Abstract
Purpose. The purpose of this study was to estimate the effect of the Cognitive Orientation to daily Occupational Performance (CO-OP) approach compared with usual outpatient rehabilitation on activity and participation in people <3 months poststroke. Methods. An exploratory, single-blind, randomized controlled trial, with a usual-care control arm, was conducted. Participants referred to 2 stroke rehabilitation outpatient programs were randomized to receive either usual care or CO-OP. The primary outcome was actual performance of trained and untrained self-selected activities, measured using the Performance Quality Rating Scale (PQRS). Additional outcomes included the Canadian Occupational Performance Measure (COPM), the Stroke Impact Scale Participation Domain, the Community Participation Index, and the Self-Efficacy Gauge. Results. A total of 35 eligible participants were randomized; 26 completed the intervention. Post intervention, PQRS change scores demonstrated that CO-OP had a medium effect over usual care on trained self-selected activities (d = 0.5) and a large effect on untrained activities (d = 1.2). At a 3-month follow-up, PQRS change scores indicated a large effect of CO-OP on both trained (d = 1.6) and untrained activities (d = 1.1). CO-OP had a small effect on COPM and a medium effect on the Community Participation Index perceived control and on the Self-Efficacy Gauge. Conclusion. CO-OP was associated with a large treatment effect on follow-up performances of self-selected activities and demonstrated transfer to untrained activities. A larger trial is warranted.
Keywords
Introduction
Individuals living with the effects of a stroke continue to experience significant challenges with their functional health, despite advances in rehabilitation. Approximately half of those living in the community after their stroke are dependent in terms of activities of daily living, 1 and the majority have not achieved their individual functional goals. 2 They experience participation restrictions,3-5 have limitations in performing meaningful activities, 6 and are significantly less active than age-matched controls. 7 Evidence suggests that treatments that incorporate repetitive task-specific training are the most effective of contemporary rehabilitation approaches to improve gait speed and upper-extremity activities and are recommended over traditional neurodevelopmental approaches.8,9 However, the effect seems to be limited to the specific tasks trained, 9 and retention of learning has not been consistently demonstrated. 10 In contrast, the Cognitive Orientation to daily Occupational Performance (CO-OP) approach, which superimposes cognitive and metacognitive elements on task-specific training, is associated with improvements in both trained and untrained activities, and the newly acquired skills are retained.11,12
CO-OP is a complex treatment approach that differs from other contemporary stroke rehabilitation approaches in that it combines theory and evidence from both motor and cognitive sciences and situates them in an educational, client-centered framework. It is defined as “a client-centred, performance-based, problem solving approach that enables skill acquisition through a process of strategy use and guided discovery.”13, p2 The clinical objectives are skill acquisition, cognitive strategy use, and generalization and transfer of learning. Elements from the motor domain are used primarily to meet the clinical objective of skill acquisition; these include practicing specific functional tasks (ie, task-specific training), usually as a whole task, such as dressing, cutting food, or walking outdoors, rather than task components such as reaching, grasping, or balance training in isolation from the task.
A limitation of task-specific training, when used alone, is that generalization to other situations and transfer to other tasks, are generally not demonstrated, 9 and improvements gained in therapy are not consistently maintained once therapy stops.10,14 In CO-OP, retention, generalization and transfer have consistently been reported.11,12,15,16 This may, in part, be because of the cognitive and metacognitive elements that are superimposed on the task-based training. In CO-OP, a global cognitive strategy, adapted from Meichenbaum’s goal-plan-do-check, 17 teaches participants to problem solve and self-monitor their own task performance. Additionally, within each task performance, therapists use a conversation-based teaching and feedback technique known as guided discovery, 18 in which participants are guided through questions, cues, and coaching to solve task performance problems on their own, rather than being given explicit instructions. Through this process, they learn to analyze their own performance and to subsequently develop performance strategies to overcome issues. This differs markedly from traditional approaches, in which it is the therapist who does the analysis of performance breakdowns, develops performance strategies, and explicitly teaches the patient how to use those performance strategies.
Preliminary evidence to support CO-OP’s benefits for individuals poststroke was first demonstrated in people more than six months poststroke.11,12,19 Two single-case experimental series showed not only improved activity performance and retention of learning following CO-OP intervention 11 but also improvement in untrained activities, suggesting the occurrence of transfer to new skills. 12 Because it is relatively well established that stroke outcomes are improved if rehabilitation occurs in the first few months following the event8,20,21 and because past CO-OP participants recommended that the strategies be taught much earlier in the rehabilitation process, 22 we sought to investigate the efficacy of the approach in subacute stroke. Therefore, the primary objective of this exploratory trial was to estimate, in people <3 months poststroke, the effect of CO-OP compared with usual occupational therapy on immediate and longer-term activity performance and participation.
Methods
A single-blind, exploratory, randomized controlled trial, with a usual care control arm, was conducted. Participants referred to outpatient stroke rehabilitation programs at 2 university-affiliated, freestanding rehabilitation centers were randomized to receive either the usual outpatient program, which included occupational therapy (usual care) or the usual outpatient program with CO-OP replacing usual occupational therapy (CO-OP).
