Abstract
Background
Accumulating evidence suggests that motor performance is affected by the cognitive control abilities of the subject. Performance in motor tasks in populations with cognitive deficits such as older adults and subjects with stroke is therefore expected to deteriorate. The goal of this study is to investigate the relationship between cognitive impairments and motor control and learning impairments in a visuomotor adaptation task in subjects with stroke.
Methods
Twenty-seven post-stroke, 31 age matched controls, and 30 young control subjects completed a sensorimotor adaptation task composed of 2 adaptation blocks separated by a washout block. Explicit learning was assessed by cueing subjects to suppress their strategy. Cognitive assessment was conducted using the Montreal Cognitive Assessment (MoCA) and a verbal learning test. Subjects with stroke performed the task with their unaffected arm.
Results
Despite the cognitive deterioration, the adaptation and savings of the stroke group and age matched controls were comparable. Adaptation and savings were smaller with respect to the young subjects. Savings was associated with a significant improvement in the explicit component across blocks. Finally, the explicit enhancement between blocks was significantly correlated with the MoCA scores in the stroke group and with the results of the verbal learning test in the young controls.
Conclusion
The lack of stroke-induced attenuation on adaptation, despite a correlation between cognitive abilities and explicit learning in adaptation, suggests that subjects with stroke have enough cognitive resources to support sensorimotor adaptation. The availability of cognitive resources for motor learning following brain damage could be utilized in the rehabilitation process.
Introduction
Cognitive control abilities can account for significant proportions of learning and retention in complex motor tasks. 1 These effects were quantified in sensorimotor adaptation tasks, where spatial and working memory and aiming strategy were shown to contribute to learning2,3 and retention.4 -10 In fact, a dominant research effort aims at studying the contribution of explicit and implicit learning mechanisms to motor learning through the examination of strategic aiming as a measure of explicit learning, and corrections that are not amenable to suppression or report as a measure of implicit learning.2,3,7,11 Another way to emphasize the difference between these components is to measure the response of the subjects to a repeated perturbation, that enhances explicit learning and attenuates implicit learning. 12 Studying explicit and implicit learning processes in clinical groups may expand the knowledge about the contribution of cognitive abilities to motor learning.
Old adults with cognitive decline show deficits in motor learning that are associated with their cognitive impairments.13 -15 For example, older adults show both an age-related decrease in the speed of processing, memory, reasoning and spatial working memory, and deficits in adaptation learning,16,17 sequence learning, 18 and balance control in dual task conditions. 19 Furthermore, in visuomotor reaching adaptation tasks, age-related deficits were shown to be associated with impairments in the explicit learning component but not with implicit adaptation. 20
Cognitive control impairments, especially in fluid abilities, are a common outcome of stroke21 -29 and are likely to contribute to impairments in motor learning.15,30,31 In sensorimotor adaptation, subjects with stroke show impaired adaption to force-field perturbations 31 and to visuomotor perturbations. 32 Furthermore, even though subjects with stroke succeeded in adapting to a gain perturbation to 1 target, they performed poorly when needed to generalize to others in comparison to control subjects. 33 Sensorimotor adaptation studies aiming to differentiate between the contribution of explicit learning and implicit processes have not been conducted in subjects with stroke.
Assessing the contribution of cognitive impairments to motor control and learning is challenging in clinical groups due to the prevalence of motor impairments in these groups. We attempted to isolate the effect of cognitive impairments on motor learning by taking advantage of the lateralization of motor impairment and focusing on the performance of the unaffected arm. Identifying the contributions of cognitive impairments to motor learning is important since this contribution is likely to increase when motor control resources are reduced, 34 and to play an important role in the response to rehabilitative treatments and in the recovery of motor functions following brain damage.
The aim of this study is to examine strategic (explicit) and implicit adaptation to a visuomotor perturbation and retention of learning in subjects with chronic stroke, age matched healthy adults, and a group of young healthy adult subjects. We hypothesize that subjects with stroke will show a greater level of impairment in adaptation than age matched control subjects due to their increased cognitive impairments. These effects will be driven by a weaker explicit adaptation and readaptation. Additionally, age matched control and subjects with stroke will show impairments in adaptation compared to the young control subjects. Finally, cognitive abilities in subjects with stroke and age matched controls will be correlated with their strategic (explicit) adaptation and with explicit enhancement. Based on recent results that show that implicit adaptation learning is intact in elderly subjects, 20 we hypothesize that the stroke group will not show impairments in implicit adaptation compared to the 2 control groups.
Methods
Subjects
Twenty-seven chronic first-ever subjects with stroke (aged between 55 and 75 years, mean age: 65.07, SD 5.96 years, 9 females; see Table 1 for demographic details for the stroke group), 31 age matched (AM) control subjects (aged between 55 and 75 years, mean age: 67.26, SD 5.22 years, 18 females), and 30 young control subjects (aged between 20 and 30 years, mean age: 25.17, SD 1.26 years, 16 females) participated in the study. Subjects with stroke were recruited at Adi Negev Nahalat Eran rehabilitation center, Israel (convenience sample). AM control and young control subjects were recruited at Ben-Gurion University of the Negev. The study was approved by the Ben-Gurion institutional Helsinki committee (for healthy subjects) and by Regional Ethical Review Board at Sheba Medical Center, Israel (Approval Number 6218-19-SMC; for subjects with stroke). All subjects signed a consent form before performing the experiment. All subjects were paid for their participation. Subjects with stroke had a motor and sensory evaluation before the beginning of the experiment (motor and sensory upper extremity Fugl-meyer assessment (FMA) and the Action Research Arm Test (ARAT)). Subjects with stroke did the experiment with their unaffected arm to differentiate the cognitive aspect of motor learning from the sensorimotor deficits of the damage (See elaboration in Discussion). The unaffected arm received a full motor and sensory score in the FMA and ARAT assessment in all the subjects with stroke. Healthy subjects (adult and young controls) were instructed to use their dominant or non-dominant arm to control for hand dominance in the stroke group (20/27 stroke, 20/31 AM control, and 15/30 young control subjects performed the task with their non-dominant arm).
Subjects With Stroke Information.
