Abstract
Background. Despite progress made in understanding functional reorganization patterns underlying recovery in subacute aphasia, the relation between recovery and changes in white matter structure remains unclear. Objective. To investigate changes in dorsal and ventral language white matter tract integrity in relation to naming recovery in subacute poststroke aphasia. Methods. Ten participants with aphasia after left-hemisphere stroke underwent language testing and diffusion tensor imaging twice within 3 months post onset, with a 1-month interval between sessions. Deterministic tractography was used to bilaterally reconstruct the superior longitudinal fasciculus (SLF), inferior fronto-occipital fasciculus (IFOF), inferior longitudinal fasciculus (ILF), middle longitudinal fasciculus (MdLF), and uncinate fasciculus (UF). Per tract, the mean fractional anisotropy (FA) was extracted as a measure of microstructural integrity. Naming accuracy was assessed with the Boston Naming Test (BNT). Correlational analyses were performed to investigate the relationship between changes in FA values and change in BNT score. Results. A strong positive correlation was found between FA change in the right ILF within the ventral stream and change on the BNT (r = 0.91, P < .001). An increase in FA in the right ILF was associated with considerable improvement of naming accuracy (range BNT change score: 12-14), a reduction with limited improvement or slight deterioration. No significant correlations were found between change in naming accuracy and FA change in any of the other right or left ventral and dorsal language tracts. Conclusions. Naming recovery in subacute aphasia is associated with change in the integrity of the right ILF.
Introduction
Aphasia is a common consequence of damage to the language-dominant left hemisphere and is prevalent in about 30% of stroke survivors. 1 Neuroimaging has increased our understanding of the reorganization patterns underlying language recovery following stroke. Most studies have focused on the integrity and activity of cortical areas in language processing. More recently, however, the importance of the white matter tracts connecting these areas has been pointed out.2,3
Current neuroanatomical models of language connectivity assume that language is processed along dorsal and ventral pathways.4,5 The superior longitudinal fasciculus (SLF), comprising the arcuate fasciculus (AF), is a major white matter tract connecting temporo-parietal with frontal language cortices within the strongly left-dominant dorsal stream. 5 The ventral stream is more bilaterally organized 4 and includes the inferior fronto-occipital fasciculus (IFOF), inferior longitudinal fasciculus (ILF), middle longitudinal fasciculus (MdLF), and uncinate fasciculus (UF). 5 Whereas the dorsal stream is assumed to support phonological processing 5 and motor-articulatory computations, 6 the ventral stream is associated with lexical-semantic processing.4,5
Diffusion tensor imaging (DTI) is a noninvasive brain imaging technique providing measures of white matter structure. To date, only a handful of studies have used this technique to investigate changes in structural connectivity in aphasia recovery.7-12 Most of these studies were performed in the chronic stage7,9-11 and provided evidence of therapy-induced structural plasticity in the left7,10 or right 9 AF within the dorsal stream. Within the ventral stream, the role of only the ILF 11 and UF 10 has been investigated. Whereas language recovery following treatment of chronic aphasia was found to be supported by changes in the integrity of the left ILF, 11 no therapy-induced structural changes have been observed for the UF in either hemisphere. 10 Recently, Jang et al 8 examined structural changes in the left and right dorsal stream from the early to the chronic stage, reporting an association between increase in volume of the damaged left AF and recovery from aphasia over the first year after stroke. Conversely, Keser et al 12 revealed that an increase in the integrity of the right AF from the acute (<2 weeks) to the chronic (6-12 months) stage was associated with poorer naming recovery. To our knowledge, the relationship between changes in structural connectivity and language recovery in the first months post onset is as yet unclear. As most improvement in language function occurs in the subacute stage, a better understanding of the changes in white matter structure underlying language outcomes at this stage of stroke recovery is warranted.
The present observational study aimed to investigate recovery of naming within 3 months post onset in relation to changes in language white matter tract integrity. Whereas most DTI studies selected specific tracts in either the dorsal or the ventral stream, we evaluated changes in dorsal and ventral language streams in the left and right hemispheres. We studied a group of aphasic patients in the subacute stage who were scanned twice with a time interval of approximately 1 month.
Methods
Participants
Ten participants (7 men; mean age: 61.8 ± 7.8 years) with aphasia due to first-ever left-hemisphere stroke completed the study. They were all enrolled in a stroke rehabilitation program, including speech-language therapy (SLT). Individually tailored SLT was provided in individual and group sessions, and comprised intensive naming treatment, 13 cognitive-linguistic therapy, communicative treatment, and/or counseling and coaching of patients and proxies. Naming therapy comprised of 2 weeks of daily 45-minute individual sessions. 13 The naming therapy protocol was based on Cueing Hierarchy Therapy. 14 For detailed description of the therapy protocol, see Spielmann et al. 13
All participants were within 3 months post onset at the time of the study, had Dutch as their native language, were right-handed prior to stroke as assessed with the Edinburgh Handedness Inventory, 15 had normal hearing, and normal or corrected-to-normal vision. None of the participants had (1) severe aphasia (Shortened Token Test 16 [TT] correct score <9 and Aphasia Severity Rating Scale 17 [ASRS] score = 0-1), (2) minimal aphasia (Shortened TT correct score >28 and ASRS score = 4-5), (3) a history of other neurological, cognitive or psychiatric disorders, (4) substance abuse, or (5) any contraindications to magnetic resonance imaging (MRI). Baseline characteristics of the participants are listed in Table 1.
Demographic and Clinical Characteristics for Each Participant.