Sampling and Randomization Procedures
Patients who had sustained an ischemic stroke (ICD-10 codes I63 and I64) referred to outpatient rehabilitation at Sunnybrook–St John’s Rehab in Toronto, ON, Canada, or The Rehabilitation Institute of St Louis, MO, between March 2011 and March 2013 were included. Exclusion criteria were the following: >3 months poststroke when starting outpatient rehabilitation, hemorrhagic stroke, other neurological diagnoses, major psychiatric illness, moderate or severe aphasia (combined scores of 6 or less on Canadian Institute of Health Information 23 items 64 and 66), or cognitive impairment (Montreal Cognitive Assessment 24 scores of 21 or less). It was estimated a priori that a sample size of 14 per group would provide 82% power to detect a between-group difference of 1.3 units on the Canadian Occupational Performance Measure (COPM). 25 To ensure balanced group sizes, a consulting statistician prepared a blocked randomization procedure stratified by site. A block size of 6 and an allocation ratio of 1:1 were used to ensure equal assignment to the treatment and control groups for every 6 patients entered in the study at each center. The random number function in Excel (Microsoft Corporation, Microsoft Excel 2010, Version 14.0.) was used to create a random sort order within each block. To ensure allocation concealment, an administrative assistant at each site, not associated with the study, created sequentially numbered sealed opaque envelopes for the study coordinator at each site. The study coordinator was not involved in the assessment or treatment of any of the participants. Treatment allocation was not completed until after consent was obtained and all inclusion/exclusion criteria had been reviewed. Because knowledge of blocking reduces the unpredictability of the next assignment, the study investigators, project coordinators, and treating therapists were all blinded to the randomization procedure and block size.
Assessment and Intervention Procedures
At time 1, participants underwent a baseline assessment battery conducted by a research assistant who was blinded to group allocation. Following that, a research occupational therapist, not involved with delivering either intervention and also blinded, conducted a goal-setting interview using the COPM. During this interview, participants selected the 4 to 6 personally meaningful functional activity goals that were the most important to them. The participants then had a third baseline assessment session in which they were videorecorded performing those self-selected activity goals.
Following the time 1 assessments, both the CO-OP and Usual Care groups received usual outpatient stroke rehabilitation with specific services based on their individual needs, such as physical therapy, speech-language pathology, or nursing. The Usual Care group received usual outpatient occupational therapy from therapists employed by the participating sites, whereas the CO-OP group received occupational therapy from CO-OP-trained occupational therapists who were part of the research team. Treatments were generally twice per week; sessions were 45 minutes long for the CO-OP group and ranged from 45 to 60 minutes long for the Usual Care group. Because the participants had a range of stroke severities and rehabilitation needs and, as is typical in usual outpatient rehabilitation, the number of sessions varied. The number of treatment sessions attended by an individual participant depended on his or her needs, the clinical judgment of the treating occupational therapist, and the institutional policies. The CO-OP group received a maximum of 10 CO-OP intervention sessions, and any additional sessions that more complex patients needed were conducted as usual care. These additional usual care sessions were tracked and counted as part of the total number of CO-OP treatments. A limitation on the number of sessions received by the usual care group was institutional policy. In Toronto, clients were limited to a maximum of 32 treatment sessions, and in St Louis, clients were limited by the number of sessions covered by their insurance provider or other means of payment.
CO-OP Intervention Description
Complete details about the theoretical underpinnings and the implementation of the CO-OP approach have been published in a textbook. 13 In this study, CO-OP treatment occurred separately from the rest of the outpatient team to avoid contamination. During the first session, the CO-OP therapist reviewed the goals previously established in the COPM interview and worked with the participant to decide on which 3 of those would become the focus of intervention sessions. Also in the first session, the CO-OP therapist used a visual presentation to teach the participant the global cognitive strategy, goal-plan-do-check. 17 In subsequent sessions, the goal-plan-do-check strategy was used iteratively as the main problem-solving framework to facilitate activity acquisition. The participant would work on one or more of the 3 goals set, guided by the therapist, to discover a plan to achieve the goal. The participant would then do the plan and subsequently check to see if the plan was implemented and if it worked—that is, whether the goal was achieved. If the goal was not achieved, the participant was guided to analyze the performance breakdown and modify or create a new plan. Within the plan phase, the therapist used guided discovery to help the participant analyze the performance breakdown and discover domain-specific strategies to overcome the particular performance problems of that client with that activity. Thus, the plan-do-check process was repeated until the performance breakdown was successfully overcome, repeatedly; then, going to the next performance breakdown until all were overcome and the goal was achieved, repeatedly. It is important to note that although in CO-OP the focus is on performing the task to be learned, there is no particular emphasis on the number of repetitions. Repetitions are never stipulated; rather, the number of times a particular task is practiced depends on the quality and consistency of the performance. Once the participant is satisfied with the performance on the particular part of the task being worked on, as determined by the check in the global cognitive strategy, the next breakdown is identified, and a new learning cycle begins. This is repeated until the goal is achieved.
Usual Care Description
Participants randomized to the usual care group received usual outpatient occupational therapy with one modification. As described above, a nontreating research occupational therapist blinded to group allocation administered the COPM to assist participants in self-selecting personally meaningful goals, prior to beginning therapy. This was a departure from usual administration of the COPM because it tends to be conducted by the treating occupational therapist if done at all. The COPM results were made available to the treating occupational therapist, but no instructions were given regarding what to do with the information.
A survey given during the preparation for this study indicated that usual care consisted of a combination of functional, task-based training and component-based training, as deemed necessary by the treating therapists.
