Abstract
Background
Temporal changes in the structural connectivity of major language tracts after stroke and their contribution to aphasia recovery are unclear.
Objective
To investigate longitudinal arcuate fasciculus (AF) integrity changes and their relationship with post-stroke aphasia recovery using diffusion tensor imaging (DTI).
Methods
Thirty-five patients with aphasia due to first-ever left hemispheric stroke underwent the Korean version of the Western Aphasia Battery and DTI at 1- and 6-month post stroke onset. Fractional anisotropy (FA), mean diffusivity (MD), radial diffusivity (RD), and axial diffusivity (AD) of both AF tracts were analyzed to evaluate the temporal changes in tract integrity and determine the correlation between changes (Δ; follow-up − initial) in DTI parameters and language scores.
Results
At 6 months post-stroke, the mean FA decreased, and mean MD and RD increased in both hemispheres; however, compared with mean AD observed after 1 month, the mean observed at 6 months increased only in the left hemisphere (P < .05). ΔFA of the left AF and proportional change in the aphasia quotient showed a significant positive correlation (r = 0.365, P = .031). No correlation was found between changes in the right AF parameters and language score. The group with increased FA in the left AF showed more significant language improvement than the group with decreased FA.
Conclusions:
During the subacute stage, the integrity of AF decreased in both hemispheres in patients with aphasia, and the change in structural connectivity of the left AF was associated with language improvement.
Introduction
Recently, there has been a growing interest in the role of white matter tracts that support functional interactions between cortical regions in language processing.1,2 In particular, advances in neuroimaging techniques, such as diffusion tensor imaging (DTI) and diffusion tensor tractography (DTT), have expanded our understanding of the role of fiber pathways in language function, beyond the contribution of cortical language areas. Several studies have demonstrated that structural connectivity damage impacts language function and its recovery.3 -6 The arcuate fasciculus (AF), a major language-associated white matter tract, is known for its traditional role of connecting and transferring information between cortical language areas and its involvement in repetition. 7 However, various recent studies have proposed a broader role for AF in spontaneous speech, comprehension, and naming.8 -11 Several studies have also suggested that the integrity of AF may play a role in aphasia recovery.12 -14
Until recently, only a few studies have used DTI to examine temporal changes in the structural integrity associated with language recovery. Additionally, most previous studies on AF included patients in the chronic stage,15 -18 and they had a relatively stable structural state. These studies have reported an increase in white matter integrity after successful treatment, such as that observed in the right AF after Melodic Intonation Therapy (MIT)15,16 or the left AF following constraint-induced language therapy 17 and anomia therapy. 18 Some studies have examined the structural changes of language-associated tracts from the acute to chronic stages,19,20 and both the left and right AF showed a trend of diminished fractional anisotropy (FA) with time. One of these studies reported an association between recovery of the damaged left AF and improvement of aphasia, 19 whereas another study revealed an association between the greater preservation of right AF and poorer naming recovery. 20 Another study on subacute stroke patients 21 found that naming recovery was associated with changes in the integrity of the right inferior longitudinal fasciculus (ILF); however, no significant correlation with changes in the left and right superior longitudinal fasciculus was found. Although the recovery mechanisms are different for different recovery stages after stroke, studies on the longitudinal left and right AF changes and their relationship with language recovery in subacute stroke patients are limited.
Thus, this study aimed to investigate the temporal changes in AF in both hemispheres in the subacute phase using DTI at 1 and 6 months after the onset of stroke. Further, we examined whether the structural changes in AF and recovery of post-stroke aphasia are linked. Based on previous findings, we hypothesized that the FA value of AF will decrease during the first 6 months post-stroke and that decrease will be associated with worse language outcomes.
Materials and Methods
Subjects
Participants were prospectively selected from a pool of patients admitted to the Department of Physical Medicine and Rehabilitation of Korea University Anam Hospital between June 2016 and December 2020 as per the following inclusion criteria: (1) age over 18 years; (2) Korean as their native language; (3) right-handedness; (4) first-ever left supratentorial stroke, confirmed by computed tomography or magnetic resonance imaging (MRI); (5) within 3 months post onset (the early subacute stage 22 ) at the time of initial assessment; and (6) presence of aphasia evaluated using the Korean version of the Western Aphasia Battery (K-WAB). The exclusion criteria were as follows: (1) bilateral cerebral hemispheric lesions and (2) known history of psychiatric disorders or hearing, cognitive, and language impairments.
Among the 501 stroke patients identified, 41 met our inclusion criteria. DTI data of these patients were processed and AF tractography was performed. However, six patients whose AF was not reconstructed either initially or at follow-up had to be excluded from the statistical analysis of DTI parameters of the AF. Finally, 35 patients with aphasia were recruited for this study with the following characteristics: sex (19 men, 16 women); mean age, (62.49 years [range, 21–88]); and initial aphasia quotient (AQ) (mean 50.70 points [range, 0–90.9]; Figure 1). Of the six excluded patients, five had middle cerebral artery infarction and one had parietal lobe hemorrhage. The mean age of the six excluded patients was 64.7 years (range, 35–78), and the initial AQ score was 33.08 points (range, 0.2–84.8), which did not yield a statistically significant difference when compared with that of the included patients (P = .189).
