Abstract
Background
There is a crucial need to devise optimum rehabilitation programs for children with cerebral palsy (CP).
Objective
This study aimed to assess the feasibility, safety, and efficacy of combining 6-Hz primed, low-frequency, repetitive transcranial magnetic stimulation (rTMS) with modified constraint-induced movement therapy (mCIMT) in improving upper limb function in children with unilateral CP.
Methods
Children aged 5 to 18 years with unilateral CP were randomized (23 in each arm) to receive 10 sessions of mCIMT with real rTMS (intervention arm) or mCIMT with sham rTMS (control arm), on alternate weekdays over 4 weeks. The primary outcome was the difference in mean change in Quality of Upper Extremity Skills Test (QUEST) scores. Secondary outcomes were changes in QUEST domain scores, speed and strength measures, CP quality of life (CP-QOL) scale scores, and safety of rTMS.
Results
All 46 children completed the trial except one. At 4 weeks, the mean change in total QUEST scores was significantly higher in the intervention arm as compared to the control arm (11.66 ± 6.97 vs 6.56 ± 4.3, d = 5.1, 95% CI 1.7-8.5, P = .004). Change in “weight bearing” and “protective extension” domain score was significantly higher for children in the intervention arm. These improvements were sustained at 12 weeks (P = .028). CP-QOL scores improved at 12 weeks. No serious adverse events were seen.
Conclusion
A 6-Hz primed rTMS combined with mCIMT is safe, feasible, and superior to mCIMT alone in improving the upper limb function of children with unilateral CP.
Trial Registration:
ClinicalTrials.gov Identifier: NCT03792789.
Abbreviations
CP, cerebral palsy; rTMS, repetitive transcranial magnetic stimulation; mCIMT, modified constraint-induced movement therapy; QUEST, Quality of Upper Extremity Skills Test; CP-QOL scale, cerebral palsy quality of life scale.
Introduction
Cerebral palsy (CP) is the most common motor disability in childhood and unilateral CP accounts for about one-third of the cases. 1 Constraint-induced movement therapy (CIMT) has been shown to result in an improvement in bimanual performance and unimanual capacity in children with unilateral CP, however, the quality of evidence is low.2,3 Moreover, the effect sizes are modest, with a mean difference (MD) of 5.44 Assisting Hand Assessment (AHA) units and MD ranging from 5.95 to 12.54 across various domains of the Quality of Upper Extremity Skills Test (QUEST). 2 Given these modest effect sizes, there is a need to explore additional neurorehabilitation measures like neuromodulation, in this population. Various modifications of CIMT have been used, with a lesser duration of daily training, lasting for 30 minutes to 3 hours, and using a sling/glove for restraining.4 -7 Choudhary et al 4 demonstrated a significant improvement in upper limb function, with modified CIMT (mCIMT; 2 hours/day for 10 days, over 4 weeks), which was sustained at 12 weeks follow-up. In addition, an increase in blood oxygen level-dependent cluster activation was recorded on functional magnetic resonance imaging (fMRI) post mCIMT. 8 A subsequent clinical trial at our center demonstrated an additive effect of virtual reality therapy (VRT) when given along with mCIMT.
Transcranial magnetic stimulation (TMS) which is based on the principle of electromagnetic induction is an upcoming non-invasive modality with a variety of diagnostic and therapeutic applications. 9 For intervention, TMS is used in a repetitive manner, which can modulate the excitability of the brain, thereby influencing motor function. Repetitive TMS (rTMS) has been shown to be beneficial in adults with hemiparesis resulting from stroke, however, the corresponding data in the pediatric population is limited.
In adults with chronic stroke, the non-lesioned primary motor cortex has been demonstrated to exert an inhibitory influence, over the lesioned hemisphere. 10 This inter-hemispheric inhibition (IHI) is exerted possibly through transcallosal pathways and could adversely influence motor recovery. Broadly, 2 types of neuro-modulation strategies could result in a better motor function in such cases: (1) excitatory rTMS to promote the success of ipsilesional or (2) inhibitory rTMS to the contra-lesional upper motor neuron systems. 11 Hence, a pathologically overactive contra-lesional hemisphere is a potential target for therapeutic neuro-modulation.12,13 However, it is prudent to note that these interhemispheric motor interactions are altered in children with perinatal stroke. Effects can be seen in both directions and may include interhemispheric facilitation rather than the normal inhibitory relationship. 14 Therefore, further work is required to develop IHI models specific to perinatal stroke, wherein adult stroke models may not be applicable.
The depression of the motor cortex by 1 Hz rTMS can be enhanced by priming with 6 Hz rTMS, as demonstrated by Iyer et al. 15 The rationale for priming is based on “metaplasticity,” which refers to the phenomenon, wherein previous cellular activity leads to a change in the ability to induce subsequent synaptic plasticity. 16 Although the safety of 6-Hz primed low frequency rTMS has been demonstrated in adults previously, 17 it was first explored in children by Gillick et al. 18 It was a phase 1 clinical trial exploring the safety and feasibility of combining 6-Hz primed 1-Hz rTMS to the contra-lesional hemisphere with CIMT, in 19 children. A significant improvement in AHA scores was noted in the real rTMS + CIMT group. Although, TMS has been shown to have additive effects when combined with CIMT, there is a paucity of data from clinical trials in children, none from a resource-limited setting so far. The only other trial (PLASTIC CHAMPS) in children, employed a factorial design, using inhibitory (1 Hz) rTMS to the contra-lateral primary motor cortex. 19 AHA gains at 6 months were additive and largest with the rTMS + CIMT group. The present trial was planned to explore the safety, and efficacy 6-Hz primed rTMS with mCIMT in improving the upper limb function of children with unilateral CP in the setting of a motor training program.
Methods
Study Design
This randomized, double-blind, placebo-controlled clinical trial was conducted at the Department of Pediatrics, All India Institute of Medical Sciences (AIIMS), New Delhi, which is a tertiary care referral center in India, from February 2019 to March 2020. A data safety monitoring board was constituted for adverse event monitoring. The institute resources were utilized and no external funding was obtained.
