Abstract
Background. In functional magnetic resonance imaging studies, picture naming engages widely distributed brain regions in the parietal, frontal, and temporal cortices. However, it remains unknown whether those activated areas, along with white matter pathways between them, are actually crucial for naming. Objective. We aimed to identify nodes and pathways implicated in naming in healthy older adults and test the impact of lesions to the connectome on naming ability. Methods. We first identified 24 cortical nodes activated by a naming task and reconstructed anatomical connections between these nodes using probabilistic tractography in healthy adults. We then used structural scans and fractional anisotropy (FA) maps in 45 patients with left hemisphere stroke to assess the relationships of node and pathway integrity to naming, phonology, and nonverbal semantic ability. Results. We found that mean FA values in 13 left hemisphere white matter tracts within the dorsal and ventral streams and 1 interhemispheric tract significantly related to naming scores after controlling for lesion size and demographic factors. In contrast, lesion loads in the cortical nodes were not related to naming performance after controlling for the same variables. Among the identified tracts, the integrity of 4 left hemisphere ventral stream tracts related to nonverbal semantic processing and 1 left hemisphere dorsal stream tract related to phonological processing. Conclusions. Our findings reveal white matter structures vital for naming and its subprocesses. These findings demonstrate the value of multimodal methods that integrate functional imaging, structural connectivity, and lesion data to understand relationships between brain networks and behavior.
Introduction
Functional neuroimaging studies of language have identified widespread temporal, parietal, and frontal brain regions that consistently activate when normal individuals perform language tasks.1-3 Correspondingly, speech and language are thought to be supported by multiple white matter tracts connecting these regions.4-6 Structural neuroimaging studies have suggested that damage to specific white matter tracts leads to deficits in specific language processes, such as fluency, phonology, or semantics.7-9 Recent diffusion tensor imaging (DTI) studies further reveal that the integrity of white matter tracts connecting lesioned or even intact cortical regions after brain damage contributes to language impairments.7,10-12
Picture naming is a key measure of word retrieval and production widely used in both neuroimaging and neuropsychology. Converging evidence from these 2 fields suggests that naming involves distributed left lateralized regions, including superior temporal gyrus, inferior parietal cortex, and prefrontal cortex.13-17 The anatomical connections between these regions that support naming remain underspecified. Recent studies have shown that lesioned pathways in the dorsal or ventral streams correlate with phonological and semantic deficits important for naming after stroke.7,9,18 However, these studies did not measure white matter integrity between specific cortical regions using diffusion imaging, instead relying on lesion overlap methods.
Although combining functional imaging, structural connectivity, and lesion methods would provide advantages over each method alone, studies using functional magnetic resonance imaging (fMRI) to identify language regions in healthy participants and those using lesion methods to examine language networks have largely been conducted separately. It is, thus, unclear if the cortical regions identified in fMRI studies and the white matter pathways between those regions are vital for language performance as measured by the behavioral effects of lesions. To address this gap, we combined fMRI and DTI data to produce a structural connectome composed of the cortical nodes and white matter tracts important for picture naming in healthy adults. Then, in left hemisphere stroke survivors, we tested for relationships between naming ability and the integrity of these nodes and tracts. This approach is similar to a series of prior studies that utilized probabilistic tractography between cortical areas activated in healthy adults to identify 2 white matter pathways involved in phonology and semantics 5 and then examined the effects of acute stroke lesions in these 2 pathways using a lesion overlap approach. 7 The current study differs from this prior work in several ways, including a focus on naming, examination of 85 intrahemispheric and interhemispheric tracts across both hemispheres, measurement of white matter tract integrity in stroke survivors using diffusion imaging, and examination of the lesion-behavior relationship in the chronic period to allow identification of tracts related either to lesion-induced deficits or to recovery.
Methods
Participants
A total of 45 chronic left hemisphere stroke survivors with history of aphasia were recruited in the study, with inclusion criteria as follows: native English speaker, at least 6 months poststroke, able to follow testing instructions, and no history of other significant neurological illnesses. See Table 1 for characteristics of the group and the Supplemental Table for the characteristics of individual participants. All patients had aphasia at the time of stroke, based on medical records and received speech-language therapy.
Demographic Data and Language Measures in Patients and Healthy Participants.
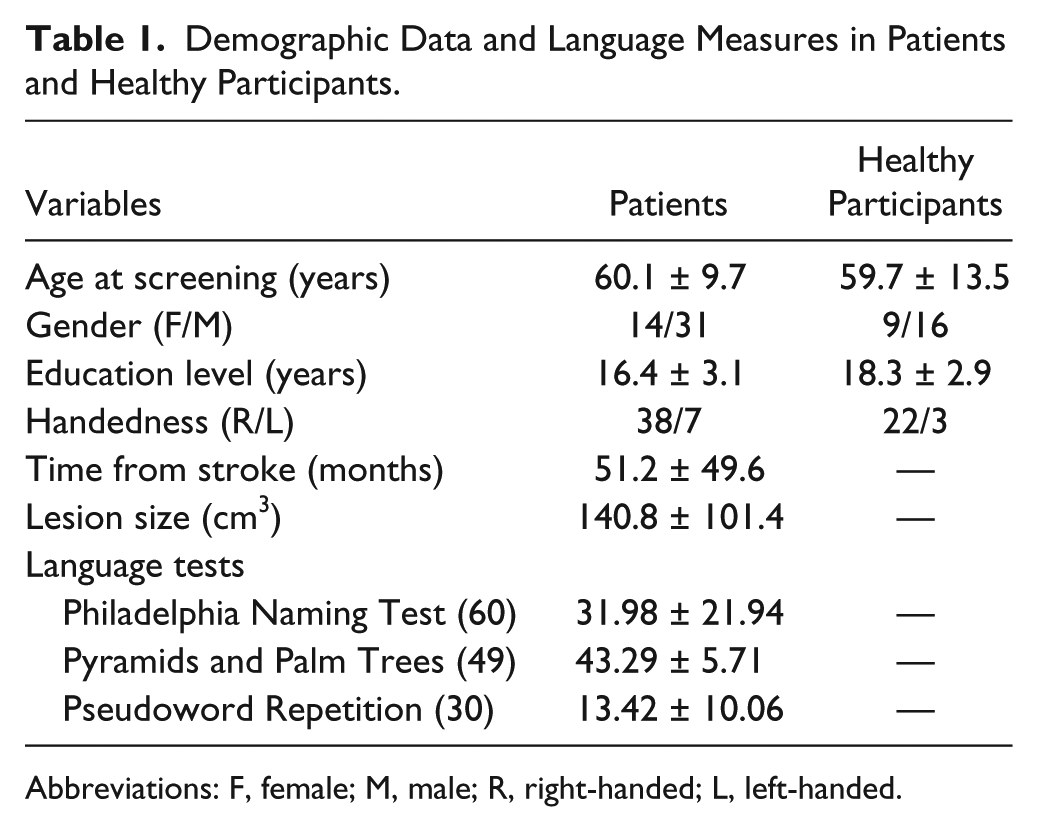
Abbreviations: F, female; M, male; R, right-handed; L, left-handed.
