Abstract
Background. Ambulatory tasks are the important components of balance training which effectively improve postural stability and functional activities in persons with Parkinson’s disease (PD). The difficulty level of an ambulatory task is usually set in the form of attention, direction, speed, or amplitude requirement. Objectives. This study aimed to explore the neural mechanisms of cerebral hemodynamic responses to the difficulty level of ambulatory tasks in persons with PD. Methods. We included ten studies that examined cerebral hemodynamic responses during ambulatory tasks at different difficulty levels in persons with PD. The change in hemodynamic responses was synthesized and meta-analyzed. Results. Patients during “ON” medication had higher relative change in oxygenated hemoglobin (ΔHBO2) in the prefrontal cortex in response to difficulty levels of ambulatory tasks, which is comparable to that in healthy elderly individuals. However, patients during “OFF” medication did not show cortical activation in response to difficulty levels. During the lower-difficulty tasks, patients during “ON” medication demonstrated higher ΔHBO2 than healthy elderly participants and patients during “OFF” medication. Factors found to significantly contribute to the heterogeneity across studies included subjects’ type and cognitive status, task duration, setting, and filter used for functional near-infrared spectroscopy (fNIRS) data pre-processing. Conclusions. The findings suggest that ambulatory task at a higher difficulty level could be necessary to train the cortical capacity of PD persons, which should be conducted during “ON” medication; meanwhile, the contributing factors to the heterogeneity of studies would be useful as a reference when designing comparable fNIRS studies.
Keywords
Introduction
Parkinson’s disease (PD) is a progressive nervous system disorder that affects movement and gradually leads to postural instability 1 and eventually limits functional activities, especially ambulatory activities, such as walking and turning. 2 Ambulatory activities are more fall-prone activities than standing and transferring in persons with PD. 3 Individuals during ambulatory activities are always required to adjust their posture and movement in speed, amplitude or direction, and/or regulate their attention to cope with various tasks or environments, which could be a key reason for ambulatory activities inducing falls more often. For example, when answering a doorbell, individuals may speed up their pace, or with more attention on the task, when turning, individuals may need to change the direction of their steps. Poor posture and movement adjustment during these activities further induce dynamic postural instability, leading to high risk of falls.4-6 Compared with other motor symptoms, postural instability has been shown to be less sensitive to mainstream treatment with dopamine supplementation. 7 The hypothesis is that the pathophysiology of postural instability in PD might not be limited to the dopaminergic basal ganglia motor loop, but may also involve other neural networks within the cortex.7,8
Physical therapy, specifically exercise training with balance-training components, has been an important intervention, in conjunction with current pharmacological treatment to improve postural stability and functional activities in individuals with PD. 9 As an exercise-training component, ambulatory activities are commonly involved. 9 The difficulty level of ambulation training is usually set in the form of speed, amplitude, direction, or attention requirement, 10 similar to the intensity of resistance training adjusted through resistance levels. 10
Clarification of the mechanisms underlying ambulatory activities, especially at different difficulty levels, is greatly helpful in designing the training protocol. Recently, real-time imaging of the cortical control of ambulatory tasks has become possible with functional near-infrared spectroscopy (fNIRS). 11 Functional near-infrared spectroscopy is a non-invasive optical imaging technique that measures the human cerebral hemodynamic response by means of the specific absorption spectra of hemoglobin in the near-infrared range. 12 Some researchers have adopted fNIRS technology to investigate the cortical control between different ambulatory tasks in persons with PD.13-19 This is the first systematic review that explores how the difficulty level of ambulatory tasks affects the cerebral hemodynamic responses in persons with PD. Since most previous studies have additionally compared the cortical responses between PD patients and age-matched healthy subjects, this was systematically reviewed as well. Furthermore, the factors that affect the difference in cerebral hemodynamic responses to difficulty levels of ambulatory tasks were analyzed when heterogeneity was present across studies.
Methods
Search Criteria
We conducted an extensive literature search on electronic databases, including PubMed, Web of Science, Cochrane Library, Academic Search Premier via EBSCOHost, PEDro database, China National Knowledge Infrastructure, and China Wanfang Database. The languages were restricted to English and Chinese, and the search was up to March 1, 2021. The keywords used to conduct the literature search were combined with the following English terms: ((Parkin*) AND (balance OR equilibrium OR walking OR gait OR gaits OR ambulation OR negotiation OR Locomotion OR turning OR mobility) AND (fNIRS OR Functional Near-Infrared Spectroscopy OR Hemodynamic OR Hemodynamics OR Hemodynamic*)). The reference lists of relevant articles were further checked to identify potential studies.
Selection Criteria
Studies eligible for this review were those that (1) adopted fNIRS to monitor the cerebral hemodynamic response during ambulatory tasks in persons with PD and (2) set different difficulty levels of ambulatory test tasks. The difficulty level of ambulatory tasks could be set in forms of attention, direction, speed, or/and amplitude. For example, a dual walking task has a higher difficulty level than a single walking task since the former requires higher attention than the latter. In other words, attention is the difficulty parameter between dual and single walking tasks. Similarly, speed is the difficulty parameter between comfortable walking and fast walking, and direction is the difficulty parameter between normal walking and turning.
Two researchers (the first and second authors) determined the eligibility of studies by screening the title, abstract, and full text. Articles were excluded if they were not original studies such as review studies, if the article did not use fNIRS to monitor the cerebral hemodynamic response during tasks, if they were not related to ambulatory tasks,20-24 and if ambulatory tasks did not have at least two difficulty levels in forms of attention, direction, speed or amplitude.25-27 Any divergence of opinion on eligibility was resolved by discussion with a third party (the corresponding author).
Data Extraction and Quality Assessment
Two researchers developed a data extraction sheet and worked independently on data extraction. The extracted data comprised the characteristics of the subjects and task paradigm, features of fNIRS setting and data analysis, and cerebral hemodynamic measures. The subjects’ characteristics involved their health status (PD or healthy), sample size, age, sex, cognition, as well as the duration and severity of PD, medication, and freezing of gait history only for subjects with PD. Based on the subjects’ characteristics, some studies divided patients into subgroups during “ON” or “OFF” medication and with or without freezing of gait. The subgroups were separated for data extraction. The features of the task paradigm included the manner and difficulty parameter of ambulatory tasks, and the phasic duration, and number of blocks. The attributes of the fNIRS measurement contained the fNIRS parameters presented in the articles, cortical regions of interest monitored, the wavelength, distance from transmitter to detector, and sampling frequency. The features of the fNIRS data analysis comprised filter frequency and baseline period referred to calculate the hemodynamic concentrations during tasks.