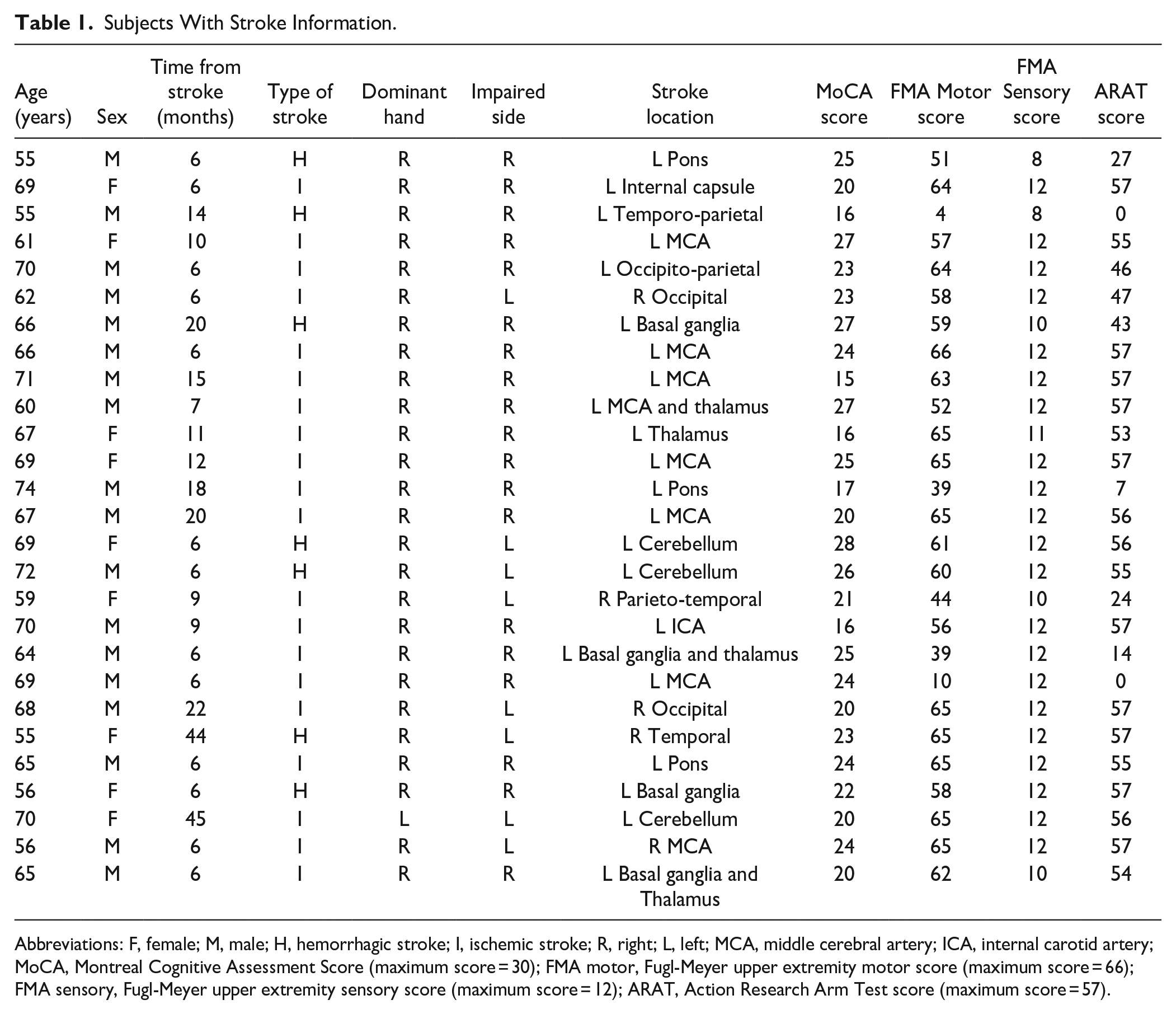
Abbreviations: F, female; M, male; H, hemorrhagic stroke; I, ischemic stroke; R, right; L, left; MCA, middle cerebral artery; ICA, internal carotid artery; MoCA, Montreal Cognitive Assessment Score (maximum score = 30); FMA motor, Fugl-Meyer upper extremity motor score (maximum score = 66); FMA sensory, Fugl-Meyer upper extremity sensory score (maximum score = 12); ARAT, Action Research Arm Test score (maximum score = 57).
Inclusion Criteria
Ability to give informed consent and understand the instructions of the tasks. Ages: Stroke and AM control subjects—55 to 75 years. Young controls’ group—20 to 40 years. Subjects with stroke with a first-ever stroke (ischemic or hemorrhagic) confirmed by CT or MRI or with a recurrent stroke, if their cognitive abilities and motor control were normal before the incidence. Time after stroke onset ≥6 months (M = 12.37 months, SD = 10.57). Exclusion criteria (All groups): history of physical or neurological condition that interferes with study procedures or assessment of motor function (eg, severe arthritis, severe neuropathy, and Parkinson’s disease). Subjects with stroke were independent before the incident without documented motor or cognitive impairments. All 87 subjects were included in the final analyses. All the subjects with stroke were enrolled in the study were after a first stroke (no recurrent strokes in the sample).
Experimental Design
The study was composed of cognitive assessments and a visuomotor adaptation session. Cognitive abilities for the AM controls and subjects with stroke were assessed using Montreal Cognitive Assessment (MoCA). 35 This test is a clinical cognitive screening tool that shows high sensitivity and specificity in identifying poststroke cognitive impairment in both ischemic and hemorrhagic strokes. 36 This 1-page assessment, tests multiple cognitive domains: visuospatial perception (5 points), naming (3 points), attention (6 points), language (3 points), abstraction (2 points), short-term memory (5 points), and orientation (6 points). The maximal score in MoCA is 30 points. 10 out of the 31 control healthy subjects did not go through cognitive assessment due to technical constraints that were not related to their cognitive abilities. In the young control group, memory was assessed using a modification of the California verbal learning test II 37 ; Young control subjects listened to a list of words twice and had to freely recall the list after each repetition. After performing the adaptation task (approximately 30 minutes) they were asked to recall the list again. Each subject had 3 scores—the number of recalled words in every attempt. We chose to use this standardized memory test because it is comparable to the memory component in the MoCA, but has more items, thereby less sensitive to a ceiling effect.
For the visuomotor adaptation task, the subject was seated at a custom-made table that housed a horizontally mounted LCD screen (22″, Dell), positioned 20 cm above a digitizing tablet (22″, CINTIQ, Wacom 22HD). The screen occluded the tablet. Stimuli were projected onto the LCD screen. The experimental software was custom written in MATLAB (Mathworks Inc, Natick MA), using cogent toolbox extensions (Cogent 2000). Subjects were seated in front of a table and were instructed to make center-out planar reaching movements by sliding an air hockey “paddle” with a custom-made handle for the tablet’s stylus. Movement trajectories were recorded at 60 Hz.
