Abstract
Background. Stroke patients with a low memory self-efficacy (MSE) report more memory complaints than patients with a high MSE. Objective. The aim of this study was to examine the effect of a memory-training program on MSE in the chronic phase after stroke and to identify which patients benefit most from the MSE training program. Methods. In a randomized controlled trial, the effectiveness of the MSE training program (experimental group) was compared with a peer support program (control group) in chronic stroke patients. The primary outcome was MSE, measured using the Metamemory-In-Adulthood Questionnaire. Secondary outcomes included depression, quality of life, and objective verbal memory capacity. Changes in outcomes over the intervention period were compared between both groups. Demographic and clinical variables were studied as potential predictors of MSE outcome in the experimental group. Results. In total, 153 patients were included: mean age = 58 years (standard deviation [SD] = 9.7), 54.9% male, and mean of 54 months (SD = 37) after stroke. Of these, 77 were assigned to the training and 76 to the control group. Improvement of MSE (B = 0.40; P = .019) was significantly greater in the training than in the control group. No significant differences were found for the secondary outcomes. An increase in MSE after training was predicted by a younger age (B = −0.033; P = .006) and a better memory capacity (B = 0.043; P = .009), adjusted for baseline MSE. Conclusions. MSE can be improved by the MSE training program for stroke patients. Younger patients and patients with a better memory capacity benefit most from the MSE training program (Dutch Trial Register: NTR-TC 1656).
Persistent cognitive disorders frequently occur as a result of stroke with long-term consequences for physical as well as cognitive functioning and quality of life.1-4 Memory deficits are present in about 30% of all stroke patients and may persist for 1 to even 10 years postonset.5-8 Current memory training programs for brain-injured patients focus on compensatory strategies and on errorless learning. 9 These approaches may be effective in traumatic brain injured (TBI) patients but seem insufficient for stroke patients because they fail to apply the compensatory techniques in their daily life.9,10
In a former study, we explored the relationship between the subjective experience of memory and actual memory deficits in stroke patients.11,12 We found that other factors such as depressive symptoms and personality may determine whether or not stroke patients experience subjective memory complaints. The subjective experience of memory functioning is an important aspect of metamemory. 13 Metamemory is broadly described as “cognitions about memory,” and entails the use of memory strategies and memory self-efficacy (MSE). 14 MSE is the feeling of mastery and control over the way one’s memory functions. TBI patients who have a negative view of their memory functioning are more likely to have depressive symptoms. 15 Stroke patients may also have a negative view of their memory deficits if they have elevated depression scores and neurotic personality traits.11,16,17 Both TBI and stroke patients seem to make less accurate judgments of their actual memory deficits if they have a significantly lower MSE, independent of existing memory deficits.12,15 Support for such a relationship can be found in interventions to improve subjective memory experience in elderly persons who visit memory clinics in the Netherlands. About 17% to 20% of these elderly people experience memory complaints but do not have an objectively decreased memory test performance. 18 An intervention focusing on improving the subjective experience of memory functioning (i.e. MSE)through psychoeducation was found to be effective in reducing memory complaints and improving memory test performance in healthy elderly people. 19
Although there are obvious differences between these healthy elderly people and stroke patients in terms of cognitive functioning, similarities are found in factors that influence the judgment of memory complaints and the lack of a relationship between subjective memory complaints and actual memory functioning.12,17 Following this line of thought, it might be possible that stroke patients with memory complaints could benefit from MSE training to overcome the negative way in which they experience their memory deficits. The focus of this study is not on improving memory deficits but on improving the subjective experience of memory functioning after stroke. If MSE could be improved by a training program, other psychosocial aspects such as quality of life and mood might improve as well. 20
The current study is a randomized controlled trial comparing the effects of MSE training with a peer support program in a sample of patients in the chronic stage after stroke. The aim of this study was to evaluate the effect of MSE training on MSE, depression, and quality of life. We expected MSE to increase in the experimental group but not in the control group. As a result of increased self-confidence in memory functioning, effects on depression and quality of life were also expected. In view of the long period since the stroke, we did not expect to find any changes in memory tests. The second aim of the study was to explore demographic and clinical factors that might predict which patients benefit most from the MSE training program.