Within 1 week of baseline functional and neuro-physiological assessment, children with unilateral CP were enrolled in a motor training program for 4 weeks. They were randomized (in a 1:1 ratio) to mCIMT + real rTMS (intervention arm) or mCIMT + sham rTMS (control arm). It comprised 10 sessions of 20 minutes of rTMS (sham or real) followed by 2 hours of mCIMT on alternate weekdays.
Standard Protocol Approvals, Registrations, and Patient Consents
The trial was prospectively registered in the database of the US National Library of Medicine (clinicaltrials.gov; NCT03792789) and conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. The protocol, informed consent (IC) forms and other documents of the study were reviewed and approved by an independent institutional ethics Committee (IECPG-574/14.11.2018.RT-32/19.12.2018). Written informed consent was taken from the parents or legally authorized representatives of the children, following regional laws/regulations. A written assent was taken by children more than 7 years of age. All participants were informed thoroughly about the study, in language and terms they were able to comprehend.
Participants
Children with unilateral CP (secondary to asymmetrical periventricular leukomalacia or stroke) were eligible for participation if they were in the age range of 5 to 18 years, had an intelligence quotient (IQ) >70 with preserved vision (able to at least see 1 in. 2 object from 1 m distance) and hearing and had a functional status as follows: gross motor function classification system (GMFCS) stage 1 to 4 and manual ability classification stage (MACS) stage 1 to 3. Unilateral CP was defined as those children with motor impairment involving 1 half of the body, with a MAS score of ≥1, with an opposite limb MAS score of 0. Children were excluded from the trial for the following reasons: a MAS score of more than 3 at shoulder/elbow/wrist or presence of contractures in the affected limb, uncontrolled epilepsy (defined by seizure frequency >1/month for preceding 3 months); severe concurrent illness or disease not associated with CP or unstable medical conditions like pneumonia; genetic or syndromic associations and diagnosis of autism spectrum disorder and any contraindications for TMS like implanted electronic device and non-removable metallic objects near the coil, for example, Pacemaker, cochlear implant. They were also excluded if they underwent a recent surgery/cast/splint or botulinum toxin/phenol block in the past 6 months or were planned to receive in the study period. Children with a severe movement disorder like dystonia, choreo-athetosis, or ballismus interfering with purposeful limb movement, those with any congenital brain malformation detected on conventional MRI brain, those receiving tone modifying agents within 2 weeks before enrolment (tizanidine, baclofen, benzodiazepines, and dantrolene), or the ones who had received mCIMT in last 6 months, were also excluded.
Study Interventions
CIMT Sessions (All Participants)
Structured mCIMT sessions were provided in groups of 3 or 4 by the principal investigator and a trained occupational therapist along with the participation of the primary caregiver at the department of physical medicine and rehabilitation (PMR). An arm sling was used for the restraint of the non-affected limb and was worn only during the intervention (an arm sling was provided free of cost to the children in both groups). The mCIMT sessions were structured and individualized for each child and conducted in a child-friendly atmosphere. During the 2-hour intervention period, they were constantly given instructions involving the specific practice of the designated task movement by the affected, unrestrained extremity. They were made to perform various repetitive and shaping activities of practicing a target movement in isolation from other movements in the given time frame. The activities were graded and selected according to the relative function. Successively the difficulty of the tasks was increased on completion of a particular task and children were given constant reinforcement throughout the session. At the end of each visit, a specific and individualized exercise plan was provided to be practiced at home for 2 hours a day on non-supervised days. The total duration of CIMT was 56 hours. Primary caregivers were asked to maintain an activity log (preferably with video records whenever feasible) to ensure compliance with the therapy on non-supervised days, which was checked by the principal investigator on each visit.
Real rTMS or Sham rTMS (1:1 Randomization)
For rTMS Sessions, the Child was seated comfortably in a reclining chair with their unaffected forearm supinated and hand supported. The elbow was positioned in 90° flexion. EMG electrodes (Ag-AgCl electrodes, solid gel) were secured on the tendon and belly of abductor pollicis brevis. EMG signals were recorded using MATRIX LIGHT amplifier (Micromed) data acquisition and amplifier system with a bandpass filter of 10 to 5000 Hz at a display sensitivity of 1 mV/division (amplifier measuring range 51.2 mV), using a recording time from stimulus onset to 500 ms after stimulus onset. The EMG data were collected using System Plus Evolution software. For hotspot identification (location of the hand area of motor cortex), a figure of 8 TMS coils (MCF B65) connected to the stimulator (Magventure Denmark, X100 with magoption) was held by hand over the approximate M1 hotspot area for the un-affected hand, tangential to the scalp with the handle pointing posterolaterally at a 450 angle to the sagittal line. It was moved systematically to find the hotspot. Single-pulse magnetic stimuli were delivered at approximately 0.1 Hz, starting at an intensity of 50% of the stimulator maximum. For the determination of resting motor threshold (RMT), stimulation intensity was adjusted systematically until a minimum intensity required to elicit Motor evoked potential (MEPs) greater than or equal to 50 µv peak-to-peak in at least 5 out of 10 trials was reached (defined as RMT).
After RMT determination, children received priming rTMS followed by 1 Hz rTMS to the contra-lesional primary motor cortex with the TMS stimulator. Evidence for inhibitory rTMS is based on studies in adults 20 as well as children.18,19,21 Priming consisted of 10 minutes of 6 Hz rTMS at 90% of RMT, delivered in 2 trains per minute with 5 seconds per train and 25-seconds intervals between trains (a total of 600 priming pulses). Priming was followed immediately by additional 10 minutes of 1 Hz rTMS at 90% of RMT without interruption (a total of 600 low-frequency pulses). At the end of each rTMS session, children were screened for any adverse event, by the rTMS administration adverse event monitoring log.
Outcomes
The primary outcome was the difference in mean change in the total QUEST score after completing 4 weeks of intervention, between the intervention and control group. There are only a few evaluation tools for assessment of upper limb function in children, to date. QUEST and AHA are the 2 most commonly used scales.2,5 AHA assesses bimanual function, while QUEST scale examines each upper limb separately, across 4 domains. QUEST has been used in multiple studies involving children with unilateral CP, involving CIMT.4,5,7,22 Additionally, the proportion of children with improvement ≥5 points in total QUEST score was compared between the 2 groups.
