Abstract
Background
Transauricular vagus nerve stimulation (taVNS) is being studied as a feasible intervention for stroke, but the mechanisms by which this non-invasive technique acts in the cortex are still broadly unknown.
Objectives
This study aimed to systematically review the current pre-clinical evidence in the auricular vagus nerve stimulation (aVNS) neuroplastic effects in stroke.
Methods
We searched, in December of 2022, in Medline, Cochrane, Embase, and Lilacs databases. The authors executed the extraction of the data on Excel. The risk of bias was evaluated by adapted Cochrane Collaboration’s tool for animal studies (SYRCLES’s RoB tool).
Results
A total of 8 studies published between 2015 and 2022 were included in this review, including 391 animal models. In general, aVNS demonstrated a reduction in neurological deficits (SMD = −1.97, 95% CI −2.57 to −1.36, I2 = 44%), in time to perform the adhesive removal test (SMD = −2.26, 95% CI −4.45 to −0.08, I2 = 81%), and infarct size (SMD = −1.51, 95% CI −2.42 to −0.60, I2 = 58%). Regarding the neuroplasticity markers, aVNS showed to increase microcapillary density, CD31 proliferation, and BDNF protein levels and RNA expression.
Conclusions
The studies analyzed show a trend of results that demonstrate a significant effect of the auricular vagal nerve stimulation in stroke animal models. Although the aggregated results show high heterogeneity and high risk of bias. More studies are needed to create solid conclusions.
Introduction
Stroke is the second leading cause of death 1 and the first leading cause of disability worldwide, causing a total economic burden of approximately of 100 billion dollars to the United States annually. 2 It can be caused either by an abrupt interruption of blood flow in the cerebral vascular territory, or blood leakage causing cerebral hemorrhage. In the ischemic stroke, the interruption of blood flow and thus shortage of glucose levels leads to the failure of energy-dependent channels, which in turn activates a series of intracellular events that culminate in increased oxidative stress. 3 The activation of inflammatory pathways is a major secondary event to the progression of brain damage.
Currently, the only available therapeutic options at the acute stage rely on chemical or surgical reperfusion techniques. Still, only a minority of the patients are eligible to receive them. 4 Also, the rapid blood reperfusion, even though crucial to restoring brain metabolic activity and reverse penumbra, may also lead to secondary lesions due to an abrupt increase in oxygen availability. 5 Neuroprotectant agents targeting pathological events may help contain the detrimental cascade resultant from both ischemia and rapid reperfusion, extend the time window for treatment, and serve as an adjunct alternative to ineligible patients to reperfusion. 6
Recently, animal studies have been investigating more accessible, less invasive interventions to deal with the consequences and complications of stroke. One of these techniques is noninvasive vagus nerve stimulation, which can be applied transcutaneously via the auricular branch of the vagus nerve or the cervical branch, with auricular vagus nerve stimulation (aVNS) being more thoroughly investigated for this condition.7,8 Recent systematically reviewed results, on the aVNS’s electroencephalographic effects, suggests the capability of aVNS in modulate cortical activity. 9 Furthermore, aVNS has been investigated in a variety of conditions, such as epilepsy, tinnitus, depression, Alzheimer’s disease, Schizophrenia, pain, migraine, and Parkinson’s disease, being considered safer, more accessible, and less invasive alternative than the surgical device.9 -12 Currently, animal studies seem to be having positive results regarding the reestablishment of motor function in rats with post stroke sequelae after aVNS.13 -22 However, there is a lack of full understanding and categorization of the current evidence, especially regarding the mechanisms behind aVNS’s effects that can be further analyzed.
Therefore, given the amplifying field of aVNS and its current potential relevancy for post stroke rehabilitation, we propose a systematic review and meta-analysis assessing the current pre-clinical evidence in aVNS in stroke aiming at understanding the mechanistic aspects of this intervention.
Methods
Data Sources and Study Selection
The team executed a systematic review of the literature and meta-analysis. The study followed the Preferred Reporting Items for Systematic Reviews and Meta-Analysis, 23 the Cochrane handbook for systematic reviews recommendations, 24 and a step-by-step guide to conduct systematic reviews of animal studies proposed by Lenaars et al. 25 This review was registered in PROSPERO, by the number 385026.
We used the search terms (“Transcutaneous vagal nerve stimulation” OR “Transcutaneous vagus nerve stimulation” OR “Auricular vagal nerve stimulation” OR “Auricular vagus nerve stimulation” OR “tVNS” OR “ taVNS” OR “aVNS” OR “VNS” OR “vagal nerve stimulation”) AND (“stroke” OR “cerebrovascular disease” OR “CVA”) OR (“poststroke” OR “post-stroke”) OR (“cerebrovasc*” OR “brain vasc*” OR “cerebral vasc*” OR “SAH”) OR (“ischemic transient attack”) OR (“ischemic stroke” OR “brain infarct*”) OR (“hemorrhagic stroke” OR “hemorrhagic stroke” OR “brain bleed”) AND ((animals) OR (rodents) OR (rats) OR (mice) OR (mouse) OR (mouse model*) OR (rodent model*) OR (rat model*) OR (mammal*) OR (animal)) in the Medline, Cochrane, Embase, and Lilacs databases. The search was performed in December 2022. The inclusion criteria were done as follows: (i) studies that used auricular vagus nerve stimulation in post cerebral vascular events in animal models and (ii) studies in English, Spanish, or Portuguese. Exclusion criteria were: (i) case reports, review articles, abstracts of conferences, letters, editorials, case control studies; (ii) ex vivo studies, in vitro studies, studies in humans or in sillico studies; and (iii) cervical vagal nerve stimulation.
The references gathered by the search were transferred to an excel for undergoing evaluation and screening. The titles and abstracts were analyzed by 2 separate authors (IR and AM). If the abstracts were not clear, a full text evaluation was retrieved. At the end, the authors reached a consensus regarding the inclusion and exclusion criteria, afterward the references underwent data extraction and analyses.
Acute ischemia/reperfusion models were considered acute with the outcomes assessed until 24 hours after the event, subacute between 24 hours and 6 months, and chronic if the outcomes were assessed after 6 months.
Data Extraction and Quality Assessment
Two authors (PSdM and JV) executed the extraction of the data on Excel, the process was supervised by a third senior investigator (FF). The authors extracted variables related to the animal models, study design, intervention, stimulation parameters, and outcomes related with neurological behavior, anatomopathological data, inflammatory markers, neuroplasticity markers, and other mechanistic outcomes disposed by the studies.
Quality Assessment
The risk of bias was evaluated by adapted Cochrane Collaboration’s tool for animal studies (SYRCLES’s RoB tool). 26 The tool allows the assessing of concerns of domains such as: sequence generation, baseline characteristics, allocation concealment, random housing, blinding (performance bias), random outcome assessment, blinding (detection bias), incomplete outcome data, selective outcome reporting, and other sources of bias. Two investigators (JV and ACG) rated the studies independently and came to a consensus regarding the evaluation, a third investigation was contacted in case of disagreement (PSdM).
Quantitative Analysis
Descriptive Data
We standardized the metrics of central tendency for mean and standard deviations (SD). When not reported, the means and SD or standard errors (SE) were extracted from the plots provided by the study using the WebPlotDigitizer software (https://automeris.io/WebPlotDigitizer/). SEs were transformed in SDs by the following formula:
Outcomes
Primary outcomes were the behavioral assessments represented by the neurological deficits scores and adhesivel removal test. Secondary outcomes included lesion and neuroplastic anatomopathological results, such as infarct size, micro capillary density, markers of angiogenesis, and BDNF levels and expression. The mean and SDs post intervention were compared between aVNS and controls for each one of the outcomes.
