Abstract
The effects of low-frequency repetitive transcranial magnetic stimulation (LF-rTMS) on treating poststroke aphasia (PSA) remain inconclusive. We aimed to evaluate the efficacy and safety of LF-rTMS on language function poststroke and determine potential factors that may affect treatment effects. Electronic databases, including MEDLINE, EMBASE, and Cochrane Library were searched to identify relevant randomized controlled trials (RCTs) concerning the effects of LF-rTMS on language performance poststroke. We adopted fixed- and random-effects models to estimate intervention effects, which were represented by the Hedges’ g and 95% CIs. Subgroup analyses regarding several factors potentially influencing the effects of LF-rTMS on language recovery were also conducted. A total of 14 RCTs involving 374 participants were included in the meta-analysis. The pooled analysis showed the positive and significant effects of LF-rTMS on language function, both short-term (Hedges’ g = 0.65; P < .05) and long-term (Hedges’ g = 0.46; P < .05). Subgroup analyses demonstrated that LF-rTMS for 20 minutes per day over 10 days yielded the largest effect size (Hedges’ g = 1.02; P < .05) and that LF-rTMS significantly improved language performance in the chronic stage after stroke (Hedges’ g = 0.55; P < .05). Patients with different native languages might have diverse responses to LF-rTMS treatment efficacy. Additionally, there were significant improvements in language subtests, including naming, repetition, comprehension, and writing. Overall, this updated meta-analysis demonstrated that LF-rTMS has significant positive effects on PSA, with moderate treatment effects. It provides additional evidence to support LF-rTMS as a promising complementary therapy to promote language recovery in PSA.
Introduction
Aphasia affects approximately 38% of survivors after stroke, and it causes serious communication disability, reduces the quality of life, and increases the risk of dependent living.1-3 Speech and language therapy (SLT), a common rehabilitation approach, can effectively restore some degrees of functional communication abilities after stroke4-6; but SLT is labor-intensive, incurring high cost to patients, with a modest effect size. 4 There is a lack of more effective treatment methods to augment SLT for poststroke aphasia (PSA); thus, searching for potential adjunctive and therapeutic strategies has become a health care imperative. Noninvasive brain stimulation (NIBS), such as transcranial direct current stimulation (tDCS) and transcranial magnetic stimulation (TMS), has been reported as a promising technique to augment traditional SLT for PSA. 7 Anodal and cathodal tDCS (or both) was administered for 20 minutes, with a current ranging from 1 to 2 mA paired with SLT to modulate cortical excitability and hence to report positive effects as compared with sham in most previous studies.8-10 However, several studies showed that tDCS had no statistically significant difference relative to sham. No current evidence demonstrated the effectiveness of tDCS for improving functional communication in people with aphasia after stroke. 11
Repetitive TMS (rTMS) has emerged as another promising NIBS tool to augment traditional SLT efficacy for PSA.7,12 rTMS can be delivered as high-frequency rTMS (HF-rTMS) with excitatory effect and low-frequency rTMS (LF-rTMS) with inhibitory effect. Up to now, only limited randomized controlled trials (RCTs) have investigated the influence of HF-rTMS on the reorganization of language functions in PSA. 7 In contrast, a growing body of research has suggested that LF-rTMS applied to the contralesional cortex may be capable of potentially facilitating language recovery after stroke. 13 However, the efficacy of LF-rTMS integrated with an SLT protocol has yet to be determined. Two published meta-analysis article were published with 8 included studies before 2015.14,15 However, a series of studies have been published with LF-rTMS in PSA since then, additional LF-rTMS parameters have been further optimized, and patient selection criteria have improved. There is a need to conduct an updated meta-analysis to reassess the summary effects of LF-rTMS on PSA. Therefore, we aimed to conduct such a meta-analysis to clarify the effects of LF-rTMS and several potential factors on language outcomes in PSA. Specifically, we sought to determine (1) whether LF-rTMS has favorable immediate or long-term effects on language performance (including naming, repetition, comprehension, and writing) and (2) how the time after stroke onset, first mother language of patients, and intervention time affect the effects of LF-rTMS on language outcomes of PSA.
Methods
Search Strategy
A comprehensive search of the literature was performed by using online databases MEDLINE, EMBASE, and Cochrane Library of human studies published in English or Chinese journals from inception up to August 31, 2019, to identify all relevant RCTs. The keywords of the search were combined with the following terms: (aphasia OR language disorders OR anomia OR linguistic disorders OR speech disorders) AND (transcranial magnetic stimulation OR TMS) AND (stroke OR cerebrovascular accident OR brain ischemia OR poststroke OR post-stroke). Additionally, reference lists from retrieved articles as an additional source were manually searched to identify other potentially relevant studies.
Study Selection
The search results were evaluated according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 16 Studies had to meet the following inclusion criteria: (1) enrollment of stroke survivors who were ≥18 years old and were diagnosed with stroke (either ischemic or hemorrhagic stroke); (2) study design was a RCT that included a clearly defined control group and an experimental condition; (3) the experimental group received LF-rTMS alone or LF-rTMS plus SLT, and the control group either underwent sham LF-rTMS alone or sham LF-rTMS plus SLT; (4) duration of training was ≥10 days to provide sufficient time for language benefits to manifest; (5) study included a validated neuropsychological test to evaluate the degree of language impairment, with data reported at baseline and postintervention; (6) studies were published in English.
Data Collection and Extraction
One investigator (ZH) conducted the primary search and excluded titles and abstracts that were not relevant to the content of the review. Two other investigators (HZ and JL) independently evaluated the remaining titles and abstracts and obtained the full text for all abstracts to determine whether these studies met the inclusion criteria. Subsequently, they independently reviewed the full-text articles and selected studies only if all inclusion criteria were met. Data from the original documents were extracted independently by 2 investigators (MY and YA) using a standard data recording and then were independently rechecked by the third investigator (BD). Any disagreement was resolved through discussion with the third reviewer (XH). The type of data systematically extracted from each eligible study were as follows: study information (design, year, inclusion criteria, number of participants), participant characteristics (age, gender, stroke duration, stroke location, and aphasia type), intervention protocol (rTMS frequency, intensity, number of pulses, and additional interventions), outcome measures (eg, Boston Diagnostic Aphasia Examination, Aachen Aphasia Test), and data regarding study quality. The primary outcome was measures to assess language function. If more than 1 measure was used in an individual trial, the primary outcome reported with a mean and SD in the results section was used. ZH also emailed corresponding authors for information that was not available in the articles; the study was excluded if authors did not respond after 2 emails were sent.
