Abstract
Background
Axial symptoms in Parkinson’s disease (PD) often respond poorly to pharmacological treatment. We evaluated whether combining repetitive transcranial magnetic stimulation (rTMS) and repetitive spinal magnetic stimulation (rSMS) is more effective than rTMS alone in improving axial and other motor disabilities in PD.
Methods
A total of 42 PD patients with axial symptoms were randomly allocated to 2 experimental intervention groups: Group I received active rTMS + active rSMS (2000 pulses; 20 Hz; 80% resting motor threshold for each motor area “M1” + 1500 pulses rSMS 10 Hz, at 50% of maximal stimulator output). Group II received active rTMS + sham rSMS with the same number of pulses. Both groups received 10 sessions (5 consecutive days/week for 2 weeks). Assessments using Freezing of Gait Questionnaire, walking speed, Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) parts II and III, and Parkinson’s Disease Questionaire-39 (PDQ-39) were performed at baseline (T0), end of sessions (T1), and 1 month later (T2).
Results
At T0, Group II showed higher walking speed. At T1 and T2, Group I demonstrated significantly greater improvements in MDS-UPDRS parts II, III, and sub-items of part III. Group I showed stronger improvement in TUG-T and average fast velocity immediately post-intervention, but this effect diminished after 1 month. PDQ-39 scores for leisure activity and walking problems were significantly higher in group I.
Conclusions
These findings indicate that combining rTMS and rSMS for 10 sessions is more effective than rTMS alone in managing PD’s motor and axial symptoms. The effect size of the outcome is large enough to be of significance in clinical practice.
Trial Registration:
The study was registered prospectively on 26/12/2021 at the clinicaltrial.gov website with the registration ID: NCT05271513, https://clinicaltrials.gov/study/NCT05271513.
Keywords
Introduction
Axial symptoms of Parkinson’s disease (PD), including freezing of gait (FOG), postural instability, trunk posture alterations, as well as axial rigidity have impact on patients’ quality of life and are difficult to treat with conventional physiotherapy and pharmacology. 1 Furthermore, deep brain stimulation (DBS) targeting the subthalamic nucleus or globus pallidus pars interna failed to ameliorate these symptoms 2 or, even, exacerbated them.2,3 Additionally, patients with cognitive problems or brain atrophy are ineligible for DBS.
Repetitive transcranial magnetic stimulation (rTMS) is a non-invasive technique that may provide a complementary approach. Prior research conducted by our lab has demonstrated that 10 sessions of high-frequency rTMS can enhance motor performance and increase serum dopamine concentration in PD. 4 However, the effect on axial muscles (gait disturbance, FOG, posture, and postural instability) was limited.
Over the last few years, limited clinical studies have reported mild improvement in posture instability and gait disturbances after epidural spinal cord stimulation (SCS) in patients with PD.5-7 SCS is an invasive neuromodulation method where a stimulator, electrodes, and wires connecting the stimulator, and electrodes are surgically placed inside the body. The electrodes are inserted into the dorsal epidural space of the spinal cord where they can activate axons in the dorsal columns as well as neurons in the dorsal horn of the cord. SCS carries the usual risks related to invasive surgery and post-surgery recovery. Improvements in Parkinsonian symptoms such as gait and imbalance have also been noted after epidural SCS for treatment of pain in other conditions such as leg ischemia or diabetic neuropathy. 8 In a comprehensive review of 19 studies on SCS for gait disturbances in PD, Opova et al 9 concluded that patients with advanced PD who demonstrate gait-related and postural problems may benefit therapeutically from SCS. SCS also has recently been effectively utilized to treat spasticity following spinal cord injury. 10
In contrast, repetitive trans-spinal magnetic stimulation (rSMS) is an emerging approach that employs transient magnetic pulses to induce electrical stimuli into conductive structures nearby. At low intensities, a stimulator placed over the dorsal spinal column is thought to activate axons in the dorsal roots which, in addition to branching into the ascending dorsal columns trans-synaptically recruits activity in the spinal grey matter both at the level of stimulation as well as in distant segments via propriospinal connections. Higher intensities of rSMS may activate axons within the cord such as the dorsal columns. rSMS, like epidural SCS has shown promise in alleviating motor symptoms of PD, with the benefit of increased safety and lower cost in comparison to SCS. 11
Menezes et al 11 in 2020, explored the safety and feasibility of stimulating the spinal cord at the fifth thoracic level using theta-burst magnetic stimulation on 5 PD patients with FOG. Patients received 3 sessions only, 60 min apart. After stimulation, patients had decreased freezing of gait by 22% (P = .040), 17.4% in the Unified Parkinson’s Disease Rating Scale (UPDRS) part III, and 48.2% in timed up and go. it was a small pilot study involving a small sample size and was an explorative study with no control group, The statistical analysis should be interpreted with caution. 11
Recently, Mitsui et al 12 studied the effect of real rSMS versus sham in matched groups of 50 patients receiving an experimental intervention protocol involving 2 days of rSMS a week for 4 weeks. All participants also received rehabilitation 5 days a week for 4 weeks. The primary outcome was the pre-post change in total UPDRS score. They observed a significantly higher improvement in scores in the group I than in the sham group immediately following the intervention (10.28 points), which gradually declined at 3 (5.04 points) and 6 months (2.38 points) after intervention.
Given these reports in the literature, we hypothesized that high frequency rTMS combined with rSMS might be a more effective intervention for PD patients with axial muscle symptoms than rTMS alone. The work aimed to evaluate if a combination of rTMS and rSMS is more effective than rTMS alone in improving axial and other motor disabilities in PD.
Material and Methods
Participants
About 42 out of 82 patients with PD were recruited consecutively from the neurology out-patients clinic at Assiut University, following the diagnostic criteria established by the UK Parkinson’s Disease Society Brain Bank, 13 during the period from May 2022 to May 2023.
Inclusion criteria: (1) Clinical diagnosis of idiopathic PD with gait disturbance and postural instability; (2) Age 40 to 75 years; (3) Stable anti-parkinsonian medication for ≥6 months. Gait/postural issues confirmed by: (a) Inability to rise from chair without using hands; (b) Impaired 10-foot walk (balance loss on turns, reduced step length, no heel strike, no arm swings); (c) Positive Retropulsion Test 14 (backward instability when pulled on shoulders).