Our visuomotor rotation paradigm was based primarily on experiments E1a of Vandevoorde and Orban de Xivry 20 and was designed to test explicit and implicit adaptation. The magnitude of perturbation was 40 degrees (clockwise; Figure 1A). The experiment consisted of a training block (9 trials), a baseline block (18 trials), an adaptation block (84 trials), a first washout (99 trials), a second adaptation block (readaptation, 84 trials), and a second washout (81 trials; Figure 1B). During baseline and washout blocks, we used 9 targets, separated by 40°. During the adaptation blocks, we used 2 targets from the previous 9 (Figure 1A). These targets were separated by 120 degrees. Targets were presented 9.25 cm from the center of the screen and were shown in blue. To start the trial subjects had to move the cursor into a black ring presented at the center of the screen. When entering the centered ring, a “beep” sound was played, marking the beginning of the trial. From the beginning of the trial the subject had 2.5 seconds to exit the central ring (begin the reaching movement). From the moment the subject left the central ring, she had 1 second to complete the reaching movement. If the subject did not start the trial after 2.5 seconds, the trial was terminated and the next 1 began. The cursor disappeared when the cursor left the central ring and reappeared at the end of the trial. If the cursor crossed the radial position of the target (with respect to the center of the screen), the trial was terminated, and the cursor was presented at that point. If the cursor did not reach the radial position of the target, the cursor was presented in its final position. After the cursor was presented, the subject had to bring it back to the central ring to initiate the next trail. The trial ended when the subject crossed the radial position of the target or when the duration of the trial was reached. The end of the trial was marked by a change of color of the target and by the reappearance of the cursor. Subjects received feedback only at the end of the trial. The feedback was a change of the target color, and a pleasant sound for successful trials. If the subject did not reach the target, the target turned red, while if the subject did reach the target, the target turned green, accompanied by a pleasant sound. No breaks were given between blocks. In the baseline and in the washout blocks the trials were without perturbation and the cursor remained a blue dot. In the adaptation blocks there were 2 kind of trials—cued+ trials (trials with a visuomotor rotation) and cued– trials (trials without a rotation; Figure 1A). In the adaptation blocks the cursor changed from a blue dot (cued– trials) to a pink triangle (cued+ trials) and vice versa. The pink triangle indicated the presence of a special trial (cued+) while the blue dot indicated a regular trial (cued−). In each adaptation block, there were 8 cued– trials (without a rotation) that appeared in fixed trial numbers across subjects (trials 20, 33, 36, 40, 46, 52, 68, and 72). To increase the awareness of subjects to the cue changes, the cueing of the trials and the feedback were based on experiment E1b of Vandevoorde and Orban de Xivry. 20 The instructions were: “First, the cursor will be a blue dot, your goal is to reach the target with the cursor. Sometimes the cursor will change to a pink triangle. These trials are special, but you still must try to reach the target with the cursor. You will be notified about the change by an attention sign that will appear, together with a sound, before the trial.” As mentioned before, the special trials (cued+) were trials where the direction of the cursor was rotated 40° clockwise with respect to hand direction.
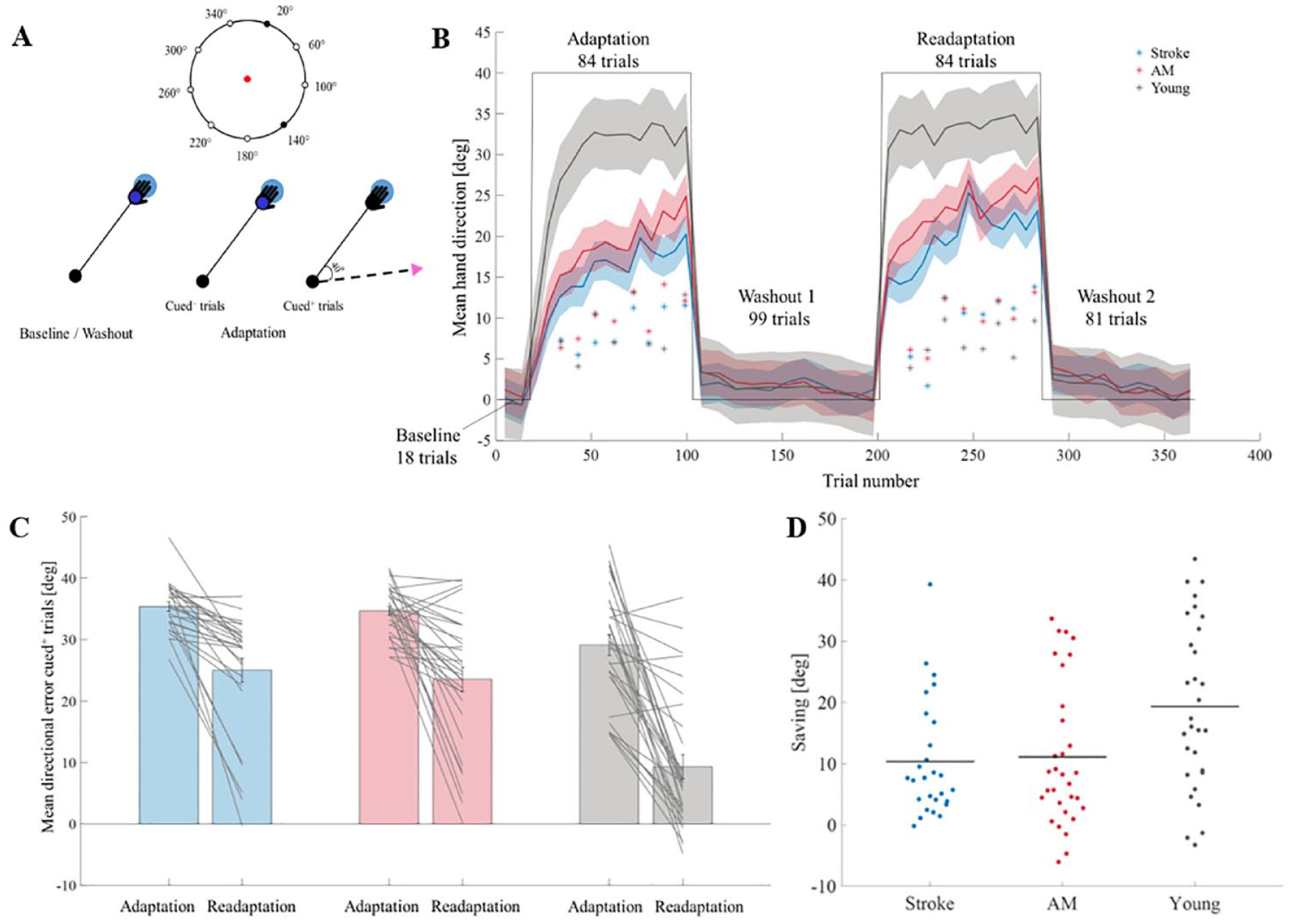
(A) Task’s description. Top—The position of the targets. Nine targets were used in the baseline and washout blocks (small circles). Two targets were used during the adaptation blocks (filled black circles). Bottom—description of trials. In baseline, washout and in the cued– trials, the hand and cursor (small darker blue dot) direction were aligned. In the cued+ trials the direction of the cursor (pink triangle) was clockwise rotated by 40° with respect to hand direction (black angle line). Directional error angle was calculated by the angle between lines connecting the center of the screen with the target (big light blue circle) and with the point where the cursor crossed the radius of the target. (B) Overall experimental design and the mean hand direction as a function of trial number. The colored lines represent the mean hand direction during the adaptation and readaptation blocks across groups—stroke (blue), AM controls (red), and young controls (gray). The solid line represents the mean hand direction in the cued+ trials of all the subjects. The shade represents confidence interval of 95%. Asterisks represent the location of the cued– trials and the mean hand direction in the cued– trials of all the 3 groups. The black line indicates the perturbation angle. (C) The mean directional error angle of the first 6 trials (cued+) in the adaptation and readaptation blocks in the 3 groups—stroke (blue), AM controls (red), and young (gray). Gray lines represent individual subjects. (D) Savings in the 3 groups. Stroke (blue), AM controls (red), and young controls controls (gray). Horizonal lines represent averages. Individual subjects are represented by dots.