Meta-Analysis
Based on the post intervention metrics, we performed a random-effects meta-analysis calculating the pooled standard mean differences between aVNS and controls and its 95% confidence intervals (CI) based on the inverse variance method. Heterogeneity were tested through I2, we considered low heterogeneity when I2 < 40%, moderate when 40% < I2 < 60%, and high when I2 > 60%. 24 A sensitivity analysis omitting each one of the studies was performed for those outcomes with more than 3 studies. The command “metacont,” for the pooled results, and “metainf,” for the sensitivity analysis, from the “meta” package in RStudio were used in this analysis. In order to test publication bias, we built a funnel plot for the main outcome using the command “funnel.meta” from the “meta” package in RStudio. 27
Results
Eligible Studies and Studies Characteristics
Primarily, we found 236 studies in all databases. Duplicates were excluded and the rest were filtered by title, abstract, and full text based on the eligibility criteria.
Finally, a total of 17 studies were selected and 9 of them were excluded because they were performed in humans. A total of 8 studies fulfilled the eligibility criteria and were included in this review (Supplemental 1).
The 8 studies were published between 2015 and 2022, they included 391 animal models. The average follow-up time of these models was 17 days ± 9.06, 7 studies analyzed subacute stroke phase and only 1 acute model. The most used model was the occlusion of the middle cerebral artery (n = 6), followed by the carotid artery occlusion (n = 2). All studies had a parallel design and analyzed the differences between active and sham aVNS after ischemia/reperfusion lesion. The majority of these studies used electroacupuncture aVNS (eaVNS; n = 5) and the rest used transcutaneous aVNS (taVNS; n = 3; Table 1).
Studies Characteristics.
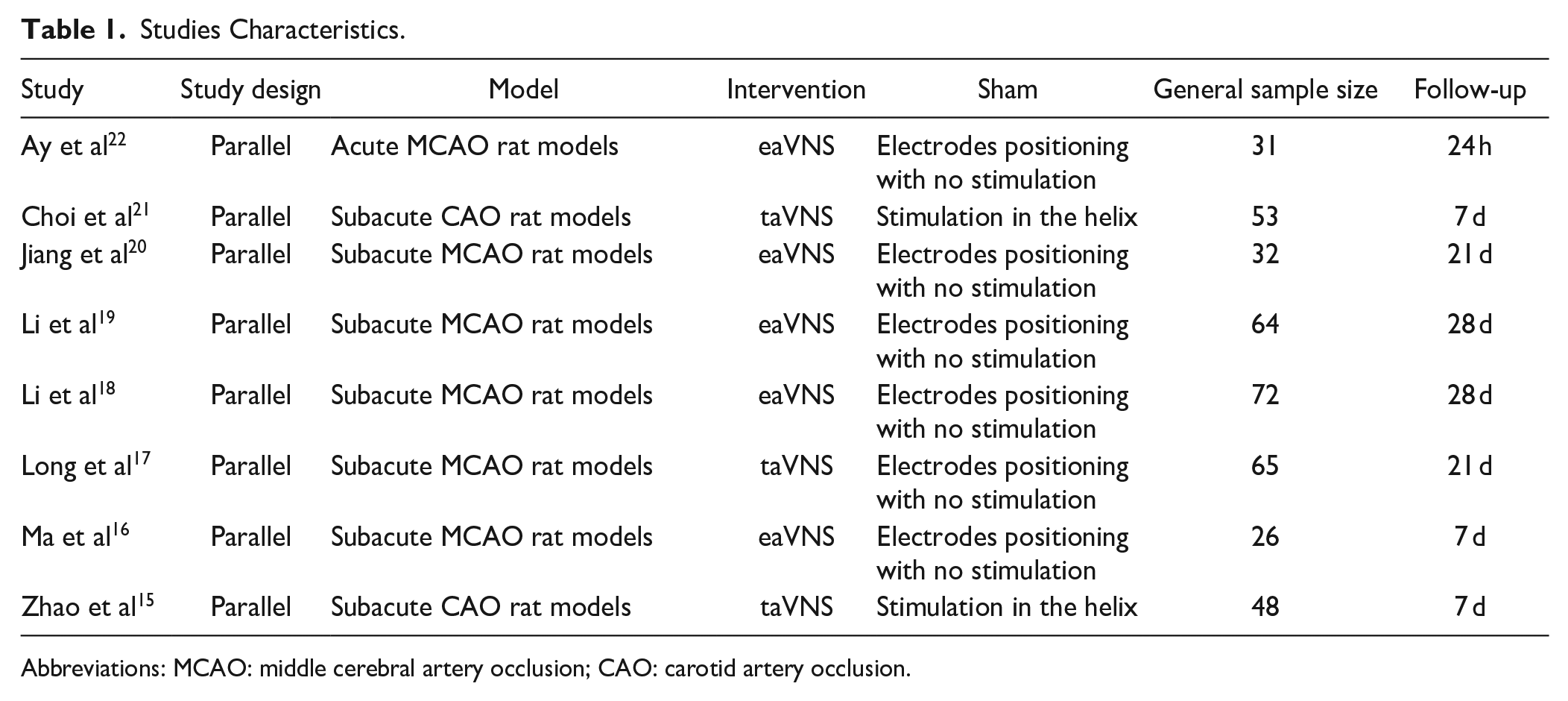
Abbreviations: MCAO: middle cerebral artery occlusion; CAO: carotid artery occlusion.
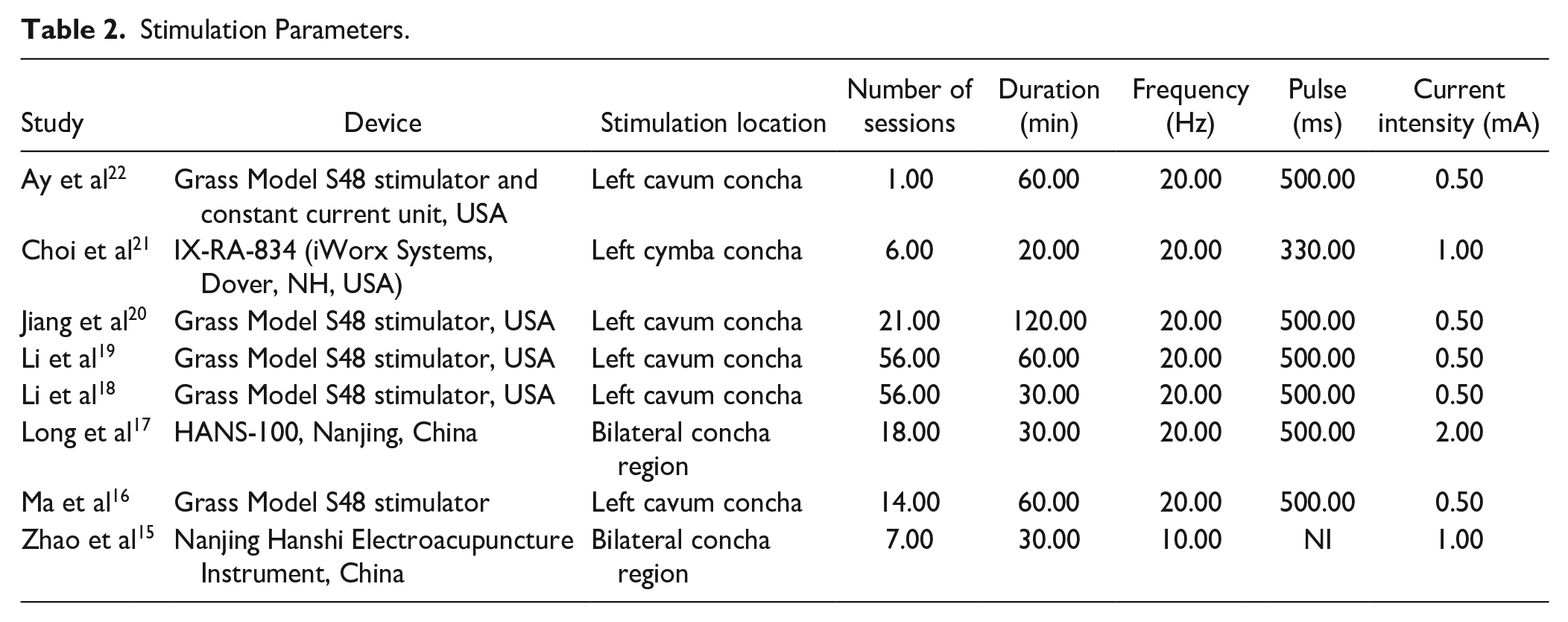
Regarding the stimulation parameters, all the studies stimulated the cavum concha. Two of them performed bilateral stimulation, while the rest stimulated the left ear. In general, the studies performed 22.38 ± 20.35 sessions, during 51.25 ± 30.18 minutes, with the frequency of 18.75 ± 3.31 Hz, the pulse width of 475.712 ± 59.49 ms, and the current intensity of 0.81 ± 0.50 mA (Table 2).