For the patient’s safety, his/her relative and the doctor are standing close to him to prevent him from falling in case of imbalance.
Exclusion criteria: Age <40 or >75 years; neurological/psychiatric disorders besides PD; moderate-severe dementia (MMSE < 21) as determined by the Mini-Mental State Examination 15 ; use of antipsychotics/antidepressants; TMS contraindications (eg, intracranial metal and seizure history); inability to provide informed consent. See Figure 1 Flowchart.
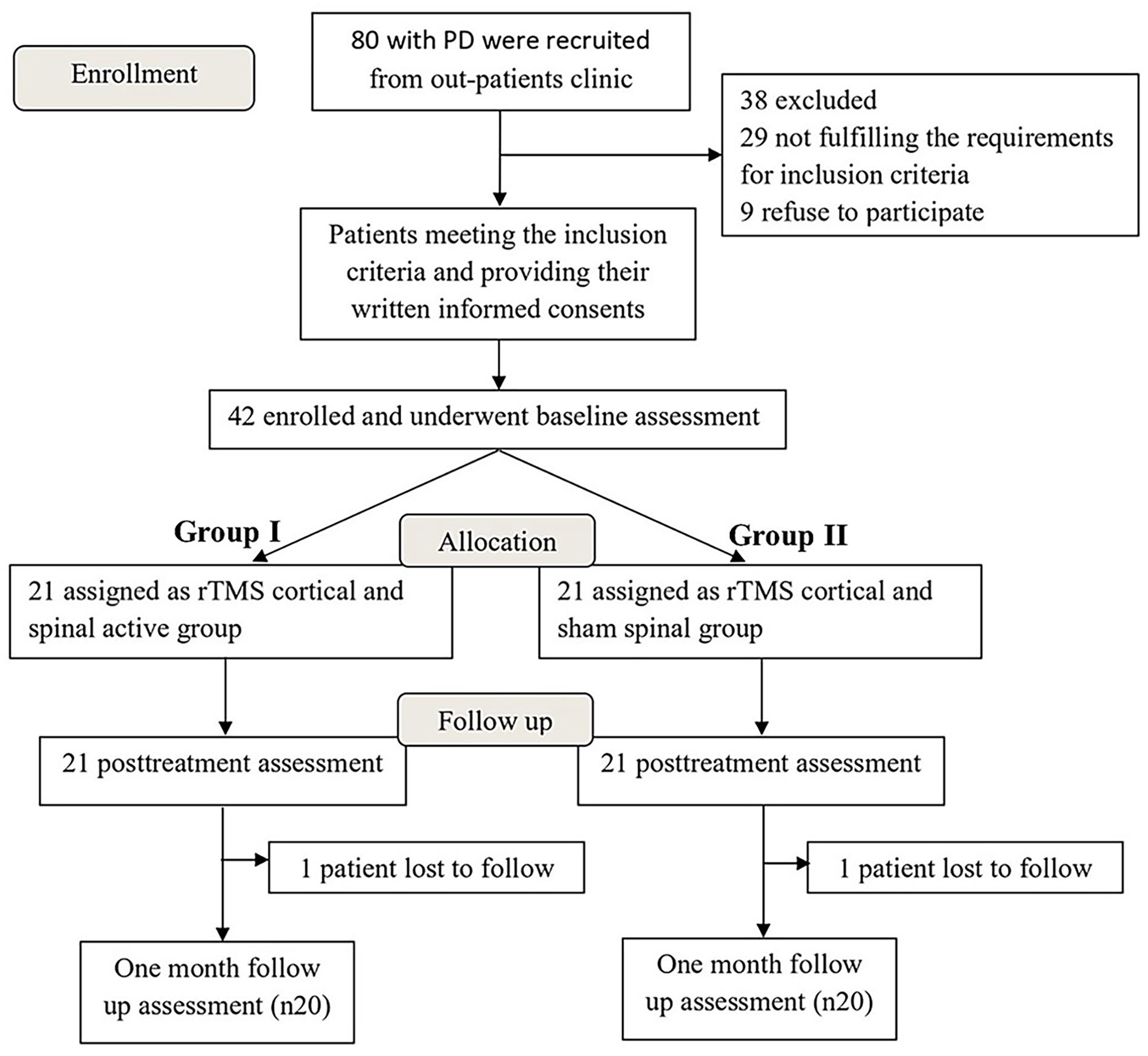
Flowchart for participants in this study (patient enrollment, allocation, and follow-up). A total of 80 patients were initially recruited; 42 met inclusion criteria and were randomized to active or sham spinal rTMS groups in addition to cortical rTMS. About 40 patients completed 1-month follow-up assessment.
Clinical Assessment
A variety of assessments were employed to evaluate the condition of each patient, including the assessment of disease severity and stage using the Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS), 16 and modified Hoehn and Yahr 17 staging. Assessments targeting the axial symptoms included using the freezing of gait questionnaire (FOG-Q), 18 the Retropulsion Test, 14 the timed up and go test (TUG),19,20 and 10-m walk test (10 MWT) including average self-selected velocity (m/s) and average-fast velocity (m/s). 21 Assessment of quality of life using the Parkinson’s Disease Questionaire-39 (PDQ-39). 22
Randomization
Patients were randomized to 1 of 2 groups utilizing opaque closed envelopes with sequential numbers. Allocation was preserved for the duration of the research. The evaluator who conducted the assessment remained blind to the specific form of stimulation. The patients were also unaware of their group allocation.
rTMS and rSMS
Procedure
Experimental intervention was performed with a figure-of-8 coil (MC-B70; MagVenture) connected with a MagPro_R20 (Medtronic Inc., USA) generator connected to a butterfly coil.
A Nihon Kohden Machine model 9400 (Tokyo, Japan) was used to amplify and record the signals. The resting motor threshold for each hemisphere was established for each patient by positioning the coil over the hot spot of the hand motor cortex (the area where the motor-evoked potentials with the highest amplitude were detected) to identify the minimum intensity required to elicit a visible movement in the first dorsal interosseous muscle of contralateral hand. 23 The magnitude of the magnetic field applied with safety considerations.
