Abstract
Background. Motor-learning interventions may improve hand function in children with unilateral cerebral palsy (UCP) but with inconsistent outcomes across participants. Objective. To examine if pre-intervention brain imaging predicts benefit from bimanual intervention. Method. Twenty children with UCP with Manual Ability Classification System levels I to III, aged 7-16 years, participated in an intensive bimanual intervention. Assessments included the Assisting Hand Assessment (AHA), Jebsen Taylor Test of Hand Function (JTTHF) and Children’s Hand Experience Questionnaire (CHEQ) at baseline (T1), completion (T2) and 8-10 weeks post-intervention (T3). Imaging at baseline included conventional structural (radiological score), functional (fMRI) and diffusion tensor imaging (DTI). Results. Improvements were seen across assessments; AHA (P = 0.04), JTTHF (P < .001) and CHEQ (P < 0.001). Radiological score significantly correlated with improvement at T2; AHA (r = .475) and CHEQ (r = .632), but negatively with improvement on unimanual measures at T3 (JTTFH r = −.514). fMRI showed negative correlations between contralesional brain activation when moving the affected hand and AHA improvements (T2: r = −.562, T3: r = −0.479). Fractional Anisotropy in the affected posterior limb of the internal capsule correlated negatively with increased bimanual use on CHEQ at T2 (r = −547) and AHA at T3 (r = −.656). Conclusions. Children with greater structural, functional and connective brain damage showed enhanced responses to bimanual intervention. Baseline imaging may identify parameters predicting response to intervention in children with UCP.
Introduction
Unilateral cerebral palsy (UCP) is characterized by motor impairment predominantly lateralized to one side of the body. This results in unimanual and often bimanual movement difficulties when performing 2-handed actions needed for independence in many daily activities. 1 UCP affects approximately 1 in 1500 births.2,3 It typically occurs as a result of unilateral brain abnormality such as brain malformation, periventricular white matter lesions, corticosubcortical lesions, or postnatal injury. 4
The pathophysiology of UCP is, however, complex and not limited to unilateral distribution, with, more typically, some influence also on less-affected hand function. 5 The type, extent, and timing of cerebral insult influences brain development and reorganization particularly affecting sensorimotor areas and the white matter tracts that arise from them, such as the corticospinal tract (CST).6-9 Large variability is demonstrated in brain imaging findings 9 and degree of hand function impairment.10,11
Classification of children with CP has tended to focus on topography of distribution of motor impairment (eg, UCP), movement type (eg, spastic), or severity of motor function (eg, Gross Motor Function Classification System), albeit with variations in the measures used to identify particular movement problems and subsequent quantification. 12
These classification models have been used to consider longitudinal outcomes in CP but have been less useful in predicting treatment response.13,14 The development of neuroimaging has enabled improved understanding of the pathophysiology in these children along with the understanding that children with UCP with similar unilateral clinical presentations demonstrate wide variations of findings on neuroimaging. 9 Yet many questions regarding etiology persist regarding the impact of etiology on function and development. 15
Advances in neuroimaging and electrophysiological methods have allowed further progress in the field of pediatric neurodisability, with appreciation of abnormalities on microstructural neuroanatomy as seen in diffusion tensor imaging (DTI), 8 on measures of real-time brain function as seen in functional MRI (fMRI), 7 and on cortical mapping and excitability as measured with transcranial magnetic stimulation (TMS). 16 fMRI signals are an indirect measure of neural activity. The BOLD (blood-oxygen-level dependent) contrast mechanism reflects changes in cerebral blood volume, cerebral blood flow, and oxygen consumption and relies on the fact that cerebral blood flow and neuronal activation are coupled. 17 In typically developed individuals, unilateral activation is expected in the primary motor cortex of the contralateral hemisphere to the hand engaged in movement (henceforth referred to in this article as typical activation). However fMRI and TMS have shown atypical activation patterns, including bilateral activation and activation in the ipsilateral hemisphere to the hand engaged in movement in UCP.18-22
Parallel to advances in neuroimaging of UCP have been developments of effective motor learning–based interventions, including unimanual and bimanual training such as constraint-induced motor therapy (CIMT) and hand arm bimanual training (HABIT).14,23-28 However, results are inconsistent across studies with respect to the extent of change and including some participants not demonstrating improvement. 29 There is a need to determine what treatment and regimen is best for whom. 3
The advances in neuroimaging, along with lack of uniform improvement in those with UCP who underwent motor learning–based interventions, have led investigators to try to identify predictors of treatment response in order to determine which children with which type of imaging findings may benefit most. Studies to date have been preliminary, with small sample sizes and limited, almost exclusively, to children undergoing CIMT.
Islam et al, 6 reporting results for children with UCP who underwent CIMT, found improved motor outcomes to be independent of the baseline corticomotor projection pattern (contralateral, mixed, ipsilateral) and structural MRI lesion characteristics. Rickards et al 31 reported on 2 studies in children with CP and adults after stroke who underwent CIMT. They found that neither integrity (fractional anisotropy [FA]) nor distorted or disrupted path of the CST predicted response to outcome; nor did it prevent participants from benefiting maximally from intervention. Friel et al 32 reported that peduncle asymmetry, an estimate of CST dysgenesis, was a poor predictor of training efficacy in children with hemiplegic CP who underwent a bimanual intervention.
Reports by Kuhnke et al 30 and Rocca et al 33 and, most recently, Manning et al, 34 identified more definitive imaging predictors for children with UCP undergoing motor interventions. For example, Kuhnke et al 30 found that children with UCP undergoing a CIMT intervention progressed differently; those whose paretic hand was controlled by preserved crossed projections made progress accompanied by a significant gain in speed, whereas patients with the paretic hand controlled by ipsilateral projections tended to show speed reduction.
The aim of our study was to examine whether structural, functional, and microstructural MR imaging may predict response to treatment in children with UCP undergoing a themed variation of HABIT. Specifically, we examined whether the degree of overall brain injury, the functional motor activation at the sensorimotor cortex, or the white matter (WM) integrity of CSTs and the corpus callosum (CC) may predict response to treatment in children with UCP undergoing a themed variation of HABIT. A better understanding of the differences in baseline imaging parameters between these children might improve our ability to target treatment protocols effectively. Our hypothesis was that both structural (MR), functional (fMRI), and microstructural (DTI) imaging biomarkers of the affected hemisphere and connecting neural pathways would predict which children with UCP would make progress following a bimanual intervention. Response to treatment was considered within the framework of the International Classification of Functioning, Disability and Health–Children and Youth Version (ICF-CY) to consider impact of movement disorder on body function and activity performance. 35
Materials and Methods
Participants
Children identified with UCP and determined to be eligible by pediatric neurologists and pediatricians were recruited from regional hospitals and/or child developmental centers in Israel and the United Kingdom. Baseline preintervention imaging data (including standard MR, fMRI, and DTI studies as noted later in the “Imaging Assessments” section) were available for 20 children with UCP (11 boys; mean age = 10 years, 11 months ± 1 year, 9 months; range = 7 years, 9 months to 16 years, 3 months) with manual capacity measured via the Manual Ability Classification System (MACS) 36 at levels I to III, who subsequently participated in one of three 2-week summer camps that used a “magic” theme-based variation of the HABIT program for improving activity performance. 14 Diagnosis, MACS level, and any coexisting conditions were documented at baseline assessment and verified via medical records. Three children were known to have attention-deficit hyperactivity disorder (participants 4, 6, and 11), 2 of whom had received pharmacological intervention (methylphenidate) for this but not during the period of intervention. Two other children were known to have some mild learning difficulties (participants 5 and 17). Participant characteristics are presented in Table 1.
Participant Characteristics: Clinical.
