Abstract
Background. Regular exercise reduces the risk of a first-ever stroke and is associated with smaller infarcts. Although evidence has suggested that therapeutic exercise following stroke is beneficial, we do not yet know whether exercise reduces stroke severity and improves functional recovery. The mechanisms underlying any benefit remain unclear. Objective. To conduct a systematic review and meta-analysis of studies testing exercise in animal models of ischemic stroke where outcomes were measured as infarct volume, neurobehavioral score, neurogenesis, or a combination of these. We also sought evidence of publication bias. Methods. We searched 3 online databases for publications reporting the use of exercise in focal cerebral ischemia. We used DerSimonian and Laird normalized random-effects meta-analysis and meta-regression to determine the impact of study quality and design on the efficacy of exercise. Results. Overall, exercise reduced infarct volume by 25.2% (95% confidence interval [CI] = 19.0%-31.3%; 65 experiments and 986 animals) and improved neurobehavioral score by 38.2% (95% CI = 29.1%-47.3%; 42 experiments; n = 771). For both outcomes, larger effects were seen when exercise preceded ischemia rather than came after it. For neurobehavioral scores, we found evidence of publication bias. Reported study quality was moderate (median score 5/10). Both model-specific (eg, type of ischemia) and exercise-specific characteristics influenced reported outcome. Conclusion. Exercise, either before or after ischemia, reduced infarct volume and improved neurobehavioral score. However, overall estimates of efficacy were higher in studies at risk of bias, and for neurobehavioral outcomes, there was evidence of a substantial publication bias.
Introduction
Physical activity contributes to good health throughout life. 1 Regular participation in mild or moderately intense physical activity prior to stroke has been associated with a reduction in the risk and initial severity of stroke and with improved functional recovery.2,3 However, much evidence comes from unreliable self-reported data. Early and more frequent physically based therapeutic activity during the first 6 months poststroke is associated with reduced disability, 4 better mobility, and greater independence,4-6 but the most effective timing and intensity of therapeutic activity is not known. Physical activity undertaken during the chronic phase of stroke recovery may improve cardiorespiratory fitness and aspects of mobility, 7 but its effects on death and disability and the most effective timing and intensity are not known.
Animal models of stroke might provide insights into the optimal dose and timing of physical activity or exercise, and this knowledge could be used to inform the design of definitive clinical trials in stroke.
Current evidence from animal studies about the effect of exercise in focal ischemia are contradictory, with studies reporting that exercise either improves8,9 or worsens 10 outcome. Recent systematic reviews have highlighted concerns regarding the internal and external validity of such studies,11-13 and these problems might explain the contradictions described above. The range and quality of research has not been described.
Systematic reviews and meta-analyses are well-established methods of characterizing the body of evidence describing interventions in animal models of stroke.14-16 A systematic review provides a comprehensive and unbiased overview of the literature, and a meta-analysis can quantify the impact of experimental design and study quality characteristics. This empirical evidence can be used to inform future experimental work and assist in the clinical development of therapies for stroke.12-14,17 Across a number of reviews of focal ischemia models, the failure to report measures to reduce bias (eg, randomization) is associated with overestimation of efficacy. 14 This prompted the development of guidelines for good laboratory practice 18 and informed the development of the Animals in Research: Reporting In Vivo Experiments (ARRIVE) guidelines 19 similar to the CONSORT reporting guidelines for human clinical trials. 20
Here, we use a systematic review and meta-analysis to study the impact of exercise, both before and after the induction of ischemia, in animal models of ischemic stroke. Our objectives were, first, to identify all studies describing the use of exercise in animal models of ischemic stroke and, second, to investigate the effects of exercise on infarct volume, neurobehavioral score, and (if there were sufficient data) neurogenesis. Finally, we aimed to quantify any influence of study quality and design characteristics (such as timing or intensity of treatment) on reported efficacy.
Methods
Identification of Relevant Studies
We searched MEDLINE, EMBASE, and Web of Science Proceedings in July 2011 for [‘exercise’ OR ‘fitness’ OR ‘locomotion’ OR ‘ambulation’ OR ‘walk*’ OR ‘run*’ OR ‘treadmill*’ OR ‘wheel*’ OR ‘swim*’ OR ‘physical therap*’ OR ‘physical exercise’ OR ‘physical conditioning’ OR ‘physical activit*’ OR ‘physical exertion’] AND [(‘stroke*’ OR ‘cva’ OR ‘aca’ OR ‘mca’ OR ‘pca’ OR ‘cerebrovascular’ OR ((‘cerebral’ OR ‘cerebellar’ OR ‘brain*’ OR ‘vertebrobasilar’) AND (‘infarct*’ OR ‘ischemia*’ OR ‘ischaemia*’ OR ‘thrombo*’ OR ‘emboli*’ OR ‘apoplexy’ OR ‘artery’ OR ‘vascular’)) OR ‘hemipleg*’ OR ‘hemipar*’ OR ‘poststroke’ OR ‘post-stroke’] AND [‘animal*’ OR ‘mice’ OR ‘mouse’ OR ‘rat*’ OR ‘rodent*’ OR ‘murine’ OR ‘cat*’ OR ‘feline’ OR ‘pig*’ OR ‘porcine’ OR ‘primate*’ OR ‘mammal*’] NOT [‘Coronary’ OR ‘myocardial’], limiting the results to animals.
Quality of Studies
Study quality was assessed using the CAMARADES 10-item study quality checklist, 14 which comprises (1) publication in a peer-reviewed journal, (2) allocation concealment, (3) randomization to treatment or control group, (4) control of temperature, (5) blinded assessment of outcome, (6) avoidance of anesthetics with marked intrinsic neuroprotective properties, (7) use of comorbid animals (hypertensive, aged, or diabetic), (8) sample size calculation, (9) statement of compliance with regulatory requirements, and (10) statement regarding possible conflicts of interest.
Inclusion Criteria
We included controlled animal studies of focal cerebral ischemia reporting the use of exercise as an intervention and reporting outcomes such as infarct volume, neurobehavioral score, or neurogenesis (measured with bromodeoxyuridine, Ki67, or 3H-thymidine or neuron number or density). We defined exercise as physical activity or movement of the body through the action of skeletal muscles that would be expected substantially to increase energy expenditure over resting levels. 21 Where animals received additional treatments (eg, stem cells), data were included if the cotreatment was administered to both the control and treatment groups. Environmental enrichment paradigms often include free access to a running wheel, but unless there was a control group with the same exposure without a running wheel, these data were excluded from further analysis.