CO-OP Intervention Fidelity
Two therapists, one in Toronto and one in St Louis, were trained in CO-OP with a standard 2-day CO-OP workshop. For ongoing training, consolidation, and fidelity purposes, the 2 CO-OP therapists were then videorecorded treating a series of pilot participants and received feedback until they were consistently scoring full marks on an intervention fidelity checklist. Once the intervention began, videos from sessions 3, 6, and 10 were reviewed and scored using the same intervention fidelity checklist to ensure ongoing fidelity.
Outcomes Measures
Table 1 provides an overview of instrument characteristics and timing of their administration. Research staff blinded to group allocation conducted assessments prior to the intervention starting (time 1), after discharge from occupational therapy or after 10 sessions (time 2), and 3 months after time 2 (time 3). Because the number of intervention sessions varied among participants based on the severity of their stroke and their individual rehabilitation needs, representatives from usual care, either therapists or administrative staff, were asked to inform the study coordinator when the participant was discharged from occupational therapy or when 10 sessions were completed, whichever came first. Time 2 assessments were performed at this point as an attempt to assess initial outcomes after a similar treatment dosage. Because many of the participants had been discharged home from acute care hospital only a few days before the time 1 assessment and because the questions posed in the participation measures are designed for people who have been in the community for at least a few weeks, these were administered only at time 2 and time 3 to ensure their validity.
Outcome Measures and Timing.
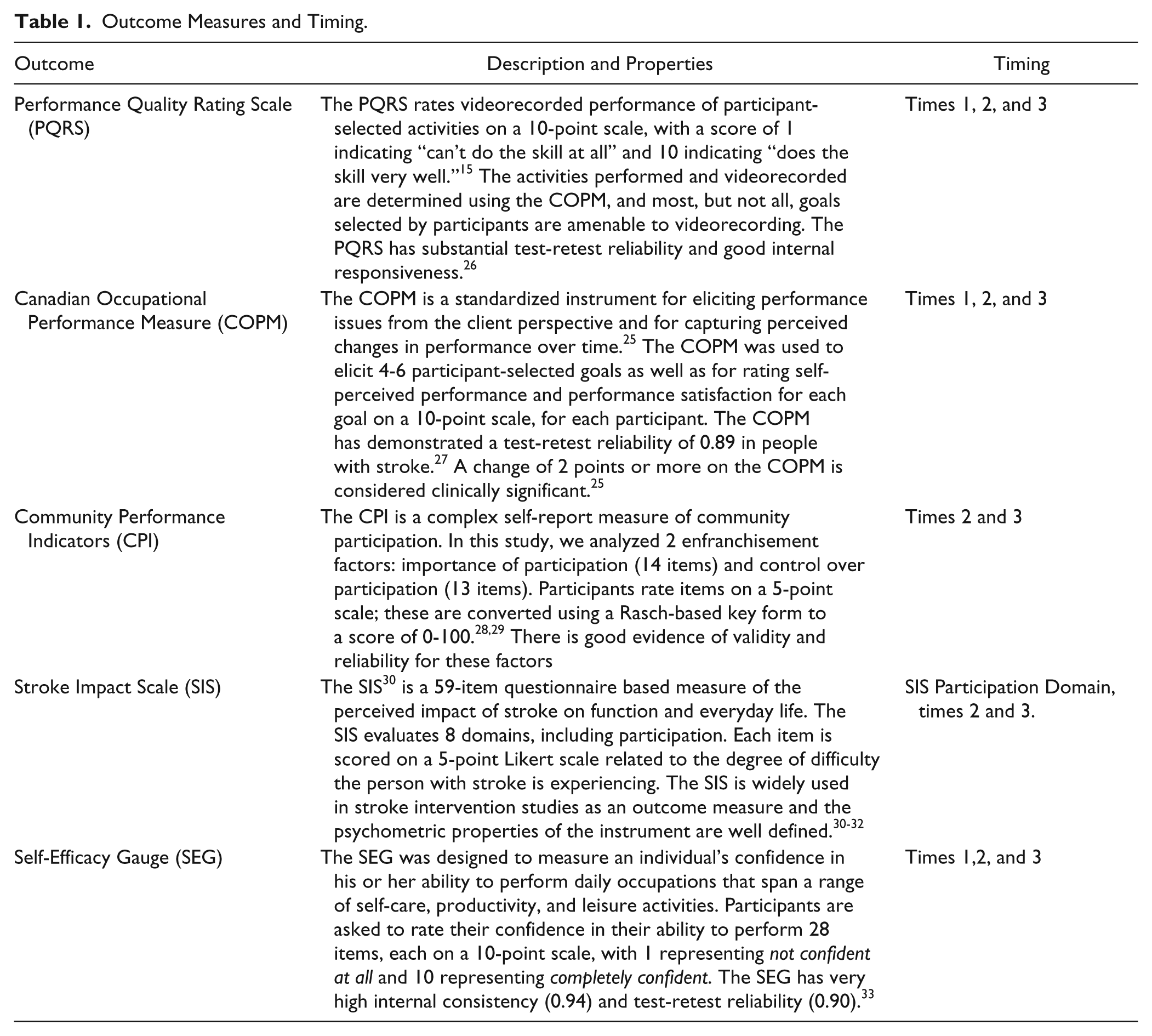
The primary study outcome was change in actual performance quality of self-selected activities, measured with the Performance Quality Rating Scale (PQRS). 15 The PQRS is a 10-point scale used to rate videorecorded performances of participant-selected activities. The participants perform the activities selected in the COPM interview in a safe environment but unaided by physical support or verbal cueing. The video recordings are rated on a scale of 1 to 10 in which 1 indicates can’t do the activity at all and 10 indicates does the activity very well. In this study, an independent, blinded observer viewed the videos in randomized, nonchronological order. The PQRS has substantial test-retest reliability and good internal responsiveness. 26
The COPM was included as an indicator of perceived performance quality and satisfaction with performance quality. The secondary study outcome was participation, assessed with the Stroke Impact Scale participation domain and the Community Participation Index. Additionally, self-efficacy was measured using the Self-Efficacy Gauge.