Quality Assessment
Two investigators (ZH and JL) independently evaluated the methodological quality of each study based on the Cochrane risk-of-bias tool, which is described in the Cochrane Handbook for Systematic Reviews of Intervention (version 5.1.0). 17 The Cochrane tool specifies 7 domains of potential bias, including sequence generation, allocation concealment, blinding of participants and personnel, incomplete outcome data, selective outcome reporting, and other biases. Furthermore, risk of bias and quality of evidence were assessed per domain and classified as low risk, unclear (insufficient detail or not reported) risk, or high risk of bias on the basis of the published study reports.
Effect Sizes Estimation
For data regarding continuous outcomes, the effect size was calculated by reporting the standardized mean difference (SMD) with 95% CIs because a single outcome measurement was assessed and reported across trials using different measurement tools. However, Hedges’ g was adopted to represent the intervention effects of each study because it is a bias-adjusted calculation of the SMD that applies an additional correction for small-sample studies that we included. Hedges’ g for effect size was given by using the mean change in performance from baseline in the control group with respect to the experimental group and dividing the difference by the pooled SD of the change from baseline.18,19 The pooled effect size was interpreted as small (≤0.2), moderate (0.5), or large (≥0.8) based on the Cohen criteria. 20
Quantitative Data Synthesis and Analysis
For quantitative synthesis, the pooled effect estimation was calculated by comparing the change from baseline to end point of the study between the intervention and control groups. For trials reporting multiple outcomes about language function measurements, we assessed whether rTMS resulted in significant language gains compared with sham samples using the total score of each scale. Besides, several subgroup analyses were performed to explore whether sample and study characteristics had the effects of rTMS on language performance using random-effects models. Recruitment sample characteristics included the length of time from stroke to initiation of the intervention (≤3 or >3 months) and first language (English, Chinese, Polish, German, Korean). We also investigated whether the duration of rTMS intervention (10, 15, 20 days) had an effect on the magnitude of the effect size.
Several subtests of language performance, including naming, repetition, comprehension, and writing, were also performed. Five of 14 studies contained follow-up visit information after an intervention. The follow-up effect of rTMS on language performance was conducted to evaluate rTMS-induced long-term benefits on PSA accordingly.
Finally, statistical heterogeneity of effect size across trials was assessed using standard χ2 (Q) and I2 statistics, in which a P value of <.10 and I2 >50% indicated statistically significant heterogeneity among studies. 17 Publication bias was conducted by using funnel plots and the Egger test. Data analyses were carried out using Stata version 13.0 (Stata Corp, College Station, TX). A P value <.05 was considered statistically significant.
Results
Characteristics of Included Studies
A total of 14 RCTs21-34 involving 374 stroke participants were identified based on the inclusion criteria by systematically screening from 2088 published manuscripts. The detailed study selection process is summarized in a PRISMA study flow diagram (Figure 1). The included studies were published from February 2011 to August 2019. Studies varied in sample size, treatment duration, and intervention protocol. Sample sizes ranged from 10 to 56, and the mean age was 63.1 years (SD 11.5). Mean age was similar across rTMS (63.1 years; SD = 11.0) and sham groups (63.1 years; SD = 12.0), and 61% of stroke participants were male, except 3 studies that had not reported gender information. One trial 23 had 2 arms, and these were counted as 2 separate comparison groups. Across 13 studies, the average time since stroke onset was about 7.8 months (SD = 10.8). Only 2 studies22,26 included information about stroke subtypes. Regarding type of PSA, 7 trials21-23,25-27,32 included patients with only nonfluent aphasia, and 7 other trials included all types.26,28-31,33,34
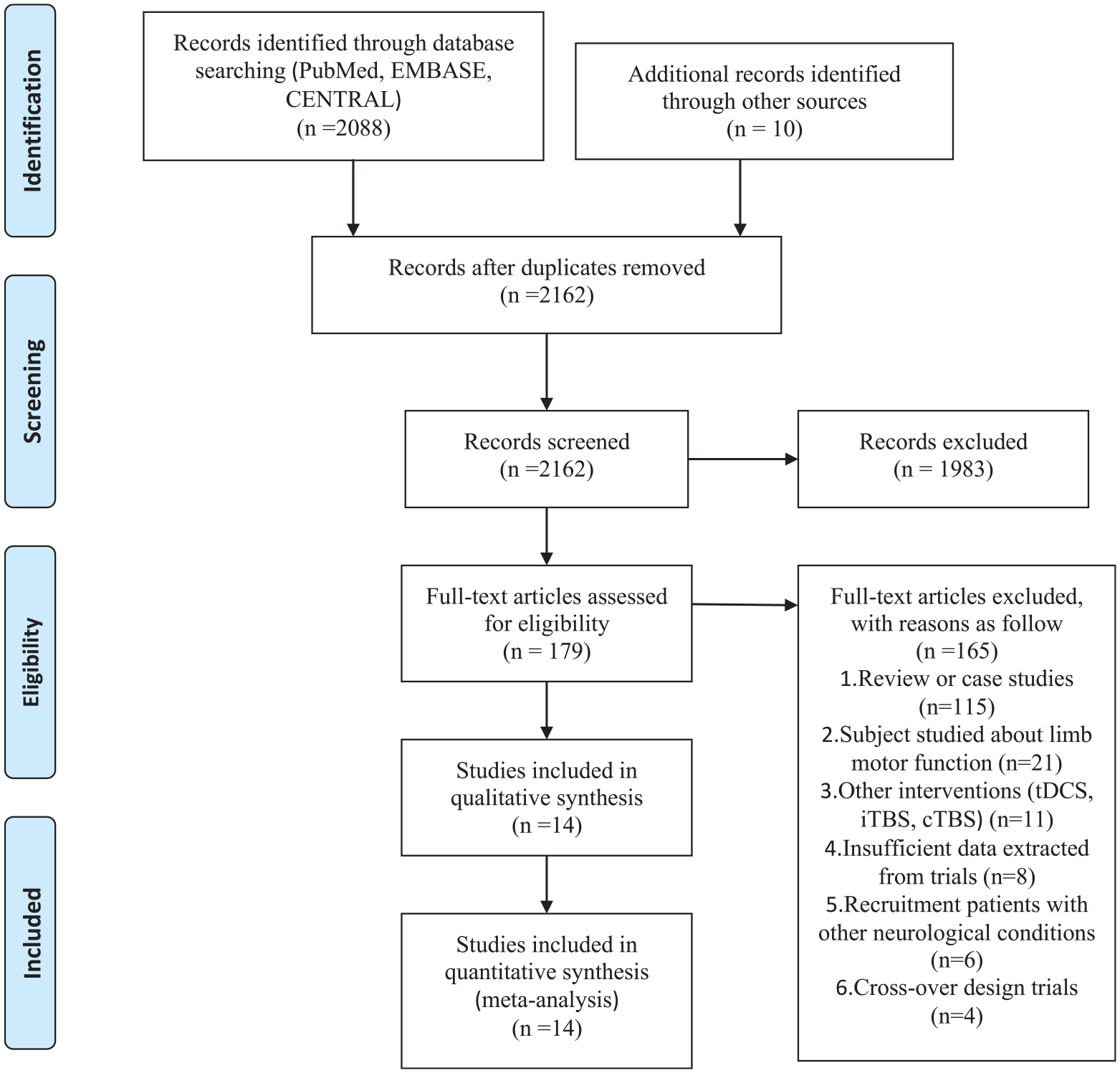
Flowchart of process for study selection following PRISMA.