The study was approved by the Georgetown University Institutional Review Board, and written informed consent was obtained from all study participants prior to enrollment in the study. A total of 25 healthy individuals without neurological and psychiatric disorders, matched to the stroke group on age, education, handedness, and gender, were enrolled in the study (Table 1).
Language Testing
Naming performance was assessed using a 60-item version of the Philadelphia Naming Test (PNT), 19 used frequently in our prior studies.20-22 Items were selected based on per-item performance across patients in the Moss Aphasia Psycholinguistics Project Database 23 to match performance on the full version of the PNT. The total number of items on the PNT that were named correctly on the first attempt was counted.
Phonological processing was assessed with a 30-item in-house pseudoword repetition (PWR) task. The Pyramids and Palm Trees Test (PPT) 24 was administered to assess nonverbal semantic processing. During the PPT, patients must match a picture to its closest associate among a set of 2 other pictures.
Image Acquisition
MRI data were acquired on a 3T Siemens Trio scanner at Georgetown University Medical Center. 3D T1-weighted images were obtained with a magnetization-prepared rapid-acquisition gradient echo sequence: repetition time (TR)/echo time (TE) = 1900/2.56 ms; flip angle = 9°; field of view (FoV) = 250 × 250 mm2; voxel size = 1 × 1 × 1 mm3; 160 contiguous sagittal slices. Diffusion-weighted images (DWIs) were acquired using single-shot echo-planar imaging: TR/TE = 7500/87 ms; flip angle = 90°; FoV = 240 × 240 mm2; voxel size = 2.5 × 2.5 × 2.5 mm3; slice thickness = 2.5 mm; sagittal slice number = 64 slices; and 60 diffusion volumes with bmax of 1100 s/mm2, 10 volumes with bmin of 300 s/mm2, and 10 volumes with no diffusion gradient (b = 0 s/mm2). FMRI volumes were acquired in healthy participants using a T2*-weighted EPI sequence: TR/TE = 2000/30 ms; flip angle = 90°; FoV = 250 × 250 mm2; voxel size = 3.2 × 3.2 × 3.2 mm3; and 38 contiguous slices.
Functional MRI Task
To identify brain regions typically involved in picture naming, we acquired fMRI data from 25 healthy participants. A previous analysis of these fMRI data was presented elsewhere, and details of the task are provided in that publication. 20 The stimuli consisted of 32 line drawings presented in pseudorandomized order with 3 stages for each long event-related trial: covert naming (7.5-9.0 s), overt naming (5.5 s), and fixation (14.0 s). The delayed response was intended to isolate word retrieval from motor/auditory speech processing 25 and remove fixed temporal associations between word retrieval and jaw motion artifacts. Images were presented using a mirror and projection system and E-Prime software (Psychology Software Tools Inc, Pittsburg, PA), and responses were recorded using a MRI-safe microphone (Opto-acoustics, FOMRI-III).
Image Data Preprocessing
Structural Data
Lesion masks were traced manually on the T1-weighted images in native space in MRIcron 26 and checked by 2 neurologists (SX and PET). The structural images were registered to an intermediate template created from images acquired from the same scanner using the Advanced Normalization Tools package (http://picsl.upenn.edu/ANTS). Lesions were masked for registration using a variant of cost function masking. The single mapping from the intermediate template space to Montreal Neurological Institute (MNI) space was subsequently conducted to register the structural images to MNI space. 18 The individual lesion map was then warped into MNI space by applying the same mapping. A lesion overlap map is shown in Figure 1.
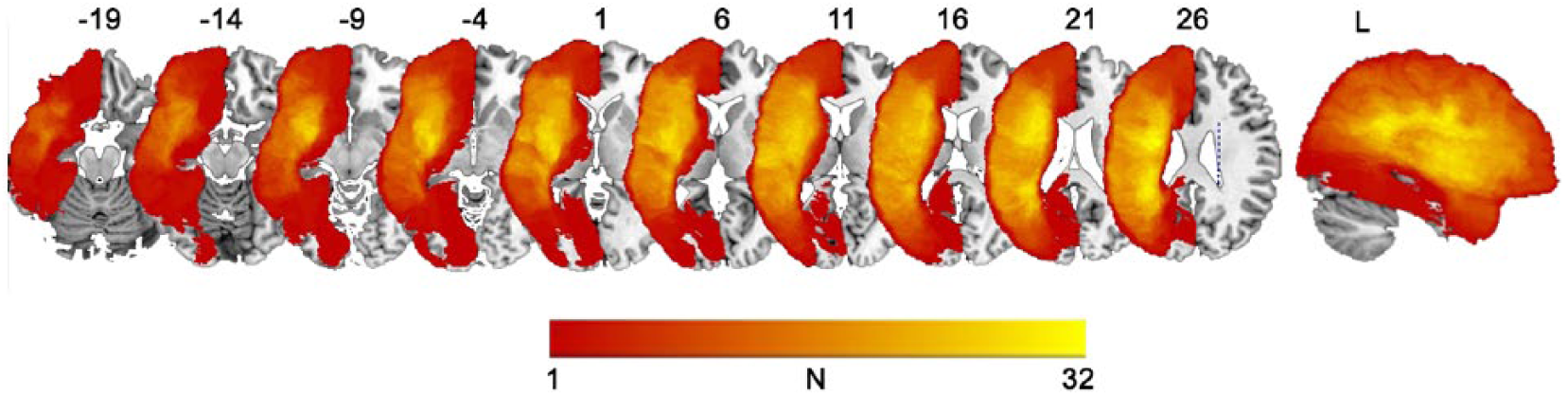
Lesion overlap map of 45 patients with chronic poststroke aphasia. N denotes the number of patients with a lesion in each voxel (maximum 32 out of 45).