Patients have received rTMS 2 hours after their last medication (on-state). Group I received active rTMS applied over each primary motor area “M1 area” (20 Hz, 80% hand motor threshold, 10 seconds per train, 20 trains given with intertrain interval 10 seconds, with a total of 2000 pulses) plus trans-spinal magnetic stimulation (10 Hz, at intensity 50% of maximal stimulator output as at this intensity the patients feel some twitches of the upper limbs, 10 seconds per train, 10 trains with 10 seconds inter-train interval with a total 1500 pulses) for 10 sessions (5 consecutive days/week for 2 weeks).
The rationale for stimulating the hand motor area is based on the fact that the PD causes postural instability and gait abnormalities that may be associated with an arm swing reduction. As the movement of the upper extremities is important for balance control in human walking, 24 it has been recognized that enhance arm swing can improve gait and balance in patients with PD.25,26
For spinal stimulation (rSMS), the center of the coil was positioned in the midline perpendicular to the lower cervical vertebral axis, with the handle pointing downward. We selected cervical stimulation because the cervical enlargement plays a crucial role in locomotor activity and contains a significant number of interneurons involved in gait coordination and rhythm generation. 27 In the cervical region, short and long axon propriospinal neurons coordinate input between corticospinal neurons, sensory input, and motoneurons in both cervical and lumbar cord. Stimulating this region can thus potentially modulate both upper and lower limb function. 28 Interneurons of the cervical enlargement are also involved in the coordination and rhythmic generation of gait,27,29 and transmit information regarding the muscle contractile state to the lateral reticular nucleus, where they can affect locomotor regions in the pontomedullary reticulospinal system. Reticulospinal neurons have long descending axons that terminate in cervical and lumbar segments. 30 Thus, the activation of cervical afferent pathways might directly facilitate gait initiation locally and via ascending projections to supraspinal locomotor centers, as well as descending projections to lumbar segments. 31
The rationale for using high-frequency rSMS, as the few studies that applied trans-spinal magnetic stimulation reported improvement after theta burst or high-frequency trans-spinal magnetic stimulation with different level of stimulations.11,12
Group II received the same protocol except that spinal stimulation was applied with the coil edge parallel to the vertebral axis to reduce the magnetic field reaching underlying neural tissue (sham).
Follow-Up of the Patients
Patients were assessed at baseline assessment (T0), end of 10 intervention sessions (T1), and 1 month after the end of sessions (T2).
Outcomes
Primary outcomes: The primary outcome was the changes in pre-post sessions (T0–T1) and pre-post 1 month later for each rating scale (T0–T2) as gait parameters, TUG time, MDS-UPDRS, and PDQ-39 questionnaire scores.
Secondary outcomes: calculation of the effect size between groups 1 month after the end of the sessions.
Ethical Approval
The study received approval from the Ethical Committee of the Faculty of Medicine at Assiut University, which was assigned the Institutional Review Board number (IRB: 04-2023-300312). The clinical trial was officially registered with the identifier NCT05271513 on ClinicalTrials.gov. Patients provided informed written consent following the Declaration of Helsinki before the investigation.
Statistical Analysis
The data analysis in this study was conducted utilizing the Statistical Package for the Social Sciences (SPSS) software for Windows, Version 26.0. (SPSS Inc., Chicago, IL, USA). The normality of the data was assessed using the Shapiro–Wilk test. The quantitative data were presented as mean ± standard deviation (SD) or mean differences between T0 to T1 and T0 to T2 for each rating score. Categorical data were expressed as percentages (%) and numbers (n). The Mann–Whitney U test was used to compare quantitative data. The Chi-square test was used to analyze categorical data, The significance level was established as P < .05.
Effect sizes are an essential metric for quantifying the practical impact of research outcomes due to their independence from sample size. In the current study, we measured the effect sizes between groups; they ranged from medium enough to be meaningful in the real world. 32 Practical significance refers to the magnitude of the difference, which is known as the effect size. Results are practically significant when the difference is large enough to be meaningful in real life.
Results
Table 1 shows that patients in Group II exhibited a significant higher walking speed (average fast velocity in ms) than those in Group I. There were no other differences between the groups at baseline.
Demographic and Clinical Data of Studied Groups at Baseline Assessment.

The data are presented as mean ± SD or numbers (%).
Pearson Chi-Square used for comparing frequencies and perecentages. Mann–Whitney U Test compare means of 2 different groups.
Abbreviations: MDS-UPDRS, Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale; FOG-Q, freezing of gait questionnaire; PDQ-39, Parkinson’s Disease Questionaire-39; TUG-T, Timed Up & Go; 10MWT, 10-m Walk Test; rTMS, Repetitive Transcranial Magnetic Stimulation; rSMS, Repetitive Trans-spinal Magnetic Stimulation; n, number; SD, standard deviation.
Number of patients with no freezing gait.
Table 2 and Figure 2 show the mean changes in scores between the T0 to T1 and T0 to T2. Group I had significantly greater mean changes compared to Group II at both assessment points with reduced total scores in MDS-UPDRS parts II and III (improvement of Group I over Group II: 4.4 (part II) and 8.9 (part III) immediately after the experimental intervention, and 4.6 and 9.6 1 month later). Total scores in the PDQ-39 also had decreased more in Group I than II, although this was significant only at T2(Group I > Group II by 9.15 points). Supplementary Tables 1S and 2S present the mean and SD values for all measurements taken at time points T0, T1, and T2 for both groups (see supplementary material).
Differences Between the Mean Changes (Pre-post Sessions and Pre-post 1 Month After Sessions) of Different Rating Scales Among Studied Groups.
