Abstract
Background. Low-frequency repetitive transcranial magnetic stimulation (rTMS) of the contralesional primary motor cortex (M1) may improve recovery in patients with hemiparetic stroke. Objective. To evaluate the effectiveness of applying 1 Hz rTMS to the contralesional M1 in addition to physiotherapy during early rehabilitation for stroke patients with hand hemiparesis in a randomized, sham-controlled, double-blind study. Methods. Forty patients with moderate upper extremity hemiparesis were randomized to receive 3 weeks of motor training (45 minutes daily) preceded by 30 minutes of 1 Hz rTMS applied to the contralesional M1 or 30 minutes of sham rTMS. Functional assessment of the paretic hand using the Wolf Motor Function Test was performed before, immediately after, and 3 months after completing treatment. Results. No statistically significant differences were found between the experimental and the control group for hand function (Wolf Motor Function Test; P = .92) or the level of neurological deficit (National Institutes of Health Stroke Scale [NIHSS]; P = .82) after treatment. Effect sizes for the experimental (d = 0.5) and the control group (d = 0.47) were small. Similar results were observed at the 3-month follow-up. Conclusions. The findings did not suggest that rTMS suppression of the contralesional motor cortex augments the effect of early neurorehabilitation for upper limb hemiparesis. Larger trials that stratify subjects based on residual motor function or physiological measures of excitation and inhibition may identify responders in the future.
Introduction
The notion of competition between hemispheres and the hypothetical negative influence of overactivity in the intact hemisphere, as a result of loss of transcallosal inhibition from the affected to the unaffected hemisphere is emerging as an important principle to understand restitution of disturbed functions after unilateral stroke. Modulation of these competitive interactions, either by increasing the excitability of the ipsilesional cortex or by decreasing excitability in the intact hemisphere, may augment neurorehabilitation training.1,2
The impact of unaffected hemisphere activity on hand paresis seems to depend on lesion site, time from onset, and size of brain lesion.3,4 Preliminary data show that low-frequency repetitive transcranial magnetic stimulation (rTMS) applied to the contralesional motor cortex decreases pathologically enhanced transcallosal inhibition toward the ipsilesional motor cortex, and thereby may improve the function of the paretic limb.5-12
Past studies of the methodological paradigm of downregulation of the nonaffected hemisphere were based on small samples, but their results are encouraging. Therefore, we designed this randomized, placebo-controlled, double-blind pilot study to evaluate the effectiveness of combination of low-frequency rTMS (applied to the intact hemisphere) and physiotherapy in patients with upper limb hemiparesis in the early stage of hemispheric stroke. These patients typically present pathologically enhanced neural activity in their unaffected hemisphere.13-15 We hypothesized that post-rTMS increase of the excitability of the affected hemisphere would boost the functional reorganization of ipsilesional motor network when combined with physiotherapy.
Methods
Patients
Written informed consent was obtained from patients, and the study was approved by the local ethics committee. Inclusion criteria were (a) male and female patients in the age range of 20 to 75 years; (b) first hemispheric stroke documented by computed tomography or magnetic resonance imaging; (c) time from onset of symptoms ≤3 months; and (d) moderate paresis of the affected upper extremity (National Institutes of Health Stroke Scale [NIHSS]—motor arm score of 1 to 3). Exclusion criteria were (a) other concomitant neurological or psychiatric illnesses; (b) history of head injury with loss of consciousness; (c) use of a drug that could have an effect on cortical excitability; and (d) severe aphasia, apraxia, dementia, perception disorders, and other serious medical conditions.
Participants were randomized by a 1:1 allocation to either an experimental (E) group receiving physiotherapy plus inhibiting rTMS or a control group (C) receiving physiotherapy combined with sham stimulation. Allocations were stored in sealed, numbered envelopes and opened only at the time of recruitment.
