Abstract
Background. Previous findings suggest that language disorders may occur in severely brain-injured patients and could interfere with behavioral assessments of consciousness. However, no study investigated to what extent language impairment could affect patients’ behavioral responses. Objective. To estimate the impact of receptive and/or productive language impairments on consciousness assessment. Methods. Twenty-four acute and subacute stroke patients with different types of aphasia (global, n = 11; Broca, n = 4; Wernicke, n = 3; anomic, n = 4; mixed, n = 2) were recruited in neurology and neurosurgery units as well as in rehabilitation centers. The Coma Recovery Scale–Revised (CRS-R) was administered. Results. We observed that 25% (6 out of 24) of stroke patients with a diagnosis of aphasia and 54% (6 out of 11) of patients with a diagnosis of global aphasia did not reach the maximal CRS-R total score of 23. An underestimation of the consciousness level was observed in 3 patients with global aphasia who could have been misdiagnosed as being in a minimally conscious state, even in the absence of any documented period of coma. More precisely, lower subscores were observed on the communication, motor, oromotor, and arousal subscales. Conclusion. Consciousness assessment may be complicated by the co-occurrence of severe language deficits. This stresses the importance of developing new tools or identifying items in existing scales, which may allow the detection of language impairment in severely brain-injured patients.
Keywords
Introduction
Language disorders such as aphasia represent a major issue for consciousness assessment. Language plays a key role in the communication of the content of consciousness and in the interaction of an individual with his or her surroundings. 1 In the absence of such a skill, the level of consciousness may be difficult to determine accurately and could easily be underestimated. Stroke is the most frequent etiology of aphasia and leads to such a deficit in up to 30% of cases (vs 15% in case of traumatic brain injury).2,3 Patients suffering from a severe brain injury due to stroke are therefore likely to present with aphasia. In brain-injured patients with disorders of consciousness, such a deficit may be difficult to detect as these patients often present few behavioral responses and are easily exhausted.4-6 However, a hypometabolism of up to 50% in areas related to language processing (ie, left temporal cortex, left inferior frontal cortex, and right inferior temporal cortex) has been observed in patients diagnosed as being in a minimally conscious state (MCS; ie, fluctuating but reproducible conscious behavioral responses).7-9 A previous study has also shown a decrease in the superior and middle temporal cortex in MCS patients as compared to controls when exposed to effortful language processing demands (ie, comprehensible vs reversed narratives). 10 Recently, Bruno et al 11 reported that MCS− patients (showing low-level behavioral responses such as visual pursuit or localization to noxious stimulation without command following) exhibited lower cerebral metabolism in left-hemispheric cortical areas, including the language network, as compared with MCS+ patients (showing high-level behavioral responses, including command following, intelligible verbalizations, or unreliable yes–no responses). Additionally, Broca’s area was disconnected from the rest of the language processing regions in MCS− patients as compared with MCS+ patients. 11 Language disorders may therefore occur in MCS patients and could interfere with behavioral assessments of consciousness. It is nevertheless almost impossible to detect language impairment in patients with disturbance in consciousness. In this context, it would be informative to assess the extent to which documented language impairments could affect patients’ behavioral responses. Thus, we investigated the impact of receptive and productive language impairments on the assessment of consciousness in patients with an established diagnosis of aphasia by administering the Coma Recovery Scale–Revised, a standardized behavioral scale commonly used to detect signs of consciousness in severely brain-injured patients.12,13
Methods
Participants
Inclusion and Exclusion Criteria
Patients were recruited in neurology, neurosurgery, and neurorehabilitation units. The inclusion criteria were (a) ischemic or hemorrhagic stroke as an etiology and (b) a diagnosis of aphasia based on standardized neuropsychological assessment (using the Aachen Aphasia Test or the Aphasia Checklist; as described below). The exclusion criteria were (a) documented period of coma (ie, Glasgow Coma Score ≤6 with E1, V1, and M1-4, the day of the admission) 14 ; (b) documented history of prior brain injury; (c) premorbid history of developmental, psychiatric, or neurologic illness; and (d) uncorrected visual or hearing impairments.