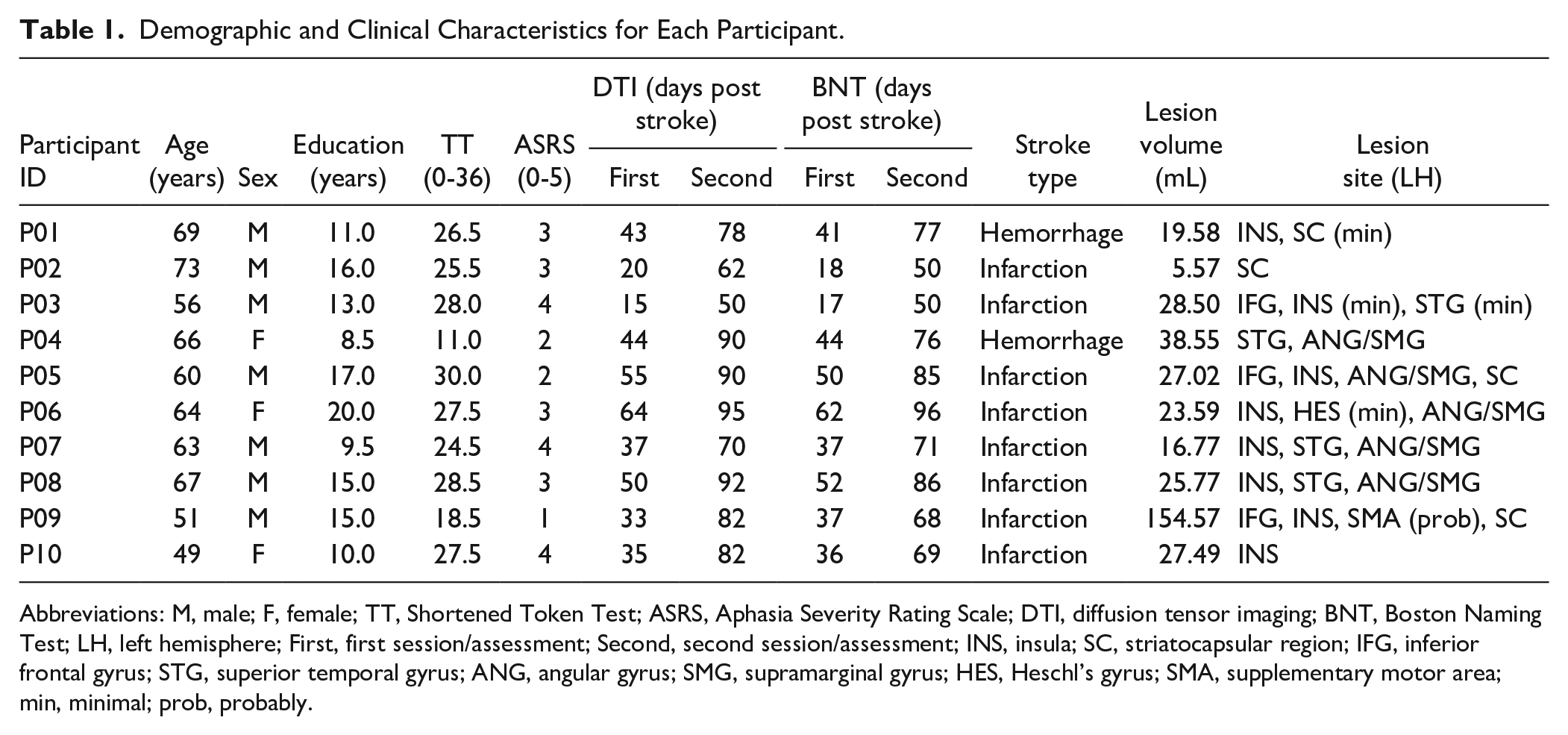
Abbreviations: M, male; F, female; TT, Shortened Token Test; ASRS, Aphasia Severity Rating Scale; DTI, diffusion tensor imaging; BNT, Boston Naming Test; LH, left hemisphere; First, first session/assessment; Second, second session/assessment; INS, insula; SC, striatocapsular region; IFG, inferior frontal gyrus; STG, superior temporal gyrus; ANG, angular gyrus; SMG, supramarginal gyrus; HES, Heschl’s gyrus; SMA, supplementary motor area; min, minimal; prob, probably.
The participants were a convenience subsample recruited from a multicenter randomized controlled trial (RCT) on transcranial direct current stimulation (tDCS) in subacute aphasia rehabilitation 13 that showed no effect of tDCS versus sham. 18 The participants in the present study were all recruited from the in- and outpatient rehabilitation clinic of Rijndam Rehabilitation, Rotterdam, The Netherlands, had no contraindications to MRI, agreed to participate in the neuroimaging part of the trial, and underwent DTI scanning twice. Figure 1 presents the flowchart of the participant selection.
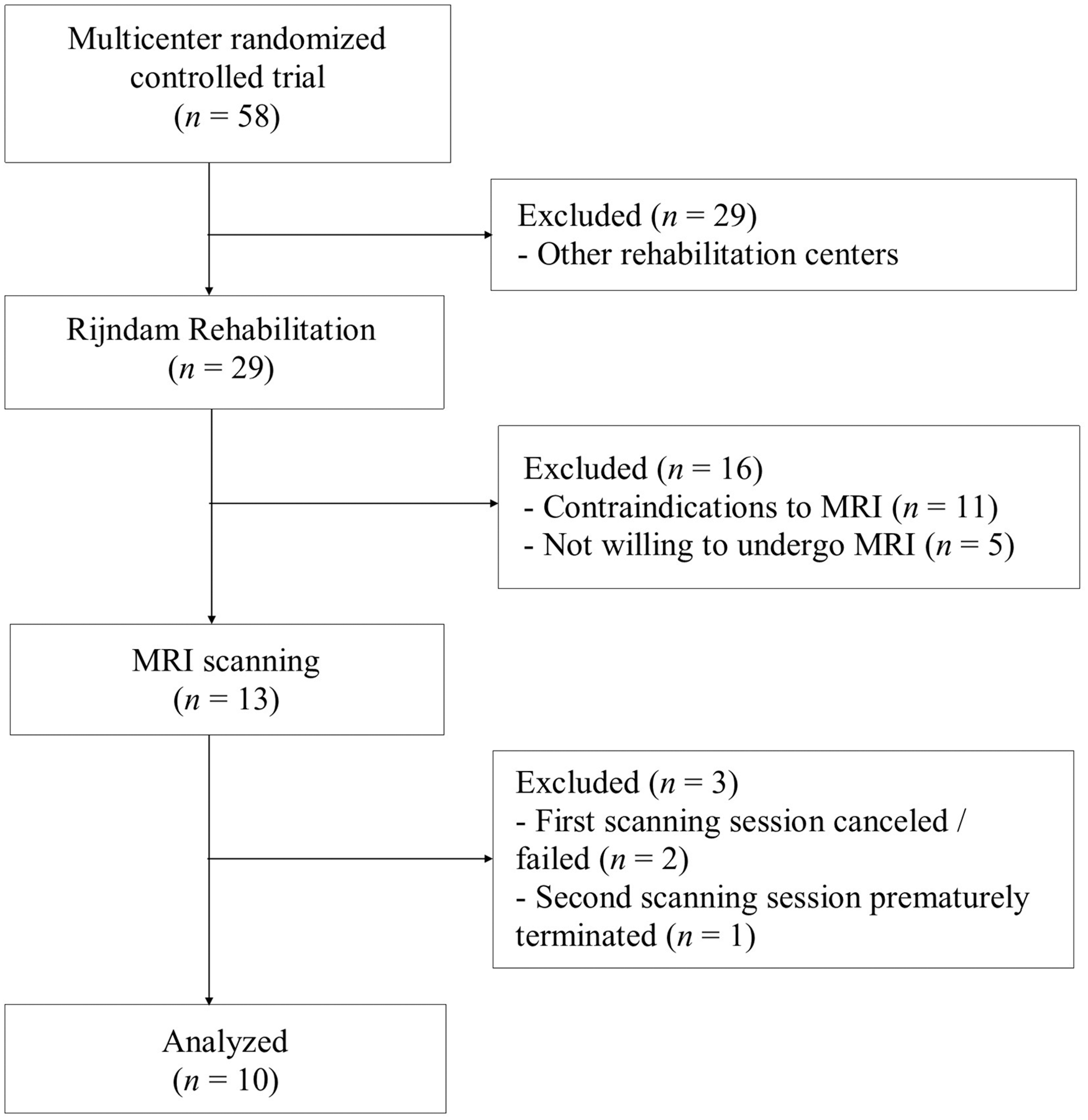
Flowchart of participant selection.