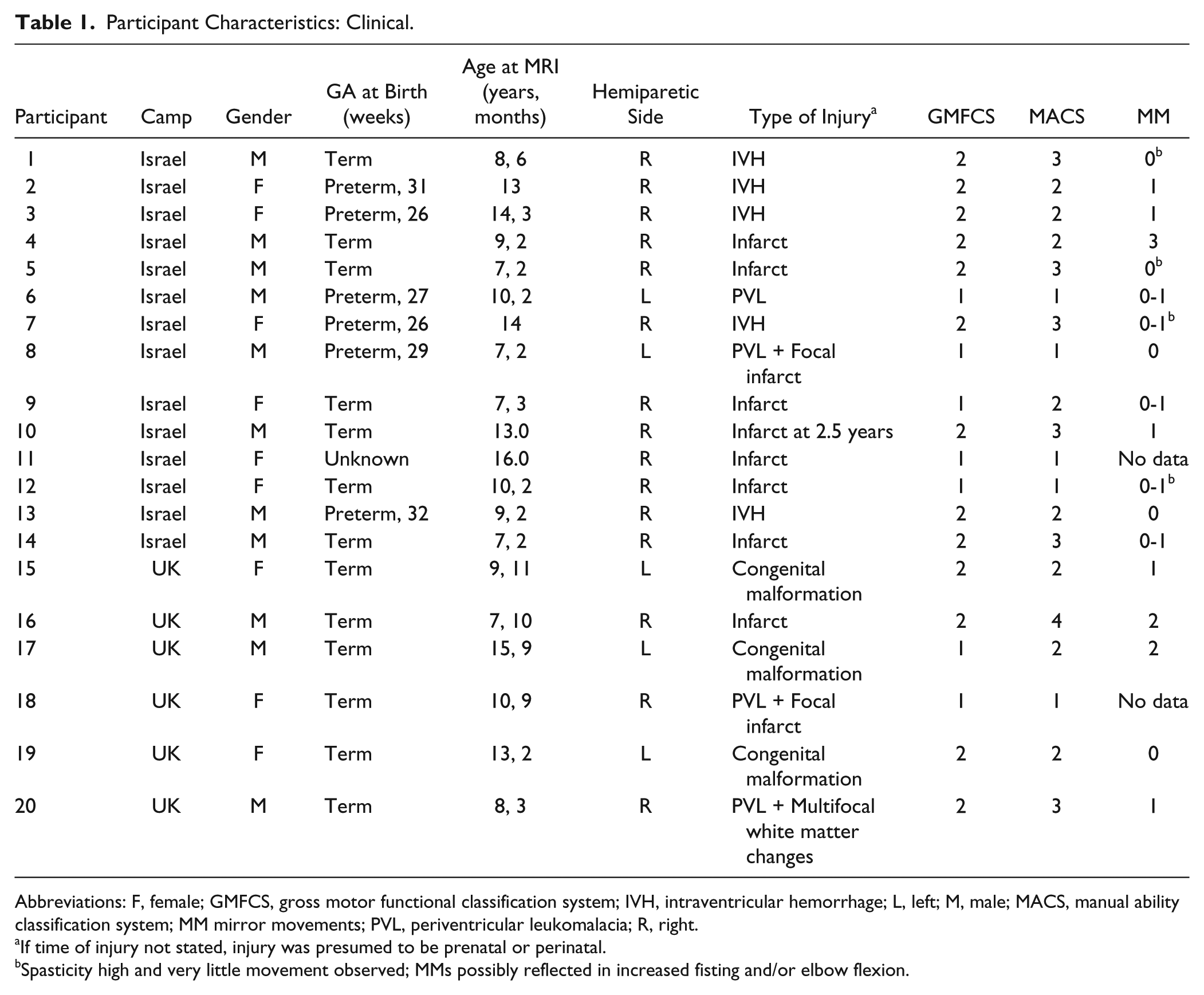
Abbreviations: F, female; GMFCS, gross motor functional classification system; IVH, intraventricular hemorrhage; L, left; M, male; MACS, manual ability classification system; MM mirror movements; PVL, periventricular leukomalacia; R, right.
If time of injury not stated, injury was presumed to be prenatal or perinatal.
Spasticity high and very little movement observed; MMs possibly reflected in increased fisting and/or elbow flexion.
Inclusion/Exclusion Criteria
Children were invited if they received age appropriate educational services and showed clinical signs of predominant spastic unilateral motor impairment caused by UCP. Based on clinical history and subsequent radiology, in the majority of cases, injury had occurred in utero or during the perinatal period; with 1 child the infarct occurred at age 2.5 years, and in 1 case the time of injury was unknown. Exclusion criteria were overt seizure activity and/or administration of motor therapy/medical/surgical treatment aimed at improving upper-extremity movements (eg, botulinum toxin injections or intensive motor learning intervention) in the previous 6 months and any contraindications to MRI. There was 1 child in the UK cohort who had participated in a 2-week intensive motor learning intervention 2 years previously (participant 19). Notably, children with severe movement limitations to active range of movement of wrist extension (<20°) or fingers (<10° flexion) or limited active grasp capacity of the affected hand were included.
Hand function and imaging assessments were undertaken in the week prior to participation in the camp (baseline-T1), in the week after the camp (postintervention, T2), and 8 to 10 weeks following the intervention (T3). The results presented here refer to baseline imaging and change in hand function assessments over the 3 time points.
Assessments of Hand Function
The Assisting Hand Assessment (AHA; version 4.3), a standardized test of spontaneous use and performance of the affected hand across 22 predefined items, uses a 4-point rating scale, during bimanual interactions in functional/play-based tasks 37 and not bimanual function per se. The AHA is scored from video recordings, with good reliability and validity reported. 13 Raw scores are transformed into logits via Rasch analysis and converted to a 0 to 100 AHA unit scale, with higher scores representing better bimanual skills. 38 The AHA unit scale is used here as recommended by the authors of the AHA. 39 Evaluations from video were made by a trained therapist blinded to treatment status and the other test results.
Unimanual skill was measured via the Jebsen Taylor Test of Hand Function (JTTHF), 40 a standardized test of speed of unimanual grasp and release (modified by eliminating the writing task because of difficulties of scoring legibility in children), with reliability and normative data reported for children. 41 Maximum time allowable to complete each task successfully was capped at 3 minutes; thus, the maximum time for all 6 items for each hand was 1080 seconds. Scores were obtained for the affected and less-affected hands.
The Children’s Hand Experience Questionnaire (CHEQ), a 29-item questionnaire, was administered to record the independent performance and skilled use of the affected/hemiplegic hand in daily bimanual activities. 42 Incorporating the CHEQ also allowed us to link outcomes to broader aspects of independence and participation of the ICF, reported as important to children and families.42,43 Children 13 years and older, or parents, completed the English version if they were fluent in English or the Hebrew translation. Scores are provided for the total number of independent activities and the number of independent activities performed using 2 hands. The percentage of independent activities performed bimanually was calculated (CHEQ percentage use).
Mirror movements (MMs) were assessed using the Woods and Teuber Scale 44 to obtain estimated measures of presence/extent of MMs (0 = no clear imitative movement to 4 = movement equal to that expected for the intended hand). Ratings were made from videos of the motor task in the MRI (Israeli group) or estimated from simultaneous movement actions recorded by sphygmometers placed in both hands during the fMRI tasks (UK group). Ratings corresponding to the Woods and Teuber Scale were calculated, in which 0% to 5% change in pressure (squeezing) from the resting state indicated no clear imitative movement (score = 0) and >5% to 25% (score = 1), >25% to 50% (score = 2), and >50% to 75% (score = 3) to changes consistent with those recorded in the “active” hand (score = 4).