Data Analysis
Data analysis was conducted using SPSS version 21 (IBM Corp. Released 2012; IBM SPSS Statistics for Windows, Version 21.0) and Microsoft Excel (Microsoft Corporation, Microsoft Excel 2010, Version 14.0). The data were cleaned, checked for accuracy and missing values, and checked for normal distribution using the Shapiro-Wilk test. Descriptive statistics were compiled for all variables, and baseline comparisons between sites and between the groups were made.
Time 1 to time 2 and time 1 to time 3 mean change scores and standard deviations were calculated for normally distributed data, and Cohen’s d effect sizes and confidence intervals were calculated. For nonnormally distributed data, medians were determined, and a nonparametric effect size r was calculated using the formula r2 = z2/N. 34
CO-OP therapist logs and institutional patient records were reviewed to establish which self-selected activities were trained during the outpatient rehabilitation program. Records from occupational therapy sessions were reviewed by occupational therapists from the research team (DC and MD) for evidence of training. A self-selected activity was considered trained if there was any indication of practicing all or part of it or any indication of discussions or education concerning the activity. If no evidence of training was found it was considered untrained. In 4 cases, only partial records were available, in which case trained and untrained goals were confirmed by examining typical therapy activities by the same therapist with other participants. For COPM and PQRS scoring and analysis, trained and untrained activity scores were grouped, summed, and averaged separately to give a single trained and a single untrained score for each participant.
The ethics review boards at the relevant institutions approved this study.
Results
Figure 1 is the CONSORT diagram depicting participant flow through the study. A total of 35 eligible participants were randomized: 20 from St Louis and 15 from Toronto. Of those, 26 completed the intervention. Table 2 provides summary scores for participant demographics by site and by group. Participants in Toronto had more years of education (P = .03), and participants in St Louis began outpatient rehabilitation approximately 17 days sooner following their stroke than did participants in Toronto (P = .04). Otherwise, there were no significant between-site differences at baseline. The 2 treatment groups were equivalent at baseline on all variables examined. After excluding 2 extreme outliers, who had more than 100 treatment sessions—one in each treatment group and both from St Louis—those in the usual care group who completed the study had an average of 13.3 occupational therapy sessions (range = 3-30); those in the CO-OP group had an average of 12.2 occupational therapy sessions (range = 5-33). CO-OP therapists scored an average of 80% accuracy on CO-OP fidelity checklists.
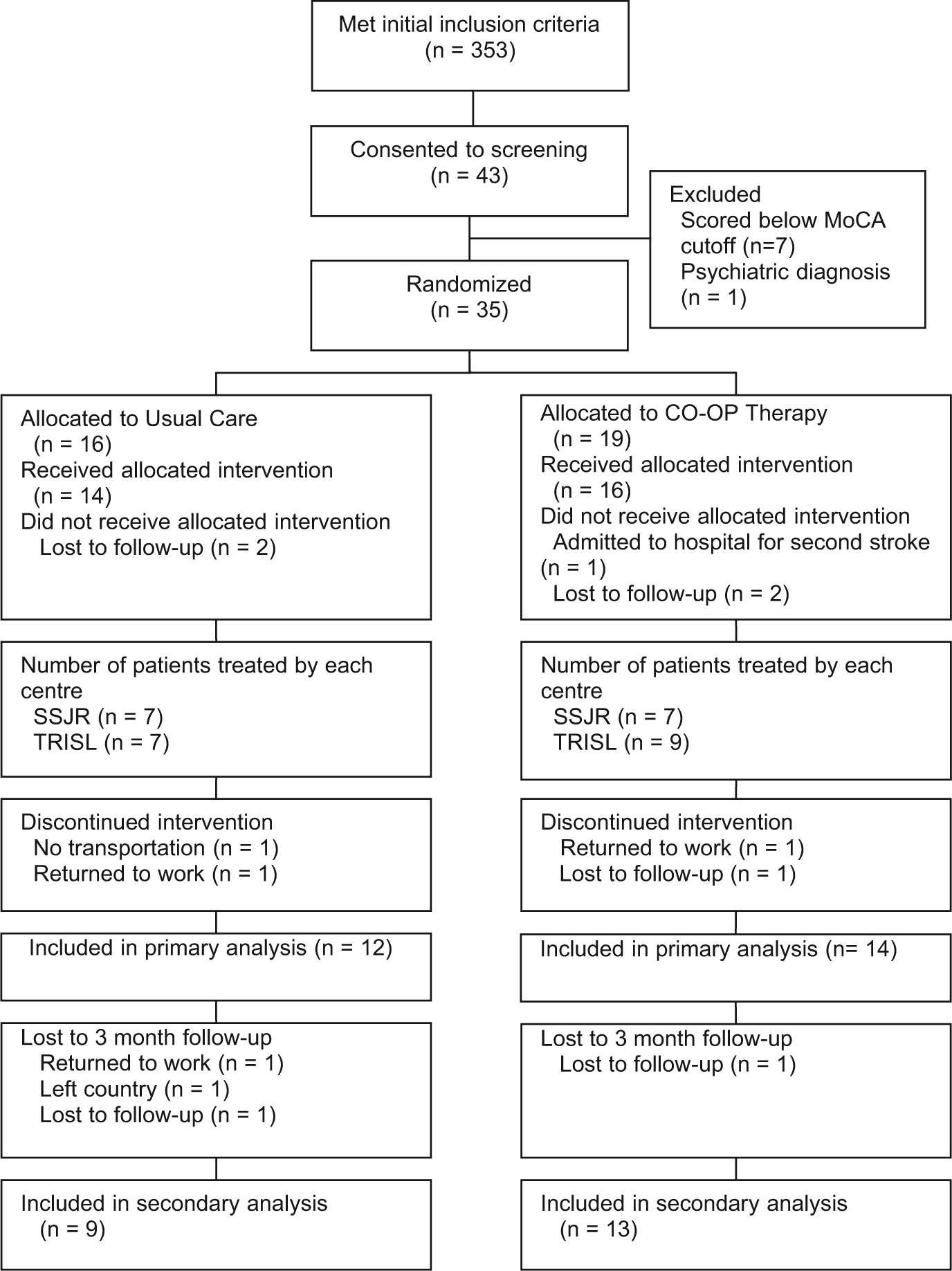
CONSORT diagram.
