Abstract
Background. Due to an increasing stroke incidence, a lack of resources to implement effective rehabilitation and a significant proportion of patients with remaining impairments after treatment, there is a rise in demand for effective and prolonged rehabilitation. Development of self-rehabilitation programs provides an opportunity to meet these increasing demands.
Objective. The primary aim of this meta-analysis was to determine the effect of self-rehabilitation on motor outcomes, in comparison to conventional rehabilitation, among patients with stroke. The secondary aim was to assess the influence of trial location (continent), technology, time since stroke (acute/subacute vs chronic), dose (total training duration > vs ≤ 15 hours), and intervention design (self-rehabilitation in addition/substitution to conventional therapy) on effect of self-rehabilitation.
Methods. Studies were selected if participants were adults with stroke; the intervention consisted of a self-rehabilitation program defined as a tailored program where for most of the time, the patient performed rehabilitation exercises independently; the control group received conventional therapy; outcomes included motor function and activity; and the study was a randomized controlled trial with a PEDro score ≥5.
Results. Thirty-five trials were selected (2225 participants) and included in quantitative synthesis regarding motor outcomes. Trials had a median PEDro Score of 7 [6–8]. Self-rehabilitation programs were shown to be as effective as conventional therapy. Trial location, use of technology, stroke stage, and intervention design did not appear to have a significant influence on outcomes.
Conclusion. This meta-analysis showed low to moderate evidence that self-rehabilitation and conventional therapy efficacy was equally valuable for post-stroke motor function and activity.
Introduction
Each year, approximately 1.1 million Europeans suffer a stroke and in 2020, there was an estimated 9.5 million stroke survivors in Europe.1,2 By 2030, stroke prevalence is expected to rise by 35%. 3 Almost 80% of these patients present motor impairments and for half, these impairments will remain permanently.4,5 This leads to activity limitation, social participation restriction, and affects quality of life. Therefore, there is an urgent need to improve stroke outcome by providing new additional and prolonged neurological rehabilitation methods. Currently, a large proportion of patients does not achieve the recommended amount of physical activity (30 min per day) and do not spend sufficient time on rehabilitation.6,7 Hospital resources and healthcare systems are often limited. 8 This has led to the development of new cost-effective rehabilitation methods such as self-rehabilitation.9-11
In this review, we define self-rehabilitation as a tailored therapy program where for most of the time, the patient performs rehabilitation exercises independently to the presence of a clinician. 12 This definition of rehabilitation also includes clinical remote support that can be provided with technology and communication methods through video or written instructions. Self-rehabilitation programs can be delivered at hospital, in rehabilitation centers, in physical therapy firms, and at home. Examples of self-rehabilitation include home-based self-guided therapies, unsupervised technologically assisted rehabilitation, and some telerehabilitation programs where patients perform exercises independently to the presence of a clinician.
To date, only one published systematic review has assessed the effects of self-rehabilitation on stroke. 13 Results showed that self-rehabilitation, which did not involve technology, appeared to be more effective than conventional therapy for arm function. However, this article assessed trials whose interventions were exclusively meant to rehabilitate upper limb motor function, rather than using the World Health Organization’s International Classification of Function, Disability, and Health (ICF-WHO) model to classify self-rehabilitation outcomes.14-16 Furthermore, few of the studies included in the review compared self-rehabilitation to conventional therapy. Since the publication of this review, several new randomized controlled trials (RCT) have emerged.
Another alternative cost-effective rehabilitation method that overlaps with self-rehabilitation is called telerehabilitation. Telerehabilitation can be defined as “the delivery of rehabilitation services via information and communication technologies.” 17 These programs of rehabilitation are often supervised by a therapist and conducted in the patient’s home through telecommunication devices (mobile phones, motion-capture systems, computers based communication, etc). 18 Telerehabilitation effects on post-stroke motor impairments remain promising, with several meta-analyses of RCTs showing similar results for telerehabilitation and conventional therapy for motor outcomes.19-22
There are several unknown factors that may influence the efficacy of self-rehabilitation on adults with stroke. First, outcomes may change depending on the location of the trial. There is evidence that race and ethnicity influence stroke incidence and recovery 23 and access to clinics can influence the intensity of rehabilitation. 24 Second, the implementation of new technologies for post-stroke self-rehabilitation may influence outcomes. Several meta-analyses have highlighted a superiority of virtual reality and robot-assisted therapy compared to conventional therapy when provided under clinical supervision.25,26 Third, the influence of delay since stroke, dose and design of intervention (in addition or in substitution to conventional therapy) on self-rehabilitation remains unknown. Lastly, the implementation of strategies to improve adherence to treatment may also influence self-rehabilitation outcomes. 27
The primary aim of this meta-analysis was to compare the effect of self-rehabilitation with conventional therapy on motor outcome and activity among patients with stroke. We included both upper and lower limb motor function and activity according to the ICF-WHO model. 14 The secondary aim was to determine the influence of trial location (continent), use of technology, stroke stage (acute/subacute vs chronic), and dose or design of the intervention (self-rehabilitation in addition or in substitution to conventional therapy) on motor function and activity.
Methods
This systematic review was performed according to the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) (Supplementary Materials 1). 28 The protocol was registered with Prospero reference CRD42020188857.
Literature Search
Search strategies were made according to the PICO format for the databases Pubmed, Embase, Cochrane Library, and Scopus (Supplementary Material 2). In addition, author hand-searching was made by examination of the grey literature (Google Scholar and PEDro) and by checking the literature citations of relevant systematic reviews.
Eligibility Criteria
Trials were included if all the following PICOs criteria were fulfilled: (P) participants were post-stroke adults (aged > 18 years; ischemic or hemorrhagic stroke; no underlying neurological disease, no restriction for stroke stage); (I) the intervention consisted of a motor self-rehabilitation program (tailored therapy program where for most of the time, the patient performs rehabilitation exercises independently to the presence of a clinician) designed for upper and/or lower limb recovery and was conducted in addition to or in substitution of conventional therapy (the treatment could be provided in the home or at the hospital, in partial or complete autonomy); (C) the control group received post-stroke conventional therapy; (O) relevant outcomes were measured to assess the ICF-WHO motor function or activity components and; (S) the study followed a RCT design, was written in English, French, or Spanish, was published before December 19th, 2020, and had a PEDro score superior or equal to 5.
Selection Process
All references issued from databases were exported into a reference management software (EndNote X9). After disposal of duplicates, a first selection based on titles and abstracts was conducted. Trials were retrieved and sorted by two independent investigators (authors G.E. and A.L.) according to the eligibility criteria. At the same time, hand-searching results were stored in a spreadsheet software. All full-text articles were then screened and selected following the same procedure. Lastly, a meeting was organized between the two reviewers to compare selection. For articles not selected by both investigators, a consensus was found through discussion.
Design Considerations to Improve Adherence to Self-Rehabilitation
Studies were assessed to determine whether their treatment intervention fulfilled considerations that improved adherence to self-rehabilitation. Based on the research reported by Argent et al., 2018, 27 five main considerations were extracted: the provisioning of real time sessions (face-to-face sessions or videoconferences), the use of sensing platforms, the provision of visual feedback performance, the definition of quantifiable objectives and the availability of written ,or video-recorded instructions. A consideration was defined to be fulfilled only when the study explicitly mentioned it.