Secondary outcomes included change in domain-wise scores of QUEST score (dissociated movements, grasp, weight-bearing, and protective extension), speed of upper limb movements (measured by the time taken to complete 9-hole peg board), muscle strength (measured by hand-held dynamometers), improvement in quality of life scores (measured by CP-QOL child and CP-QOL teen as per the age of the child), and compliance to therapy. The subjects were assessed at 4 weeks and 12 weeks from baseline (Figure 1B). Neurophysiology parameters were assessed at baseline and 4 weeks. Two types of dynamometers were used—one for measuring hand-grip strength and the other one for push-pull strength. The un-affected upper limb function was assessed and compared, pre- and post-intervention by using 9-hole peg board and dynamometers. Adverse events were recorded using rTMS administration adverse event monitoring log maintained from enrolment till the end of the follow-up period.
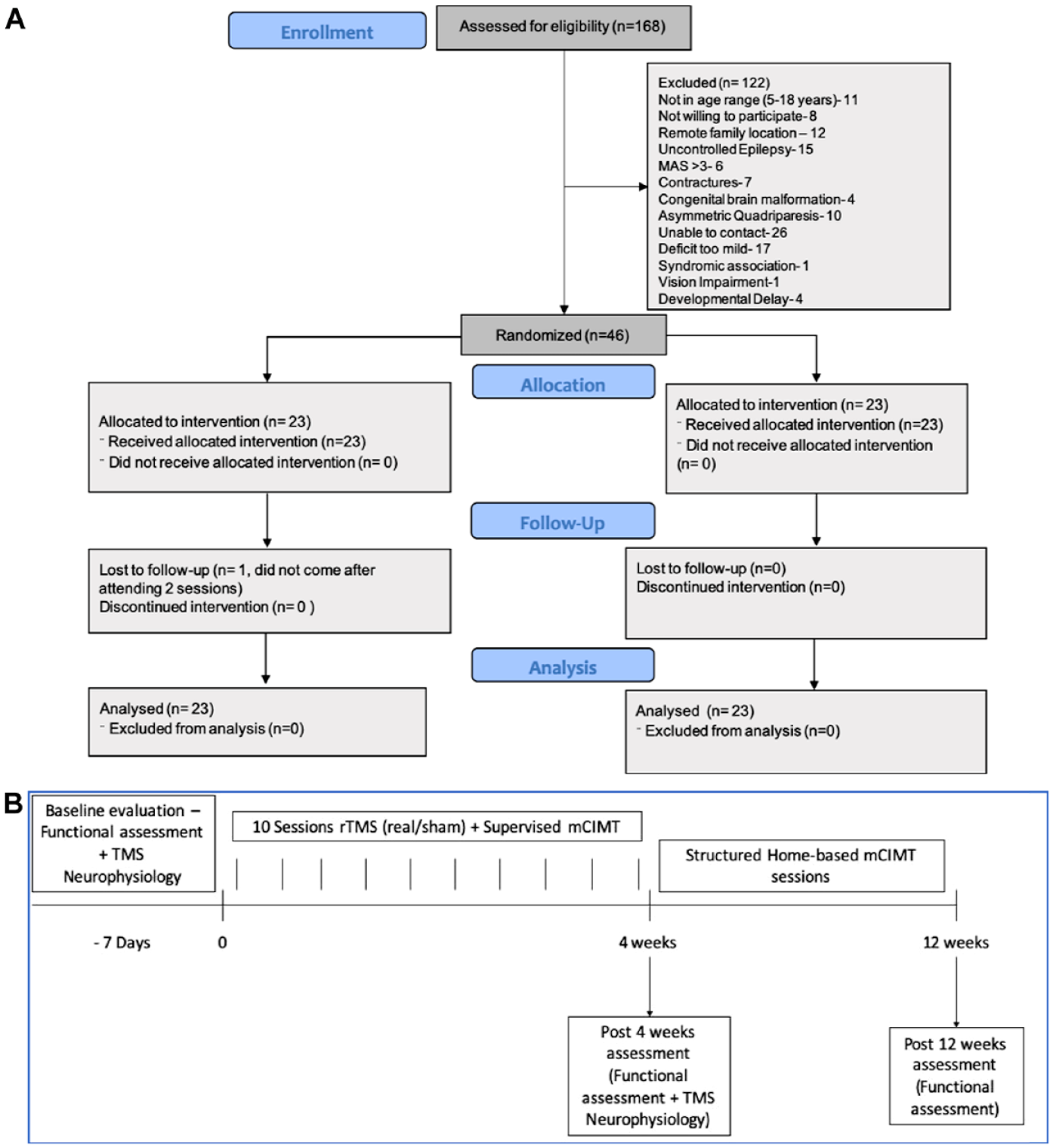
(A) CONSORT diagram depicting flow of the study and (B) flow of the trial.