The studies that met our inclusion criteria but did not provide sufficient data (sample size, mean, and a measure of variance) for meta-analysis were included in the assessment of study quality. Three independent reviewers (KJE, SS, and HJ) screened titles and abstracts and, where appropriate, full text articles; where a consensus could not be reached on inclusion of data or of an individual article, a fourth reviewer was consulted.
Data Extracted
We identified individual comparisons where the outcome in a group of animals receiving exercise was compared with that of a control group. For each comparison, we extracted data for the number of animals per group, mean outcome, and variance. Where an outcome was measured serially, only the last data were analyzed. Where numerical data were unavailable after e-mailing authors, we measured values from graphs.
We extracted details of study design, including details of animal (species, strain, and sex), surgery (anesthetic, method of ventilation, type of stroke model, and duration of ischemia, where transient), intervention (type of exercise, time of day for exercise, duration [total minutes performed over entire study], exercise details and duration [total days of study period], and the time of the start of treatment [days preischemia or postischemia]), and outcome assessment (the time period between the end of exercise and sacrifice or other assessment). Exercise was categorized as either “voluntary”—where the animal was presented with the opportunity to engage in exercise but without any “motivating” stimulus (eg, free access to a running wheel)—or forced—where the animal was subjected to a noxious stimulus (eg, electric shock) if it did not perform the exercise or was restrained such that participation was inevitable. We considered the situation where animals had unrestricted access to the exercise apparatus as “continuous exercise” and that where access was regulated as single or multiple exercise. Where there were multiple treatment combinations, we compared groups receiving the cotreatment without exercise and those receiving the cotreatment and exercise.
Data Analysis
For each comparison, we calculated a normalized effect size (the percentage improvement in outcome relative to control) and a corresponding standard error. Where multiple neurobehavioral outcomes were measured in the same cohort of animals, we used fixed-effects meta-analysis to combine data to give an overall estimate of outcome. We then aggregated effect sizes using DerSimonian and Laird random effects meta-analysis. 22 To assess the impact of study quality and the likelihood of publication bias, we analyzed the entire data sets for each outcome. For study characteristics, we considered exercise interventions before or after stroke separately. We assessed the presence and impact of publication bias using funnel plotting, 23 Egger regression, 23 and Trim and Fill. 24 When more than one outcome was measured in a single cohort of animals, the process of combining these to give an overall outcome could confound the assessment of publication bias, and so we used individual rather than pooled outcomes from these cohorts for this analysis.
Factors Modifying the Effects of Exercise
We assessed the impact of study characteristics and quality on outcomes using both stratified meta-analysis and metaregression. The significance of differences between n groups was assessed by partitioning heterogeneity and using the χ2 distribution with n − 1 degrees of freedom (df). We considered P values <.05 to be statistically significant. Correction for the number of comparisons made gave critical P values of .05 for the analysis of preischemic versus postischemic exercise, .004 for the analysis of study characteristics, and .017 for the analysis of study quality. We used metaregression to assess the impact of continuous variables, with a critical value for P of .008.
Results
A total of 40 publications met our inclusion criteria and were included in this review (supplementary appendix 1). Of these, 35 reported data suitable for inclusion in the meta-analysis (see Figure 1, supplementary appendix 2). These studies described 107 experiments. Data for infarct volume were reported for 65 experimental cohorts (n = 986 animals); for neurobehavioral score, 73 outcomes were reported for 42 experimental cohorts (n = 771 animals). There was significant heterogeneity for both infarct volume (χ2 = 557; df = 64; P = 9 × 10−80) and neurobehavioral score (χ2 = 189.3; df = 41; P = 6 × 10−21). The number of experiments reporting neurogenesis outcomes (7 experiments; n = 114 animals; χ2 = 31.1) was small, and because different outcome measures were reported, these were not analyzed further.
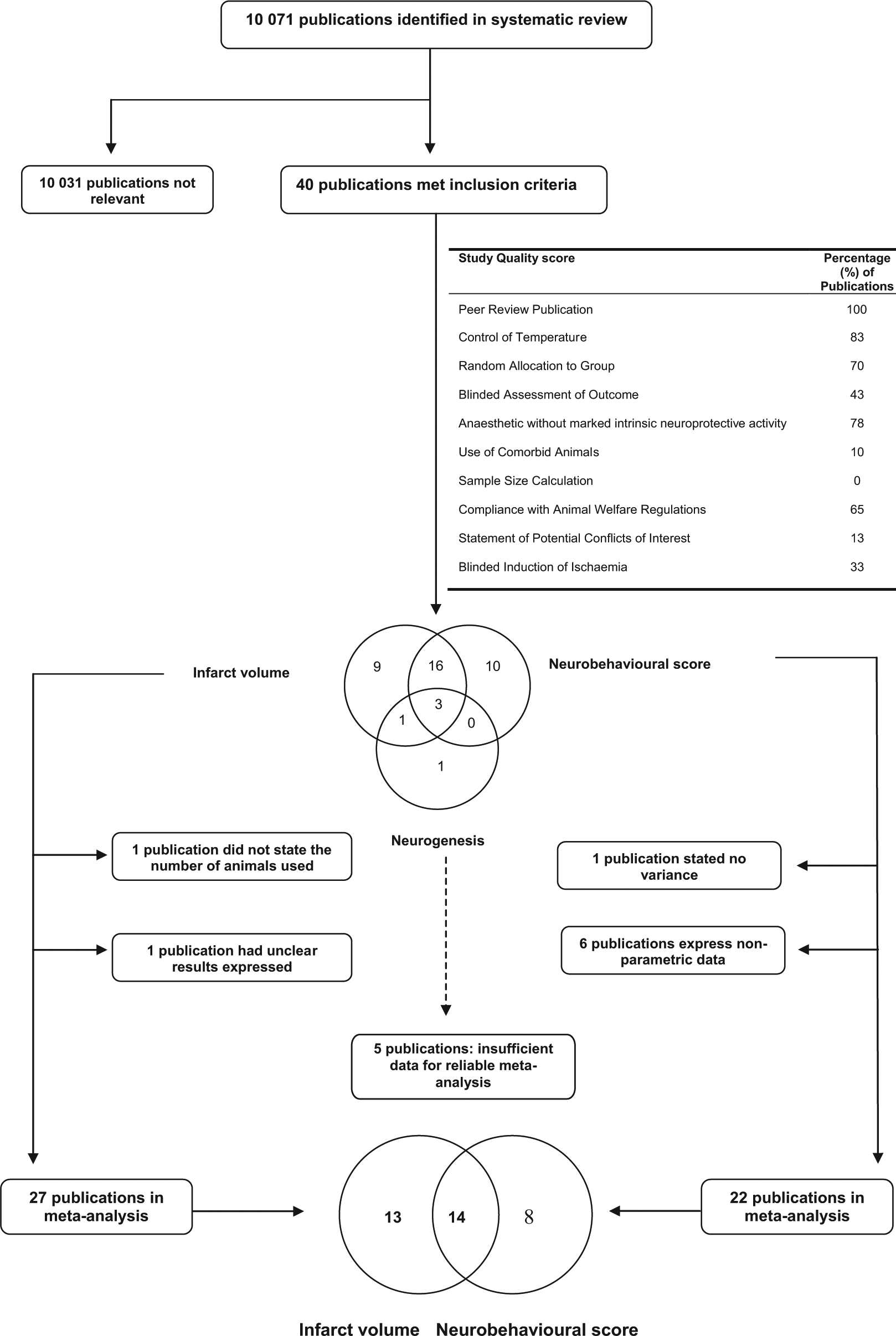
The number of publications identified from the systematic search alongside the number that met inclusion criteria. The Venn diagram at the center demonstrates the number of publications that reported infarct volume, neurobehavioral score, and neurogenesis outcomes, whereas the lower Venn diagram demonstrates the number of publications included in the meta-analysis for both infarct volume and neurobehavioral score. Within the figure, the table summarizes the percentage of studies that report study quality items as described in the Methods section.