Data Extraction
Prior to data extraction, the methodological quality of each study was evaluated using the Cochrane Collaboration’s Risk of Bias and the PEDro checklist. 29 The following characteristics were then collected: country in which the trial was conducted, number of participants, patient’s mean age and stroke stage (acute: <7 days after stroke, subacute: 7 days to 6 mo after stroke and chronic: >6 mo after stroke), 30 type and duration of the therapy for both intervention and control groups, adherence to self-rehabilitation (measured by the time spent performing exercises or the amount of patients that reached the number of recommended sessions), type of outcome measures according to the ICF-WHO framework and mean improvements (with standard deviations). For body function: the Fugl–Meyer, modified Ashworth Scale, Chedocke Arm and Hand Inventory, grip strength, Hand Function subscore of the Stroke Impact Scale and kinematic data such as stability indicators were considered. For activity limitation: the Action Research Arm Test, Box and Block Test, Wolf Motor Function Test, Barthel Index, Functional Independence Measure, Nine Hole Peg Test, EQ-5D, 10 Meter Walking Test, 6 Minutes Walking Test, 2 Minutes Walking Test, Berg Balance Scale, and Timed Up and Go were considered. For participation, no information was extracted since too few studies measured it.31,32 When the information was not found in the full-text, authors were contacted.
Statistical Analysis
All studies whose motor outcomes were available and measured according to the ICF-WHO framework were considered for quantitative synthesis. Mean improvements were calculated from the pre-post intervention score differences. When several time-points were mentioned, the latest-one was retained for the analysis. Related standard deviations were then estimated according to the Cochrane Handbook. If a study reported standard error values, standard deviations values were computed by multiplying the standard error values by the square root of the sample size within a group.
The RevMan 5.3 software was used to generate forest plots from trials included in the meta-analysis. Given that the scales used to assess outcomes were very heterogeneous, a standardized mean difference (SMD), and 95% confidence interval was calculated for each study. The SMD represents the ratio between the difference of mean effect between groups and the pooled standard deviation of the effect among participants.
Heterogeneity between the different trials was estimated using the I2 test. As suggested by the Cochrane Handbook, heterogeneity was rated as non-significant for I2 < 30%, moderate for a I2 between 30% and 50%, substantial for a I2 between 50% and 75% and considerable for I2 > 75%. In case of heterogeneity, a further analysis was done using a random-effect model. Publication bias was evaluated visually with funnel plot graphic representation. In case of funnel plot asymmetry or presence of outliers, a sensitivity analysis was conducted. The GRADEpro approach was used to assess the strength of the body evidence. This method enables a level of certainty regarding the overall results by taking into consideration the number of studies included in the analysis, the global study design, the risk of bias assessment, and the heterogeneity, directness, precision, and standard mean difference of the results.
Subgroup analyses were conducted to see whether the location of the trial, the use of technology, the stroke stage (acute/subacute vs chronic), the total training duration (superior vs inferior or equal to 15 h), and the design of the intervention (self-rehabilitation in addition or in substitution to conventional therapy) had an influence on outcome regarding the activity component of ICF-WHO framework. Additional subgroup analyses were performed to test difference between upper and lower limb self-rehabilitation effects on motor function and activity. Programs were not considered to be technologically assisted when the technology was only used to encode data, play video instructions or contact the therapist.
Results
The search identified a total of 9293 records. After removing duplicates, 4661 articles were screened based on their titles and abstracts. This resulted in the inclusion of 306 studies for full-text screening. Among these, 34 trials9,10,33-64 were retained for the qualitative synthesis and 30 were selected for the meta-analysis. A flow chart diagram illustrating the selection process is presented in Figure 1. Selection process represented through a flow chart diagram.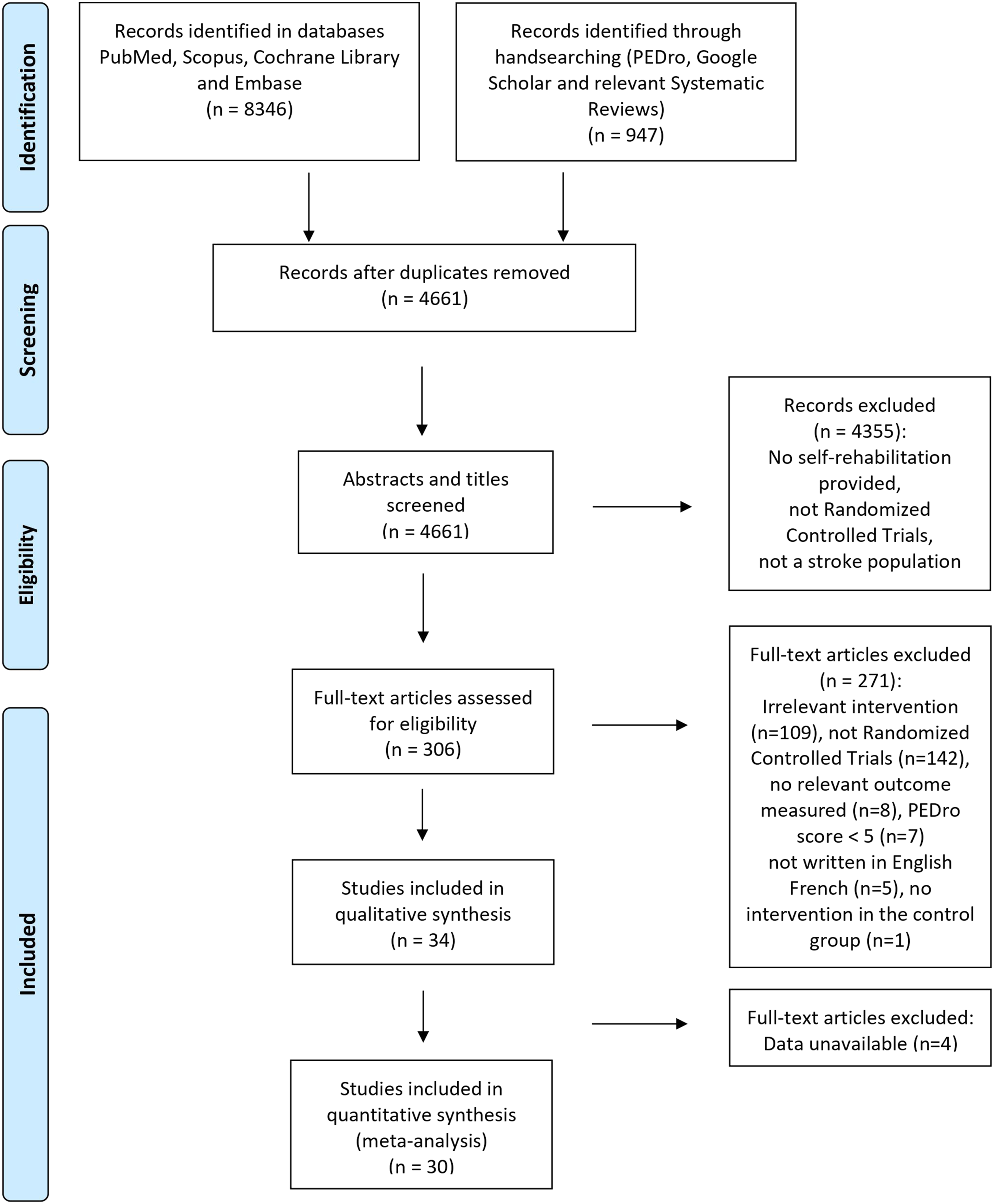
Methodological Quality of the Included Studies
All randomized controlled trials (RCTs) were assessed with the PEDro checklist and Cochrane Collaboration Risk of Bias (Supplementary Materials 3 and 4). Trials had a median PEDro Score of 6.5 [6–8] and 85% of these were superior or equal to 6. The Cochrane Collaboration Risk of Bias assessment revealed that twenty-three RCTs (67%) reported the blinding of assessors, one RCT (3%) the blinding of participants and no RCTs the blinding of therapists. Among trials included in qualitative analysis, half declared using concealed allocation during the randomization process.