Imaging Assessments
MRI scans were performed at baseline on a 3-T gradient echo (GE) according to our previously published protocol. 22 Structural images included sagittal T1-weighted spin echo sequence; T2-weighted fast spin echo; FLAIR; high-resolution anatomical 3D fast spoiled gradient echo sequence, and DTI, which was acquired along 19 diffusion gradient directions for the Israeli group and 16 diffusion directions for the UK group (b = 1000 s/mm2), and one was acquired without an applied diffusion gradient. To control for possible effect of different DTI parameters (number of diffusion gradients), we performed a t test between the DTI parameters of the CC, posterior limb of the internal capsule (PLIC), and CST between the UK and Israeli groups. No significant differences in DTI values were found. The 2 MR scanners used at the 2 sites (Israel and UK) were both GE scanners. An identical detailed protocol was used at both locations to ensure that the MRI parameters of the scans were identical.
A pediatric radiologist, blinded to medical history and hand function assessments, scored conventional MRI using a quantitative scoring method for brain injury based on topographical MR imaging patterns of the brain. The score, which ranges from 0 for a normal scan to a maximum of 18 for each hemisphere, covers 7 aspects across 4 domains: (1) number of affected lobes, (2) volume and type of white matter injury, (3) extent of gray matter damage, and (4) major white matter tract injury, per our published protocol. This scoring will be referred to as the radiological score and is consistent with classification recommended by the Surveillance of Cerebral Palsy Europe (SCPE) consensus. 9 Scores have been demonstrated to correlate with clinical function. 45
fMRI was performed with T2*-weighted GE-echo planar imaging sequence: slice thickness/gap = 3.5/0.3 mm; field of view/matrix = 240 mm/128 × 128; repetition time/echo time/flip angle = 2250/29 ms/79°. The fMRI motor task was a block design involving clenching and extending all fingers of 1 hand in synchrony with 2-Hz paced tones, alternating between affected and less-affected hands.46-48 Children were asked to do their best to move only 1 hand without moving the opposite hand. Range of movement was limited by an inflated sphygmometer (UK) or a soft sponge ball (Israel) placed in children’s hands, depending on the research site. This allowed for movement with minimal effort but controlling/limiting large variations in range of movement.
The choice of fMRI task was intentionally kept simple with auditory cues to minimize difficulties of analyses from more complex motor planning tasks or motion artifacts caused by eye movements, respectively. Although generalized activation patterns through attentional networks may influence results, we aimed to minimize the impact of these by replication of the tasks in the active and passive conditions.
fMRI analysis was performed using BrainVoyager QX2 software package (http://www.brainvoyager.com). Preprocessing and fMRI analysis were previously described. 22 Two contrasts were studied: (1) activation when moving the affected hand versus baseline activation and (2) activation when moving the less-affected hand versus baseline activation. A false discovery rate procedure was used for the selection of thresholds, and the false discovery rate (q value) of 0.005 was chosen. The number of voxels in sensorimotor areas around the central sulcus within left and right hemispheres in each condition was quantified separately. The sensorimotor activations that were measured consisted of the numbers of voxels activated around the central sulcus, which anatomically consist of primary motor and sensory areas. This definition was intentionally broad to account for possible brain plasticity, including significant shifts in brain structures that have been shown to occur following brain injury early in life. Because of the variability in the brains caused by the lesions, we did not coregister the brains to a standard space; therefore, we did not use a predefined region of interest (ROI) but rather manually traced the ROI on each individual brain.
A laterality index (LI)20,49 was determined for each participant to take into account relative contralateral and ipsilateral activation when moving 1 hand. The LI was calculated according to the formula LI = (Contralateral − Ipsilateral)/(Contralateral + Ipsilateral), where contralateral and ipsilateral equal the total number of voxels activated above threshold in areas around the central sulcus contralateral or ipsilateral to the moving hand. LI values closer to 1 indicate a more typical unilateral pattern of activation; values closer to 0 indicate a more bilateral pattern of activation; and values closer to −1 indicate more atypical ipsilateral activation.
DTI analysis was performed using DTIStudio software (Johns Hopkins University, Baltimore, MD). Preprocessing of diffusion data and DTI tractography analysis were previously described. 22 Briefly, the CC and CST were reconstructed with streamline fiber tracking using the Fiber Assignment by Continuous Tracking (FACT) algorithm. 50 The CC was further segmented into the genu, midbody, and splenium based on an adaptation of the Witelson segmentation scheme. 51 Because of the relatively small sample size and the large brain lesions in our cohort, we combined areas of the original 7 subdivisions of the Witelson protocol to create 3 simplified regions as previously performed by Wilde et al. 52 Based on findings from our previous study, 22 we excluded the genu from further analysis. ROI analysis was performed for the left and right PLIC using ROIEditor software (Johns Hopkins University, Baltimore, MD). Mean values of axial diffusivity (Da), radial diffusivity (Dr), mean diffusivity (MD), and FA were calculated from these fiber tracts and ROIs. Higher Da, Dr, and MD and lower FA values reflect reduced tract integrity and, thus, more abnormality or damage.
The PLIC region is part of the CST fiber tract. However, when performing tractography, we measured the diffusivity indices within the entire CST and obtained mean values across the entire tract. Because this may have masked differences in specific areas within the CST and because the PLIC is in an area known to be sensitive for detection of injury within the CST,53,54 we also included it as a separate ROI in this study.
Intervention Procedures
Children attended one of three 2-week camps, 6 hours daily for 10 days over 2 weeks during the summer holidays. Additional homework tasks were set and reviewed each morning with practice and demonstration of previously learnt activities.
The camp program followed the procedures summarized by Charles and Gordon, 55 using the HABIT principles to promote intensive practice and repetition in part and whole task movements. A magical theme was incorporated into the program such that children learned specifically selected/modified magic hand tricks in collaboration with professional magicians and prepared costumes and props for a magic show performed at the end of the camp. Details of the protocol are outlined in Green et al. 14 Magic tricks were scaled to challenge increasingly complex bimanual skills either in timing, accuracy, or fine manipulation. Staffing numbers were equitable across camps (3 staff; 4 participants) and consisted of undergraduate and graduate students of physiotherapy and occupational therapy.
Statistical Analysis
This analysis sought to determine the relationship between the degree of improvement (Δ) on hand function measures postintervention at time points T2 and T3 and imaging characteristics at baseline. Improvement was calculated as the percentage of change between precamp and postcamp (T1 − T2) and precamp and follow-up (T1 − T3) to enable similar comparisons across hand function measures. In cases where prescores on the CHEQ were 0 (eg, no activity performed independently), these were transformed to 0.5, and 0.5 accordingly added to postscores to enable a percentage change score to be calculated. For ease of comprehension, percentage increase (performance scores) was used for AHA and CHEQ and percentage decrease (time) for JTTHF, so that all Δs represented improvement. CHEQ analyses were based on CHEQ percentage use, with Δs calculated as the difference between scores, which were already in percentage form.
Pearson’s correlation was assessed between Δ on hand function measures and the following baseline imaging variables: radiological score, fMRI (number of voxels in ipsilateral and contralateral hemispheres when moving the affected and less-affected hands), and DTI (FA, MD, Da, and Dr) in the midbody and splenium of the CC and in the affected and less-affected PLIC and distal CST. In view of the mix of ordinal and interval scales, the Spearman ρ was calculated with similar results, and therefore, Pearson correlations are reported. Because of differences in measurement of MM, correlation analyses of fMRI results were run with and without children identified with MM rather than running partial correlations.
Statistical analysis was performed using SPSS software (SPSS 17.0, Chicago, IL). The study was approved by the institutional review board of Tel Aviv Sourasky Medical Center (0204-10-TLV and 0239-11-TLV) and the UK National Research Ethics Committee (H-10/H080/40/AM01), conforming to the Helsinki Declaration of research sites, and fully informed consent was obtained from parents and assent from children.