The measures of fNIRS comprised relative change (Δ) in oxygenated hemoglobin (HBO2) and deoxygenated hemoglobin (HHB) concentrations in specific brain functional regions during certain ambulatory tasks by subtracting the baseline concentration. The data for each brain functional region were extracted separately.
After data extraction, the two researchers compared the accuracy of the data.
Quantitative Data Synthesis and Analysis
The hemodynamic measures consisted of ΔHBO2 and ΔHHB in diverse cortical regions during ambulatory tasks. Only one hemodynamic measure in each of the eligible studies was pooled for meta-analysis. To minimize heterogeneity across studies, ΔHBO2 in the prefrontal cortex (PFC) was chosen for this meta-analysis because it is a unique measure presented in all studies.13-19,27-29
Comprehensive Meta-Analysis software (Version 3, Biostat, Englewood, New Jersey) was used to perform the meta-analyses. To investigate how the difficulty levels of ambulatory tasks affected the cerebral hemodynamic response in persons with PD, the ambulatory tasks were divided into two levels of difficulty that were higher or lower in the form of difficulty parameters. Some studies had one more difficulty parameter for comparison although only one difficulty parameter was pooled for meta-analysis. The pooling priority of the difficulty parameters was ranked from attention and direction to speed, based on the frequency used in these studies. The effect size for difference in cerebral hemodynamic responses to difficulty levels of ambulatory tasks was calculated using Hedges’ g by dividing the difference of means of response between ambulatory tasks at each difficulty level through the estimated pooled standard deviation. The pooled standard deviation of cerebral hemodynamic response was estimated by the standard deviation of response during ambulatory tasks at each difficulty level, with bias corrected from the sample size in each group.
The cortical hemodynamic responses to difficulty levels of ambulatory tasks were meta-analyzed separately in subgroups of PD patients during “ON” and “OFF” medication and healthy subjects. The difference between subject groups was further analyzed using subgroup analysis. I2 statistics were used to assess statistical heterogeneity across studies with different subject groups. If there was significant heterogeneity across studies, the random-effects model of meta-analysis was used; otherwise, the fixed-effects model was used to analyze the training effects. 9 Egger’s test was used to analyze publication bias to determine whether there was a correlation between the effect size and sample size.