Diagnosis of Aphasia
The patients hospitalized in neurorehabilitation units were diagnosed based on the Aachen Aphasia Test (AAT). 15 This test is an assessment tool for the diagnosis, classification, and quantification of aphasic disorders that has been translated into English, Dutch, Italian, and Portuguese,16-19 and is one of the most widely used aphasia batteries in Europe. The AAT consists of 6 subtests assessing spontaneous and written language, repetition, naming, and comprehension of words/sentences and of commands (ie, Token Test). Based on the nature and severity of receptive and expressive language deficits, performance is categorized as Broca’s, Wernicke’s, global, anomic, or mixed-type aphasia. Patients hospitalized in neurology and neurosurgery units were diagnosed based on the Aphasia Checklist (ACL), which consists of 7 subtests assessing language comprehension and production, reading, writing, repetition, word generation, and also verbal communication. 20 The total score varies from 0 to 148 with a cutoff of 135, below which a diagnosis of aphasia is established. The resulting profile of receptive and expressive language deficits establishes the aphasia subtype. The ACL takes approximately 30 minutes to administer and is therefore well-suited for the acute population in whom fatigue may negatively influence test performance. Finally, we documented each patient’s brain lesions according to their available magnetic resonance imaging or computed tomography scan reports.
Behavioral Assessment
The Coma Recovery Scale–Revised (CRS-R) was administered once to each participant after a 30-minute period of rest without nursing care. The CRS-R consists of 23 hierarchically arranged items that comprise six subscales assessing arousal, auditory, visual, motor, oromotor/verbal, and communication functions. In each subscale, the highest item represents cognitively mediated behaviors while the lowest item represents reflexive activity. Table 1 presents each item of the scale and specifies whether each can be associated with receptive and/or productive language. In this prospective study, we administered all the items of each subscale. Scores related to the highest item achieved in each subscale were summed to obtain the total score which varied between 0 and 23. The CRS-R also allowed us to determine whether patients met existing diagnostic criteria for disturbance in consciousness (ie, MCS) based on the presence or absence of 2 specific target behaviors, functional object use, and functional communication. 12