Demographics.
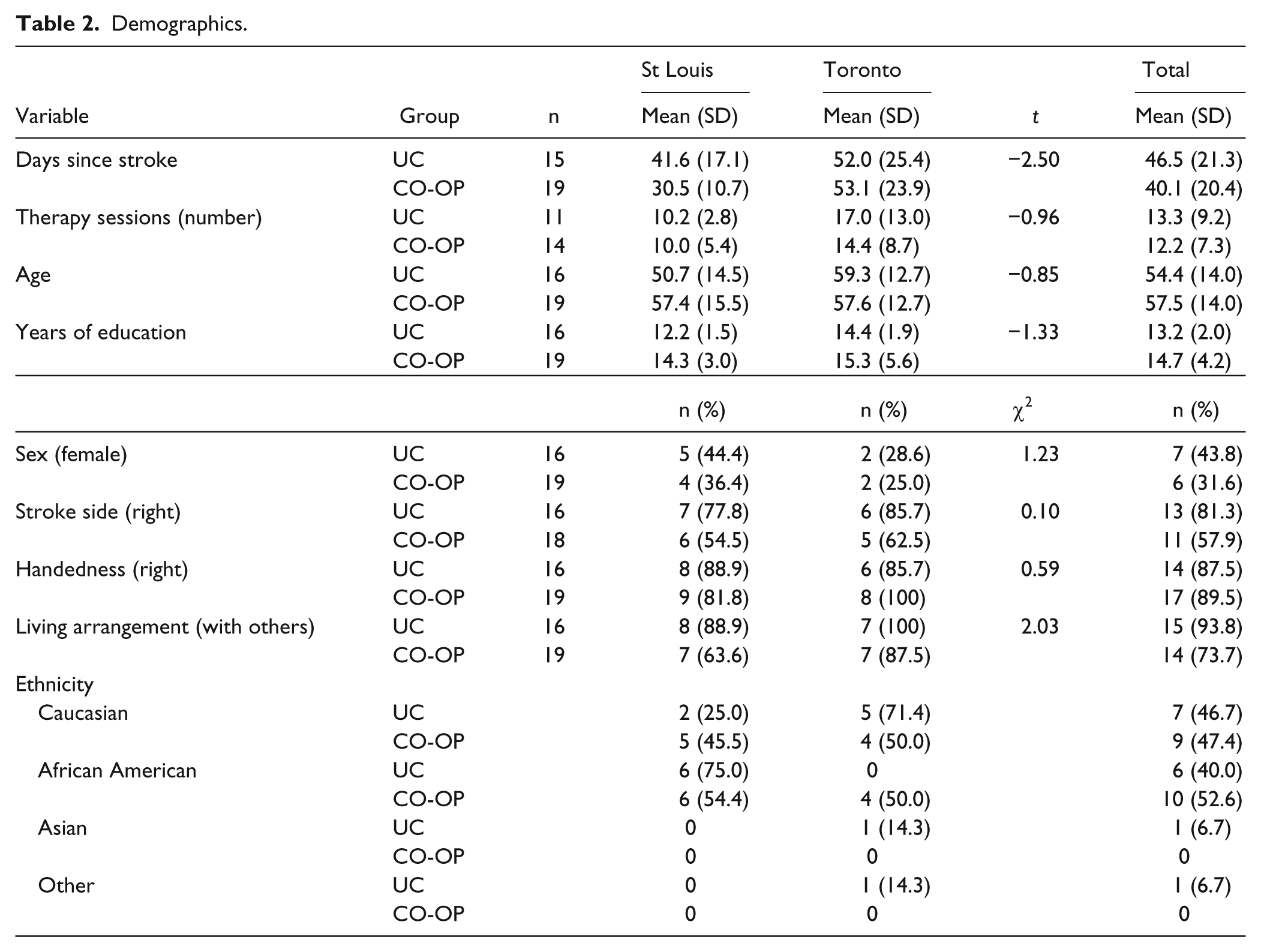
Table 3 provides an overview of the 178 goals selected during the COPM interviews. A total of 23 of the 178 goals, such as “weight management” or “traveling to see family” could not be videorecorded and thus did not have PQRS scores associated with them. As an individual’s PQRS score was based on an average of scores from all his or her self-selected activities, the loss of some activities that could not be videorecorded did not cause missing PQRS data; all participants who completed the intervention had at least 1 trained and 1 untrained activity that could be included in the analysis.