For the heterogeneity on hemodynamic response to the difficulty level of ambulatory tasks, a three-step meta-regression was conducted to detect contributing resources. First, each potential factor about the subjects, task paradigm, and fNIRS setting and data analysis was entered into single-variable regression model. Then, the potential factors with significant or near-to-significant contribution in the single-variable model were entered into a mixed-variable regression model. Last, the potential factors with significant or near-to-significant contribution in the mixed regression model or with significant change by the test of change were entered into the final mixed-variable regression model.
Before entering into the first mixed-variable model, correlations between each potential factor were analyzed by Pearson’s test using SPSS (Statistical Product and Service Solutions version 25.0). Among those with strong correlations (r > .70), only one factor was entered into the first mixed-variable model, which had the highest proportion of variance explained using its single-variable model.
In addition, the response during less difficult ambulatory tasks between PD patients and age-matched healthy subjects were meta-analyzed to better understand the precondition of their cerebral adaptation on less difficult ambulatory tasks.
The level of significance was set at P < .05 for all statistical tests.
Results
Trial Flow and Study Characteristics
A total of 257 potentially eligible articles were identified by database searches, and no gain was identified by checking the reference lists of relevant articles. After removing 81 duplicated articles, 176 articles remained and were further screened by the title, abstract, and full text of the article. Finally, 10 articles were included in the data synthesis and meta-analysis (Figure 1). PRISMA flow diagram showing the trial flow.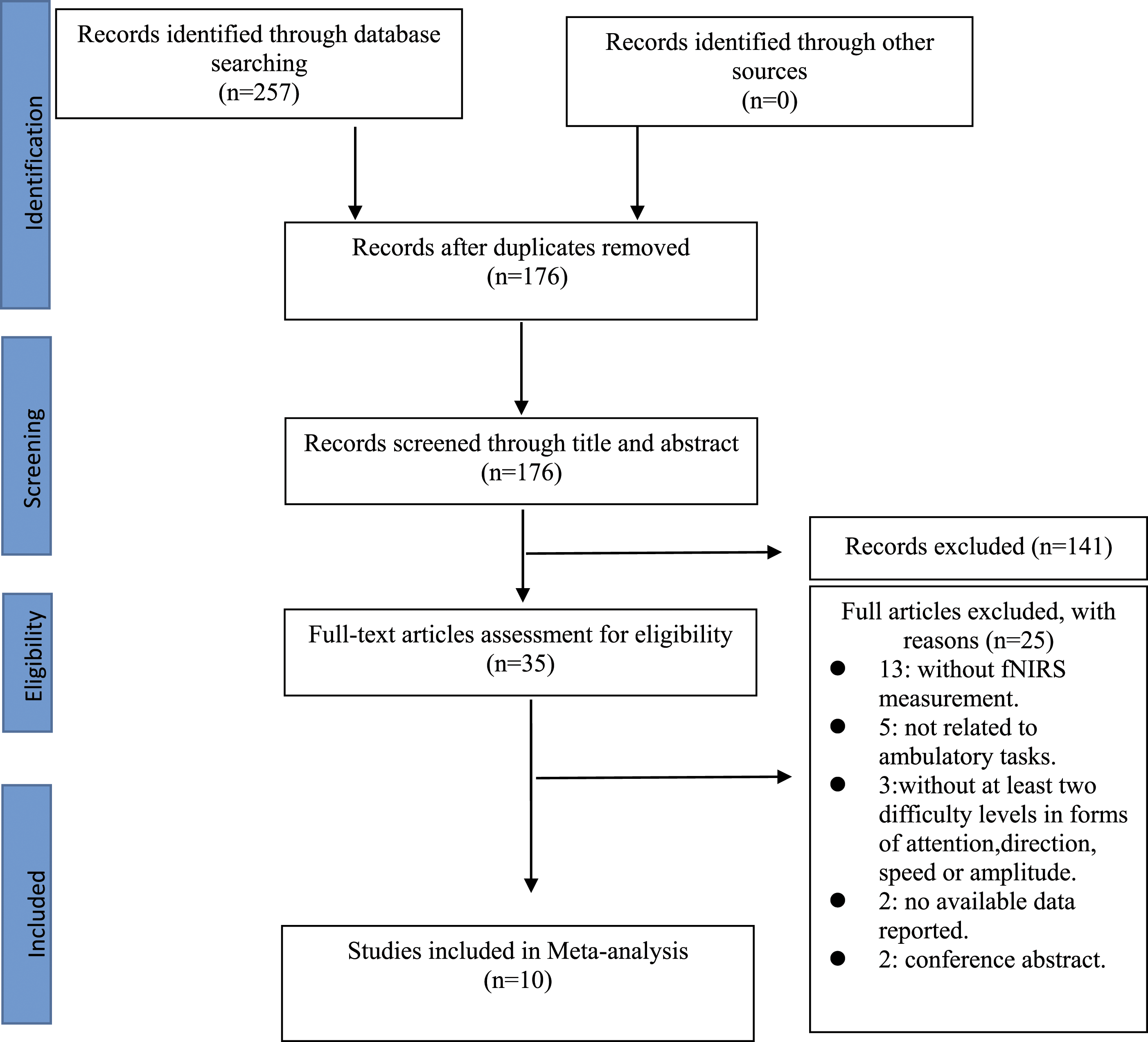
Among them, six studies targeted age-matched healthy subjects as well as PD patients, who were included to further explore the response in healthy subjects and difference between the two subject groups.13-15,17,19,27
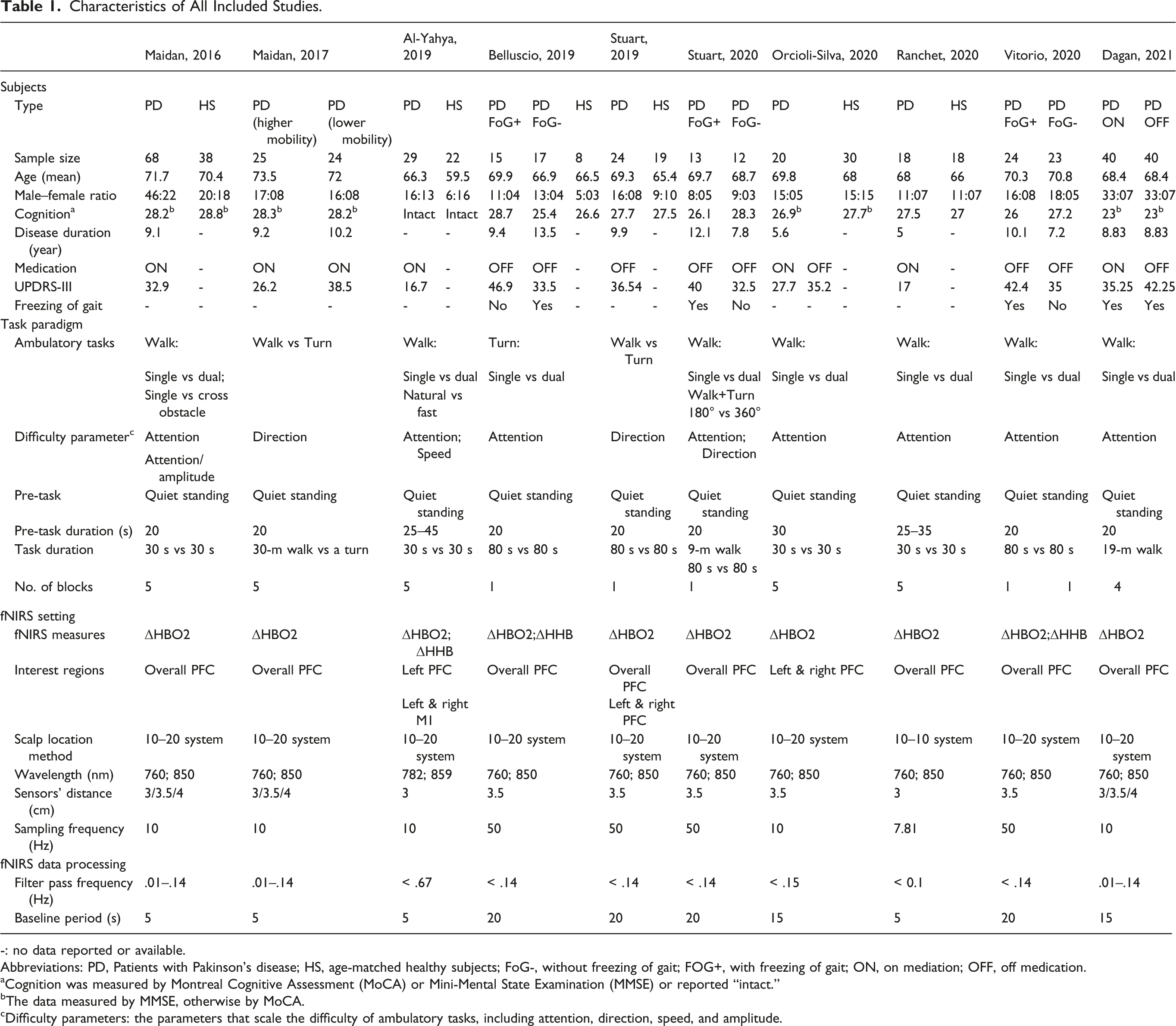
A total of 392 persons with PD and 135 healthy elderly were included in these studies. The mean age ranged from 66.3 to 73.5 years in persons with PD and 59.5 to 70.4 years in healthy elderly. The sex ratio (male:female) ranged from 1.23 to 4.7 in persons with PD and from .38 to 1.67 in healthy persons. For cognition, all studies set the ability to follow instruction or intact cognition as one of the selection criteria. All studies adopted the Mini-Mental State Examination or Montreal Cognitive Assessment to quantify cognition, except one that excluded subjects with cognitive impairment through selection criteria. 14 For PD patients, the disease duration ranged from 5.6 to 13.5 years. The severity measured by the Unified Parkinson’s Disease Rating Scale III ranged from 26.9 to 46.9 points of the mean. Seven groups of PD patients were assessed during “ON” medication, whilst nine groups during “OFF.” A history of freezing of gait was reported in four studies.
