Abstract
Background
People with Parkinson’s disease (PD) have difficulties generating quick and accurate steps in anticipation of and/or in response to environmental hazards. However, neural mechanisms underlying performance in cognitively demanding stepping tasks are unclear.
Objective
This study compared activation patterns in cognitive and motor cortical regions using functional near-infrared spectroscopy (fNIRS) between people with PD and age-matched healthy older adults (HOA) during stepping tasks.
Methods
Fifty-two people with PD and 95 HOA performed a simple choice stepping reaction time test (CSRT) and 2 cognitively demanding stepping tests (inhibitory CSRT [iCSRT] and Stroop stepping test [SST]) on a computerized step mat. Cortical activation in the dorsolateral prefrontal cortex (DLPFC), Broca’s area, supplementary motor area (SMA), and premotor cortex (PMC) were recorded using fNIRS. Stepping performance and cortical activity were contrasted between groups and between the CSRT and the iCSRT and SST.
Results
The PD group performed worse than the HOA in all 3 stepping tests. A consistent pattern of interactions indicated differential hemodynamic responses between the groups. Compared with the CSRT, the PD group exhibited reduced DLPFC activity in the iCSRT and reduced SMA and PMC activity in the SST. The HOA exhibited increased DLPFC, SMA, and PMC activity when performing the SST in comparison with the CSRT task.
Conclusions
In contrast to the HOA, the PD group demonstrated reduced cortical activity in the DLPFC, SMA, and PMC during the more complex stepping tasks requiring inhibitory control. This may reflect subcortical and/or multiple pathway damage with subsequent deficient use of cognitive and motor resources.
Introduction
Approximately two-thirds of people with Parkinson’s disease (PD) fall each year,1,2 with a large proportion falling recurrently. 3 Postural stability and particularly the ability to generate quick and accurate steps to negotiate environmental hazards are significantly impaired in PD. 4 For example, in a choice stepping reaction time (CSRT) task requiring participants to step as quickly as possible in response to visual targets, people with PD showed significantly increased stepping reaction time variability compared with healthy age-matched controls. 5 Furthermore, it has been shown that people with PD step significantly slower and make more mistakes (e.g., step onto a wrong target) than healthy age-matched controls in a Stroop stepping test (SST), which requires response inhibition. Although poorer performance in these complex stepping tasks has been correlated with worse global cognitive function, 5 underlying neural mechanisms are unclear. For example, it is not known whether deficits in motor or cognitive brain regions impair performance in challenging stepping tasks in people with PD.
Functional near-infrared spectroscopy (fNIRS) is a portable optical neuroimaging technique that enables investigation of cortical activity while participants move freely.6-8 It has been reported that compared with healthy age-matched people: people with PD show an increase in prefrontal cortex (PFC) activity during quiet standing, 9 a smaller relative increase in PFC activity between usual walking and walking while performing a cognitive task, 10 and a similar relative increase in PFC activity between usual walking and obstacle crossing. 10 However, the examination of cortical activity patterns using fNIRS involving complex stepping tasks has been restricted to healthy young people. 11 In Huppert et al. study, PFC activity was found to be significantly higher when participants performed a lateral stepping reaction time task with an incongruent stimulus (arrow direction and location conveyed opposite meanings) compared with when they performed a lateral stepping reaction time task with a congruent stimulus (arrow direction and location conveyed the same meaning). 11
As CSRT tasks accurately reflect the ability to step quickly and appropriately to avoid obstacles while walking and have been shown to be underpinned by neuropsychological, sensorimotor and balance systems important for balance control, 12 a better understanding of cortical activation patterns in people with PD when performing such complex stepping tasks may assist in informing interventions to preserve safe mobility in this population. Therefore, the aims of this study were to compare cortical activation in cognitive (dorsolateral PFC [DLPFC] and Broca’s area) and motor (supplementary motor area [SMA] and premotor cortex [PMC]) cortical regions using fNIRS between people with PD and healthy age-matched controls during cognitively demanding stepping reaction time tasks. We examined cortical activity in these regions of interest because the DLPFC has a well-documented role in the control of executive functions, including inhibitory control; components of the cognitively demanding stepping tests used here; the SMA contributes to executive planning and motor coordination; and the PMC is involved in controlling executive planning and motor sequencing, planning and movement initiation (PMC): All functions that are usually impaired in people with PD. 13
We hypothesized that compared with healthy controls, people with PD would exhibit greater activity in all cortical areas assessed when undertaking a simple CSRT task but exhibit no or a less marked increase in activity (compared with healthy controls) in all cortical areas when completing more complex stepping tasks (ie an inhibitory CSRT test [iCSRT] and an SST). Furthermore, the above would be accompanied by a nonsignificant between-group difference in response times in the CSRT task but significantly slower stepping responses (response and movement time) in the PD group (compared with healthy controls) in the more complex stepping tests (iCSRT and SST). This pattern of neural and behavioral responses would be consistent with previous studies undertaken in clinical groups with balance disorders 6 and indicate that people with PD are near their cognitive and motor capacities during a relatively simple stepping test and therefore cannot perform as well as healthy controls when the complexity of the stepping task increases. Our secondary hypothesis was that within both PD and control groups, increased variability of stepping response times (particularly in the complex stepping tasks) would be associated with increased cognitive and motor cortical activity.
Methods
Study Registration
We registered our hypotheses on the Open Science Framework on September 11, 2019, prior to statistical analysis of the data.
Participants
Fifty-two people with PD (mean [SD] age = 70.2 [8.4] years, 35 men) and 95 healthy older people (mean [SD] age = 71.3 [4.9] years, 24 men) living in the Sydney metropolitan area comprised the study sample. The recruitment methods for the 2 groups are outlined below.
PD group: this sample comprised participants of a randomized controlled trial of step training (ACTRN12618001515280). They were recruited from the community through the Neuroscience Research Australia volunteer registry, Parkinson’s NSW, the outpatient department at Calvary Hospital, and through advertisements in social media and newsletters. Inclusion criteria were: diagnosis of idiopathic PD according to the UK PD Brain Bank criteria, 14 aged 40 years or older, living independently and able to communicate in English. Exclusion criteria comprised other progressive neurological disorders, unstable medical and psychiatric conditions, and a Montreal Cognitive Assessment (MoCA) 15 score <19 (indicative of marked cognitive impairment) and being color-blind.