Based on a study by Lin et al, 16 who report effect sizes of change scores for the Wolf Motor Function Test (WMFT) as high as 0.44, we calculated that a sample size of 129 patients would be required to detect changes in functional motor ability with a power of 80%. However, strict eligibility criteria substantially reduced the number of subjects from a total of 686 stroke patients admitted to our unit during 4 years of the duration of the project. Because the participants were recruited from patients subjected to intense treatment programs, we expected higher effect sizes than that found by Lin and colleagues. Figure 1 shows the participant flow throughout the study.
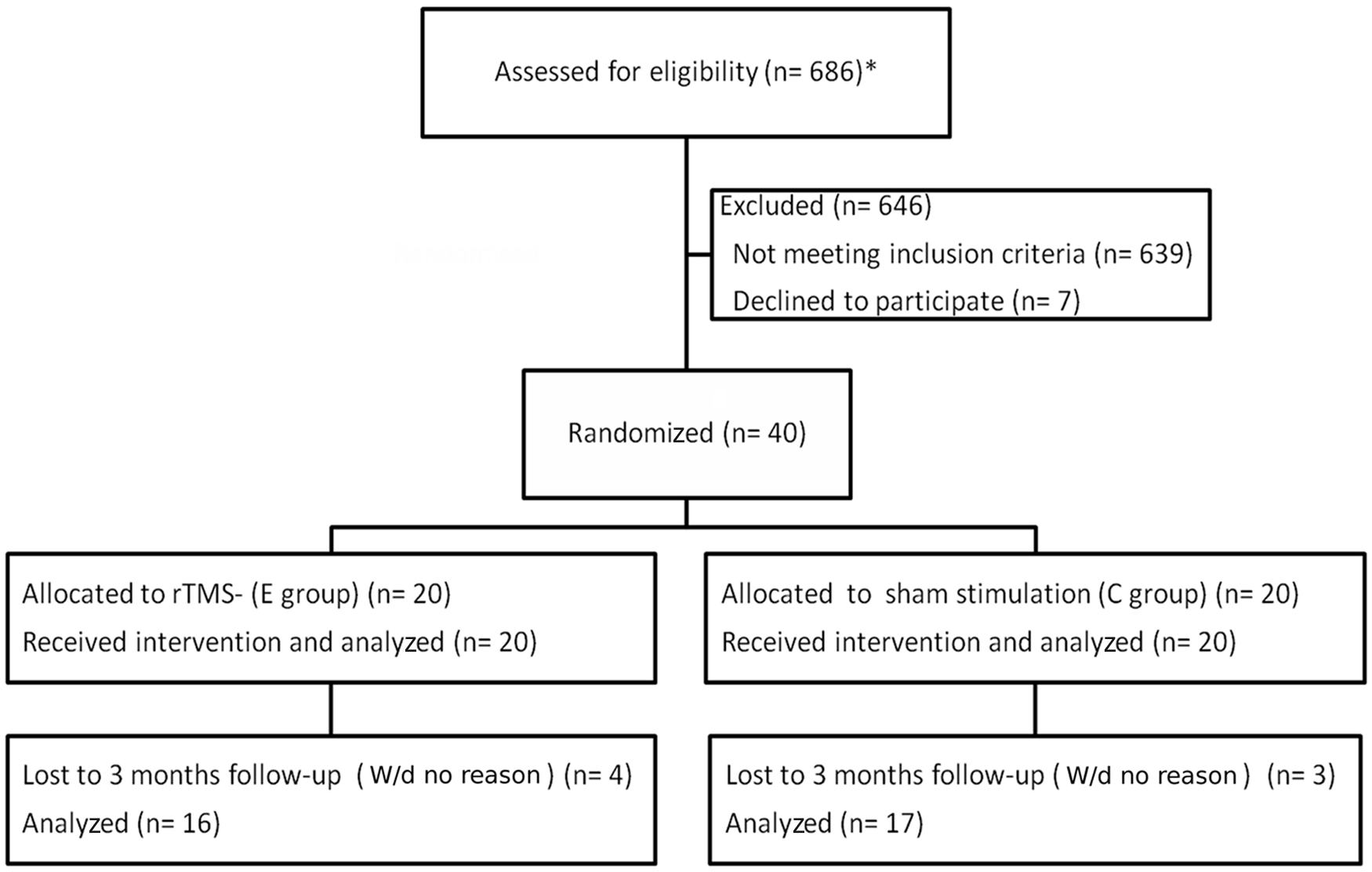
CONSORT diagram monitoring participant flow through the successive phases of the trial. *Total number of stroke patients admitted to the neurorehabilitation unit of the Institute of Psychiatry and Neurology between February 2007 and August 2010.