Adaptation was measured as the change in hand direction in the perturbed trials. The explicit component was measured by the change in hand direction induced by the cue. In the cue− trials, subjects were expected to switch off any conscious strategies they were using during the perturbation trials. Targets were presented pseudo randomly; in the learning blocks each target appeared 3 times—within each 6-trial block. In the baseline and washout blocks each target was presented once—within 9-trail block. The order of appearance was random. The implicit component was measured by the change in the hand direction in the cued− trials.
Preregistration
This study is a part of a larger study that was preregistered online https://osf.io/2y5jx. Due to difficulties in recruitment, data from only 27 subjects with stroke were tested (instead of the 30 that were planned in the preregistration). The group of the young subjects was not pre-registered.
Power Analysis
Effect size was computed based on explicit adaptation estimates in young vs. old adults in experiment E1a in Vandevoorde and Orban de Xivry
20
(
Preprocessing
All analyses and statistical calculations were performed in MATLAB 2020a (Mathworks Inc., Natick, MA) and SPSS 21 (IBM SPSS statistics). For each reaching movement, the hand direction (relative to target angle) was the angle between lines connecting the center of the screen with the target and with the point where the cursor crossed the radius of the target. In trials where the cursor did not reach the radius of the target, cursor’s angle was defined based on the position of the cursor at the end of the trial. The hand direction was the primary dependent variable in the implicit and explicit components. We refer to the hand direction with respect to the target as the directional error angle (deviation from the target). Directional error angle above 80° or trials where the cursor did not complete 30% of distance to the target were considered outliers and were removed from the analysis. The number of outliers was 3.4% of all trials.
Statistical Analysis
Adaptation Analysis
Adaptation was examined through analysis of the hand directions in cued+ trials during the first (adaptation) and second (readaptation) blocks separately. Statistical comparison was performed using a 2-way ANOVA, with a within subject effect of trial and between-subject effect of group (AM controls, stroke, or young controls). In case of sphericity correction, we used the Greenhouse–Geisser correction. Statistical comparison was also performed using 2 sample t-test for post-hoc comparisons, with a Bonferroni correction for multiple comparisons with a factor of 2. Effect size for significant results was reported using partial eta square (
Saving Analysis
Savings were quantified using a comparison of the hand directions in the first 6 trials of the adaptation and readaptation blocks. We chose to evaluate savings using multiple trials since we were concerned that the context change may confuse the subjects, and that their first response will be excluded. 6 trials were used based on the design of the experiment (3 repetitions of each target per 6 trial block). Statistical comparison was performed with 2-way ANOVA. The within-subject factor was block and the between-subject factor was the group (AM controls, stroke, or young controls). In case of sphericity correction, we used the Greenhouse–Geisser correction. Statistical comparison was also performed using 2 sample t-test for post-hoc comparisons with a Bonferroni correction for multiple comparisons (with a factor of 2). Effect size for significant results was reported using partial eta square (
Explicit Component Analysis
The explicit component was calculated by subtracting hand direction angle in the cued– trials from the cued+ trials that appeared before them. All 8 new trials represented by the difference between the cued+ trials (that preceded the cued– trials) and the cued– in each block were examined together. Statistical comparisons within group were performed using a 1 sample t-test. Statistical comparisons across groups were performed using a 1-way ANOVA and a 2-way ANOVA (in case of sphericity correction, we used the Greenhouse–Geisser correction) and 2 sample t-test for direct and post-hoc comparisons (Bonferroni correction for multiple comparisons with a factor of 2 was applied). The between-subject factor was the group (AM controls, stroke, and young controls). When looking on the explicit component over trials, we examined the difference between the 8 difference measures (the difference between the cued+ trials that followed cued– trials and the cued–) in each block separately. Statistical comparisons across groups were performed using a 2-way ANOVA. The within subject factor was trial (8 in each block) and the between-subject factor was the group (AM controls, stroke, and young controls). Effect size for significant results was reported using partial eta square (
Implicit Component Analysis
The implicit component was analyzed from the cued– trials in each learning block (8 cued– trials per block). The amount of implicit learning was calculated per learning block as the mean of the 8 cued– trials in each block. Statistical comparisons within group were performed using a 1 sample t-test. Statistical comparison was performed using a 1-way ANOVA and a 2-way ANOVA (in case of sphericity correction, we used the Greenhouse–Geisser correction) and 2 sample t-test for direct and post-hoc comparisons (Bonferroni correction for multiple comparisons with a factor of 2 was applied). The between-subject factor was the group (AM controls, stroke, and young controls). When looking at the implicit component over trials, we examined the difference between the 8 cued– trials in each block separately. Statistical comparisons across groups were performed using a 2-way ANOVA. The within subject factor was trial (8 in each block) and the between-subject factor was the group (AM controls, stroke, and young controls). Effect size for significant results was reported using partial eta square (
Explicit Enhancement and Saving Analysis
We quantified the overall explicit learning by subtracting the explicit component in the adaptation block from the explicit component in the readaptation block. We refer to this variable as explicit enhancement. To create a savings parameter, we subtracted the mean hand direction angle in the first 6 trials of the adaptation from the mean of the first 6 trials in the readaptation blocks. Correlations were conducted using Pearson’s correlation.
Explicit Enhancement Association With Cognitive Assessments
The associations between cognitive abilities and explicit enhancement were conducted using Pearson’s correlation except of the sub-domains of the MoCA, where Spearman correlation was used due to the ordinal nature of the sub-scales.
Implicit adaptation association with cognitive assessment
The associations between cognitive abilities and implicit adaptation were conducted using Pearson’s correlation, based on the implicit estimate of the adaptation block.