All 14 studies investigated the effect of LF-rTMS combined with SLT versus sham LF-rTMS combined with SLT except 1 study. 27 The protocol of SLT of each included study was not identical. For example, a duration of 10 sessions of SLT was chosen in 8 studies21,24,25,28,29,31,32,34 and 15 sessions in 3 studies.23,30,33 Most SLT contents of studies emphasized individual linguistic symptoms. In all, 12 trials applied 1-Hz rTMS with an intensity equaling 90% of the daily defined individual resting motor threshold (RMT); 1 trial 21 used 1-Hz rTMS with 100% RMT, and 1 trial 14 used 1-Hz rTMS with 80% RMT. rTMS treatment sessions were 5 days per week for 10 days for 10 studies,21,22,24,25,27-29,31,32,34 15 days for 3 studies,23,30,33 and 20 days for 1 study. 26 Regarding the stimulus site of rTMS, 14 studies targeted the triangular part of the right inferior frontal gyrus, whereas 1 study 23 stimulated the right superior temporal gyrus. One RCT reported by Hu et al 22 compared the effects of HF-rTMS and LF-rTMS targeting the mirror area within the right hemispheric Broca’s area. There were 2 methods to deliver sham stimulation: using an air-cooled sham coil over the targeted area or placing a real rTMS coil over the vertex region. Additionally, there were 7 studies24,25,27,29,31,32,34 using MRI-guided navigation to determine the stimulation position for the TMS coil when applying on the language-related regions. Safety is an indispensable consideration because rTMS can induce several potential adverse effects, such as seizures, headaches, and skin pain. Thus, we reported adverse effects in the present meta-analysis. For the 8 studies22,25,26,30-34 that mentioned adverse events, only 2 of them22,25 reported dizziness and dull pain during the rTMS intervention sessions. For all trials, 8 of 203 (3.9%) participants in the LF-rTMS group and 6 of 171 (3.5%) in the sham group dropped out of the study during the training. Further information regarding study characteristics are shown in Table 1.
Summary Characteristics of Included Studies.
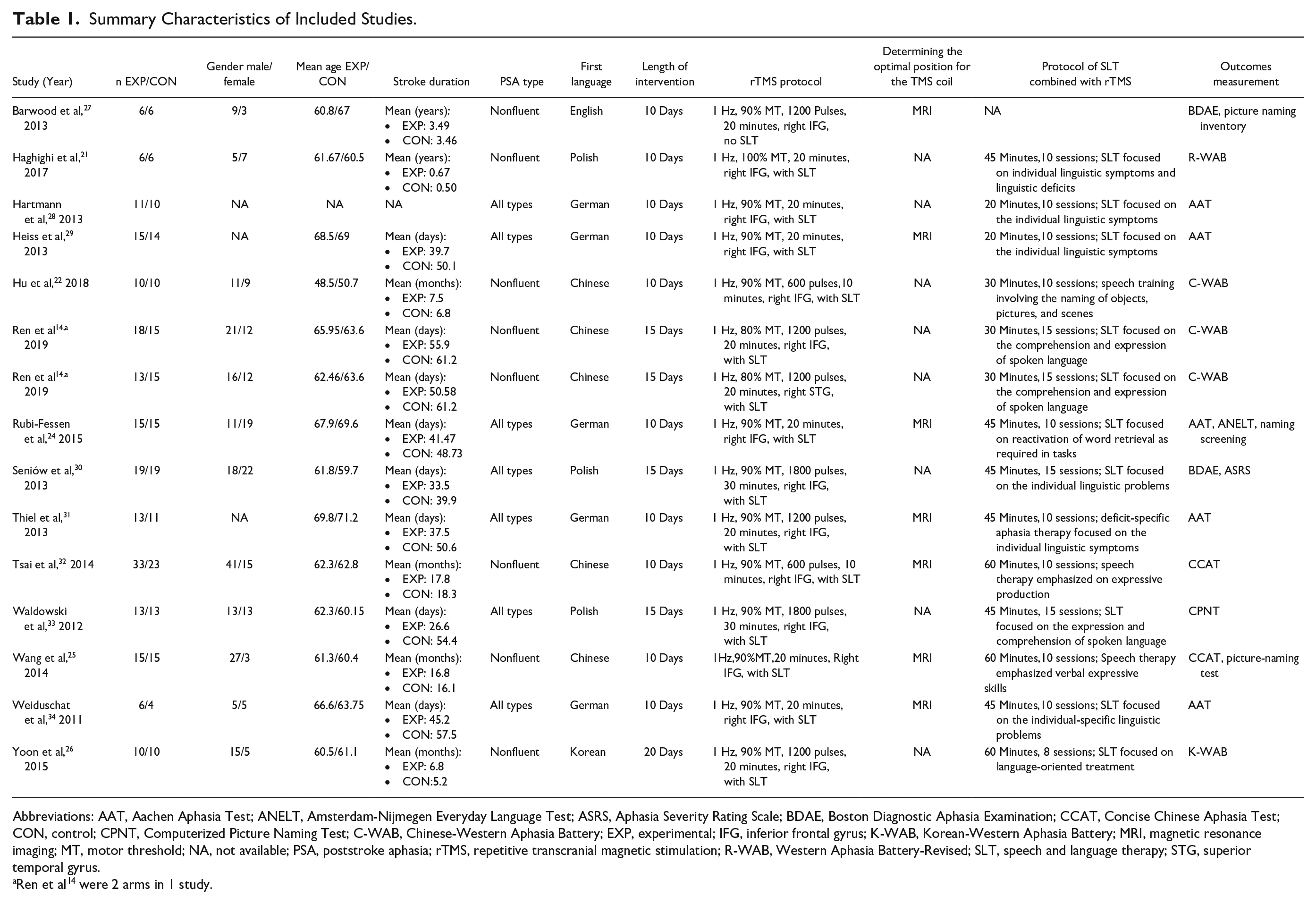
Abbreviations: AAT, Aachen Aphasia Test; ANELT, Amsterdam-Nijmegen Everyday Language Test; ASRS, Aphasia Severity Rating Scale; BDAE, Boston Diagnostic Aphasia Examination; CCAT, Concise Chinese Aphasia Test; CON, control; CPNT, Computerized Picture Naming Test; C-WAB, Chinese-Western Aphasia Battery; EXP, experimental; IFG, inferior frontal gyrus; K-WAB, Korean-Western Aphasia Battery; MRI, magnetic resonance imaging; MT, motor threshold; NA, not available; PSA, poststroke aphasia; rTMS, repetitive transcranial magnetic stimulation; R-WAB, Western Aphasia Battery-Revised; SLT, speech and language therapy; STG, superior temporal gyrus.