Among the studies, the ambulatory tasks included walking with different speeds, dual walking, or turning. The resting duration before tasks was 20 s in most studies, 30 s and 45 s in one study,13,27 or 25 to 45 s arranged in a pseudo-random order. 14 The task duration was 30 s,13-16,27 80 s,17-19,29 or time of a walking bout.18,28 The block paradigm for ambulatory tasks was designed in six studies.13-16,27,28
Characteristics of All Included Studies.
-: no data reported or available.
Abbreviations: PD, Patients with Pakinson’s disease; HS, age-matched healthy subjects; FoG-, without freezing of gait; FOG+, with freezing of gait; ON, on mediation; OFF, off medication.
aCognition was measured by Montreal Cognitive Assessment (MoCA) or Mini-Mental State Examination (MMSE) or reported “intact.”
bThe data measured by MMSE, otherwise by MoCA.
cDifficulty parameters: the parameters that scale the difficulty of ambulatory tasks, including attention, direction, speed, and amplitude.
Hemodynamic Response to Difficulty Level of Ambulatory Tasks Among Subject Groups
Ten studies with 22 subgroups were included in the meta-analysis. There were three kinds of subject groups including nine subgroups of patients during “OFF” medication, seven subgroups of patients during “ON” medication, and six subgroups of healthy subjects. The heterogeneity of effect size was large across each subject group, I2 = 98% in patients during “ON” medication, I2 = 83% in patients during “OFF” medication, and I2 = 92% in healthy subjects. A study of Maidan et al. 16 with patients during “ON” medication as subjects showed large residual standard deviation of the effect size (>2), which is ultra-low probability data (<5%) and was therefore excluded from the further subgroup analysis and meta-regression analysis. After removing the study, 16 the heterogeneity across studies with patients during “ON” medication reduced to I2 = 93% (P < .05).
The meta-analysis showed a positive effect of the difficulty level on ΔHBO2 in the PFC (P < .05) in patients during “ON” medication (g = .945, 95% confidence interval [CI]: .074-1.817, P < .05) and healthy subjects (g = 1.759, 95% CI: .706-2.811, P < .05), but negative effect in patients during “OFF” medication (g = −.481, 95% CI: −1.001-.038, P > .05) (Figure 1). Subgroup analysis showed significant difference across three kinds of subject groups (P < .05), which existed in patients during “OFF” medication compared with patients during “ON” medication or healthy subjects (both P < .05), but did not exist between patients during “ON” medication and healthy subjects (P > .05) (Figure 1).
Factors Influencing Hemodynamic Response to the Difficulty Level of Ambulatory Tasks
Contributing Factors to Heterogeneity of Effect Sizes using 3-step Meta-Regression.
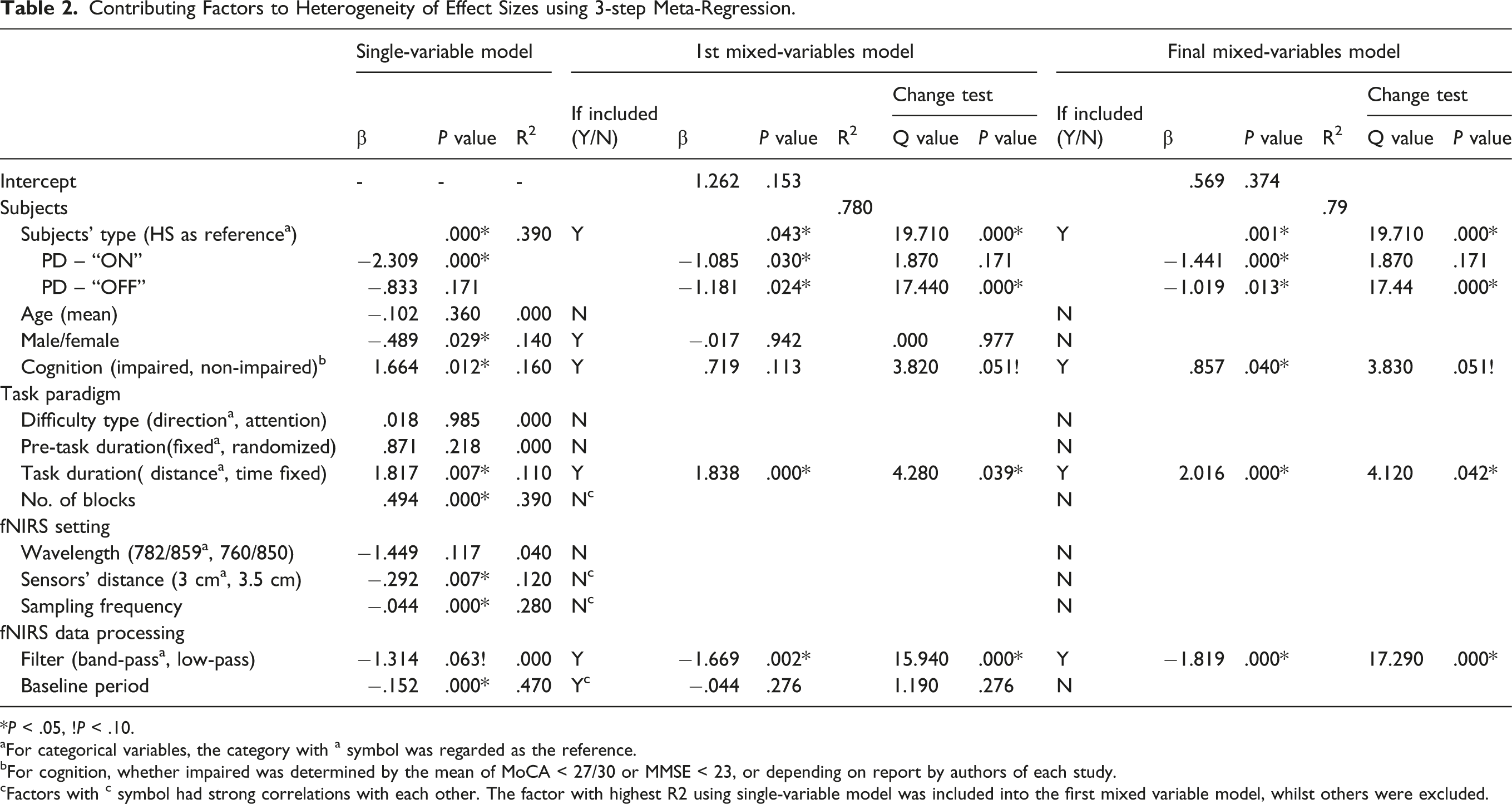
*P < .05, !P < .10.
aFor categorical variables, the category with a symbol was regarded as the reference.
bFor cognition, whether impaired was determined by the mean of MoCA < 27/30 or MMSE < 23, or depending on report by authors of each study.
cFactors with c symbol had strong correlations with each other. The factor with highest R2 using single-variable model was included into the first mixed variable model, whilst others were excluded.