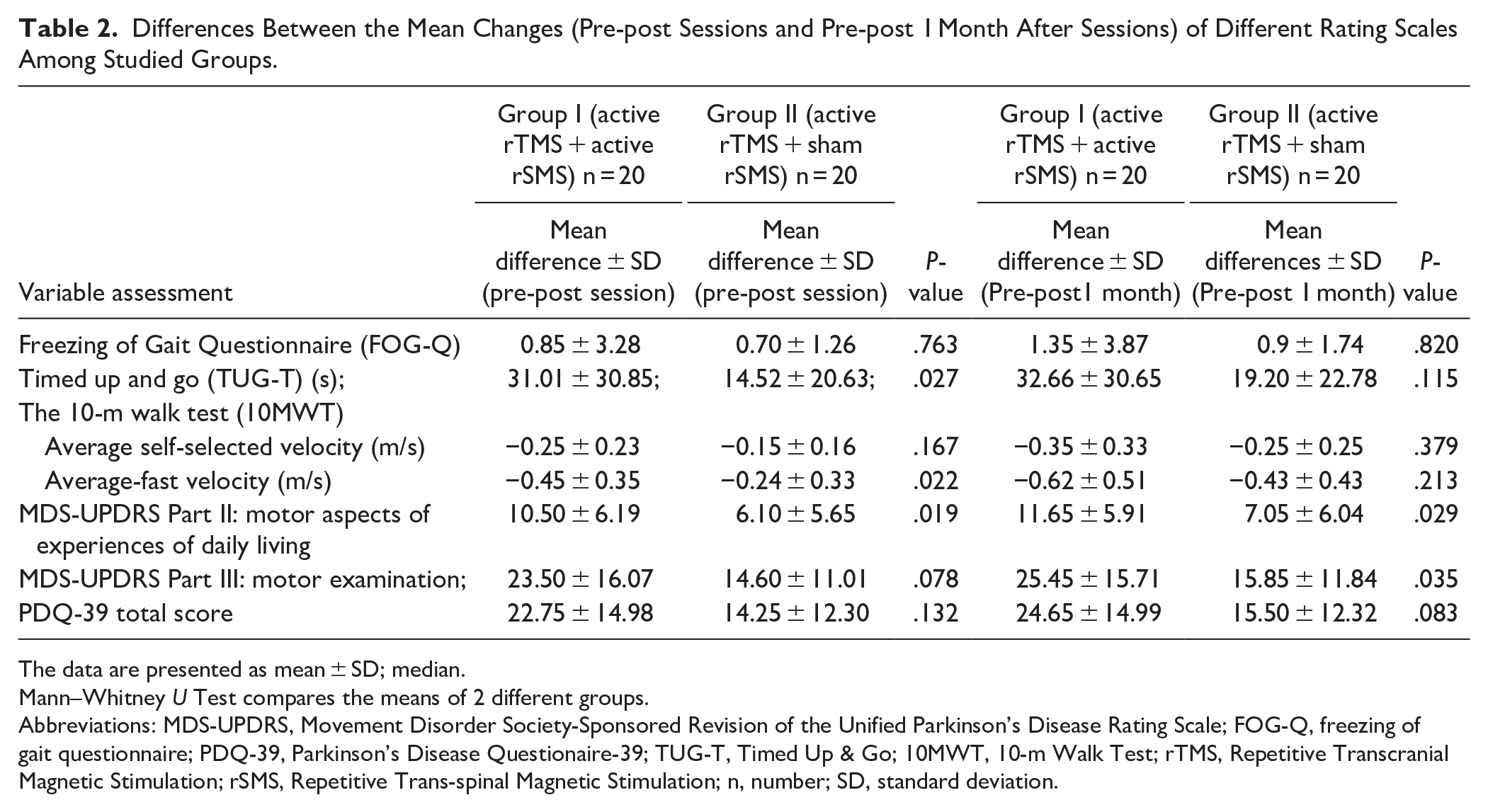
The data are presented as mean ± SD; median.
Mann–Whitney U Test compares the means of 2 different groups.
Abbreviations: MDS-UPDRS, Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale; FOG-Q, freezing of gait questionnaire; PDQ-39, Parkinson’s Disease Questionaire-39; TUG-T, Timed Up & Go; 10MWT, 10-m Walk Test; rTMS, Repetitive Transcranial Magnetic Stimulation; rSMS, Repetitive Trans-spinal Magnetic Stimulation; n, number; SD, standard deviation.
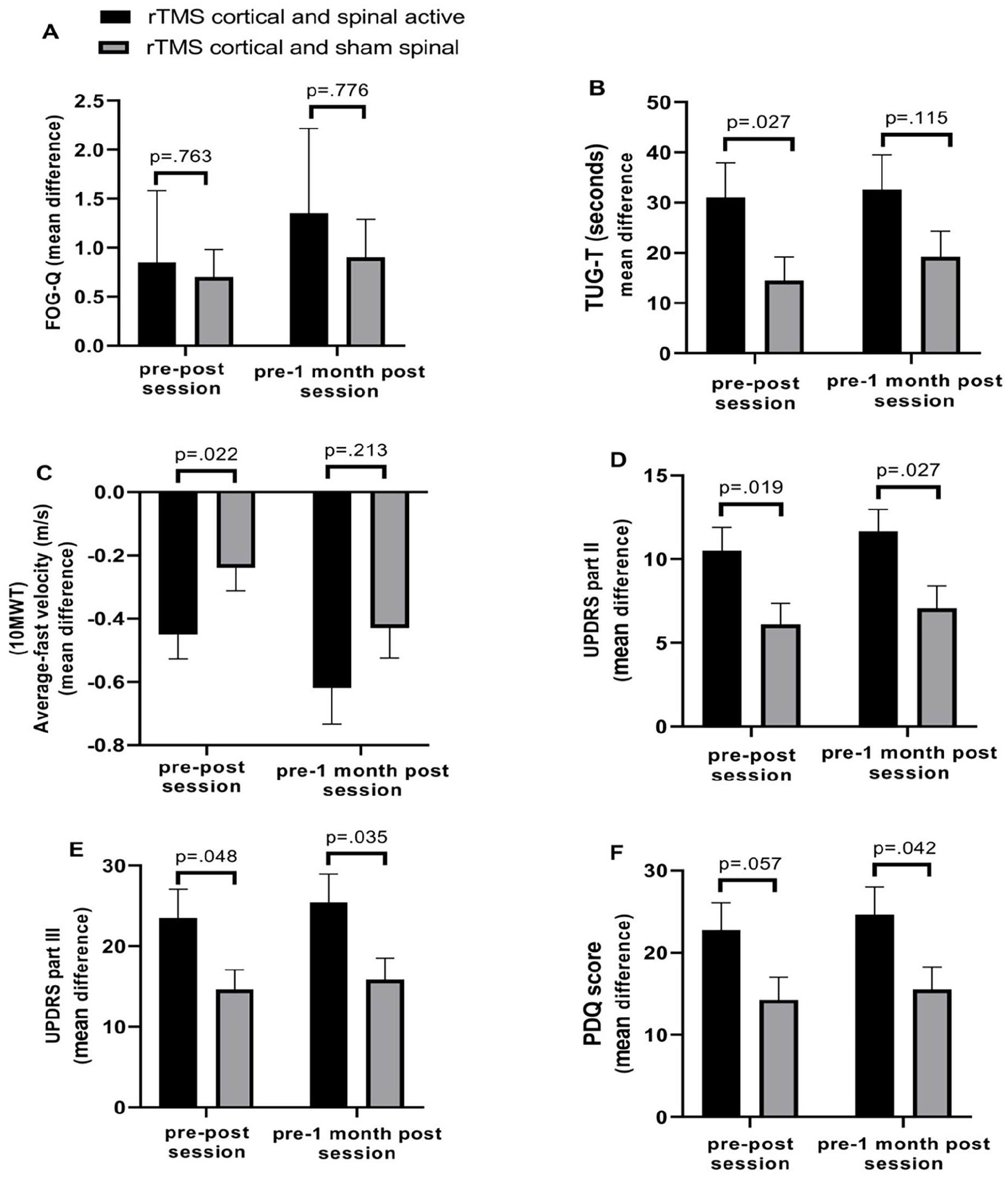
There is no significant mean difference between the 2 groups for FOG-Q either in the pre-post session assessment or the pre-post 1-month assessment (A). There is a significant mean difference in the pre-post session assessment between the 2 groups for TUG-T; however, in the pre-post 1-month assessment, the mean difference is not statistically significant (B). Regarding the 10-m Walk Test, the only mean difference in the average fast velocity was statistically significant in the pre-post session assessment (C). In terms of the MDS-UPDRS Part II and Part III (Motor Examination), there are significant mean differences between the 2 groups in both assessments, indicating better outcomes for Group I (D and E). For the PDQ-39 Total score, there is a significant mean difference in the pre-post 1-month assessment between the 2 groups (F).