Evaluation Tools
Quality of Upper Extremity Skills Test
This is a validated and reliable tool devised for measuring the upper limb functions and the quality of movements in children with spastic CP in response to therapy and is valid for children from 18 months to 8 years.23,24 Further studies have also validated the score in children aged 2 to 12 years. 25 It mainly examines each limb separately, in the 4 domains of function, namely, dissociated movements, grasp, protective extension, and weight bearing, and has 36 test items. Combined scores for 2 limbs are calculated and the maximum score in each domain is 100. A child with 1 fully functioning arm and another completely hemiplegic arm would be scored 50. All the scores from the individual domains are then averaged to obtain the final percentage score. This scoring system has been validated against the Peabody development scale and has high inter-observer reliability ranging from 0.51 to 0.96. The test-retest reliability of QUEST and its domains ranged from 0.75 to 0.95. 23 The QUEST has been used in several previous trials involving CIMT.4,5,23,25
Nine-Hole Peg Board Test
It is a tool for assessing the fine motor function and dexterity of the hand, and the ability to manipulate small objects (grasp and release). It has been validated against the Purdue pegboard test and normative data for children aged 4 to 19 years are available.26,27 In this test, both hands are tested separately. The time taken to insert and remove the nine pegs into the holes in the peg board with first the dominant hand and then the non-dominant hand is calculated. The best of 2 trials for the time taken to insert all the pegs and then remove them from the board were taken. This test has a high inter-rater agreement of >0.99 and moderately high test-retest reliability (rs = .81 and .79). 26
Hand Held Dynamometer
Hand-held dynamometers are used for the assessment of muscle strength in children as well as adults. Pull strength was assessed using Baseline push-pull dynamometer and pinch strength was assessed by a Baseline hydraulic pinch gauge in this study. Validity and reproducibility of hand-held dynamometry have high reproducibility and discriminative power in children which has been illustrated using the Jamar dynamometer in the past. 28 Reference values of isometric muscle force by hand-held dynamometry have also been established for children aged 4 to 16 years. 29 The test-retest reliability of the electronic push-pull dynamometer for measurement of extensor and flexor muscle strength has been found to be 0.85 to 0.99. 30
CP Quality of Life Questionnaire for Children (CPQOL-Child) 31
This is a questionnaire devised for the assessment of the quality of life for children with CP. It assesses the quality of life on the basis of 7 main domains—social well-being and acceptance, functioning, participation and physical health, emotional well-being, access to services, pain and impact of disability, and family health. It can be used for 4 to 12 years old children. There are 2 versions of CP-QOL child: the parent-proxy version (for parents of children aged 4-12 years) which comprises 65 items and the child self-report version (for children aged 9-12 years) which comprises 53 items. It has a high internal consistency (.74-.92) and 2-week test-retest reliability (.76-.89). 32
Further details regarding assessment tools and evaluation scales are provided in Supplemental Appendix A.
Statistical Analysis
For sample size calculation, the anticipated increase in total QUEST scores at 4 weeks in the mCIMT group was taken as 10.7 ± 5.2 (mean ± SD) based on a previous trial by Choudhary et al. 4 The anticipated increase in total QUEST scores at 4 weeks in the mCIMT + rTMS group was taken as 15.0 ± 5.0 (mean ± SD) based on the gain in hand function reported in the PLASTIC CHAMPS trial. 19 Considering alpha error as 0.5 and power as 90% the calculated sample size was 30 in each group.
Participants were randomized in a 1:1 ratio to mCIMT + real rTMS (intervention arm) and mCIMT + sham rTMS (control arm), using the block randomization method. Randomization was performed with the use of computer-generated, non-stratified sequences, and assignments were prepared in sequentially numbered, sealed, opaque envelopes. Randomization was performed by persons not involved in the trial. For sham rTMS, a sham coil was used which simulated the tactile and auditory effect of the real coil but did not generate magnetic pulses. The subjects, caregivers, and outcome assessor were blinded to the subject’s group allocation. Structured application of outcome parameters was performed by a separate physical therapist, blinded to subject characteristics and treatment allocation.
Statistical analysis was done using Stata 14.0 statistical software. For comparing baseline characteristics, chi-square and Fisher’s exact tests were used for categorical variables and the Student t-test/Wilcoxon rank-sum test was used for continuous variables. A P value of less than .05 was considered to indicate statistical significance. Intention to treat analysis and a secondary per-protocol analysis was done for the primary and all the secondary outcomes. For patients who were lost to follow-up, the last observation was carried forward. None of the following intention-to-treat analysis conclusions were altered by the secondary per-protocol analysis.
The primary outcome of the difference in mean change in QUEST score from baseline at 4 weeks was analyzed using a student t-test. For comparing the proportion of subjects with change ≥5 point change in QUEST score amongst the 2 study arms, the test of proportions was used. For analysis of secondary outcomes, the student t-test/Wilcoxon rank sum test was used. We used repeated measures ANOVA along with post hoc comparison with the Bonferroni test for within-the-group analysis of various parameters across three-time points (baseline, 4, and 12 weeks).
The study protocol and statistical analysis plan is available for further reference in Supplemental Appendix A.
Results
Characteristics of the Study Population
During the enrolment period (February 2019-March 2020) 168 children were screened for eligibility; of these 122 children were excluded (reasons in Figure 1A) and 46 met the inclusion criteria and were randomized into the 2 study groups (23 in each arm). All children completed the study except 1 child in the intervention arm who did not return for further visits after 2 sessions. The CONSORT flow diagram for the study is illustrated in Figure 1A. Trial enrolment had to be discontinued due to the national lockdown enforced during the beginning of the COVID-19 pandemic. There were no significant differences in the baseline characteristics of the 2 groups (Table 1). All the participants either had GMFCS level I or II, with no difference in distribution amongst the study arms. The distribution for co-morbidities of movement disorder, behavioral problems, ophthalmological co-morbidities, hearing impairment, and epilepsy was also similar.