Procedure
One researcher (SI) was responsible for the random assignment of subjects to E or C groups and data protection. The neurologist (MB) conducted examinations and used outcome measures to assess motor function. A licensed physiotherapist led motor training. Both did not know subjects’ group allocations. Another researcher (KW, trained in TMS application) was responsible for rTMS delivery.
All participants received individual physiotherapy in accordance with the neurodevelopmental treatment/Bobath concept, which is the preferred therapeutic approach at our rehabilitation unit. 17 Motor training was performed by the same physiotherapist to minimize the effect of therapist characteristics. Although the general nature of the therapeutic method was similar for all patients, the level of difficulty and type of exercises varied depending on the severity of neurological deficit to avoid tasks that were too easy or too difficult. Motor training included active and active-assistive exercises of the affected hand (reaching, grasping, moving, and releasing various objects). The program also included gait training and practice of activities of daily living. Immediately before a 45-minute physiotherapy session, patients from E group were subjected to 30 minutes of rTMS applied to the hand area of M1 in the unaffected hemisphere to suppress its excitability. The C group received placebo stimulation. Patients did not know whether they were receiving real or sham stimulation. During rTMS, participants were repeatedly asked about their self-feeling and experienced sensations to maintain their alertness. The rehabilitation program consisted of 15 once-daily sessions, 5 days per week for 3 weeks. After the program (between posttreatment and follow-up assessments) we controlled the amount of physiotherapy that was provided in formal settings. However, all patients were encouraged to continue motor training at home after discharge. The number of hours of this individual exercise was not monitored.
Functional assessment of the paretic hand was performed using the same scales (considered as outcome measures) 3 times: before therapy, immediately after therapy, and 3 months after completion of therapy (follow-up examination). Test performance was videotaped and later rated by the same clinician experienced in motor assessment. The WMFT, 18 which consists of 15 tasks, was used to assess upper extremity function. The WMFT is highly reliable and sensitive. 19 Test outcomes were assessed in 2 respects: functional motor ability assessment (FAS; each item on a 6-point rating scale, with a maximum total score of 75) and performance time (TIME, reported in seconds).
Along with WMFT, the upper extremity part of the Fugl-Meyer Motor Assessment (FMA) 20 and the NIHSS were used as secondary outcome measures. Similarly, they were performed in pretreatment, posttreatment, and follow-up assessments. The modified Rankin Scale and Barthel Index were used only to compare the baseline disability and functional status.
Transcranial Magnetic Stimulation Procedure
Repetitive transcranial magnetic stimulation was performed by a single investigator (KW), using a Magstim Rapid Stimulator (Magstim Company, Whitland, UK) equipped with an air-cooled figure-of-eight coil (each loop 70 mm in diameter). The coil was placed tangentially to the scalp over the hand area of the primary motor cortex. The exact site of stimulation was defined as the location where stimulation of a slightly suprathreshold intensity elicited the largest motor evoked potential (MEP) in the first dorsal interosseous (FDI) muscle. The placement of the coil was maintained by marking the actual point on the skin of the head.
Magnetic stimulation was applied at 90% of the resting motor threshold (rMT) at 1 Hz frequency. A total of 1800 pulses were generated during each session. Dosing parameters were based on previous studies.5,21 The rMT was determined in each subject once before treatment and was defined as the minimum stimulus intensity able to elicit MEP of at least 50 mV in at least 5 of 10 consecutive stimulations. MEPs were recorded in a belly–tendon (respectively, cathode–anode) montage on the skin overlying the FDI muscle of the unaffected hand. The FDI muscle is relatively large, easily distinguishable from surrounding muscles, and enables close-spaced electrode placement. It is also routinely used in accordance with clinical guidelines. 22 The electromyography signal was not amplified, filtered, or digitized for further analysis.