Study Quality
Important measures to minimize potential sources of bias were frequently reported; randomization was reported in 28 of 40 publications (70%) and the blinding of the assessment of outcome by 17 of 40 (43%; Figure 1). However, the details of randomization (coin toss, random number, and computer generated random number) were not reported. The median number of study quality checklist items scored was 5 out of a possible 10 (interquartile range [IQR] = 4-6). The aggregate study quality score explained a significant proportion of the heterogeneity both for infarct volume (χ2 = 126; P < .02; Figure 2A) and for neurobehavioral score (χ2 = 74.0; P < .02; Figure 2B). For neurobehavioral score, efficacy was lower in the highest-quality studies. For infarct volume, efficacy was significantly higher in studies that reported random allocation to treatment group compared with those that did not (28.2% [21.3%-35.0%], 51 experiments, 737 animals [randomized], vs 15.8% [4.5%-27.0%], 14 experiments, 249 animals [not randomized]; χ2 = 60.9; P < .02; Figure 2C). For neurobehavioral score, there was little difference (39.6% [27.2%-51.9%], 27 experiments, 428 animals [randomized], vs 35.2% [23.2%-47.3%], 15 experiments, 343 animals [not randomized]); χ2 = 0.75; P > .02; not shown. Blinded assessment of outcome accounted for a significant proportion of heterogeneity for both infarct volume and neurobehavioral score. For infarct volume, efficacy was higher in studies that reported the blinded assessment of outcome (43.9% [20.0%-67.8%], 11 experiments, 128 animals, vs 22.8% [16.4%-29.2%], 54 experiments, 858 animals; χ2 = 12.8; P < .02; figure not shown), but for neurobehavioral score, the reverse was seen (blinded 28.4% [16.5%-40.3%], 17 experiments, 276 animals, vs unblinded 43.7% [31.9%-55.5%], 25 experiments, 496 animals); χ2 = 48.2; P < .02; Figure 2D.
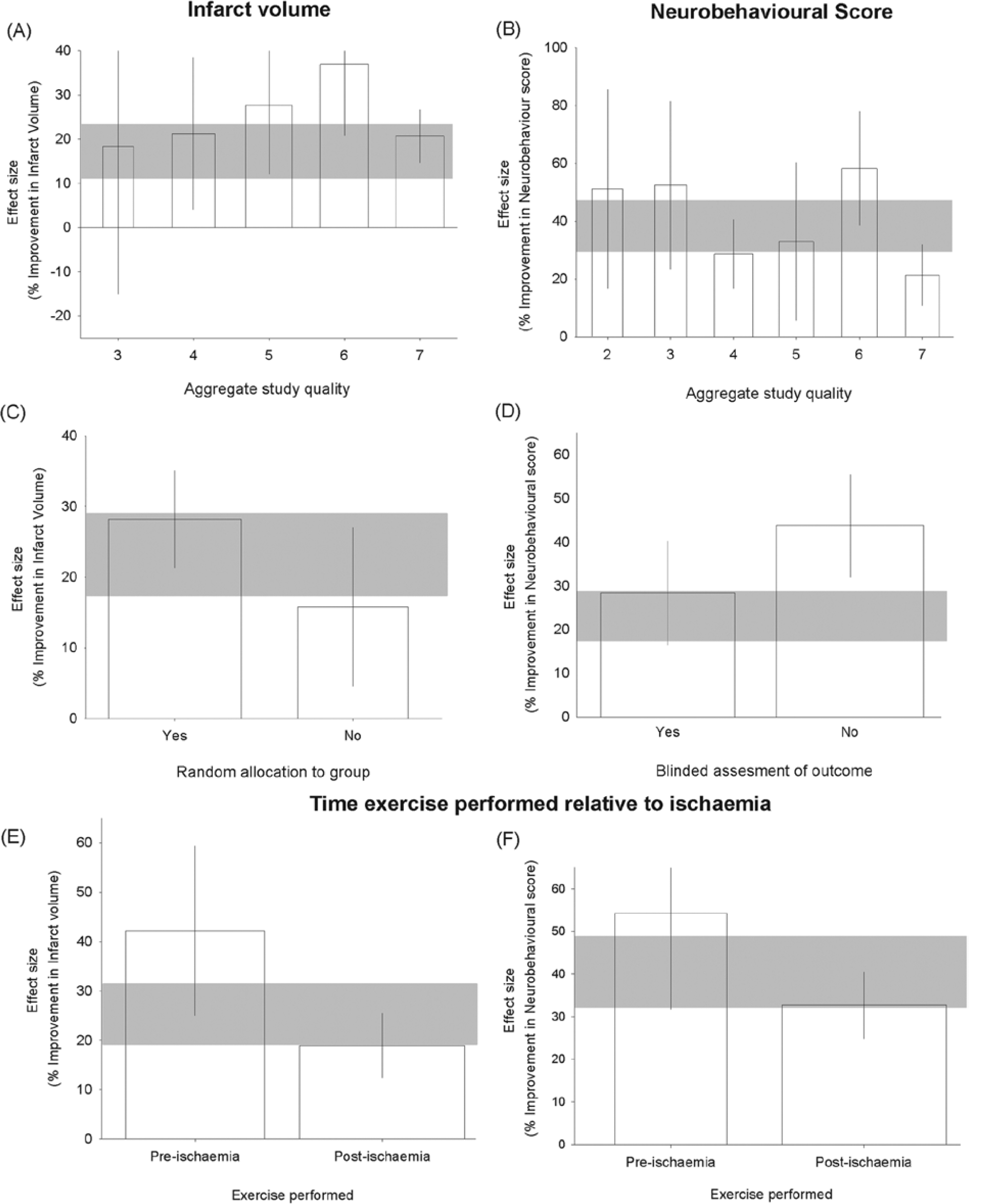
Effect size and 95% confidence intervals (CIs) for effect of exercise on infarct volume stratified by (A) aggregate study quality score and (B) presence of randomization and neurobehavioral score stratified by (C) aggregate quality score and (D) blinded assessment of outcome. E. Reported effect sizes for infarct volume according to preischemic or postischemic exercise. F. Neurobehavioral outcomes according to exercise preischemia or postischemia. Error bars represent 95% CI of individual estimates. Bar width represents the log of the number of animals, and the gray bar represents the 95% CI of the global estimate.