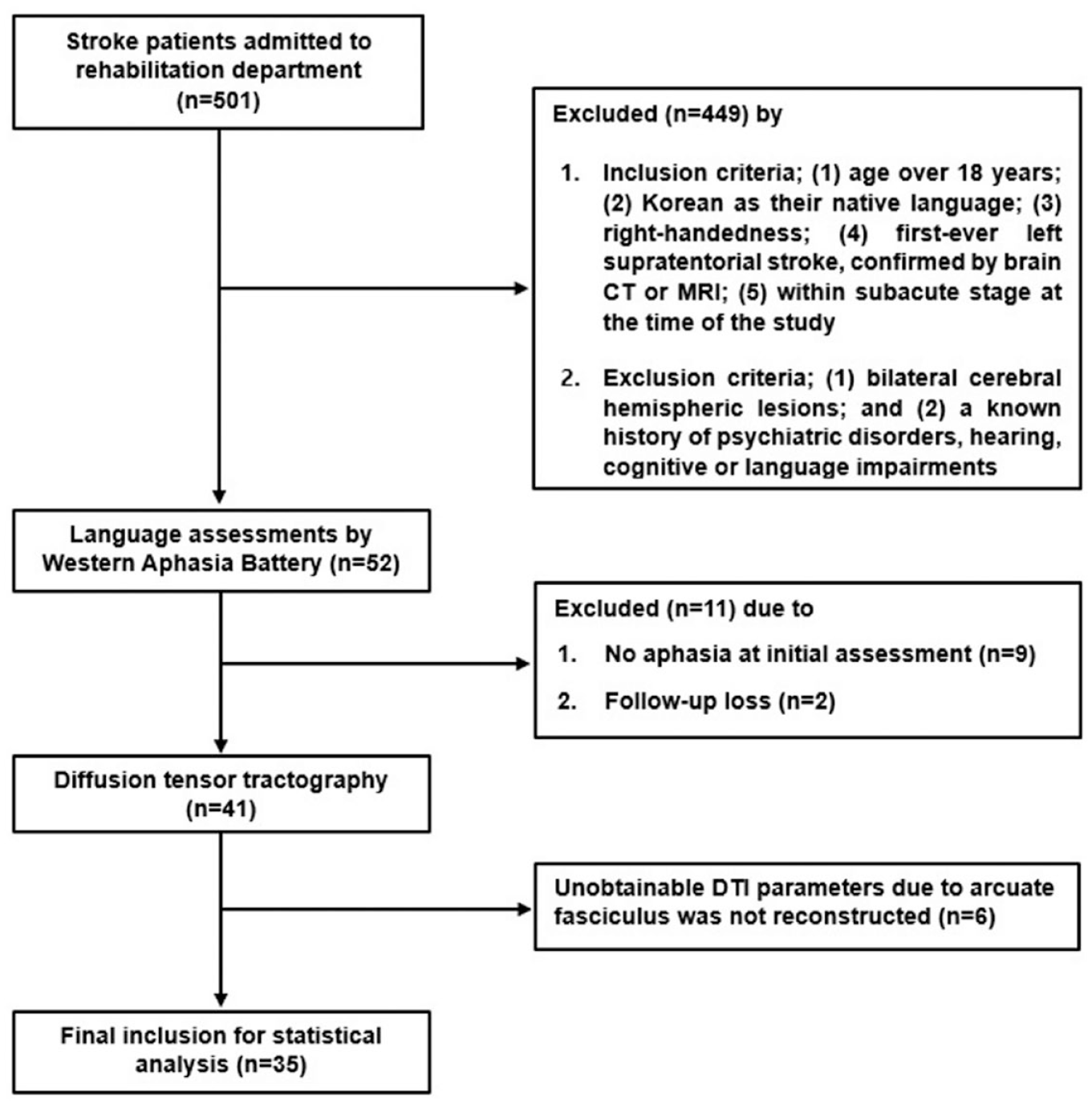
Flowchart of Participants Screening and Recruitment.
Written informed consent was obtained from all participants. This study was approved by the Institutional Review Board of Korea University Anam Hospital (no. 2017AN0005) and conducted in accordance with the Declaration of Helsinki.
Language Assessment
Patients underwent a comprehensive K-WAB assessment on admission (mean, 29.40 days) and at 6 months (mean, 181.77 days) after the onset of stroke. The K-WAB consists of subtests evaluating four oral language areas: spontaneous speech, auditory comprehension, repetition, and naming. The AQ score ranges from 0 to 100, which is twice the sum of the four subtest scores (fluency: 0–20, comprehension: 0–10, repetition: 0–10, and naming: 0–10). To diagnose aphasia, cut-off scores determined by age and education levels were used. 23 Aphasia recovery was calculated as a proportional change of AQ score (ΔAQp), defined as (AQ6mon–AQinitial)/(100–AQinitial).24,25 The proportional change rather than difference in two time points (ΔAQ; AQ6mon–AQinitial) was used to reduce the ceiling effect of initial AQ scores in patients with mild aphasia.
Magnetic Resonance Image Acquisition
MRI scanning, using a 3.0T Prisma MRI scanner (Siemens, Erlangen, Germany) with a 64-channel head coil, was performed during admission (mean, 36.06 days) and at 6 months (mean, 185.57 days) after stroke onset. DTI data were acquired using a single-shot spin-echo planar imaging pulse sequence. Image acquisition parameters were as follows: number of diffusion gradient directions = 64, matrix = 112 × 112, field of view = 224 × 224 mm2, slice thickness = 2.0 mm, voxel size = 2.0 × 2.0 × 2.0 mm3, echo time = 55 ms, repetition time = 6500 ms, b = 1000 s/mm2, and flip angle = 90°.
A T1-weighted image was acquired with the following parameters: repetition time (TR) = 2020 ms, echo time (TE) = 2.91 ms, flip angle = 9°, filed of view (FOV) = 217 × 166 mm2, and voxel size = 0.34 × 0.8 × 0.34 mm3. The T1-weighted images were coregistered to the DTIs using SPM12 toolbox (https://www.fil.ion.ucl.ac.uk/spm/software/spm12/) implemented in MATLAB (version 9.1.0; R2016b, The MathWorks, Inc., Natick, Massachusetts, United States) and used to display the AF tractography results.
Diffusion Tensor Tractography of the Arcuate Fasciculus
Pre-processing of the images and AF tractography was performed using DTI studio software (H. Jiang, S. Mori, Department of Radiology, Johns Hopkins University, Baltimore, Maryland, USA). 26 Head motion and eddy current were corrected using an automated image registration (AIR) program, and the diffusion directions were rotated during the AIR eddy current correction process.27,28 Significant motion artifacts were automatically checked by DTI Studio, but no images were excluded for severe head motion. To minimize head motion, the patient’s head was immobilized by a head strap with foam padding.
Lesion identification was conducted manually on the b = 0 image, first volume of DTI, using ITK-SNAP ver. 3.8.0., to identify the lesion location of stroke in each patient. 29 After identification, the lesion’s binary image was coregistered with the individual T1 image, and the coregistered images were spatially normalized to the standard MNI152 brain template. Image processing was conducted using the SPM12 toolbox. The lesion overlay map was calculated using an image calculation tool from SPM12 by summing every lesion’s binary image. The spatial relationship between the lesions and AF tract was visualized with the lesion frequency map and the standard AF tract mapped on the MNI152 brain template using MRIcroGL (https://www.mccauslandcenter.sc.edu/mricrogl). As the patients’ AF tracts were already damaged or lost owing to the lesion, a standardized AF image was used to accurately identify the spatial overlap between the lesions and the AF pathway. The standardized AF image was downloaded from NatBrainLab (https://www.natbrainlab.co.uk/atlas-maps) and overlaid on the lesion overlay map. 30
Fiber tracking was based on fiber assignment by a continuous tracking (FACT) algorithm, a commonly used deterministic post-processing algorithm for fiber tracking. 31 This algorithm computes fiber trajectories between regions of interest (ROIs) defined by the rater based on the largest (primary) eigenvector ε1 at each voxel. A two-ROI approach32,33 was used to reconstruct each fiber tract, and only fiber tracts passing through both ROIs were obtained (Figure 2 A, B). All ROIs were placed on the color FA map of each patient to identify the tract direction using three colors: green (anterior–posterior), blue (superior–inferior), and red (left–right). The first ROI was set on the anteroposteriorly oriented periventricular white matter shown in green on the color map on the coronal plane, running alongside the corticospinal tract (CST). The second ROI was placed to find the descending fibers of the AF connected to the fibers defined by the first ROI, and the ROI was drawn on the craniocaudally oriented fibers (shown in blue on the color map) passing through the periventricular fibers on the axial plane at the level of the anterior commissure. 34 All the fibers that were clearly not a part of the AF were removed (e.g., fibers running outside the brain or several branches running to the prefrontal cortex). The thresholds used for fiber tracking were FA > 0.15 and tract-turning angle <70°. When no fiber was reconstructed, we lowered the threshold of FA to 0.1 to ensure the absence of the tract. AF was reconstructed manually by expert researchers (YN and MC); the inter-rater reliability coefficients (Cronbach’s alpha) calculated for the left AF and right AF were 0.961 and 0.994, respectively.