Stimulation Parameters.
Behavioral Outcomes
Most of the studies analyzed neurological deficits scores (n = 6), followed by the adhesivel removal test (n = 3). Choi 2022 analyzed the effects of taVNS in cognition using the NOR and Y-maze test and Long dysphagia analyzing body weight, food, and water intake. All studies were able to show an effect of auricular stimulation on those outcomes compared to controls (Table 3).
Behavioral Outcomes and Results.
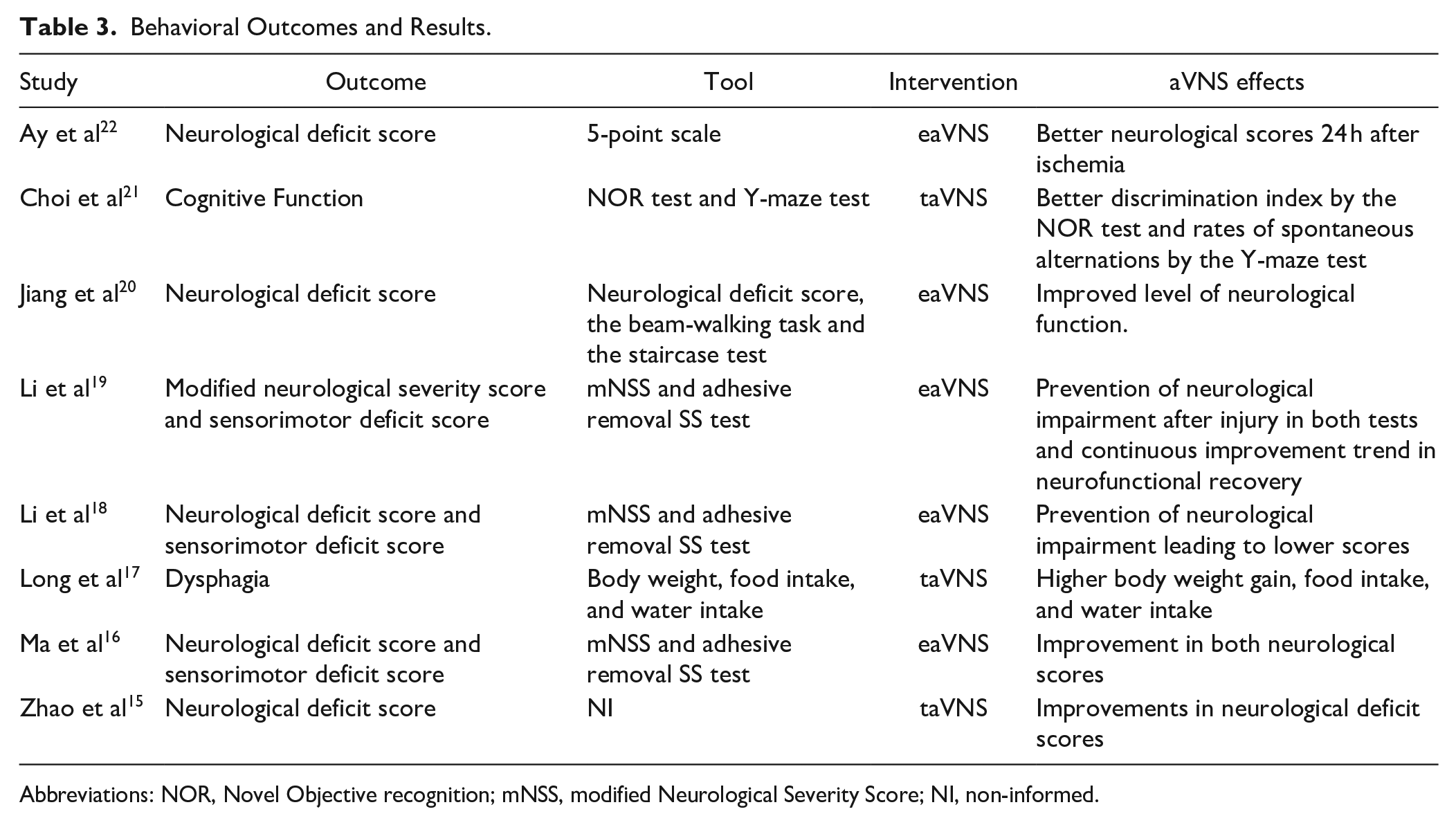
Abbreviations: NOR, Novel Objective recognition; mNSS, modified Neurological Severity Score; NI, non-informed.
Quantitative Analysis
Based on that, we could synthesize the data from the neurological deficits scores and the adhesive removal test.
For the neurological deficit score, 6 studies using I/R models were included in the analysis, showing an effect of aVNS in reducing neurological deficit in comparison to controls, the studies demonstrated a moderate heterogeneity. (SMD = −1.97, 95% CI −2.57 to −1.36, I2 = 44%). A sensitivity analysis was performed omitting each one of the correspondent studies that did not impact the result (Supplemental 2A).
Regarding the adhesive removal test, 3 studies results were synthesized, the aVNS pooled effect reduced the time of the test, however detected a high heterogeneity between the studies (SMD = −2.26, 95% CI −4.45 to −0.08, I2 = 81%; Figure 1).