Methods
Participants
Patients were recruited in the Rijndam Rehabilitation Centre, Rotterdam, and the Heliomare Rehabilitation Centre, Wijk aan Zee, in the Netherlands. Patients were included in the study if 18 months or more had elapsed since their first and only stroke, if they were between 18 and 80 years old and were living independently, and if they reported subjective memory complaints. Subjective memory complaints were assessed using a semistructured telephone interview. Memory complaints were rated using several questions such as “Do you experience hindrance from your memory deficits in daily life?” “Do you think your memory could be improved?” and “Do you feel ashamed or embarrassed if you forget appointments or information?” Patients who experienced memory complaints were included. Patients who reported memory deficits but nevertheless were able to adequately deal with these deficits by using memory aids were excluded. Other exclusion criteria were progressive neurological disorders such as dementia or multiple sclerosis, insufficient knowledge of the Dutch language, alcohol or drug abuse, and subdural hematomas or subarachnoid hemorrhages. The Medical Ethics Committee of the Erasmus University Medical Centre in Rotterdam, the Netherlands, approved the study. All patients signed an informed consent form.
Intervention
The MSE training was adapted from a program developed by Verhey and Ponds, 21 which aimed at improving subjective memory experience in healthy elderly people to improve memory functioning. This training focuses on altering negative beliefs regarding memory functioning in general. Participants are taught to use memory strategies and are educated on the influence of negative perceptual bias in memory-demanding situations. The effort put into situations demanding memory function is partly determined by subjective beliefs, and this relationship is discussed as well. These concepts are used to optimize goal setting in situations demanding memory function. This MSE training for stroke patients consisted of 4 parts: (1) a general introduction on memory and stroke, including the consequences of actual memory deficits and how to cope with these; (2) a training in internal and external memory strategies to improve compensating abilities (eg, visualization, diary use, and taking notes); (3) psychoeducation on the influence of beliefs, anxiety, memory-related worries, and motivation on memory performance; and (4) realistic goal setting regarding memory-demanding tasks, using cognitive behavioral therapeutic aspects. The training program involved 9 twice-weekly group sessions of 1 hour. Patients were encouraged to discuss the information during the sessions and to relate to familiar situations. Patients received a booklet with information about the sessions. At the end of each session, homework assignments were given, which took about half an hour to complete. These were discussed in the next session.
The control group also followed a schedule of 9 twice-weekly, 1 hour sessions. It was a peer support group in which general education on causes and consequences of stroke was provided. Patients were invited to share problems experienced in their daily life. No active therapeutic interventions were performed in the control group. A psychologist was present to provide basic information on stroke and to moderate the sessions. After completing the 9 sessions, patients received a booklet with handouts of the sessions. The patients did not receive homework assignments in the control group. Both groups were moderated by a trained psychologist.
Study Design
Patients were randomly allocated to either the MSE intervention or the peer support group. All patients were informed on the scope of the study, without details on the contents of both interventions. Thus, patients were not informed on the type of intervention (MSE training vs peer support group) they had been allocated to. All patients were assessed at home within 3 weeks prior to the intervention (T0) and within 10 days after the intervention (T1) by trained research psychologists who were blinded to group allocation. The data were entered into a database by the blinded research psychologists.
Measurement Instruments
MSE was measured using 3 subscales of the Metamemory-In-Adulthood questionnaire (MIA). 22 This questionnaire was translated and validated for a Dutch population by Ponds and Jolles. 23 The questionnaire consists of 74 questions related to subjective memory experiences in daily living. The items are entered on a 5-point Likert scale. The score range for each subscale is 1 to 5. The subscales change, capacity, and anxiety are used to compute an MSE score (range = 3-15). A higher score on the MSE subscale indicates more confidence in capacity and change and lower anxiety regarding memory functioning. The MIA has been used in a large cohort study in the Netherlands, studying cognitive aging. 11 The questionnaire was previously used in neurological populations.15,24
Depression was measured using the Center of Epi-demiological Studies–Depression Scale (CES-D). 25 This scale is used to measure reactive depression. The score ranges from 0 to 60. Higher scores indicate more depressive symptoms. A score of 16 or higher is regarded as a probable clinical depression.