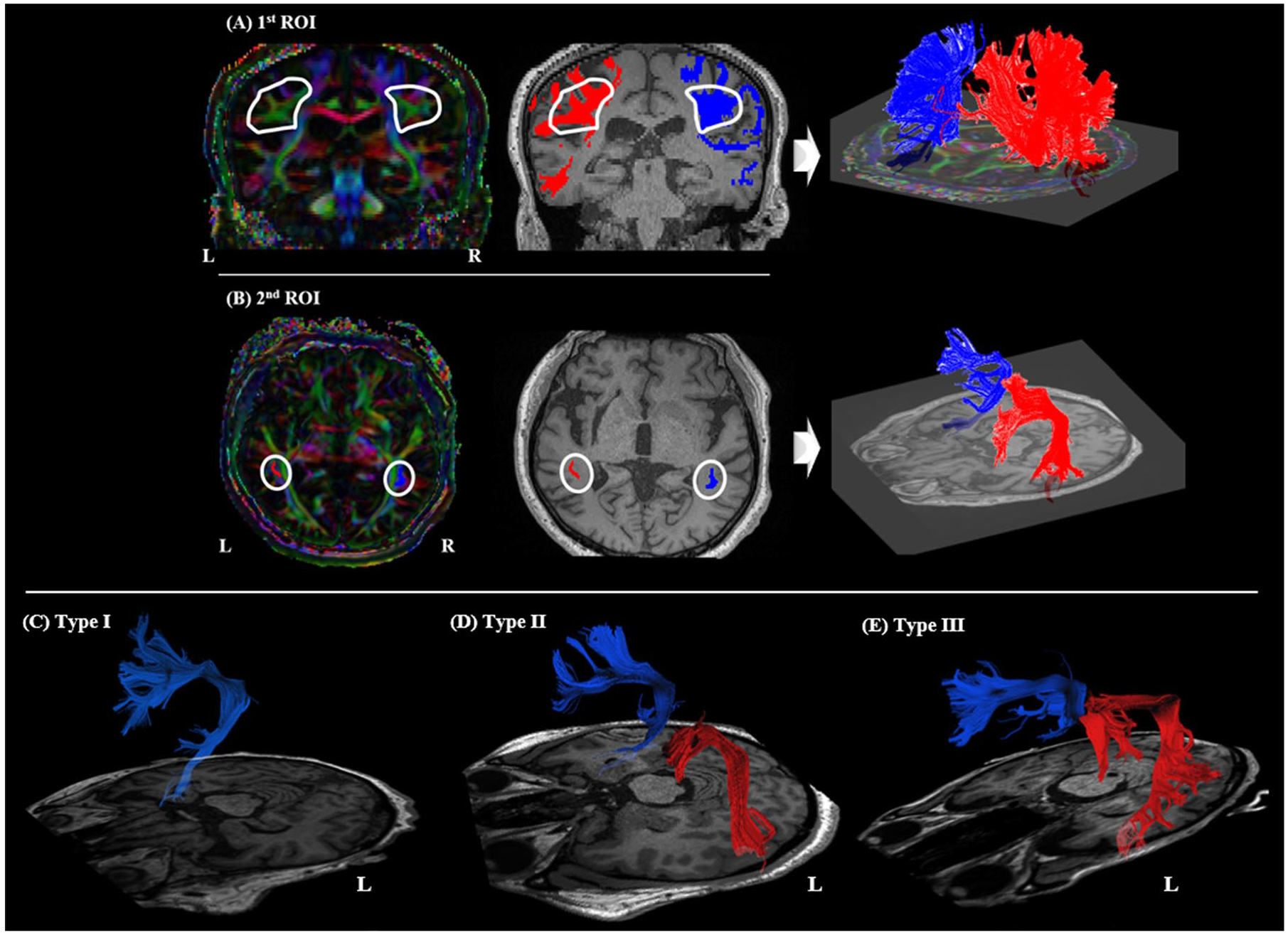
Region of interest (ROI) placement for arcuate fasciculus (AF) reconstruction and types of reconstructed AF. (A) The first and (B) the second ROI placements on the color map and fibers passing each ROI shown on the T1 coregistered image (blue, right AF; red, left AF). Three types of tractography (I-III) depending on the severity of left AF damage. (C) In Type I, fibers of the AF are not visualized; (D) in Type II, the fibers of AF are disrupted between Wernicke’s and Broca’s areas; (E) in Type III, the AF is preserved around the brain lesion. L, left; R, Right.
We classified the reconstructed left AF into three types based on its morphology and degree of damage 35 : Type I: lack of AF reconstruction; Type II: damage to fibers observed between Wernicke’s and Broca’s areas showing disruption, discontinuation, or shallowness; and Type III, AF preservation (Figure 2 (C)-(E)). DTI parameters, including FA, mean diffusivity (MD), radial diffusivity (RD), and axial diffusivity (AD), were measured across the entire identified AF tract; all parameters were separately measured in each hemisphere. The change in DTI parameters (Δ) was calculated for each value by subtracting the initial mean from the follow-up mean. As described earlier in the ‘Subjects’ section, six patients whose AF was not reconstructed (type I) were excluded from the study because the Δ values could not be calculated. In addition, we classified patients into two groups according to the direction of Δ FA values, negative (meaning longitudinal decrease; group 1) or positive (meaning longitudinal increase; group 2) to compare the amount of language function changes between the groups.
Statistical Analyses
Statistical analyses were performed using the IBM Statistical Package for Social Sciences (SPSS) version 24 (Armonk, NY: IBM Corp.). A paired-sample t-test was used to analyze changes in DTI parameters and K-WAB scores between the initial and follow-up assessments. Univariable regression analysis was used to identify the candidate factors related to aphasia recovery (ΔAQp). Variables with a P value < .8 were selected and used in the stepwise selection multivariable regression analysis. Only determinants with a significance level <.1 were included in the final model. Statistical significance was set at P < .05. Additionally, Pearson’s correlations were calculated to investigate the relationship between changes in DTI parameters of the left AF and changes in the K-WAB subtest scores. The p values calculated for Pearson’s correlations were corrected for multiple comparisons (p/4).