Items of the Coma Recovery Scale–Revised.
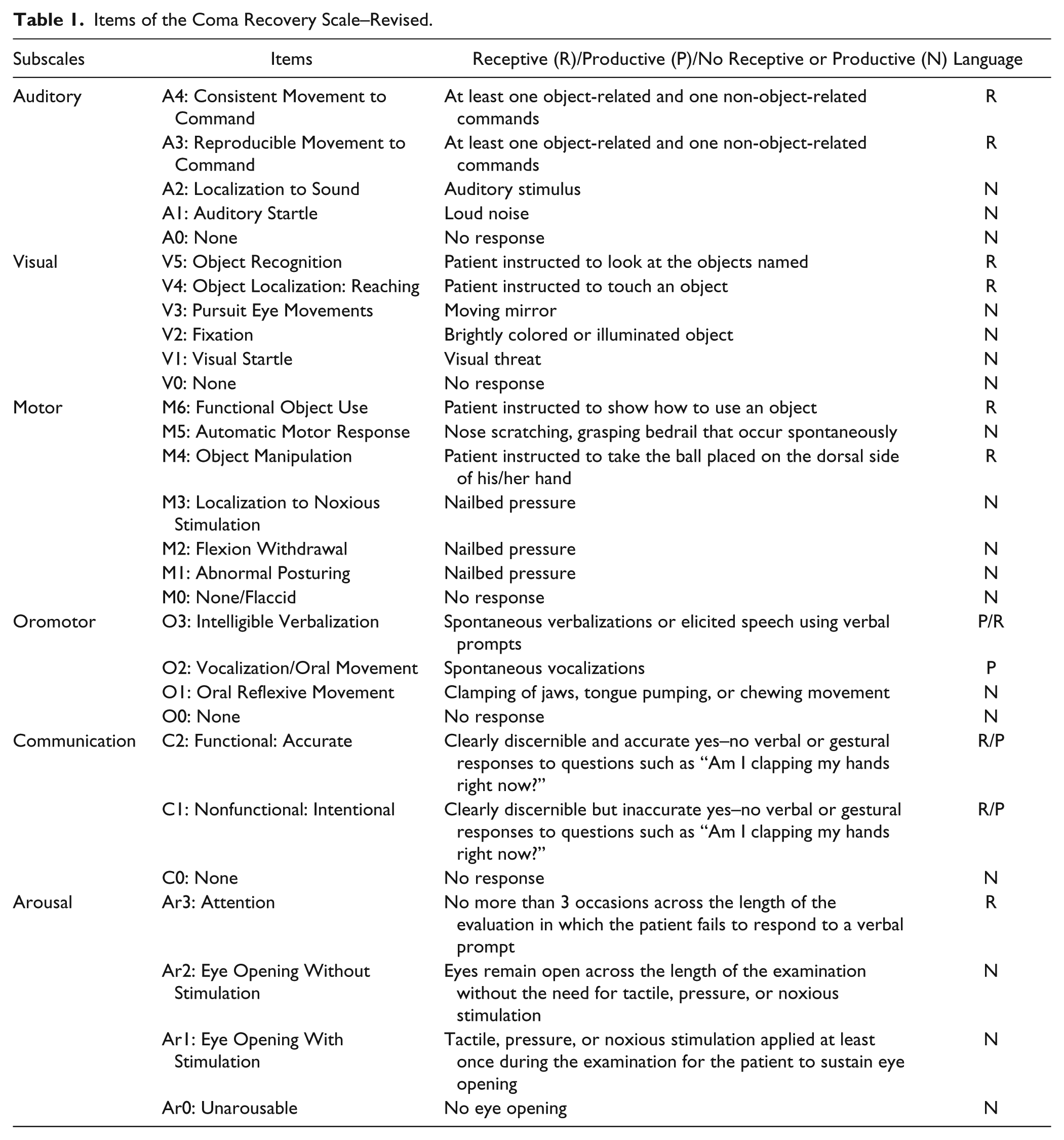
The CRS-R and aphasia scales (ie, the AAT and the ACL) were administered by 3 independent raters who were trained speech therapists. This prospective study was approved by the ethics committee of the Faculty of Medicine of the University of Liege. The study was performed in collaboration with the University Hospital of Cologne and the Rehanova neurorehabilitation center, Cologne-Merheim, Germany. Written informed consent was obtained from the patient or from his or her legal surrogate.
Statistical Analyses
The Mann–Whitney U test was used to investigate whether CRS-R total scores and subscores differed according to the type of aphasia. Chi-square tests were also used to detect which items of the CRS-R were failed most frequently. Spearman’s rank correlations were computed to test whether CRS-R total scores correlated with the patients’ age and the length of time poststroke. Results were considered significant at P value ≤.05.
Results
We assessed 30 patients with a diagnosis of aphasia. Three patients with a premorbid history of neurologic illness (ie, Alzheimer’s disease, Parkinson’s disease, and delirium, respectively), 2 patients with traumatic and epileptic etiologies, and 1 patient with a documented period of coma were excluded. Therefore, 24 stroke patients (mainly hemorrhagic; n = 20) were included in our analyses (17 males, 66.29 ± 12.94 years old, 3-111 days poststroke). Aphasic subtypes were as follows: global (n = 11), Broca (n = 4), Wernicke (n = 3), anomic (n = 4), and mixed (n = 2). Ten acute patients (<6 weeks poststroke) 21 were tested with the ACL and 14 subacute patients (≥6 weeks poststroke) were tested with the AAT before our CRS-R assessment. Three subacute and 3 acute patients could not complete either the AAT or the ACL because of poor performance on subtests assessing receptive and productive language and were consequently categorized as globally aphasic (eg, the subacute patients were not credited with any points on the Token Test of the AAT as they responded incorrectly to all 4 items). 15 Our patients did not present with any medical complication that led to a period of unconsciousness after the initial stroke, and none had disturbance in consciousness at the time of testing. Demographic data are provided in Table 2.
Demographic Data of Each Patient With Aphasia.