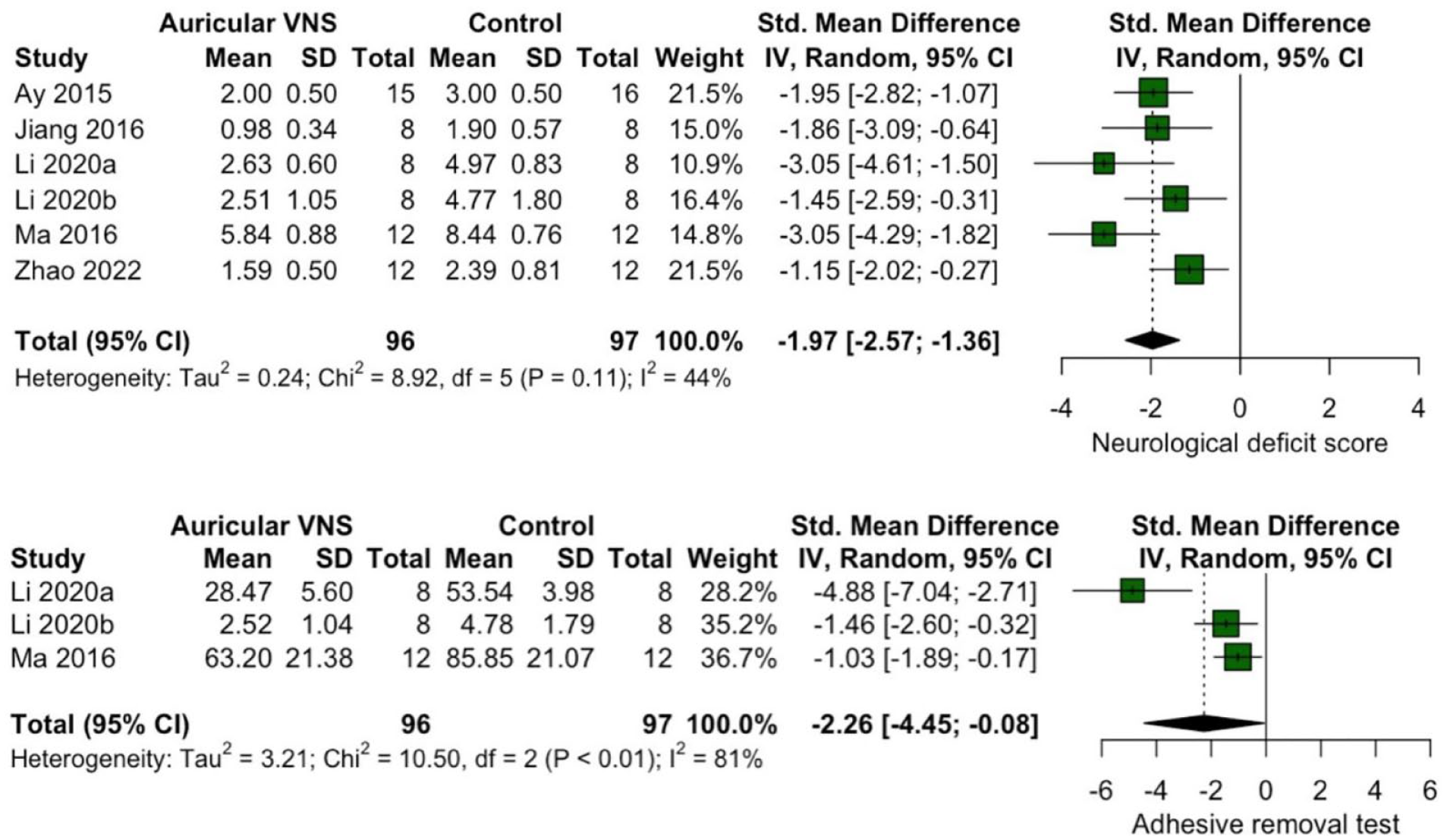
Forest plots of the aVNS effect on the neurological deficit score and adhesive removal test.
Anatomopathological/Inflammatory Outcomes
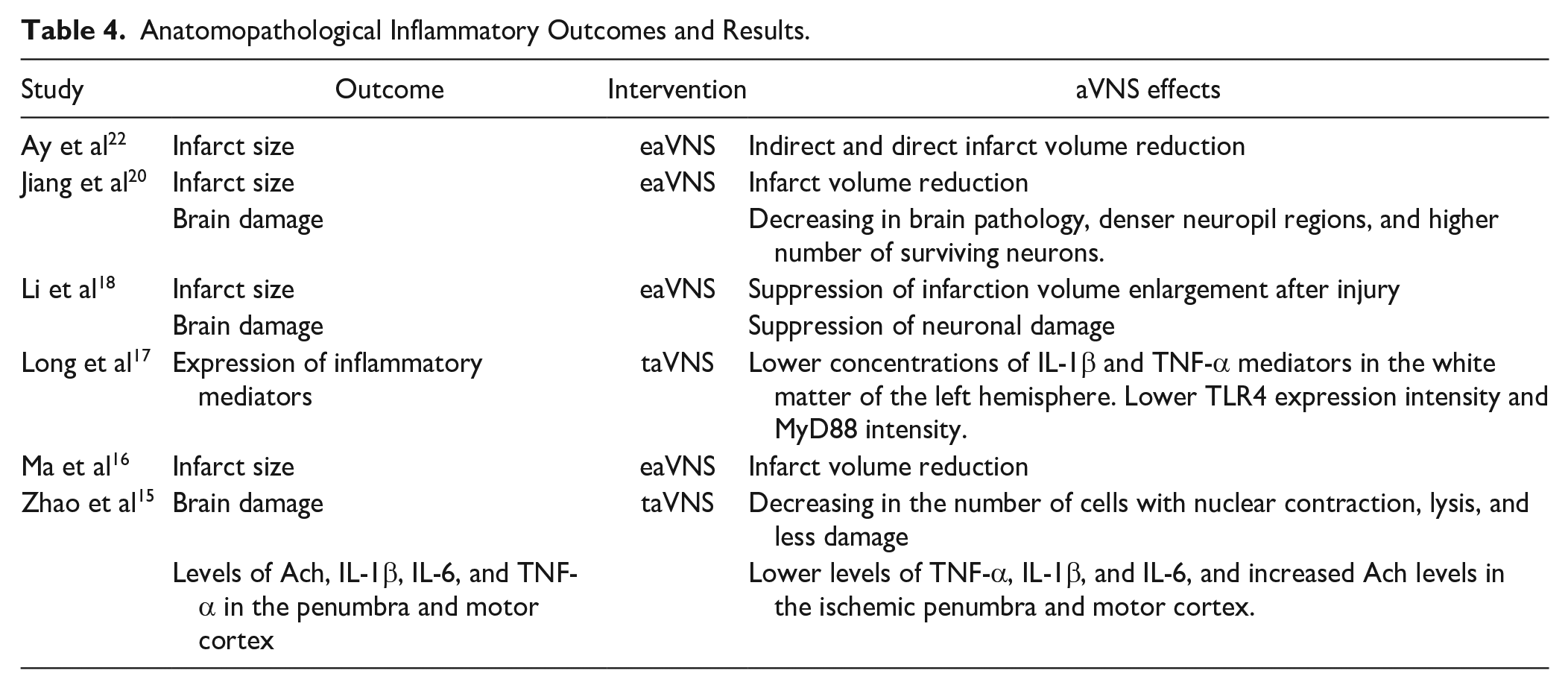
The authors included in this review analyzed anatomopathological outcomes to study the mechanistic effects of aVNS. Most of the studies analyze the infarct size (n = 4), the brain damage (n = 3), and inflammatory cytokines expression (n = 2). All the studies could detect a reduction in infarct size, brain damage, and inflammatory cytokines induced by aVNS compared to control after I/R (Table 4).
Anatomopathological Inflammatory Outcomes and Results.
Quantitative Analysis
Regarding infarct size, we could synthesize the data and calculate the pooled standardized mean difference between aVNS and control. Overall, aVNS decreased the infarct size after I/R compared to controls, however, the studies presented a modern high heterogeneity between them (SMD = −1.51, 95% CI −2.42 to −0.60. I2 = 58%; Figure 2). A sensitivity analysis was performed omitting each one of the correspondent studies that did not impact the result (Supplemental 2B).