No study reported a sample size calculation. The studies were generally small; for infarct volume, the median numbers of animals for control and treatment groups were 7 (IQR = 5-9) and 8 (IQR = 6-10), respectively, and for neurobehavioral score, the median numbers of animals for control and treatment groups were 7 (IQR = 3.3-8) and 10 (IQR = 8-12), respectively.
Publication Bias
For infarct volume, there was evidence of publication bias from Egger regression but not from funnel plotting or trim and fill. For neurobehavioral score, both funnel plot and Egger regression analyses were consistent with publication bias, and this finding was confirmed using Trim and Fill analyses, which imputed an additional 14 unpublished individual outcomes and an adjusted efficacy of 31.0% (23.0-39.1) compared with the unadjusted summary estimate of 40.0% (32.4-47.8, 73 individual outcomes).
Effect of Exercise on Infarct Volume
Exercise reduced infarct volume by 25.2% (19.0%-31.3%; χ2 = 557; P < .001). Preischemic exercise reduced infarct volume by 42.2% (25.0%-59.3%, 18 experiments, 277 animals), and postischemic exercise reduced infarct volume by 18.9% (12.4%-25.5%, 47 experiments, 709 animals, Figure 2E). Whether exercise was initiated preischemia or postischemia accounted for a significant proportion of the observed heterogeneity (P < .05).
Effect of Exercise on Neurobehavioral Score
Exercise improved neurobehavioral outcome by 38.2% (29.1%-47.3%, 42 experiments, 771 animals; χ2 = 189; P < .001). Preischemic exercise was more effective (54.2% [31.7%-76.7%], 6 experiments, 86 animals) than postischemic exercise (32.7% (24.9%-40.5%), 36 experiments, 685 animals; P < .03; Figure 2F).
Effects of Preischemic Exercise
For these 18 experiments (277 animals), stratifying outcome by the anesthetic used (χ2 = 35.4; P < .004; Figure 3A; chloral hydrate was associated with larger effect sizes) or species (χ2 = 79.7; P < .004; Figure 3B) explained a significant proportion of the observed heterogeneity.
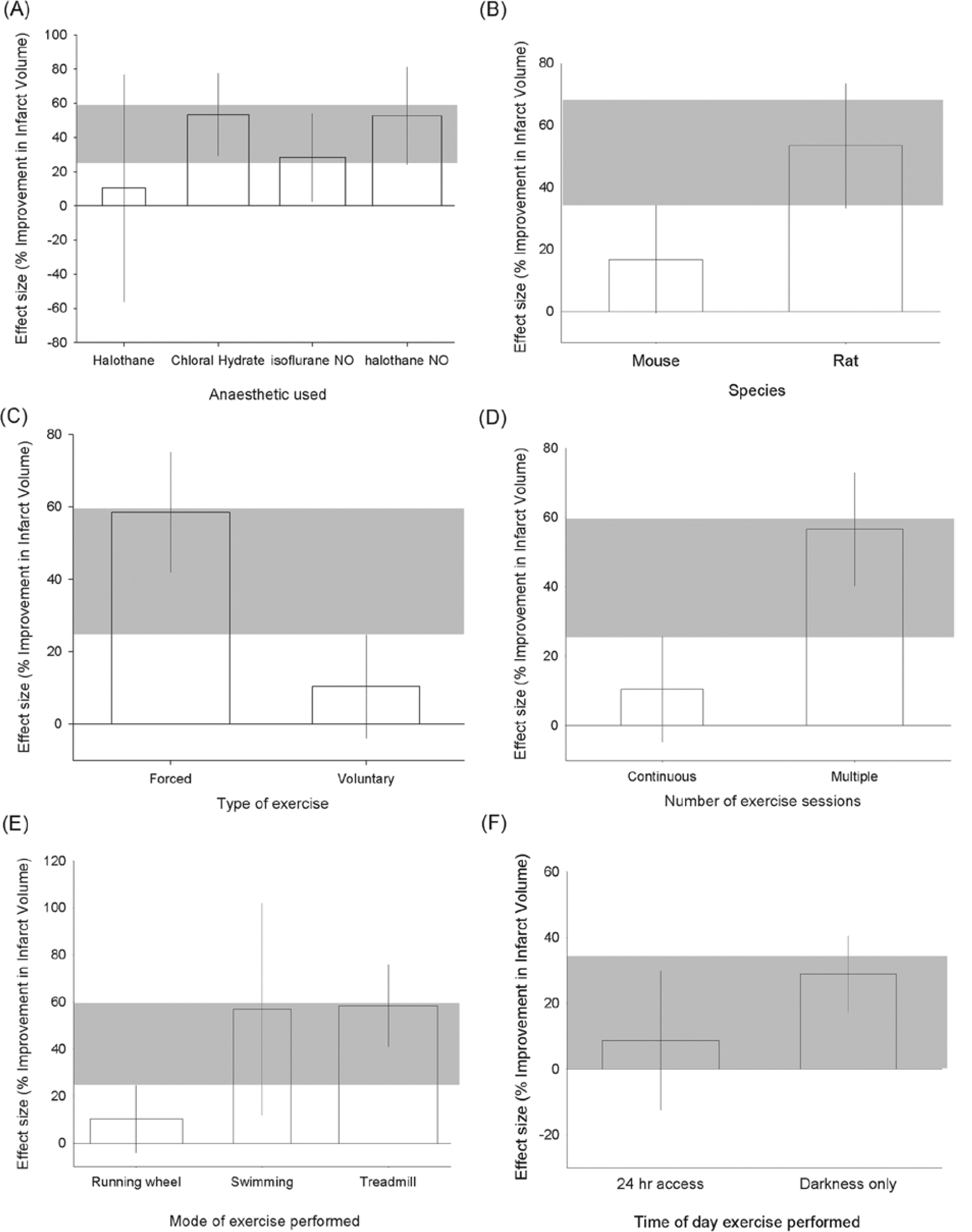
Effect size and 95% confidence intervals (CIs) for effect of prestroke exercise by (A) anesthetic used, species used, (C) type of exercise, (D) number of exercise sessions, (E) mode of exercise performed, and (F) time of day exercise is performed. Bar width represents the log of the number of animals; gray bar represents the 95% CI of the global estimate.