Summary of Participant-Selected Goals.
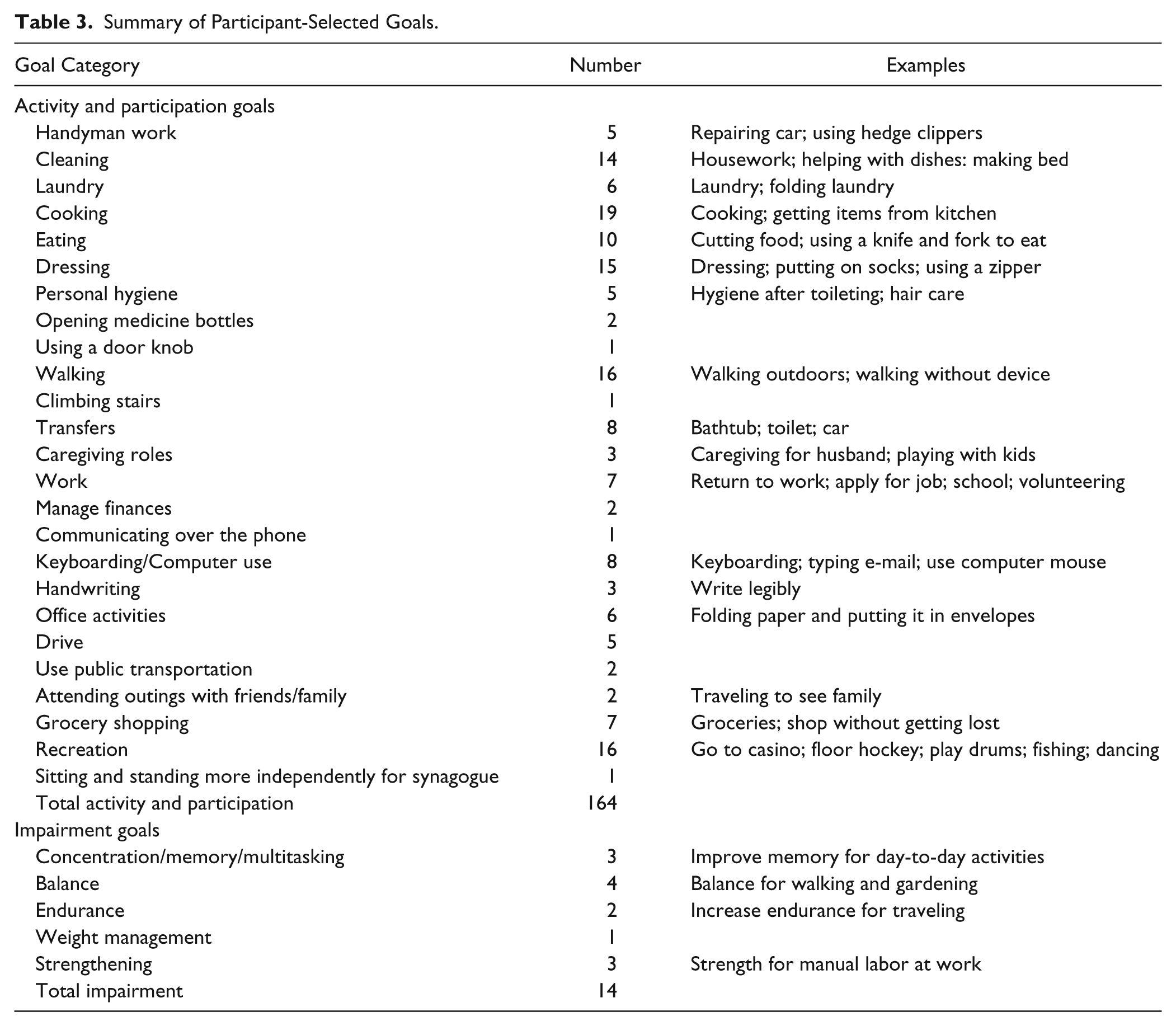
Table 4 displays summary statistics, change scores, and effect sizes. The effect size of Cohen’s d can be interpreted as follows: 0.2 represents a small effect; 0.5 represents a medium effect; and 0.8 represents a large effect. 35 CO-OP’s effect over usual care at time 2 was medium for PQRS-trained activities (d = 0.5) and large for PQRS untrained activities (d = 1.2). Large effects for time 3 change scores were found for both PQRS trained (d = 1.6) and untrained activities (d = 1.1). There was no effect of CO-OP over usual care on time 2 COPM change scores, a small effect at time 3 for changes in COPM performance and satisfaction of untrained goals, and a small effect for changes in performance of untrained goals. At time 3, CO-OP had a medium effect over usual care for change in CPI perceived control (d = 0.7) and the SEG (d = 0.7).