Healthy older adult (HOA) group: this sample comprised participants of a randomized controlled trial of cognitive-motor interventions to prevent falls (ACTRN12616001325493). 16 Inclusion criteria comprised individuals aged 65 years or older, living independently in the community and able to communicate in English; able to walk 10 m without the use of a walking aid; willingness to provide informed consent and comply with the study protocol. Exclusion criteria comprised progressive neurological disorders, unstable medical or psychiatric conditions, a Pfeiffer Short Portable Mental Status Questionnaire 17 score <8 (indicative of severe cognitive impairment), being color-blind, residing in residential aged care, currently participating in a fall prevention trial.
The assessments were conducted in two 60-minute sessions over 2 days for the PD participants. They underwent disease-related and sensorimotor tests (not reported in this article) in the first session and performed the stepping tests with the fNIRS monitoring in the second session. For all assessments, the PD participants were in their typical “on” phase of the levodopa treatment cycle, that is, testing commenced between 30 and 60 minutes after levodopa intake, and participants confirmed that this time period corresponded to their optimal “on” phase. The HOA participants completed demographic/health questionnaires and performed the stepping tests with the fNIRS monitoring in a single 60-minute session. The University of New South Wales Human Research Ethics Committee approved this study and all participants gave informed consent prior to study participation.
Assessments
Demographic, Sensorimotor, Falls, and PD-Related Assessments
The age and sex of all participants were recorded, and all participants were asked if they had any falls in the past 12 months. Falls were defined as unexpected events that resulted in unintentionally coming to the ground, floor, or other lower level. 18 The PD participants were assessed using the Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) 19 with parts II (activities of daily living score) and III (motor score) used as descriptive data. Participants’ stage of the disease using the Hoehn and Yahr scale was extracted from the MDS-UPDRS. 20 Duration of disease since date of diagnosis and levodopa equivalency daily dosage intake were also recorded. 21 Global cognition was measured using the MMSE in the HOA group 22 and the Montreal MoCA in the PD group. 15 In order to compare global cognition between groups, MoCA scores were converted to MMSE scores using the calibration of van Steenoven et al. 23
All participants also completed the Physiological Profile Assessment (PPA), which comprises 5 validated measures of physiological function related to balance control 24 : visual contrast sensitivity, lower limb proprioception, knee extension strength, simple reaction time, and postural sway when standing on a compliant surface with eyes open for 30 seconds. A weighed-score algorithm is used to compute an individual composite fall risk score. 24
Stepping Tests
Three stepping tests (CSRT, iCSRT, and SST) were conducted using a customized system comprising a computerized mat (150 × 90 cm) and a computer screen. 25 The mat contained 8 panels: 2 central stance panels, a left panel, a right panel, 2 front panels, and 2 back panels (Figure 1).
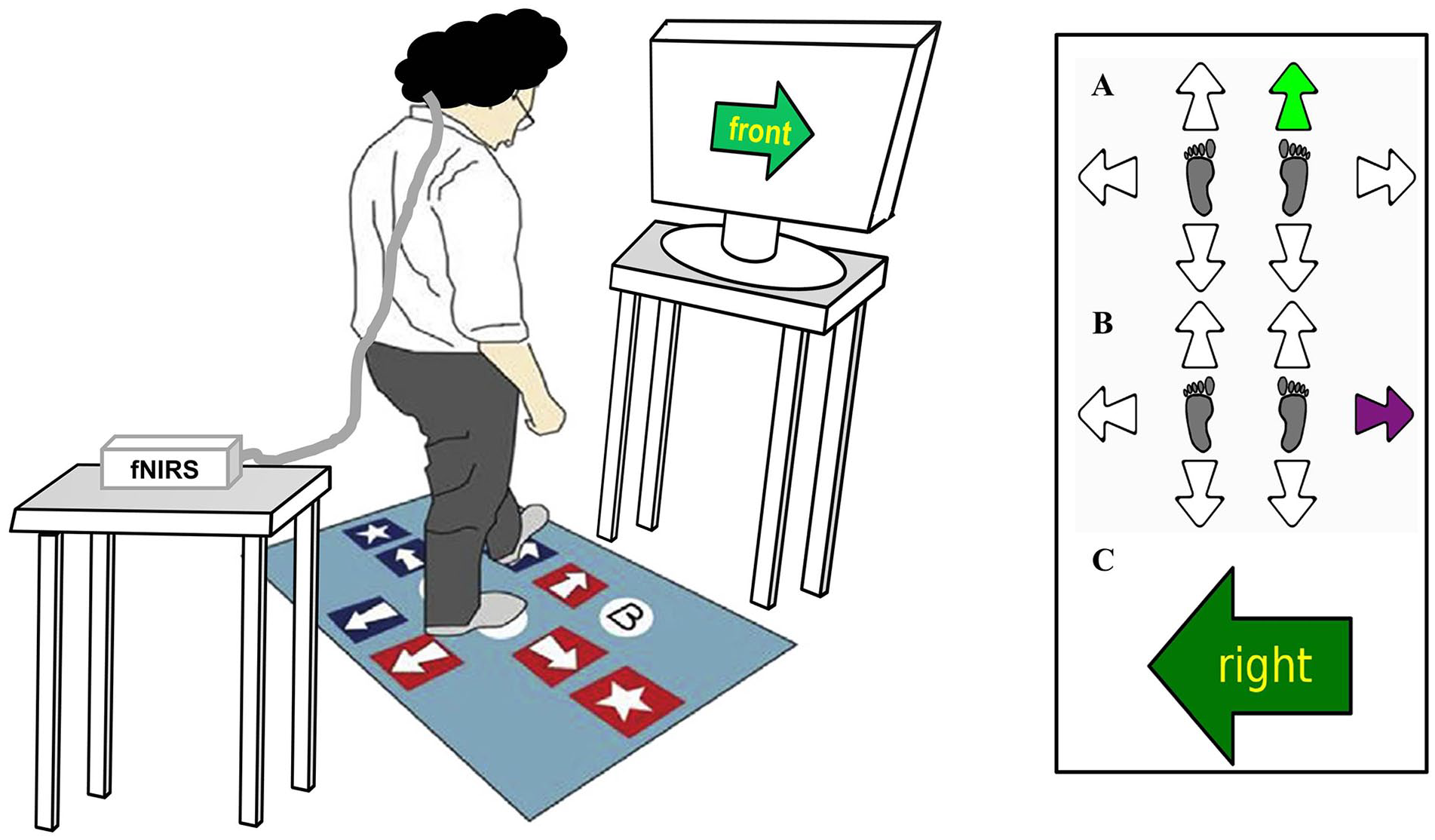
Stepping test setup, including computerized stepping mat and functional near-infrared spectrocopy (fNIRS) monitoring (left panel) and stepping tests display (right panel). Left panel: Illustration of a participant performing the Stroop stepping test. Right panel: Monitor displays of (A) choice stepping reaction time test: participants are required to step as quickly as possible onto the stepping mat panels corresponding to the location of the green arrow appearing on the screen (here, right/forward arrow on the mat); (B) inhibitory choice stepping reaction time test: participants are required to refrain stepping when a purple arrows appears on the screen (inhibitory component) (here, refrain stepping on the right panel) and only step as quickly as possible onto mat panels corresponding to the location of the green arrow; (C) Stroop stepping test: participants are required to step as quickly as possible on the panel corresponding to the direction defined by the word in the arrow and not the direction of the arrow itself (here, step on the right panel).