Diffusion Data
Preprocessing of the diffusion images was performed using the FMRIB software Library (FSL; http://www.fmrib.ox.ac.uk/fsl/). For DWI data, eddy current distortions and motion artifacts were corrected by registering each diffusion volume to the nondiffusion volume with an affine transform. Following tensor estimation, spatial normalization was performed using a nonparametric, diffeomorphic deformable image registration technique, 27 which incrementally estimates its displacement field using a tensor-based registration formulation. It is designed to take advantage of similarity measures comparing tensors as a whole via explicit optimization of tensor reorientation and includes appropriate reorientation of the tensors following deformation. 28 We measured the degree of damage to white matter tracts reconstructed below using the standard tensor-derived fractional anisotropy (FA).
Functional MRI Data
FMRI data were preprocessed and analyzed using FSL tools. Preprocessing included high-pass temporal filtering, correction for head motion using MCFLIRT, slice timing correction, and intensity normalization across volumes. Registration and normalization to the MNI template brain was carried out using FLIRT in FSL. The functional images were finally spatially smoothed to 5 mm full width at half maximum. At the first level, a canonical double-gamma hemodynamic response function was constructed for the duration of the event in each trial. Motion parameters were included as covariates in the model. Covert naming plus overt naming conditions were contrasted against fixation. Although the task design allowed isolation of these 2 processes, the activity for the 2 conditions overlapped substantially in comparison to fixation; so for simplicity, we analyzed all naming activity compared to fixation to identify all cortical areas involved in naming. At the second level, a gray matter mask was applied to all contrasts, and a group contrast was carried out. The resultant group activation map was thresholded at P < .001, uncorrected to ensure sensitivity to all areas involved in naming.
Construction of Naming Processing Network in Healthy Participants
Definition of Cortical Regions of Interest (ROIs) in the Normal Naming Network
To define cortical regions involved in normal picture naming in healthy controls, we identified locations of peak activation in the group-level fMRI analysis. In total, 12 bilateral homologous regions were identified in each hemisphere as the naming-relevant cortical regions, including various subregions of the frontal, parietal, and temporal lobes (Table 2). Spherical seed regions with 5-mm radius were then generated centered on each of the fMRI peak coordinates. These seed ROIs were then transformed back to the native DWI space using the inverse normalization parameters obtained during the normalization of the anatomical image with FNIRT. Prior to normalization and inverse warping, the FA image was coregistered to the corresponding anatomical image using FLIRT in FSL.
MNI Coordinates of Peak Voxel in Regions of Interest Defined From Naming-Activated fMRI Maps.
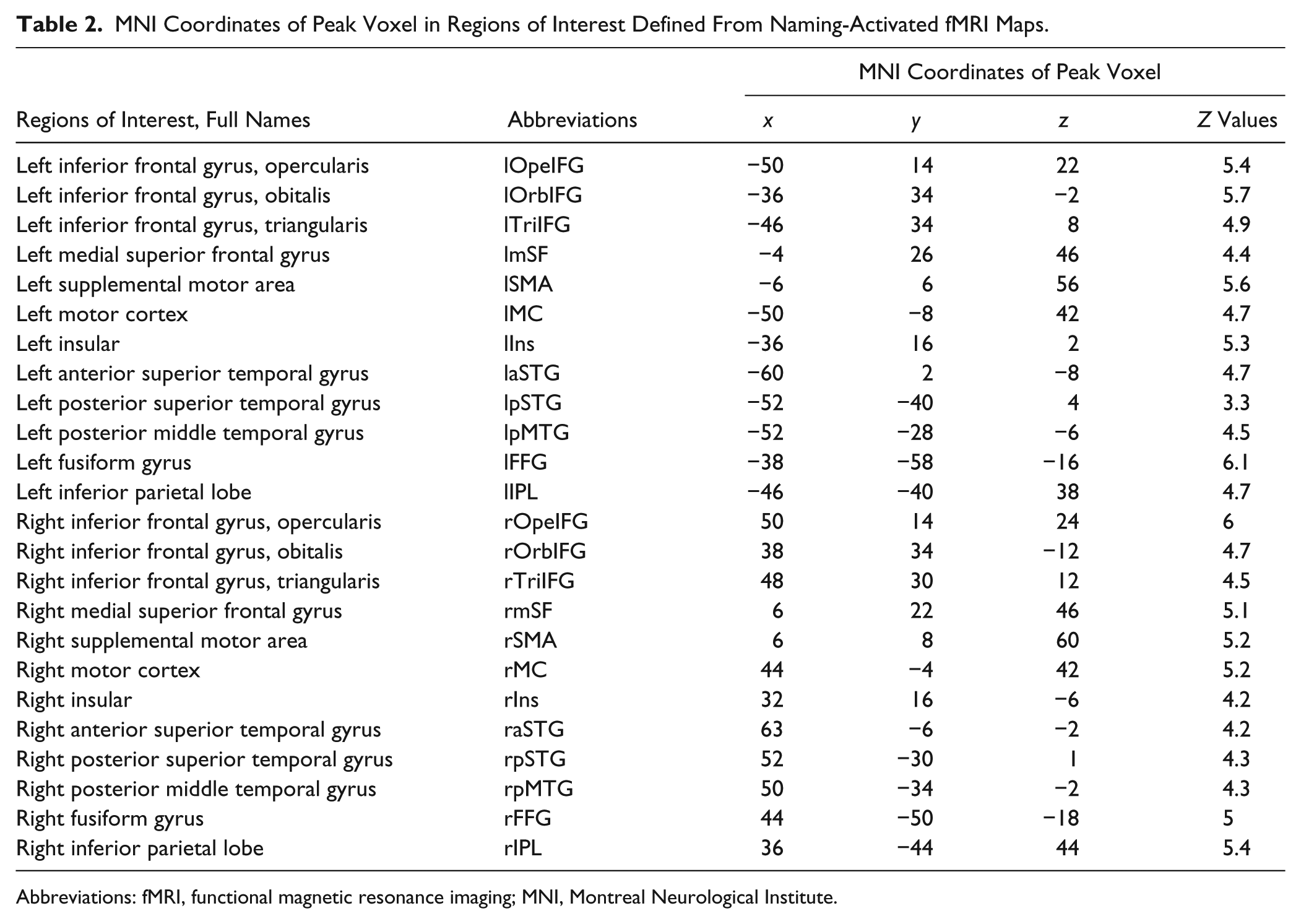
Abbreviations: fMRI, functional magnetic resonance imaging; MNI, Montreal Neurological Institute.