In the present study, we explored the relationship between naming recovery and changes in language white matter tract integrity irrespective of tDCS condition, because the RCT showed no effect of tDCS versus sham, neither immediately after the intervention nor at 6 months follow-up. 18 The decision to collapse the data was further justified by the fact that the participants in this study, when grouped into 2 groups (tDCS and sham), also showed no significant differences in naming recovery (Supplementary Table 1) or in change in tract integrity for any of the tracts of interest (Supplementary Table 2).
The study was approved by the Medical Ethics Committee of the Erasmus MC, University Medical Center Rotterdam, The Netherlands, and all participants provided written informed consent in accordance with the Declaration of Helsinki.
Language Measure
Naming accuracy was assessed with the 60-item Boston Naming Test 19 (BNT). In the Dutch version, item 57 (“trellis”) is not included, due to its low naming agreement in a Dutch norm group. 20 In deviation from the standard BNT administration procedure, 19 incorrect responses were not followed up with cues. The spontaneous responses were scored as either correct or incorrect, based on a list with target names and synonyms. 21 In accordance with the Dutch scoring system, 21 semantically correct responses with at least 2/3 of the phonemes produced correctly were scored correct. The language outcome measure was change in BNT scores. The first assessment was performed at a mean of 39.4 ± 14.1 days post onset (range: 17-62 days post onset) and the second assessment at a mean of 72.8 ± 14.8 days post onset (range: 50-96 days post onset).
Image Acquisition and Processing
The first scanning session was performed at a mean of 39.6 ± 15.0 days post onset (range: 15-64 days post onset) and the second scanning session at a mean of 79.1 ± 14.5 days post onset (range: 50-95 days post onset).
Images were acquired on the same whole-body 3.0-T Discovery MR750 system (GE Healthcare), using an 8-channel head coil, at Erasmus MC, University Medical Center Rotterdam. DTI data were acquired using an optimized single-shot spin-echo echo-planar imaging pulse sequence (repetition time [TR] = 7925 ms; echo time [TE] = 81.9 ms; field of view [FOV] = 240 mm; matrix size = 128 × 224; slice thickness = 2.5 mm; in-plane pixel size = 1.9 × 1.9 mm2; 59 contiguous sagittal slices; acquisition time = 4:53 minutes). Diffusion gradients were applied in 32 noncollinear directions (b = 1000 s/mm2) and 4 nondiffusion weighted images were acquired.
Pre- and postprocessing of the DTI data were performed using ExploreDTI (http://www.ExploreDTI.com) by an experienced rater (MV), who was blinded to the language test results of the participants. Preprocessing consisted of subject motion and eddy current correction. During this step, the b-matrix was corrected for the rotational component of subject motion. 22 The data quality summary as implemented in ExploreDTI and the native data were visually inspected to check for apparent motion artefacts. Subsequently, the diffusion tensors were estimated using nonlinear least squares fitting. Whole brain tractography was performed using a uniform 2 mm seed point resolution, and the following termination thresholds were applied: fractional anisotropy (FA) <0.2, angle <30°, and fiber tract length <50 mm. Deterministic tractography was used to reconstruct the dorsal and ventral tracts for both hemispheres in native space. Using the FA color-coded maps, regions of interest (ROIs) were drawn manually on the coronal and axial slices based on a priori anatomical knowledge. The ROIs were defined according to the ROI reconstruction protocols of Catani et al 23 for the SLF; of Wakana et al 24 for the IFOF, ILF, and UF; and of Makris et al 25 for the MdLF. When necessary, NOT ROIs were placed to exclude fibers from neighboring tracts. The ROIs were defined as follows:
For the SLF (Figure 2a), the axial slice where the anterior and posterior parts of the corpus callosum meet, was identified. Then, a single SEED ROI was drawn in the white matter region located in the posterior portion of the slice and lateral to the corona radiata. Only voxels with an anterior-posterior directionality were included in the ROI. 23
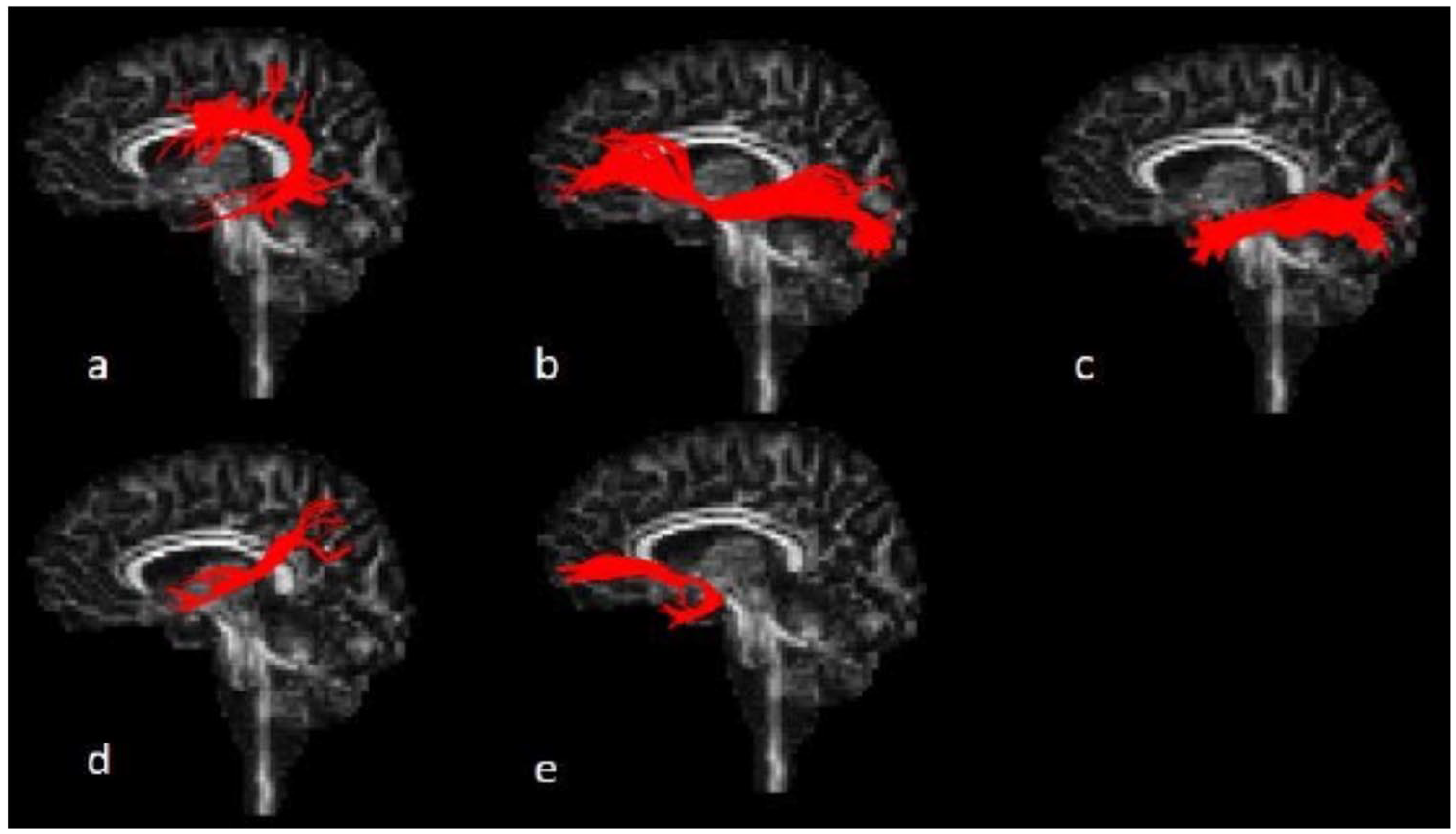
Reconstruction of left-hemisphere dorsal and ventral language white matter tracts using deterministic tractography: (a) superior longitudinal fasciculus (SLF); (b) inferior fronto-occipital fasciculus (IFOF); (c) inferior longitudinal fasciculus (ILF); (d) middle longitudinal fasciculus (MdLF); (e) uncinate fasciculus (UF); Figure courtesy of M. Verly (2017; doctoral thesis, p. 303).