Means, Standard Deviations, Change Scores, and Cohen’s d Effect Size. a
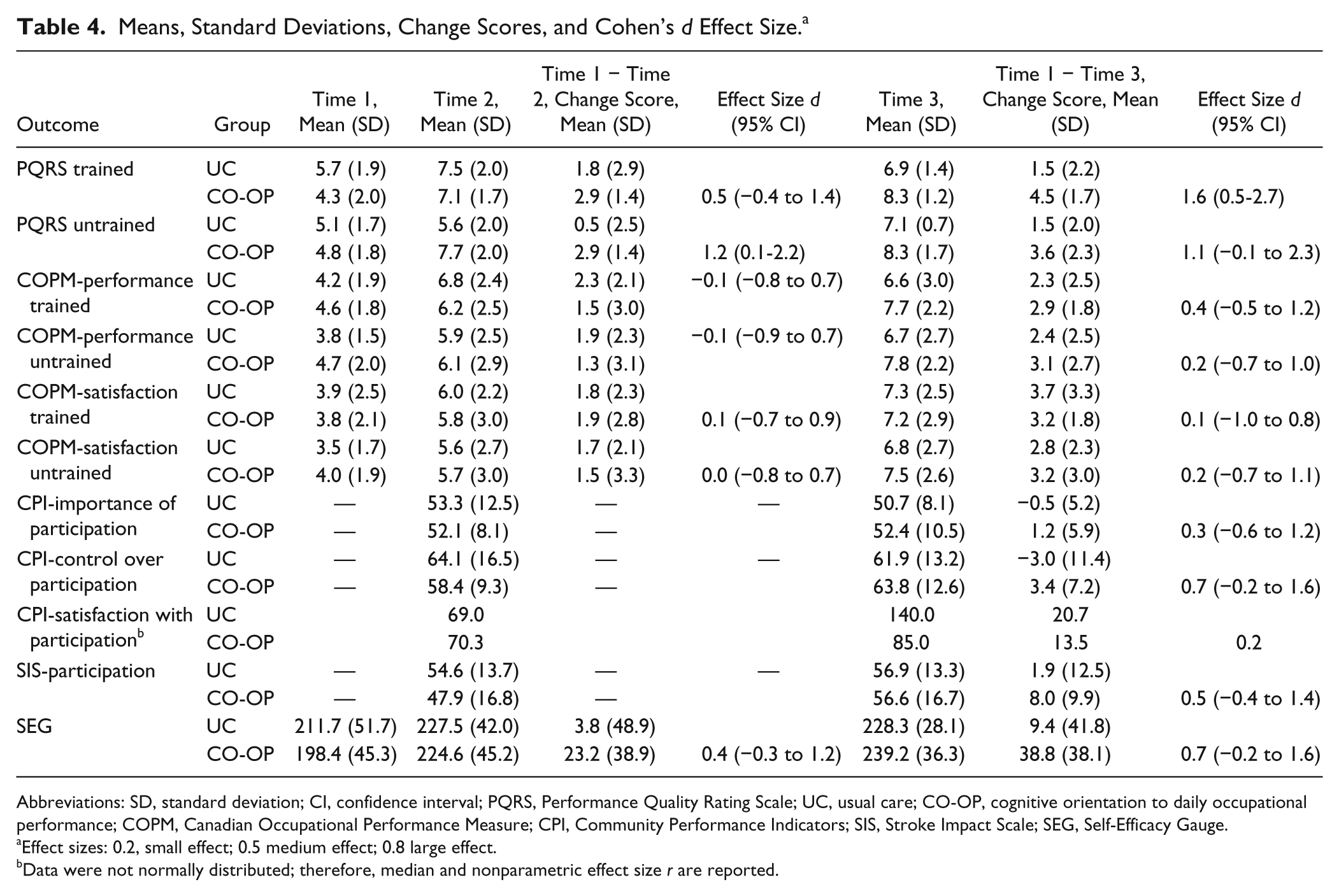
Abbreviations: SD, standard deviation; CI, confidence interval; PQRS, Performance Quality Rating Scale; UC, usual care; CO-OP, cognitive orientation to daily occupational performance; COPM, Canadian Occupational Performance Measure; CPI, Community Performance Indicators; SIS, Stroke Impact Scale; SEG, Self-Efficacy Gauge.
Effect sizes: 0.2, small effect; 0.5 medium effect; 0.8 large effect.
Data were not normally distributed; therefore, median and nonparametric effect size r are reported.
Discussion
Incorporating the CO-OP approach as part of an outpatient rehabilitation program is associated with a large effect at follow-up on actual performance of trained and untrained self-selected functional activities compared with programs incorporating usual occupational therapy. This suggests not only improved performance on skills trained in rehabilitation but also transfer of cognitive strategy training to permit those living with the effects of stroke to learn new skills outside of the rehabilitation setting as the need arises. This discussion elaborates on the features of the CO-OP approach that may contribute to transfer of learning to untrained activities, on the small effect of perceived performance, and on study limitations.