Probabilistic Tractography to Identify White Matter Connections in the Normal Naming Network
For each healthy participant, white matter connections between the 12-seed ROIs within each hemisphere and interhemispheric connections between homologous regions were extracted using a probabilistic tractography algorithm implemented in the FSL toolbox (probtrackx) based on Bayesian estimation of diffusion parameters (Bedpostx). 29 Briefly, fiber tracking was performed between each pair of ROIs with one as seed and another as target. For each voxel within the seed region, we generated 5000 streamline samples with a step length of 0.5 mm and a curvature threshold of 0.2 to map the probabilistic connection pattern. The tract density map was obtained by dividing by the total number of streamline samples. Fiber tracking was performed in both directions from seed to target and backward, and the connection maps were averaged. To estimate the dorsal and ventral connectivity between ROIs, we then created dorsal and ventral masks based on the Johns Hopkins University white-matter tractography atlas 30 and used them as the excluded masks to constrain the tracking algorithm to fibers passing through the ventral or dorsal pathways. The resultant density maps were thresholded at 1% to exclude spurious connections and normalized to MNI space. Probabilistic connection maps were created by summing the thresholded density maps across all healthy participants. Each resultant group-wise connection map was thresholded at a level of 50%, indicating that at least 13 of the 25 participants consistently showed the connections within each voxel of the tract. The reconstructed connections in MNI space were then nonlinearly transformed to the group-wise FA template using FNIRT in FSL. Finally, the FA values of reconstructed connections were extracted across patients for the statistical analyses.
Statistical Analysis
To examine the behavioral impact of lesions to the cortical nodes in the normal naming network, partial correlations were conducted between lesioned voxels within seed regions and PNT scores in patients, controlling for total lesion volume and demographic factors. Similarly, to examine the impact of lesions to the white matter connections in the normal naming network, partial correlations were conducted between FA values of reconstructed tracts and PNT scores in patients. To further clarify how specific tracts contribute to phonological and semantic processes important for naming, partial correlations were conducted between FA values of tracts and PWR or PPT scores, controlling for the same variables. For each of these analyses, multiple comparisons were corrected at a threshold of q <0.05 with false discovery rate (FDR).
Results
Reconstructed Tracts Related to Naming Processing in Healthy Individuals
A total of 12 paired cortical seeds in each hemisphere were identified based on the naming-related fMRI activity in the healthy controls. Probabilistic tractography resulted in a total of 85 connections between these seed regions, including 45 tracts in the left hemisphere and 32 tracts in the right hemisphere, and 8 interhemispheric tracts (Table 3, Figure 2). The average size of tracts was 3218 mm3 (SD = 1337 mm3; range = 118-7122 mm3).
Partial Correlations Between Integrity of Reconstructed Tracts and PNT Scores in Patients, Controlling for Age, Gender, Education Level, Handedness, Time From Stroke Onset, and Total Lesion Volumes. a
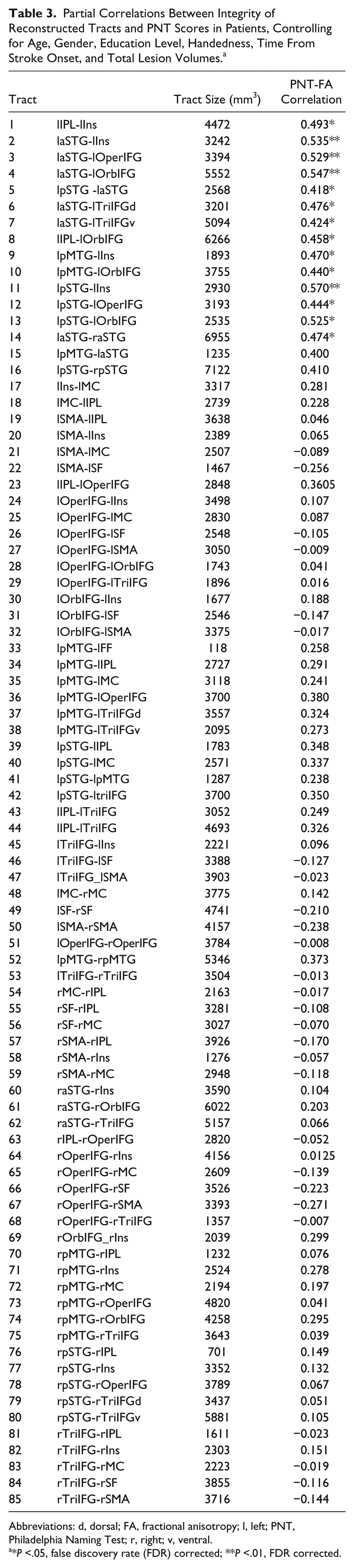
Abbreviations: d, dorsal; FA, fractional anisotropy; l, left; PNT, Philadelphia Naming Test; r, right; v, ventral.