For the CSRT test, participants were asked to stand on the 2 central panels. They were then instructed to step onto a panel as quickly as possible when the corresponding arrow on the screen changed color from white to green (Figure 1A). Participants first undertook 6 practice trials followed by 24 randomly presented trials (4 trials each for the 6 stepping panels). An error was defined as a step to an incorrect panel.
The iCSRT test was similar to the CSRT (go trials indicated by green); however, no-go trials were also presented as purple arrows. Participants were instructed step as quickly as possible in response to the presented green arrows but not to step if a purple arrow was presented (Figure 1B). Participants completed 6 practice trials followed by 24 randomly presented trials (4 trials for each of the 6 panels, including 18 green and 6 purple arrows). Errors comprised steps onto incorrect panels and steps taken in response to the purple arrow presentations.
In the SST, a large arrow was presented in the center of the screen pointing in 1 of 4 directions (up, down, left, and right) that matched the 4 possible step directions (forward, backward, left, and right). A word indicating a different direction was written inside the arrow. Participants were instructed to “step by the word” and therefore had to inhibit the response indicated by the arrow’s orientation. Four practice trials, followed by 20 randomly presented trials (5 trials for each direction) were administered. Errors comprised any steps taken that were not by the word.
The order of the step tests (CSRT, iCSRT, SST) was randomized for all participants. Stepping performance measures included means and intraindividual variability (standard deviations) of response and movement times computed across 24 trials in the CSRT, 18 trials in the iCSRT (calculated only for the green arrows presented), and 20 trials in the SST. Response time of each stepping performed was defined as the time from stimulus onset to foot lift-off. Movement time of each stepping performed was defined as the time between foot lift-off and touchdown on the correct step panel.
fNIRS Data Acquisition and Analysis
Cortical activity was recorded by a continuous-wave fNIRS system (NIRSport, NIRx). This wearable device contains 8 LED sources that emit 760 and 850 nm frequency-modulated wavelengths and 8 detectors. The sampling rate was set at 7.81 Hz. The 16 optodes, making up 18 channels, were placed on a lightweight cap based on the 10-10 international system. The fNIRS Optodes’ Location Decider (fOLD) toolbox 26 and the Brodmann area atlas 27 were used to define the following regions of interest: DLPFC (Brodmann area 9), Broca’s area (Brodmann area 45), SMA (Brodmann area 8 or frontal eye fields, which is also covered by part of the SMA), and PMC (Brodmann area 6). Due to a limited number of optodes, we were only able to cover part of these cortical areas (Supplementary Figure S1). Optode positions, associated channels, anatomic landmarks and their specificity are outlined in Supplementary Table S1. We considered coverage of ≥50% of a region of interest sufficient. 26 Caps (sizes 54, 56, or 58 cm) were positioned on the participants’ head, the Cz position was considered as the reference, centered between the nasion and the inion (anteroposterior measurement) and between the left and right preauricular points (mediolateral measurement). The optodes were covered by an opaque black cap to reduce interference from external lights.
The data were recorded using NIRStar 15-2 software. Prior to each trial, the equipment was calibrated to determine the optimal amplification factor within an optimal range (0.4-7.0 V). During the calibration, the participants were asked to stand still looking at the screen positioned 1 m ahead. The quality of the signals was then evaluated by the amplification gain and signal level. The differential path length factor was adjusted according to each participant’s age. 28 Each test commenced with a brief practice (6 stepping trials for the CSRT and iCSRT tests [1 in each stepping direction] and 4 stepping trials for the SST [1 in each stepping direction]) so participants could familiarize themselves with the test requirements. This was followed by a baseline period (30 seconds of data collection in which participants were instructed not to move) to bring the haemodynamic status as close to a resting state as possible, and immediately on completion of this, the stepping test. In line with recommendations for reporting fNIRS data 7 block average of the oxygenated hemoglobin (HbO), deoxygenated hemoglobin (HbR), and total hemoglobin (HbO – HbR) concentrations levels during the stepping tests relative to the baseline periods were then computed.
The fNIRS data were analyzed using Homer2 open source software in MATLAB. The following steps were performed: (1) raw data were converted to optical density data; (2) if mean data were <0.01 or >300, if mean data divided by its standard deviation <2, if the source-detector separation was <0 mm or > 45 mm, then the software excluded the channel as an active channel; (3) motion artifacts defined as signal changes greater than a set parameter (standard deviation threshold = 10; amplitude threshold = 0.3) were removed; (4) wavelet transformation of the optical density data was performed to identify motion artifacts8,29,30 (interquartile range = 0.1); (5) data were filtered with a high-pass filter at 0.01 Hz and a low pass filter at 0.14 Hz (to remove physiological responses, ie, heart rate) 8 ; (6) the optical density data were converted to HbO, HbR, and total concentrations; (7) a correlation-based signal improvement of the concentration changes was performed to correct for motion artifacts 31 ; (8) the length of each stepping test data collection was standardized for each individual to the shortest stepping test duration (limited up to 60 seconds); (9) changes in HbO, HbR, and total concentrations during each stepping test relative to the preceding baseline period were computed and block average data were calculated.
Statistics
Continuous data were inspected for right skewed distributions and log transformed if required to allow parametric analyses (stepping time variables were log-transformed). Outliers were replaced with the next highest value (3 cases with very slow times). Student t tests and chi-square tests were used to assess between-group differences for the demographic, clinical, and sensorimotor data. Mixed models analyses of variance (repeated-measures ANOVAs) tests were performed for the stepping performance and hemodynamic data with stepping condition (CSRT vs iCSRT; CSRT vs SST) as within-subject factors and group (PD vs HOA) as a between-subject factor. All ANOVAs were performed with age and sex as covariates. Chi-square tests were performed to examine between-group differences in the proportion of participants who made one or more errors in each stepping test. Pearson correlation tests were used to examine the associations between decision and movement time intraindividual variability for each stepping test (log-10 transformed variables) and the hemodynamic variables for each cortical region of interest. Despite the multiple comparisons made, P values were set at <.05 in this exploratory study. Ninety-five percent confidence intervals (CI) were reported for the significant post hoc tests. The data were analyzed using SPSS version 24 for Windows (IBM Corp).