Sham stimulation was performed with a coil that imitates the sound of a real TMS coil. The stimulation parameters were chosen in accordance with the current safety guidelines for rTMS. 23
Statistics
All analyses were performed with the SPSS software package. Normality of distribution of dependent variables was tested with the Kolmogorov–Smirnov statistic. If normality was confirmed, differences between groups were analyzed with Students t test or—when scores in subsequent assessments had to be compared—mixed model of analysis of variance (with time of assessment as a within-group factor and intervention as a between-group factor). In case of violation of the normality assumption, the Mann–Whitney U statistic was used to evaluate between-group differences, and the Wilcoxon signed rank test was used to determine the level of significance of changes between tests’ scores in the same group. Additionally, to evaluate effect size of the rTMS and sham stimulation, Cohen d (mean change score divided by pooled standard deviation) was calculated twice for each group in every analysis. The first effect size refers to the change between pretreatment and posttreatment assessment, whereas the second reflects long-term effects assessed as a change from pretreatment to follow-up assessment. According to Cohen criteria, 24 effect sizes of 0.2 to 0.5 are small, 0.5 to 0.8 are moderate, and greater than 0.8 are large. Categorical data referring to baseline characteristics with sufficient numbers of observations were assessed using the χ2 test. Additional analyses were performed to investigate differences in the proportion of WMFT tasks that could not be completed in 2-minute limit both at baseline and posttreatment. The generalized estimating equation model for the analysis of repeated binary data (completed or not completed) was used for each of the 15 items and for the total number of incomplete tasks according to the procedure described by Wolf et al. 25 The 2 factors, time and intervention, as well as their interaction were assessed. All tests were 2 sided and P < .05 was considered statistically significant, unless multiple comparisons were performed. In that case a Bonferroni correction was applied.
Results
Forty participants (39 right-handed and 1 left-handed) with upper extremity paresis undergoing rehabilitation were entered (Table 1). There were no significant differences between the 2 groups (E and C) in either demographic and clinical characteristics (Table 1) or average scores of outcome measures at baseline (Table 2).
Demographic and Clinical Characteristics
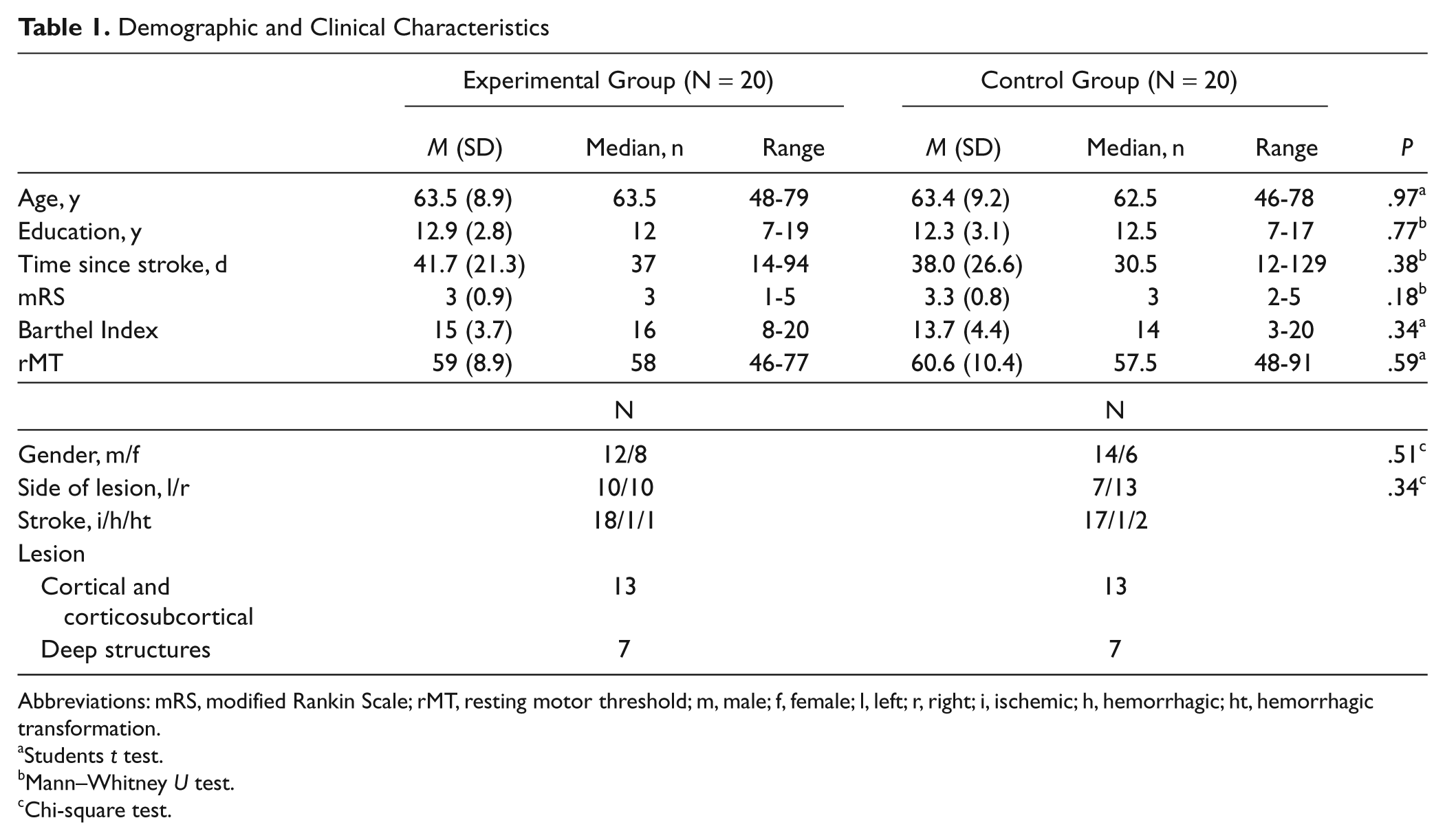
Abbreviations: mRS, modified Rankin Scale; rMT, resting motor threshold; m, male; f, female; l, left; r, right; i, ischemic; h, hemorrhagic; ht, hemorrhagic transformation.
Students t test.
Mann–Whitney U test.
Chi-square test.
Results of Clinical Scales in the Whole Group (N = 40)
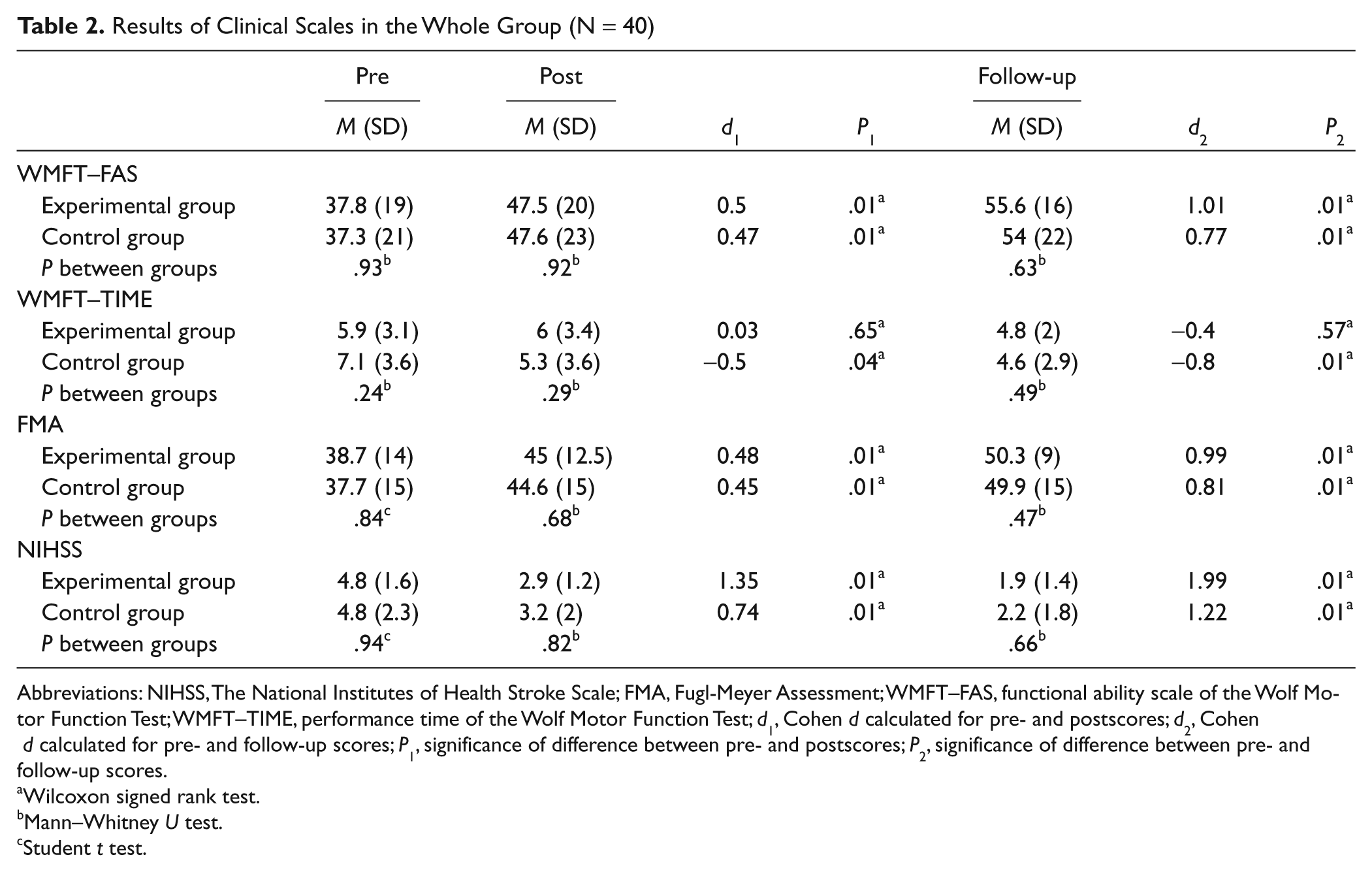
