manifested during stimulation, we conducted a post hoc analysis between A-tDCS and S-tDCS for the online performances in the picture naming and fluency tasks. No significant effects were found (all P > .125).
Discussion
To our knowledge, this is the first study stimulating the prefrontal executive control network and looking at modulatory effects on several language production tasks in chronic poststroke aphasia. We used a S-tDCS controlled, double-blind, within-subjects design to control for possible expectation effects of the participant and/or experimenter. No differences between side effects or changes in mood following A-tDCS and S-tDCS were found. As such, the present tDCS protocol (intensity: 1 mA; current density: 0.04 mA/cm2; duration: 20 minutes) can be used without major concerns about safety and blinding, and it is unlikely that the effects of stimulation on language can be attributable to a change in mood.
Prefrontal A-tDCS Improves Phonemic Fluency
We found an effect of stimulation on phonemic fluency, indicating that left prefrontal tDCS can improve processes that depend on an interplay between language and executive control functions. This interplay has been shown to be crucial for functional communication in everyday life in aphasia.46,47 In a study conducted by Sarno and Levita, 48 the most marked difference between aphasic patients who improved most and those who improved least in general language functioning could be found in phonemic fluency, when compared with several other language measures (naming, sentence comprehension, and Token Test). Because prefrontal A-tDCS seems to be able to enhance phonemic fluency, it could thus be that strengthening the prefrontal executive control network in addition to speech-language therapy might also have beneficial effects on more general communication functioning in aphasia. This issue could be taken up in future studies by adding a functional communication measure.
Interestingly, all the participants who reported language-related handicap in the baseline-screening showed an improvement in phonemic fluency after anodal stimulation, whereas most of those who did not report any language-related handicap did not improve after A-tDCS. This subjectively perceived language-related handicap did not show any relationship with the objective performance in the language tasks assessed during the baseline screening (ie, naming, repetition, and comprehension). According to Fridriksson et al 46 and Ramsberger, 47 such discrepancies between impairment-based tests of aphasia and language-related abilities in everyday life could be partly explained by different amounts of executive function involvement. It might thus be that participants who feel less handicapped by their language impairments rely more strongly on executive control functions to improve their everyday-life communication. This could explain why prefrontal stimulation mainly improved the performance of participants who possibly do not rely sufficiently on executive control functions to compensate for their language impairments and who could, thus, benefit from an enhancement of prefrontal activation. However, this explanation remains speculative because it could also be that participants who are less aware of their handicap report having no disability despite, or even because of, executive difficulties.
Contrasting the results from Baker et al, 49 revealing that frontal tDCS in aphasia is more effective when the lesion location is close to the stimulation site, our results did not reach significance. However, because the visual inspection of the t-map shows a similar pattern as reported by Baker et al, 49 it would be interesting to take up such an analysis in future large-scale studies. We found no differences between A-tDCS and S-tDCS during stimulation (online). This finding is in agreement with previous evidence from studies with healthy participants demonstrating that A-tDCS over the left DLPFC improves verbal fluency only after, but not during, stimulation. 50 It seems conceivable that increases in the excitability of the underlying cortex as a consequence of A-tDCS develop over time instead of arising straight away. Consequently, counterbalancing the order of tasks in the present study might have led to a “wash-out” of possible online effects.
Prefrontal A-tDCS Improves Picture Naming in High-Frequency Words Only
In contrast to studies with healthy participants,15-17 we found no overall effects of prefrontal stimulation on picture naming. However, an effect of stimulation on RT was found for pictures with high-frequency target words. tDCS-induced changes in cortical activity might, thus, only be sufficient for modifying frequently used words, which have higher resting levels of activation51,52 and stronger representations 53 in the language system. The same rate of changes in neural activity sufficient for improving the access of high-frequency words may, thus, not be sufficient to achieve the necessary activation level for the production of lower-frequency words. The idea that single sessions of tDCS might only improve frequently used words is in line with studies showing that A-tDCS enhances the retrieval of trained (ie, frequently used) but not untrained items.54,55 A transfer to untrained items was previously found only after repeated stimulations in combination with high-intensive naming therapy, 56 which could also be the case for low-frequency words.