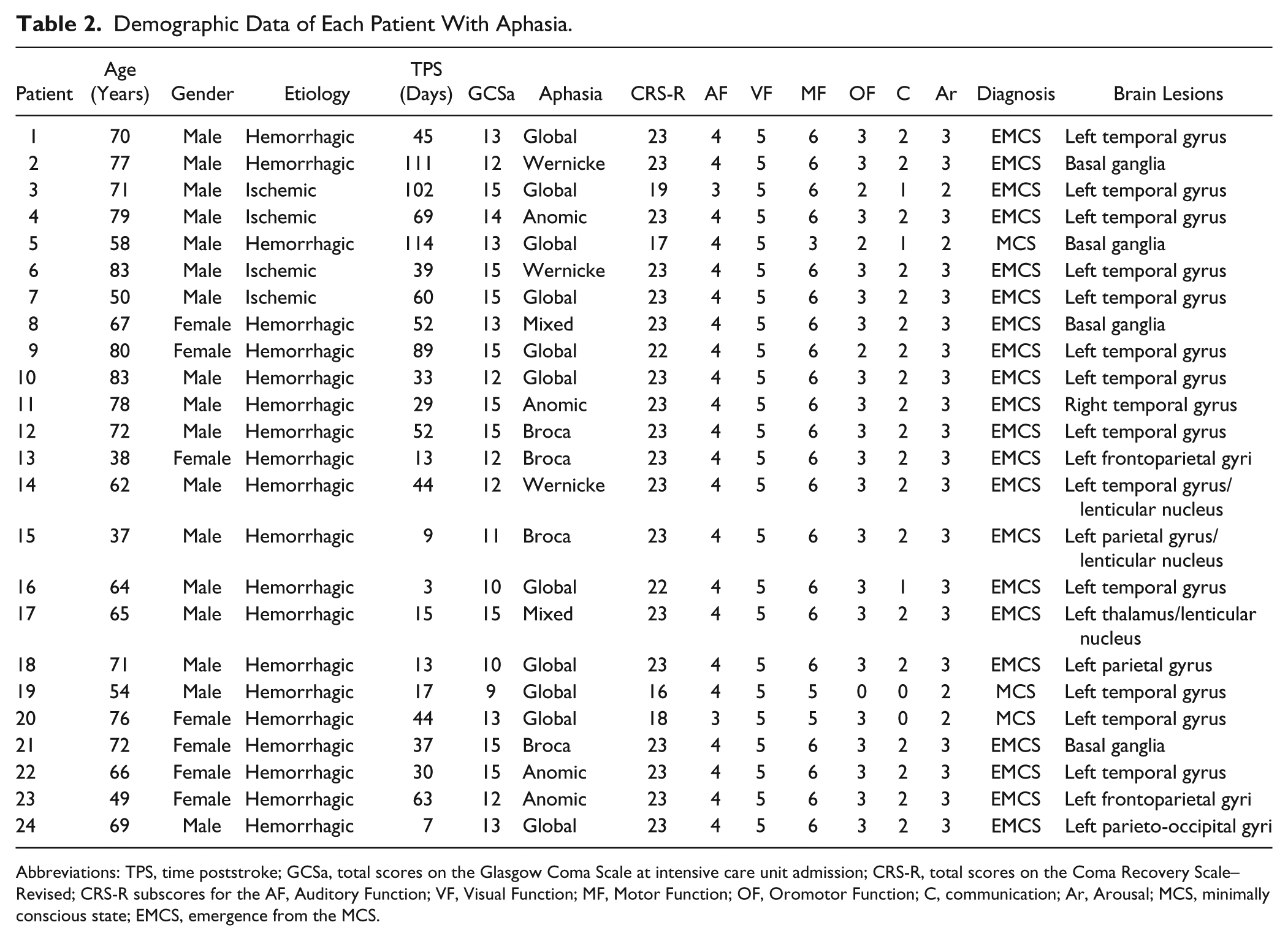
Abbreviations: TPS, time poststroke; GCSa, total scores on the Glasgow Coma Scale at intensive care unit admission; CRS-R, total scores on the Coma Recovery Scale–Revised; CRS-R subscores for the AF, Auditory Function; VF, Visual Function; MF, Motor Function; OF, Oromotor Function; C, communication; Ar, Arousal; MCS, minimally conscious state; EMCS, emergence from the MCS.