Changes in TUG-T and 10 meters walk test varied between individuals. Times in both tests improved more after the intervention in Group I than II, although the effects were only significant at T1 (decreased in Group I over Group II: 16.49 seconds for TUG-T and −0.21 m/s for 10 m walk at fast velocity) but not at T2.
Table 3 shows the mean changes in scores in the T0 to T1, as well as the T0 to T2 for various sub-items of UPDRS and PDQ. Group 1 had a decreased scoring in most of the sub-items Part II of MDS-UPDRS atT2, with no significant differences between groups when evaluated immediately post-intervention (T1). Significant differences were recorded between groups in most of the subitems of MDS-UPDRS part III at both assessment times (rigidity of lower limbs, arising from chair, gait problems, postural instability, posture, and Hoehn and Yahr Stage). Subitems of the PDQ for walking half a mile and difficulty in leisure activity scored lower indicating improvement in Group I than Group II.
Difference Between Mean Changes (Pre-post Sessions and Pre-post 1 Month After Sessions) of Sub-items of Different Scales Related to Axial Motor Symptoms (MDS-UPDRS) and PDQ-39 Among Studied Groups.
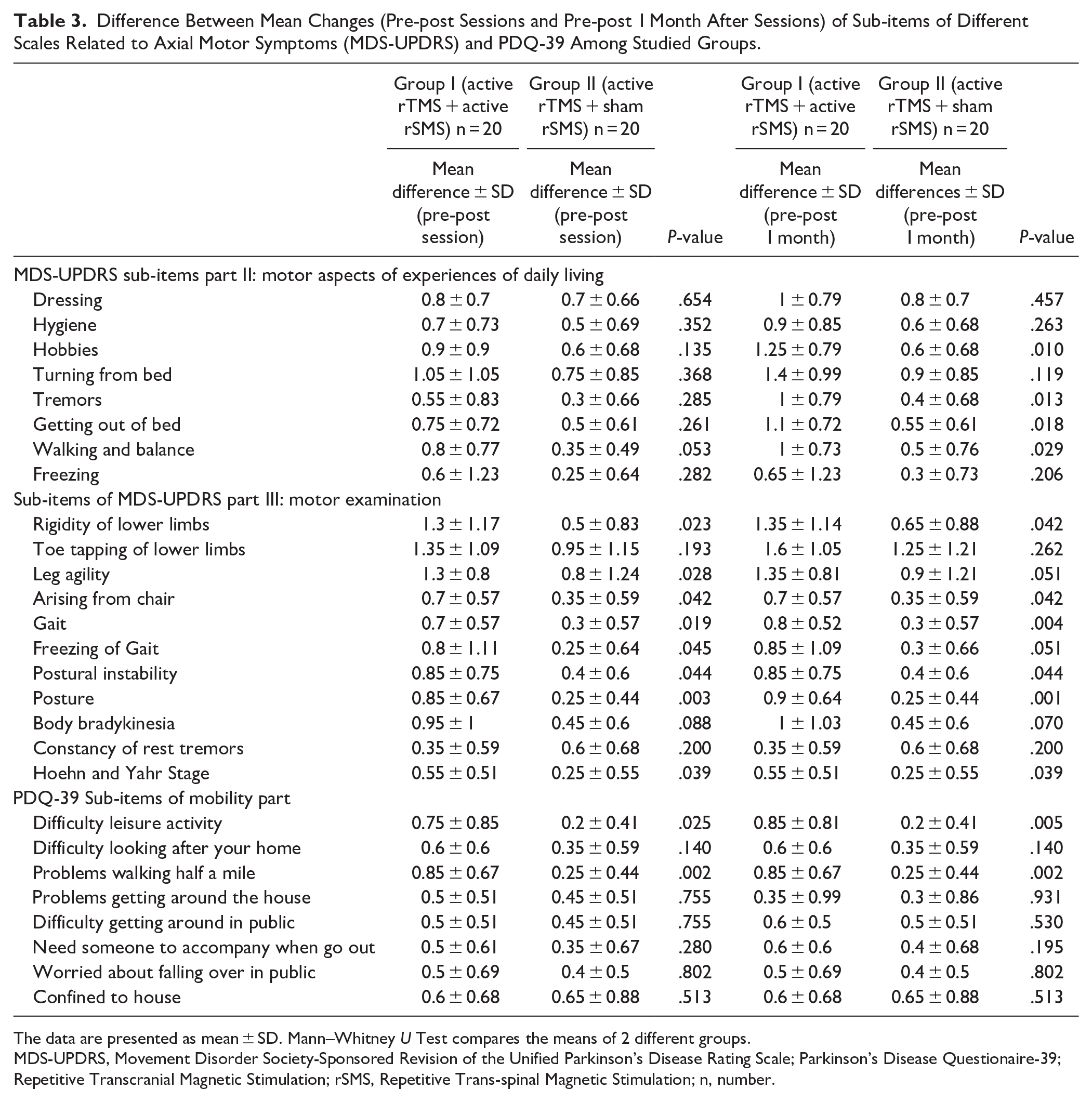
The data are presented as mean ± SD. Mann–Whitney U Test compares the means of 2 different groups.