Results
Adaptation and Savings in Stroke and Age Matched Controls
Subjects with stroke showed reduced cognitive abilities (ie, MoCA scores) compared to the 21 AM control subjects that performed the MoCA test (22.15 for the stroke group and 24.95 for AM control group, t(46) = 2.79, P < .01, d = 0.82).
To study the effect of cognitive ability on adaptation, we examined the ability of the stroke and AM control subjects to adapt to a visuomotor perturbation. We found a significant effect of trial on directional error both during the adaptation and readaptation blocks (adaptation, F(5.9,331.63) = 29.00, P < .001,
Next, we examined differences between the groups in the ability to show enhanced adaptation during the readaptation block (savings). We therefore compared the mean directional error in the first 6 trials of the adaptation and readaptation blocks.
Directional error was significantly smaller in the readaptation block than in the adaptation block (F(1,56) = 57.60, P < .001,
Age Related Changes in Adaptation and Savings
To measure adaptation and savings in the context of age-related changes, we examined adaptation and readaptation in an additional group of young control subjects (N = 30, age = 25.17 years ± 1.26 SD). In the adaptation block, a significant difference between the directional errors across groups was found (F(2,85) = 23.71, P < .001,
To measure if the group effect was constant along the block, we also estimated the interaction between adaptation across trials and group. This interaction was significant (F(13.5,573.78) = 1.93, P = .02,
Similar results were found in the readaptation block; a significant effect of group (F(2,85) = 16.92, P < .001,
Regarding age related changes in savings we found a significant group × block interaction (F(2,85) = 5.23, P < .01,
Explicit Learning
To investigate the mechanisms that underlie the differences between groups, we examined the explicit control over learning, under the hypothesis that the reduced cognitive abilities of the subjects with stroke will lead to impairments in their explicit control over the adaptation process.
The explicit component was calculated by subtracting hand direction in the cued– trials from the cued+ trials that appeared before them. We averaged these values for all cued– 8 instances in each block. There was a significant explicit component to adaptation (when compared to 0; adaptation block—stroke, t(26) = 3.36, P < .01, AM controls, t(30) = 3.77, P < .01, and young controls, t(29) = 7.98, P < .001; readaptation block—stroke, t(26) = 5.76, P < .001, AM controls, t(30) = 5.97, P < .001, and young controls, t(29) = 12.41, P < .001). Explicit learning in the adaptation block was significantly different between the groups (F(2,85) = 19.68, P < .001,
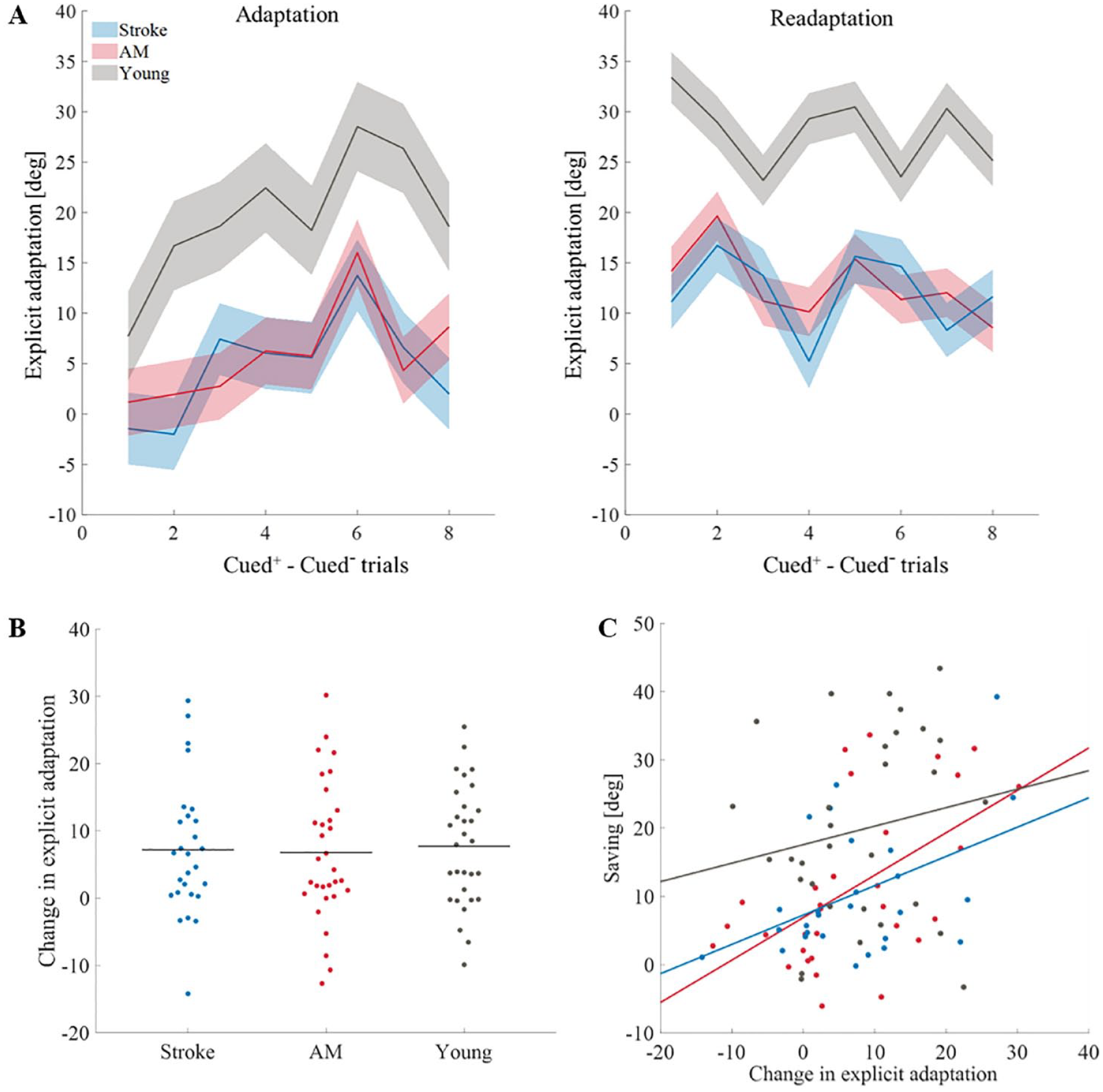
Explicit learning. (A) Explicit learning (calculated by subtracting hand angle direction in the cued– trials from the cued+ trials that appeared before them, see Figure 1B for the location of the cued– trials) along the adaptation block and readaptation block in all of the groups (stroke (blue), AM controls (red), and young controls (gray)). The solid line represents the mean hand angle of the explicit components of all the subjects. The shade represents confidence interval of 95%. (B) Change in explicit adaptation (readaptation block–adaptation block) across groups. Horizontal lines represent means. Dots represent individual subjects: stroke (blue), AM controls (red), and young controls (gray). (C) Correlation between explicit learning and savings across all groups. Dots represent individual subjects: stroke (blue), AM controls (red), and young controls (gray). Trendlines are derived using a regression analysis for visualizing the linear associations between variables.