Health-related quality of life was measured using the EuroQol EQ5D questionnaire. This scale consists of 5 questions, which can be transformed to a utility score and a Visual Analogue Scale (VAS). Both the utility score, which represents the social perspective on quality of life, and the VAS score, which represents the patients’ perspectives on quality of life, are used as outcome measures. The utility score has a range score from 0 (worst possible health) to 1 (best possible health). The VAS score ranges from 0 to 100, with higher scores indicating a better health perception. Besides these unidimensional measures, quality of life was also measured using the multidimensional WhoQol Bref questionnaire. 26 This questionnaire consists of 26 questions on the quality of life in 4 dimensions: physical quality of life, psychological quality of life, social factors of quality of life, and environmental factors of quality of life. Psy-chological quality of life and social factors of quality of life were used as outcome measures in this study. These domains can be transformed into a scale ranging from 4 to 20, and higher scores indicate a better perception of quality of life in the specific domain.
To assess actual memory capacity, we used the Dutch version of the Auditory Verbal Learning Test (AVLT) 27 and Story Recall from the Rivermead Behavioural Memory Test (RBMT). 28 The AVLT was used prior to and immediately after the intervention in the same form. The score of the AVLT ranges from 0 to 60 in the immediate recall condition and from 0 to 15 in the delayed recall condition (maximum number of remembered words). Parallel versions of the Story Recall of the RBMT were used before and after the intervention. The score of the RBMT stories ranges from 0 to 42 (maximum number of story elements of 2 stories). We used the delayed recall score of the AVLT and the delayed recall score of the RBMT as outcome measures.
Other Measurements
We used several measurements to determine predictive factors of MSE in the experimental group. Based on previous findings, we identified potentially predictive factors, including demographic variables, coping, personality traits, and executive functioning.8,9 Patient charts were obtained, and semistructural interviews were performed to gather information on age, gender, type and localization of stroke, comorbidity, time postonset (time from stroke until baseline measurement), education level, marital status, and work status. Coping was measured using the Assimilation-Accommodation Coping Scale. 29 Personality was measured using the Eysenck Personality Questionnaire Brief version, using the subscales that measured extraversion and neuroticism. 30 The presence of aphasia was assessed using the Token Test, Boston Naming Test and Word Fluency Test.31-33 The latter was also used to assess executive functioning. The Trail Making Test, a visual scanning task, and 2 subtests (zoo map and key finding) of the Behavioural Assessment of the Dysexecutive Syndrome were used to assess executive functioning as well.34,35
Procedure
All tests and questionnaires were administered in a fixed order, starting with the MIA, to minimize the effect of the memory tests on subjective memory experiences. All neuropsychological tests were performed in the same order during both measurements. Patients were randomized following a block design per group and per center in fixed blocks of 10 groups. When patients enrolled in the study, they were placed on a waiting list. If at least 4 patients had been included, a new group was started. The allocation of the group to either the experimental or control condition was performed by an independent investigator who was not otherwise involved in this study. If more patients were included prior to the start of the group, a maximum of 2 patients could be added to the group, resulting in a maximum group size of 6 patients. All groups consisted of 4 to 6 participants, allowing sufficient interaction but also limiting distraction. The time per session was set at a maximum of 1 hour taking into account possible attention deficits.
Statistical Analyses
All statistical analyses were performed using SPSS PASW version 18 for Microsoft Windows. Potential differences between groups in the baseline characteristics were analyzed using independent t tests for continuous variables and χ 2 tests for categorical variables. Changes over time in continuous outcome measures were studied for each group using paired t tests. The differences between the groups over the intervention period were analyzed using linear regression analyses. Each outcome after the intervention period was entered as a dependent variable in the regression model adjusted for the baseline measurement, which was entered as a covariable. For the group comparisons, a second covariable was added to the model that indicated to which group the participant was allocated (1 for the experimental, 0 for the control group), following the intention-to-treat principle. Significance of this grouping variable indicates a statistically significant difference in outcome between the 2 groups. In case of missing follow-up data, the data of the T0 measurement was imputed at T1, according to the intention-to-treat principle (last observation carried forward method; LOCF). A P value of <.05 was used for statistical significance.