Results
Demographic, Clinical, Sensorimotor, and PD-Related Characteristics of Participants
Table 1 presents the demographic, clinical, sensorimotor, and disease-related characteristics of the participants. The PD group comprised people in mild to moderate Hoehn and Yahr stages (1-3) with a mean disease duration (SD) of 6.9 (4.8) years. The PD group comprised more men and more past fallers than the HOA group. PD participants also had higher PPA scores indicating increased physiological fall risk, worse contrast sensitivity, slower hand reaction time, and greater postural sway. The mean (SD) MMSE scores for the HOA (29.2 (0.9)) and PD groups (28.8 (1.3)) indicate that both group were cognitively intact.
Demographic, Cognitive, Sensorimotor, and Disease-Related Measures for the Healthy Older Adults and the Parkinson’s Disease Groups. a
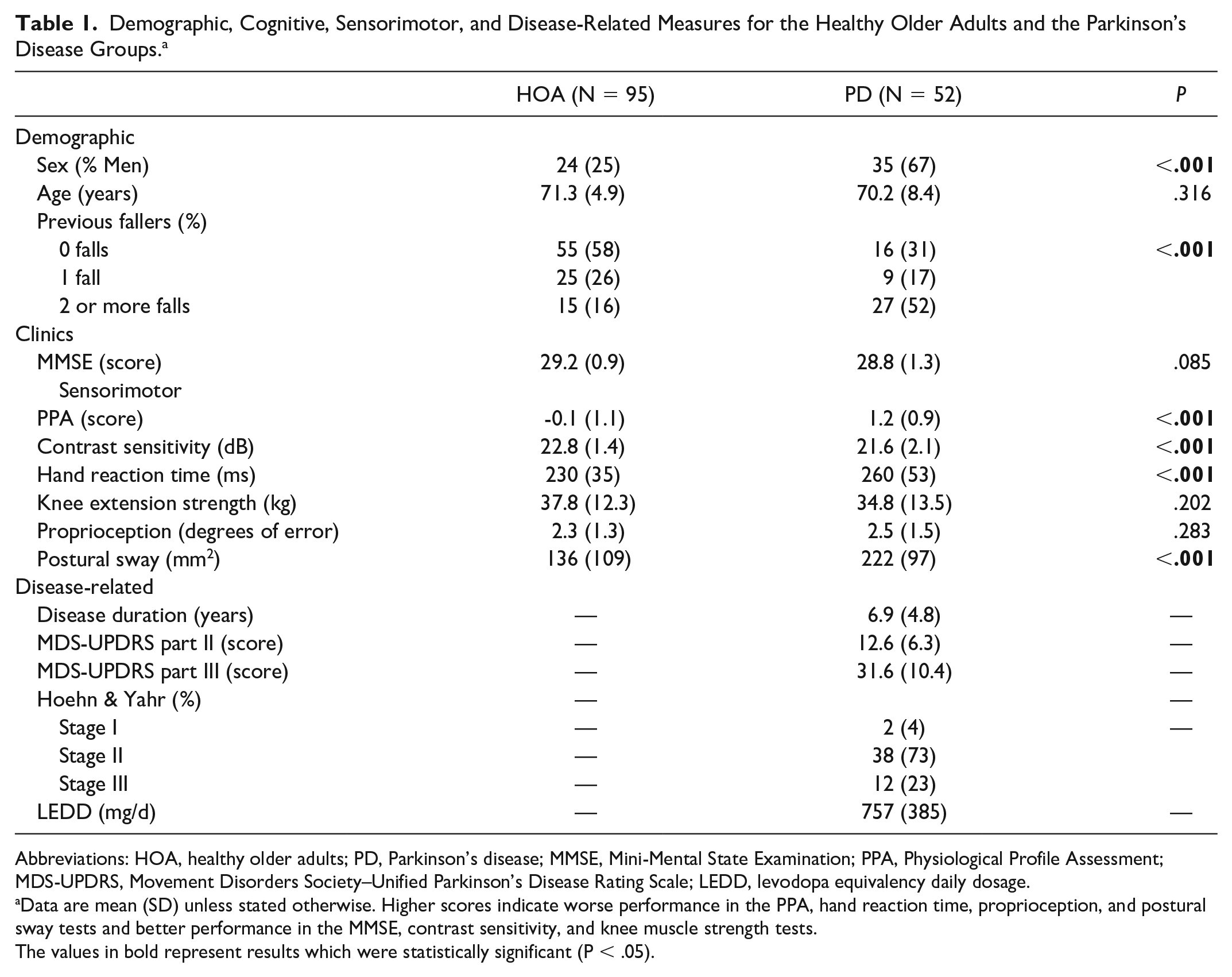
Abbreviations: HOA, healthy older adults; PD, Parkinson’s disease; MMSE, Mini-Mental State Examination; PPA, Physiological Profile Assessment; MDS-UPDRS, Movement Disorders Society–Unified Parkinson’s Disease Rating Scale; LEDD, levodopa equivalency daily dosage.
Data are mean (SD) unless stated otherwise. Higher scores indicate worse performance in the PPA, hand reaction time, proprioception, and postural sway tests and better performance in the MMSE, contrast sensitivity, and knee muscle strength tests.
The values in bold represent results which were statistically significant (P < .05).
Stepping Responses
Table 2 presents the mean and intraindividual variability for the step responses for each stepping test. For the comparison between the CSRT and iCSRT conditions, no group by condition interactions were observed. However, group main effects were evident for mean and intraindividual variability for response and movement time, indicating the PD group showed slower and more variable stepping performance than HOA. Condition main effects were also observed for mean response time, indicating the participants responded slower in the iCSRT test compared with the CSRT test.