Characteristics of the exercise itself seemed to be important: forced exercise was associated with a substantially greater reduction in infarct volume than voluntary exercise (58.5 [41.8-75.1], 12 experiments, 170 animals, vs 10.4% [−4.0% to 24.8%], 6 experiments, 107 animals); χ2 = 119; P < .004 (Figure 3C). Continuous exercise was less effective in reducing infarct volume than multiple-session exercise (10.5% [−4.7% to 25.7%], 5 experiments, 88 animals, vs 56.6% [40.2%-72.9%], 13 experiments, 189 animals); χ2 = 117; P < .004 (Figure 3D). Swimming and treadmill exercise reduced infarct volume more than the use of a running wheel (χ2 = 119; P < .004; Figure 3E). However, the same group of experiments involved the running wheel, voluntary exercise, and (with 1 exception) continuous exercise, so which of these characteristics might be responsible for the lower observed efficacy is not clear.
Only 5 of 18 experiments reported the time of day at which exercise was performed; exercise at night (in darkness) was significantly more effective (28.9% [17.3%-40.5%], 2 experiments, 26 animals) than where animals had 24-hour access to exercise (8.7% [−12.4% to 29.8%], 3 experiments, 54 animals); χ2 = 13.8; P < .004 (Figure 3F). Interestingly, efficacy was substantially higher in those experiments where the timing of exercise was not stated (53.4% [33.3-73.6], 13 experiments, 197 animals).
Using metaregression there was no association between reported efficacy and the time of starting treatment, amount of exercise, or time of outcome assessment (see Table 1). For neurobehavioral score only 6 experiments using 87 animals reported the effects of treatment started before ischemia, and these data were not analyzed further.
Continuous data were analyzed using metaregression techniques. Both Infarct Volume and Neurobehavioral score were analyzed according to whether exercise was preischemia or postischemia.
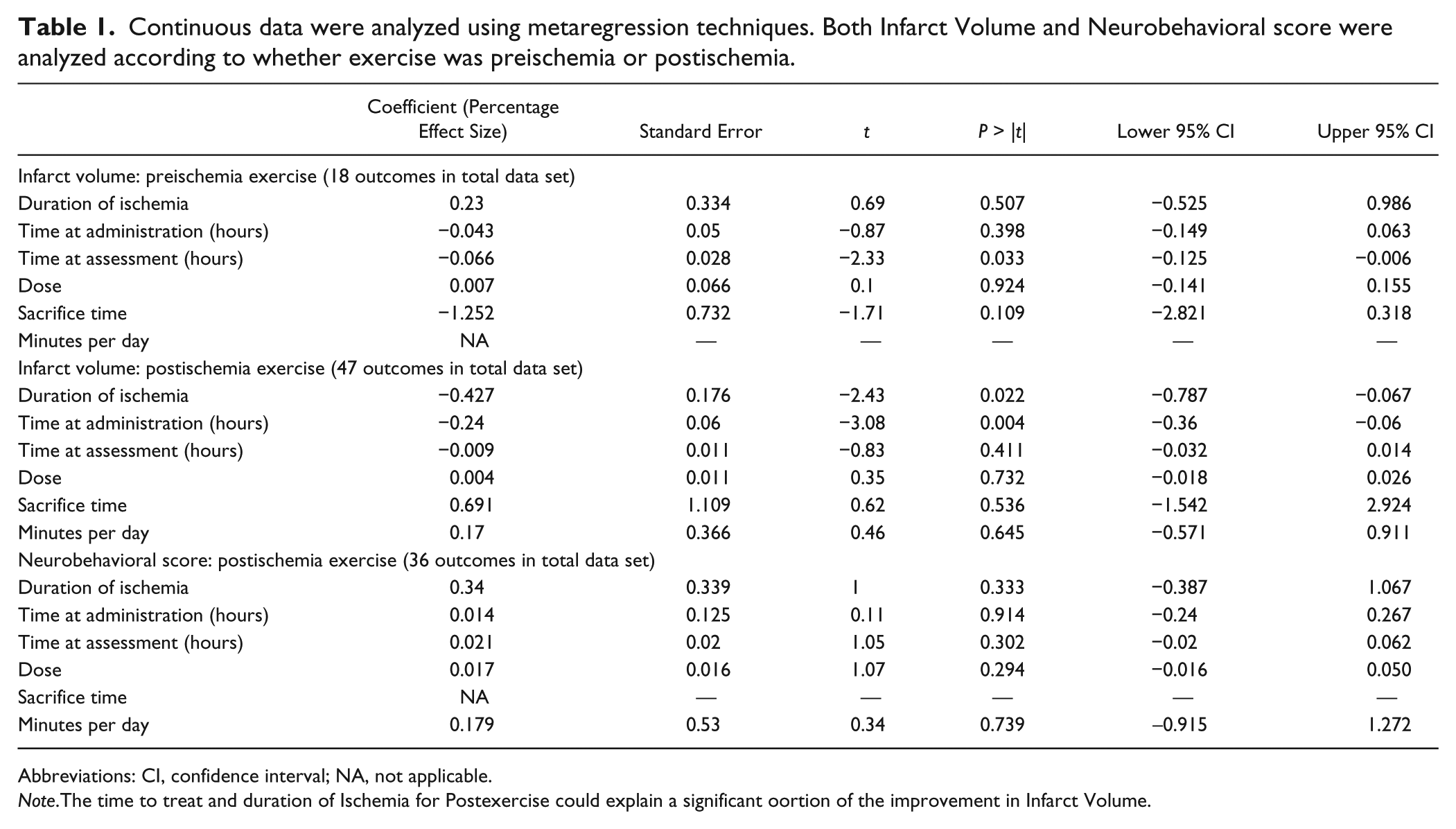
Abbreviations: CI, confidence interval; NA, not applicable.