Results
Clinical Characteristics, Language Assessments, and DTI Parameters of AF
Among the 35 patients, 20 had cerebral infarctions. The mean lesion volume decreased longitudinally to 32163.97 mm3 from 34862.86 mm3, which was statistically insignificant (P = .459). The most overlapping lesions were found in the basal ganglia and inferior frontal gyrus, which interrupted the path of the AF, especially the anterior part (Supplementary Figure 1). Of the 35 patients, 27 (77.14%) still had aphasia at the 6-month follow-up assessment, whereas 8 showed full recovery. In DTT of AF, including six patients classified as type I at either time point, the initial types of left AF were as follows: type I, 4 (9.8%); type II, 10 (24.4%); and type III, 27 (65.8%). The types of AF at follow-up changed in seven patients (17.1%); five of them showed more disrupted morphology at follow-up, two showed change from type II to type I, and three from type III to type II (Supplementary Figure 2). The other two showed more improved morphology at follow-up, one showed change from type I to type III, and one from type II to type III (Table 1).
Demographic, Clinical, and Neuroanatomical Characteristics (n = 35).
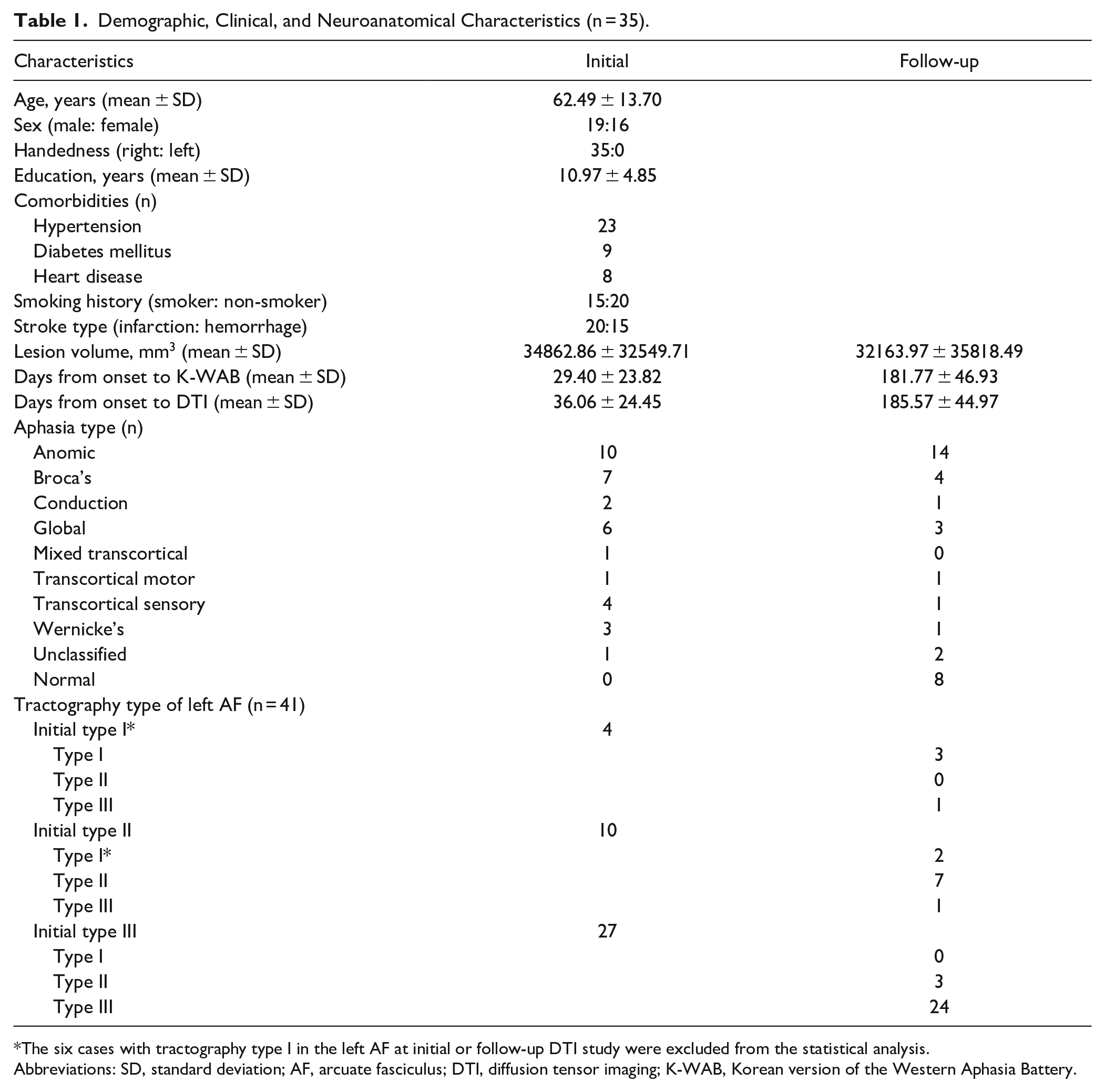
The six cases with tractography type I in the left AF at initial or follow-up DTI study were excluded from the statistical analysis.
Abbreviations: SD, standard deviation; AF, arcuate fasciculus; DTI, diffusion tensor imaging; K-WAB, Korean version of the Western Aphasia Battery.
The severity of aphasia significantly improved as shown by K-WAB assessment at the 6-month follow-up compared to that initially in terms of AQ and all subtest scores; the mean AQ score improved to 70.73 points from 50.7 points (P < .001) and mean ΔAQp was 0.42. At 6 months post-stroke, both AFs showed statistically significant changes in several DTI parameters. The left AF showed a significant decrease in FA (P = .027) and an increase in diffusivity indices (AD, MD, and RD) (P < .01). The right AF showed a significant decrease in FA (P = .002) and an increase in MD (P = .048) and RD (P < .001) but no significant change in AD (P = .436) (Table 2).
Changes in Aphasia Severity and Diffusion Tensor Imaging Parameters of Bilateral Arcuate Fasciculus.