Study Characteristics
Characteristics of Included Trials.
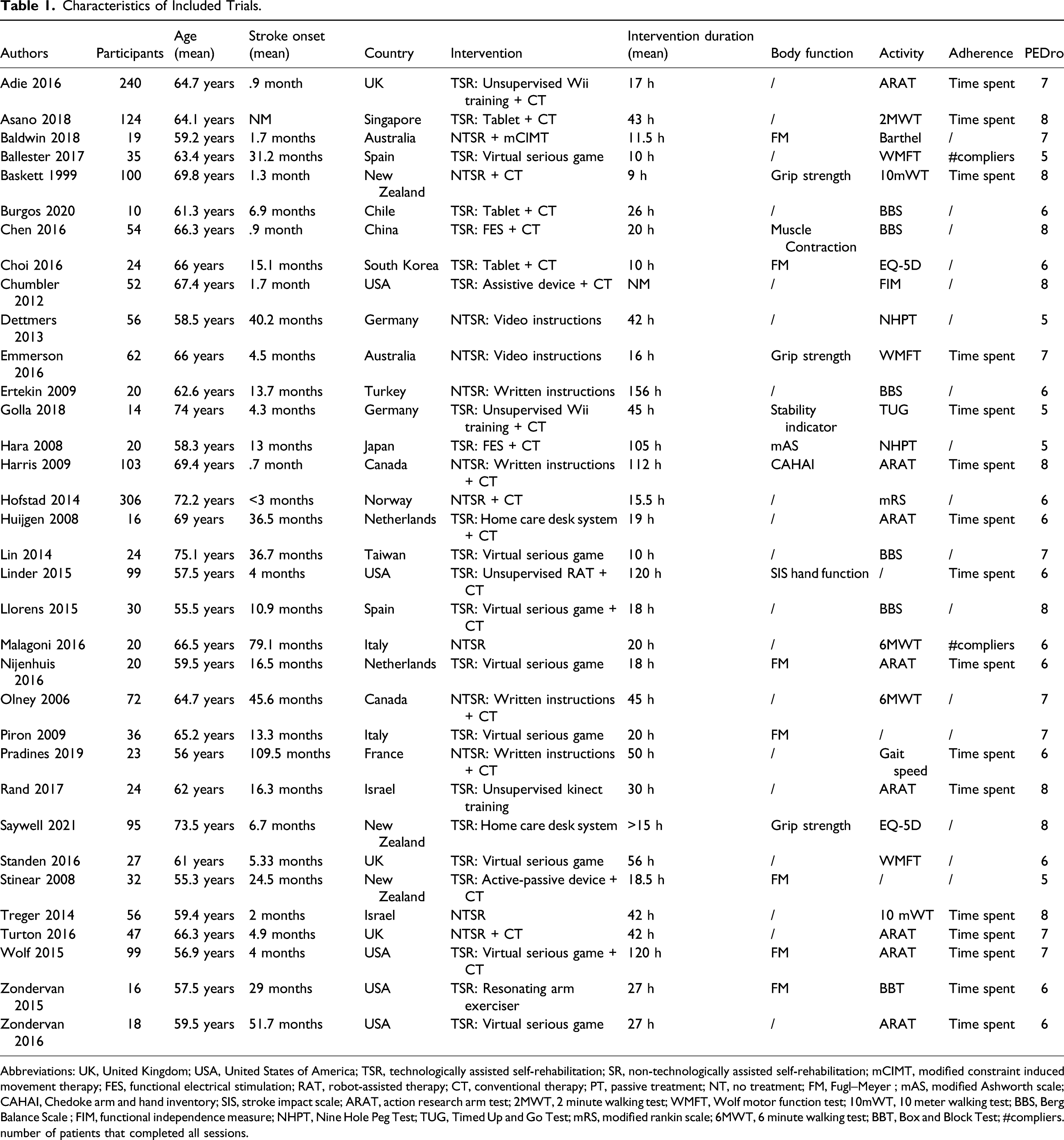
Abbreviations: UK, United Kingdom; USA, United States of America; TSR, technologically assisted self-rehabilitation; SR, non-technologically assisted self-rehabilitation; mCIMT, modified constraint induced movement therapy; FES, functional electrical stimulation; RAT, robot-assisted therapy; CT, conventional therapy; PT, passive treatment; NT, no treatment; FM, Fugl–Meyer ; mAS, modified Ashworth scale; CAHAI, Chedoke arm and hand inventory; SIS, stroke impact scale; ARAT, action research arm test; 2MWT, 2 minute walking test; WMFT, Wolf motor function test; 10mWT, 10 meter walking test; BBS, Berg Balance Scale ; FIM, functional independence measure; NHPT, Nine Hole Peg Test; TUG, Timed Up and Go Test; mRS, modified rankin scale; 6MWT, 6 minute walking test; BBT, Box and Block Test; #compliers. number of patients that completed all sessions.
Technologically assisted self-rehabilitation was provided in 23 RCTs (63%). Among these, fourteen (60%) focused on upper limb recovery, six (27%) on lower limb recovery, and three (13%) on full body recovery. Devices were tablets, computers, robots, non-immersive virtual reality systems, and functional electrical stimulation machines. In 20 RCTs, participants were instructed to practice their exercise at home according to therapist instructions. Non-technologically assisted self-rehabilitation was prescribed in twelve RCTs (35%). Among these, six (50%) focused on upper limb recovery, two (17%) on lower limb recovery, and four (33%) on full body recovery. Different types of therapy were provided: balance and walking programs, bimanual exercises, functional reaching tasks, functional grasping tasks, and unsupervised constraint-induced movement therapy. In twelve RCTs, participants were instructed to practice their exercise at home according to written or video guidance. Among the trials, 7 (58%) supplemented their intervention with conventional therapy. The total intervention dose ranged from 9 to 156 h with a mean duration of 40.5 ± 38.2 h and a total intervention duration from 2 weeks to 1 y. Twenty-nine trials (83%) provided interventions with a total duration superior or equal to 15 h, five (14%) provided less than 15 h of intervention, and one (3%) study was not precise enough to determine the total hours of intervention.
Effect of Self-Rehabilitation on Motor Function
Fifteen trials (44%) measured motor function according to the first ICF component. Among these, thirteen reported quantitative outcomes; eleven regarding upper limb, one regarding lower limb, and one regarding full body. Upper limb motor function assessments consisted of measuring control of voluntary movements (movement quality), muscle power (grip strength and lower limb muscular contraction), and motor reflex (spasticity) according to the ICF core sets.