Predictors for MSE outcome after the intervention were determined using multivariable regression analyses with backward variable selection, adjusted for MSE at baseline. Only baseline determinants that predicted MSE outcome in a bivariable analysis adjusted for baseline MSE, were entered in the multivariable model if the P value was <.10. Potential predictors included patient and stroke characteristics, time postonset, aphasia, executive functioning, personality, depressive symptoms, coping style, quality of life, participation, and memory capacity, all measured at baseline. Only patients who entered the intervention group were included in these analyses. A P value <.05 was used as the significance level for inclusion of variables and >.10 for removal of variables.
Results
Study Population
The inclusion of patients is described in Figure 1; 6 patients decided to withdraw from the study after providing informed consent, and 5 patients were excluded because of the existence of dementia (n = 2) or severe language disorders (n = 3). A total of 153 patients enrolled in either the experimental or the control condition.
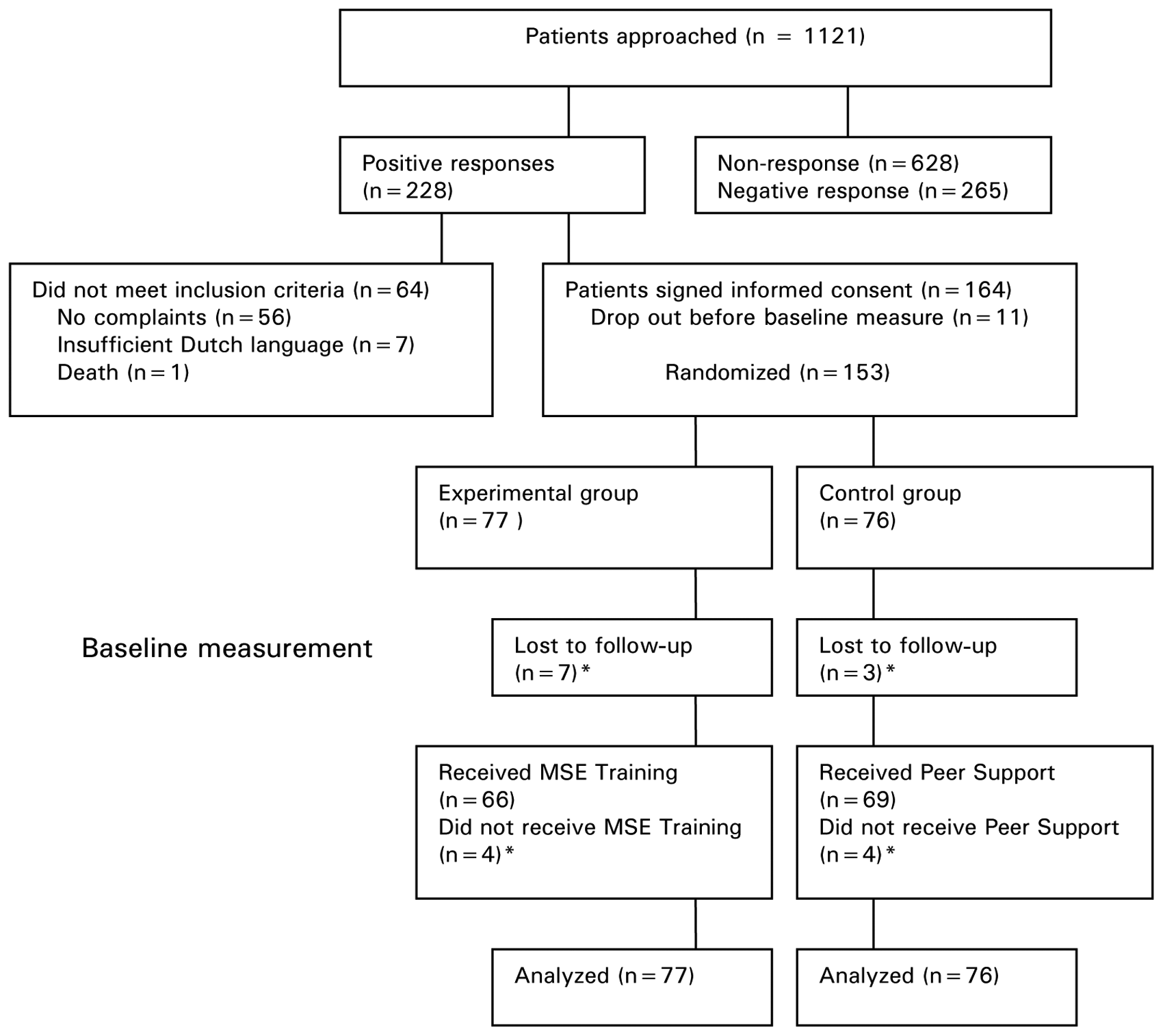
Flowchart of patient inclusion
Patient Characteristics
The mean age of the total study population was 58 years and 54.9% were male. Mean time postonset was 53.9 months (standard deviation [SD] = 37.2 months). In 57% of the population, the stroke was left sided, and the type of stroke was ischemic in 68% of the population. Table 1 presents the patient characteristics and baseline outcomes of the 2 separate groups. There were no significant differences between the experimental and the control group in any of these variables, except for the social component of quality of life (P < .04).
Baseline Characteristics a
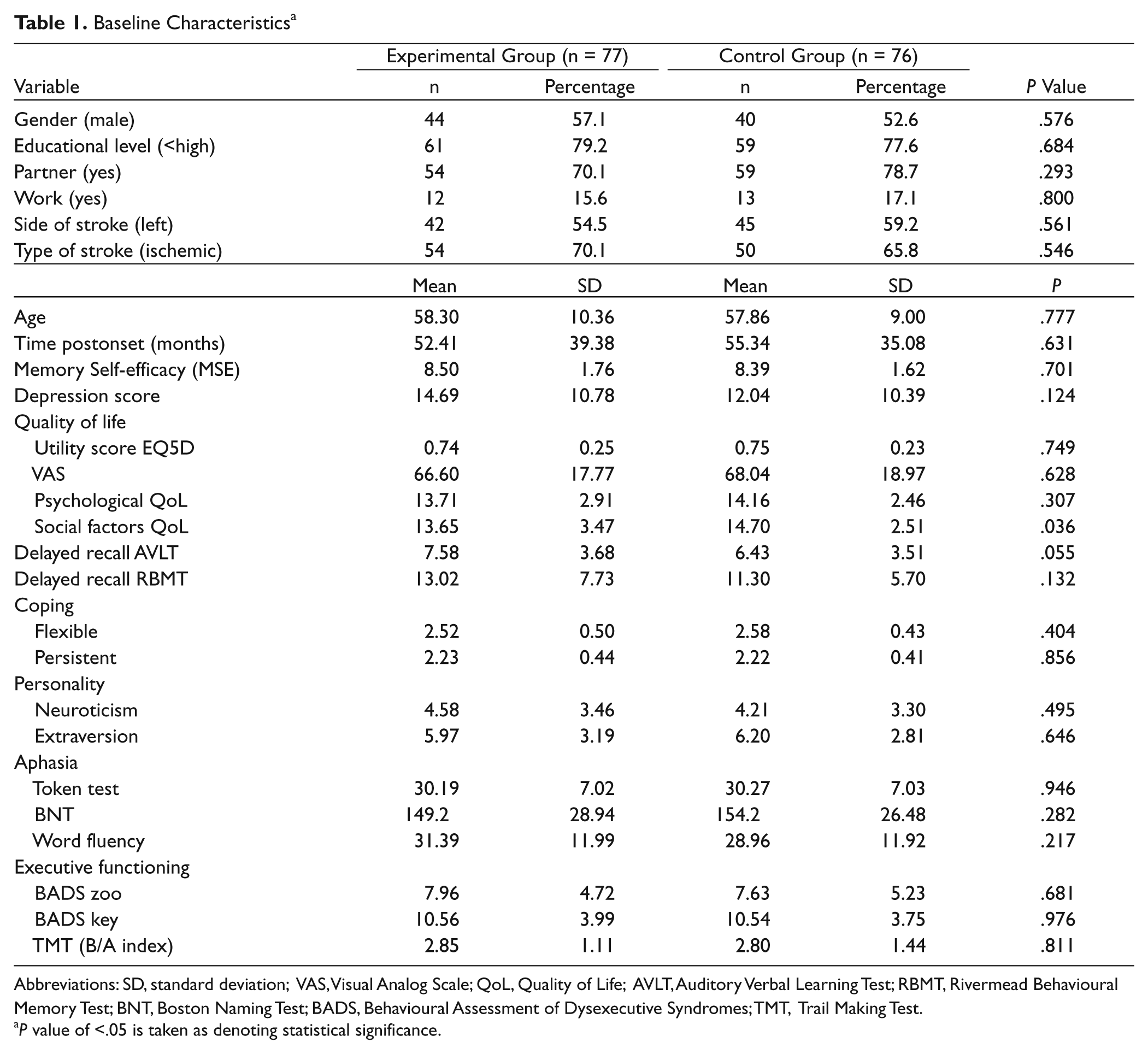
Abbreviations: SD, standard deviation; VAS, Visual Analog Scale; QoL, Quality of Life; AVLT, Auditory Verbal Learning Test; RBMT, Rivermead Behavioural Memory Test; BNT, Boston Naming Test; BADS, Behavioural Assessment of Dysexecutive Syndromes; TMT, Trail Making Test.