MDS-UPDRS, Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale; Parkinson’s Disease Questionaire-39; Repetitive Transcranial Magnetic Stimulation; rSMS, Repetitive Trans-spinal Magnetic Stimulation; n, number.
Tables 4 and 5 showed the effect sizes to illustrate the practical significance of these outcomes. In most cases, these lie in the moderate range.
Effect Size of Outcome Between Groups at the End of Sessions and 1 Month Later in Different Rating Scales Among Studied Groups.
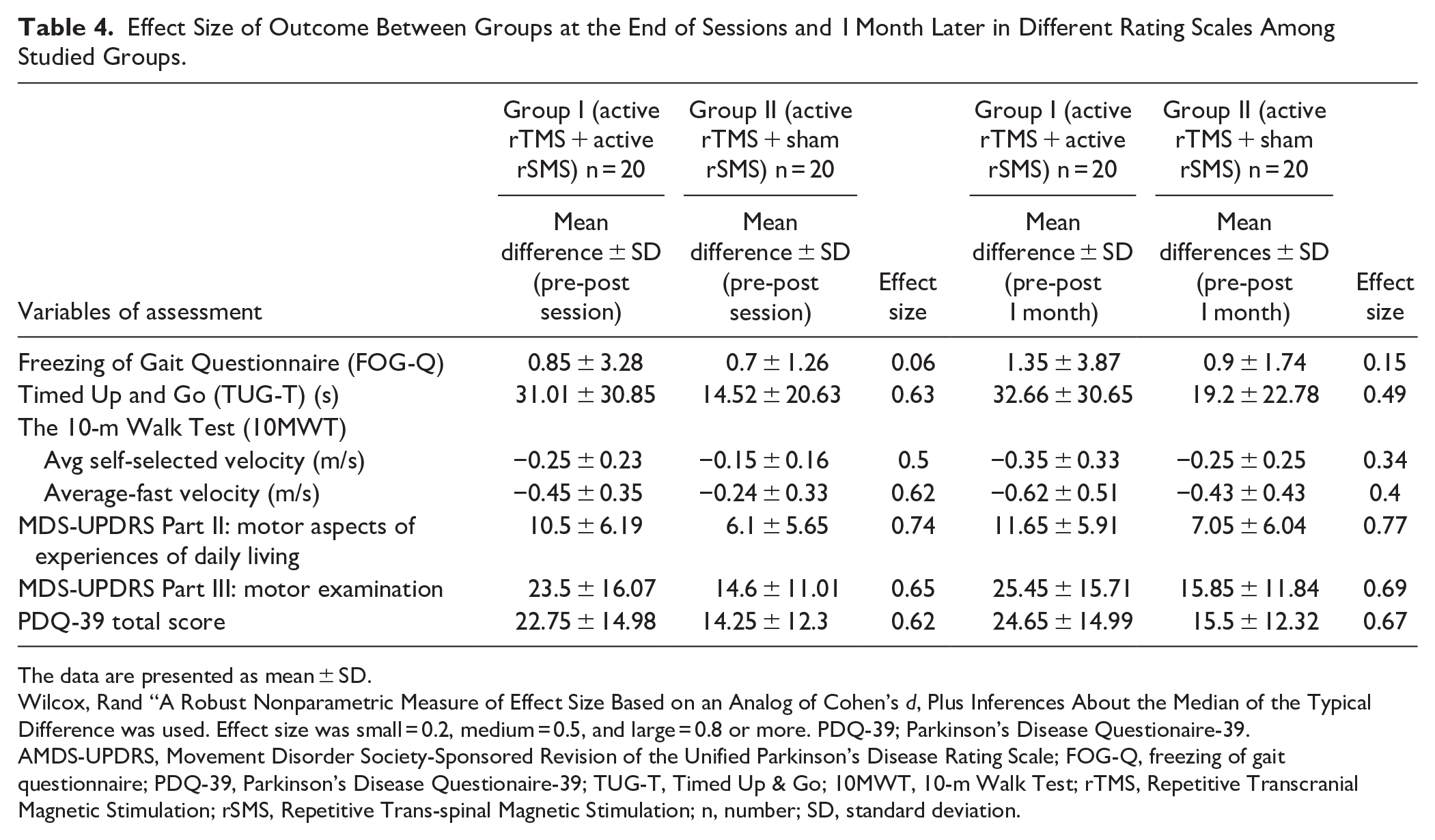
The data are presented as mean ± SD.
Wilcox, Rand “A Robust Nonparametric Measure of Effect Size Based on an Analog of Cohen’s d, Plus Inferences About the Median of the Typical Difference was used. Effect size was small = 0.2, medium = 0.5, and large = 0.8 or more. PDQ-39; Parkinson’s Disease Questionaire-39.
AMDS-UPDRS, Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale; FOG-Q, freezing of gait questionnaire; PDQ-39, Parkinson’s Disease Questionaire-39; TUG-T, Timed Up & Go; 10MWT, 10-m Walk Test; rTMS, Repetitive Transcranial Magnetic Stimulation; rSMS, Repetitive Trans-spinal Magnetic Stimulation; n, number; SD, standard deviation.