*P <.05, false discovery rate (FDR) corrected; **P <.01, FDR corrected.
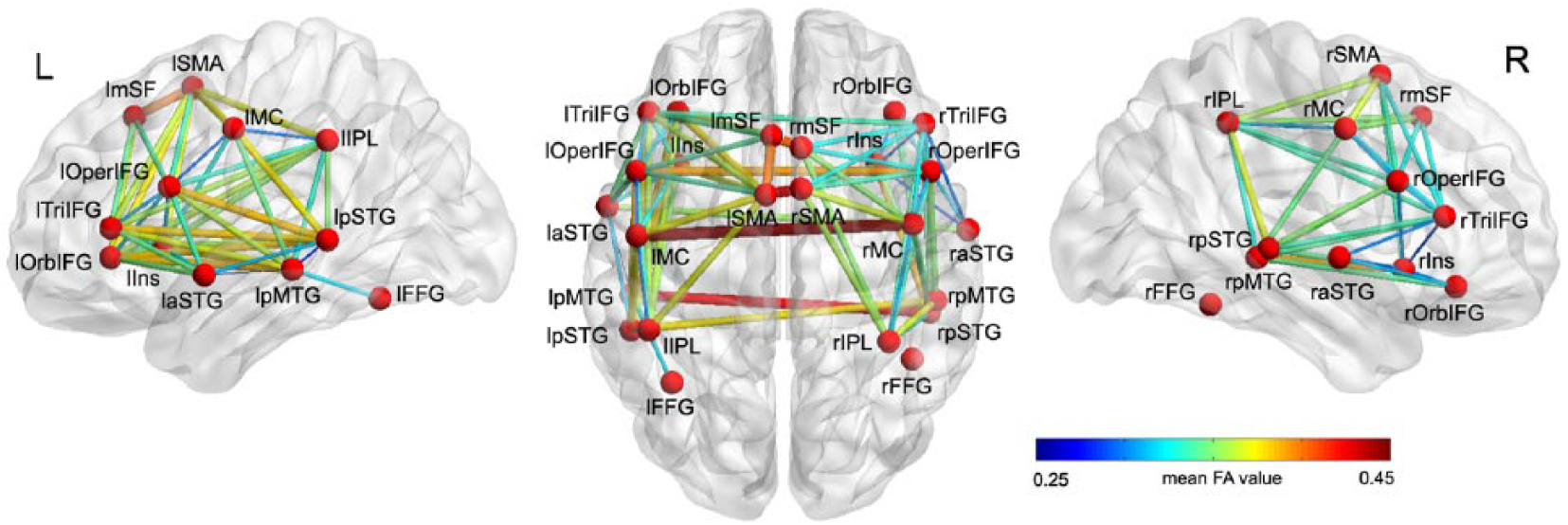
White matter connections between regions of interest activated in response to naming performance in healthy participants. A total of 85 tracts were successfully reconstructed between 24 seeds in 25 age-matched healthy participants. Among them, there were dorsal and ventral connections between 3 seed pairs (laSTG/lTriIFG, lpMTG/lTriIFG, and rpSTG/rTriIFG). The thickness and color of the tracts indicate the mean fractional anisotropy values. Full names of seeds and tracts are listed in Tables 2 and 3.
Impact of Lesions to Cortical Nodes on Naming Ability in Patients
To determine the behavioral impact of lesions to the cortical nodes in the normal naming network, partial correlations between the lesion load on each seed in left hemisphere and PNT scores were examined. The lesion load of left hemispheric seeds on average was 29.32% (SD = 24.62%; range = 2.02% in lFF to 49.25% in lIns). The percentage of patients with a lesion in each left hemispheric seed was 6.67% in lFF to 68.89% in lIns. When controlling for age, gender, education, handedness, and time from stroke, PNT performance was significantly negatively related to lesion load on 7 left hemisphere seeds (lIPL: partial r = −0.41, P = .0086; lIns: r = −0.51, P = .0074; lMC: r = −0.48, P = .0017; laSTG: r = −0.35, P = .026; lOperIFG: r = −0.43, P = .0053; lOrbIFG: r = −0.47, P = .0022; lTriIFG: r = −0.41, P = .009; FDR corrected with q < 0.05). However, when adding total lesion volumes as an additional covariate, no significant correlations between lesion loads on left hemisphere seeds and PNT performance were found (all P > .05, FDR corrected). Thus, no specific left hemisphere cortical ROIs activated in normal controls were identified in which local damage affected naming performance above and beyond the effect of total lesion size.
Impact of Lesions to the White Matter Tracts in the Normal Naming Network on Naming Ability in Patients
Next, to examine whether the observed tracts connecting paired naming-activated seeds are functionally related to overall naming performance, we conducted separate partial correlation analyses to test the relationships between FA values for each tract and PNT scores in patients. The results showed that FA values for 14 tracts were significantly correlated with PNT scores (partial r: 0.42-0.57; FDR corrected with q < 0.05; Table 3, Figure 3A), controlling for age, gender, education level, handedness, time from stroke, and total lesion size. Among the identified tracts, 13 were left hemisphere frontoparietal, frontal-temporal, and intratemporal connections, including 3 dorsal stream tracts and 10 ventral stream tracts; 1 tract was an interhemispheric connection (Table 3, Figure 3B). No significant relationships were found between PNT scores and FA values of tracts in the right hemisphere. These results indicate that naming outcomes primarily rely on the residual white matter network in the left hemisphere in chronic left hemisphere stroke survivors.