P value of <.05 is taken as denoting statistical significance.
Effects of MSE Training
Over the intervention period, the MSE score, the psychological health component of quality of life, and the delayed recall score of the AVLT significantly improved in the experimental group (Table 2). In the control group, the delayed recall score of the AVLT and the RBMT improved significantly over time. Comparing the changes over the intervention between both groups, we found that MSE increased significantly more in the experimental group than in the control group (Table 2).
Mean Differences Over the Intervention Period in the Experimental and Control Groups and Differences Between the Groups
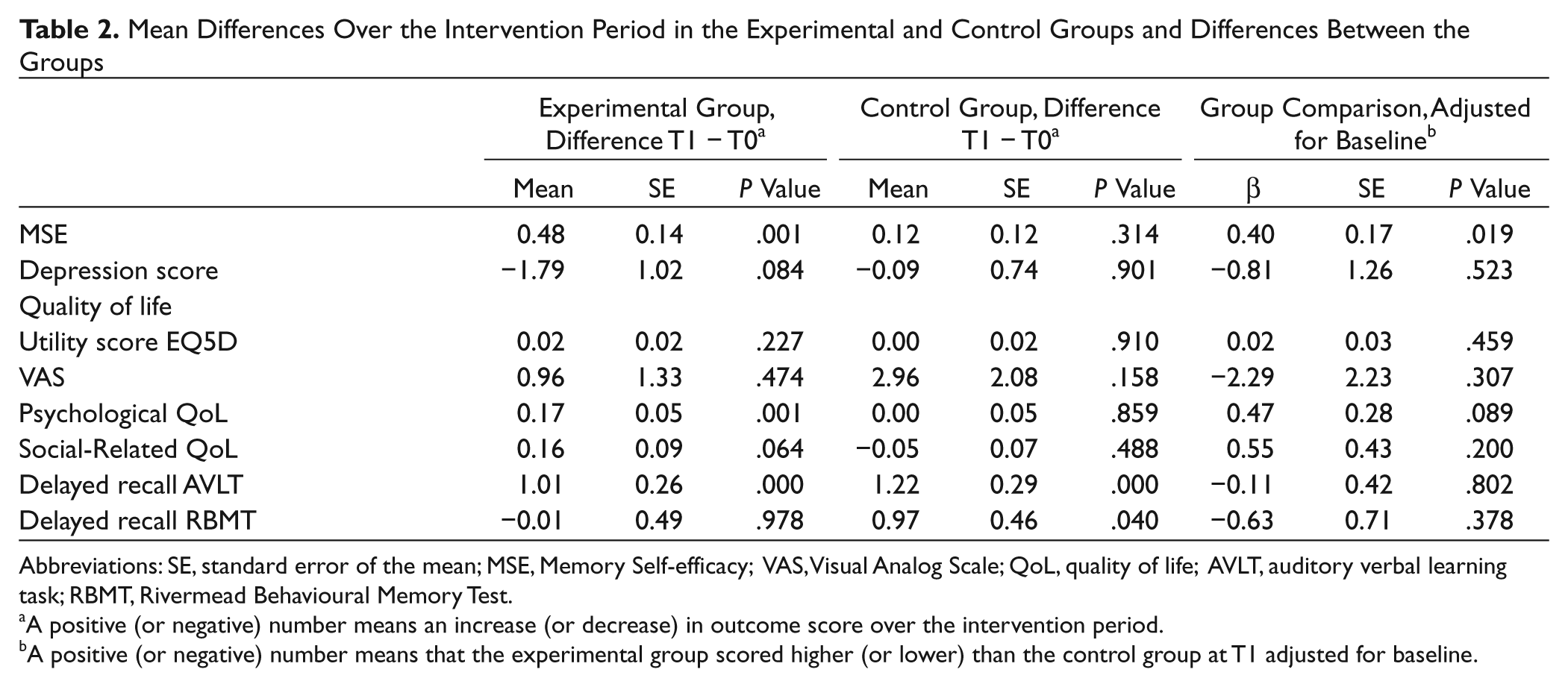
Abbreviations: SE, standard error of the mean; MSE, Memory Self-efficacy; VAS, Visual Analog Scale; QoL, quality of life; AVLT, auditory verbal learning task; RBMT, Rivermead Behavioural Memory Test.
A positive (or negative) number means an increase (or decrease) in outcome score over the intervention period.