Effect Size of Outcome Between Groups at the End of Sessions and 1 Month Later in Sub-Items of Different Rating Scales Among Studied Groups.
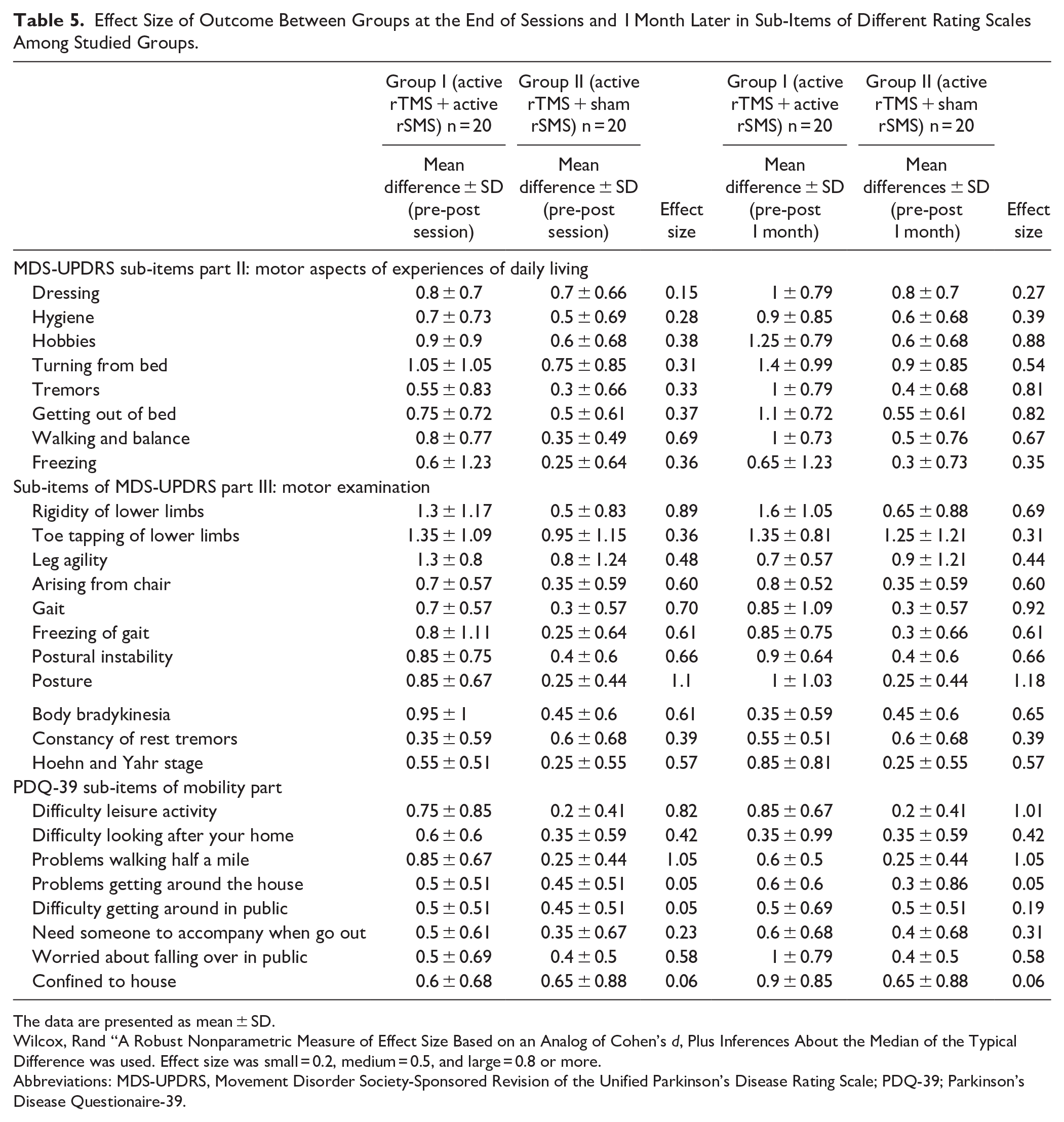
The data are presented as mean ± SD.
Wilcox, Rand “A Robust Nonparametric Measure of Effect Size Based on an Analog of Cohen’s d, Plus Inferences About the Median of the Typical Difference was used. Effect size was small = 0.2, medium = 0.5, and large = 0.8 or more.
Abbreviations: MDS-UPDRS, Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale; PDQ-39; Parkinson’s Disease Questionaire-39.
Discussion
Patients with advanced PD experience axial motor symptoms such as gait disturbance, FOG, and postural instability,33,34 which are typically resistant to dopamine replacement therapy.2,35,36 Previous studies showed improved motor function in PD after 10 sessions of high frequency of rTMS.4,37,38 However, the effect of rTMS on axial muscles (gait disturbance, FOG, posture, and postural instability) was limited. Murray et al 39 studied the effect of rSMS on cortical and subcortical neuroplasticity in healthy volunteers. Their findings supported the hypothesis that rSMS induces surround inhibition in the spinal cord and nerve axons while diminishing the impact of corticocortical inputs on corticospinal neurons. 39 This innovative technique might serve as an effective neuromodulation tool by modulating excitability at the cortex and subcortical levels in neurological illnesses.
To our knowledge, this is the first study to use a combination of rTMS and rSMS as a therapeutic approach to improve motor function in PD patients. The main result was that after intervention, scores of MDS-UPDRS parts II and III decreased indicating more improvement in Group I patients (combined rTMS and rSMS) than those in Group II (rTMS + sham rSMS) both at T1 and T2. Inspection of the sub-items of each scale of part III showed that these improvements occurred mainly in axial motor symptoms such as rigidity of lower limbs arising from a chair, gait problems, postural instability, and posture at T1 and T2. Interestingly, the sub-items of part II were only significant 1 month after the end of therapy but not immediately afterward. The TUG-T and timed 10-m walk (at high speed) improved more in Group I than Group II only when tested at T1, but this was not sustained at T2.
Arii et al 40 reported that rSMS improved symptoms of camptocormia and other motor symptoms for a few days after the intervention. Takeuchia et al 41 reported an immediate effect of intermittent rSMS (5 Hz in 8 trains of 1-second duration (40 total stimuli delivered)) on camptocormia in PD. The effect increased after twice-weekly sessions for 4 weeks.