Abbreviations: NIHSS, The National Institutes of Health Stroke Scale; FMA, Fugl-Meyer Assessment; WMFT–FAS, functional ability scale of the Wolf Motor Function Test; WMFT–TIME, performance time of the Wolf Motor Function Test; d1, Cohen d calculated for pre- and postscores; d2, Cohen d calculated for pre- and follow-up scores; P1, significance of difference between pre- and postscores; P2, significance of difference between pre- and follow-up scores.
Wilcoxon signed rank test.
Mann–Whitney U test.
Student t test.
In posttreatment assessment, the results obtained by both groups in all 3 outcome measures were significantly better than baseline scores. However, they were still similar, with no significant between-group differences in WMFT–FAS (U = 196.5, P = .92), WMFT–TIME (U = 161; P = .29), FMA (U = 185; P = .68), or NIHSS (U = 192; P = .82). Similarly, effect sizes for both groups were small (near the upper point) except for NIHSS, in which the effect size was large for the E group and moderate for the C group (Table 2). The proportion of tasks that could not be completed declined in both groups, with total number of incompletes dropping from 79 (26%) to 44 (15%) in the E group and from 81 (27%) to 47 (16%) in the C group. However, the generalized estimating equation model analysis did not show any statistically significant interactions (none of the P values reached the .003 level following the Bonferroni correction). There were 4 occurrences (in both groups) of unsuccessful performance in posttreatment assessment, although the same tasks could be performed at baseline.