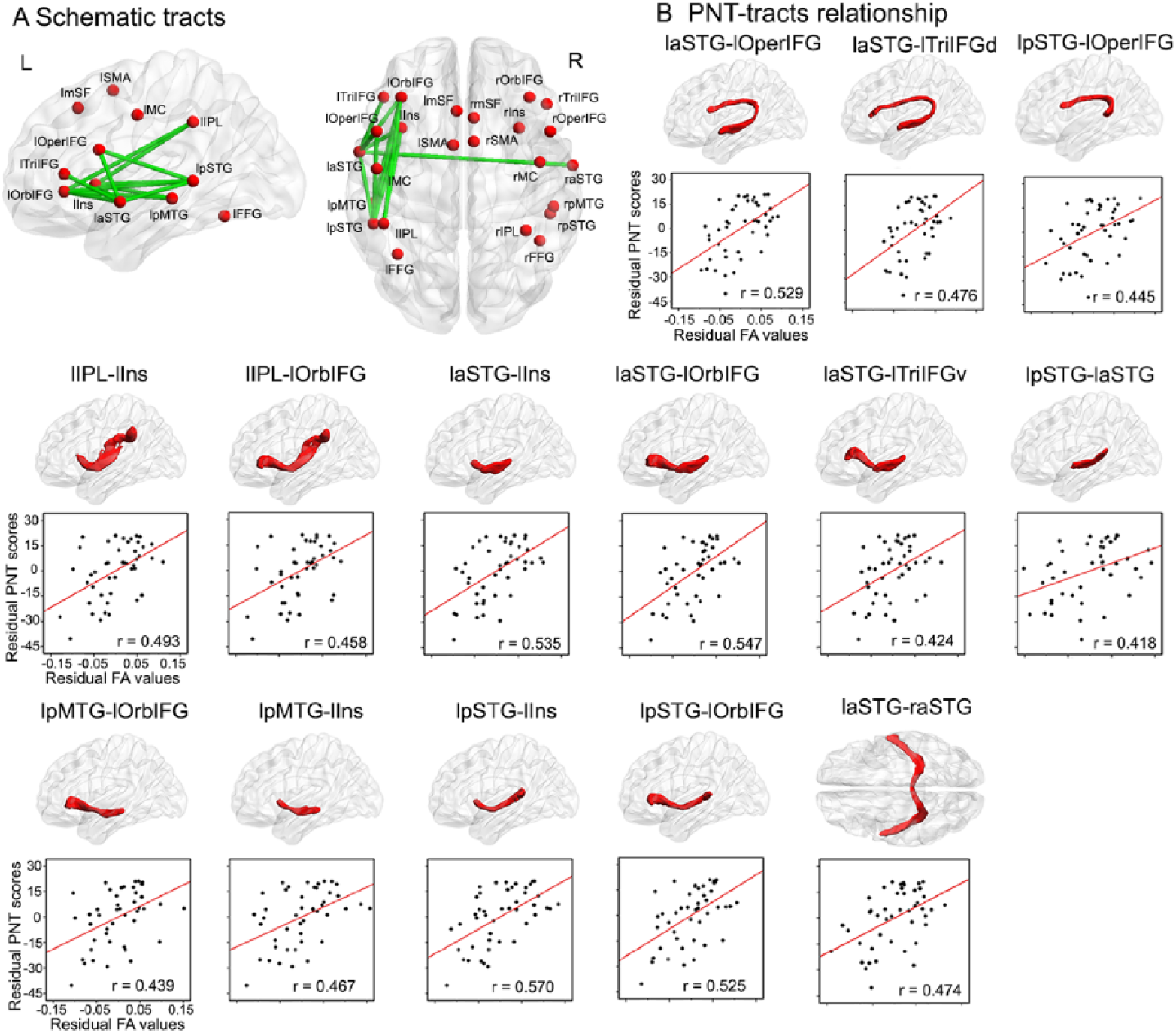
Relationships between the mean fractional anisotropy (FA) of reconstructed white matter tracts and overall naming performance in left hemisphere stroke survivors. (A) Schematic of the reconstructed white matter tracts related to overall picture naming performance among 45 patients. (B) The partial correlations of the mean FA values of 14 white matter tracts with picture naming performance. The upper column shows the corresponding white matter structures, and the lower column shows the respective partial correlations of FA with picture naming scores controlling for age, gender, level of education, handedness, time from stroke onset, and total lesion volumes (q < 0.05 with false discovery rate correction). Full names of seeds and tracts are listed in Tables 2 and 3.
Impact of Lesions to White Matter Pathways on Specific Naming Subprocesses
To clarify the contribution of the tracts implicated in naming above to specific components of naming processing, separate partial correlation analyses were performed to test the relationship of the integrity of the 14 tracts identified above with measures of phonology (PWR scores) and semantics (PPT scores) in patients. When controlling for the same variables as above, the mean FA values of the lpSTG-lOperIFG tract significantly related to PWR performance (partial r = 0.48, FDR corrected with q < 0.05; Figures 4A and 4B). PPT scores were significantly correlated with the mean FA values of 5 left hemispheric tracts after FDR correction (Figures 4A and 4C): laSTG-lOrbIFG (partial r = 0.43; q < 0.05), laSTG-lTriIFG (partial r = 0.47; q < 0.05), laIPL-lIns (partial r = 0.50; q < 0.01), lIPL-lOrbIFG (partial r = 0.51; q < 0.01), and lpSTG-lOrbIFG (partial r = 0.46; q < 0.05).