Results
Whereas 28 children participated in the 3 camps, imaging data were available for 20 children with UCP (11 boys; mean age = 10 years, 11 months ± 1 year, 9 months; range = 7 years, 9 months to 16 years, 3 months) with Manual Ability Classification System level I to III (see Table 1). Imaging data were not obtained for 4 children: 1 who had dental braces at the time of imaging; 1 who had a peritoneal shunt and, therefore, did not go in the 3T scanner; and 2 who were unavailable for logistical purposes. Unclear scanning sequences were obtained in 4 cases because of excess movements; therefore, these children were excluded, leaving 20 children with usable imaging data.
Of the 20 children, all with clinical diagnosis of UCP, 2 children were found to have some form of bilateral brain injury on structural MR imaging. Child 7 had both clinical and imaging findings involving both lower extremities and the right upper extremity. Child 20 had bilateral damage across nonmotor areas of the frontal lobe without clinical indication of involvement of the less-affected hand.
All 3 children (4, 16, and 17) with significant MM had evidence of bilateral activation on fMRI in the sensorimotor areas. However, there were 3 additional children with LI, suggesting bilateral activation, of whom 2 (6, 19) did not have significant MM and 1 (18) had no MM data available. Further details regarding bilateral activation are noted in Table 2.
Participant Characteristics: Imaging. a
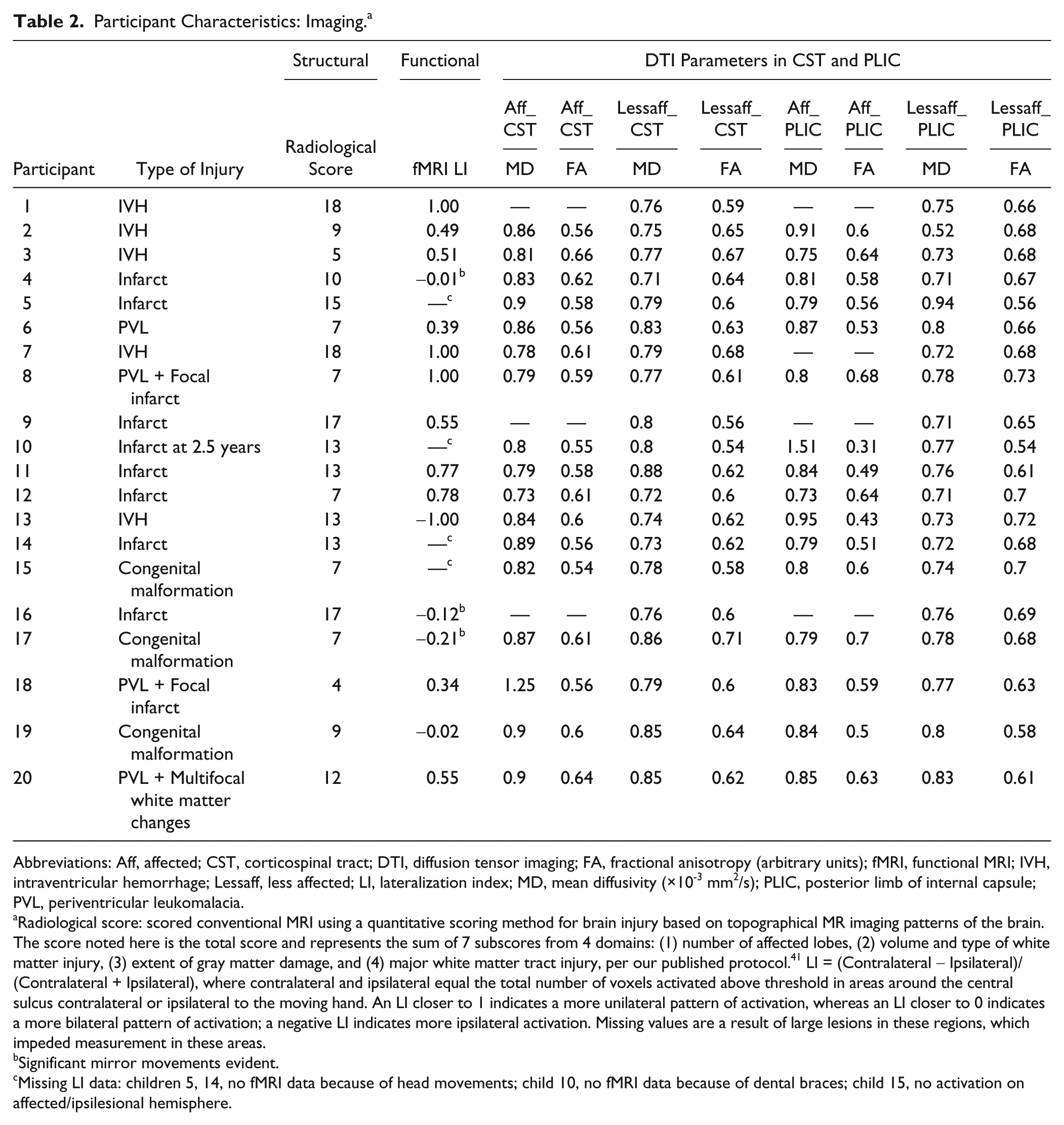
Abbreviations: Aff, affected; CST, corticospinal tract; DTI, diffusion tensor imaging; FA, fractional anisotropy (arbitrary units); fMRI, functional MRI; IVH, intraventricular hemorrhage; Lessaff, less affected; LI, lateralization index; MD, mean diffusivity (×10-3 mm2/s); PLIC, posterior limb of internal capsule; PVL, periventricular leukomalacia.
Radiological score: scored conventional MRI using a quantitative scoring method for brain injury based on topographical MR imaging patterns of the brain. The score noted here is the total score and represents the sum of 7 subscores from 4 domains: (1) number of affected lobes, (2) volume and type of white matter injury, (3) extent of gray matter damage, and (4) major white matter tract injury, per our published protocol. 41 LI = (Contralateral − Ipsilateral)/(Contralateral + Ipsilateral), where contralateral and ipsilateral equal the total number of voxels activated above threshold in areas around the central sulcus contralateral or ipsilateral to the moving hand. An LI closer to 1 indicates a more unilateral pattern of activation, whereas an LI closer to 0 indicates a more bilateral pattern of activation; a negative LI indicates more ipsilateral activation. Missing values are a result of large lesions in these regions, which impeded measurement in these areas.
Significant mirror movements evident.
Missing LI data: children 5, 14, no fMRI data because of head movements; child 10, no fMRI data because of dental braces; child 15, no activation on affected/ipsilesional hemisphere.
Hand Function Measures
Tables 3 and 4 provide details of individual and group data changes across time points. It should be noted that our cohort contains data from 7 children that were reported on previously. 14 Analysis of the data shows that as a group, children made overall progress immediately after the intervention for all measures used: bimanual (AHA), F(2, 36) = 3.58, P = .04, η2 = 0.166; bimanual independence (CHEQ), percentage use, F(1.50, 24.00) = 14.96, P < .001, η2 = 0.483; and unimanual (JTTHF), F(1.27, 22.89) = 11.43, P = .001, η2 = 0.388. Improvement was maintained at follow-up except for performance on the AHA (Table 3). Considerable interindividual variation was noted between the children along the different time points as well as on the different measures (unimanual vs bimanual). Given the significant change criteria of >4 logits on the AHA or >20% on the JTTHF, observation of Table 3 shows that one group, consisting of 8 children (2, 3, 6, 8, 11, 12, 13, and 19), made progress on both the AHA and the JTTHF after the intervention, though only 3 of these maintained that improvement at follow-up. A second group, consisting of 11 children (1, 4, 5, 7, 10, 14, 15, 16, 17, 18, and 20), made progress on either AHA or JTTHF after the intervention, though this included 3 (4, 17, and 20) who actually showed poorer scores on the AHA. A third group, consisted of 1 child (9), who showed no progress on either the AHA or JTTHF but did show improvement on the CHEQ.