The explicit component increased from adaptation to readaptation (F(1,85) = 48.25, P < .001,
Investigating the influence of the explicit component further, we looked at the explicit component over trials (within each block). We found that the effect of trial is significant; the explicit component increases along the adaptation block but not during readaptation (adaptation block, F(5.2,261.12) = 4.99, P < .001,
Considering our results, we wanted to examine if there is a connection between the explicit enhancement across blocks and savings. A Pearson’s correlation test revealed a moderate and significant association between these components only in the AM controls (r(29) = .55, P <0.01) and subjects with stroke (r(25) = .45, P = .02; young controls, r(28) = .14, P = .45; Figure 2C). This result is consistent with previous reports suggesting that savings is associated with the retrieval of explicit strategy.7,20
Implicit Learning
Next, to examine change in implicit adaptation, we analyzed the data from the cued– trials in each learning block (8 cued– trials per block). The amount of implicit learning was calculated per learning block as the mean of the 8 cued– trials in each block. The amount of implicit learning was increased during adaptation; hand direction in the cued– trials in the adaptation block deviated from the hand direction during the previous baseline (stroke, t(26) = 8.22, P < .001, AM controls, t(30) = 12.01, P < .001, and young controls, t(29) = 6.93, P < .001). Additionally, implicit leaning was also increased during readaptation; hand direction in the cued– trials in the readaptation block deviated from the hand direction during the previous washout (stroke, t(26) = 9.84, P < .001, AM controls, t(30) = 8.97, P < .001, and young controls, t(29) = 6.81, P < .001). These results indicate that there was an implicit component to adaptation.
Implicit learning did not differ between groups in the adaptation (F(2,85) = 0.89, P = .42,
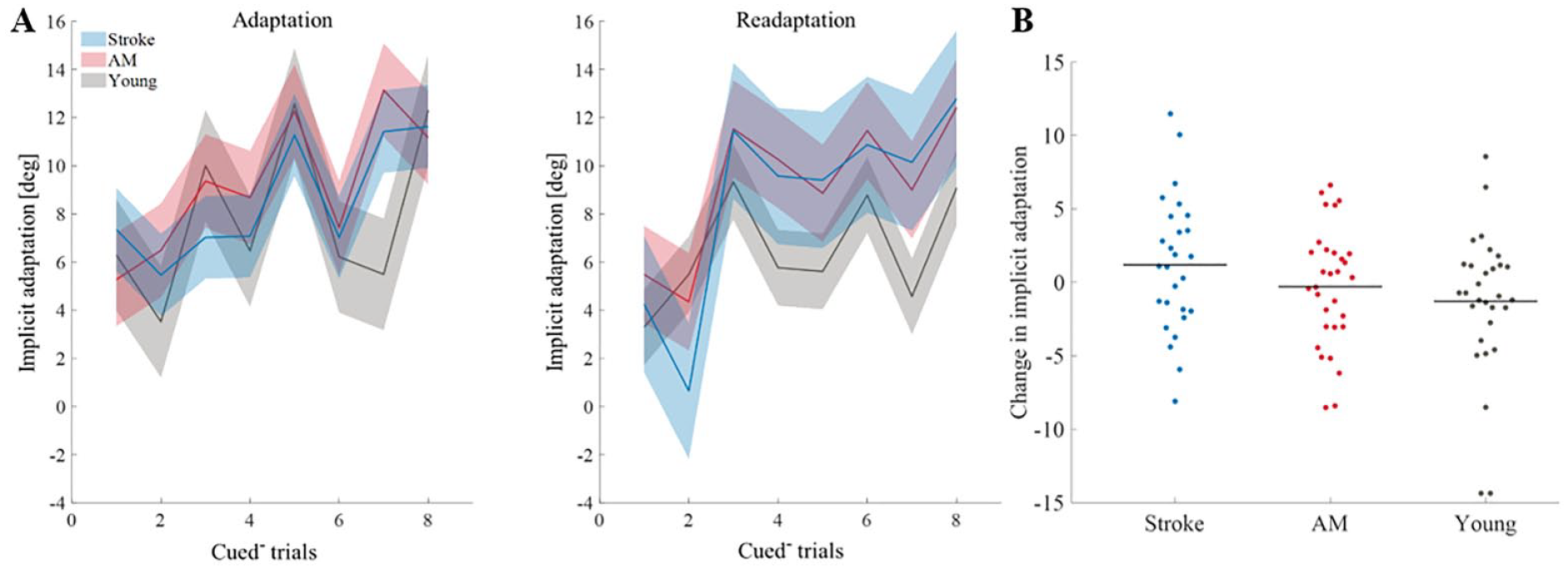
Implicit learning. (A) Implicit learning through time in adaptation block and readaptation block in all of the groups (stroke (blue), AM controls (red), and young controls (gray)). The solid line represents the mean hand angle of the implicit components of all subjects. The shade represents the ± confidence interval of 95%. (B) Change in implicit adaptation (implicit component during adaptation–implicit component during readaptation). Horizonal lines represent averages. Dots represent individual subjects: stroke (blue), AM controls (red), and young controls (gray).
To further investigate the implicit component, we looked at the implicit learning across trials; implicit learning increased across trials in both learning blocks (adaptation block, F(5.5,409.96) = 8.45, P < .001,
Explicit Enhancement Association With Cognitive Decline
We next examined if adaptation deficits are associated with cognitive decline, through the association between age-related impairments in explicit learning in adaptation and cognitive abilities (assessed by MoCA). The correlation between cognitive decline and explicit learning during the adaptation block was not significant in both groups (stroke r(25) = .07, P = .72; AM control group r(19) = -.01, P = .95). Deficits in explicit learning in the readaptation block were found to be moderately correlated with cognitive decline in the stroke group (r(25) = .46, P = .02) but not in the AM control group (r(19) = .18, P = .43). When examining the correlations between the change in explicit learning (explicit enhancement), we also found a moderate association between explicit enhancement and cognitive impairments in the stroke group (r(25) = .44, P = .02) but not in the AM control group (r(19) = .24, P = .30; Figure 4A). Finally, we explored the contribution of sub-cognitive domains to explicit enhancement. We chose to focus on explicit enhancement since it is more interpretable than the performance in the readaptation block, which involves both adaptation and recall abilities. We therefore conducted a Spearman correlation between explicit enhancement and the sub-domains of the MoCA. The cognitive abilities that were mostly associated with explicit enhancement in the stroke group were perception ( ρ(25) = .55, P < .01), short-term memory (ρ(25) = .48, P = .01) and attention (ρ(25) = .41, P = .03). To examine if this association increases with age, we searched for an association between explicit enhancement and performance in a verbal learning test, as an estimate of declarative memory, 39 in the young group. A weak positive correlation between the verbal learning scores at the first 2 repetitions (but not at the final recall) and explicit enhancement were detected (first repetition, r(28) = .37, p = .04; second repetition, r(28) = .38, P = .04; final recall, r(28) = .30, P = .10; Figure 4B).