Of these 24 patients, 25% (n = 6) did not reach the maximal CRS-R score of 23. Total scores varied between 16 and 22 and all presented with global aphasia. Based on their performance on the CRS-R, 3 out of these 6 patients would have been diagnosed as being in MCS as they did not meet the criteria for emergence from MCS because of the absence of both functional communication and functional object use. 12 Using the Mann–Whitney U test, we compared CRS-R total scores and subscores obtained in patients diagnosed with or without a global aphasia. We obtained significant differences for the total scores (U = 32.5; P = .003) as well as for the motor (U = 52; P < .05), oromotor (U = 45.5; P = .02), communication (U = 39; P = .008), and arousal (U = 45.5; P = .02) subscores. We did not obtain any difference for the auditory (U = 58.5; P = .12) and visual subscores (U = 58.5; P = .3). Within the global aphasia group, χ2 tests were performed to assess which items of the CRS-R were the most frequently failed in patients incorrectly diagnosed as being in a MCS, as compared to patients correctly diagnosed as not being in MCS having emerged from the MCS. We obtained a significant difference for the following items: M6—functional object use (χ2 = 200; P < .001) and M4—object manipulation (χ2 = 57.81; P < .001), on the motor subscale; O3—intelligible verbalization (χ2 = 35.51; P < .001) and O2—vocalization (χ2 = 100.75; P < .001), on the oromotor subscale; C2—functional communication (χ2 = 120; P < .001) and C1—nonfunctional communication (χ2 = 100.75; P < .001), on the communication subscale; and Ar3—attention (χ2 = 57.81; P < .001), on the arousal subscale (see Figure 1). We did not obtain any significant correlation between CRS-R total scores and patients’ age (ρ = 0.06; P > .05) or the time poststroke (ρ = 0.20; P > .05).
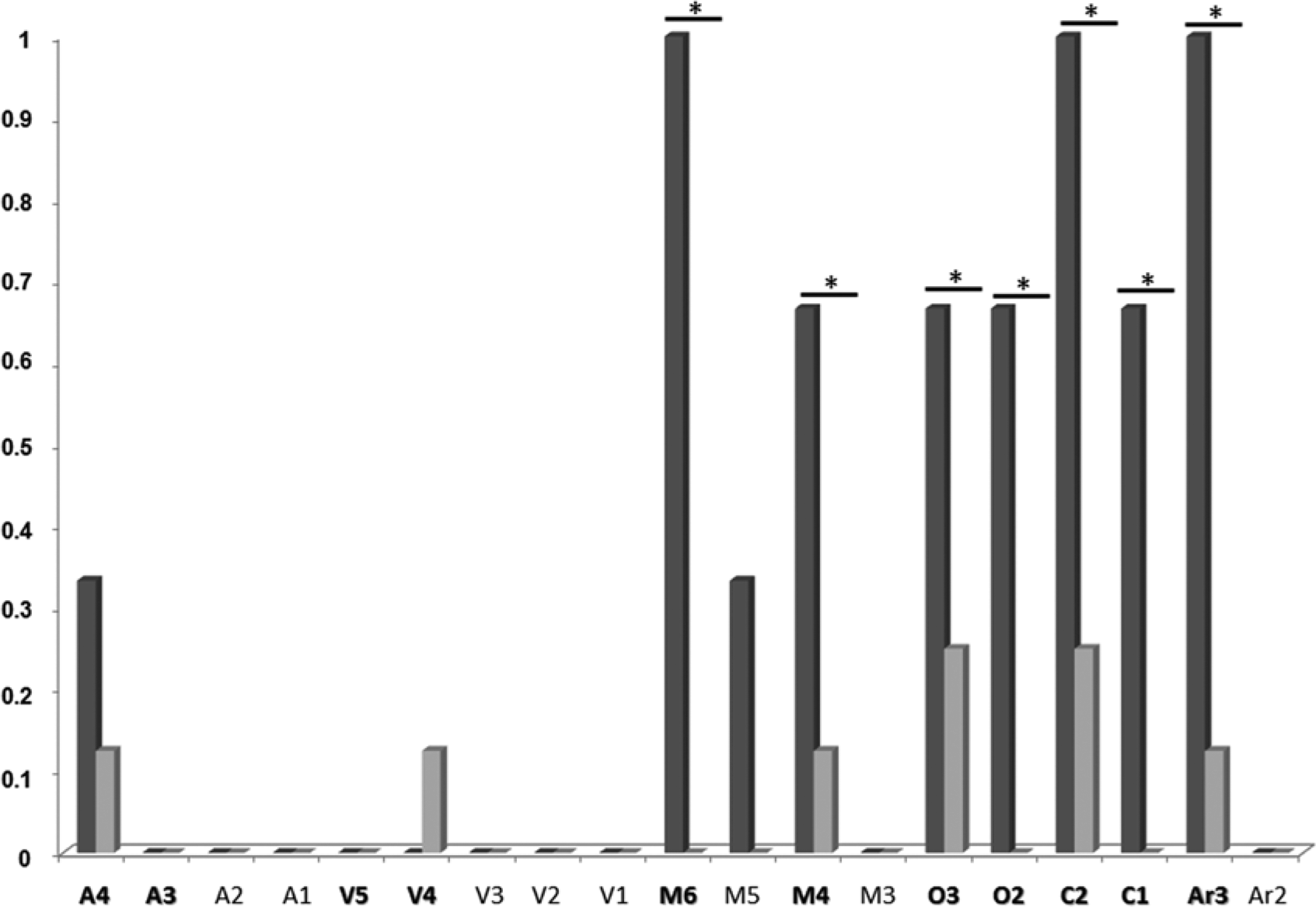
Proportion of patients with global aphasia who failed on Coma Recovery Scale–Revised (CRS-R) items.
Discussion
We found that 25% (6 of 24) of stroke patients with a diagnosis of aphasia and 54% (6 of 11) of patients with a diagnosis of global aphasia did not reach the maximal CRS-R total score of 23. An underestimation of level of consciousness was observed in 3 patients with global aphasia who could have been misdiagnosed as being in a MCS. That these cases are indeed instances of misdiagnosis is confirmed by the fact that while patients suffering from disorders of consciousness typically experience a period of coma following the injury, 22 this was not the case for any of our aphasic patients (see Glasgow Coma Scale scores in Table 2). Finally, lower CRS-R subscores were observed on the communication, motor, oromotor, and arousal subscales.