Motor Function Data at 3 Time Points Over the Study Period.
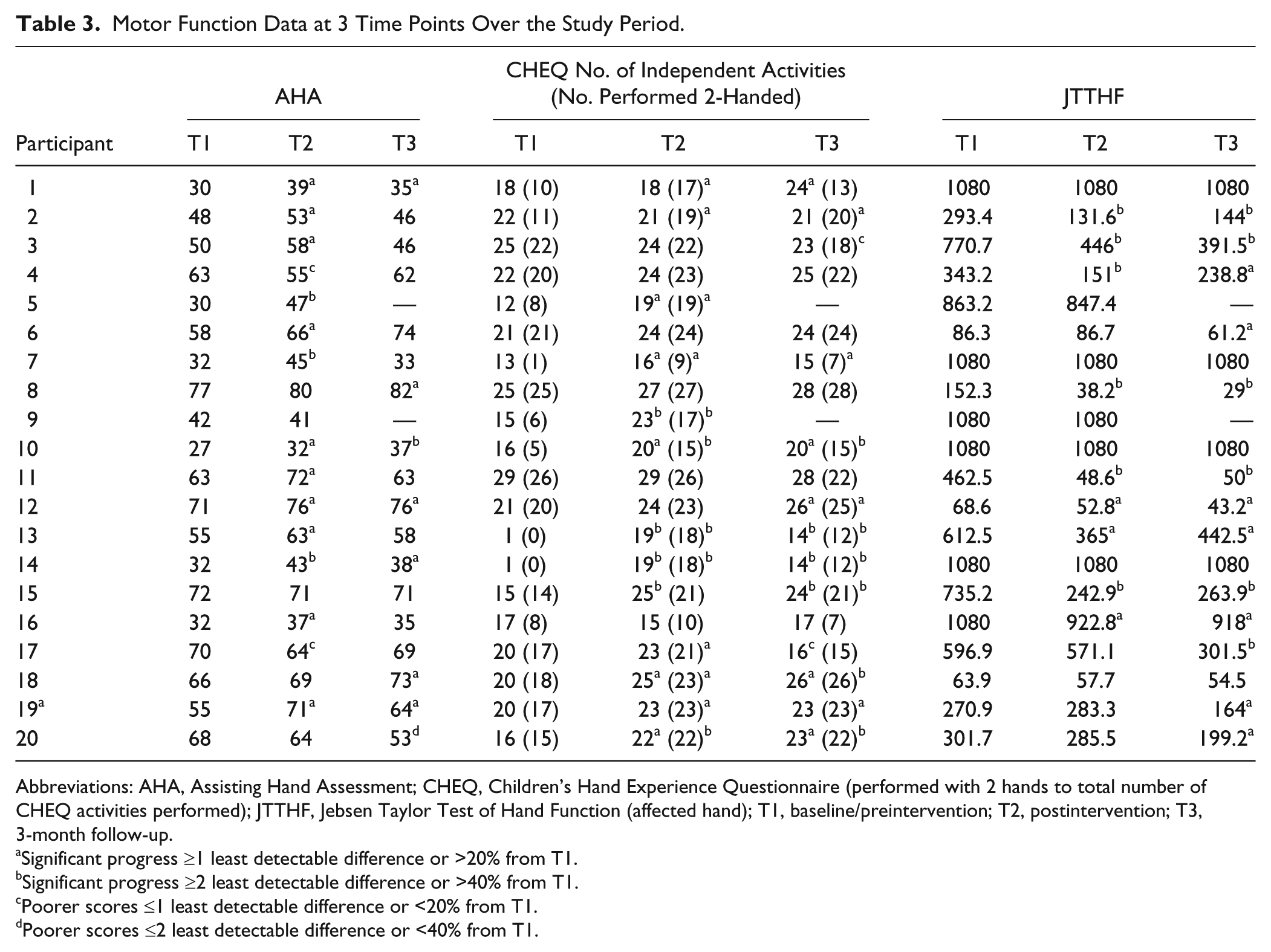
Abbreviations: AHA, Assisting Hand Assessment; CHEQ, Children’s Hand Experience Questionnaire (performed with 2 hands to total number of CHEQ activities performed); JTTHF, Jebsen Taylor Test of Hand Function (affected hand); T1, baseline/preintervention; T2, postintervention; T3, 3-month follow-up.
Significant progress ≥1 least detectable difference or >20% from T1.
Significant progress ≥2 least detectable difference or >40% from T1.
Poorer scores ≤1 least detectable difference or <20% from T1.
Poorer scores ≤2 least detectable difference or <40% from T1.
Performance on Each Outcome Measure at Precamp, Postcamp, and 3-MFU.
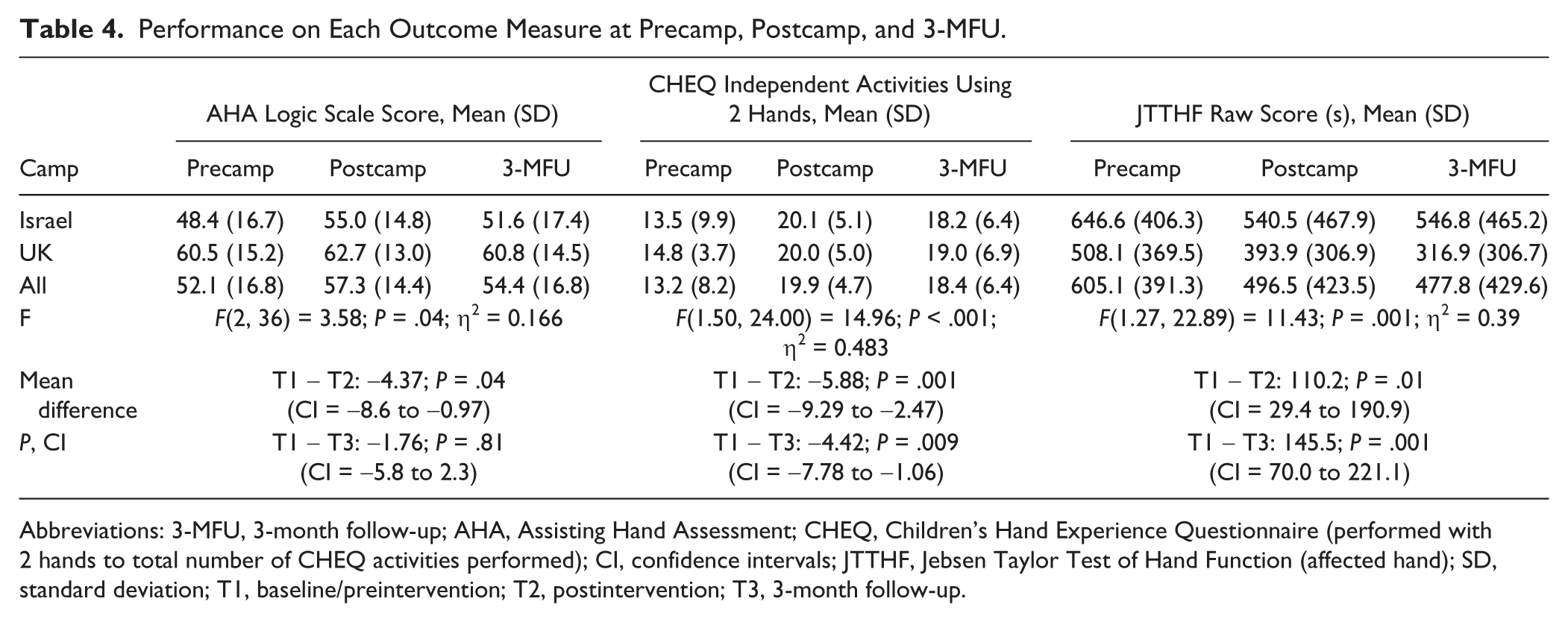
Abbreviations: 3-MFU, 3-month follow-up; AHA, Assisting Hand Assessment; CHEQ, Children’s Hand Experience Questionnaire (performed with 2 hands to total number of CHEQ activities performed); CI, confidence intervals; JTTHF, Jebsen Taylor Test of Hand Function (affected hand); SD, standard deviation; T1, baseline/preintervention; T2, postintervention; T3, 3-month follow-up.