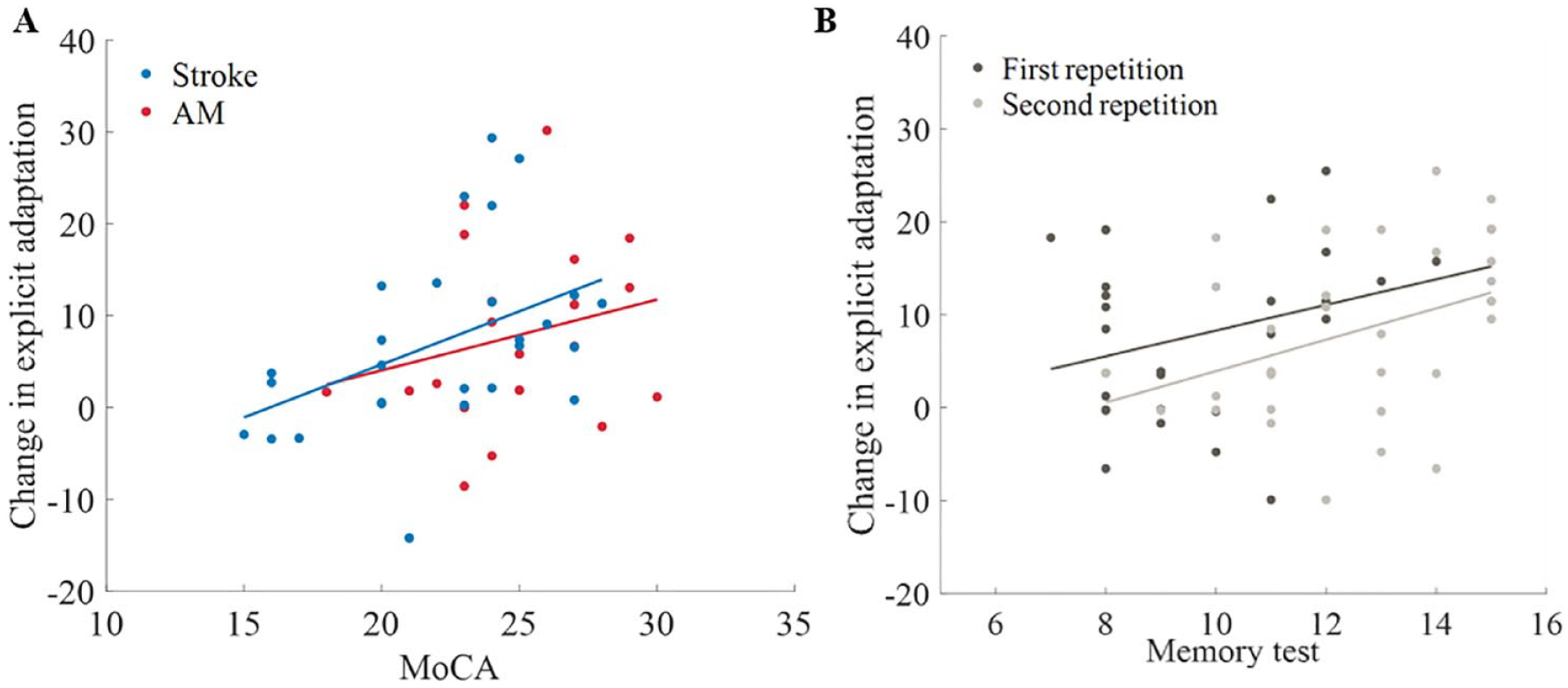
Cognitive performance and explicit adaptation. (A) Correlation between explicit enhancement and MoCA final score in the stroke (blue), AM controls (red). Dots represent individual subjects. (B) Correlation between explicit enhancement and the scores from the first repetition (dark gray) and the second repetition (light gray) in the modification of the California verbal learning test II in the young controls group. Dots represent individual subjects. Trendlines in A and B are derived using a regression visualizing the linear associations between variables.
The Association Between Implicit Adaptation and Cognitive Abilities
Finally, we explored if implicit adaptation is correlated with the cognitive abilities of the stroke and AM controls. We did not find a significant correlations between implicit adaptation in the adaptation block and the MoCA scores of the stroke (r(25) = .28, P = .15) and AM control groups (r(19) = -.06, P = .80). This result further supports the finding that cognitive abilities affect adaptation through the explicit process.
Discussion
We report that compared to age-matched control subjects, subjects with chronic stroke do not show attenuation of adaptation, savings, explicit, and implicit learning. This similarity was observed despite the increased cognitive impairments in the stroke group. Both stroke and AM controls showed inferior performance when compared to a group of young control subjects: their adaptation, savings, and explicit learning were decreased, whereas their implicit adaptation was comparable. The explicit enhancement in the stroke and young controls’ groups was correlated with their performance in the cognitive assessments, supporting the dependency of explicit learning on cognitive abilities.
Age Influence Adaptation and Saving
Aging is associated with deterioration of adaptation rates and savings.20,40 -42 This deterioration was shown to be associated with the reduction of explicit learning20,43 and with age-related cognitive decline. Based on these studies, we predicted that subjects with stroke, that have more severe cognitive impairments, will show a greater attenuation of adaptation rates. Nevertheless, although the stroke group showed reduced cognitive abilities with respect to their AM control group, they did not present adaptation attenuation.
The lack of a significant reduction in adaptation performance in the stroke group, despite their cognitive deficits, may indicate that the aging effect on adaptation is not driven by their cognitive decline. This explanation is unlikely since our correlation analysis shows that cognitive abilities and adaptation are related. MoCA scores were moderately correlated with explicit control for the subjects with stroke. Furthermore, the cognitive components that were specifically correlated with adaptation were attention, perception, and short-term memory. Attention processes influence the ability to store, manipulate visuospatial information and are important in monitoring movements.44,45 When altering perception by increasing uncertainty to the visual feedback of subjects during adaptation, subjects adapt slower 46 so if perception is impaired, impairments in adaptation are expected. Memory was also found to be connected to the explicit component in adaptation for young and old adults.43,47 In this study, the connection between working memory and explicit enhancement was also found in the young control subjects, suggesting that the dependence of explicit learning in adaptation on cognitive abilities is not specific to the stroke population. Moreover, we found that savings was associated with the explicit enhancement component, providing further evidence for the role of cognitive deterioration in aging in motor learning.