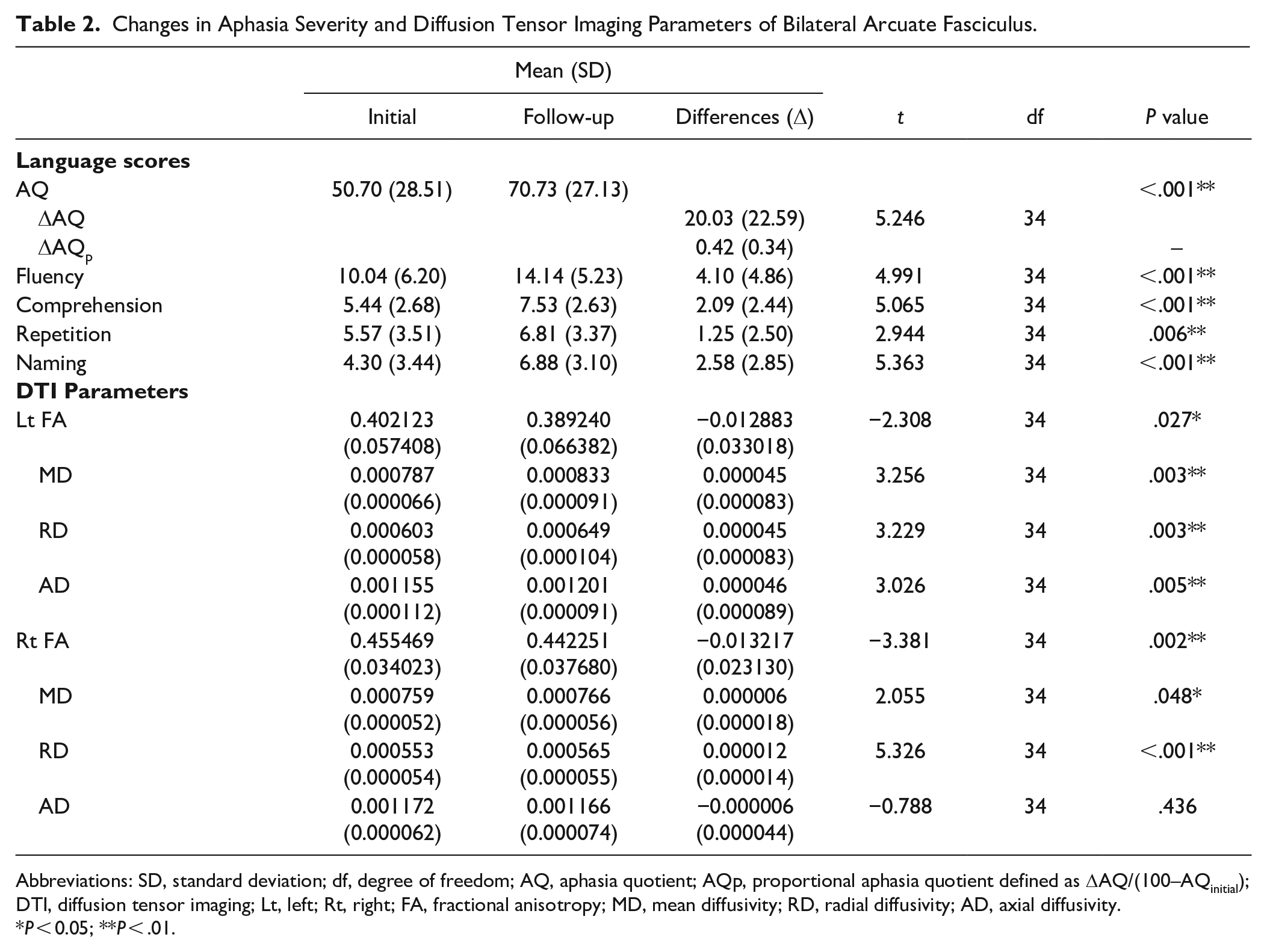
Abbreviations: SD, standard deviation; df, degree of freedom; AQ, aphasia quotient; AQp, proportional aphasia quotient defined as ΔAQ/(100–AQinitial); DTI, diffusion tensor imaging; Lt, left; Rt, right; FA, fractional anisotropy; MD, mean diffusivity; RD, radial diffusivity; AD, axial diffusivity.
P < 0.05; **P < .01.
Correlations between DTI Parameters and Language Recovery
The following variables with a P value < .8 in univariable analysis were selected for multivariable analysis: age, stroke type, years of education, Δlesion volume, initial AQ, left ΔFA, and right ΔFA. The p values of ΔMD, ΔRD, and ΔAD in the left AF were also <0.8; since ΔFA, ΔMD, ΔRD, and ΔAD were highly correlated (Spearman’s Rank correlation coefficients ≥0.8), only ΔFA with the largest beta coefficient was included in the analysis to avoid multicollinearity. In multivariable analysis, ΔFA of the left AF showed a statistically significant correlation with ΔAQp (β = 0.365, P = .031) (Table 3 and Figure 3). Among the K-WAB subtests, fluency (ΔFp) and repetition (ΔRp) improvements showed a correlation of borderline significance with ΔFA of the left AF (P = .017 and P = .022, respectively) (Supplementary Table 1). There was no significant correlation between ΔFA of the right AF and language recovery (β = .217, P = .211).
Univariable and Multivariable Linear Regression Analysis with ΔAQp as Outcome Measure.
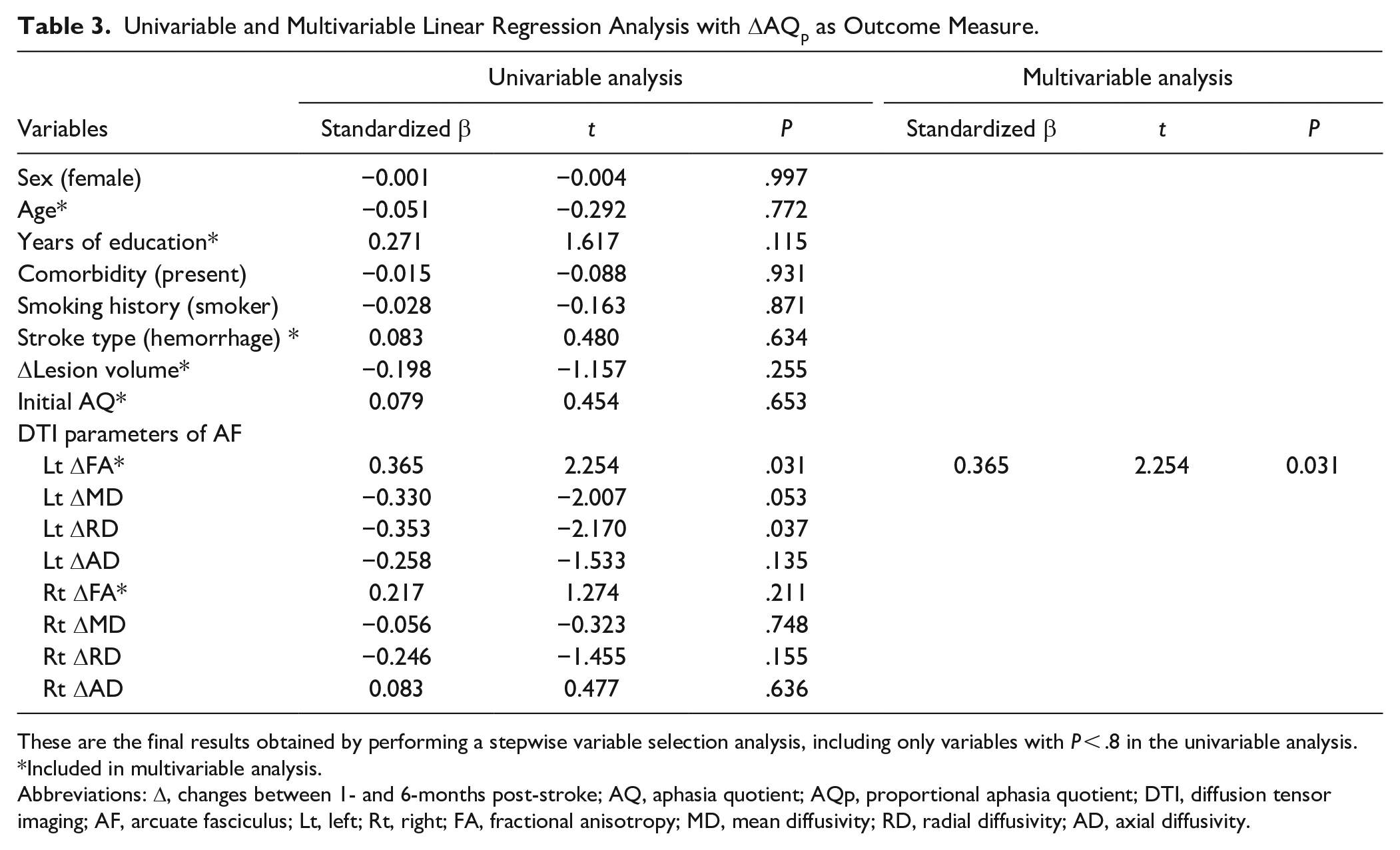
These are the final results obtained by performing a stepwise variable selection analysis, including only variables with P < .8 in the univariable analysis.