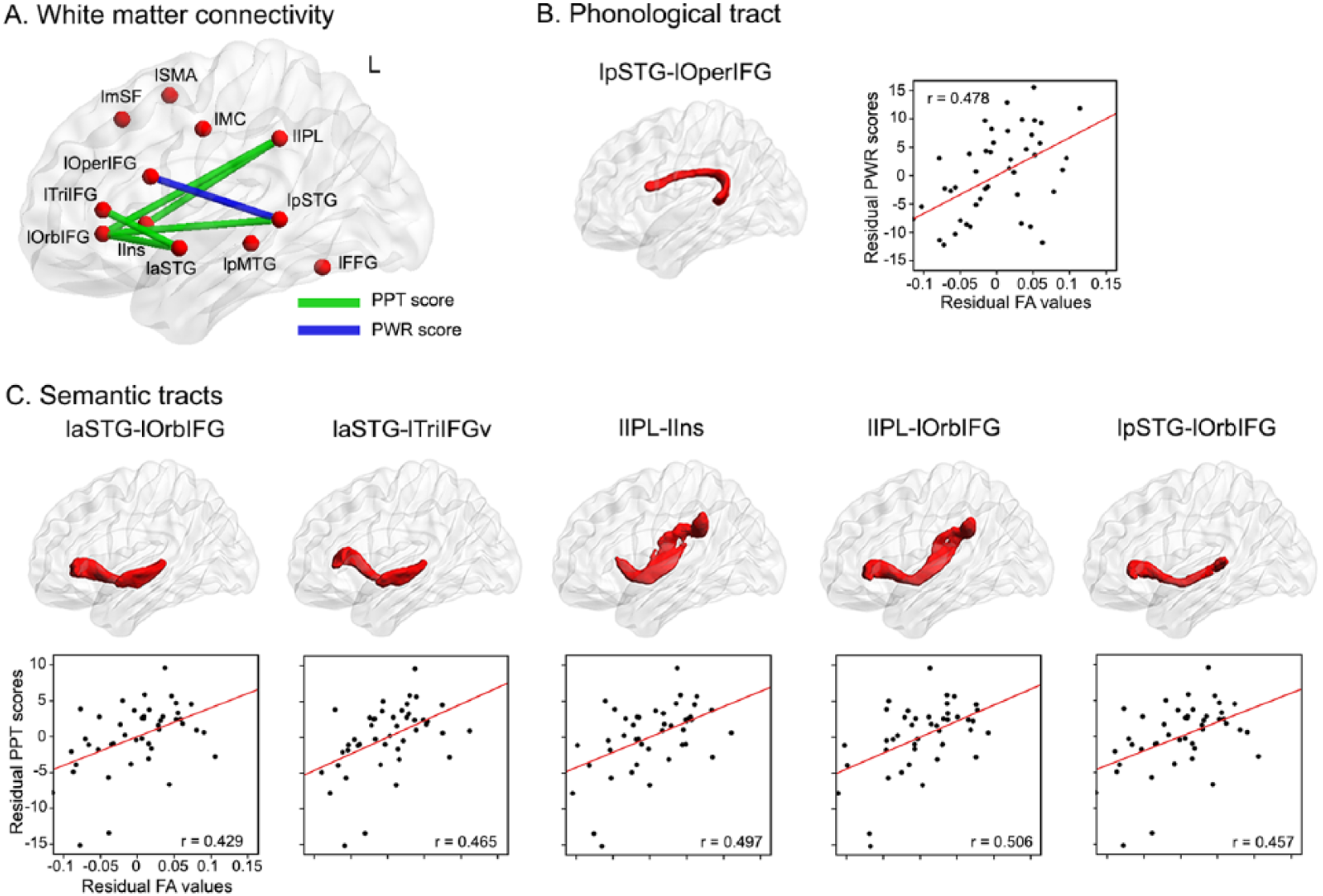
Results of analyses relating specific naming processing to the mean FA of white matter tracts in left hemisphere stroke survivors. (A) Schematic of the white matter tracts specifically associated with phonological and semantic performance in patients. The white matter structures and partial correlations of FA with phonological and semantic processing are shown in (B) and (C) controlling for age, gender, level of education, handedness, time from stroke onset, and total lesion volumes (q <0.05 with false discovery rate correction). Full names of seeds and tracts are listed in Tables 2 and 3.
Finally, we considered whether the observed relationships between these white matter tracts and either phonology or semantics were specific to these processes. There was no significant relationship between PWR and PPT scores controlling for the above variables (partial r = 0.01; P > .05), indicating the independence of these 2 measures. We then reperformed the partial correlations above adding the PPT as an additional covariate in the analyses examining relationships between white matter tracts and PWR and adding PWR as a covariate for the analyses examining PPT. When controlling PPT scores in addition to other covariates, the relationships between lpSTG-lOperIFG tract integrity and phonological performance remained significant (partial r = 0.47, FDR corrected with q < 0.05). Likewise, when partialling out PWR scores, the partial correlates of the 5 semantic processing-related tracts were still significant (partialr: 0.48-0.52; FDR corrected with q < 0.05). These results suggest that different white matter connections in the normal naming network uniquely support phonological and semantic processing.
Discussion
By defining a naming-specific connectome based on fMRI and DTI data in normal adults, we have delineated a network crucial for picture naming and found that naming performance in left hemisphere stroke survivors depends on specific white matter tracts between gray matter nodes in this network. In contrast, damage in specific gray matter nodes of the network did not relate significantly with naming performance after accounting for total lesion size and other individual differences. Finally, connections in the dorsal and ventral streams related to phonological and semantic processing abilities, respectively. These findings suggest the causal roles of white matter structures that functionally support distinct components of naming subprocesses.
Some of the early aphasiologists emphasized the importance of white matter connections for proper speech and comprehension. 31 Karl Wernicke 32 highlighted the importance of a connection between the motor and sensory areas and asserted that damage to that connection results in conduction aphasia, a deficit characterized by relatively intact fluency and comprehension but poor repetition.32-34 Although damage to the arcuate fasiculus, the dorsal connection between the temporal and frontal cortices, may not be the primary cause of conduction aphasia, 35 this connectivist view lives on in many ways. Current models emphasize the role of multiple white matter streams between sensory and motor areas for proper phonological and semantic processing of speech.4,6
Our work builds on this history of relating language deficits to damaged connections. We demonstrate that naming performance depends on long-range connections between the temporal, inferior parietal, and inferior frontal cortices. This finding fits in with a number of studies reporting that severe aphasia is associated with deep white matter lesions.11,36 Some of these studies, like our own, emphasize the contribution of white matter for naming performance over gray matter. For instance, Bonilha et al 11 examined connections between BA 22, 37, 44, and 45 and found that naming performance depended on white matter fibers going to BA 45, even after controlling for necrosis in that area. We add that this relationship between white matter integrity and naming persists after controlling for total lesion volume, whereas the correlation between lesion loads on gray matter and naming disappears after lesion volume is controlled. Furthermore, our study extends the prior literature by identifying additional white matter pathways implicated in naming ability after stroke.