Radiological Score
Figures 1A to 1C show the correlations between radiological score at T1 (preintervention) and changes on hand function measures at T2 and T3. Radiological score positively correlated with improvement on the AHA (r = 0.475; P = .034) and CHEQ percentage use (r = 0.632; P = .003) at T2 (ie, immediate posttest) but negatively correlated with improvement on the JTTHF (affected hand: r = −0.514; P = .024) at T3 (ie, 3-month follow-up). No relationship was found for the AHA or CHEQ at T3.
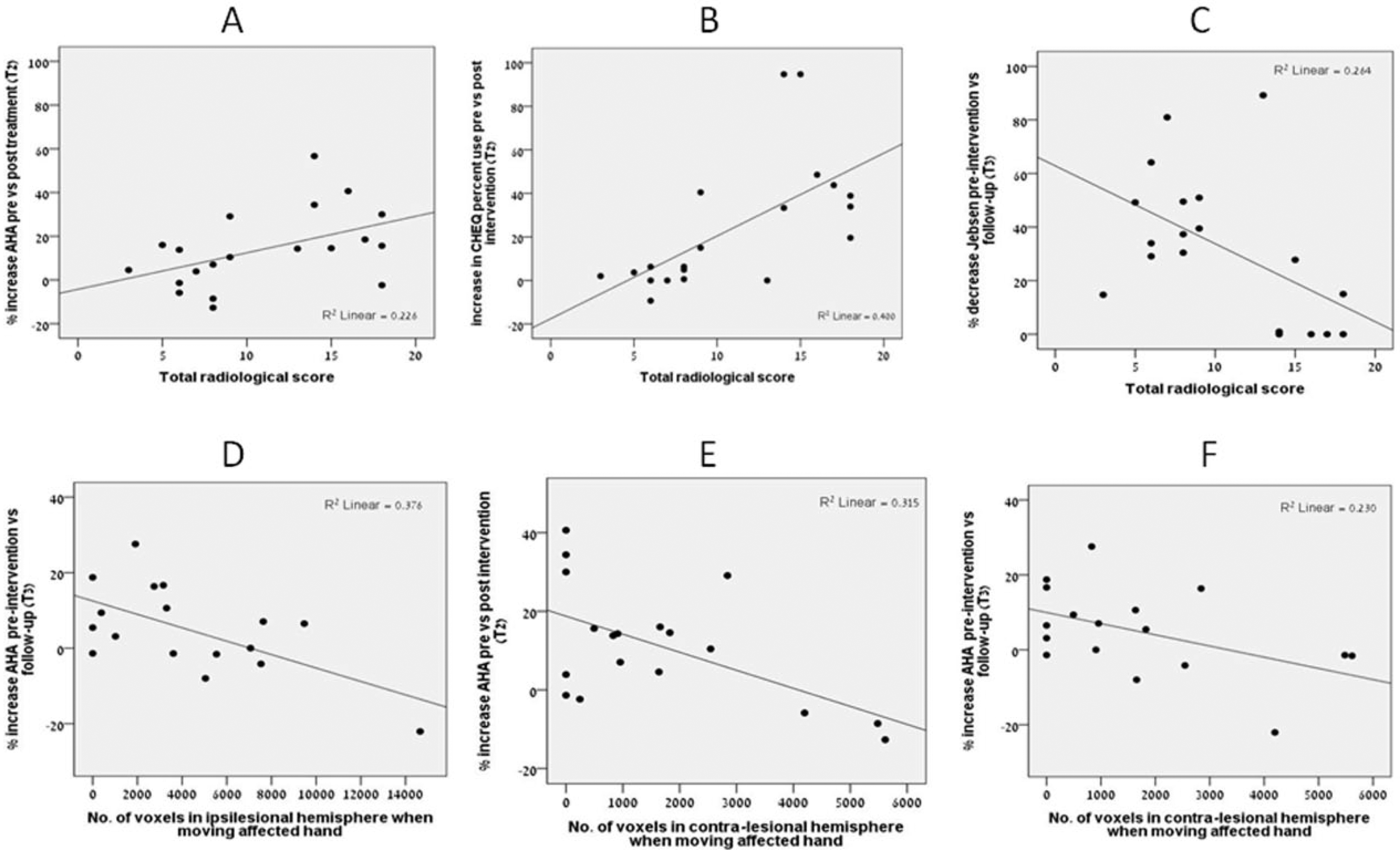
Scatterplots showing correlations between baseline radiological score and motor improvement postintervention on the (A) AHA, (B) CHEQ at T2 and (C) JTTHF at T3; and between fMRI parameters in the ipsilesional (D) and contralesional (E, F) hemispheres and motor improvement on the AHA.
fMRI Measures
Figures 1D to 1F show the significant relationships between baseline fMRI measures and changes on hand function measures. A negative correlation was noted between activation of the ipsilesional hemisphere when moving the affected hand and progress on the AHA at T3 (r = −.613, P = .009; Figure 1D) but not at T2. A negative correlation was also noted between activation of the contralesional hemisphere when moving the affected hand and progress on the AHA at T2 (r = −.562, P = .015; Figure 1E) and at T3 (r = −.479, P = .052; Figure 1F). We note that there was an outlier on the AHA outcomes (Table 3, participant 20) at T3; removing this score from the analysis resulted in a reduction in the significance between the activation of the ipsilesional hemisphere and progress on the AHA (r = 0.361; P = .17) but did not affect the significance of other findings. No relationships were found for the CHEQ or JTTHF at either T2 or T3.
In a post hoc analysis, we checked the extent to which atypical activation of the ipsilateral (contralesional) hemisphere when moving the affected hand may have been a result of MM. As noted earlier, our cohort included 3 children with significant MM (see Table 1; children 4, 16, and 17). One of these children was seen to be a significant outlier for activation of the ipsilesional hemisphere and was visibly seen to have significant and functionally impairing MM of both hands. When running the correlations without these 3 children, correlations between ipsilesional activation and AHA were no longer significant, whereas the correlation between activation in the contralesional hemisphere and AHA at T2 maintained the same trend (r = −.482; P = .069). Of interest, correlations emerged between the number of voxels in the ipsilesional hemisphere during movement of the affected hand and progress in unimanual function on the JTTHF, both postintervention and at follow-up (T2: r = 0.54, P = .04; T3: r = 0.63, P = .02, respectively).
Diffusion Tensor Imaging
Figure 2 shows the significant correlations between changes on hand function measures and the FA values in the affected and less-affected CST and in the PLIC. DTI measures of the CC did not correlate with change on the AHA, CHEQ, or JTTHF at T2 or T3. DTI measures of the CST were mixed with no correlation for any of the hand function assessments at T2. However, at T3, FA in both the affected and less-affected CST demonstrated a negative correlation with progress on the AHA (affected: r = −0.594, P = .015; less affected: r = −0.497, P = .036; Figures 2A and 2B). DTI measures of the PLIC showed correlations with outcome measures at both T2 and T3. At T2, FA in the affected PLIC correlated negatively with progress on CHEQ percentage use (r = −0.547, P = .038; Figure 2C). At T3, FA in the affected PLIC correlated negatively with improvement on the AHA (r = −0.656, P = .008; Figure 2D).