Note.The time to treat and duration of Ischemia for Postexercise could explain a significant oortion of the improvement in Infarct Volume.
Effects of Postischemic Exercise
Infarct Volume
Where exercise began after ischemia, the type of ischemia explained a significant proportion of the observed heterogeneity, with larger effect sizes in models of temporary ischemia (χ2 = 24.9; P < .004; Figure 4A). The means of induction of ischemia (χ2 = 123, P < .004; ligation was associated with larger estimates of effect sizes; Figure 4C) and anesthetic used (χ2 = 161, P < .004; effect size was greatest with chloral hydrate; Figure 4B) both explained a significant proportion of observed heterogeneity. Animal sex was also a significant source of heterogeneity, in that the significant improvement in outcome seen in males (21.8% [15.3-28.3], 43 experiments, 655 animals) was not seen in females (1.1% [−4.4% to 6.7%], 3 experiments, 39 animals); χ2 = 85.5; P < .004 (Figure 4D). Efficacy in aged animals (20.2% [7.2%-33.3%], 3 experiments, 40 animals) was comparable to that in animals without comorbidities (20.3% [13.1%-27.6%], 42 experiments, 635 animals), but in hypertensive animals, there was no significant benefit of exercise (−4.6% [−13.3% to 4.1%], 2 experiments, 34 animals); χ2 = 47.7; P < .004 (Figure 4E).
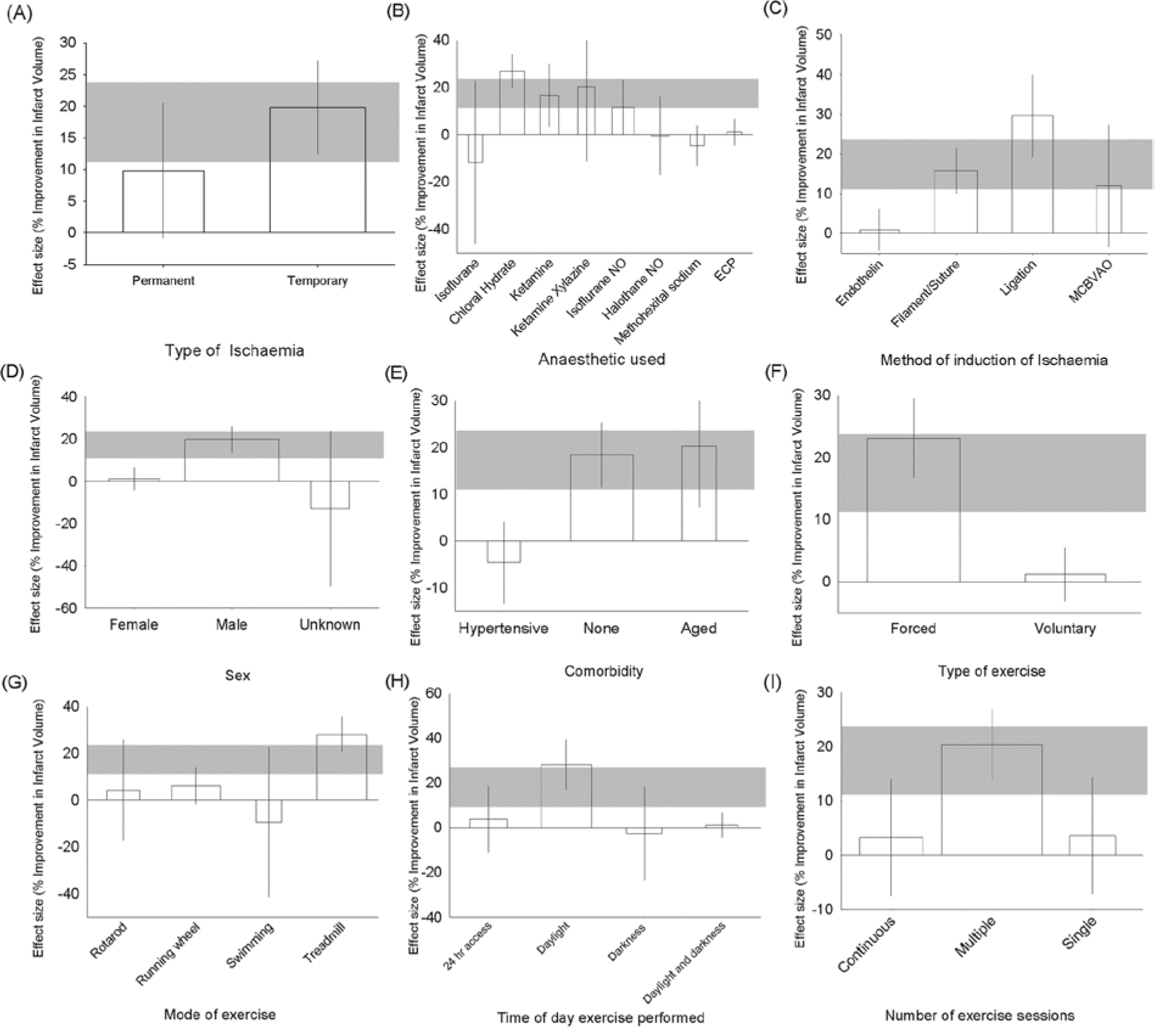
Effect size and 95% confidence intervals (CIs) for effect of poststroke exercise on (A) type of ischemia, (B) anesthetic used, (C) method of induction of ischemia, (D) sex, (E) comorbidity, (F) type of exercise, (G) mode of exercise, (H) time of day exercise was performed, and (I) number of exercise sessions. Bar width represents the log of the number of animals; gray bar represents the 95% CI of the global estimate.
Forced exercise was again more effective than voluntary exercise (23.8% [17.4%-30.2%], 39 experiments, 606 animals vs 1.0% [−3.4% to 5.4%], 8 experiments, 103 animals); χ2 = 136; P < .004 (Figure 4F). Of the different exercise types, treadmill exercise was the only one to significantly improve outcome (χ2 = 126; P < .004; Figure 4G). The time of day at which exercise was performed was described in 31 experiments, and for these, training in daylight only was associated with greater efficacy (χ2 = 104; P < .004; Figure 4H); the number of exercise sessions performed also appeared to be important, with multiple exercise sessions having greatest efficacy (χ2 = 60.6; Figure 4I). There was no impact of whether the intensity of exposure to exercise was constant or increased through the period of treatment.