Ren et al 14 were 2 arms in 1 study.
Risk-of-Bias Assessment
The risk of bias is shown in Table 2 and Figure A.10. Generally, 4 of 14 studies23,24,30,32 scored positively on all quality criteria, and several quality criteria were not mentioned or discussed in the remaining studies. Except 2 studies,22,26 the remaining 12 (86%) studies utilized adequate random sequence generation and were classified as low-risk bias. Seven (50%) studies21,23-25,30,32,34 described details of allocation concealment using either a sealed envelope or a confidential random numbers tag; however, the other 7 studies22,26-29,31,33 did not report information of allocation concealment. Blinding of participants and study assessors was reported in 12 (86%) and 13 (93%) of the studies, respectively. One trial 26 was rated as high-risk bias because participants were not blinded, and 2 trials29,34 were assessed as high-risk bias because reasons for the drop-outs were not reported. Information on selective outcome reporting was not mentioned in 3 (21%) studies21,28,33 because of lack of a research protocol, and other bias of study was not reported in 5 (36%) studies25,27-29,34 because baseline balance was not mentioned.
Assessment Quality of Intervention Trials According to the Cochrane Risk-of-Bias Tool.
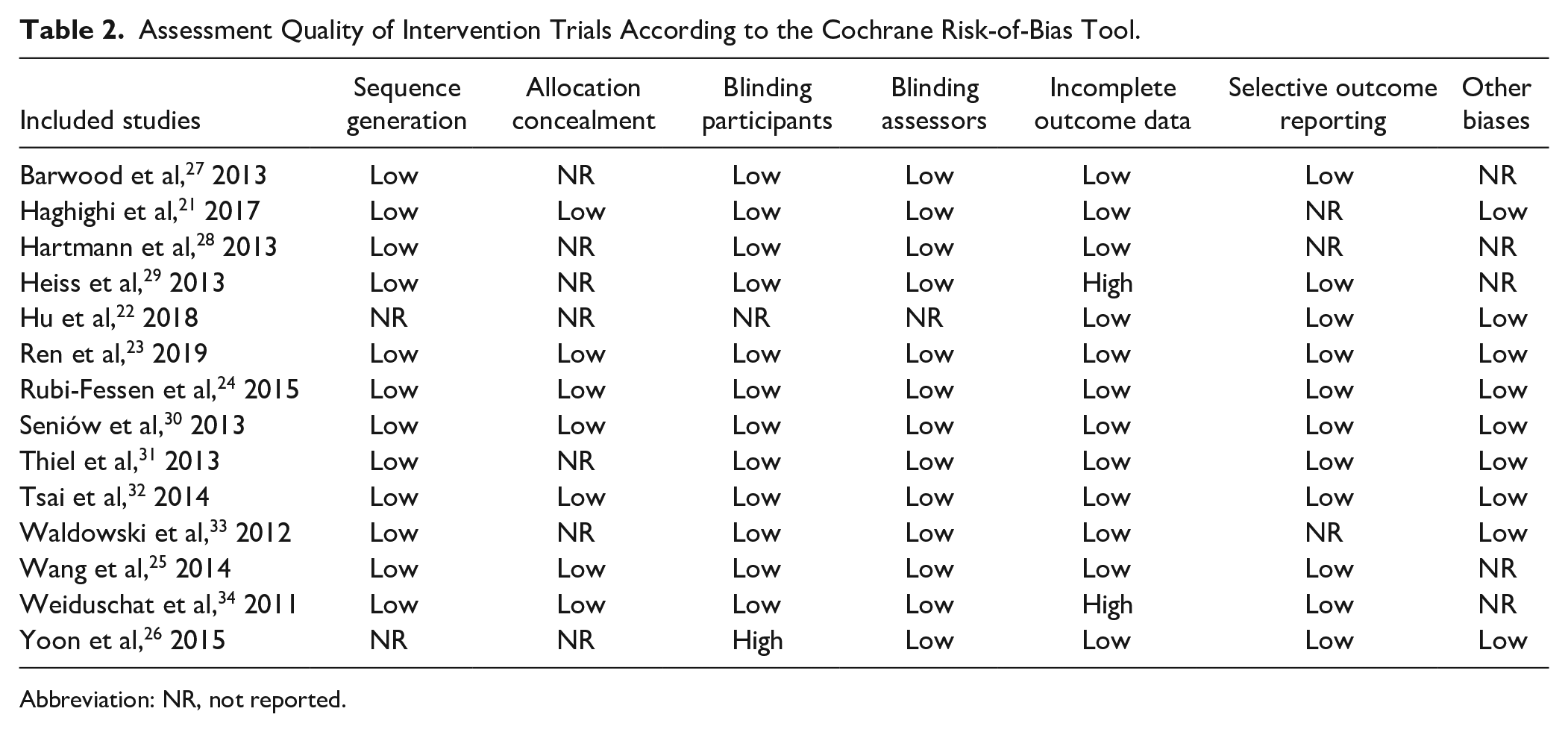
Abbreviation: NR, not reported.