For the IFOF (Figure 2b), the most anterior coronal slice where the left and right genu of the corpus callosum join, was identified. Then, on this slice, the whole hemisphere was selected using a SEED ROI. Following this, the midpoint between the posterior edge of the cingulum and the posterior edge of parieto-occipital sulcus was identified in the sagittal plane. An AND ROI was drawn around the full occipital lobe on the corresponding coronal slice. 24
For the ILF (Figure 2c), a coronal slice was selected at the posterior edge of the cingulum, identified on a sagittal slice. Then, a SEED ROI was selected comprising the entire hemisphere. The second ROI —an AND ROI— included the entire temporal lobe on the most posterior coronal slice where the temporal lobe is not connected to the frontal lobe. 24
For the MdLF (Figure 2d), SEED ROIs were drawn in the superior temporal gyrus white matter, inferior from the SLF fibers and superior to the ILF fibers, on 3 coronal slices. The first slice was the most anterior slice on which the middle cerebellar peduncle is still visible. The second slice was the most anterior slice where the corticospinal tract covers the middle cerebellar peduncle. The third slice was the most anterior slice showing both the transverse pontine fibers and the superior cerebellar peduncle. 25
For the UF (Figure 2e), the most anterior coronal slice on which the fornix is still visible was identified. Then, a SEED ROI was placed in the anterior-posterior white matter inferolateral from the anterior portion of the internal capsule. Finally, on the same slice, an AND ROI was drawn in the anterior-posterior white matter of the full temporal lobe. 24
Along each white matter tract, the mean value for FA was extracted as an indirect measure of microstructural integrity. FA describes the degree of directional dependence of water diffusion in fiber tracts, with values ranging from 0 (isotropic diffusion) to 1 (diffusion in one direction); higher values indicate a higher degree of directional organization and coherence of fiber tracts. FA is thought to reflect myelination and/or axonal density 26 and is a measure that is very sensitive to microstructural changes. 27
Statistical Analysis
Statistical analyses were performed using IBM Statistical Package for Social Sciences for Windows, version 23. To analyze changes in FA values for each of the white matter tracts from the first to the second scanning session and to analyze change in BNT scores from the first to the second assessment, we used paired-samples t tests. To investigate the relationship between changes in FA values and change in BNT score, we calculated Pearson’s correlations. The significance level was set at α = .05; probability values calculated for Pearson’s correlations were corrected for multiple comparisons (10×).
Results
Deterministic tractography was successful for all tracts in all participants, except for 1 tract; due to the lesion, the left SLF could not reliably be identified in 3 participants (P06, P08, and P10), and, therefore, analyses for the left SLF were performed on 7 participants.
At the group level, no significant changes in FA were found for any of the left and right hemisphere white matter tracts from the first to the second scanning session (Table 2). Figure 3 presents the individual tractography results at the first and second scanning session.
Mean (SD) Fractional Anisotropy for Each White Matter Tract at First and Second Scanning Sessions, and Comparisons Between Fractional Anisotropy at First and Second Scanning Sessions.
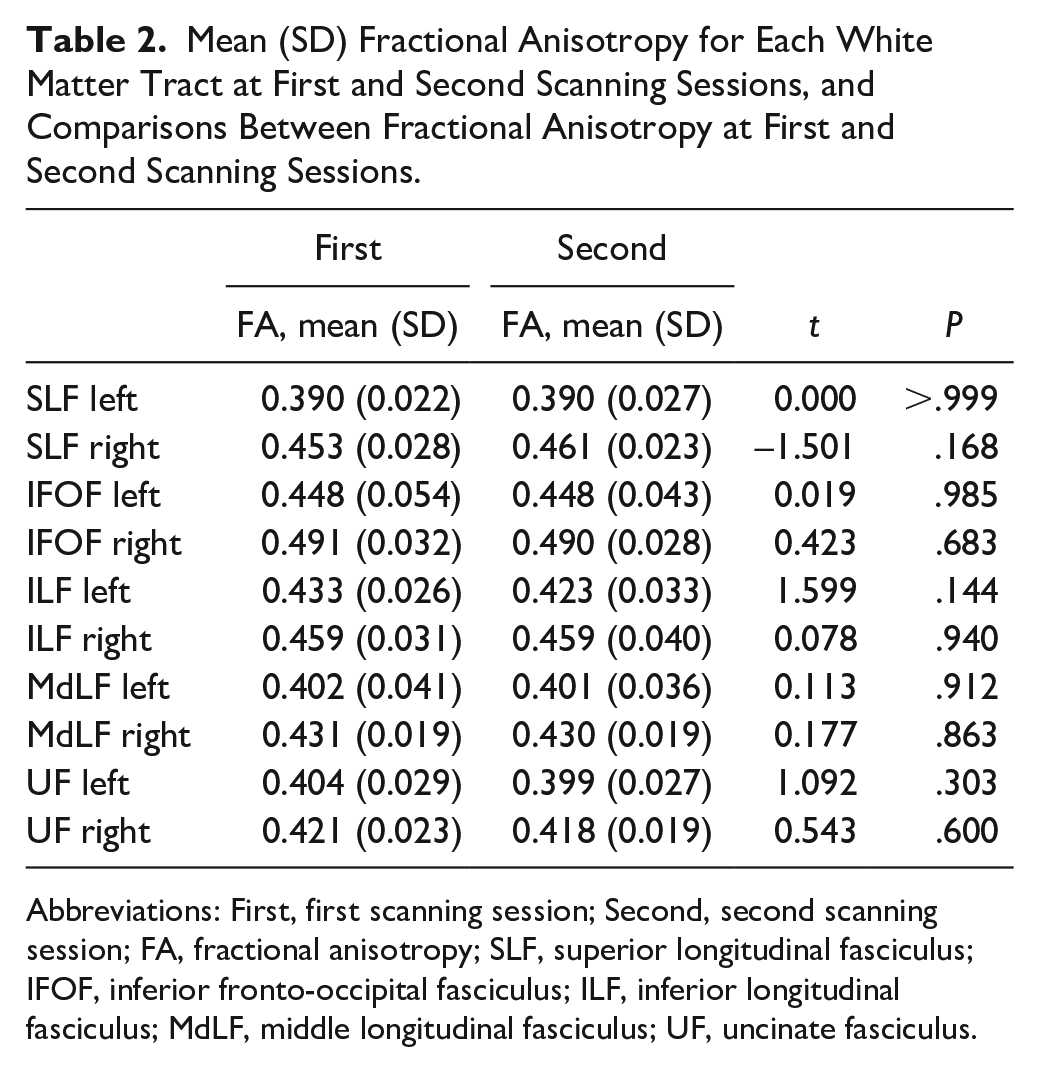
Abbreviations: First, first scanning session; Second, second scanning session; FA, fractional anisotropy; SLF, superior longitudinal fasciculus; IFOF, inferior fronto-occipital fasciculus; ILF, inferior longitudinal fasciculus; MdLF, middle longitudinal fasciculus; UF, uncinate fasciculus.