Transfer of skills learned in rehabilitation to novel skills is necessary to achieve optimal long-term functional health because rehabilitation programs are unable to teach clients all the activities they may need at home and in the community or even all variations of a single activity. Measurement of transfer of cognitive strategy training to real-world situations has been accomplished by the assessment of untrained tasks, by standardized assessment of daily tasks, or by participant or proxy self-report of daily-life situations. 36 In this study, the primary indicator of transfer was change in performance on self-selected tasks or activities not addressed during the intervention sessions with the therapist (untrained tasks). This type of far transfer (transfer to a completely unrelated task) is expected with cognitive strategy training because the therapist’s primary emphasis is on teaching problem solving skills rather than on teaching the particular functional skill itself. 36
Therapists trained to use the CO-OP approach are explicitly taught to work toward generalization and transfer. 13 Other CO-OP features may also contribute to transfer, including guided discovery, performance analysis, and self-discovery of performance strategies. Transfer is reported to be more closely linked to variable practice than blocked practice. 37 One theory for explaining this phenomenon is the learned-variability model of skill transfer, in which learning a skill involves learning how to do the skill in different ways and learning when to alter it. 38 In CO-OP, learned variability occurs through the use of guided discovery combined with performance analysis within the plan and check phases of the goal-plan-do-check framework. The client is taught to check or self-analyze his or her performance breakdowns and is guided to identify different plans or strategies until settling on one that works best to perform the task at hand, learning different ways to do the skill as part of the process. Additionally, clients are guided to identify their own strategies rather than being assigned strategies by the therapist. There is experimental evidence to suggest that providing time to attend to performance issues and subsequently self-select a strategy is more strongly associated with transfer than being given a strategy. 39
Evidence from a neuroimaging study suggests that the combined training of motor and cognitive systems has a positive impact on transfer. Olsson et al 40 conducted an experiment comparing motor practice, mental practice, and combined motor and mental practice to learn a finger tapping task and unexpectedly discovered improvements on a novel transfer task only in the combined training group. Functional magnetic imaging data indicated overall broader cortical activity in the combined training group and showed that the posterior cerebellar hemisphere was involved in transfer.
Transfer is also linked to high self-efficacy, 41 and high self-efficacy is linked to better functional outcomes in stroke. 42 Stevens et al 41 demonstrated that practice of an easier task rather than a more difficult task was associated with higher self-efficacy, and subsequently, that higher self-efficacy predicted better ability on a transfer task. These authors speculated that self-efficacy, rather than implicit learning, is a mediator of transfer to a similar but more difficult task, although they are cautious to emphasize that further research is required to confirm this finding. Interestingly, in this CO-OP study, an unexpected medium effect of CO-OP on self-efficacy was shown. We speculate that following CO-OP, self-efficacy in a broad range of daily activities likely comes from having experienced success with the self-selected activities practiced in therapy, the attribution of that success to the newly learned ability to problem solve performance issues, and the subsequent willingness to try new activities at home independently. Thus, it is plausible that improved self-efficacy is an outcome of CO-OP, and self-efficacy then mediates transfer.
Self-efficacy and the ability to transfer new learning from rehabilitation to the real world may both be mediators of participation. It may be that improvements in self-efficacy need to be in place before measurable improvements in participation are seen. For example, the changes in the Community Participation Index for the CO-OP group were higher for the perceived control over participation domain than for involvement in living situations domain, suggesting a degree of confidence in the ability to participate but less actual involvement. This may be because the transition from confidence and ability to actual doing takes more time.
Limitations
This was an exploratory study with the objective of estimating CO-OP’s effect relative to a control treatment in preparation for a larger, more definitive trial. As such, the sample size was too small to find statistically significant differences for most outcomes or to stratify groups on potential key confounders, such as stroke severity. The sample size also limited the statistical analysis to univariate procedures without the capacity to control for the effect of potential confounders, such as site and dosage.
The decision to have a usual care control meant that the comparison treatment was unstandardized. Both control programs were in university-affiliated hospitals linked to well-regarded academic programs, and the control treatment is believed to have been close to current best practice—that is, interdisciplinary treatment, largely based on repetitive functional task training, with treatment of impairments and components when deemed appropriate by the therapist.
For the purposes of blinding and consistency between the 2 treatment groups, the COPM was administered by a research therapist with no clinical relationship with the patient, rather than by the treating therapist as is usually done. This may have resulted in a disconnect between the participant and the treating therapist regarding the self-selected goals, may have had implications for the relatively smaller effect of CO-OP on COPM, and may be something to reconsider in future studies.
A final important limitation of this study was the relatively short follow-up period, which was just 3 months after the postintervention assessment and an average of 7 months following the stroke. To get a better estimate of the stability of the treatment effects and the impact on participation, future studies should follow participants for at least 1 year after discharge.
Conclusion
CO-OP, incorporated as part of a usual outpatient stroke rehabilitation program, was associated with a large treatment effect compared with usual outpatient rehabilitation alone on follow-up performances of both trained and untrained self-selected activities. Results also suggest a medium effect on changes in participation from postintervention to follow-up and on self-efficacy. A larger-scale trial is warranted.
Footnotes
Acknowledgements
The authors acknowledge the assistance of Alisa Grigorovich, Tanya Ramsey, Carin Roth, Tammy Craig, Kimberly Walker, and Duana Russell-Thomas as well as the staff in the Performance, Participation, and Neurorehabilitation Laboratory in the Program in Occupational Therapy at Washington University in St Louis for their assistance with assessment, evaluation, and treatment.
Authors’ Note
ClinicalTrials.gov Identifier: NCT01309165.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a Canadian Institutes of Health Research Open Operating Grant (FRN#111200) and was supported by the St John’s Rehab Hospital and The Rehabilitation Institute of St Louis. SM’s salary support came from the St John’s Rehab Foundation. TW received salary support from the National Center for Medical Rehabilitation Research (NCMRR) in the National Institute of Child Health and Human Development (NICHD) of the National Institutes of Health under award numbers K12HD055931 and K23HD073190. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