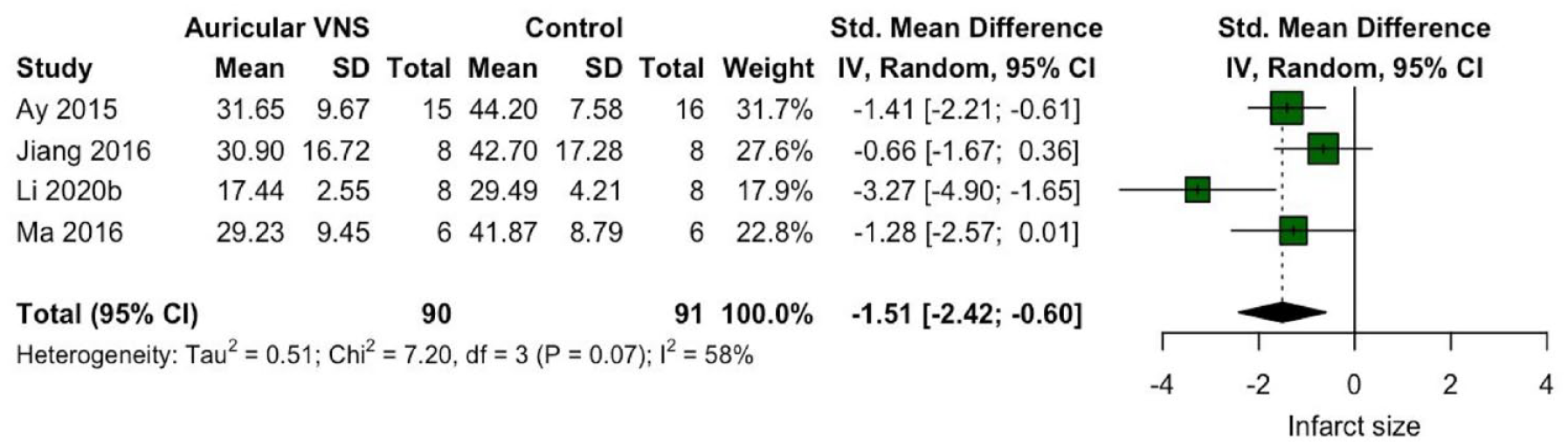
Forest plot of the aVNS effect on infarct size.
Neuroplasticity Markers Outcomes
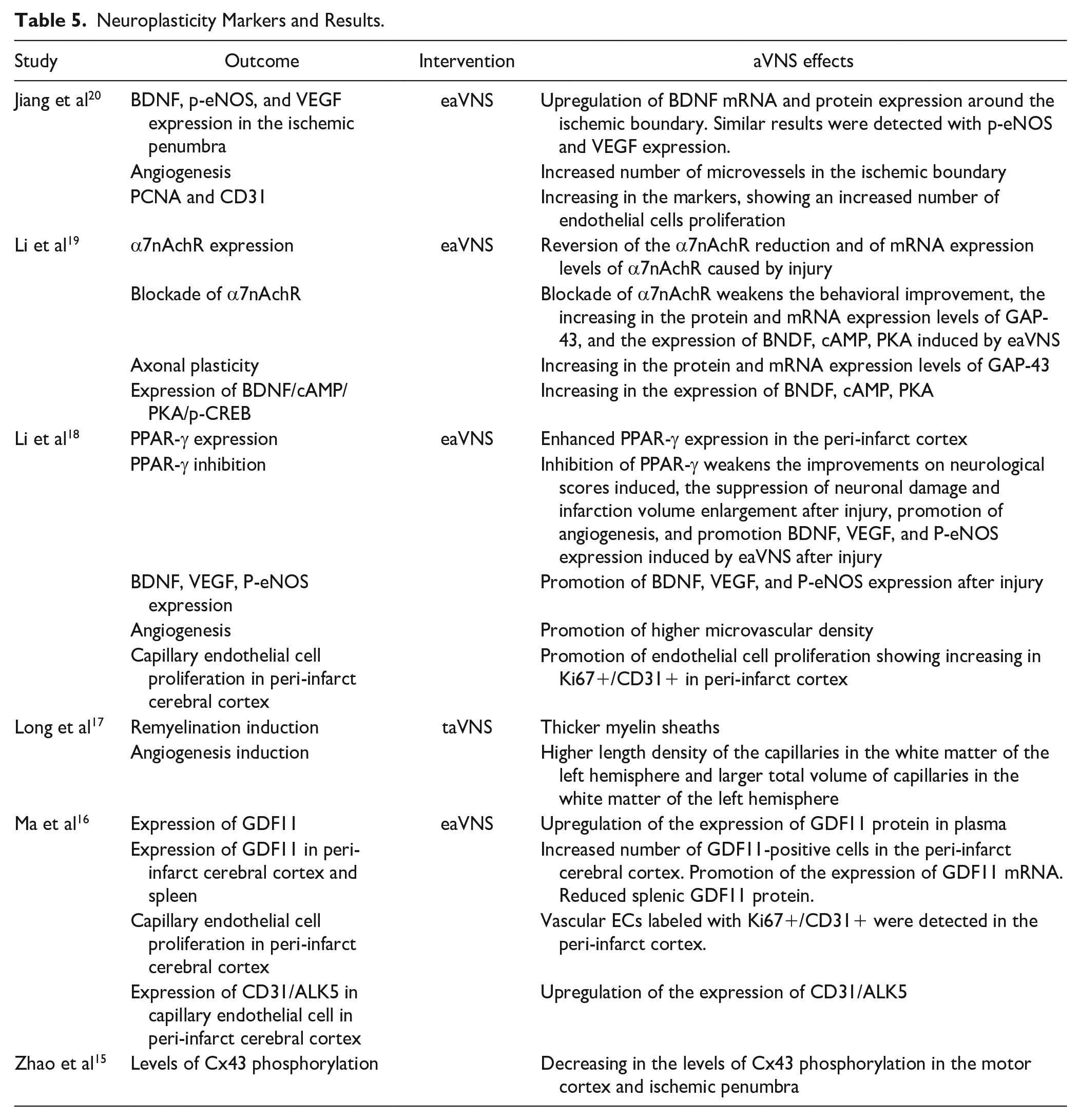
Different types of neuroplasticity markers were measured in the studies included in this review. Three studies described an increase in BDNF levels and expression in peri-infarct regions induced by aVNS. Other 3 have shown an increased microcapillary density and CD31/PCNA/KI67+/ALK5 levels suggesting an increase in angiogenesis promoted by aVNS after I/R. Other markers as phospho-endothelial nitric oxide synthase (p-eNOS), vascular endothelial growth factor (VEGF), α7nAch receptor, GAP-43, peroxisome proliferator activated receptor-γ (PPAR-γ), cAMP, PKA, p-CREB, growth differentiation factor 11 (GDF11), Cx43 expression, and length of myelin sheath were also measure. In general, aVNS was able to upregulate these metrics suggesting higher neuroplastic changes and increase in the microvasculature in the intervention group after injury. Li et al have tested the blockade of the α7nAch receptor and PPAR-γ, showing a reduction in the positive aVNS effects (Table 5).
Neuroplasticity Markers and Results.