Correlation between Potential Covariates in Meta-analyzed Studies (20 subgroups).
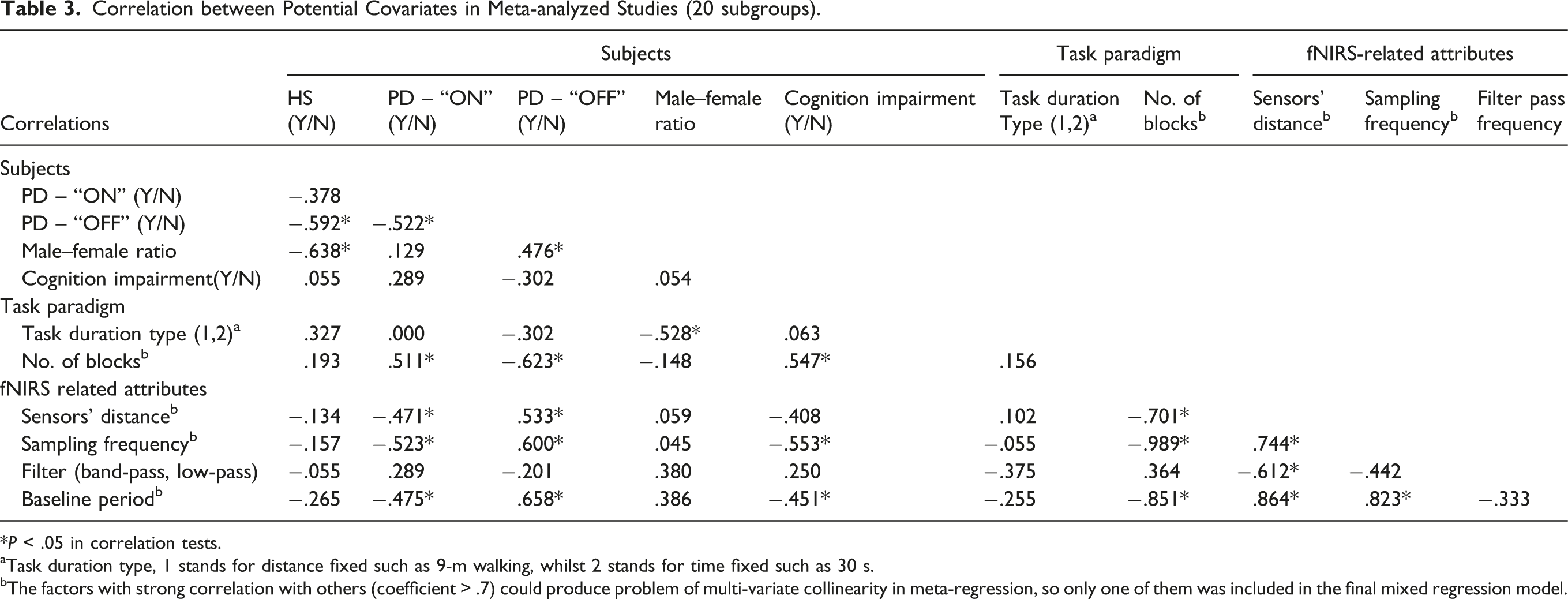
*P < .05 in correlation tests.
aTask duration type, 1 stands for distance fixed such as 9-m walking, whilst 2 stands for time fixed such as 30 s.
bThe factors with strong correlation with others (coefficient > .7) could produce problem of multi-variate collinearity in meta-regression, so only one of them was included in the final mixed regression model.
Hemodynamic Concentration During Less Difficult Ambulatory Tasks Among Subject Groups
The hemodynamic concentration during the less difficult ambulatory task was higher in patients during “ON” medication (mean = .385, 95% CI: 0.256–.514) than in healthy subjects (mean = .124, 95% CI: 0.044-.204) and patients during “OFF” medication (mean = .028, 95% CI: −.040-.095) (between-group difference: P < .05). No difference was observed between the healthy subjects and patients during “OFF” medication (between-group difference: P > .05) (Figure 2). Meta-analysis results of hemodynamic response to less difficult ambulatory tasks among patients with PD and healthy subjects.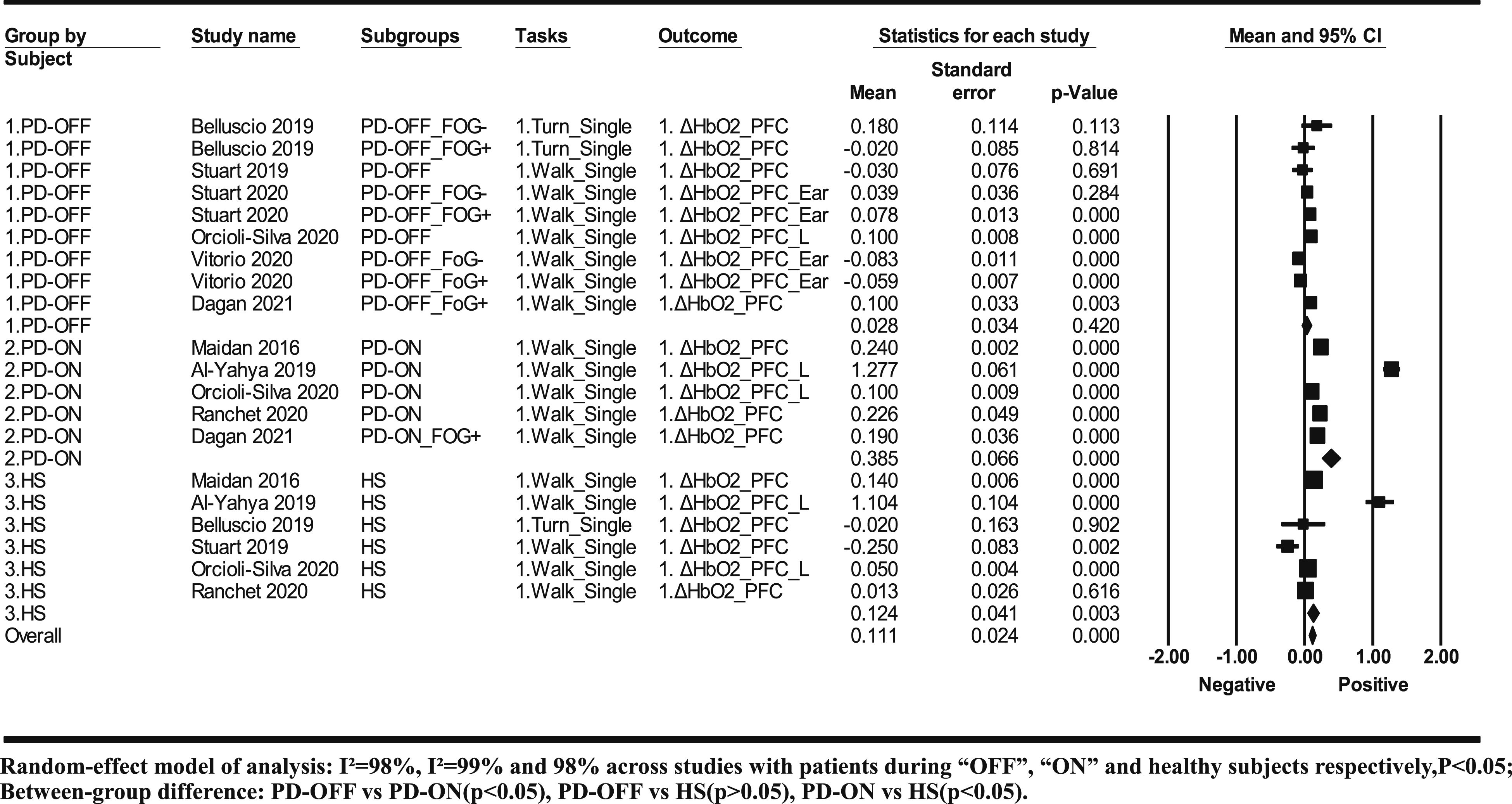
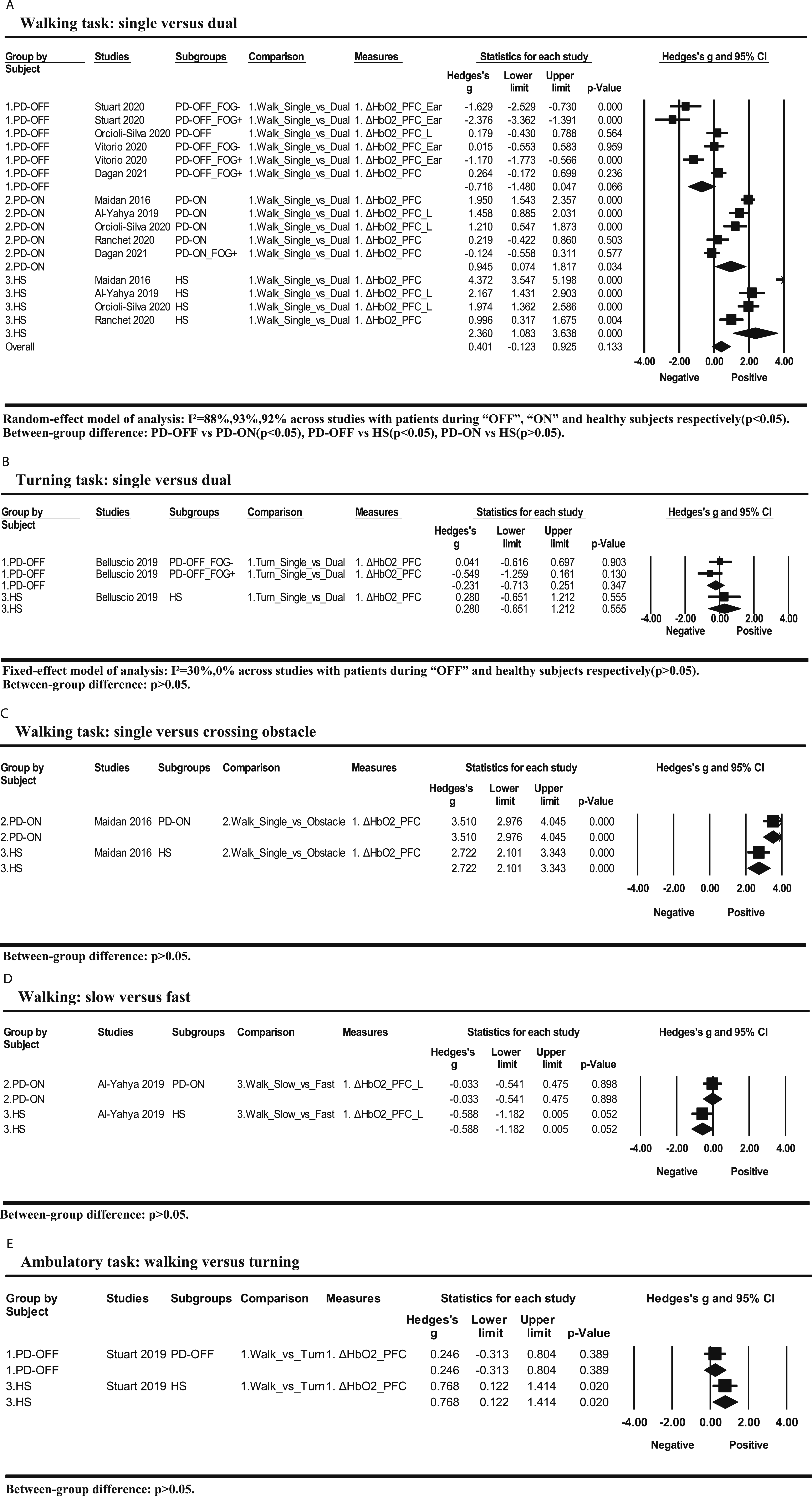