Descriptive Statistics and ANOVA Results of Stepping Response Results by Group in Relation to CSRT, iCSRT, and SST Performance. a
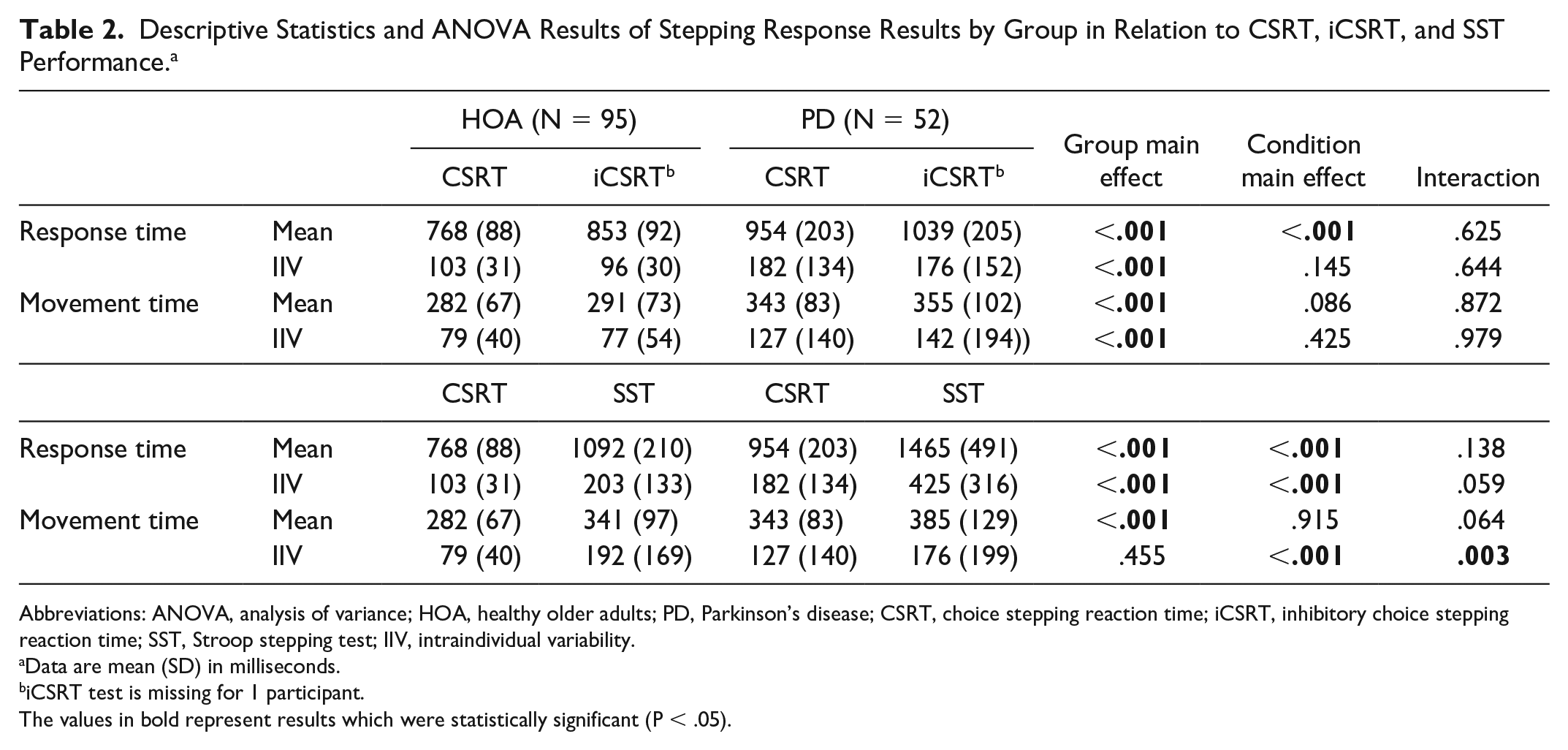
Abbreviations: ANOVA, analysis of variance; HOA, healthy older adults; PD, Parkinson’s disease; CSRT, choice stepping reaction time; iCSRT, inhibitory choice stepping reaction time; SST, Stroop stepping test; IIV, intraindividual variability.
Data are mean (SD) in milliseconds.
iCSRT test is missing for 1 participant.
The values in bold represent results which were statistically significant (P < .05).
For the comparison between the CSRT and the SST conditions, a group by condition interaction was observed for intraindividual variability of movement time. Post hoc tests showed that the PD group was more variable than HOA in the CSRT (intraindividual variability mean difference [95% CI] = 0.149 [0.058-0.241], P = .002). In addition, the HOA were more variable in the SST condition compared with the CSRT condition (mean difference [95% CI] = 0.297 [0.208-0.386], P < .001). A group main effect was observed for mean response and movement time (the PD group stepped more slowly than HOA) as well as intraindividual variability of response time (the PD group were more variable in their stepping response time overall). Condition main effects were observed for both mean and intraindividual variability of response time (participants responded more slowly and were more variable in the SST condition compared with the CSRT condition).
Stepping Errors
Table 3 presents the number (%) of participants among the PD and HOA groups who made 1 error, 2 or more errors, or none in each stepping test as well as the error rates (number of errors per participant) in each group/stepping test. More PD participants than HOA participants made at least 1 error in the CSRT test (χ2 = 7.23, P = .007), the iCSRT test (χ2 = 17.45, P < .001), and the SST (χ2 = 14.30, P < .001).