Quantitative Analysis
We were able to quantitatively synthesize the aVNS effects in microcapillary density, CD31 proliferation, and BDNF protein levels and RNA expression from the studies. aVNS showed to increase microcapillary density (SMD = 2.15, 95% CI 0.34 to 3.96, I2 = 78%), increase CD31 proliferation (SMD = 3.16, 95% CI 0.59 to 5.73, I2 = 87%), and BDNF protein levels (SMD = 4.11, 95% CI 1.01 to 7.22, I2 = 89%) and RNA expression (SMD = 4.51, 95% CI 1.25 to 7.78, I2 = 88%), showing a high heterogeneity between studies in all results (Figure 3).
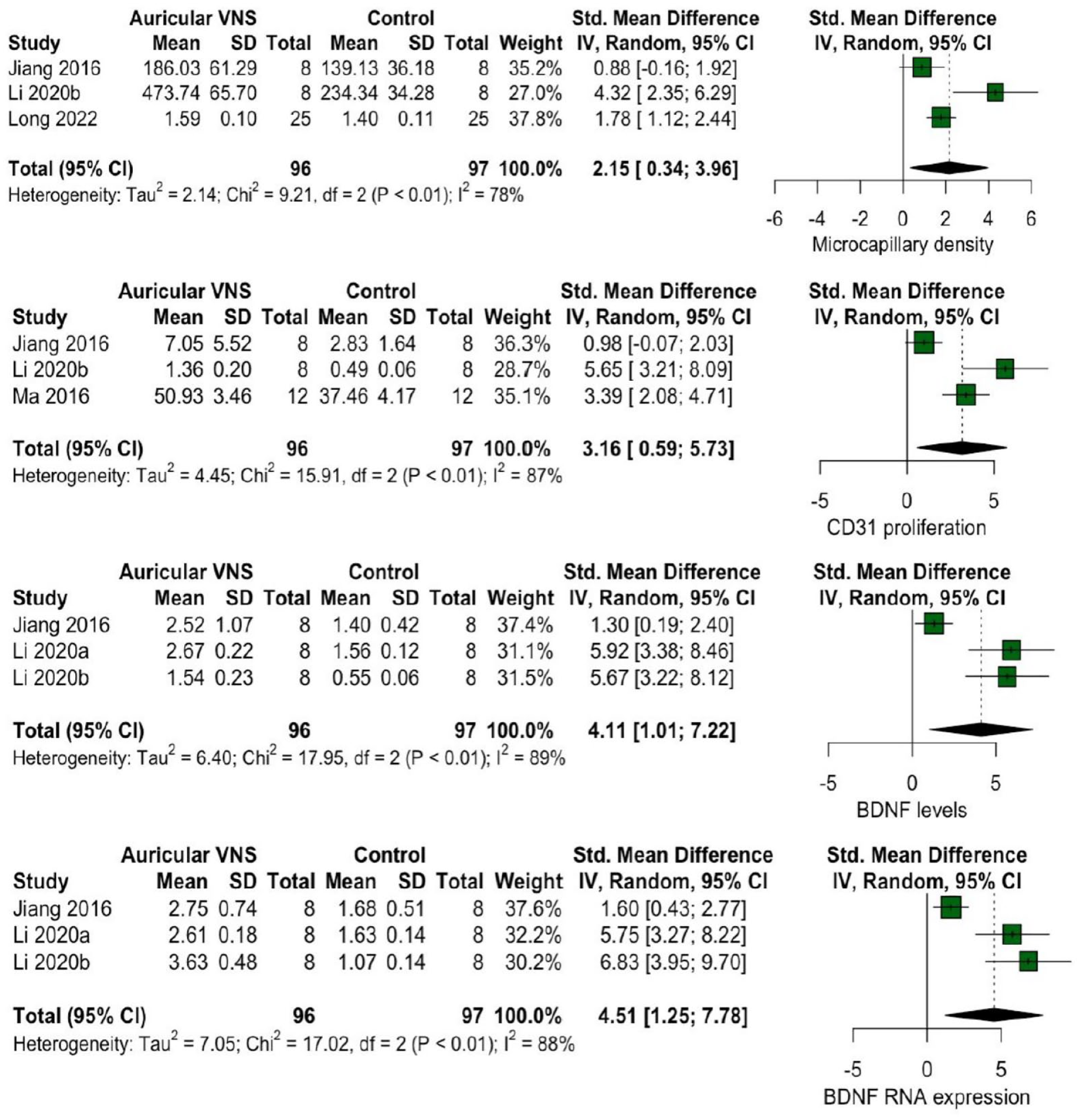
Forest plots of aVNS effects on microcapillary density, CD31 proliferation, BDNF protein levels and RNA expression.
Other Mechanistic Outcomes Related With aVNS Effects
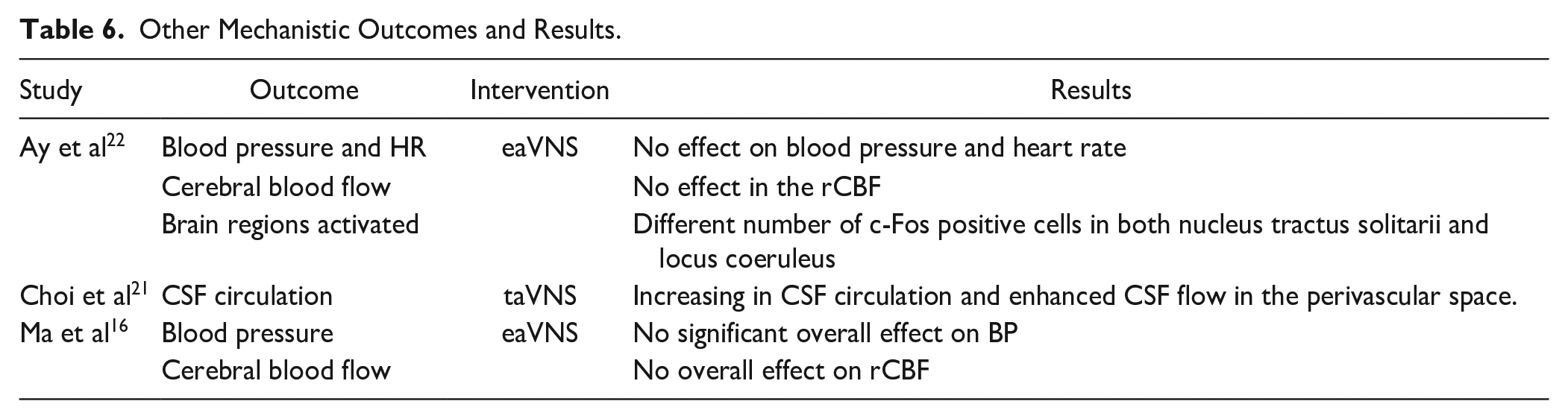
Other ungrouped mechanistic outcomes included blood pressure and HR, cerebral blood flow, brain regions activated, and cerebrospinal fluid (CSF) circulation. Ay et al and Ma et al could not detect an eaVNS effect on blood pressure, HR, and cerebral blood flow. Choi et al have demonstrated an increase in CSF circulation induced by taVNS. Finally, Ay et al demonstrated the activation in nuclei tractus solitarii and locus coeruleus induced by eaVNS (Table 6).
Other Mechanistic Outcomes and Results.
Publication Bias
Regarding the main outcome, neurologic deficit scores, we aimed to assess the presence of publication bias. All the 6 studies that provided data on the score were included in the analysis. The funnel plot built shows a slight asymmetry in the button of the graph suggesting that a publication bias may be present overestimating the intervention effect. However, this analysis may be compromised by the low number of studies (Figure 4).