Included in multivariable analysis.
Abbreviations: Δ, changes between 1- and 6-months post-stroke; AQ, aphasia quotient; AQp, proportional aphasia quotient; DTI, diffusion tensor imaging; AF, arcuate fasciculus; Lt, left; Rt, right; FA, fractional anisotropy; MD, mean diffusivity; RD, radial diffusivity; AD, axial diffusivity.
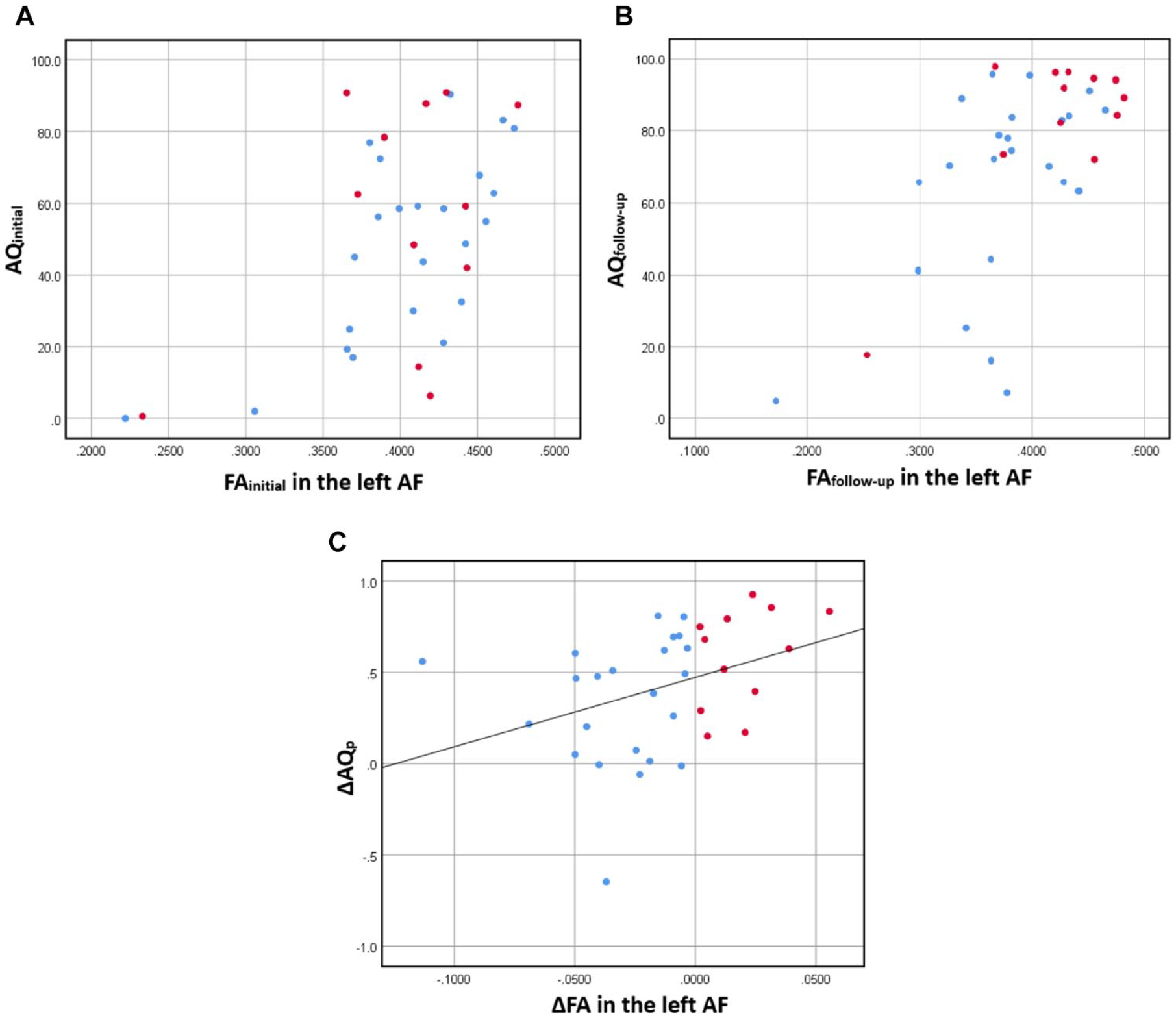
Relationship between Aphasia Quotient (AQ) Score and Fractional Anisotropy (FA) Value of Left Arcuate Fasciculus (AF). Scatter Plot between AQ Score and FA Value at Initial (A) and Follow-Up Evaluation (B), and (C) Regression Plot between change in FA (ΔFA; FAfollow-up– FAinitial) in the left AF and Proportional Change in AQ (ΔAQp; (AQ6mon–AQinitial)/(100–AQinitial)). Blue dots: Patients with Decreased FA (Group 1); Red Dots: Patients with Increased FA (Group 2).
At the group level, the left AF showed a significant decrease in the FA value over time. However, in individual patients, the FA value longitudinally decreased in 23 patients (group 1) but increased in 12 patients (group 2). For K-WAB scores, most patients showed improvement irrespective of an increase or decrease in the FA value in the left AF; only 4 out of 35 patients showed a decline in language functions. Patients with an increased FA in the left AF (group 2) showed more significant ΔAQp improvement than those with decreased FA (group 1) (Figure 4). Since the degree of change in ΔAQp is much larger in group 2 than that in group 1, it was interpreted as showing a positive correlation between ΔFA and ΔAQp at the group level.