Effects of LF-rTMS on Aphasia
The pooled result showed a significant and positive effect of LF-rTMS on language performance in terms of immediate benefits, with a moderate to large mean effect size (Hedges’ g = 0.65 [0.43-0.86], P < .0001; Figure 2), and no heterogeneity was observed (Q = 13.27; df = 13; I2 = 2%; P = .427). The effect size did not change significantly even when any 1 trial was removed (Figure A.11). Regarding the long-term effect of LF-rTMS, 5 studies22,25,30,32,33 reported follow-up information. One study 22 performed the language outcome assessment after 2 months of the rTMS intervention; 2 studies25,32 performed it at the 3-month follow-up; and 2 other studies30,33 conducted it after 15 weeks of rTMS treatment. A small to moderately significant effect size (Hedges’ g = 0.46 [0.16-0.77], P = .003; Figure 2) without heterogeneity (Q = 2.21; df = 4; I2 = 0%; P = .698) was found with data from 5 trials.
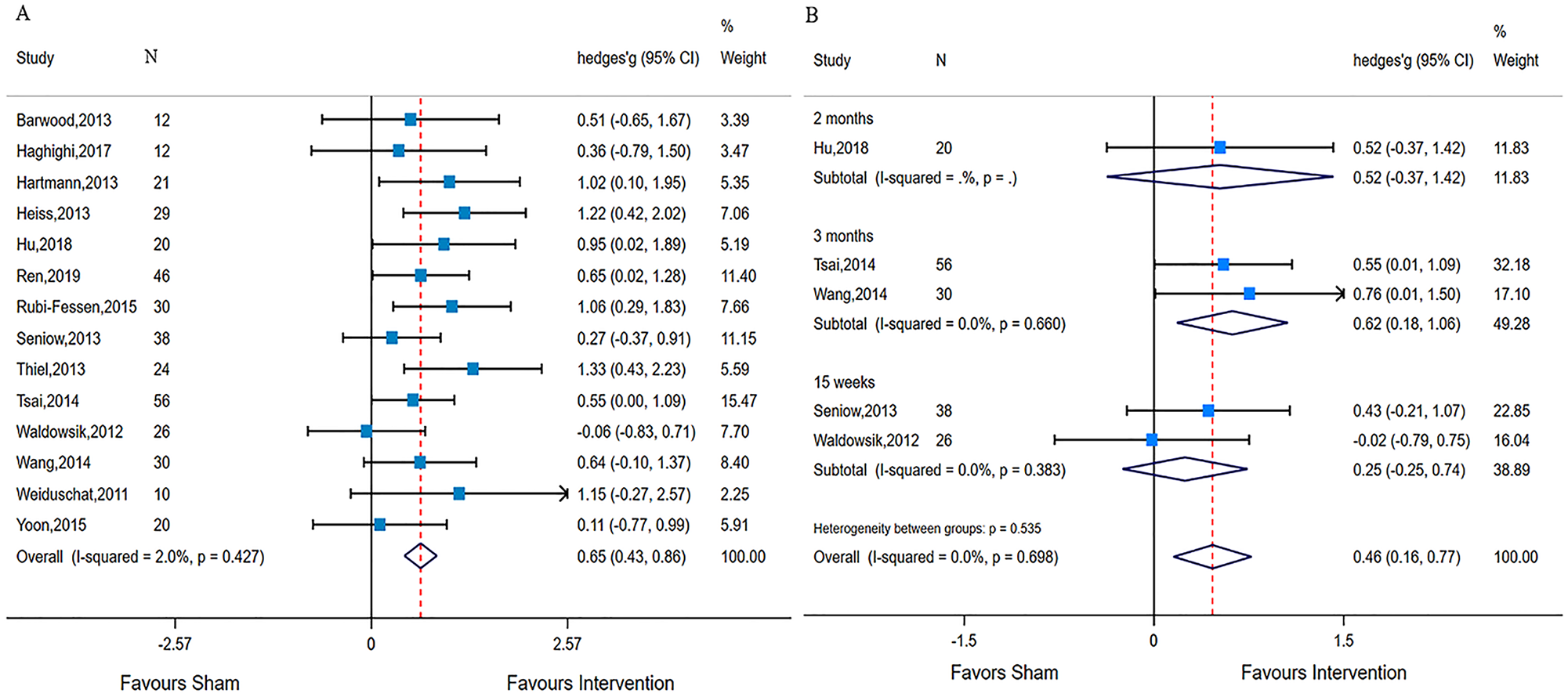
Forest plot showing the intervention effects of low-frequency repetitive transcranial magnetic stimulation on poststroke aphasia relative to sham using Hedges’ g and 95% CIs: A. Immediate effect. B. Long-term effect.
Effects of LF-rTMS on Language Subtests
First, we evaluated expressive language, which contains naming (13 studies), repetition (11 studies), and writing (4 studies). The meta-analysis of expressive language skills indicated statistically significant effects for naming (Hedges’ g = 0.53 [0.31-0.75]) with small heterogeneity (I2 = 3.8%; P = .409), for repetition (Hedges’ g = 0.44 [0.20-0.67]) without heterogeneity (I2 = 0%; P = .984), and for writing (Hedges’ g = 0.74 [0.31-1.16]) without heterogeneity (I2 = 0%; P = .994), respectively. Receptive language was measured by using comprehension (11 studies) and the Token Test (4 studies). LF-rTMS resulted in significant and small to moderate effects on comprehension (Hedges’ g = 0.45 [0.22-0.68]) with small heterogeneity (I2 = 3.9%; P = .406), and it had moderate effects on the Token Test (Hedges’ g = 0.60 [0.18-1.02]) without heterogeneity (I2 = 0%, P = .868; Figure 3). Detailed data are shown in Table 3 and Figures A.1 to A.5 in the Supplemental Material.