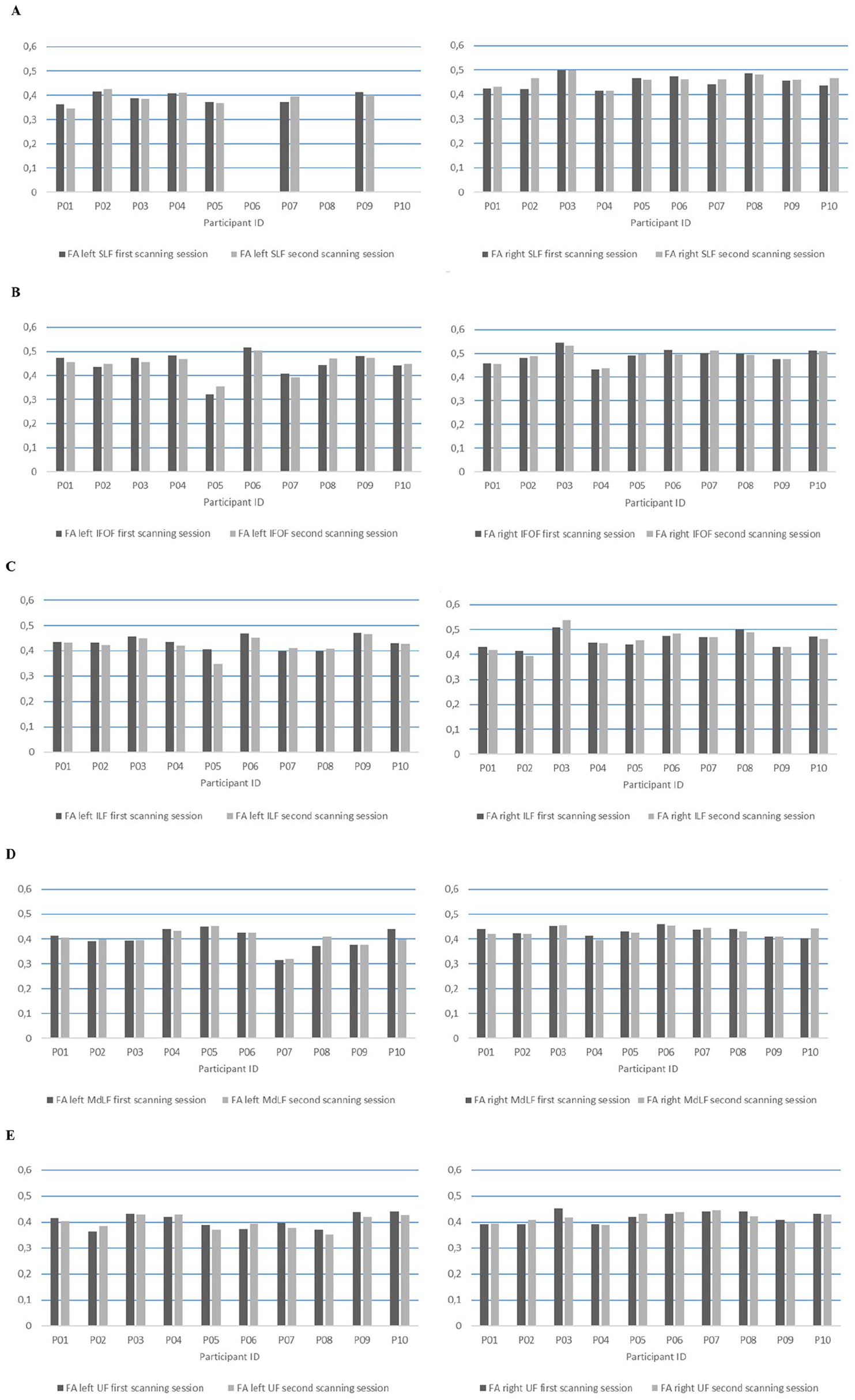
Fractional anisotropy (FA) values of the (A) left, respectively right, superior longitudinal fasciculus (SLF), (B) left, respectively right, inferior fronto-occipital fasciculus (IFOF), (C) left, respectively right, inferior longitudinal fasciculus (ILF), (D) left, respectively right, middle longitudinal fasciculus (MdLF), and (E) left, respectively right, uncinate fasciculus (UF) for each participant at first (dark gray) and second scanning session (light gray).
For the left SLF (Figure 3A, left panel), 2 participants (P02, P07) showed an increase in FA (FA change: 0.001 and 0.025, respectively), 4 participants (P01, P03, P05, P09) showed a decrease (range FA change: −0.017 to −0.004), and 1 participant (P04) showed no FA change. For the right SLF (Figure 3A, right panel), 6 participants (P01, P02, P03, P07, P09, P10) showed an increase (range FA change: 0.001 to 0.045), 3 participants (P05, P06, P08) showed a decrease (range FA change: −0.012 to −0.004), and 1 participant (P04) showed no FA change.
For the left IFOF (Figure 3B, left panel), 4 participants (P02, P05, P08, P10) showed an increase (range FA change: 0.008 to 0.032), and 6 participants (P01, P03, P04, P06, P07, P09) showed a decrease (range FA change: −0.017 to −0.007). For the right IFOF (Figure 3B, right panel), 4 participants (P02, P04, P05, P07) showed an increase (range FA change: 0.003 to 0.012), and 6 participants (P01, P03, P06, P08, P09, P10) showed a decrease (range FA change: −0.018 to −0.001).