Proportion of Participants Who Made an Error and Error Rates in Relation to the CSRT Test, the iCSRT Test, and the SST Performance. a
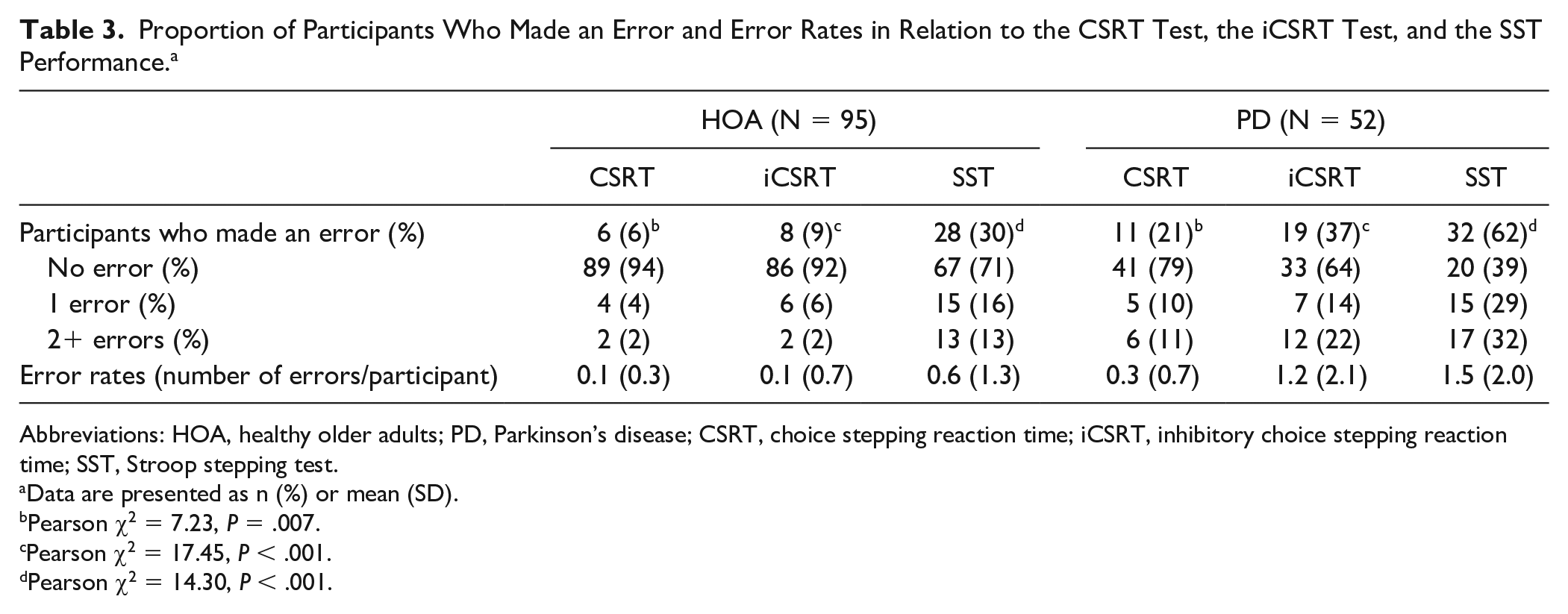
Abbreviations: HOA, healthy older adults; PD, Parkinson’s disease; CSRT, choice stepping reaction time; iCSRT, inhibitory choice stepping reaction time; SST, Stroop stepping test.
Data are presented as n (%) or mean (SD).
Pearson χ2 = 7.23, P = .007.
Pearson χ2 = 17.45, P < .001.
Pearson χ2 = 14.30, P < .001.
Cortical Activity
For simplicity, and because there is evidence indicating HbO values are more reliable and sensitive to locomotion-related cerebral blood flow than HbR values, 32 only the HbO concentration data are presented in the main body of this article, with the data for HbR and total hemoglobin concentrations presented as supplementary material (Supplementary Tables S2 and S3).
Table 4 and Supplementary Figure S2 show the HbO concentrations recorded in the 3 stepping conditions. For the comparison between CSRT and iCSRT, group by condition interactions were observed in the DLPFC and Broca’s area. Post hoc tests revealed in these regions of interest, HbO concentrations were significantly reduced (indicative of decreased cortical activity) in the iCSRT condition compared with the CSRT condition in the PD group (DLPFC: mean difference [95% CI]: −2.44 [−4.32 to −0.55], P = .012; Broca’s area: mean difference [95% CI]: −2.58 [−4.84 to −0.32], P = .026), while no significant changes were observed for the HOA group.
Relative Oxyhemoglobin (HbO) Concentration (nmol/L) in the Cortical Regions of Interest by Group in the CSRT Test, the iCSRT Test, and the SST. a
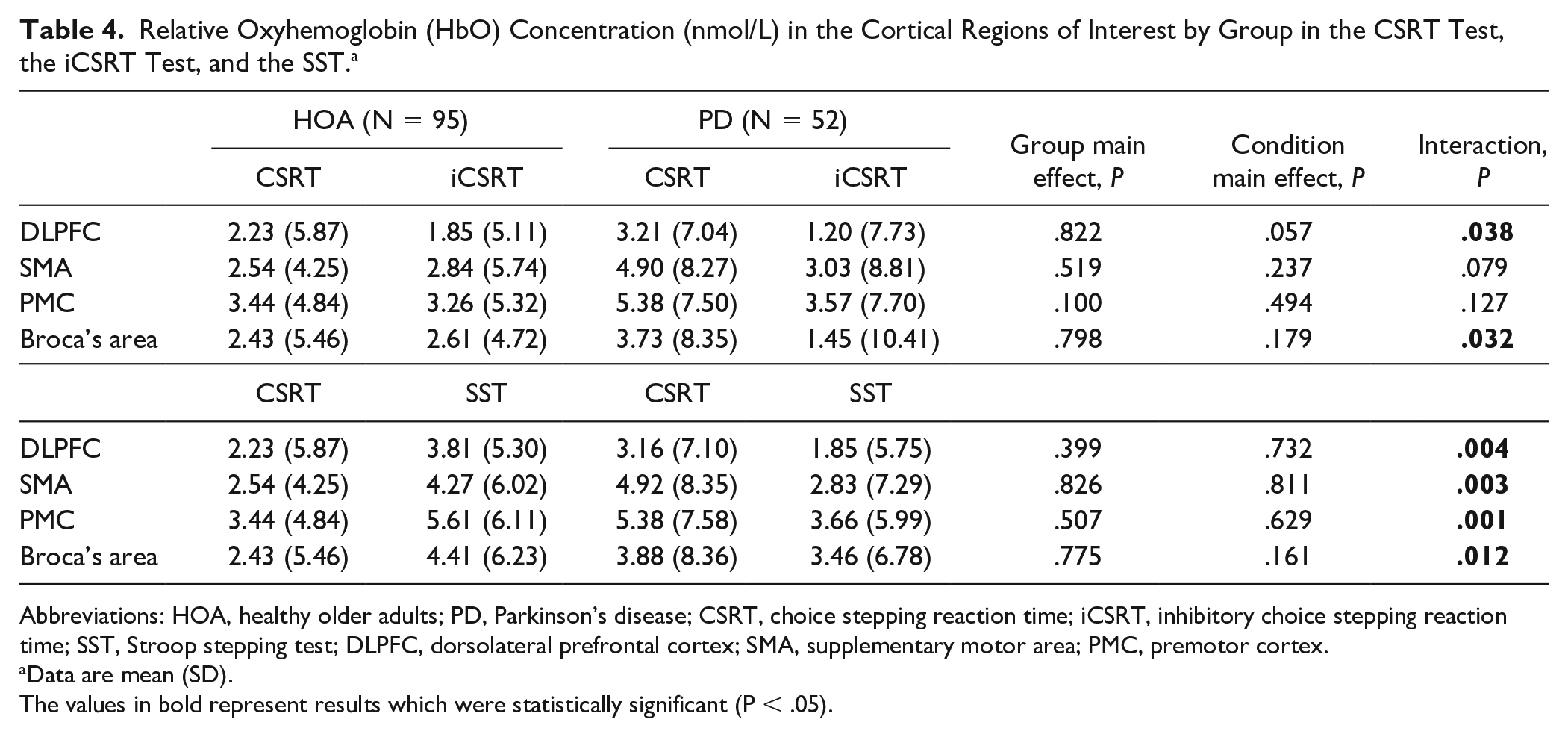
Abbreviations: HOA, healthy older adults; PD, Parkinson’s disease; CSRT, choice stepping reaction time; iCSRT, inhibitory choice stepping reaction time; SST, Stroop stepping test; DLPFC, dorsolateral prefrontal cortex; SMA, supplementary motor area; PMC, premotor cortex.