Therefore, the analysis revealed that type of treatment had no significant impact on the clinical or functional state of our patients.
Four subjects from the E group and 3 from the C group were lost to follow-up. The comparison of the scores in the follow-up assessment demonstrated additional, significant improvement from baseline in both groups; however, no significant between-group differences were observed in WMFT–FAS (U = 122.5; P = .63), WMFT–TIME (U = 117; P = .49), FMA (U = 116; P = .47), or NIHSS (U = 124; P = .66). Large or near-large effect sizes were observed in all outcome measures, with slightly higher Cohen d in the E group (Table 2).
E group received more sessions of formal physiotherapy between posttreatment and follow-up assessments than C group (mean = 25.6, SD = 18 vs mean = 15.5, SD = 15); however, the difference was not statistically significant (U = 95.5; P = .14).
Additional Analyses
Subgroup with dominant-hand paresis
The E group consisted of 11 patients (6 men and 5 women) and the C group consisted of 7 patients (3 men and 4 women) with paresis of the dominant hand. One person from the E group was left-handed, whereas all others (in both groups) were right-handed. No significant between-group differences were observed in baseline characteristics.
Both groups’ scores improved significantly posttreatment, with Cohen d ranging from 0.32 to 0.55 for both WMFT–FAS and FMA, indicating small to moderate effect sizes. In NIHSS, the effect size of change scores was large for both groups. Again, there were no significant between-group differences in WMFT–FAS (U = 33.5; P = .66), WMFT−TIME (U = 28; P = .34), FMA (U = 36; P = .82), or NIHSS (U = 35.5; P = .78).
Three patients from the E group and 1 from the C group were lost to follow-up. Analyses revealed no significant differences in WMFT–FAS (U = 21; P = .7), WMFT–TIME (U = 15; P = .25), FMA (U = 22.5; P = .85), or NIHSS (U = 21.5; P = .74) between the 2 groups. Effect sizes were large for all outcome measures in the E group, whereas in the C group the only large effect size was observed in NIHSS. E group received significantly more days of rehabilitation (mean = 30.9, SD = 17.6) than the C group (mean = 9.5, SD = 12.6) between posttreatment and follow-up exams (U = 6; P = .02), which might have had an impact on the results of the follow-up assessment.
Subgroup with nondominant-hand paresis
The E group consisted of 9 nondominant-hand paretic patients (6 men and 3 women), and the C group had 13 (11 men and 2 women) such patients. No significant differences were observed between groups at baseline.
Posttreatment WMFT–FAS scores did not differ significantly between the 2 groups (U = 46; P = .4); neither did WMFT−TIME (U = 25; P = .33), FMA [F(1, 20) = 0.001; P = .97] or NIHSS [F(1, 20) = 1.019; P = .33].
One patient from the E group and 2 from the C group were lost to follow-up. There were no significant between-group differences in the amount of posttreatment rehabilitation, t(17) = 0.215; P = .83. No differences were observed in WMFT–FAS (U = 40; P = .74), WMFT−TIME (U = 15; P = .25), FMA (U = 41; P = .8), or NIHSS (U = 53.5; P = .46).
Subgroup with cortical and corticosubcortical lesions (without deep ganglia)
After excluding patients who lacked cortical lesions, the E group consisted of 13 patients (6 men and 7 women) and the C group also comprised 13 patients (8 men and 5 women). There were no significant differences between groups at baseline.
Mixed model analysis of variance revealed that the type of therapeutic intervention had no significant effect on WMFT–FAS scores, F(1, 24) = 0.099; P = .76, in this subgroup of patients. Likewise, no significant difference between groups was observed in WMFT−TIME (U = 80; P = .82), FMA (U = 69; P = .43), and NIHSS (U = 64; P = .28).