A positive (or negative) number means that the experimental group scored higher (or lower) than the control group at T1 adjusted for baseline.
Predictors of MSE Outcome
Significant predictors of MSE outcome in bivariable analyses adjusted for MSE at baseline were age (B = −0.026; P = .037), delayed recall score of the RBMT (B = 0.039; P = .021), and stroke located in the brainstem (B = −0.855; P = .030). In the multivariable regression analyses, we found that patients with a better delayed recall score of the Story Recall of the RBMT (B = 0.043; P = .009) and patients who were younger (B = −0.033; P = .006) had a significantly higher MSE score after the MSE training (Table 3). All other demographic and clinical variables had no predictive value for MSE outcome after the MSE training when adjusted for MSE scores at baseline.
Multivariable Prediction Model (Final) for MSE Outcome After the Intervention Period (R2 = 0.574) a
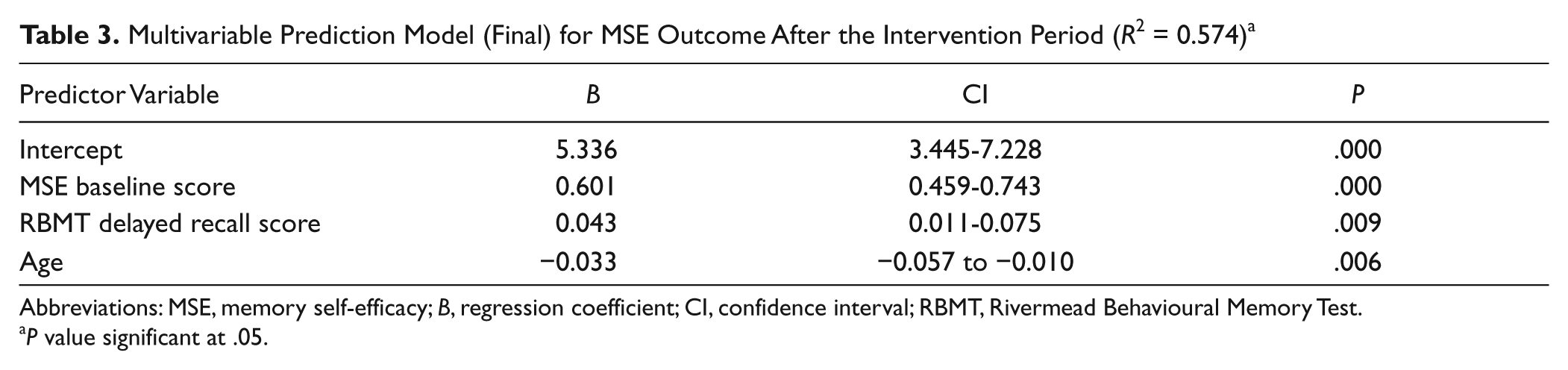
Abbreviations: MSE, memory self-efficacy; B, regression coefficient; CI, confidence interval; RBMT, Rivermead Behavioural Memory Test.