Data are mean (SD).
The values in bold represent results which were statistically significant (P < .05).
For the comparison between SST and CSRT, group by condition interactions were observed for all regions of interest (Table 4 and Supplementary Figure S3). Post hoc tests showed lower HbO concentrations, statistically significant for the SMA (mean difference [95% CI]: −2.25 [−4.35 to −0.16], P = .035) and the PMC (mean difference [95% CI]: −1.96 [−3.85 to −0.07], P = .042) and a statistical trend for the DLPFC (P = .087), in the SST condition compared with the CSRT condition in the PD group. In contrast, significantly higher HbO concentrations (indicative of increased cortical activity) for the DLPFC (mean difference [95% CI]: 2.05 (0.57-3.53), P = .007), SMA (mean difference [95% CI]: 1.94 [0.30-3.59], P = .021), PMC (mean difference [95% CI]: 2.52 [1.04-4.00], P = .001), and Broca’s area (mean difference [95% CI]: 2.65 [1.04-4.25], P = .001] were observed in the SST condition compared with the CSRT condition in the HOA group. Furthermore, the PD group had significantly lower HbO concentrations in the DLPFC (mean difference [95% CI]: −2.63 [−4.68 to −0.59], P = .012) and PMC (mean difference [95% CI]: −2.85 [−5.07 to −0.64], P = .012) than the HOA group in the SST condition.
The total hemoglobin and HbR concentration findings were generally in line with the HbO findings whereas overall no significant interactions or main effects of group and condition were observed for HbR concentrations (Supplementary Tables S2 and S3).
Associations Between Intraindividual Variability Stepping Response and Cortical Activity
Some weak associations (r < 0.3) were observed between response time intraindividual variability in the SST and SMA and PMC activity levels only for HOA group. No associations between intraindividual variability of response or movement times and hemodynamic variables in either of the stepping tests for any of the areas of interest were observed for PD and HOA groups (Supplementary Table S4).
Discussion
Our hypothesis that people with PD would exhibit greater cognitive and motor cortical activity when undertaking the CSRT task but exhibit no or a less marked increase in activity (compared with HOA) in these cortical areas when undertaking the iCSRT and SST tasks was only obliquely confirmed. We found that the PD group exhibited similar cognitive and motor cortical activity when undertaking the CSRT task and that the HOA showed increased cognitive and motor cortical activity in the most complex (SST) stepping task. However, rather than exhibiting increased cortical activity with more complex stepping tasks, the PD group exhibited significantly reduced cortical activity when undertaking the iCSRT and SST tasks, relative to the CSRT task. Regarding stepping performance, we observed the PD group made more errors and had significantly slower and more variable response times in all stepping tasks in comparison with HOA. This pattern of neural and behavioral findings could suggest not only that the PD group participants were at near cognitive and motor capacity during the simple stepping condition but also that they were unable to fully utilize their cognitive and motor cortical resources when required to undertake complex stepping tasks.
Both the iCSRT and the SST tap into the inhibitory control of executive function, which is impaired in PD. The two more complex stepping tasks involve complementary aspects of executive function: slow SST times indicate reduced ability to inhibit the step response indicated by the arrow’s orientation whereas increased iCSRT reaction times to the green (go) arrow trials reflect proactive slowing in the context of the probability that a purple (no-go) arrow trial, and stepping errors reflect ability to stop the stepping response. The findings that participants with PD stepped slower and made more mistakes than their healthy age-matched peers across both cognitively-demanding stepping tests suggests reduced inhibitory control of executive function despite prospective slowing and no indication of a differential speed-accuracy trade-off. These behavioral findings in PD are also consistent with the premise that tasks requiring volitional input 8 rely more strongly on the indirect locomotor pathway (basal ganglia, DLPFC, SMA, and PMC), as opposed to more automatic tasks, such as walking. Our results extend the well-documented associations between reduced activity in the DLPFC and PMC with deficits in executive functions (attention, response inhibition, sequential planning), working memory, and anticipatory postural adjustments.33-35 Based on the brain activity responses of the HOA, it appears the SST was more demanding than the iCSRT. For this group, the iCSRT (primarily requiring response inhibition) did not appear to be sufficiently challenging to tap into their “cognitive reserve.” 36 In contrast, the HOA group exhibited significantly greater DLPFC, SMA, and PMC activity in the SST which may reflect this task requiring set-shifting and attention in addition to response inhibition. It might be that the PD group had no cognitive reserve beyond that required for the simpler CSRT task; hence, it appears the more difficult tasks overwhelmed the PD group, as indicated by their reduced activity in both cognitive and motor cortical regions and significantly worse stepping performance.
The finding that the PD group exhibited both reduced cortical activity in the cognitive and motor areas when performing the complex stepping tasks suggests a discrete cortical deficiency is not responsible for the impaired stepping performances observed. Indeed, reduced activity was also evident for Broca’s area (CSRT vs iCSRT), the cortical region with functions related primarily to speech production—a function not relevant to the stepping tasks performed. Instead, it suggests impaired complex stepping requiring inhibition results from subcortical and/or multiple pathway mechanisms. This may involve degeneration of basal ganglia and subthalamic nucleus networks,37,38 as defective subcortical networks are known to downregulate cortical activity during tasks requiring inhibitory control. 39 Impaired cholinergic pathways have also been shown to produce cognitive and motor deficits in PD. 40 For example, the Meynert nucleus has neural cholinergic connections with the DLPFC (affecting cognitive processing) and the pedunculopontine nucleus has cholinergic and noncholinergic connections with the basal ganglia and thalamus (affecting motor control and attention), and both are impaired in PD.40,41 Deficits in these structures impair fronto-striatal circuit function, and in consequence cognitive processing.40,42 Thus, the modulation of attention that occurs when the PFC activates the Meynert nucleus in cognitively demanding tasks where Meynert nucleus should directly generate acetylcholine release 43 may not occur in people with PD. In consequence, people with PD may exhibit decreased cortical activity when the opposite is required to perform a demanding cognitive-motor task.