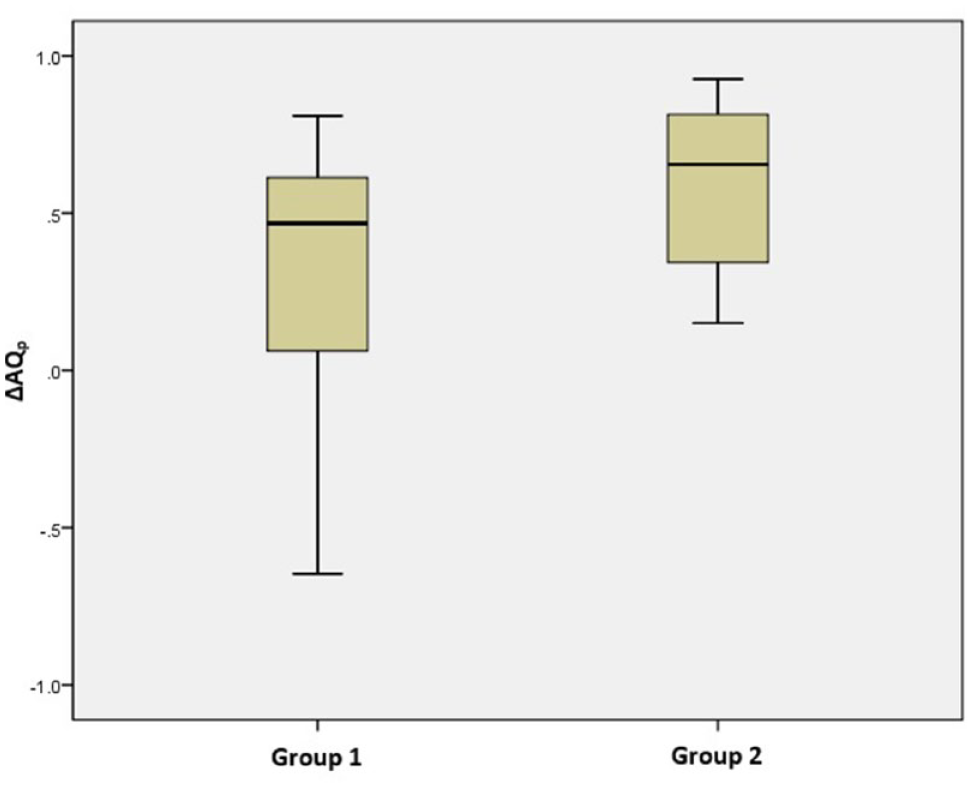
Comparison of Proportional Change in Aphasia Quotient (ΔAQp) between Groups with Decreased FA (Group 1) and Increased FA (Group 2) in the Left Arcuate Fasciculus.
Discussion
The neural mechanisms underlying aphasia recovery and the contribution of language-associated white matter tracts remain unresolved. Although research on functional connectivity has improved our understanding of neuroplasticity in language networks, studies on structural connectivity, especially in the subacute stage where spontaneous recovery is most prominent, are still lacking. In this study, we aimed to explore changes in white matter tract integrity, specifically in AF during the subacute phase, and to determine whether these changes correlated with language recovery in patients with post-stroke aphasia. Using longitudinal DTI analysis, we identified temporal changes in AF in both hemispheres. On analysis, significantly decreased FA and increased diffusivity indices (MD and RD) in both left and right AF were noted. The FA and RD changes in the left AF were significantly correlated with improvement in the AQ score, but the right AF was not.
Degeneration of the white matter tract is accompanied by the degradation of axons or myelin sheaths. The surrounding microstructural environment changes, and water molecules become mobile. Altered diffusion can be measured using DTI parameters.36,37 FA is the most commonly used parameter that is highly sensitive to changes in the overall tract integrity; however, it is not very specific to the type of changes.36,37 In contrast, RD and AD reflect specific relationships with white matter pathology, demyelination, and axonal degeneration, respectively.8,36,38,39 Destruction of the myelin sheath allows water molecules to diffuse more freely in the direction perpendicular to the axons, which can be reflected as increased RD.8,36,38 On the other hand, AD often decreases in the acute phase after axonal damage, followed by an increase during transition from subacute to chronic phases.8,39 Additionally, an increase in MD (the average amplitude of diffusion) is usually consistent with an increase in water content in conditions such as inflammation, edema, and cerebral hemorrhage. 40 Therefore, decreased FA and increased MD along with increases in both AD and RD may reflect impairment in the overall axonal and myelin integrity of the tract.8,36,41 However, interpretation of changes in DTI parameters is complex and requires caution as it can be influenced by various factors, including partial volume effect and regions of crossing tracts.36,42 Furthermore, as the lesion size decreases with resorption of hemorrhage and edema over time, temporal evolution of DTI parameters may also influence the interpretation.43,44 In this study, longitudinal alterations in ipsilesional structural connectivity were detected, FA decreased, and all three diffusivity indices (AD, RD, and MD) increased in the left AF. These changes might be explained by a direct effect of the lesion, which possibly induced Wallerian degeneration of the AF. Previous studies have suggested the diminished integrity of the left AF in patients with aphasia using FA value.17,18,20 Ivanova et al. reported significantly higher MD, RD, and AD values in addition to lower FA of left AF in these patients compared to those in healthy controls, reflecting profound impairment in tract integrity. 8 A trend similar to our results has also been reported for CST. A longitudinal study investigated Wallerian degeneration of the ipsilesional CST at five consecutive time points after stroke and demonstrated an increase in all diffusivity indexes with a decrease in FA. 45 Another study has also shown similar pattern of diffusivity alterations in CST after stroke compared to that in controls. 41
In our study, longitudinal degenerative changes were also detected in the contralesional right AF but showed slightly different features from the ipsilesional side. FA, MD, and RD of the right AF showed similar changes to those of the left AF, but AD was preserved. The absence of significant changes in AD implies a pathophysiology different from the direct axonal damage seen in the left AF. Structural changes of the contralesional hemisphere can be explained by transneuronal degeneration. Transneuronal degeneration refers to the structural deterioration of areas remote from but connected to the initial pathology. 46 Interactions can be disrupted by reduced trophic support or altered neuronal stimulation from the injured area. Degeneration of remote regions can show various features such as neuronal shrinkage, reductions in dendrite and synapse number, and alterations of axonal myelin content and fiber number. 46 Umarova et al 47 conducted a longitudinal study in patients with post-stroke neglect to investigate whether dysfunction of an area remote from a stroke lesion accompanies structural changes in remote white matter tracts. They presented alterations in neglect-related white matter networks that were primarily preserved from the lesion. The remote alterations represented an increase in RD without a corresponding change in AD, unlike perilesional changes in which FA decreased and all diffusivity indices increased.