Baseline Characteristics of the Study Population. a
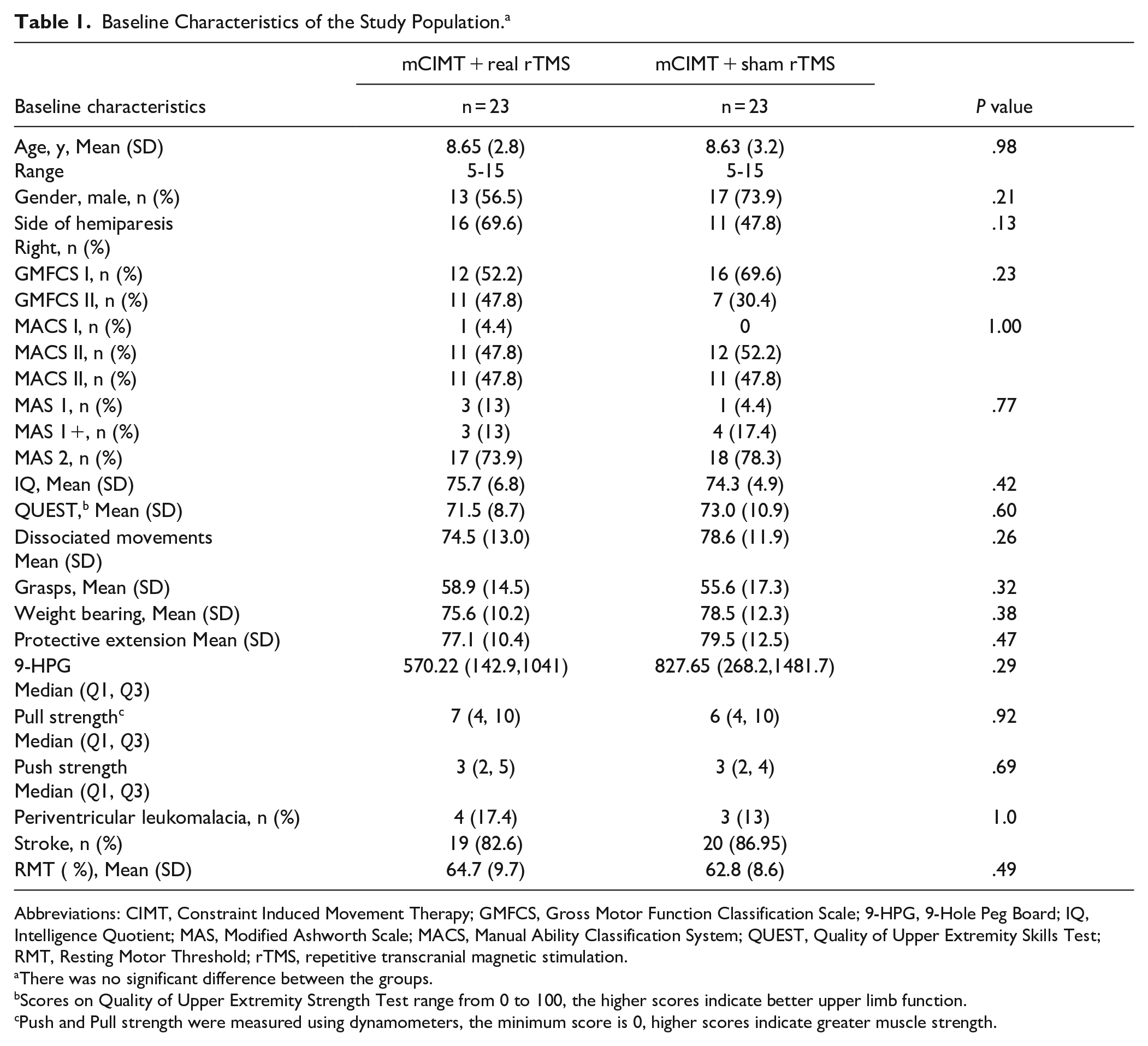
Abbreviations: CIMT, Constraint Induced Movement Therapy; GMFCS, Gross Motor Function Classification Scale; 9-HPG, 9-Hole Peg Board; IQ, Intelligence Quotient; MAS, Modified Ashworth Scale; MACS, Manual Ability Classification System; QUEST, Quality of Upper Extremity Skills Test; RMT, Resting Motor Threshold; rTMS, repetitive transcranial magnetic stimulation.
There was no significant difference between the groups.
Scores on Quality of Upper Extremity Strength Test range from 0 to 100, the higher scores indicate better upper limb function.
Push and Pull strength were measured using dynamometers, the minimum score is 0, higher scores indicate greater muscle strength.
Primary outcome: Change in Upper Limb Function at 4 weeks
The mean change in total QUEST score after completing 4 weeks of therapy, was significantly higher in the intervention arm as compared to the control arm [absolute difference (d), 5.1; 95% Confidence Interval (CI), 1.7-8.5; P = .004; Table 2, Figure 2]. The proportion of subjects with an increase in QUEST total score ≥5 was significantly higher in the intervention arm [absolute difference (d), 30.4%; 95% CI, 4.8-56.0; P = .027].
Change in Functional Scales at 4 Weeks From Baseline.
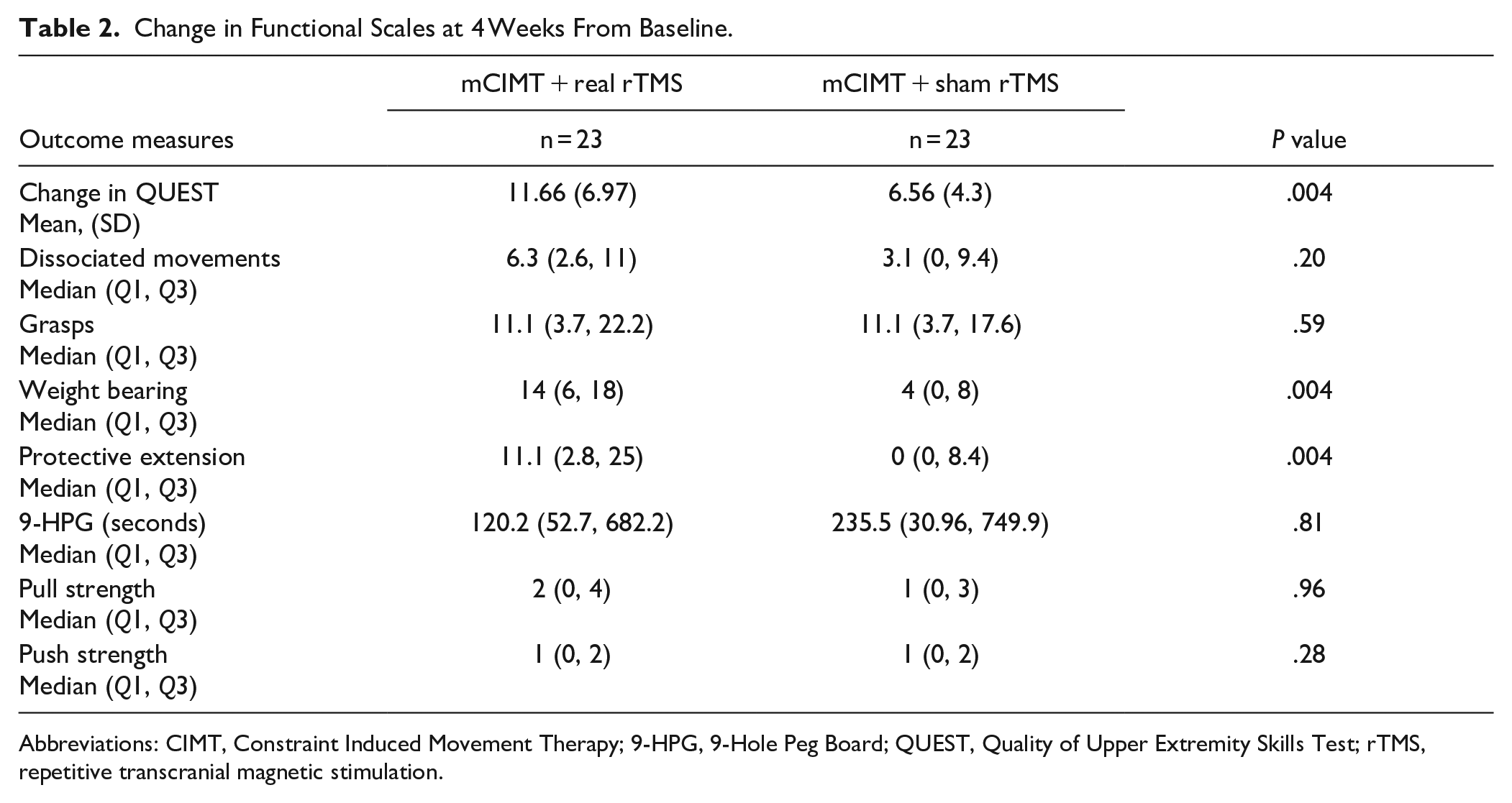
Abbreviations: CIMT, Constraint Induced Movement Therapy; 9-HPG, 9-Hole Peg Board; QUEST, Quality of Upper Extremity Skills Test; rTMS, repetitive transcranial magnetic stimulation.