For the left ILF (Figure 3C, left panel), 2 participants (P07, P08) showed an increase (FA change: 0.01 and 0.009, respectively), and 8 participants (P01, P02, P03, P04, P05, P06, P09, P10) showed a decrease (range FA change: −0.059 to −0.001). For the right ILF (Figure 3C, right panel), 3 participants (P03, P05, P06) showed an increase (range FA change: 0.011 to 0.028), and 7 participants (P01, P02, P04, P07, P08, P09, P10) showed a decrease (range FA change: −0.021 to −0.001).
For the left MdLF (Figure 3D, left panel), 6 participants (P02, P03, P05, P07, P08, P09) showed an increase (range FA change: 0.001 to 0.037), and 4 participants (P01, P04, P06, P10) showed a decrease (range FA change: −0.042 to −0.001). For the right MdLF (Figure 3D, right panel), 4 participants (P03, P07, P09, P10) showed an increase (range FA change: 0.001 to 0.039), 5 participants (P01, P04, P05, P06, P08) showed a decrease (range FA change: −0.019 to −0.005), and 1 participant (P02) showed no FA change.
For the left UF (Figure 3E, left panel), 3 participants (P02, P04, P06) showed an increase (range FA change: 0.008 to 0.021), and 7 participants (P01, P03, P05, P07, P08, P09, P10) showed a decrease (range FA change: −0.021 to −0.002). For the right UF (Figure 3E, right panel), 5 participants (P01, P02, P05, P06, P07) showed an increase (range FA change: 0.003 to 0.017), and 5 participants (P03, P04, P08, P09, P10) showed a decrease (range FA change: −0.036 to −0.003).
At the group level, the BNT score improved significantly from 34.8 (SD = 15.7) at the first assessment to 40.5 (SD = 15.1) at the second assessment, t(9) = −3.0, P = .015. Figure 4 presents the individual naming results at the first and second assessment. Six participants (P1, P3, P5, P6, P7, P9) showed improvement on the BNT (range BNT change score: 5 to 14), 2 participants (P4, P10) showed limited improvement (BNT change score: 2 and 1, respectively), and 2 participants (P2, P8) showed a slight decline (BNT change score: −3 and −1, respectively).
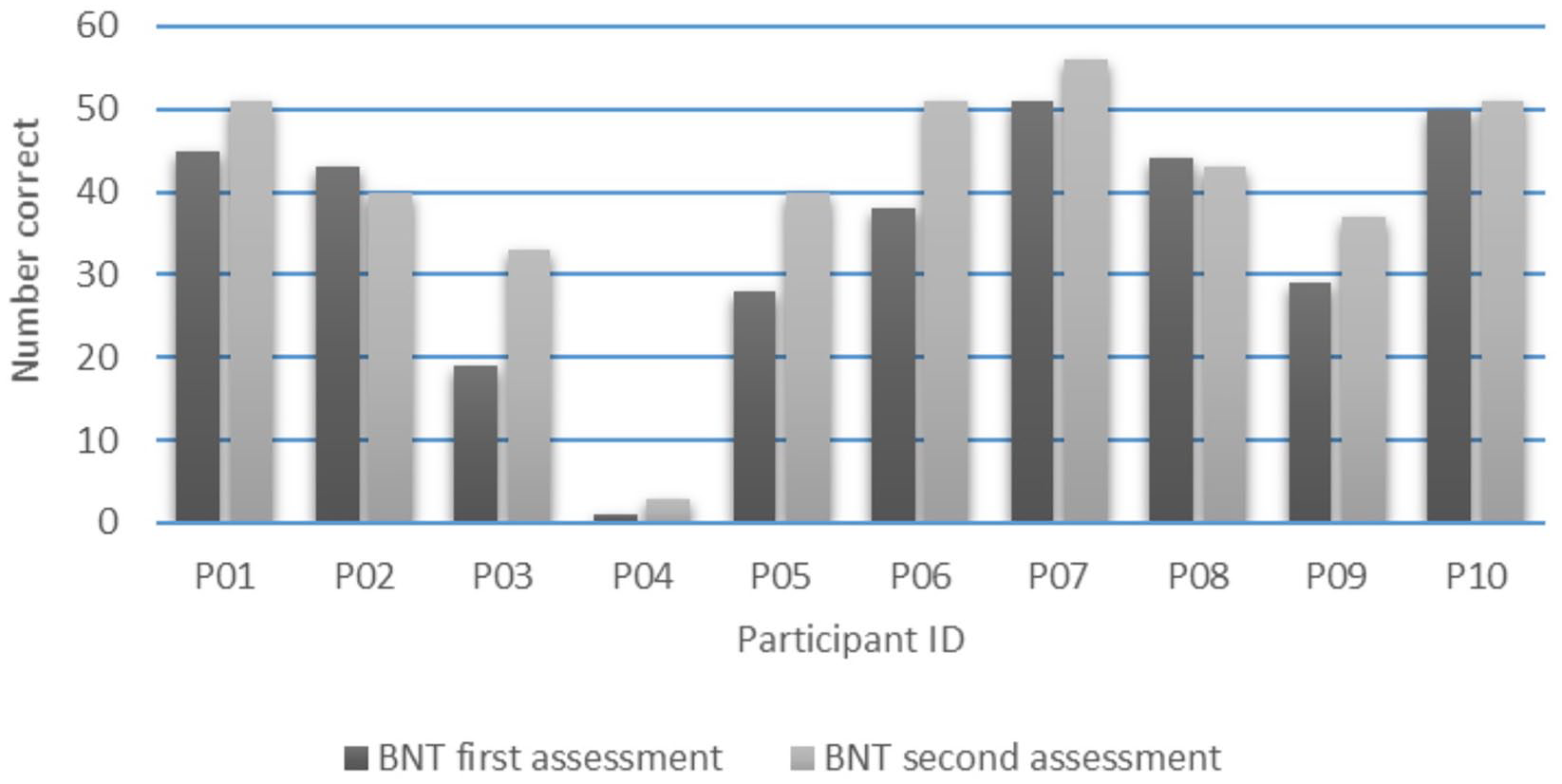
Boston Naming Test (BNT) scores for each participant at first (dark gray) and second assessment (light gray).