Outcomes of the intervention group improved in 12 trials (92%) and all trials showed an improvement in the control group. As presented in Figure 2, self-rehabilitation showed similar effectiveness to conventional therapy for motor function recovery (SMD = .09; 95% CI = −.11 to .28; P = .39). Subgroup analysis (P = .34) revealed that upper limb self-rehabilitation (SMD = .12; 95% CI = −.1 to .33; P = .29) and lower limb self-rehabilitation (SMD = −.15; 95% CI = −.66 to .36; P = .56) were similarly effective. Self-rehabilitation effect on motor function for upper and lower limb represented through a forest plot.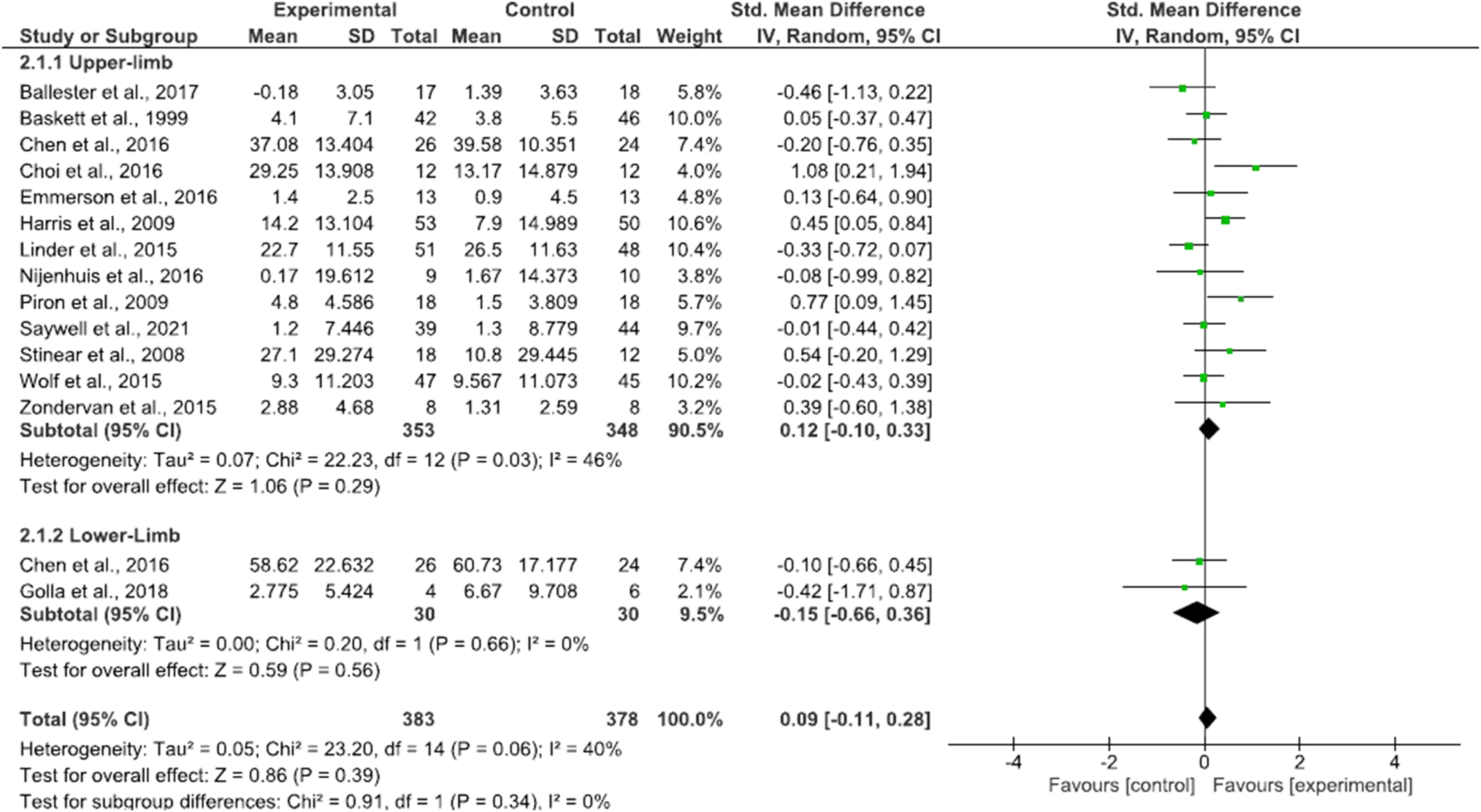
The Grade Approach disclosed a low certainty of evidence regarding self-rehabilitation effect on motor function (Supplementary Material 5) related to the fact that few studies assessed motor function and that heterogeneity between results was moderate (I2 = 40%). The effect size quantified by the SMD was considered as low. 65
Effect of Self-Rehabilitation on Activity
Thirty-one trials measured activity according to the second ICF component. Among these, twenty-six reported quantitative outcomes. Selected outcomes were assessed according to the ICF set scores for hand and arm use (reaching and grasping), walking (gait speed and balance), and mobility. Funnel plot graphic representation revealed asymmetry and the presence of one outlier.
52
Sensitivity analysis after removal of the outlier showed no significant difference (Supplementary Material 6). From the remaining trials, twenty-four (96%) showed an improvement in the intervention group and twenty-one (88%) in the control group. Meta-analysis showed that self-rehabilitation was as effective as conventional therapy to improve activity outcomes (SMD = .08; 95% CI = −.04 to .19; P = .18) (Figure 3). Subgroup analysis (P = .23) showed no superiority of self-rehabilitation using technological devices (SMD = .01; 95% CI = −.14 to .16; P = .88) compared to self-rehabilitation not using technology (SMD = .15; 95% CI = −.01 to .31; P = .07). Technologically assisted and non-technologically assisted self-rehabilitation effect on activity represented through a forest plot.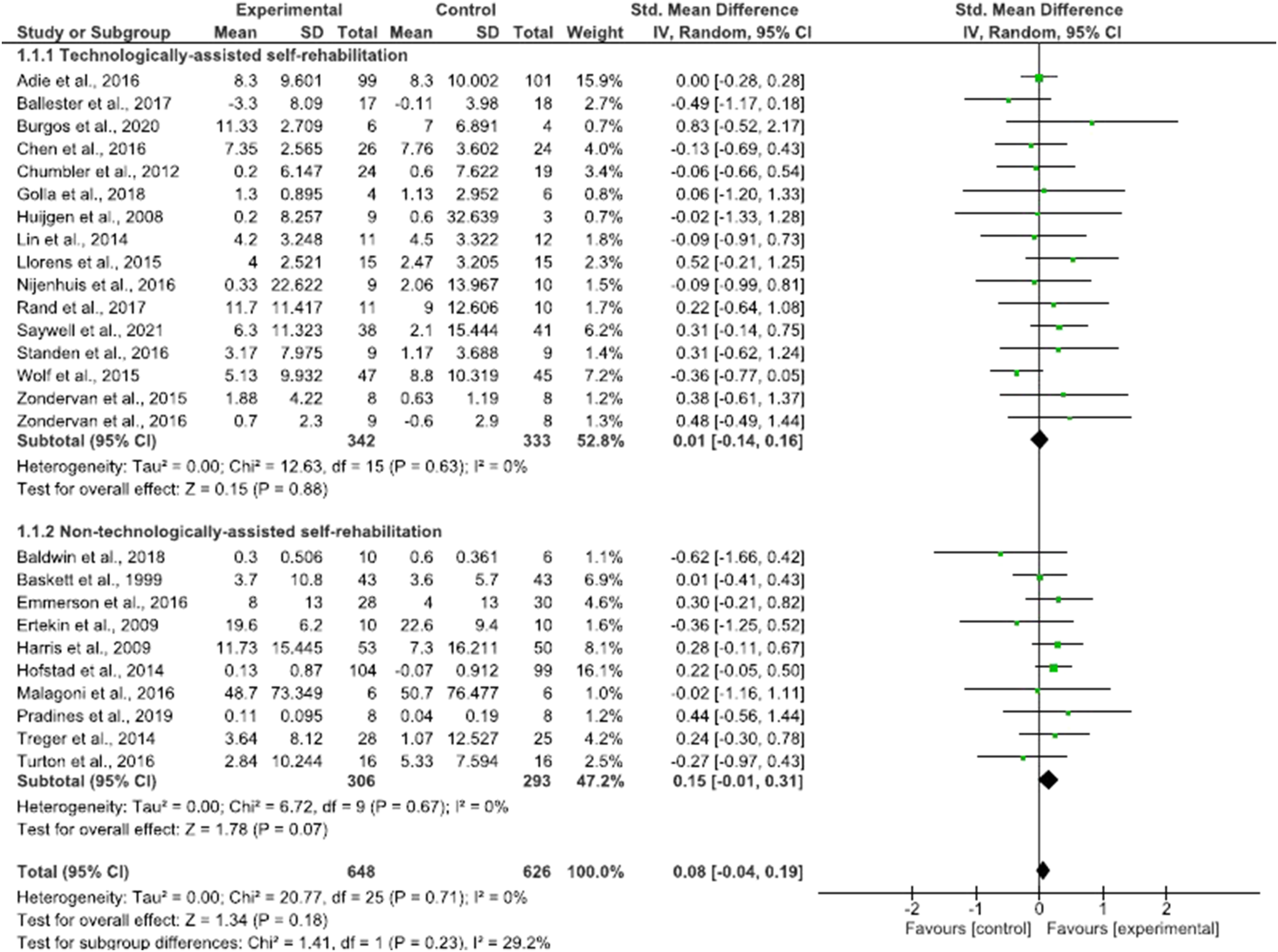