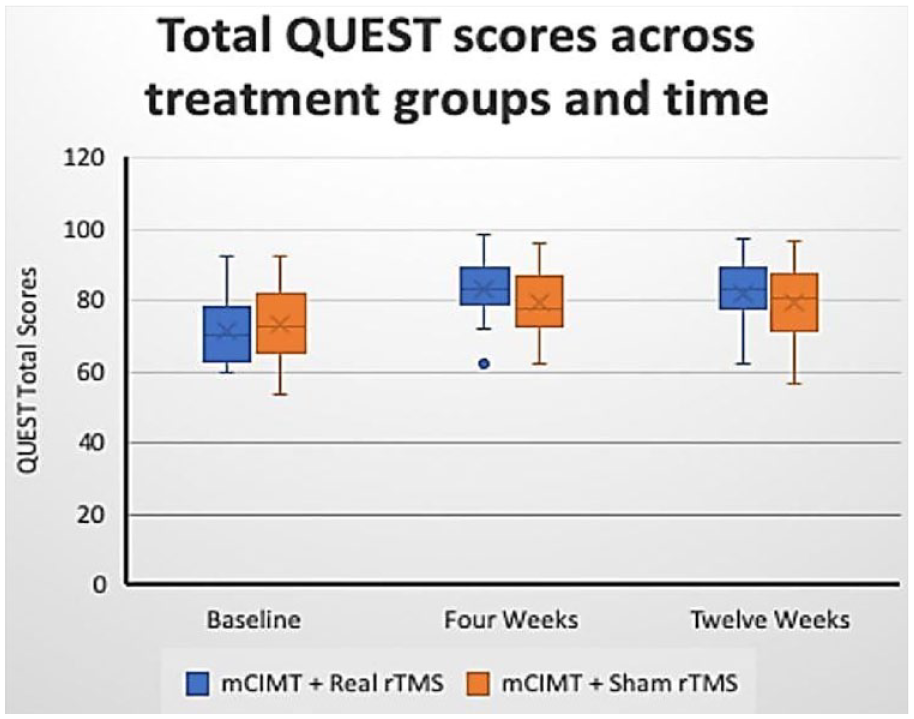
Primary outcome. Change in QUEST total score across time and treatment groups.
Change in “weight bearing” and “protective extension” domain score was significantly higher for children in the real rTMS + mCIMT arm as compared to those in sham rTMS + mCIMT arm [14 (Q1, Q3—6, 18) vs. 4 (Q1, Q3—0, 8), P = .004] and [11.1 (Q1, Q3—2.8, 25) vs. 0 (Q1, Q3—0, 8.4), P = .004] respectively (Table 2). Change in the “dissociated movements” domain score was not found statistically significant amongst both arms (P = .20). Change in the “grasps” domain score was similar across both the study groups pre- and post-therapy (P = .59). Change in upper limb speed by 9-HPG (P = .81), pull strength (P = .96), and push strength (P = .28) by dynamometry was not found to be significantly different amongst both the study groups (Table 2).
Change in Upper Limb Function at 12 weeks
The mean change in total QUEST scores at 12 weeks (8 weeks after stopping supervised interventions) was significantly higher in the real rTMS + mCIMT arm as compared to the sham rTMS + mCIMT arm (d = 3.8, 95% CI 0.4-7.2, P = .028). The improvement in QUEST domain scores of weight bearing (P = .002) and protective extension (P = .005) was sustained as well (Figure 3).