P value significant at .05.
Discussion
This study showed that the MSE training program for patients in the chronic stage after stroke is effective in improving MSE. The second aim of the study was to identify potential predictors of MSE outcome. Younger patients and/or those with a better verbal memory capacity may benefit more from attending the MSE training program. These findings are useful in targeting patients for an MSE-intervention. Elderly patients might need additional sessions, individual attention, or different training forms to achieve the same results as younger patients. Age could also be a limiting factor. Similar changes in the training program might be needed for patients with more memory deficits.
Over the intervention period, the psychological component of quality of life improved significantly in the experimental group but not in the control group. This suggests that improving MSE may result in a better psychological quality of life, but the difference in improvement between the 2 groups was not statistically significant. Nevertheless, this may indicate that further development of the MSE training program will possibly result in increased psychological quality of life of the patients.
Cicerone and others 9 recommend a combination of strategy training, errorless learning, and metacognitive training for TBI patients to enhance memory performance. For stroke patients, external compensation strategies are recommended as a practice standard for remediation of memory deficits. 9 Nair and Lincoln, 36 however, state that there is insufficient evidence for memory rehabilitation for stroke patients because of the low methodological quality of current studies. Our study is, to our knowledge, the first large trial on metacognitive memory training for stroke patients. 9
No significant improvement in depression and quality of life was found between both groups. Several factors may explain this. First, long-term results might be needed to achieve improvements on the domains of psychological and social quality of life. More time may be required to integrate newly learned information into daily life routines in order to have an effect on depression or quality of life. Second, the quality-of-life measure used is a self-report scale and subject to coping and response shifts. In future research, measures for social participation or other domains of daily functioning should be included.
There are several limitations to the study. The inclusion criteria resulted in a heterogeneous study population, as shown in the large SDs of the mean values of the outcome measures. Small improvements are difficult to detect in such a heterogeneous population. Possibly, training effects increase if treatment is targeted at specific subpopulations. Furthermore, we did not perform a Bonferroni or other correction method for multiple testing because the use of these methods would increase the chance of a type II error: missing actual treatment effects. However, we did use a conservative way of data analysis with the LOCF (last observation carried forward) method, assuming that MSE training in dropouts would not have been effective. If we had performed a per protocol analysis, the group differences would have been larger. Finally, we included patients in the chronic stage after stroke, in which no change or even decline of functioning may be expected. Therefore, the change in MSE, however small, may be considered as promising and relevant. Taking all limitations into account, our findings could indicate that memory complaints in stroke may be reduced with an intervention aimed at MSE. Future research is still warranted and should be aimed at earlier intervention, long-term follow-up, broadening of the outcome measures, and adjusting the intervention for specific groups of stroke patients.
Conclusion
This study is the first to investigate the integration of MSE into memory programs for stroke patients in a large randomized trial and to show that MSE can be improved in a chronic stroke population. The training program seems particularly effective in younger patients and in patients with a reasonable level of cognitive functioning and also seems to improve the psychological component of quality of life. Long-term investigations should reveal whether the effects fade out over time or whether other aspects of functioning and quality of life may improve.
Footnotes
Acknowledgements
The researchers thank all participating patients; Marieke Visser, Arlette Thiellier, Wendy Boerboom, and Anushka Wardenaar for performing measurements; and Henk de Geus for his help with improving the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by the Kinder Fonds Adriaanstichting (Children’s fund Adriaanstichting) Grant Number: 07.03.28-2007/0082. This study is registered with the Dutch Trial Register (NTR) TC 1656.