Self-rehabilitation appeared equally effective when delivered in addition to (SMD = .06; 95% CI = −.07 to .19; P = .36) or in substitution of (SMD = .11; 95% CI = −.09 to .31; P = .29) conventional therapy (Figure 4), irrespective of whether patients were in the subacute (SMD = .07; 95% CI = −.06 to .20; P = .27) or chronic stages (SMD = .09; 95% CI = −.14 to .31; P = .44) (Figure 5), and for programs of different duration (SMD = .12; 95% CI = .00 to .24; P = .06) (Figure 6). Sensitivity analysis revealed no significant difference between upper and lower limb self-rehabilitation on activity (Figure 7). Influence of trial location was also found to be non-significant (P = .94) (Supplementary Material 7). Effect of self-rehabilitation in addition or substitution to conventional therapy on activity represented through a forest plot. Effect of self-rehabilitation on acute vs chronic patients with stroke on activity represented through a forest plot. Effect of total duration of self-rehabilitation programs on activity represented through a forest plot. Self-rehabilitation effect on activity for upper and lower limb represented through a forest plot.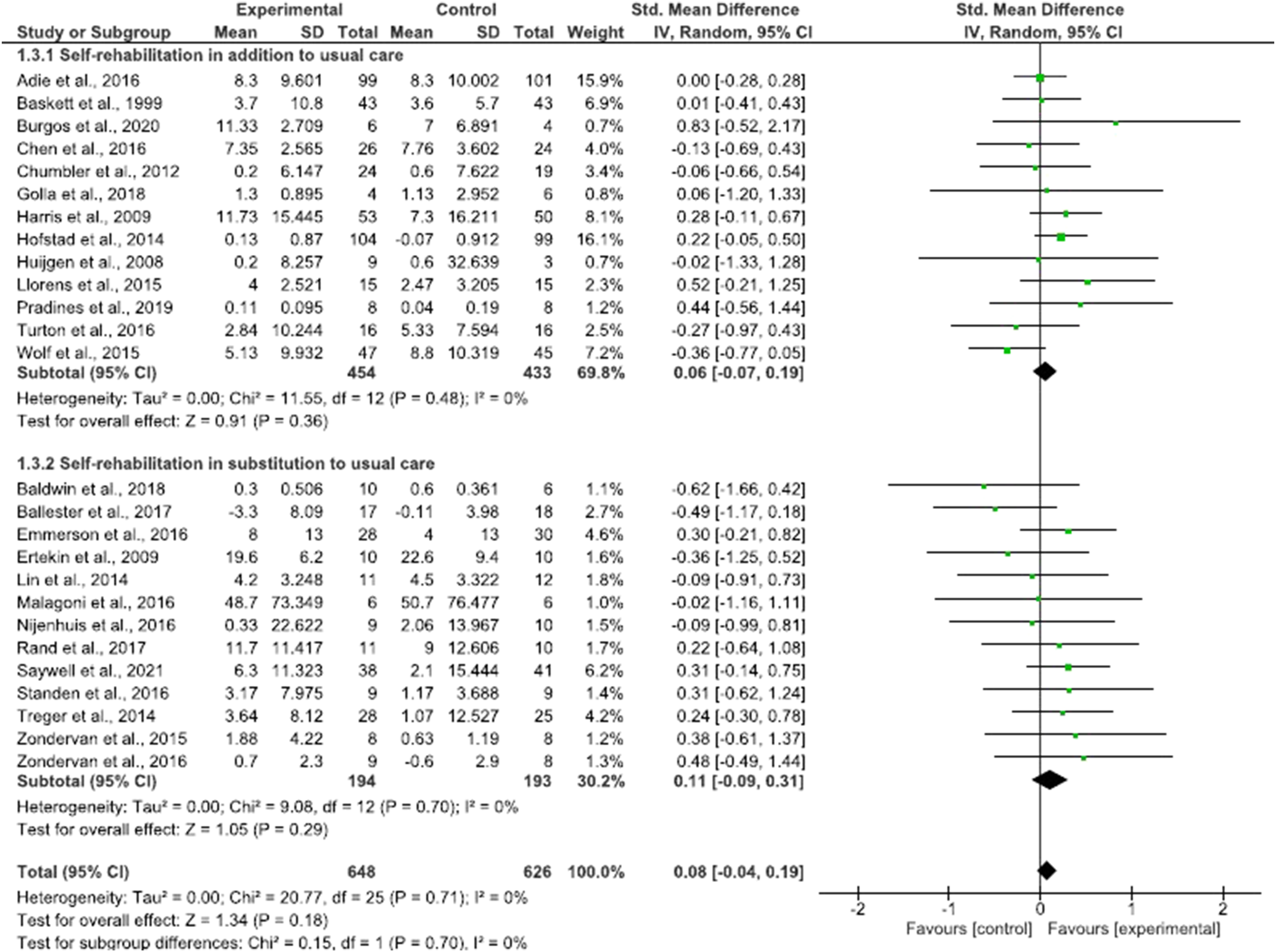
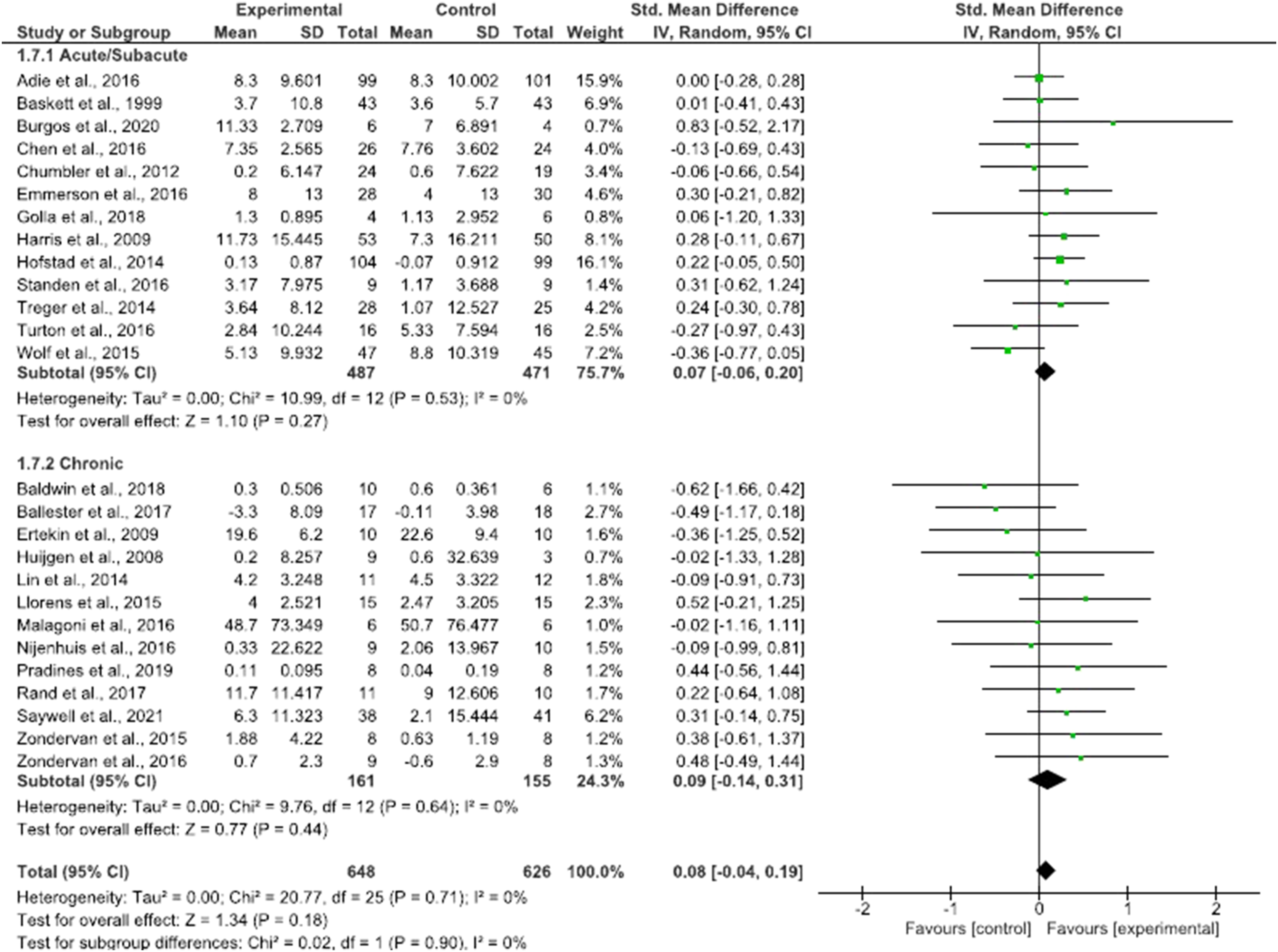
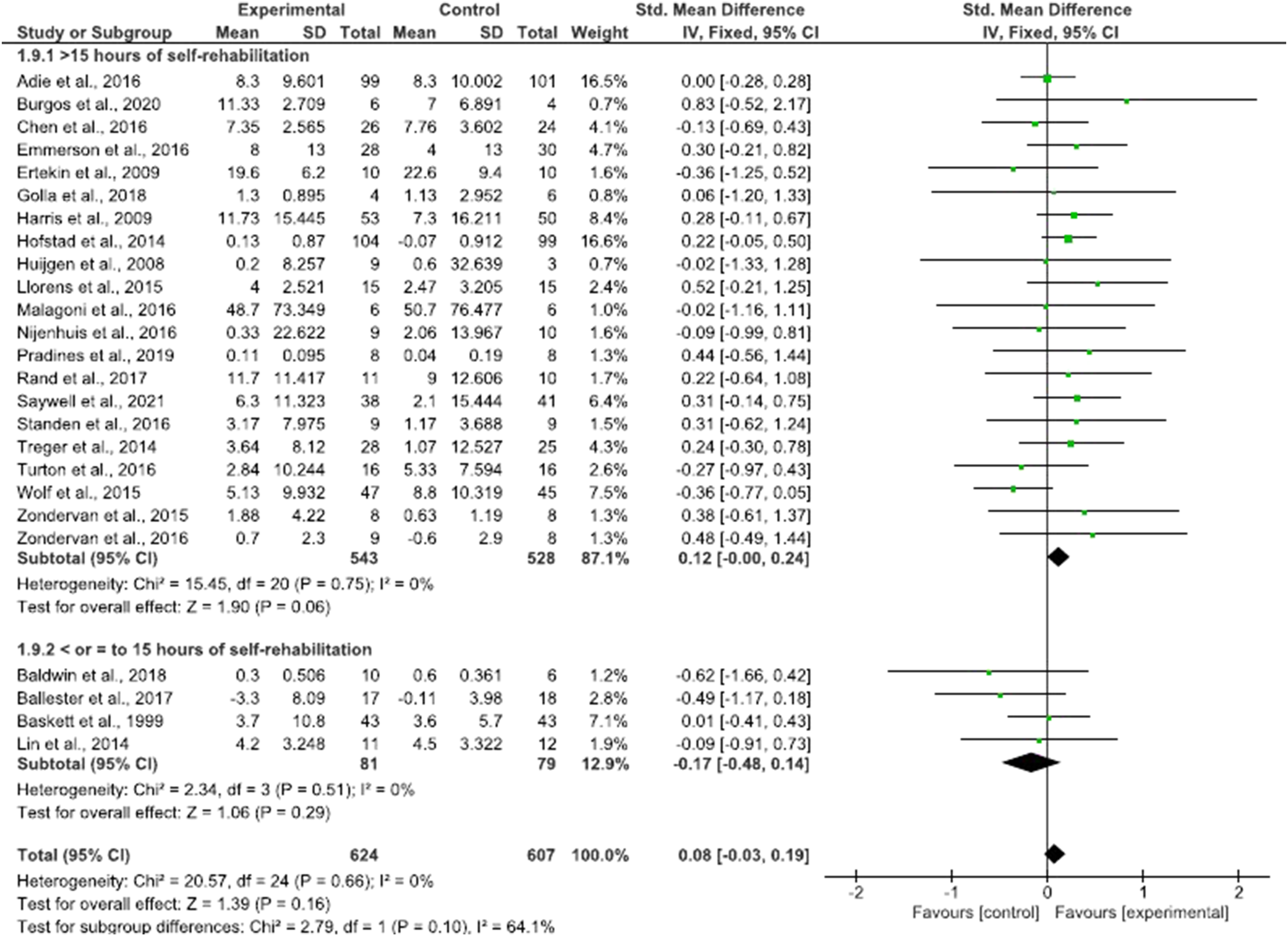
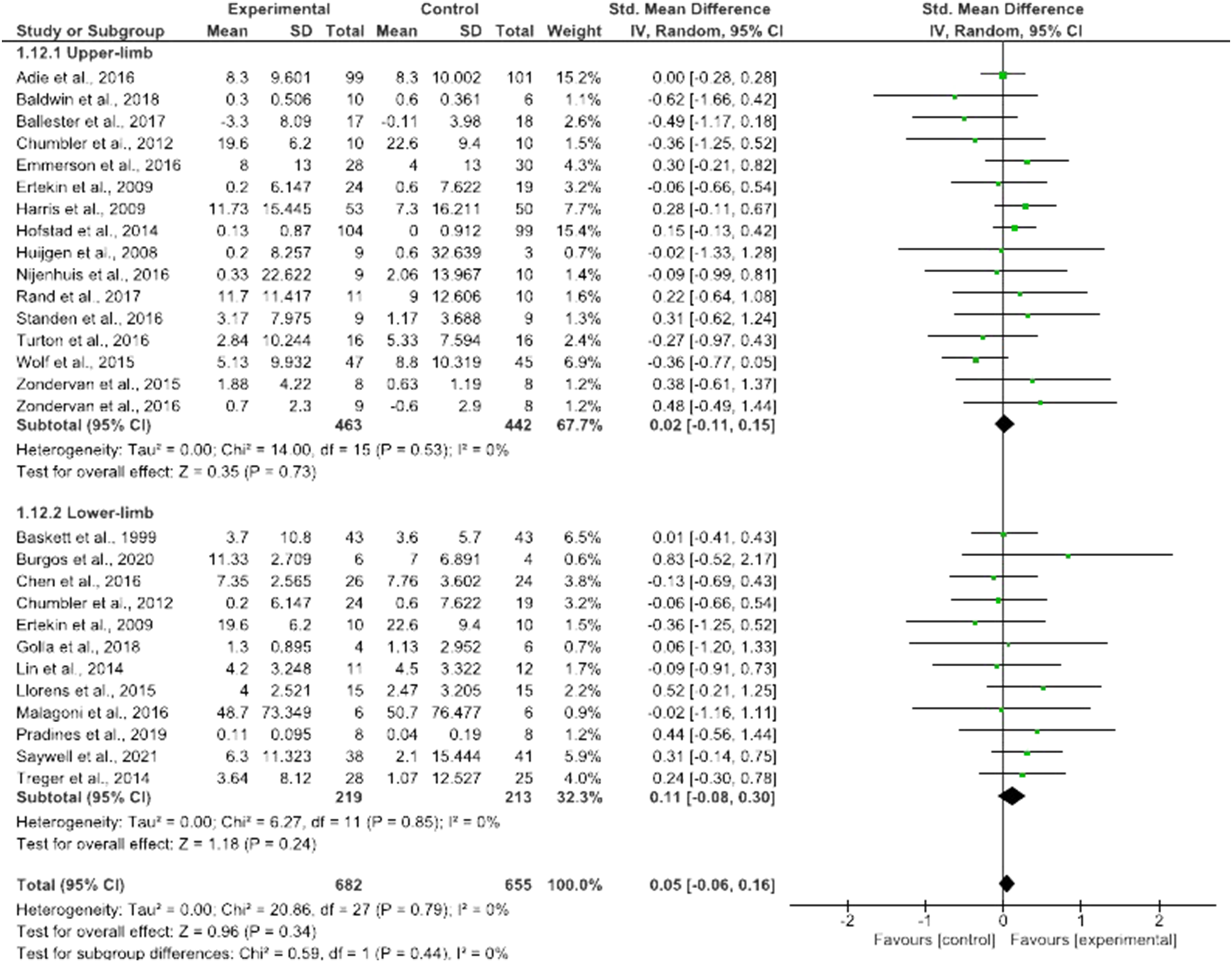
The certainty of evidence regarding the self-rehabilitation effect on activity was qualified as moderate according to the Grade Approach (Supplementary Material 8). Inconsistency, indirectness, and risk of bias were judged to be low for all subgroup analyses. In addition, heterogeneity between studies was found to be non-significant (I2 = 0%). The effect size quantified by the SMD was considered as low. 65
Adherence to Self-Rehabilitation
Eighteen trials reported measures related to treatment adherence. Six trials showed that patients spent the same or more time performing exercises in the intervention than control groups. However, seven trials showed that patients from the intervention group spent less time performing exercises than patients from the control group. Five trials reported that 15 to 40% of patients did not reach the total amount of sessions.