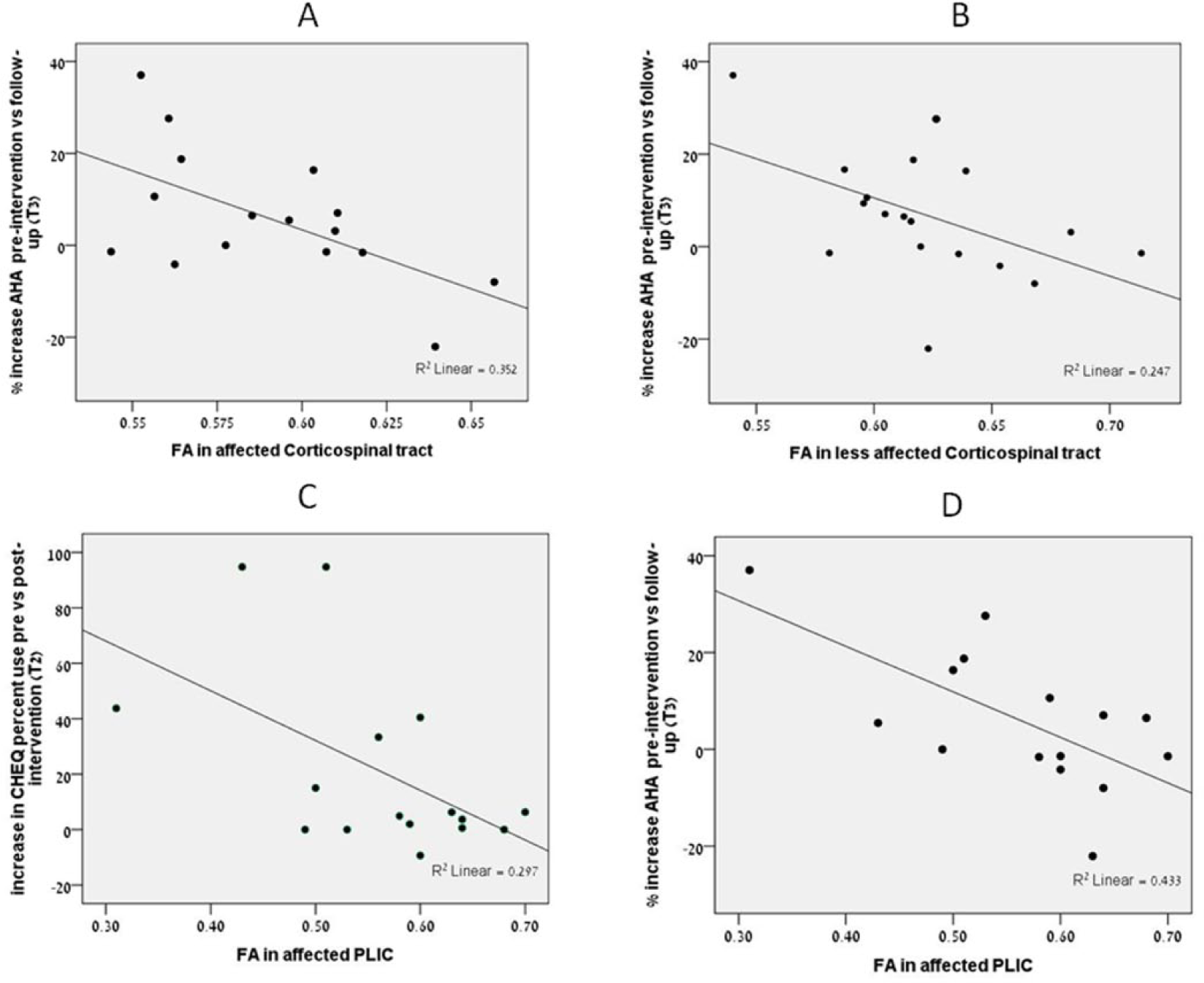
Scatterplots showing correlations between baseline DTI parameters in the CST (A, B) and the PLIC (C, D) and motor improvement postintervention.
Discussion
In this study, we examined the ability of MR and DTI characteristics of brain injury in children with UCP to predict hand function following a bimanual intervention. Our findings revealed a varying set of relationships between the imaging findings and change in hand function. Most clear was the finding that children with greater overall severity of brain injury at baseline, as indicated by higher radiological score and decreased cortical activation of the affected hemisphere on fMRI, showed greater improvement on bilateral hand function but less improvement on unimanual function. Consistent with this was the finding that DTI values in the PLIC and CST, indicating more white matter damage (both to the affected and less-affected sides), were also associated with greater improvement on bimanual function but no improvement on unimanual assessment. These data point to the overall consistent finding that children with UCP with greater overall brain damage made greater progress on bimanual function following a bimanual intervention. Given that degree of brain damage correlates with the severity of hand function impairment, 45 we may conclude that children with UCP with greater severity of motor impairment may make greater progress on bimanual function following a bimanual intervention. Collinearity between measures of unimanual and bimanual function prevents a more definitive statement on this latter point or comparisons between unimanual or bimanual intervention. Although a number of individual studies have attempted to determine best responders within their limited sample, it remains largely unknown who is likely to benefit. 29 We explored the extent to which the relative lack of improvement of children with less-severe motor impairments may have been the result of a ceiling effect. We did not identify any child who did not have further capacity, of at least 1 least-detectable difference (beyond the standard error of measurement), for change across measures following intervention.
Despite the above initial conclusions, our results also contained data suggesting that other elements are at play in the equation of which children with UCP make progress after intervention. We identified a recurring theme suggesting that severity of impairment and type of connectivity in both affected (ipsilesional) and less-affected (contralesional) hemispheres predicted degree of improvement after a bimanual intervention. These findings held true mostly regarding bimanual function, but there was some suggestion of an effect on unimanual function on the less-affected side. Arguably, some improvement in bimanual function (and independence) may in part be a result of improved skills of the less-affected hand.
We found on post hoc analysis that more typical activation of the ipsilesional, and not contralesional, hemisphere during movements of the affected hand corresponded to better progress in unimanual function when analyses excluded children with significant MM. This is consistent with our DTI results corresponding to the integrity of the CST and PLIC. We further noted that whereas all 3 children with significant MM had evidence of atypical bilateral activation, there were 2 other children with atypical bilateral activation who did not have significant MM and 1 for whom no MM data were available (see Table 2). These findings are consistent with the idea that bilateral activation may be a result of either ipsilateral reorganization and or presence of MM. 56 Clearly, further research is required, with more robust measures of MM, to determine the extent to which atypical contralesional activation is caused by MM and the impact this has on bimanual performance. Indeed, it is not possible to state at this stage whether children with MM may benefit more from a combined unimanual and bimanual therapy program. 57
We had 2 children who at presentation showed inconsistent findings between structural imaging and clinical data. One made good progress that was not sustained at follow-up. The other made no progress on bilateral function but good progress on unimanual function. Additionally, there were 3 children with significant MM who demonstrated worsening scores on the AHA together with improvement on unilateral function. What remains to be clarified is the functionality of these findings, including the impact on learning rates and maintenance of skills in the affected hand of children with unilateral motor impairments.