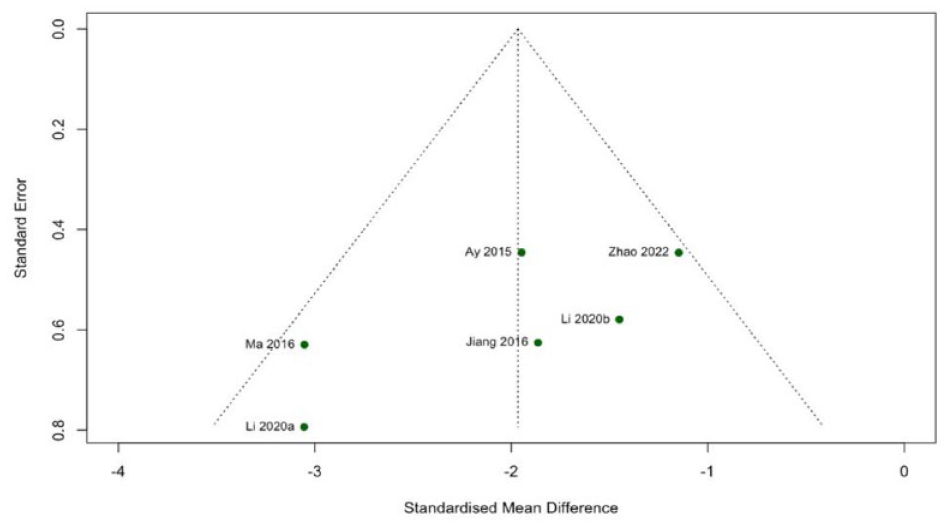
Funnel plot based on Neurological Deficit Scores (main outcome) to assess publication bias.
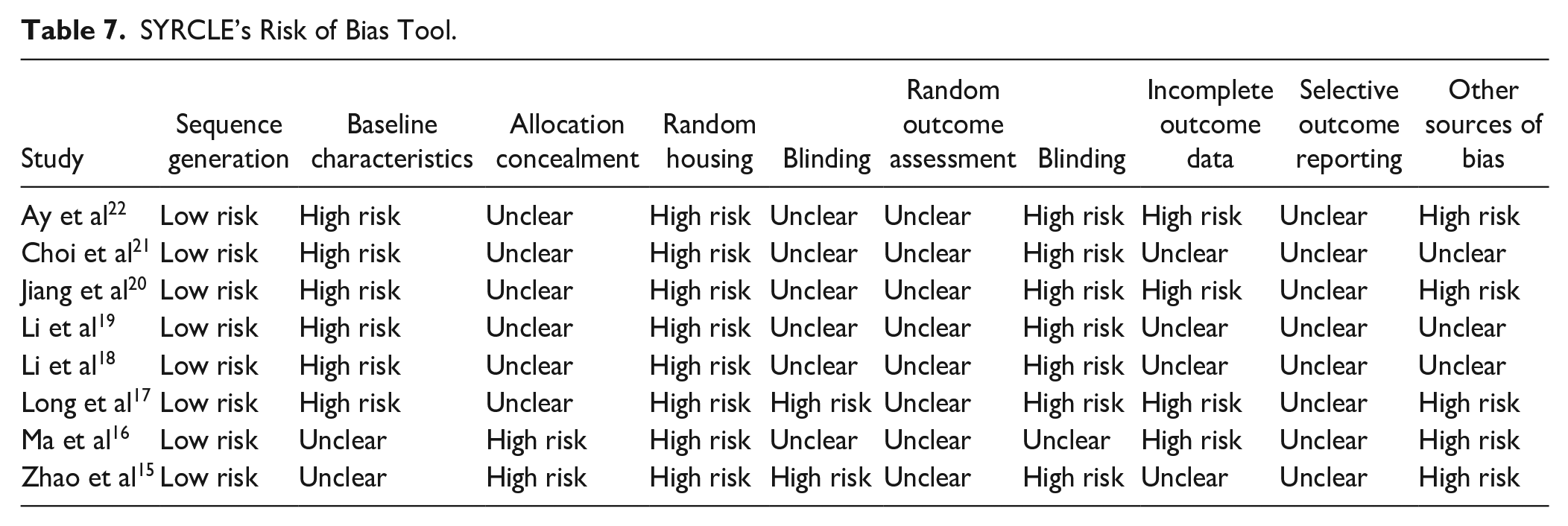
Risk of Bias
The risk of bias was evaluated by the SYRCLE’s risk of bias tool, classifying 10 domains in low risk, unclear risk, or high risk of bias. As demonstrated in Table 7, the domain with lower risk of bias was the sequence generation. On the other hand, random housing was the domain with more studies classified as a high risk of bias. In general, all studies presented at least 3 domains classified as a high risk of bias and most of the domains presented an unclear risk (Table 7).
SYRCLE’s Risk of Bias Tool.
Discussion
In this review, we analyzed studies testing the effects of auricular vagus nerve stimulation in an induced ischemic-reperfusion animal models, the pooled results suggest that the intervention reduce motor and sensory deficits, infarct size, inflammatory outcomes, and induces changes in neuroplastic in the post-stroke recovery period. A total of 8 studies were included in the review, 6 of which investigated neurological deficit scores. A pooled analysis of these 6 demonstrated that aVNS stimulation reduced neurological deficits in I/R models. These studies had a moderate heterogeneity (44%) and a significant standardized mean difference with a relatively small CI (SMD = −1.97, 95% CI −2.57 to −1.36). Regarding the 3 studies analyzing the adhesive removal test, the animals that underwent active stimulation had significantly improved sensitivity and dexterity compared to the ones that had sham (SMD = −2.26, 95% CI −4.45 to −0.08), although the pooled results had a high heterogeneity (81%). There was also a decrease in infarct size (SMD = −1.51, 95% CI −2.42 to −0.60), with a moderate heterogeneity (58%). An increase in microcapillary density (SMD = 2.15, 95% CI 0.34), CD31 proliferation (SMD = 3.16, 95% CI 0.59 to 5.73), an increasing in BDNF peri-infarct enzyme concentration (SMD = 4.11, 95% CI 1.01 to 7.22) and RNA expression SMD = 4.51, 95% CI 1.25 to 7.78) was also present. All these results had high heterogeneity.
A sensitive analysis of the neurological deficit and infarct size was made omitting each study at time, the results did not change significantly in none of these situations, creating an understanding of congruence between the trials. Also, the risk of bias was analyzed by 2 separate authors that needed to agree with the evaluation. The SYRCLES’s RoB tool for animal studies was used analyzing 10 domains regarding the risk of bias in the trials. The domain with less concerns was the one testing the allocation sequence generation and the one with higher risk of bias was the domain testing the randomness of the animal’s selection. In general, all studies had an unclear to high risk of bias in most of the domains.
When compared to previous studies in invasive and non-invasive cervical stimulation, this meta-analysis conveyed similar clinical, neuroprotective, and anti-inflammatory results regarding aVNS.28 -32 However, aVNS has been demonstrated to be a more feasible, accessible, and a safer therapy. 12
AVNS Mechanisms of Behavioral Improvement
As demonstrated in our pooled results, behavioral outcomes were improved after auricular vagal nerve stimulation.
The direct comparison between infarct size of animals that underwent aVNS after the I/R model suggests that the intervention creates a morphological environment of neuroprotection. All the studies show an anatomically smaller lesion in the active groups when compared to the controls.