Seventeen RCT (50%) explicitly revealed providing real time sessions, eighteen (53%) used sensing platforms, nine (26%) supplied performance visual feedback, seven (20%) defined clearly measurable objectives, and twenty-two (65%) gave written or video-registered instructions.
Discussion
The present meta-analysis showed low to moderate certainty of evidence that self-rehabilitation efficacy was equally effective to conventional therapy for post-stroke motor function and activity. Most of the included RCTs reported improvements in both intervention and control groups for lower and upper limb outcomes. Subgroup analyses revealed that trial location, use of technology, intervention design, dose and stroke stage classification were all statistically non-significant. The results of this meta-analysis are globally in line with previous reviews that assessed self-rehabilitation 13 or telerehabilitation19-22,66,67 effects on motor function or activity of adults after stroke. In 2018, Da Silva et al., conducted a meta-analysis to assess self-rehabilitation efficacy on post-stroke arm function. 13 Results revealed that constrained-induced movement therapy, electrical stimulation, and non-technological programs were superior to other technological self-rehabilitation programs. Treatment dose and stroke stage were shown to significantly influence post-stroke motor outcomes. However, some of these findings differed from our results. This might be explained by the fact that the review of Da Silva et al., only focused on arm rehabilitation and included non–RCTs (50% studies) and with a high risk of bias.
The 2019 meta-analysis of Rintala et al 21 compared telerehabilitation to other programs that could improve physical functioning after stroke. Results showed that online video monitoring programs, telephone interventions, and DVD exercises were equally effective for daily living activity outcomes. A recent Cochrane review suggested that telerehabilitation could not be considered as inferior to conventional therapy to improve balance, upper limb function, and daily living activity. 19 Two other meta-analyses showed similar results on both upper and lower limb motor function, activity, and participation.20,22 However, none of these papers focused on non-technological interventions.