Only a few previous studies have reported associations between cortical activity in the PFC and sensorimotor cortical areas with stepping or gait variability. In young samples, Koren and colleagues 44 reported increased DLPFC activity was significantly associated with increased stride-time variability in perturbed walking conditions and Groff and colleagues 45 observed that increased precentral gyrus activation corresponded to increased stride-time variability during simple walking. Vitorio and colleagues 46 also showed increased PMC and primary motor cortex activity were associated with increased step length variability in young adults, but that these associations were reversed in HOA. Contrary to our hypothesis, we found only a few weak and inconsistent correlations between cortical activity in our regions of interest and variability of stepping times in both the PD and HOA groups. It is possible external factors relating to cognitive and sensorimotor function in both groups,4,5 as well as disease-specific factors (eg, bradykinesia, hypokinesia) in the PD group may have overshadowed any relationships between cortical activity and stepping reaction time variability.
Strengths of our study include registration of our hypotheses on the Open Science Framework 47 prior to any statistical analyses, and the conduct of the study in line with published recommendations,6-8 particularly regarding the identification of appropriate regions of interest based on Brodmann atlas classification 26 and the selection of relevant and appropriate stepping tasks that do not require verbal responses. 48 We also acknowledge some limitations. First, between-subject variability in the hemodynamic measurements was high. However, such variability seems to be inherent for these neural data, mainly due to cognitive interference. 49 Moreover, the sole reliance on anatomical landmarks as opposed to magnetic resonance imaging scans for optode placement might have resulted in inaccuracies in the estimation of boundaries of cortical regions of interest. In addition, the use of data-based filters at the processing stage as opposed to short-separation channels (which measure the extracerebral activity alone, so that it may be removed from the total fNIRS signal) means that we cannot guarantee the complete removal of physiological and motion artefacts. Second, our fNIRS system allowed for only limited coverage (18 channels) of our selected regions of interest and precluded us from comparing cortical activity between hemispheres (less impaired hemisphere vs more impaired hemisphere). While this would not have affected our between-group comparisons, future studies using more fNIRS channels could investigate cortical lateralization during stepping in people with PD. Third, we acknowledge that the CSRT does not represent a true control or baseline condition for our SST, as is the case for the Stroop color reading and Stoop color word tests. However, the two tests have distinctly different levels of cognitive difficulty while maintaining the same step responses, and therefore provide an appropriate paradigm to investigate our study hypothesis.
Implications
This study has important clinical implications. First, the reduced cortical activity in cognitive and motor areas observed in people with PD while they performed complex stepping tasks may inform strategies for improving mobility and balance in this population. On the one hand, this could include direct practice in challenging stepping tasks comprising a cognitive component, and on the other hand, educational programs regarding the need to primarily attend to safe mobility in complex everyday environments, that is, to slow down and concentrate. 50 Second, the finding that people with PD had increased variability of response times and more stepping errors than their healthy peers in all stepping tests provides insight into why people with PD are at increased risk of falls5,51 as well as the situations in which they may fall.
Conclusions
People with PD performed worse than HOA in stepping reaction tasks, regardless of their complexity. Reductions in cortical activity in DLPFC, SMA, and PMC within the PD group during the more complex stepping tasks requiring inhibitory control compared with the simple task may reflect subcortical and/or multiple pathway damage with subsequent deficient use of cognitive and motor resources.
Supplemental Material
Figure_S1 – Supplemental material for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control
Supplemental material, Figure_S1 for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control by Paulo H. S. Pelicioni, Stephen R. Lord, Yoshiro Okubo, Daina L. Sturnieks and Jasmine C. Menant in Neurorehabilitation and Neural Repair
Supplemental Material
Figure_S2_R2 – Supplemental material for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control
Supplemental material, Figure_S2_R2 for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control by Paulo H. S. Pelicioni, Stephen R. Lord, Yoshiro Okubo, Daina L. Sturnieks and Jasmine C. Menant in Neurorehabilitation and Neural Repair
Supplemental Material
Figure_S3_R2 – Supplemental material for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control
Supplemental material, Figure_S3_R2 for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control by Paulo H. S. Pelicioni, Stephen R. Lord, Yoshiro Okubo, Daina L. Sturnieks and Jasmine C. Menant in Neurorehabilitation and Neural Repair
Supplemental Material
Table_S1_R1 – Supplemental material for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control
Supplemental material, Table_S1_R1 for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control by Paulo H. S. Pelicioni, Stephen R. Lord, Yoshiro Okubo, Daina L. Sturnieks and Jasmine C. Menant in Neurorehabilitation and Neural Repair
Supplemental Material
Table_S2_R1 – Supplemental material for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control
Supplemental material, Table_S2_R1 for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control by Paulo H. S. Pelicioni, Stephen R. Lord, Yoshiro Okubo, Daina L. Sturnieks and Jasmine C. Menant in Neurorehabilitation and Neural Repair
Supplemental Material
Table_S3_R2 – Supplemental material for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control
Supplemental material, Table_S3_R2 for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control by Paulo H. S. Pelicioni, Stephen R. Lord, Yoshiro Okubo, Daina L. Sturnieks and Jasmine C. Menant in Neurorehabilitation and Neural Repair
Supplemental Material
Table_S4_R1 – Supplemental material for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control
Supplemental material, Table_S4_R1 for People With Parkinson’s Disease Exhibit Reduced Cognitive and Motor Cortical Activity When Undertaking Complex Stepping Tasks Requiring Inhibitory Control by Paulo H. S. Pelicioni, Stephen R. Lord, Yoshiro Okubo, Daina L. Sturnieks and Jasmine C. Menant in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We acknowledge Bethany Halmy and Rui Liu for helping with data collection, Dr Rodrigo Vitorio for his advice on data analysis and the participants who volunteered themselves to take part in this research.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Parkinson’s NSW seed grant. Paulo H. S. Pelicioni was a recipient of a Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) PhD scholarship (Grant number: BEX 2194/15-5). Stephen R. Lord is supported by NHMRC Research Fellowship.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