Using metaregression, we found that more than a fifth of the observed variation in outcome could be explained by the interval between the start of ischemia and the start of treatment, with improvement in infarct volume being larger the earlier treatment was initiated (adjusted R2 = 21.6; df = 55; P < .008; Table 1). Individually, there was no significant impact of either the total duration of exercise or the time from ischemia onset to outcome assessment.
Neurobehavioral Score
For postischemic exercise, the effect on neurobehavioral score was significantly higher in models of transient rather than permanent ischemia (40.7% [27.3%-54.08%], 23 experiments, 361 animals vs 24.1% [15.1%-33.1%], 11 experiments, 260 animals); χ2 = 14.66; P < .004 (Figure 5A). The anesthetic agent explained a significant proportion of the observed heterogeneity (χ2 = 26.0; P < .004; Figure 5B), but there was no significant effect of species (Figure 5C). In contrast to the effects on infarct volume, efficacy was highest with voluntary (48.7% [35.5%-61.9%], 6 experiments, 82 animals) rather than forced (30.7% [22.8%-38.6%], 30 experiments, 603 animals) exercise paradigms (χ2 = 13.2; P < .004; Figure 5D). The type of exercise explained a significant proportion of the observed heterogeneity, but confidence limits around most of the point estimates were large; there was again an apparent superiority of the treadmill (38.1% [27.8%-51.4%], 16 experiments, 313 animals) over the running wheel (26.0% [13.7%-38.2%], 12 experiments, 188 animals). Continuous exercise appeared to be more effective (50.1% [36.3%-63.9%], 2 experiments, 32 animals) than multiple-session regimes (32.5% [24.7%-40.2%], 33 experiments, 632 animals), and only 1 experiment tested the efficacy of a single exercise session (5.4% [−5.5% to 16.3%], 20 animals); χ2 = 25.2; P < .004 (Figure 5F). The circadian phase during which exercise was performed was described for 14 experiments; efficacy was highest with 24-hour access (49.4% [35.3%-63.5%] 1 comparison) compared with daylight training (35.1% [19.4%-50.7%] 6 comparisons), nighttime training 17.5% (−17.3 to 52.2, 4 comparisons), and training both in the daytime and nighttime for less than 24 hours (14.8% [−87.7 to 117.2], 3 comparisons; χ2 = 13.3).
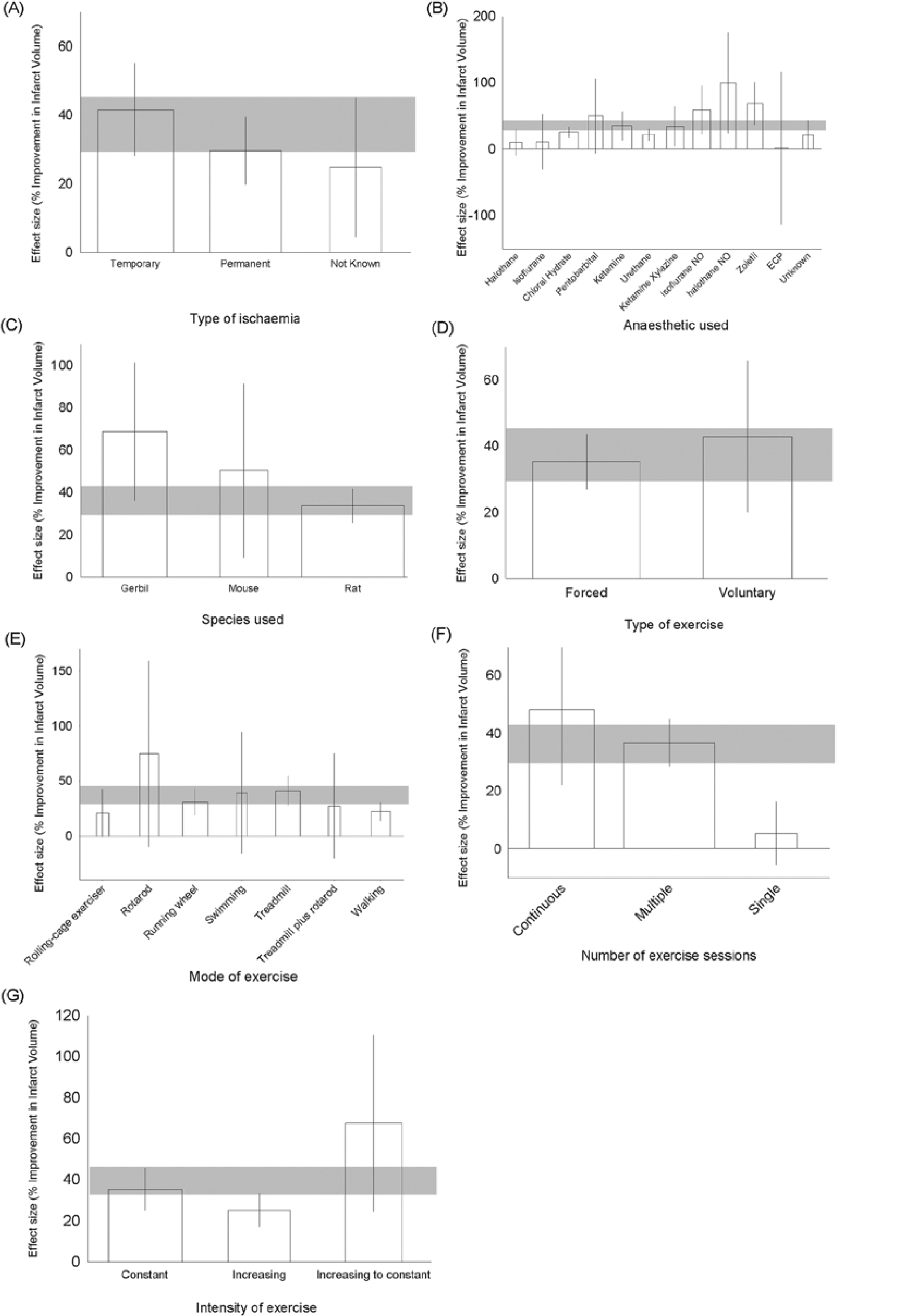
Effect size and 95% confidence intervals (CIs) for effect of poststroke exercise by (A) type of ischemia, (B) anesthetic used, (C) species used, (D) type of exercise performed, (E) mode of exercise, (F) number of exercisesessions, and (G) intensity of exercise. Bar width represents the log of the number of animals, gray bar represents the 95% CI of the global estimate.