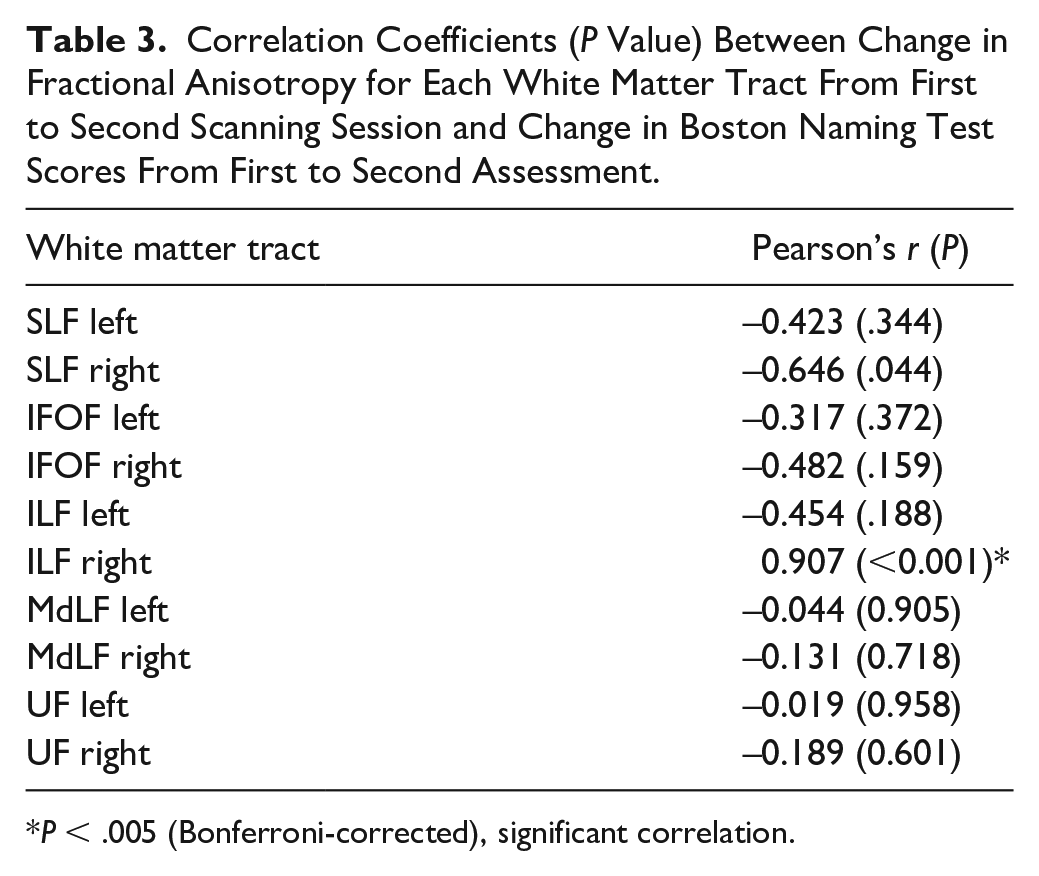
No significant correlations were found between FA change in the left hemisphere tracts and change in BNT score. For the right hemisphere tracts, a moderate negative correlation was found between FA change in the SLF and change in BNT score (r = −0.65, P = .044). However, this correlation did not survive a correction for multiple testing. A strong positive correlation was found between FA change in the ILF and change in BNT score (r = 0.91, P < .001; Figure 5); an increase in FA was associated with a considerable improvement on the BNT (range BNT change score: 12 to 14), a decrease in FA was related to a limited improvement or slight decline in BNT scores. Table 3 shows the results of the correlational analyses. See Supplementary Figure 1 for the scatterplots of the correlational results.
Correlation Coefficients (P Value) Between Change in Fractional Anisotropy for Each White Matter Tract From First to Second Scanning Session and Change in Boston Naming Test Scores From First to Second Assessment.
P < .005 (Bonferroni-corrected), significant correlation.
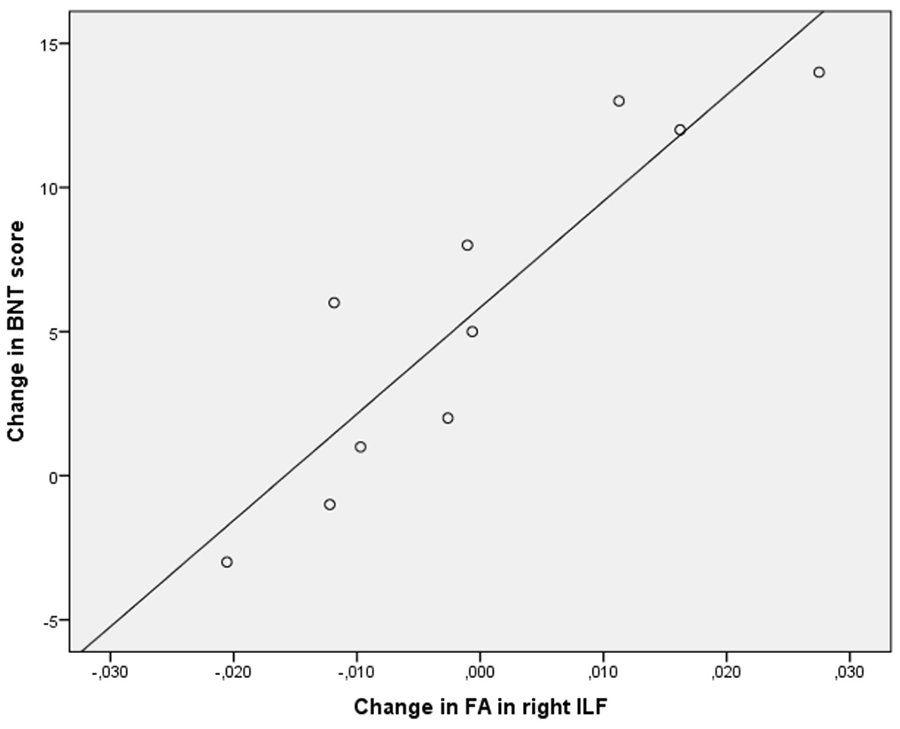
Scatterplot showing the correlation between change in fractional anisotropy (FA) in the right inferior longitudinal fasciculus (ILF) from first to second scanning session and change in Boston Naming Test (BNT) score from first to second assessment.
Discussion
In this observational study, we investigated changes in tract integrity in the left and right dorsal and ventral language streams in relation to naming recovery in subacute aphasia. Naming recovery appeared to be related to FA change in the ILF within the ventral stream in the unaffected right hemisphere, and unrelated to changes in both the ventral and dorsal streams in the left hemisphere.
The ILF is an important component of the ventral semantic system, transferring visual information from the occipital to the anterior temporal lobe, 5 and is proposed to be involved in visual object recognition5,28 and in mapping lexical labels onto object representations. 28 Our finding that the right ILF was involved in naming recovery is in line with a functional neuroimaging study in poststroke aphasia reporting activation of right hemisphere cortical areas that are connected by the ILF. 29 Abel et al 29 showed that during in-scanner picture naming cortical activation extended to the right hemisphere, including visual cortex and areas in the temporal lobe.