No Effect of Prefrontal A-tDCS on Repetition
For word repetition, we found no effect of stimulation. Based on the assumption that prefrontal stimulation supports lexical retrieval,9-11 the lack of an effect in repetition can be explained by the strong involvement of the nonlexical route when repeating auditory words. This is further supported by the lack of a word frequency effect in the repetition task, which suggests that our participants relied strongly on the nonlexical route in repeating auditory words.
Limitations
Because of the relatively small sample size as well as the low statistical power, the results reported should be interpreted cautiously. Moreover, recent reviews have brought forward the issue of unexplained intrasubject and intersubject variability in NIBS, a factor whose influence on the current results cannot be ruled out.57,58 In addition to these limitations, the participants included in the present study differed regarding their lesion localization, language difficulties, and time elapsed since the stroke. Despite using an electrode montage that has been shown to exhibit high current density in the frontal lobes, 35 we do not know how each lesion topography could have influenced the current spread of tDCS. However, this heterogeneity of the sample allowed us to investigate the variability of tDCS effects, which could encourage future research in the field of NIBS and aphasia.
Conclusions
In conclusion, we found that anodal stimulation of the intact left DLPFC modulates performance of aphasic participants in a verbal fluency task, supporting the idea that increasing prefrontal excitability might have beneficial effects on language functioning in aphasia. Moreover, despite no overall effects on picture naming and repetition, prefrontal stimulation had a facilitative effect on naming high-frequency words. The results of our study indicate that strengthening executive control functions after stroke could complement speech and language–focused therapy. This notion can be used to guide the development of future therapies in aphasia rehabilitation.
Supplemental Material
Supplementary_material_A – Supplemental material for Effects of Prefrontal Transcranial Direct Current Stimulation on Lexical Access in Chronic Poststroke Aphasia
Supplemental material, Supplementary_material_A for Effects of Prefrontal Transcranial Direct Current Stimulation on Lexical Access in Chronic Poststroke Aphasia by Maria I. Pestalozzi, Marie Di Pietro, Chrisovalandou Martins Gaytanidis, Lucas Spierer, Armin Schnider, Leila Chouiter, Françoise Colombo, Jean-Marie Annoni and Lea B. Jost in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_material_B – Supplemental material for Effects of Prefrontal Transcranial Direct Current Stimulation on Lexical Access in Chronic Poststroke Aphasia
Supplemental material, Supplementary_material_B for Effects of Prefrontal Transcranial Direct Current Stimulation on Lexical Access in Chronic Poststroke Aphasia by Maria I. Pestalozzi, Marie Di Pietro, Chrisovalandou Martins Gaytanidis, Lucas Spierer, Armin Schnider, Leila Chouiter, Françoise Colombo, Jean-Marie Annoni and Lea B. Jost in Neurorehabilitation and Neural Repair
Supplemental Material
Supplementary_material_C – Supplemental material for Effects of Prefrontal Transcranial Direct Current Stimulation on Lexical Access in Chronic Poststroke Aphasia
Supplemental material, Supplementary_material_C for Effects of Prefrontal Transcranial Direct Current Stimulation on Lexical Access in Chronic Poststroke Aphasia by Maria I. Pestalozzi, Marie Di Pietro, Chrisovalandou Martins Gaytanidis, Lucas Spierer, Armin Schnider, Leila Chouiter, Françoise Colombo, Jean-Marie Annoni and Lea B. Jost in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank Dr Camille Chavan for her help in data collection, which was very much appreciated.
Authors’ Note
Jean-Marie Annoni and Lea B. Jost share last authorship.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Swiss National Science Foundation (Grant numbers 325130_156937, 320030_175469).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