The improvement mechanisms can be explained by the secondary outcomes analyzed in this review. The 2 main hypotheses behind the aVNS effects on subacute stroke are the induced neuroprotection and neuroplasticity. The immune system plays an important role in the physiopathology of the acute and subacute ischemic stroke producing and releasing different cytokines and chemokines, such as interleukin 1b (IL-1b), interleukin 6 (IL-6), and tumor necrosis factor alpha (TNF-α).33,34 In the subacute phase, local leukocytes release these factors, amplifying the brain-inflammatory response, more leukocytes activation, disruption of the BBB, brain edema, and neuronal death.33,34 The vagal nerve stimulation has been demonstrated to modulate the immune system inducing anti-inflammatory states. A recent systematic review and meta-analysis conducted by our group detected the role of the vagal nerve stimulation in modulating IL-1b, IL-6, IL-10, and interferon gamma (IFN-g) in humans. We also could detect a role of the vagal stimulation in reducing C-reactive protein, a protein related with inflammatory states and widely used in clinical practice (A mechanistic analysis of the neural modulation of the inflammatory system via vagus nerve stimulation: A systematic review and meta-analysis—in submission).
The other mechanism behind the behavioral effects of the vagal nerve stimulation may be through the neuroplasticity induction. Plasticity is the ability to adapt in a changing environment. In pathological circumstances, the lack of neuroplasticity or a maladaptive reaction may prevent recovery. Regarding ischemic stroke, the reorganization of cortical representations on the surroundings or contralateral hemisphere occurs.35,36 This reorganization can be directed to functional recovery enhancing the specificity of the readaptation targeting renormalize the circuitry that suffered maladaptation. 37 Different neuromodulatory techniques have shown to be able to induce neuroplasticity in different neurological conditions, such as traumatic brain injury, chronic pain, and stroke.38 -40 One of the biomarkers widely related and used to measure neuroplasticity is the BDNF,41 -43 responsible for neuron survival. 44 When compared to sham, our meta-analysis conveyed that, animals receiving active stimulation presented higher expression of BDNF enzyme in the periphery of the ischemic area. This neurotrophic factor is associated with the development of dendrites structures in the neurons, increasing their size and quantity. 45 It has also been demonstrated that BDNF is crucial for adult neurogenesis, with infusion of BDNF in the hippocampus of rats demonstrating an increased number of newborn neurons. 46 This BDNF induced neurogenesis has also been linked to increases of long-term potentiation, facilitating communication between the new cells and their functioning. Therefore, the fact that this substance is increased in the periphery of an ischemic area after vagal stimulation, is very likely one of the mechanisms by which the intervention increases neuroplasticity. Although the BDNF quantification was taken only from peri-infarct tissue, it is possible to rationalize that the increased transcription of this enzyme is present throughout the nervous system. This is possible because the intervention is systemic, therefore it is not likely that only the infarct tissue has an increase in this protein.
In addition, our analysis conveyed that, animals in the aVNS group showed higher microcapillary density and an increase in CD31 together with different markers after stimulation. CD31 has been demonstrated to be a sensitive and specific marker in vascular differentiation.47,48 Therefore, the increase of this marker together with the microcapillary density may be associated with a greater endothelial proliferation and vascular support that could be protecting the neurons from the ischemic event.
Individual studies demonstrated other factors modulated by VNS not included in the meta-analysis that corroborate with our main hypothesis, such as increasing in VEGF, p-eNOS, PPAR-γ, and GDF11, important peptides related to cellular growth, angiogenesis, vascular relaxation, suppression platelet aggregation, proliferation of vascular smooth muscle, and neuroprotection.49 -52 Furthermore, some studies have demonstrated aVNS as a modulator of transcription factors such as cAMP, PKA, p-CREB, and α7nAChR, involved neurogenesis, increased axonal regeneration and reorganization, and reinforcing the hypothesis behind the aVNS neuroplastic induction.19,53 -57 However, in humans there are studies only testing the chronic effects of aVNS, and not the acute or subacute effects explored in this review. Therefore, this hypothesis and the demonstrated effects must be tested in future mechanistic randomized clinical trial in humans combined with the currently applied gold standard interventions.
Future Perspectives of aVNS
TaVNS is a promising intervention since it is non-invasive, affordable, usable, and it is effective to modulate vagal tone which would induce potential neural reorganization, and anti-inflammatory effects, allowing neuroplastic changes and reflecting the reduction of neurological deficits. According to several studies,58 -60 taVNS is a safe alternative to the previous surgical device. Our recent meta-analysis of 167 studies assessing 5,827 subjects showed that this technique is a safe and feasible option for intervention, with a low incidence of adverse events and no severe adverse events reported associated with the stimulation. 12
TaVNS can affect many of the bodily processes that the vagus nerve is engaged in, meaning it could be an intervention to a broad range of diseases, such as depression, diabetes, Alzheimer’s disease, arthritis, and stroke. For instance, a recent systematic review performed by our group has demonstrated the taVNS electroencephalographic effects current stage of evidence and the lack of homogenous studies and standardized metrics in the field. 9 Thus, well designed, and multimodal mechanistic studies to explore the ways that taVNS works are required. A possible mechanism that has been studied recently is the modulatory effects of vagus stimulation in the cholinergic anti-inflammatory pathway, the microbiota-gut-brain axis, or the hypothalamic-pituitary-adrenal axis.61,62 In one of our mechanistic meta-analyses on the field, we found that VNS significantly affected inflammatory cytokines and biomarkers including CRP, IL-10, and IFN-g. Additionally, taVNS had a modulatory influence on IL-1b, IL-6, and IL-10, according to subgroup analysis. (A mechanistic analysis of the neural modulation of the inflammatory system via vagus nerve stimulation: A systematic review and meta-analysis—peer review.) In the stroke field, to clarify taVNS’s therapeutic benefits on stroke, translational clinical trials in humans focusing on how the stimulation is able to reduce behavioral/neurological deficits acutely, subacutely, and chronically are necessary. To further examine the long-term efficacy and long-lasting mechanistic consequences, investigations with a larger number of sessions and assessments after longer periods are required.
Conclusion
This systematic review and meta-analysis gather the preclinical evidence of the effects of aVNS on stroke in rats. We demonstrated that aVNS reduced neurological deficits and improved sensorimotor performance after I/R induction in comparison to controls. These findings were corroborated by a reduction of infarct size, induction of angiogenesis, and increased neuroplastic markers promoted by aVNS, indicating the 2 main mechanisms of neuroprotection and neuroplasticity behind the behavioral neurological improvements. However, the results presented a high heterogeneity and risk of bias. Therefore, future well designed randomized clinical trials are needed in order to translate the pre-clinical effects of aVNS on stroke.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231177595 – Supplemental material for Understanding the Neuroplastic Effects of Auricular Vagus Nerve Stimulation in Animal Models of Stroke: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-nnr-10.1177_15459683231177595 for Understanding the Neuroplastic Effects of Auricular Vagus Nerve Stimulation in Animal Models of Stroke: A Systematic Review and Meta-Analysis by Paulo S. de Melo, MS, João Parente, MS, Ingrid Rebello-Sanchez, MS, Anna Marduy, MS, Anna Carolyna Gianlorenco, PhD, Chi Kyung Kim, PhD, Hyuk Choi, PhD, Jae-Jun Song, PhD and Felipe Fregni, PhD in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
CKK, HC, and JS are directly associated with Neurive Co, a company developing neuromodulation technologies, such as taVNS, to treat common brain diseases.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: ACG is supported by the Federal University São Carlos and is a consultant for Neurive. PSdM JV, IR-S, and AM are consultants for Neurive. FF is supported by NIH grants, by a research grant and gift from Neurive to Spaulding Rehabilitation Hospital, and is a consultant for Neurive.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