Parameters That Influence Self-Rehabilitation Effect on Activity
This review showed that technologically assisted compared to non-technologically assisted self-rehabilitation had equal effects on motor function outcome measures relative to control conditions. This neutral finding may be explained by the use of new-technologies on post-stroke outcomes having heterogeneous effects that differ between the types of technologies such as virtual reality vs robot-assisted therapy and the specific methods of each therapy (e.g.,, immersive vs computer screen virtual reality).25,68 For example, a recent meta-analysis showed that virtual reality and serious game implementation led to superior outcomes compared to conventional therapy when provided in hospital. 26 Similarly, another systematic review with meta-analysis revealed that the outcomes of robot-assisted therapy were superior to any other therapy. 25 However, these results are not supported by a recent RCT that did not show significant effects of robot-assisted therapy on upper limb motor function and activity when compared to conventional therapy of the same frequency and duration. 69 For comparison between the present paper and the first two papers reporting superior effects of technology use on outcomes, the absence of supervision from a therapist when patients were self-rehabilitated with technological devices may have decreased outcome performance making self-rehabilitation equally valuable to conventional therapy made with a therapist. The differences between the three papers and the present paper also may reflect subtle differences in using different types of technologies and differences in the user-friendly interface design of the technology. Irrespective of whether or not technology enhances rehabilitation relative to conventional therapy, the results of the present paper lend support to the use of technologically assisted self-rehabilitation for prolongation of effective treatment following hospital-based rehabilitation.