If a cortical region is strongly activated during naming in healthy individuals, why might lesions of that region not cause naming deficits? First, it is possible that lesion status is not as sensitive a measure as tract integrity, and perhaps, a behavioral effect of lesions in gray matter nodes would be observed with a larger sample size. Second, cortical plasticity might account for the negative finding in the gray matter. Recruitment of perilesional cortical areas and possibly homotopic right hemisphere regions can compensate to some degree for damage in primary language regions.20,37 Alternatively, perhaps, no specific cortical node is truly necessary for naming, in that information flow through the network may be diverted through other nodes when one is damaged. Perhaps, then, only damage to combinations of nodes or to the connections between nodes causes severe, lasting deficits.
Naming performance depends on both phonological and semantic processing.18,38,39 As expected, our naming connectome includes pathways in the dorsal and ventral streams thought to be responsible for phonological and semantic processing.6,40 Furthermore, we find that naming performance in stroke survivors depends on tracts in both these streams. Although it would have been informative to test the relationship of dorsal and ventral pathways to phonological and semantic paraphasias, robust error analyses require a far greater number of naming items than the 60 used here. We, therefore, examined associations between the dorsal and ventral pathways implicated in naming and performance on classic tests of phonology and semantics. When these streams were tested for their canonical roles, the FA values in certain connections within the dorsal and ventral streams correlated with performance on tasks of phonological and semantic processing, respectively. Given the multiple operations involved in naming, and the other processes involved in the semantic or phonological measures used here, these measures may not perfectly reflect the specific deficits leading to anomia in the patients. Ideally, multiple measurements along with assessment of paraphasias would help probe varied domains of impairment in patients. Still, our findings confirm the canonical dual stream model and demonstrate that specific pathways in both streams are necessary for naming.
It should be noted that the naming activity elicited by our fMRI task in healthy adults was bilateral, suggesting possible right hemisphere contributions to naming. Recent transcranial magnetic stimulation studies have demonstrated that inhibition of right hemisphere language network homotopes can disrupt phonological processing41,42 and naming in adults. 43 These studies suggest that the right hemisphere activity observed in our functional imaging data may be causally important for naming performance. In a lesion study, a cohort of right hemisphere stroke survivors would be needed to sensitively address this issue. However, some recent DTI studies of left hemisphere stroke survivors have found that white matter pathways in the right hemisphere contribute to aphasia recovery.44,45 We, therefore, examined the integrity of the right hemisphere pathways in the naming connectome in our left hemisphere stroke survivors but found no significant relationships between right hemisphere white matter and naming outcomes. The prior studies addressing this issue examined major right hemisphere white matter fasciculi as a whole, whereas we examined more specific connections between network nodes, perhaps explaining the negative result here. Notably, we did identify 1 interhemispheric connection (left aSTG–right aSTG) that was associated with naming ability, indicating a potential contribution of interhemispheric connectivity to naming, if not intrahemispheric processing within the right hemisphere.
One limitation of the present study is that white matter pathways were not reconstructed with tractography in individual patients. Rather, tracts were reconstructed in a group of healthy controls and then used to test mean FA of these pathways in patients. Similar strategies to measuring tract integrity have been used previously in patient populations,10,46 but under this circumstance, the tractography results in healthy individuals may not directly correspond to true white matter connections in stroke patients, especially in the lesioned hemisphere. 47 This is because errors in spatial normalization may lead to imperfect alignment of patient FA maps with the tracts, and the FA may relate partially to fibers from other nearby pathways that travel with the fibers connecting the naming nodes or may relate partially to crossing fibers. Therefore, additional work will be useful to confirm these results using direct point-to-point connectivity measures in patients.
Conclusions
We reconstructed a structural network consisting of white matter tracts between areas that activate in healthy individuals during naming and demonstrated that naming performance after chronic left hemisphere stroke depends on the integrity of specific tracts within this network. These findings do not provide evidence that the exact areas activated in response to naming in healthy individuals are functionally necessary for performance, at least in the chronic phase of stroke recovery, but demonstrate that the white matter between these areas is crucial. Our findings bridge 2 disparate literatures on brain networks for naming using fMRI in healthy participants and lesion analysis in stroke survivors and extend our current knowledge about the neural basis of naming processing by identifying regions of white matter that are essential for naming ability.
Supplemental Material
Supplemental_Table – Supplemental material for Behavioral Effects of Chronic Gray and White Matter Stroke Lesions in a Functionally Defined Connectome for Naming
Supplemental material, Supplemental_Table for Behavioral Effects of Chronic Gray and White Matter Stroke Lesions in a Functionally Defined Connectome for Naming by Shihui Xing, Ayan Mandal, Elizabeth H. Lacey, Laura M. Skipper-Kallal, Jinsheng Zeng and Peter E. Turkeltaub in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We thank Katherine Spiegel, Mackenzie E. Fama, Rachael Harrington, Alexa Desko, Lauren Taylor, Laura Hussey, Jessica Friedman, and Molly Stamp for contributing to data collection and our participants for their involvement in the study.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Center for Advancing Translational Sciences via the Georgetown-Howard Universities Center for Clinical and Translational Science (KL2TR000102), the Doris Duke Charitable Foundation (Grant 2012062), NIDCD (R01DC014960), and the Vernon Family Trust (donation to PET), the National Key R&D Program of China (2017YFC1307500), the National Natural Science Foundation of China (81000500), the Natural Science Foundation of Guangdong Province of China (2015A030313049, 2017A030303011), the Science and Technology Planning Project of Guangdong Province of China (2017A050506024), the Guangdong Provincial Key Laboratory for Diagnosis and Treatment of Major Neurological Diseases (2014B030301035), and the Southern China International Cooperation Base for Early Intervention and Functional Rehabilitation of Neurological Diseases (2015B050501003).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