A second explanation for the lack of difference is that the effect of aging between the ages of 25 and 67 years on cognitive abilities is much greater than the effect of stroke in our study group. Indeed, this is evident when considering the MoCA scores: assuming young control subjects will have the maximal score in MoCA (30 points), the difference due to aging (~6 points) is substantially bigger than the difference due to stroke (~2 points). While we assumed that the effect of aging will be greater than the effect of stroke in the power analysis, our design may still not be sensitive enough to capture the effect of stroke. Furthermore, it could be that the relationship between cognitive abilities and adaptation and explicit learning is not linear. Thus, the effect of cognitive decline associated with aging may have a stronger effect on adaptation performance than the additional deterioration that is caused by stroke.
A third alternative explanation is that the lack of effect is a result of increased inter-subject variability in the AM and stroke groups. One way to approach the increased variability in the stroke group is by stratification of subjects with stroke based on symptoms, or lesion site. For example, Mutha et al 48 showed that subjects with stroke with lesions to the left parietal cortex show adaptation deficits. Our results are not consistent with this result since most of the subjects with stroke in this study (22/27, see Table 1) had stroke due to injuries in their left hemisphere. In fact, when running our analyses only on the left lesioned subjects, the results of the subjects with stroke are still not differentiable from the AM control subjects (adaptation block, F(1,51) = .47, P = .50; readaptation block, F(1,51) = 0.97, P = .33). We therefore speculate that the lack of difference is not a result of the increased variability in the stroke group.
The inconsistency between our results and the results of Mutha may stem from the small number of subjects in Mutha’s study (5 in each group) or from the fact that the subjects with stroke in Mutha’s studies used their affected hand, whereas our subjects used their unaffected hand. This conjecture is supported by a recent paper that demonstrate adaptation impairments with the affected arm, 32 suggesting that adaptation impairments may be effector specific. This claim is also supported by a meta-analysis by Kal et al, 49 that point to differences in explicit and implicit learning after stroke when using the affected arm in comparison to the unaffected arm. Further research focusing on the performance of the unaffected arm 50 should be conducted to characterize the cognitive aspect of motor learning (implicit or explicit) and to minimize the contaminating effects of sensory or control impairments.
Importantly, the dependence of performance in motor tasks on cognitive abilities increases with aging and following brain damage to compensate for motor and cognitive impairments. 51 Previous studies have demonstrated that in motor learning tasks, old adults rely more on their cognitive abilities to perform the task52,53 than young adults. Thus, it may be that the significant correlation between cognitive abilities and explicit learning in the subjects with stroke and not in the AM controls group indicate a greater reliance of the subjects with stroke on their cognitive abilities. The increase reliance also suggests that the subjects with stroke had cognitive resources to compensate for their adaptation deficits.
Motor Learning and Stroke
The effect of stroke on motor learning was examined in other tasks as well. Results from sequence learning tasks are inconclusive; some studies show that stroke leads to motor learning impairments54,55 and some show that it does not.56 -59 For example, Vakil et al, 55 discovered that healthy controls learned a motor sequence significantly better than the stroke group, while Shin et al., 58 found no difference between stroke and healthy controls in their motor sequence learning abilities. When isolating explicit learning in stroke, our results are consistent with results of studies that evaluated explicit learning using the unaffected arm in subjects with stroke.57,59
Implicit Control
Implicit learning was decreased in the readaptation block in the young controls group compared to the AM control and stroke groups. This result is consistent with the results of Vandevoorde and Orban de Xivry 20 that showed increased implicit involvement in elderly. This increase may stem from the decreased explicit involvement in these groups. This dependence between explicit and implicit learning may indicate that they are driven by the same feedback.9,40,60 Alternatively, the difference could be related to the task, and reflect a better ability of the young control subjects to suppress their strategy in the cued– trials. In fact, a central limitation of this study is the assumption that all experimental groups react in the same manner to the cues. It could be that the stroke and the AM controls do not react to the cues as consistently and effectively as the young controls. In that case, the estimate of the implicit learning will be contaminated by explicit learning in these groups, and the measures of explicit learning will be an under-estimation of their explicit strategy. This limitation is exaggerated by recent reports about inconsistencies between different methods for estimating explicit and implicit learning. 61 Admittedly, other available paradigms for estimating explicit learning, such as the aiming task,3,7 could provide a direct measure of explicit learning, but their implementation in elderly and subjects with stroke was not successful in pilot studies in our lab. We conclude that additional attempts to measures explicit and implicit learning should be made to corroborate the results presented here.
Other Limitations
An additional limitation of this study is that the lack of stroke effect on adaptation is a result of a low sensitivity of our measure, or a ceiling effect of the estimation of the explicit component. Another limitation is that the subjects with stroke may had minor motor impairments in their unaffected arm that were not captured by the clinical (FMA) assessment. We call for replicating our results and for conducting a follow up experiment in a population with stroke that have greater cognitive deficits. An additional approach could be to further stratify subjects in the stroke groups according to their cognitive impairment level and damage location. For example, a follow up study with a group of subjects with right hemisphere damage (without neglect) is important to establish that our results are not specific to damage in the left hemisphere.
Implications
Our results indicate that motor learning abilities are not susceptible to cognitive decline following stroke, thereby suggesting that strategic learning should be incorporated into rehabilitation protocols even in cases where subjects have cognitive deficits. The correlation between explicit enhancement and cognitive ability in the stroke group suggests that this group utilizes cognitive resources for its learning more than the age-matched control group. The implication of this result is that cognitive load during rehabilitation should be monitored to free cognitive resources to support explicit strategies in rehabilitation.
Conclusions
Our results support the involvement of cognitive processes in adaptation learning and savings by showing that populations with cognitive impairments show reduced adaptation that is attributed to explicit learning. The lack of additional adaptation attenuation in the stroke group despite its cognitive impairments, suggests that the relationship between adaptation learning, and specifically, explicit adaptation and enhancement, and cognitive abilities is not linear, and that reported adaptation impairments following stroke may be driven by contralesional motor deficits of the subjects. Future studies should concentrate on the connection between cognition, motor learning and motor recovery in the acute phase of stroke, where most of the recovery and rehabilitation treatment take place.
Footnotes
Acknowledgements
We thank Rotem Bachar for helping us with the data collection. We thank Dr. Oren Barzel and Dr. Shilo Kramer for their clinical assistance. We thank the reviewers for their constructive comments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the United States-Israel Binational Science foundation grant 2015327, and by ISF grant 1244/22 to LS.