Not all ambulatory tasks were included in the overall meta-analysis (Figure 3). The hemodynamic responses to the difficulty level of each ambulatory task were presented (Figure 4), which will be systematically discussed. Meta-analysis results of hemodynamic response to difficulty levels of ambulatory tasks among patients with PD and healthy subjects. Hemodynamic responses to the difficulty level of different ambulatory tasks among patients with PD and healthy subjects.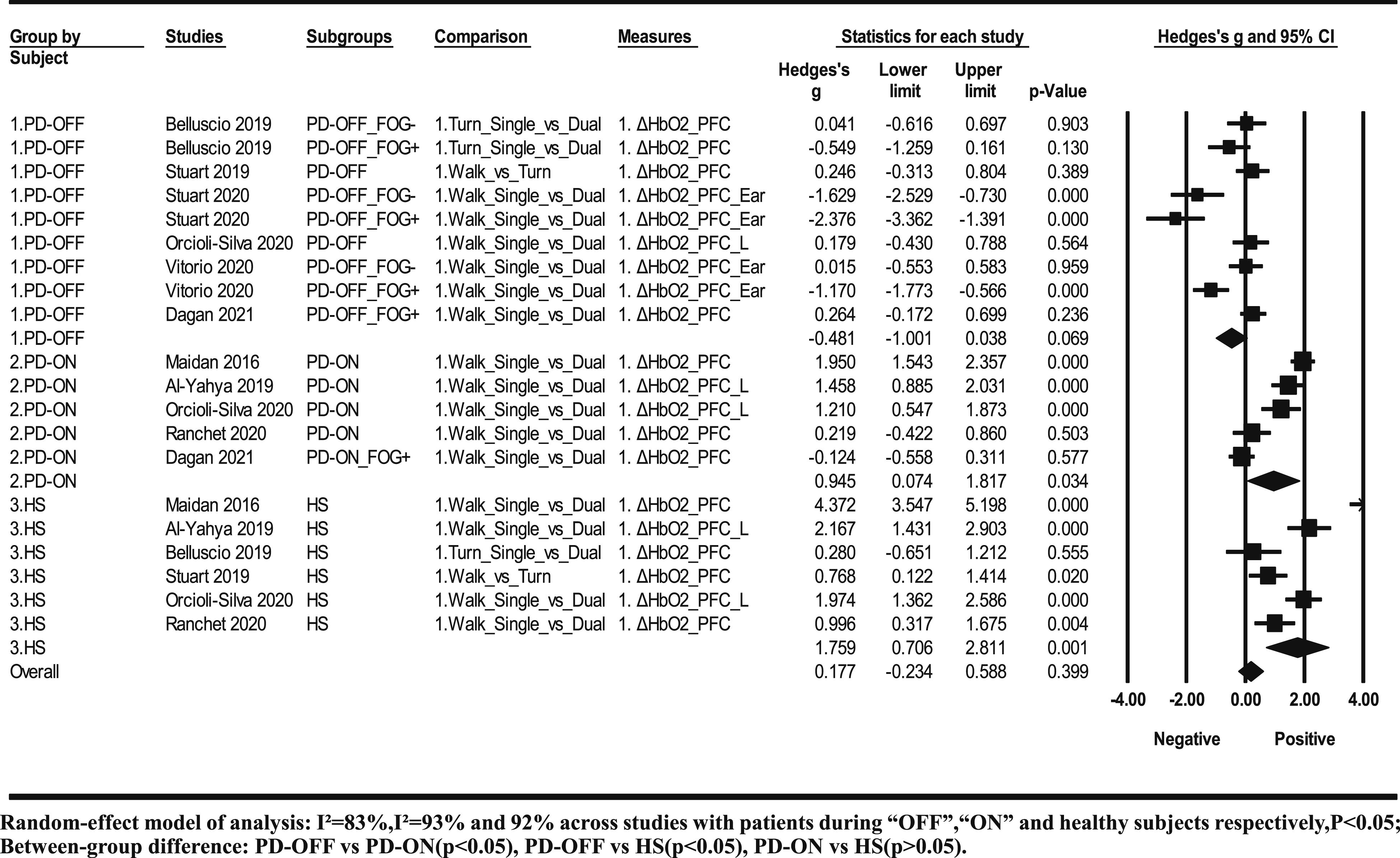
Discussion
This is the first systematic review to investigate the change in cerebral hemodynamic response to difficulty levels of ambulatory tasks in persons with PD using meta-analysis. The results showed that persons with patients during “ON” medication showed significant changes of cerebral hemodynamic response to the difficulty levels of ambulatory tasks as the age-matched healthy people (Both P < .05), whilst persons with PD during “OFF” medication did not change their hemodynamic response (P > .05). This indicates that PD impairs the capacity of cortical response to a more difficult ambulatory task, which supports the capacity limitation hypothesis. 30 Our results imply the effectiveness of PD medication in repairing the cortical capacity to meet the requirement of a more difficult ambulatory task. On the other hand, ambulatory tasks at some difficulty level could be necessary to train the cortical capacity of PD patients, which should be conducted during “ON” medication.
Although a comparable response to the difficulty level of an ambulatory task in patients during “ON” medication with the healthy elderly, persons with patients during “ON” medication showed more cortical activation than the healthy elderly during simpler ambulatory tasks. The results support another theory of neural inefficiency: this theory postulates that PD patients during “ON” medication show increased utilization of brain function associated with poorer or equivalent behavioral outcomes in comparison to the healthy elderly. 30 The neural inefficiency actually exists in healthy elderly as well while comparing with young persons. 31 The increased utilization of the PFC is suggested to be a neural compensatory strategy for functional impairment of neural circuits involving the basal ganglia. 22 Compared with “ON” medication, PD patients during “OFF” medication show an insufficient neural compensatory strategy at simpler ambulatory tasks.
Beside PFC, the M1 demonstrated similar neural inefficiency but sufficient capacity for more difficult ambulatory tasks in patients during “ON” medication, which was shown only in one study. 14 The premotor cortex and supplementary motor cortex are important neural components in balance control, which are suggested to form a motor optimization loop with the basal ganglia and cerebellum to produce mature action plans. 32 However, none of the studies included them as the regions of interest. Future studies are needed to look at these regions to gain insights into the cortical mechanism of balance control.