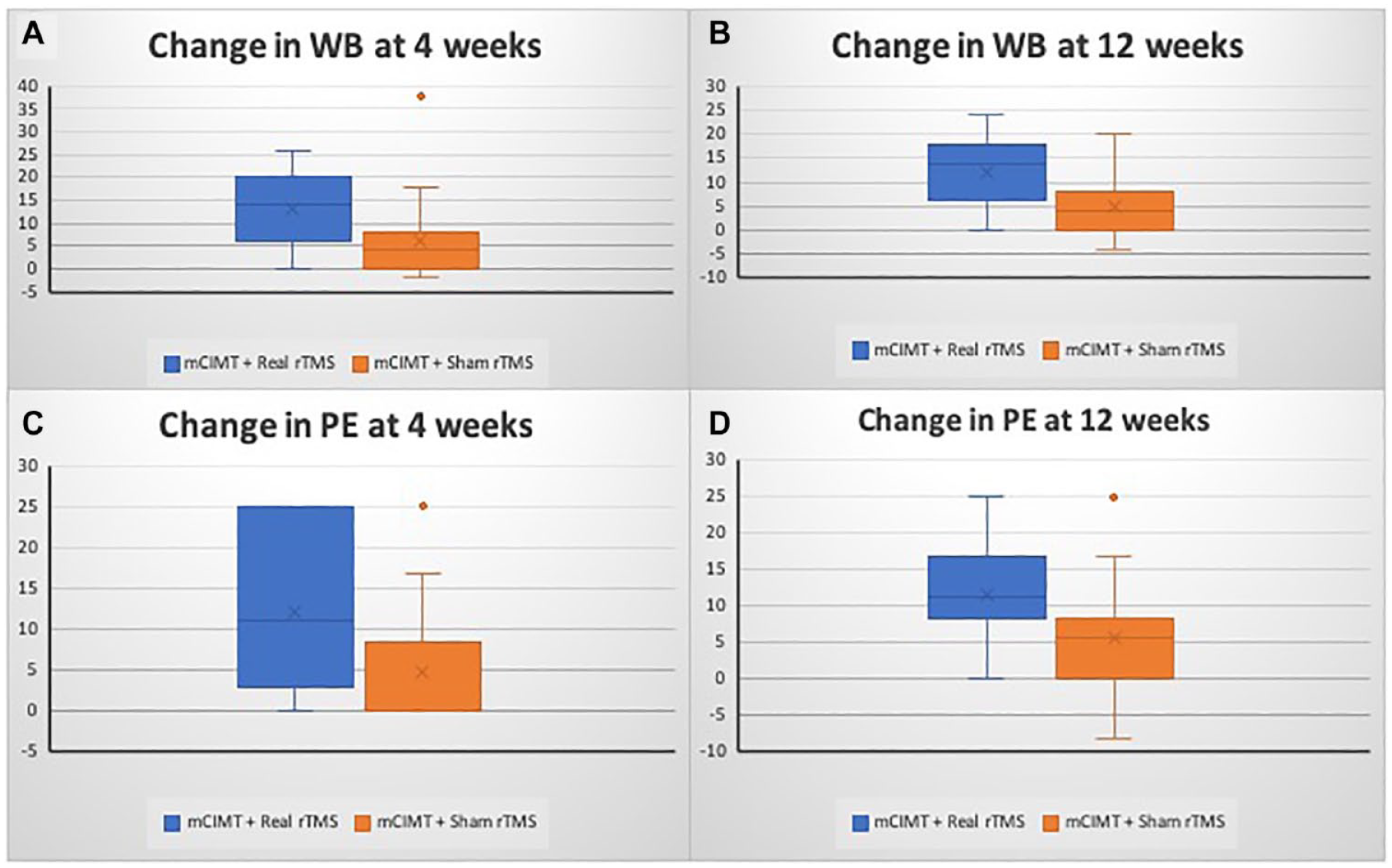
Secondary outcomes. (A) Change in Weight Bearing Domain (WB) scores at 4 weeks. (B) Change in WB Domain scores at 12 weeks. (C) Change in Protective Extension (PE) Domain at 4 weeks. (D) Change in PE Domain at 12 weeks.
Within the group comparison, both groups showed statistically significant improvement in QUEST total scores, domain-wise scores, time to complete 9-HPG scores and Pull strength and Push strength scores from baseline at 4 weeks (P < .001). This improvement was sustained at 12 weeks which is 8 weeks after stopping supervised therapy (P < .001).
CP-QOL Scores
Significantly better improvement in CP-QOL scores for “feelings about functioning” (Median [Q1, Q3], 12.5 [6.25, 12.5] vs 6.25 [6.25, 7.29], P = .01) and “participation and physical health” (Median [Q1, Q3], 9.37 [6.25, 12.5] vs 6.25 [6.25, 11.94], P = .02) domains was seen at 12 weeks in the intervention arm as compared to the control arm.
The Function of the Un-Affected Upper Limb
There was no decrease in the function of the unaffected upper limb as measured by speed and strength assessments using a 9-hole peg board and dynamometers (Figure 4).
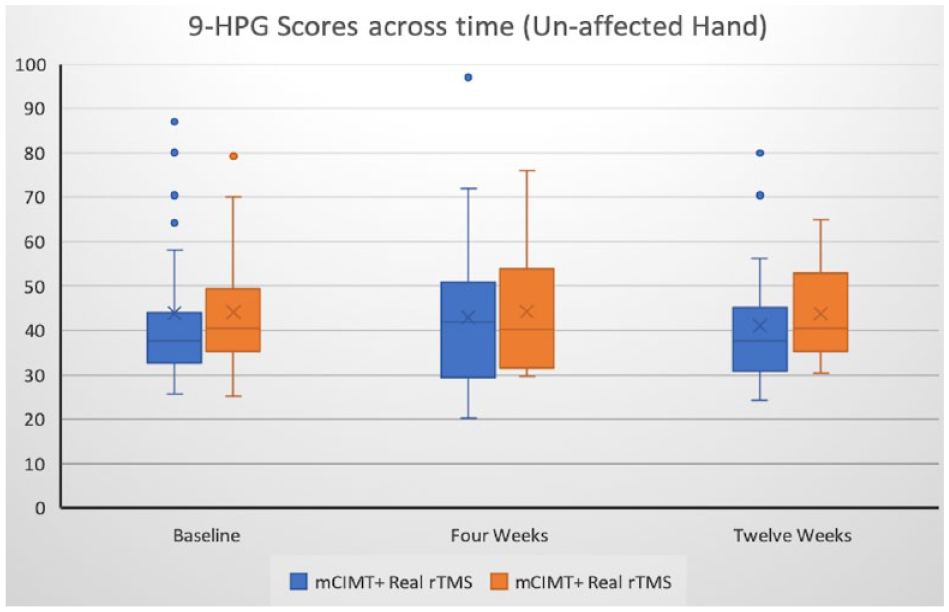
Unaffected hand function did not decrease with intervention- 9-Hole Peg Board Scores across time.
Safety
No serious adverse events were seen. Minor adverse events seen were mild and brief headache and light-headedness reported by 1 subject each which was self-remitting within a few minutes. One child, who was a known case of epilepsy, had an episode of vacant stare lasting for a few seconds, a few hours after the intervention. This child was in the sham group and this episode of brief breakthrough seizure was reported to the ethics committee.