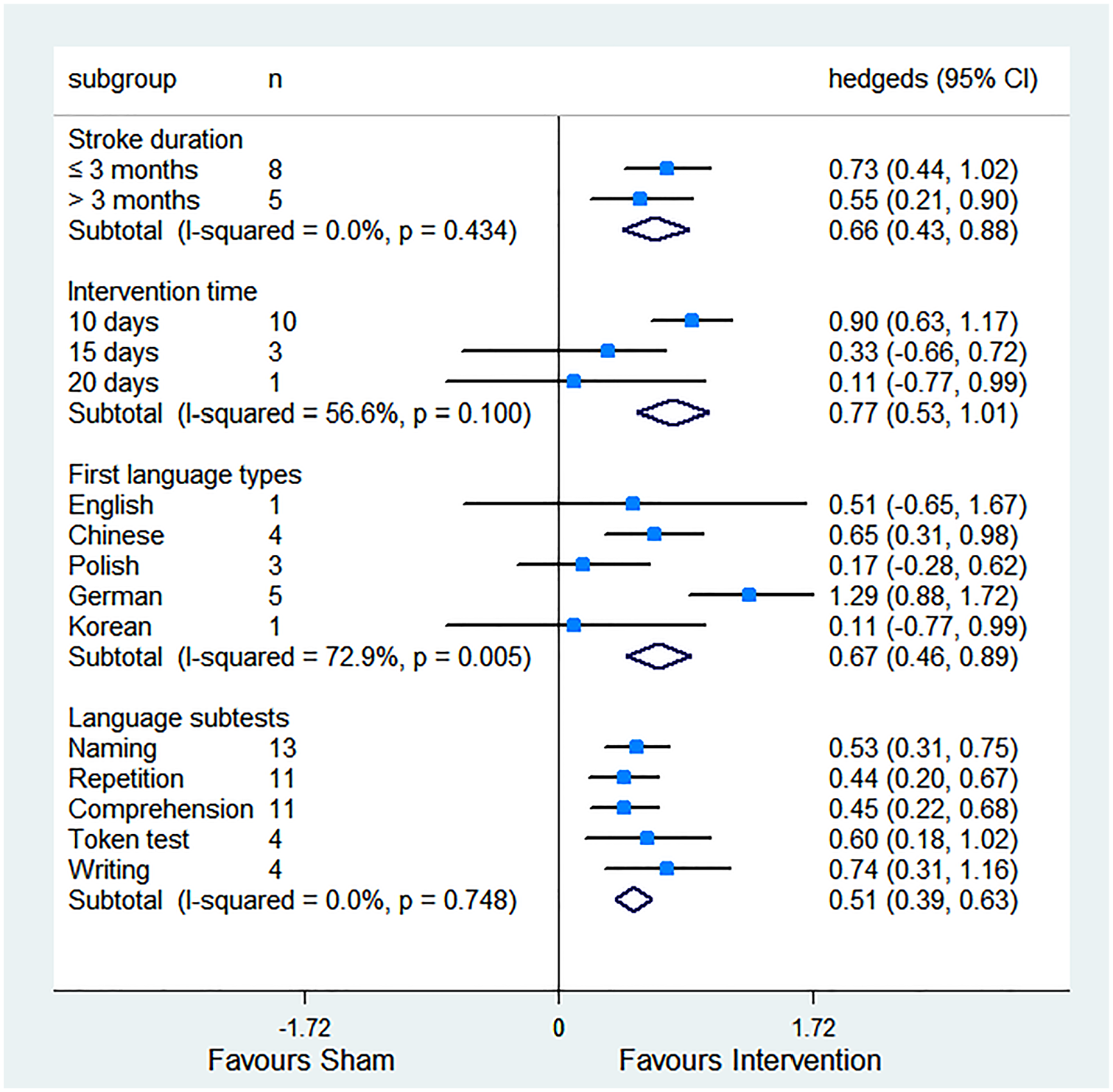
Forest plot showing results from subgroup analyses, including pooled effects for each language subset.
Meta-analytic Results From Included Studies. a
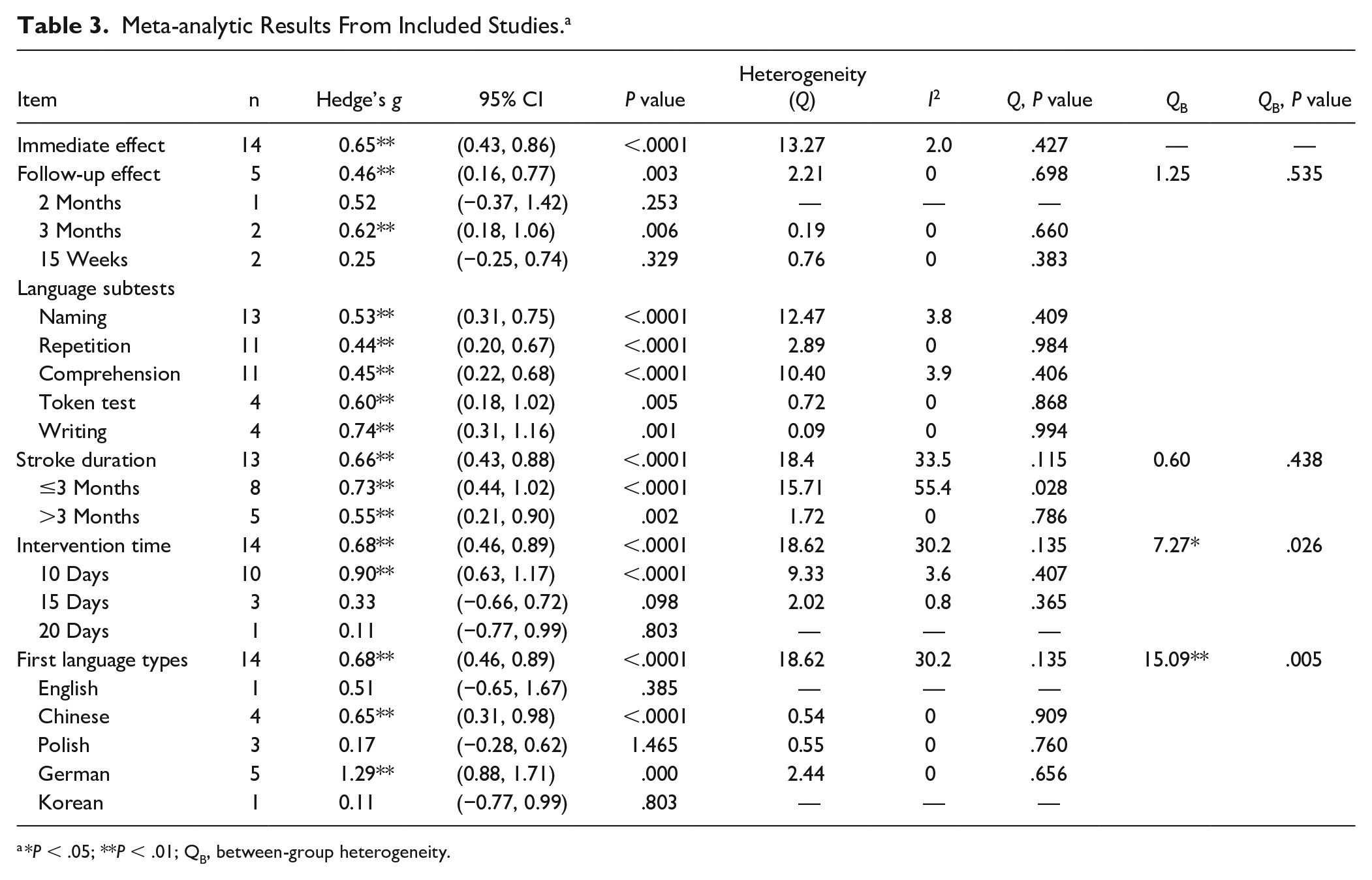
*P < .05; **P < .01; QB, between-group heterogeneity.