Certain trials included in this review reported a lack of adherence to self-rehabilitation programs. Poor treatment adherence may be responsible for non-significant outcomes in rehabilitation. In their article of 2018, Argent et al 27 identified several factors that may influence adherence to self-rehabilitation. Psychological and cognitive factors have been highlighted as potential reasons for reduced treatment compliance. External locus of control, feeling pain during exercises, having a low level of physical activity, and not being supported by family or friends were also reported as contributors to bad treatment adherence. In the same review, the authors suggested a few solutions to consider in order to improve treatment compliance. First, by providing real time sessions (face-to-face or by video conference) and with sensing platforms such as cameras or inertial measurement units that measure activity during the patient’s home exercises. These measures provide the clinician with a better understanding of the patient’s behavior and problems, and they would therefore be able to give feedbacks and adapt the program to an appropriate level of difficulty for the patient, thereby maintaining treatment compliance. Second, the identification of clear measurable objectives and the implementation of visual feedback (such as graphics) on patient’s weekly performance (e.g., level of physical activity and time spent in rehabilitation) were considered as parameters moderating treatment adherence. Lastly, patient’s education on their pathology and the provision of written operating instructions on the exercises to perform improved intervention compliance.
Our meta-analysis suggests that self-rehabilitation can be performed in addition to or in substitution of conventional therapy, in adults with subacute and chronic stroke. These results are not in line with the meta-analysis of Da Silva et al., 2018, since the authors showed that self-rehabilitation had a superior effect for arm recovery when performed in patients more than 12 months post-stroke compared to more recent time since stroke onset. 13 This could be explained by the fact that our review assessed different outcomes at different times after stroke.
Cost-Effectiveness of Self-Rehabilitation
Self-rehabilitation may be far less expensive than conventional therapy in terms of time saved for the patient, care partner or clinician. In this review, only four studies evaluated the cost of intervention.9,33,59,63 Among these, one study performed a cost-effectiveness analysis and showed that its program was more cost-effective than conventional face-to-face therapy. 9 This finding is supported by previous work that conducted cost-effectiveness analysis to compare telerehabilitation programs to conventional therapy in other pathologies.70,71
Implications for Clinical Practice
Our results indicate with a low to moderate body of consistent evidence that self-rehabilitation of motor function and activity is equal to conventional therapy. Self-rehabilitation should be considered as an additional method allowing to increase rehabilitation intensity or duration. The rehabilitation team could implement the rehabilitation plan depending on the patient’s functional assessment, delay since stroke, and contextual factors. Self-rehabilitation is thus complementary to face-to-face practice and should not replace it. Indeed, by providing self-rehabilitation in addition to conventional therapy, the burden of healthcare professionals will be reduced and facilitated. Furthermore, self-rehabilitation may help to increase therapy time and offer treatment for patients who may not otherwise have a treatment opportunity, whatever the delay since stroke. A recent RCT conducted in Benin showed great potential to provide such programs in developing countries. 72 Lastly, the new arrival of the COVID-19 crisis has imposed new constraints to both therapists and patients. 73 For both these scenarios, developing the means to provide self-rehabilitation (whether supplied with technological devices or not) must be considered to increase treatment opportunities as a potential solution when access to treatment is reduced.
Regarding the content of self-rehabilitation, future programs should use new low-cost technologies (such as smartwatches, tablets, smartphones, and virtual reality) to improve and assess adherence to treatment. The contents of these structured programs should be designed by a rehabilitation professional defining agreed objectives with the patient, giving clear instructions (written or video), and providing coaching during real time sessions. The content should be based on intense physical exercises that are progressively adapted in difficulty, promoting use of the affected limb and providing feedback. 26
Recommendations for Future Research
There is a need for future research to precisely define the content (in terms of duration and intensity) of all group interventions and to select appropriate and recommended outcomes. 32 To this end, future trials should measure the duration and intensity of activity during self-rehabilitation and conventional therapy using objective measures. We recommend the use of actimetry measures of heart rate, smartwatches, and armbands that measure activity during rehabilitation. 74 It is also recommended to analyze movement kinematics with inertial sensors or cameras in order to objectively measure and better understand patient activity and recovery. Adherence and cost-effectiveness should also more often be assessed to better understand their impact on self-rehabilitation. Moreover, patient’s recruitment should be improved by assessing intersubject differences and providing concealed allocation. 30 Future RCTs should also compare self-rehabilitation to the best evidence synthesis and match the dose and duration of intervention in all groups. Lastly, self-rehabilitation programs should be defined based on prognostic factors and tailored according to patient’s objectives. 75
Strengths and Limitations
This meta-analysis brings new information regarding the effects of self-rehabilitation on motor function and activity in adults with stroke according to the ICF-WHO framework. Further, this review conducted innovatory subgroup analyses to test the influence of location, stroke stage classification, type of treatment and dose, design and duration on the self-rehabilitation effect. Only RCTs of good methodological quality (PEDro score ≥5) were selected.
However, some limitations must be mentioned. First, the definition of self-rehabilitation and its differentiation from conventional therapy may be subjective and difficult to quantify. Furthermore, there is no consensus on the definition of self-rehabilitation and relative to other commonly used terms such as telerehabilitation, self-directed therapy, and home-based rehabilitation. Second, although hand-searching was conducted with PEDro and Google scholar databases in order to avoid publication bias, some unpublished RCTs available in the Grey Literature may have been missed. In addition, publication biases were only assessed visually using funnel plot symmetry. No statistic indicators were considered to exclude trials responsible for the funnel plot asymmetry. Third, the limited available data regarding outcomes such as the Functional Independence Measure and the Barthel Index prevents to review the effect of self-rehabilitation on activity of daily living. Lastly, some RCTs of good quality may have not been included in the review due to unclear methodology or unpublished data.
Conclusion
This systematic review and meta-analysis provides low to moderate quality of evidence that self-rehabilitation and conventional therapy are equally effective for post-stroke motor recovery and activity. The use of technology, the total duration, and design of the intervention (self-rehabilitation in addition to vs in substitution of conventional therapy), the treated stroke population and the location of the trial did not have any influence on outcomes.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683211048773 – Supplemental Material for Self-Rehabilitation for Post-Stroke Motor Function and Activity–A Systematic Review and Meta-Analysis
Supplemental Material, sj-pdf-1-nnr-10.1177_15459683211048773 for Self-Rehabilitation for Post-Stroke Motor Function and Activity–A Systematic Review and Meta-Analysis by Gauthier Everard, Alexandre Luc, Ioannis Doumas, Khawla Ajana, Gaëtan Stoquart, Martin Gareth Edwards and Thierry Lejeune in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgments
We would like to thank Sophie Patris, professional librarian at UCLouvain, for her help in the elaboration of the search strategy.
Authors contributions
AL and GE independently conducted the selection process and extracted data and performed analyses. GE wrote the manuscript. TL, ME, KA, ID, GS and AL contributed to data interpretation and manuscript revisions. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We would like to thank the Région Wallonne, the SPW-Economie-Emploi-Recherche and the Win2Wal Program (convention n°1810108) for their support. This publication also benefits from the support of the French Community of Belgium within the framework of the financing of a FRIA grant.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