Functional (ie, consistent and reliable yes–no responses) and nonfunctional (ie, inconsistent and/or unreliable yes–no responses) communication were among the most frequently failed items of the CRS-R. The difficulty in successfully accomplishing these items may be explained by their morphosyntactic complexity. A positive rating on these items requires a reproducible verbal or gestural “yes” or “no” responses to personal or situational orientation (eg, “am I clapping my hands right now?”) questions. Language processing demands associated with yes–no questions are more complex than those required for the command-following items that appear on the auditory and visual subscales. The latter subscales require verbal comprehension (eg, “close your eyes” for consistent or reproducible movement to command and “look at the cup” for object recognition), but have no decision-making demands and, as such, require less complex language processing. Patients with global aphasia are known to respond to very simple commands even in presence of severe receptive language impairments. 23 Therefore, unsurprisingly, auditory and visual subscores were not significantly impacted in those patients. Besides the communication subscores, the motor subscores were also significantly lower. As for functional communication, the “functional object use” item requires more complex language processing as patients are asked to show how to use an object placed in their hand (eg, a brush or a cup). One could argue that a deficit in this item could reflect the presence of apraxia (ie, loss of the ability to execute learned purposeful movements), 24 which is frequently present after a stroke and often associated with aphasia. 25 Nevertheless, a failure on this item cannot exclusively be explained by such a deficit since all these patients also failed on the “functional communication” item, which also involves higher language processing. The complexity of the verbal command may therefore have played a significant role in the failure of this item. The intelligible verbalization and vocalization items of the oromotor subscale were also among the most frequently failed items, suggesting the presence of productive language impairments. Finally, arousal subscale scores were significantly lower with the “attention” item failed most frequently. While this item might seem unrelated to language processing, a positive rating is only given if there are no more than 3 occasions across the length of the assessment in which the patient fails to respond to a verbal prompt. Responding to verbal prompts may be difficult to achieve for patients with severe receptive language impairments or with global aphasia. A failure in such an item may therefore reflect language impairments rather than impaired attention or impaired arousal level. All patients were able to sustain eye opening during the entire assessment (ie, score of 2 on the arousal subscale), which is inconsistent with an arousal impairment.