High heterogeneity is a common problem across fNIRS studies. 33 It has been proposed to result from three categories of potential factors, including subjects’ characteristics, task paradigms, as well as fNIRS settings and analysis methods. 33 This is the first study to explore the contributing factors of heterogeneity using meta-regression. Three steps of meta-regression were conducted to explore the contributing factors, considering their interaction between potential factors.
Four contributing factors of heterogeneity were found, including subject types, cognition impairment, task duration, and filter type for fNIRS data processing. Their prediction model could explain 79% of the variance across studies. The strongest contributing factors were subject types (PD – “OFF” compared with healthy subjects) and filter pass types (low-pass compared with band-pass), which had the highest Q value in the change test of the final mixed model (Q = 17.44 and 17.29, respectively). For subject types, the result of meta-regression further affirms the result of sub-group analysis, which has been discussed above. For filter types, our result indicates that low-pass is less sensitive to detect changes of cerebral hemodynamic response to the difficulty level of ambulatory tasks than the band-pass. Low-pass and band-pass filters are the commonest methods used for fNIRS data pre-processing. A recent review study on fNIRS signal filtering methods indicated that low-pass filter is not effective in removing the physiological very low frequency components in the fNIRS signals, leading to higher inter-subject variability than band-pass. 34 An ambulatory task has very low frequency components such as irregular postural adjustment, which cannot be removed by a low-pass filter, therefore resulting in lower sensitivity to detect meaningful results. A recent consensus guide to use fNIRS in posture and gait research has highlighted the necessity to compare the different filter methods since no consensus on the best filter method to remove motion artefact in posture and gait tasks exists. 35 Our finding should provide timely evidence to support the superiority of the band-pass filter.
Cognition impairment showed a negative effect on the changes of cerebral hemodynamic response to the difficulty level of ambulatory tasks. The finding concurs with a previous study exploring the effect of cognition impairment on cerebral hemodynamic response to cognitive tasks with different complexity levels. 36 For task duration, the method with fixed distance was found to be less sensitive to detect the changes of cerebral hemodynamic response to the difficulty level of ambulatory tasks than the fixed time. The mean of hemodynamic response was commonly used for data analysis, which was calculated through dividing by time. Tasks with fixed distance would lead to different task duration which could weaken the sensitivity of detecting difference.
The difficulty type has not showed significant influence on the changes of cerebral hemodynamic response to the difficulty level of ambulatory tasks. Based on priority order, the difficulty type included in meta-analysis was attention, followed by direction, which could limit observing ambulatory tasks with other difficulty types. We further included all kinds of ambulatory tasks to discuss through a systematic review. There were three kinds of ambulatory tasks with different attentional requirement, including single versus dual walking task, single versus dual turning task, and single walking versus walking with obstacle crossing. The comparison between the single and dual walking task showed similar meta-analysis results of overall ambulatory tasks mentioned above. Comparison between single and dual turning tasks was investigated by only one study with three subgroups. 20 However, the difference between patients during “OFF” medication and healthy subjects did not reach to significant level, but showed a similar trend as the overall meta-analysis that patients during “OFF” medication respond less than healthy subjects. Specifically, patients with a freezing of gait demonstrated poorer cortical response than those without freezing of gait. Comparison between single waking and walking with obstacle negotiation was investigated by only one study as well. 15 The study adopted two comparisons, single walking versus dual walking and versus walking with obstacle negotiation. They found that compared with healthy subjects, patients during “ON” medication had higher activation during single walking, less increase of activation during the dual task, but higher increase of activation during walking with obstacle negotiation. The results on single and dual walking tasks of the study concur with our meta-analysis result. Higher activation during obstacle negotiation but not during dual task in patients could indicate different cortical activation between the two tasks. Besides attention, speed was set as the difficulty parameter in one study. 14 Surprisingly, a high speed did not increase cortical activation in both patients and healthy subjects, which indicates different cortical activation between difficulty settings as well. Therefore, the meta-analysis finding that the difficulty parameter did not show influence on effect size of the cortical activation change in response to difficulty levels of ambulatory tasks should be interpreted with caution, which needs to be further tested with more studies by including other parameters. Comparison between walking and turning was conducted by a unique study as well. 17 It found a higher increase of cortical action during turning in healthy subjects than in patients during “ON” medication, which concurs with our overall meta-analysis results.
For the study of Maidan et al., which was excluded from the final meta-analysis due to its ultra-low probability data, the setting of the task could be the main cause resulting in the extreme difference with others. The study designed a 30-m walk followed by a 180° turn in a task block. The baseline and the task duration of walking and turning tasks were incomparable in this study, 16 in contrast with other studies.13-15,17-19
Despite its significant findings, this study has several limitations. First, publication bias existed in studies targeting persons with PD during “OFF” medication. We used Hedges’ g as the effect size for change in hemodynamic response to task difficulty level, which was adjusted with the sample size to reduce influence resulting from publication bias. Second, some of the included studies reported their results not in the form of data but figures. We had to estimate the data from these figures.13,18,27,28 Third, beside ΔHBO2, three studies have reported ΔHHB with significant findings.14,19,28 It could be not convincing evidence since fishing for significant results was the reason underlying the reporting of ΔHHB in those studies. Fourth, although PFC is the common region of interest across studies, the translation of optodes positioning to precise cortical regions remains a challenge. The international 10-20 system or 10-10 system is a common approach to define scalp locations as a percentage of the individual’s head size, which could minimize the influence of variability in brain morphology among individuals.
In conclusion, PD impaired the cortical capacity of increasing the activation level of patients in response to a more difficult ambulatory task. The impaired capacity could be repaired to match healthy elderly by PD medication. However, compared with healthy elderly, persons with PD still need to use an increased activation level of PFC to compensate for the insufficient neural function of ambulatory tasks even at lower difficulty levels. The findings suggest that an ambulatory task at a higher difficulty level could be necessary to train the cortical capacity of PD patients, which should be conducted during “ON” medication. A series of contributing factors detected by meta-analysis would be useful to direct the design of the fNIRS study to ensure comparability with previous studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China grant number 81802240.