Finally, we examined the effect of changing the intensity of exercise; most studies used constant intensity (30.2% improvement [19.5%-40.9%], 22 experiments, 388 animals). Efficacy was lower where training intensity increased throughout the experiment (24.1% [18.3%-29.8%], 8 experiments, 182 animals) and higher where training intensity increased to begin with but then plateaued (62.0% [25.9%-98.0%], 6 experiments, 115 animals); χ2 = 11.45; P < .004 (Figure 5G). Using metaregression, there was no impact of the time of start of treatment, duration, and time of outcome assessment (Table 1).
Discussion
In this systematic review and meta-analysis of exercise in animal models of focal cerebral ischemia, we showed that exercise, either before or after ischemia, is associated with significant improvements in infarct volume and neurobehavioral function.
In contrast to systematic reviews of other interventions in animal models of stroke, we found that the reporting of study quality items was relatively high,12,25 specifically for randomization (70%) and blinding (43%). However, the methods of randomization were not described, and we cannot judge the adequacy of the methods used. Although the relationship between study quality and effect size was less clear than that seen in other analyses, the smallest effect on neurobehavioral outcome was seen in the highest-quality studies. For neurobehavioral score, there was evidence of a substantial publication bias. Small study effects may be observed for reasons other than publication bias, but as we have argued before, 26 few of these are relevant in animal models of stroke.
Our stratified analyses suggest that both model- and exercise-specific methodology can influence observed outcomes. For both infarct volume and neurobehavioral function, exercise was more effective when administered before ischemia, and postischemic exercise improvements in infarct volume were greatest with shortest delays to treatment. For neurobehavioral outcomes, efficacy was independent of the delay to treatment. For both outcomes, there was no effect of the intensity of exercise.
For experiments where exercise was initiated after ischemia, treatment was more effective in transient focal ischemia models than in permanent ischemia models. This is reflected in the observation that the largest reductions in infarct volume were seen when a luminal filament was used to induce ischemia; these findings are typical of neuroprotective interventions. 27
The optimal exercise regime is not clear. For infarct volume, forced exercise regimens were more effective than voluntary exercise, whereas the reverse was true for neurobehavioral outcome. For both outcomes, treadmill exercise appeared to be the most effective. However, other exercise regimens were frequently tested in continuous rather than intermittent paradigms, and in this univariate analysis, it is not clear which of these factors is responsible for the observed differences in efficacy. There were insufficient data to analyze any effect of the amount of exercise undertaken or to allow multivariate analysis of the influence of study design factors. It is important to note that although some exercise paradigms may be ineffective, we found no evidence of harm in any of our analyses.
These findings contrast with those of a previous systematic review and meta-analysis, which found that environmental enrichment led to a small but significant increase in infarct volume. However, both meta-analyses suggest a substantial improvement in neurobehavioral outcome. It may be that the beneficial effects of exercise involve effects both on the response to ischemia and on regeneration and repair, whereas the effects of environmental enrichment may be limited to regeneration and repair.
Methodological limitations
As we identified evidence of a substantial publication bias, it could be that our conclusions regarding the influence of study design and quality may change if the unpublished data were made available for inclusion in our study. Furthermore, although our assessment of study quality was based on reported measures to reduce bias, some studies might have taken such measures but not reported them, and others that did report such measures might have used inadequate methods (eg, randomization). This highlights the importance of improved reporting of study methodology. Whereas we sought to include all relevant outcomes, the meta-analysis methodology used can only use parametric data, and for this reason, data from 2 publications were excluded. Where serial neurobehavioral data were presented, we only used last time-point data. 13 Although this approach does not capture transient improvements, we believe that long-term outcome is the most clinically relevant. For behavioral analyses, we combine data across multiple paradigms. Our rationale for this general approach is that although the experimental approach and the details of reporting may differ, all these measures represent improvement in functional recovery after an ischemic event and would, therefore, be of interest to the clinical setting. This is a common limitation in meta-analysis: whereas one gains detail in splitting, one also loses statistical power. Furthermore, using exercise duration as the “amount of exercise” only accounts for one facet of the amount of exercise; our analysis is not sensitive to other potentially important variables (eg, total distance intensity or schedule). In addition, limited data meant that we could not analyze measures of neuroplasticity—clearly an important area for stroke recovery. Finally, and most important, our work is in essence an observational study, and our findings should only be considered as hypothesis generating.
Clinical Implications
Although our results can only be considered as hypothesis generating, we believe that they may have important clinical implications. First, the effects of exercise are most pronounced when it occurs before ischemia, consistent with findings in human stroke. 28 Second, regarding the optimal timing of exercise-based therapies, exercise was more effective when given before than when given after ischemia, and when given after ischemia, exercise was more effective when given earlier rather than later. Taken together, the animal data do not support the hypothesis that early postischemia exercise is harmful. For neurobehavioral outcomes, there was no effect of delay to initiation of treatment, and for infarct volume, the benefit was greatest with shorter delays to treatment. The animal data therefore suggest that early mobilization following stroke 29 is likely to be beneficial. Our findings suggest that physical exercise has beneficial effects on unrelated neurobehavioral tasks. Further high-quality studies in animals testing the most effective type and intensity of exercise and the relative benefits of forced compared with voluntary exercise could provide important data to inform the development of human trials. For patients with stroke, it would be helpful to establish which exercise-based treatments are effective if initiated once the stroke is mature. However, there are significant challenges in defining maturation (structural component, method of induction, and species), and thus, we could not address this question within the work described.
Summary
Exercise improves outcome in animal models of stroke. Reported study quality was good, but there was evidence of publication bias. The most effective intensity and timing of and motivational drive for exercise might usefully be addressed in future well-conducted animal studies.
Footnotes
Authors’ Note
KJE and HJ contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: KE and MM are supported by the MRC Trials Methodology Hub. NS was supported by an Australian NHMRC career development fellowship (APP1035465). HJ was supported by a Postgraduate Scholarship from the National Heart Foundation, Australia and the Emlyn and Jennie Thomas Post Graduate Medical Research Scholarship. LL was supported by a PhD studentship from The University of Edinburgh Centre for Cognitive Ageing and Cognitive Epidemiology, part of the cross council Lifelong Health and Wellbeing Initiative (G0700704). Funding from the Biotechnology and Biological Sciences Research Council (BBSRC), Engineering and Physical Sciences Research Council (EPSRC), Economic and Social Research Council (ESRC), and Medical Research Council (MRC) is gratefully acknowledged.