In the current study, the language score of the participants improved significantly over time, and the improvement correlated positively with the FA changes in the left AF; a greater increase in FA resulted in greater language improvement. However, the left AF showed a degenerative temporal change characterized by reduced FA at the group level. To understand this discrepancy, we performed additional analysis, dividing participants into two subgroups according to the direction of change in the left FA. We compared the amount of language recovery between the two groups. Patients in group 2 (red dots in Figure 3) showed more significant ΔAQp improvement compared with those in group 1 (blue dots in Figure 3). Because the degree of change in ΔAQp was much larger in patients with an increased FA, ΔAQp and ΔFA consequently showed a positive correlation at the group level. There were significant language improvements in both groups, which may be related to activation of the cortical regions. Saur et al 48 mapped the dynamic changing patterns of language activation using functional MRI and represented the mechanisms of language recovery longitudinally. A reduced activation in the acute phase is followed by bilaterally increased activation in the early subacute phase as a result of reperfusion of the penumbra and resolution of diaschisis, which correlate with language improvement.49,50 However, marked language improvements, which were shown in the group with an increased FA, suggest a role for white matter plasticity in aphasia recovery.
Several previous studies examining the relationship between structural plasticity in white matter and aphasia recovery in patients in the chronic stage reported an increased fiber integrity correlating with language improvement. Two of these studies reported the remodeling of the right AF after MIT in patients with large left hemisphere lesions.15,16 Schlaug et al 15 found a significant increase in the number of AF fibers and AF volume after successful MIT, possibly due to increased myelination of axons and additional axon growth or collaterals. Wan et al 16 reported a significant FA reduction in the white matter underlying the right inferior frontal gyrus and its association with improvements in speech production. Although it may seem contradictory to our results that an increase in FA is associated with better language recovery, a decrease in FA was interpreted as an improvement in fiber integrity by more axonal sprouting and more branching in close proximity of the cortical target regions. Further three longitudinal studies investigated remodeling of the left AF or left ILF following intensive language therapy.17,18,51 Two of them17,18 showed training-induced improvement in fiber integrity characterized by an increase in FA of the left AF following constraint-induced language therapy and anomia therapy, respectively. The other study 51 reported an association between semantic improvement and structural plasticity of the left ILF. The overall trend of change in white matter tracts in these studies differed from those of our study, which demonstrated longitudinal degenerative changes of either ipsilesional or contralesional AF over time. This discrepancy may suggest that the direction of change in white matter may depend on the time after stroke, that is, the difference between the subacute and chronic stages. Treatment-induced plasticity might be reflected in chronic-stage disease in which the structural state is relatively stable. However, our findings establish that an increase in fiber integrity is associated with behavioral improvement, which corroborates with the results of the aforementioned studies.
To our knowledge, there is only one observational study conducted in the subacute stage where changes in the dorsal and ventral language tracts in the left and right hemispheres were investigated in relation to naming recovery in patients with aphasia. 21 Contrary to our results, however, language improvement assessed with the Boston Naming Test (BNT) showed no significant correlation with FA change in the left AF, but a significant correlation was found with FA change in the right ILF. One explanation for the discrepancy is the difference in the language assessment tools used. AF is known to be critical for phonological processing, whereas ILF is important for lexical-semantic processing, 8 which is well reflected in the BNT. In our study, improvement in fluency (ΔFp) and repetition (ΔRp) among the K-WAB subtests showed a higher correlation with ΔFA of the left AF than improvement in naming (ΔNp). Interestingly, this study showed that an increase in FA in the right ILF was associated with a considerable improvement in naming scores, whereas a decrease was related to a limited improvement or slight decline. Although the type of tracts and side of hemisphere were different, their results were consistent with our study where we established that improvement of structural integrity is related to the recovery of aphasia in the subacute stage. It remains unknown why some participants showed an increase in the integrity of the left AF, although, overall, the group showed a trend of diminished integrity. There might be different underlying mechanisms, but the variables in our dataset could not explain this difference. Further studies with larger sample sizes are required.
In our results, there was no statistically significant correlation between DTI parameters of the right AF and language improvement, which was an unexpected result. Although the role of the right hemisphere in aphasia recovery remains debatable, many studies have reported a facilitating role of the right hemisphere.50,52 -56 In addition, a previous DTI study on AF in patients with aphasia 12 reported that the volume of the right long segment of AF was a predictor of aphasia recovery. Therefore, the contribution of the right hemisphere language network was expected but not elicited in this study.
The limitations of this study include the small sample size. We recruited patients with aphasia for an extended period, but many patients were lost at the follow-up. Second, the time from stroke onset at which the language and DTI assessments performed showed large deviations across the included patients. As significant recovery occurs rapidly in the subacute stage, 57 the possibility of the impact of time on the outcome cannot be excluded. Third, we excluded data from six patients with severe aphasia because their AF was not reconstructed. Therefore, changes in AF parameters in patients with severe aphasia were not analyzed in this study. Fourth, as DTI-based tractography is unable to properly reconstruct crossing fibers, the possibility of false negatives, that is, missing existing connections, cannot be ruled out; this implies that some AF fibers may be missed in the current reconstructions.36,58,59 Fifth, we had no control on the aphasia therapy during the follow-up period. On admission to our department, patients underwent a comprehensive rehabilitation program for 4 weeks, including individualized language therapy. Then, some patients were discharged home while others were transferred to another hospital; thus, speech therapy could not be controlled. Therefore, the contribution of potentially variable aphasia therapy to the parameters of AF changes could not be investigated. Lastly, our study was based on the anatomical model proposed by Catani et al, 60 and we analyzed only the long segment of the model and not the indirect segments. The number of patients included in our study was not large enough to enable subgroup analysis according to the location of the damaged segments. Therefore, we focused on the overall integrity of the AF tract and its relationship with the overall language improvement.
Conclusion
Our findings demonstrated that the FA values representing the integrity of the bilateral AF decreased and the diffusivity index increased 6 months post-stroke compared with those at 1 month after stroke. Among the DTI parameters, FA changes in left AF were significantly and positively correlated with language improvement. Patients in the increased FA group showed more significant language improvement than those in the decreased FA group. These findings help to understand the natural course of structural connectivity changes in the language network in aphasia patients in the subacute phase after stroke and the underlying mechanisms of language recovery.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683221121752 – Supplemental material for Structural Changes in the Arcuate Fasciculus and Recovery of Post-stroke Aphasia: A 6-Month Follow-up Study using Diffusion Tensor Imaging
Supplemental material, sj-docx-1-nnr-10.1177_15459683221121752 for Structural Changes in the Arcuate Fasciculus and Recovery of Post-stroke Aphasia: A 6-Month Follow-up Study using Diffusion Tensor Imaging by Cho Rong Bae, Yoonhye Na, Minjae Cho, Yu Mi Hwang, Woo-Suk Tae and Sung-Bom Pyun in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank all the participants and colleagues of the Department of Rehabilitation Medicine at Korea University Anam Hospital for their contributions to this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. 2022R1A2B5B02001673).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