Discussion
In this single-center, randomized, controlled trial, combining rTMS with mCIMT was found significantly better in improving upper limb function in comparison to mCIMT alone, in children with unilateral CP in the setting of a motor training program. All children enrolled in the program had a significant improvement in upper limb function which was sustained at 12 weeks follow-up. To the best of our knowledge, this is the first such study from a low-to-middle-income country. Two previous trials have shown the additive effect of rTMS and CIMT in children with congenital hemiparesis.18,19 In the PLASTIC CHAMPS trial, an improvement of 5.91 AHA logit units was seen at 6 months, in the rTMS + CIMT group as compared to the group receiving neither. Gillick et al, had reported a mean gain of 3.67 AHA logit units. A mean gain of 5.10 (1.7-8.5) in the QUEST total score was noted in the current trial, which is comparable to the previous studies.
This trial employed 6-Hz primed low frequency rTMS to the contra-lesional motor cortex. rTMS at frequency >1 Hz has been observed to enhance synaptic transmission (long-term potentiation), while low frequency rTMS (<1 Hz) has been demonstrated to decrease the efficacy of synaptic transmission (long-term depression). 33 However, there can be high inter-individual variability in response to various neuromodulation paradigms. 34 For the use of priming, we were guided by the trial by Gillick et al, in 19 children with hemiparesis. The rationale for the use of priming is based on the Bienenstock-Cooper-Munro theory. 35
In the current study’s total duration of CIMT was 56 hours, over 4 weeks. This was a relatively low dose, however, was comparable to previous studies.4,18,19 It was 10 hours in the trial by Gillick et al (5 sessions of 2-hour each), and 80 hours (20 hours of individualized, 5.5 hours of group, and 5 hours of rTMS) in the PLASTIC CHAMPS trial. In a Cochrane review, the average length of CIMT programs was 4 weeks, with a frequency of twice weekly to 7 days per week, and the mean total number of hours was 137 hours (range 20-504). 2
In addition to motor benefits, significant improvement in quality of life (CP-QOL) scores was seen. Children had to work in groups for mCIMT sessions. Most had never met another child with unilateral CP. It may be speculated that these sessions stimulated mutual interaction, and healthy competition and instilled confidence amongst children and caregivers. It would be interesting to capture this aspect of a group training program, on a subjective scale.
None of the children had any serious adverse event, which re-enforces the safety of rTMS in children as demonstrated in prior studies. 1 Only 1 child who was a known case of epilepsy and was in the mCIMT + Sham rTMS group had a brief breakthrough seizure which was self-aborted. Minor side-effects like mild headache and lightheadedness post rTMS sessions were seen in 1 child each. This is similar to past studies.36,37 Gillick et al 37 reported headache in 50% and 89% of children receiving real and sham rTMS respectively. Mild TMS-related headache was reported in 40% of children with perinatal stroke as compared to 13% of healthy children in a study reporting data from 3.5 million stimulations. 36
Most children with unilateral CP have average intelligence38,39 and it is the motor disability that poses a major challenge in performing activities of daily living. An optimum motor rehabilitation regimen can result in a significant improvement in their quality of life. There are several trials and systematic reviews on the usefulness of CIMT in this population, however, the data on TMS and its combination with CIMT is scarce.4,22,40 -42 Moreover, the data on whom to give rTMS, the optimum dose and duration of therapy is further lacking.
Besides, the number of studies from developing countries is minuscule, owing to various reasons such as the cost of the TMS machine, limited centers with TMS facilities, and a paucity of trained personnel to provide therapy, especially to children. Our institute is a tertiary care, teaching institute, which caters to multiple states in the northern part of the country. As many children would be visiting from neighboring states, it would pose a challenge to stay and travel to the hospital for therapy sessions. Hence, families were provided with free or low-cost accommodations near the institute, managed by social workers or non-government organizations.
This study has some limitations. First, the enrolment had to be closed due to a nation wide lockdown amidst the COVID-19 pandemic. Second, the follow-up could be done at 12 weeks, a longer follow-up would better estimate the long-term outcomes. Third, the last observation was carried forward for missing data, but the effect is minor as only 1 patient did not complete the trial intervention. Fourth, no individualized goal-setting (or measurement of subjective goal achievement) was done, apart from CP-QOL scores, and the subjects were not asked to guess their treatment allocation.
In conclusion, this study provides Class II evidence that combining rTMS with mCIMT was superior in improving upper limb function in children with unilateral CP as compared to mCIMT alone, which was sustained at 12 weeks.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683231174222 – Supplemental material for Brain Stimulation and Constraint Induced Movement Therapy in Children With Unilateral Cerebral Palsy: A Randomized Controlled Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683231174222 for Brain Stimulation and Constraint Induced Movement Therapy in Children With Unilateral Cerebral Palsy: A Randomized Controlled Trial by Juhi Gupta, Sheffali Gulati, Upinder Pal Singh, Atin Kumar, Prashant Jauhari, Biswaroop Chakrabarty, Ravindra Mohan Pandey, Renu Bhatia, Suman Jain and Achal Srivastava in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank the patients and their families for participation in the trial, Lily Farhat Parveen and Raman Kumar Singh for their contribution in constraint induced movement therapy; Ashish Upadhyay, Hem Chandra Sati, for their assistance in the interpretation of statistical results and the latter in randomization procedure.
Author Contributions
Juhi Gupta: Conceptualization, Methodology, Investigation, Data curation, Project administration, Writing—original draft, review and editing, Formal analysis, Visualization. Sheffali Gulati: Conceptualization, Methodology, Investigation, Writing—review and editing, Visualization. U Singh: Supervision, Investigation. Atin Kumar: Methodology, Investigation. Prashant Jauhari: Methodology, Visualization, Supervision. Biswaroop Chakrabarty: Methodology, Visualization. R M Pandey: Methodology, Formal Analysis. Renu Bhatia: Investigation. Suman Jain: Investigation, writing-original draft. Achal Srivastava: Methodology, Supervision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
The full trial protocol is available as an online appendix and is also freely available from the US National Library of Medicine (clinicaltrials.gov; NCT03792789). A deidentified dataset will be archived and upon request will be available from the corresponding author at the All India Institute of Medical Sciences, New Delhi, India.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