Subgroup Analyses
Time From Stroke Onset
We intended to identify whether the magnitude of benefits of rTMS treatment varied in the course of stroke recovery. The time from stroke to intervention ≤3 months including 8 studies ranged from 11 to 81 days. Five studies included chronic stroke survivors, with the average stroke duration >3 months, ranging from 6 to 42 months. The subgroup analyses indicated that initiated rTMS treatment ≤3 months poststroke resulted in a significant and nearly large effect on language function (Hedges’ g = 0.73 [0.44-1.02]) with moderate heterogeneity (I2 = 55.4%; P = .028), whereas there was a significant and moderate effect size for patients in the chronic stroke phase (Hedges’ g = 0.55 [0.21-0.90]) without heterogeneity (I2 = 0%; P = .786). However, no statistical significance in between-groups difference was observed (QB = 0.60, P = .438; Figure 3). (QB, between-group heterogeneity). Detailed data are shown in Table 3 and Figure A.6.
Intervention Duration
Stroke participants with a 10-day course of rTMS treatment experienced the largest benefits (Hedges’ g = 0.90 [0.63-1.17]) with small heterogeneity (I2 = 3.6%; P = .407). Longer interventions (15 days or 20 days more) were not associated with more significant benefits of language function relative to brief interventions (Hedges’ g = 0.33 [−0.66 to 0.72]; Hedges’ g = 0.11 [−0.77 to 0.99]). Further analysis showed that studies with 20 minutes per day of LF-rTMS treatment had a larger effect size of 1.02 (Hedges’ g = 1.02 [0.69-1.36]) than studies with 10 minutes per day (Hedges’ g = 0.65 [0.18-1.12]). Overall, the between-groups difference showed statistical significance (QB = 7.27, P = .026; Figure 3). Detailed data are shown in Table 3 and Figure A.7.
Patient Characteristics
The mother language of stroke survivors was different, although all studies were published in English. Patients with German as the first language experienced the largest benefit (Hedges’ g = 1.29 [0.88-1.71]), and studies with Chinese showed significant, moderate, and positive effects on language function (Hedges’ g = 0.65 [0.31-0.98]). However, there was no significant difference observed in patients whose mother language was English or Polish or Korean, although the between-groups difference did reach statistical significance (QB = 15.09; P = .005). Detailed data are shown in Table 3 and Figure A.8.
Publication Bias Assessment
As shown in the funnel plots in Figures A.9A to A.9G in the Supplemental Material, no publication bias was observed for the immediate efficacy, the long-term efficacy, naming, repetition, comprehension, Token test, or writing (Egger test: P = .365, P = .741, P = .291, P = .497, P = .669, P = .13, and P = .12, respectively).
Discussion
This updated meta-analysis included 6 additional published studies and revealed that LF-rTMS improved language performance in stroke survivors with various types of aphasia. The largest effect size of rTMS was observed in stroke survivors who received rTMS for 20 minutes per day over 10 sessions.
Ren et al 14 conducted the first meta-analysis evaluating the effects of LF-rTMS on PSA and observed a positive and significant benefit of LF-rTMS with 7 included RCTs. Another meta-analysis published by Li et al 15 indicated that LF-rTMS enhanced the naming by analyzing 4 RCTs. A total of 8 RCTs27-34 were included in these 2 meta-analyses. With 6 additional RCTs21-26 being included, we were able to investigate the immediate effect as well as the long-term effect of LF-rTMS. Notably, chronic stroke survivors receiving LF-rTMS treatment about 1.29 years after stroke still exhibited positive and moderate treatment effect. Only 1 RCT 22 of present studies concerning HF-rTMS (10 Hz) reported that the HF-rTMS group showed significantly improved repetition scores compared with the sham stimulation group, but it demonstrated superior therapeutic effect of LF-rTMS over HF-rTMS on naming. Regarding HF-rTMS, another case study 35 found that HF-rTMS targeting on the left IFG improved naming and comprehension in nonfluent patients. Because of relatively few studies, more large-scale RCTs should be conducted to explore the effect of HF-rTMS on PSA.
The highlight of our study was that 10 sessions of LF-rTMS interventions (as compared with 15 or 20 days) had the maximal effect (Hedges’ g = 1.02) on language outcomes. We have to point out that there could be insufficient data to detect the effect in 15 days (3 trials) and 20 days (1 trial). Additionally, it is of note that studies with a 10-day session included stroke patients ≤3 months from stroke onset, whereas none of the studies with 15- or 20-day sessions included stroke patients ≤3 months from stroke onset. We further analyzed and found that introducing 10 days rTMS ≤3 months from stroke onset demonstrated a larger treatment effect than >3 months (Hedges’ g = 1.22 vs 0.63; Figure A.7C). It is possible that rTMS harnessed the dynamic and heightened neuroplasticity in the subacute phase after stroke.
We identified that LF-rTMS treatment resulted in a small to moderate significant efficacy at the 3-month follow-up, whereas follow-ups at 2 months and 15 weeks did not reach statistical significance. However, the results should be interpreted with caution because only 5 included RCTs provided detailed follow-up data. Mounting evidence demonstrated that LF-rTMS improved long-term language prognosis for nonfluent aphasia in chronic stroke survivors.36-39 Consistent with previous findings, 2 RCTs25,32 with 86 chronic stroke survivors conducting 3 months of follow-up of LF-rTMS resulted in a moderate and positive efficacy on nonfluent PSA. Therefore, these findings may provide support to the mechanism by which LF-rTMS produces a lasting efficacy pertaining to the induction of plastic neural changes when LF-rTMS is applied to nonfluent PSA. Interestingly, stroke survivors with different mother languages appear to respond differently to LF-rTMS treatment; however, several factors such as educational background in patients, quality of rTMS application, batteries for evaluation of language performances, size of lesions, and severity of aphasia might be related to this finding.