In contrast to our study, most previous DTI studies on aphasia recovery established a relationship between language recovery and changes in structural connectivity within the left7,8,10 or right dorsal stream.9,12 Although 4 of these 5 studies differed from our study in the tasks used to investigate aphasia recovery,7-10 3 of these studies reported on a relationship between structural changes and spoken language production,7,9,10 including naming7,10 or picture description. 9 The fourth study 8 found a relationship with a summary score for spoken language comprehension and production. Unlike our study, most of these studies examined therapy-induced white matter plasticity in the chronic stage7,9,10 and/or exclusively focused on the dorsal stream,7-9,12 which may explain the discrepancy in findings. It is worth noting that, although our finding regarding the right SLF did not reach significance after multiple testing correction, it is consistent with the observational study by Keser et al, 12 demonstrating that increased tract integrity within the right dorsal stream was associated with less improvement on the BNT.
One previous study has investigated the role of the left and right ILF in recovery of naming after stroke and reported an association between therapy-induced naming improvement in chronic aphasia and structural plasticity in the ILF in the lesioned left hemisphere. 11 While this study pointed to the left ILF, we found a relationship with structural changes in the right ILF. This discrepancy may be related to a difference in time after stroke for the participants. Whereas the study by McKinnon et al 11 was performed in patients who were at least 12 months poststroke, we studied a group of patients who were within 3 months post onset. Based on functional neuroimaging studies, it has been suggested that hemispheric contributions to aphasia recovery may change over time.30,31 In a longitudinal study, Saur et al 30 observed strongly reduced activation in both hemispheres in the first days post onset, followed by increased bilateral activation with recruitment of right homologue language areas during the next 2 weeks, and restoration of left-hemisphere language dominance after 4 to 12 months. Van de Sandt-Koenderman et al 31 reported that patients in the subacute stage of stroke (<3 months post onset) showed bilateral or right-lateralized language activation, whereas chronic patients (>12 months post onset) showed bilateral or left-lateralized language activation patterns.
Our findings underscore that the bilateral representation of the ventral stream enables the unaffected right hemisphere to compensate for compromised language function in the left hemisphere. 4 This is in line with functional neuroimaging studies, reporting that the right hemisphere may play a role in compensating for language deficits in the first months after stroke.32,33 Several studies have suggested that right hemisphere recruitment may depend on lesion volume32-34 and lesion site.32-35 In the present study, the involvement of the right ILF seems to be independent of lesion volume, as the 3 participants (P3, P5, P6) who showed considerable naming improvement and an increase in FA had similar lesion volumes as 2 (P8, P10) of the 3 participants who showed a decrease in FA and limited improvement or a slight decline in naming accuracy. The participant (P2) who showed the largest FA decrease and the largest deterioration of naming accuracy had the smallest lesion volume of all participants. Whether the relationship between naming recovery and change in right ILF integrity was driven by lesion site remains unclear given the heterogeneous lesion profiles of the participants.
Interestingly, the changes in the right ILF found in the present study appeared to go in 2 directions. Whereas an increase in FA was associated with a considerable improvement on the BNT, a decrease in FA was related to a limited improvement or even a slight decline in BNT scores. A reduction in tract integrity in the unaffected right hemisphere was an unexpected finding. However, Ivanova et al 36 observed a lower mean FA in several right-hemisphere white matter tracts, including the ILF, in chronic aphasic patients too; a finding thought to be a reflection of remote neurodegeneration effects after stroke. Our finding is also in line with a study in a general population of stroke patients, reporting a relationship between reduced white matter integrity in the nonlesioned hemisphere at 3 months post onset, which was attributed to demyelination, and poor cognitive recovery. 37 Hence, our finding that some participants showed a decrease versus others showing an increase in integrity of the right ILF suggests that there might be 2 different underlying neural mechanisms at play: remote degeneration with a reduction in tract integrity and lack of recovery in some participants, and an increase in integrity in others, which might reflect adaptive white matter plasticity in the unaffected hemisphere. White matter plasticity underlying behavioral improvements has also been found in the healthy adult brain and has been suggested to reflect activity-dependent myelination.38,39 It remains unknown what drives remote degeneration in some participants and white matter plasticity in others.
Within the ventral stream, none of the other language white matter tracts examined, including the UF, appeared to play a role in subacute naming recovery. The absence of results for the UF are in accordance with the results found by Van Hees et al 10 in chronic aphasia, showing no structural changes in the left or the right UF following naming treatment.
This study has several limitations. A first limitation is the small sample size. Our results, providing the first indication of a role of the right ventral stream in early naming recovery, need to be confirmed with larger samples. Another limitation is that the participants were predominantly male (7 men, 3 women). Given the evidence for sex differences in white matter tracts related to language,40-42 this imbalance might have introduced bias. This is particularly prominent for the analyses of the left SLF. Due to the presence of the lesion in the SLF, the left SLF could not be reconstructed in 3 participants, which excluded 2 of the 3 female participants’ data. A third limitation is that the time post onset at which the behavioral and tractography data were obtained varied across individuals and between measurements. These differences in the timing of data collection hamper the interpretation of the structure-function relationships, as both aphasia recovery 43 and changes in structural integrity 3 may occur rapidly in the first months after stroke. A fourth limitation is that some of the participants received tDCS, whereas others did not, which might potentially have influenced the behavioral and neural results at the individual level. However, when grouped into 2 groups (tDCS and sham), the participants showed no significant differences in naming recovery (Supplementary Table 1) or in change in tract integrity for any of the tracts of interest (Supplementary Table 2). Furthermore, when examining the scatterplots showing the correlation between FA change in each of the tracts and change in BNT score with visualization of experimental condition (Supplementary Figure 1), we found no evidence of confounding by tDCS, as the participants assigned to either the experimental or control condition were randomly distributed across each of the plots. Last, DTI tractography provides only indirect measures of white matter tissue properties, and, hence, uncertainty exists about the correspondence between DTI measures and underlying neuroanatomical factors. 44
In conclusion, the present findings provide preliminary evidence of a relationship between change in white matter integrity in the right ventral stream and naming recovery in subacute aphasia. Whereas an increase in the integrity of the right ILF was associated with a considerable improvement of naming accuracy, a reduction was related to a limited improvement or a slight deterioration of naming accuracy. The present findings add to the understanding of the neural mechanisms underlying recovery from aphasia and highlight the need to examine changes in the bilateral ventral white matter pathways in studies investigating recovery of language after stroke.
Supplemental Material
Supplementary_material_Blom-Sminketal_revision2020_cleancopy – Supplemental material for Change in Right Inferior Longitudinal Fasciculus Integrity Is Associated With Naming Recovery in Subacute Poststroke Aphasia
Supplemental material, Supplementary_material_Blom-Sminketal_revision2020_cleancopy for Change in Right Inferior Longitudinal Fasciculus Integrity Is Associated With Naming Recovery in Subacute Poststroke Aphasia by Marieke Blom-Smink, Marjolein Verly, Kerstin Spielmann, Marion Smits, Gerard M. Ribbers and Mieke W. M. E. van de Sandt-Koenderman in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank the MRI technologists for their contribution to the project. Special thanks are given to the participants for their commitment to the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Erasmus MC Cost-Effectiveness Research (Grant Number: 2012-12112) and the Dutch Brain Foundation (Grant Number: 2013(1)-97).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