As noted earlier, studies examining imaging biomarkers as predictors for improvement in children with UCP who underwent intervention for the upper extremity are emerging in the literature.6,31,32,34,58 Our findings, suggesting that certain imaging biomarkers predict improvement in children with UCP, are in contrast to the studies noted earlier by Islam et al, 6 Rickards et al, 31 and Friel et al, 32 that did not identify such biomarkers. Our study is partially consistent with that of the study by Kuhnke et al, 30 in that atypical activation of the nonlesioned hemisphere when moving the affected hand corresponded to less progress regarding speed. This suggests that either ipsilateral reorganization and/or transcallosal inhibition may be linked to behavioral factors such as MMs and/or developmental disregard, subsequently impeding acquisition of bimanual function. However, it should be noted that fMRI findings (as used in our study) are less definitive for mapping the strength of corticospinal connectivity than those obtained using TMS. 18
Our findings are at odds with those of Rocca et al, 33 in that we found fiber integrity (FA) in the lesion area to be negatively associated with outcome after an intervention, although intervention type differed. Manning et al, 34 consistent with our findings, recently reported higher MD in the PLIC of the affected tract corresponding to prediction of better progress in unimanual skills, following a unimanual therapy, and a more unilateral (typical) activation pattern associated with greater improvements in clinical scores.
Our study adds to the previous literature, most notably by examining predictors of change after a bimanual intervention, which has received little attention. Only 1 study 32 used a bimanual intervention, and in contrast to that study, we identified some imaging biomarkers that predicted improvement. Previous studies enrolled children with less-severe hand function, whereas we included those with MACS levels I to III.
A number of our findings warrant further discussion. Our results showed that children with greater overall brain damage, based on a quantitative scoring method of MR images (radiological score), made the most progress on bimanual tasks. This is consistent with a report that examined best responders among children with UCP who received either a unimanual (CIMT) or a bimanual intervention and found that children with poorer hand function (suggesting greater brain pathology) made the most progress on unilateral tasks. 29 However, the question remains as to why this is so? Is this a case of regression to the mean, 59 whereby those children with more extreme values at baseline tend to be less extreme at follow-up? This appears less likely given that our findings were true across multiple measures and analyses. Alternatively, those with poorer hand function may have greater latent function to be activated/greater capacity for change once they undergo an effective intervention. Furthermore, our assessments of hand function focused predominantly on the activity level of the ICF-CY, whereas outcomes of importance to children and families are directed more to participation in community and social life, emotional well-being, and gaining independence. 43 Finding discrete indicators that may predict outcomes of intervention for these broader dimensions of disability in childhood is outside the scope of this study; however, the increase in independence documented on the CHEQ provides a promising indication of the impact of the therapy. Future studies should extend measures to capture outcomes of importance to children and families.
Of all the predictors in our study, those of the affected PLIC appear to be most consistent with previous literature. For example, a recent study of stroke patients proposed that FA in the PLIC may be a useful biomarker for prediction of upper-limb motor recovery. 53 This finding should be compared against the recent report by Marumoto et al, 54 who, in a small sample of stroke survivors with hemiplegia who underwent a unimanual intervention (CIMT), found the ratio of FA between the affected and less-affected PLIC to be positively correlated with motor improvement after intervention. This is also consistent with recent findings of more typical patterns of activation and more significant white matter immaturity of the PLIC corresponding with unimanual progress in children with UCP. 34 However, as noted earlier, others have found that white matter integrity was not associated with motor outcome or improvement after intervention, specifically relating to a unimanual intervention in CP and stroke patients.31,60 Given the heterogeneity between the studies, these conflicting findings await clarification by future studies.
We found that children with increased contralesional (atypical) activation, associated with active movements of the affected hand, made less progress on bimanual function, whereas those with increased contralesional activation during movements of the less-affected hand made more progress on unimanual function of the affected hand (eg, more typical activation of the hemispheres during movements of the affected or less-affected hand). This perhaps suggests that improvements in bimanual skills may partly be a result of improved skills of the less-affected hand. 61 Previous work has demonstrated that different types of reorganization, including intrahemispheric and interhemispheric connectivity, such as ipsilateral reorganization identified via TMS, may influence treatment response to unimanual interventions. 62 Lack of TMS data in our study preclude us from making definitive statements regarding CST reorganization but do suggest a complex interaction between lesion severity, reorganization, and brain activation patterns and unimanual and bimanual function. The issues yet to be unraveled include the functionality of bilateral brain activation and the relative impedance of ipsilateral reorganization or transcallosal inhibition in the acquisition of skills in the affected hand of children with unilateral motor impairments. It is evident that better means for categorizing neural pathology need to consider not only the etiology, location, and severity of brain lesions, but also patterns of functional connectivity and brain activation.
Limitations
Our study, like others in this field, contains limitations related to the small sample size and varied lesions causing UCP in the participants. Our finding of an outlier, possibly affecting results at time 3 but not consistent with other findings, is an example of where a large sample size might have clarified the meaning of this outlier. The development of hand function involves a complex interplay between motor, sensory, cognitive, and motivational factors, often presenting as coexisting conditions 63 that were beyond the scope of our study to explore. Missing also are data related to mastery motivation, persistence and resilience, and quality of life in these children, all of which may contribute to intervention response. Thus, our findings should be considered preliminary and hypothesis generating, leading to studies with larger samples and well-defined comparison groups. Despite the relatively small sample size, our study compares favorably to others6,31,32,34,58 in terms of methodological strength and is the first to report identification of imaging predictors of progress using a bimanual intervention.
We also note some limitations to the hand function assessments used in our study. There were 4 children who did not show progress on the JTTHF at all time points. Additionally, there was 1 child who did not show progress on the JTTHF but for whom data were only available at time points T1 and T2. This lack of progress may have been a result of the floor effect, with a maximum response time achieved if the child failed to perform all the tasks in the item (eg, turn all 5 cards over). However, some of these children may have managed to increase the number achieved but without obtaining an improved time.
Despite using the same scanners and identical protocols at the 2 sites, there may have been small differences in results between these sites because of software updates. Similarly, although MMs were addressed in this study, there is a report stating that by focusing attention, mirror activity can be voluntarily suppressed. 64 This might have led to an underestimation of the MM scoring and might have influenced the fMRI results. Similar to other pediatric imaging studies, the logistical and ethical issues regarding imaging and neurophysiology assessment need to be considered as a significant limiting factor in data acquisition. These issues include the young age of many of the children and frequent movement artifacts in imaging data.
Conclusions
In conclusion, we found that children with greater brain damage, as demonstrated on structural, functional, or DTI, showed enhanced responses to a bimanual intervention. Thus, baseline imaging may identify parameters that predict which children with UCP will make progress after a bimanual intervention. Defining reliable and homogeneous subgroups is essential to predict future clinical outcomes and best responders to treatment. Further studies with larger samples, with unified methods of classifying and quantifying neuropathology, are necessary to better delineate these findings and inform as to which interventions are most suited for each individual child.
Footnotes
Acknowledgements
We are extremely grateful to the children and their families, and magicians and young magicians, who gave so much of their time to participate in this study, as well as Geoff Charles-Edwards, Clinical Scientist Guy’s and St Thomas’ NHS Foundation Trust (GSTFT) and Gareth Barker of the Department of Neuroimaging, Kings College, London, for providing the data from the UK participants. We would also like to thank therapy students from Oxford Brookes University and Tel Aviv University, staff of Guys and St Thomas’ NHS Trust and the Tel Aviv Sourasky Medical Centre, and Becca Krom for their support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by grants from Guy’s and St Thomas’ Charity, Breathe Arts Health Research, and Marnie Kimelman Trust, and funding from ILAN, the Israeli Association for Disabled Children and Beit Issie Shapiro provided a venue and funding for one of the summer camps. DG was supported by a grant from the Department of Immigration and Absorption during 2010-2011.