To summarize, the behavioral pattern demonstrated by patients with language impairments seems to be associated with (a) a low performance on items with higher morphosyntactic complexity (ie, communication subscore of 0 and motor subscore of 5) and good performance on items with lower morphosyntactic complexity (ie, auditory subscore of 4 and visual subscore of 5), (b) a low performance on items related to productive language (ie, oromotor subscore of 0 or 1), and (c) impaired performance on the “attention” item of the arousal subscale (ie, arousal subscore of 2), probably because of difficulty comprehending verbal prompts. This prospective study suggests that a CRS-R total score of 16 or 17 may represent a potential threshold to detect and avoid misdiagnosis due to language impairments. Such a threshold will nevertheless have to be replicated in a bigger population of patients with global aphasia in order to test its accuracy. Additionally, our results suggest that both criteria for the emergence from the MCS (ie, functional communication and functional object use) are affected by the presence of language impairments. Thus, it might be important to redefine such criteria in order to avoid misdiagnosis and to accurately detect recovery of consciousness.
The interpretation of our results should nevertheless be nuanced. Indeed, if 25% (6 out of 24) of the patients with aphasia did not reach a maximal score of 23, it also means that 75% succeeded in reaching such a score. Moreover, all patients scoring less than 23 on the CRS-R were diagnosed with global aphasia, suggesting that only very severe language impairments are likely to confound the assessment of consciousness, and lead to misdiagnosis. Identifying performance patterns in CRS-R assessments in patients with known language impairment and no impairment in the level of consciousness proved to be an effective strategy. This approach allowed us to unambiguously evaluate how language impairments per se can affect responses to the CRS-R items. Studying the impact of language impairment directly in patients with disorders of consciousness would make it impossible to determine the root cause of failure in any CRS-R item as this could be secondary to either impairment in consciousness or to aphasia.
Future studies should also include additional testing to better understand the influence of other cognitive deficits on CRS-R performance. Even though standardized scales were administered to diagnose aphasia in our patients, the presence of other deficits such as impaired attention, impaired memory, or—more specifically—apraxia could also have had an impact on the patients’ behavioral responses. The frequent failure observed on the object manipulation item could well be related to an underlying apraxia. Adding such information would help flesh out the degree to which language or other cognitive impairments influence CRS-R performance. In parallel with these findings, more precise information with regard to lesion size and/or location should be collected. In our study, the diagnosis of aphasia was only based on standardized neuropsychological assessment using the AAT and ACL. It is important to determine whether brain lesions associated with aphasia are predictive of CRS-R performance.
Conclusion
The limited behavioral repertoire presented by severely brain-injured patients requires the use of highly sensitive and standardized behavioral scales, such as the CRS-R, for assessing level of consciousness.6,13 However, we showed that this assessment may be complicated by the co-occurrence of language impairments. One could argue that the solution would be to create a scale excluding language components. It is nevertheless difficult to assess consciousness without involving verbal comprehension. If the aim is to assess high-level responses (eg, communication or object recognition) that go beyond basic perceptual processing (eg, visual pursuit or localization to sound), verbal commands have to be used. Furthermore, the use of common tests for aphasia is often impossible when dealing with patients suffering from disorders of consciousness. Our results stress the importance of developing new tools, and identifying items on existing tools that can detect language impairments and differentiate these deficits from disturbance in consciousness. Such a development would help clinicians to better characterize a patient’s neurobehavioral profile and may affect rehabilitation. The development of a tool specifically designed to assess receptive and productive language would also inform the process of language recovery in MCS patients. It has been suggested that the language network is impaired in MCS− patients who may retain the same level of consciousness as MCS+ patients but are simply unable to express it to the same extent. 11 More investigations are needed in order to better understand remnant language abilities in this challenging population.
Footnotes
Authors’ Note
The data related to this study can be made available on request by the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Funds for Scientific Research.