Four patients from the E group and 2 from the C group were lost to follow-up. The 2 groups received similar amounts of rehabilitation between posttreatment and follow-up assessment, t(18) = 1.309; P = .21. Although both groups significantly improved their outcomes, there were no significant between-group differences in WMFT–FAS (U = 41.5; P = .54), WMFT–TIME (U = 47; P = .85), FMA (U = 43; P = .62), or NIHSS (U = 35.5; P = .28). All effect sizes in the E group were large, whereas in the C group effect sizes were large for NIHSS but moderate for WMFT–FAS and FMA.
Discussion
Our study addressed the issue of combining 1 Hz rTMS applied to the contralesional motor cortex with physiotherapy for rehabilitation of upper extremity hemiparesis. Compared with previous studies, we attempted to verify the effectiveness of rTMS on a relatively large sample of patients in a randomized, placebo-controlled, double-blind trial early after stroke.
The employed rTMS protocol is considered to reduce overactivity in the contralesional motor cortex and subsequently to enhance the neural activity in ipsilesional M1.12,26 The mechanisms underlying the inhibitory rTMS-induced effect and its influences on functional interactions within the entire motor network are not yet fully understood. We expected that behavioral motor improvement might result from enhancing the activity of the remaining neurons in the primary motor cortex and unmasking some preexisting, functionally latent, neural connections around the stroke lesion. This strategy had appeared to be effective in prior single or consecutive multisession trials,5-9,12,27 although most of those trials involved participants who were further removed from the time of onset and involved rather small samples.
Nevertheless, the results of our study do not confirm the preliminary hypothesis that the application of low-frequency rTMS inhibiting the contralesional motor cortex significantly improves restitution of paretic hand function. The lack of significant differences in behavioral motor performance between our experimental and control groups was confirmed by posttreatment measurement, as well as in the follow-up assessment. Similarly, we did not observe any differences after distinguishing patients with dominant- and nondominant-hand paresis, or patients with cortical and corticosubcortical lesions. Spontaneous recovery and motor training were the more powerful factors influencing gains, while rTMS had an imprecise if any impact on improvement.
Limitations include the lack of neurophysiological data to confirm the assumed overexcitability of the unaffected hemisphere, the lack of measurement of the degree of its suppression by rTMS, and reduced statistical power of the trial. However, because effect sizes (concerning WMFT–FAS scores) in our sample proved higher than expected, resulting in statistical power of 0.43 to 0.46 for pretreatment to posttreatment comparisons and 0.7 to 0.88 for pretreatment to follow-up comparisons, our analyses appear fairly reliable, at least for long-term effects.
An optimal means of functional reorganization involving both hemispheres in patients with poststroke hand hemiparesis still remains to be found. One should take into consideration that, because structural and functional pathology may extend into secondary or tertiary cortical areas, impairments on the level of higher order organization of motor activities (e.g., cognitive or executive regulation) may co-occur and affect the quality of motor performance. For example, involvement of the ventral premotor cortex for planning and execution of hand movement, 28 or the supplementary motor cortex for the temporal organization of movements, 29 could alter the response to rTMS or the TMS could dysregulate connected structures.21,26,30,31 Handedness may also affect how the hemisphere executes movements. 32 The intensity of stimulation is another factor that might have influenced our results. We used 90% of rMT, whereas other researchers usually applied higher values. We assumed, after Takeuchi et al, 5 that subthreshold stimulation may act by local inhibition of the stimulated site, while suprathreshold stimulation may also change the opposite homogeneous motor areas via transcallosal inhibition. Functional effects of rTMS inhibition over the contralesional motor cortex might also depend on the size and spatial distribution of the brain lesion, as well as on the initial degree of functional impairment. 33
Appropriate correction of these neurophysiological processes requires complete knowledge about mutual interactions between elements of the neural functional network and their significance for the quality of complex motor activities.
Conclusion
Our study suggests that suppression of contralesional motor cortex activity by rTMS is not yet an evidence-based method during early neurorehabilitation for patients such as those in our trial. Larger trials that stratify participants, perhaps, based on individual patterns of cortical activation or by degree of hand motor function, may be warranted.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by a grant from the Polish Ministry of Science and Education Nr S 006/P-N/2007/01.