LF-rTMS targeting the triangle of the right inferior frontal gyrus (IFG) significantly improved language performance on naming, repetition, writing and comprehension, which was consistent with previous studies.14,15 There are several possible mechanisms by which LF-rTMS influences language function after stroke. Previous literature39-41 considered that rTMS-enhancing recovery of language function was ascribed to reconstruction of the language network, and the inhibitory LF-rTMS facilitated language recovery by reducing interhemispheric inhibition. Several clinical studies42-44 have demonstrated that interhemispheric compensation should be possibly involved in the process of language function recovery after a lesion. Unfortunately, this method easily leads to language dominance transferring to the right hemisphere and is less efficient for language function recovery.45,46 Thus, several functional imaging studies47,48 have reported that applying LF-rTMS on the nondominant hemisphere facilitated the reactivation of the residual functional networks in the dominant hemisphere by reducing transcallosal inhibition after damage of primary language areas of the dominant hemisphere. Furthermore, another imaging study 49 applying functional magnetic resonance found that LF-rTMS coupled with SLT may influence cerebral blood flow and contribute to the mitigation of language impairment in chronic poststroke aphasic patients. Taken together, the application of inhibitory LF-rTMS leading to a reduction of overactivation in the IFG of the unaffected hemisphere might be a satisfactory strategy to enhance the susceptibility of language areas to SLT in the affected hemisphere.
The effect size was 0.44 for repetition and 0.45 for comprehension tests, which indicated that they did not achieve minimal clinically important difference effects of active LF-rTMS compared with sham LF-rTMS. 50 The reason may be that the efficacy of LF-rTMS on language function probably depended on the stimulus site. A recent study 23 first compared the influences of 2 LF-rTMS stimulus sites on language recovery in PSA, showing that stimulation of the right superior temporal gyrus significantly improved language recovery in terms of comprehension and repetition, whereas activation of the IFG significantly enhanced spontaneous speech. These findings elucidated that the modulation of comprehension or repetition may be regulated by the temporal lobe; thus, they would be less likely to be affected by LF-rTMS treatment.
Impairments in spoken language limit stroke survivors in activities of daily living. 3 Assessing spontaneous spoken language in life situations, therefore, is an important indicator of therapeutic success. However, most assessment in our included studies either focused on spoken language performance in the clinic or laboratory, following prompting from an examiner, or evaluated general communication efficacy during everyday activities, without emphasis on spoken language characteristics themselves. 51 Therefore, further RCTs may utilize patient-centered measures and validated methods to evaluate real-world communication in PSA, such as the Verbal Activity Log.
We observed deficiency in safety assessments in the included studies. For example, only 8 trials22,25,26,30-34 reported adverse events in the article. Two studies22,25 reported dizziness and dull pain during the intervention sessions. Although LF-rTMS is likely a safe neuromodulatory technology, the safety of LF-rTMS should be fully assessed and reported, especially because stroke participants belong to a vulnerable population.
There were several limitations to this study. First, we were unable to examine the influence of several patient characteristics, such as age, aphasia severity, and comorbidity, on the effects of LF-rTMS. Notably, aphasia severity, as one of the critical predictors of response to treatment, may also affect the magnitude of LF-rTMS effect sizes; however, we were underpowered to assess this in the present study. Similarly, much of the SLT intervention information, for example, the history of, nature of, and intensity of SLT interventions that were conducted before, during, or after LF-rTMS, was only sporadically reported, and therefore precluded our ability to examine the impact of LF-rTMS across studies. Second, although no or little heterogeneity was noted, it is still important to know that the LF-rTMS protocols and the outcome measures can vary differently in different languages. Third, for studies with small sample size, or with subgroups containing only <5 participants, the results may need to be interpreted with caution. In addition, the language outcomes in included studies were psycholinguistic outcomes rather than functional language outcomes. In some degree, it fails to translate to functional language, and the dearth of functional language outcomes assessments will exaggerate the results. Finally, we also had concerns about the quality of published studies. For example, only 4 out of 14 studies met low-risk bias in each item quality assessment.
In conclusion, our updated meta-analysis with 14 included studies indicated that LF-rTMS combined with SLT had favorable immediate as well as long-term effects on language recovery (specifically on naming and writing) in patients with PSA. We also observed that intervention duration of 20 minutes per day for 10 days (compared with 15 or 20 days) or treatment in subacute (rather than chronic) stroke had the largest effect size. These findings suggest that LF-rTMS is a viable neuromodulatory intervention to improve language functions in patients with PSA. A large well-designed randomized clinical trial is required to validate and confirm these findings.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-docx-1-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-1-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-1-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-10-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-10-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-11-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-11-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-12-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-12-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-13-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-13-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-14-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-14-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-15-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-15-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-16-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-16-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-17-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-17-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-18-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-18-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-2-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-2-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-3-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-3-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-4-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-4-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-5-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-5-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-6-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-6-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-7-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-7-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-8-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-8-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Supplemental Material
sj-tif-9-nnr-10.1177_15459683211011230 – Supplemental material for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis
Supplemental material, sj-tif-9-nnr-10.1177_15459683211011230 for Effects of Low-Frequency Repetitive Transcranial Magnetic Stimulation on Language Recovery in Poststroke Survivors With Aphasia: An Updated Meta-analysis by Zhongqiu Hong, Haiqing Zheng, Jing Luo, Mingyu Yin, Yinan Ai, Baomei Deng, Wuwei Feng and Xiquan Hu in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to thank the School of Public Health, Sun Yat-Sen University, for assistance in data analysis for this article.
Authors’ Note
ZH, HZ, and XH initiated the study concepts and design. ZH, HZ, and BD reviewed references and conducted the meta-analysis. ZH, HZ, and JL drafted the manuscript. ZH, JL, MY, and YA reviewed references and interpreted data. ZH, WF, and XH carried out the statistical analysis, and WF revised the manuscript. All authors read and approved the final manuscript and contributed to the drafting and revising of the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) received grant support from the National Natural Science Foundation of China (Grant No. 81871847, 81702232, and 81672261), the Natural Science Foundation of Guangdong Province (Grant No.2017A030313493), the Basic and Applied Basic Research Foundation of Guangdong Province (Grant No.2019A1515011106), the Science and Technology Planning Key Project of Guangzhou (Grant No. 201803010119), and the Key Technologies Research and Development Program of China (Grant No. 2018YFC2001603).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