Menezes et al 11 found a significant improvement in the MDS-UPDRS part III (P = .042) and borderline improvement in TUG-T after rSMS (P = .060), but the improvement tended to return to baseline by 28 days. However, only 5 patients were included in this pilot study. An important question is whether changes after rSMS are better than those of conventional rehabilitation. Sawada et al 42 found no differences in the effect on total MDS-UPDRS score and TUG-T in a group receiving rSMS compared to a group who received rehabilitation.
Mitsui et al 12 studied the effect of real rSMS versus sham in matched groups of 50 patients receiving protocol involving 2 days/week for 4 weeks. Patients also received rehabilitation 5 days/week for 4 weeks. They observed a significantly higher improvement in scores in Group I than sham group immediately following the end of sessions (10.28 points), which gradually declined at 3 and 6 months after sessions.
In contrast to our findings, Menezes et al recently published a double-blind clinical trial in March 2024 that investigated the efficacy of intermittent θ burst trans-spinal magnetic stimulation on 33 patients with PD with gait and balance disorders. They found that the intervention had no significant effect on total TUG gait or balance in PD patients and failed to find significant differences between groups. 43 The discrepancy between our findings and those of Menezes may be attributed to multiple differences between them: the present study combined 2 interventions (cranial and spinal), whereas Menezes et al and all previous studies applied spinal only. In the present study we applied 10 sessions at lower cervical while Menezes applied 5 sessions at third thoracic vertebra. In addition, the center of the coil was positioned in the midline perpendicular to the lower cervical vertebral axis, with the handle pointing with total number of pulses 1500 while in Menezes et al the cable pointing to right side total 1200 pulses.
Possible Mechanism of Action of rTMS and rSMS
Many studies using functional magnetic resonance imaging, TMS, and other methods have found that rTMS of the motor cortex in PD can modulate the stimulated area’s local excitability and modify connectivity to distant areas that are part of a wider motor network.44,45 It has been postulated that such changes lead to the clinical effects accompanying rTMS. The rationale for using high-frequency rTMS and rSMS was that previous work by Lomarev et al 46 and Khedr et al,38,47 who reported that high-frequency rTMS enhances motor performance in patients with PD. Lomarev et al 46 documented a gradual reduction in the execution times of walking and complex hand movement tasks. Khedr et al 47 reported that rTMS with a high frequency was more effective in PD than with a low frequency. In addition, Khedr et al 4 showed that 6 sessions of high-frequency rTMS could elevate serum dopamine concentration in PD, improving clinical symptoms.
Single pulses of SMS activate spinal dorsal root axons at the lowest threshold, followed by axons in the ventral roots as the intensity is increased. 48 Dorsal root activation evokes synaptic activity in both local spinal segments and supraspinal projection targets of ascending axons. In contrast, activity in ventral roots can antidromically activate recurrent pathways in the ventral cord, such as Renshaw cells. Thus, the long-term effect of repetitive stimulation could occur by activating many possible pathways. According to a study by Murray et al, 39 rSMS may induce surround inhibition in the spinal cord and impair the efficacy of corticocortical inputs to corticospinal neurons. The effectiveness of rSMS in treating neuropathic pain is comparable to that of SCS; however, less is known about the effects of rSMS on ascending sensory pathways. Lo et al 49 suggest that rSMS may activate these pathways. Indeed, epidural SCS has been shown to disrupt akinetic corticostriatal activity.
We speculate that rTMS and rSMS might operate by the separate mechanisms outlined above and then combine (in a bottom-up and top-down manner), influencing both the mesencephalic locomotor generator and corticospinal mechanisms.
One interesting finding was the significant difference in the mean changes in group I than group II in TUG-T and average fast-velocity of the timed 10-m walk test at T1 and loss of the differences between groups at T2 (short-term effect of rSMS for certain activity) in addition most of subitems of MDS-UPDRS part II which concerned with motor aspects of experiences of daily living as hobbies, tremors, getting out of bed and walking and balance in group I at T2 but not T1 (late effect). This finding may reflect patients’ experiences in evaluating their daily living activity, or it may be related to the attenuation of the efficacy of rTMS at T2 with more sustained efficacy of rSMS.
Limitations of the Study
The principal limitations of the study were the limited sample size and the brief duration of follow-up.
Conclusion and Recommendation
Our findings indicate that axial muscle symptoms in PD are more significantly ameliorated in individuals with PD who are treated with a combination of rSMS and rTMS instead of rTMS alone. Additional research with bigger sample sizes and longer-term follow-ups is required to find optimal stimulation protocols.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683241300547 – Supplemental material for In PD, Non-Invasive Trans-Spinal Magnetic Stimulation Enhances the Effect of Transcranial Magnetic Stimulation on Axial Motor Symptoms: A Double-Blind Randomized Clinical Trial
Supplemental material, sj-docx-1-nnr-10.1177_15459683241300547 for In PD, Non-Invasive Trans-Spinal Magnetic Stimulation Enhances the Effect of Transcranial Magnetic Stimulation on Axial Motor Symptoms: A Double-Blind Randomized Clinical Trial by Eman M. Khedr, Nourelhoda A. Haridy, Mohammad A. Korayem, Ahmed Mamdouh Tawfik and Ahmed A. Hamed in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors extend their gratitude to all participants who contributed to this study. The authors would like to thank Professor John C Rothwell, Professor of Human Neurophysiology, UCL Institute of Neurology, for his valuable comments and revision of this manuscript.
Author Contributions
Eman M. Khedr: Conceptualization; Formal analysis; Investigation; Methodology; Project administration; Supervision; Validation; Writing—original draft; Writing—review & editing. Nourelhoda A. Haridy: Data curation; Formal analysis; Methodology; Visualization; Writing—original draft; Writing—review & editing. Ahmed Mamdouh Tawfik: Data curation; Formal analysis; Investigation; Methodology; Writing—review & editing. Mohamed A. Korayem: Data curation; Methodology; Writing—review & editing. Ahmed A. Hamed: Data curation; Formal analysis; Methodology; Software; Visualization; Writing—original draft; Writing—review & editing.
Availability of Data and Materials
Any data generated or analyzed during this study can be obtained from the corresponding Author upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
