Abstract
Background
Robot-assisted therapy and noninvasive brain stimulation (NIBS) are promising strategies for stroke rehabilitation.
Objective
This systematic review and meta-analysis aims to evaluate the evidence of NIBS as an add-on intervention to robotic therapy in order to improve outcomes of upper-limb motor impairment or activity in individuals with stroke.
Methods
This study was performed according to the PRISMA Protocol and was previously registered on the PROSPERO Platform (CRD42017054563). Seven databases and gray literature were systematically searched by 2 reviewers, and 1176 registers were accessed. Eight randomized clinical trials with upper-limb body structure/function or activity limitation outcome measures were included. Subgroup analyses were performed according to phase poststroke, device characteristics (ie, arm support, joints involved, unimanual or bimanual training), NIBS paradigm, timing of stimulation, and number of sessions. The Grade-Pro Software was used to assess quality of the evidence.
Results
A nonsignificant homogeneous summary effect size was found both for body structure function domain (mean difference [MD] = 0.15; 95% CI = −3.10 to 3.40; P = 0.93; I2 = 0%) and activity limitation domain (standard MD = 0.03; 95% CI = −0.28 to 0.33; P = 0.87; I2 = 0%).
Conclusions
According to this systematic review and meta-analysis, at the moment, there are not enough data about the benefits of NIBS as an add-on intervention to robot-assisted therapy on upper-limb motor function or activity in individuals with stroke.
Keywords
Introduction
The mean global lifetime risk of stroke increased from 22.8% in 1990 to 24.9% in 2016. 1 Only 38% of individuals who present flaccid arm plegia after stroke recover dexterity to some extent, and only 11% achieve full functional recovery. 2 Upper-limb function is directly related to the satisfactory performance of self-care activities, leisure, and labor. 3 Upper-limb function also affects mental health 4 and perception of quality of life. 5
The identification of more effective interventions to enhance arm and hand function is a priority of neurorehabilitation research, reflected by an increasing number of randomized clinical trials. 6 In this context, robotic therapy (RT) has evolved rapidly. 7 Intensity of practice and task-specific training are key elements of successful RT.8,9 Although a Cochrane review (45 trials, 1619 participants) reported that RT improved arm function, activities of daily living, and muscle strength scores, 10 a recent multicenter trial (770 participants) suggested that further research is needed to find ways, or combined therapy strategies, to translate the improvements in upper-limb impairment after RT training into improvements in upper-limb function and activities of daily living. 11
In this context, noninvasive brain stimulation (NIBS) has emerged as a potential add-on tool to rehabilitation interventions by boosting adaptive plasticity mechanisms. 12 NIBS is based on the principle of application of external stimuli to modify brain activity. NIBS techniques include transcranial direct current stimulation (tDCS) and transcranial magnetic stimulation (TMS), among other interventions.13,14
This systematic review and meta-analysis aims, for the first time, to evaluate the evidence of the association of RT and NIBS for upper-limb rehabilitation in stroke. We analyzed randomized clinical trials that compared the association of RT and active NIBS or sham for the improvement of upper-limb motor recovery on body function and structure as well as on activity according to the International Classification of Functioning, Disability and Health (ICF). 15
Methods
This study was performed according to the PRISMA Protocol 16 and was approved by the Sao Paulo University Medical School Research Ethics Committee in March 2017 (No. 0085/17). The protocol was registered on the PROSPERO Platform (CRD42017054563).
Search Strategy
Seven scientific databases were systematically searched: MEDLINE (Medical Literature Analysis and Retrieval System Online; through the PubMed interface), EMBASE (Excerpt Medical Database), Cochrane Central Register of Controlled Trials (CENTRAL), LILACS (Latin American & Caribbean Health Sciences Literature; through the Virtual Health Library—Bireme interface), CINAHL (Cumulative Index to Nursing and Allied Health Literature through the EBSCO interface), DORIS (Database of Research in Stroke), and PEDro (Physiotherapy Evidence Database). The articles were manually retrieved. In addition, the following online archives of theses or trial registers were searched: ClinicalTrials, Digital Library of Theses and Dissertations of São Paulo University, DORIS, Digital Brazilian Library of Theses and Dissertations, Public Domain Portal, and CAPES (Coordination for the Improvement of Higher Education Personnel) Thesis and Dissertation Bank.
The following keywords were used: stroke, robot, transcranial direct current stimulation, and transcranial magnetic current stimulation. The full search strategy can be found in Supplementary Material (SM), Table SM1. No publication date or language restrictions were imposed. The search included all studies published until July 2019.
Study Selection
Titles and/or abstracts of studies retrieved using the search strategy and those from additional sources were independently assessed by 2 reviewers. The 2 reviewers read the full-text articles and selected studies according to the inclusion and exclusion criteria. Disagreements between the 2 reviewers were resolved by consensus.
Eligibility Criteria
Types of studies: Randomized clinical trials with parallel or crossover design were included. Authors were contacted to provide missing data of abstracts and nonpublished studies.
Types of participants: People with upper-limb paresis caused by stroke were included. No restrictions were imposed regarding age, residual upper-limb motor-function, time since last stroke, type of stroke, or history of previous strokes. Studies that included individuals with cerebellar strokes or strokes in cerebellar pathways were not included.
Types of interventions: Trials that added active NIBS before, during, or after RT to improve upper-limb outcomes were included. Four types of NIBS were assessed: tDCS, transcranial alternating current stimulation, transcranial random noise stimulation, and TMS.
Types of controls: RT associated with sham NIBS or RT alone.
Types of outcome measures: Upper-limb performance measured by the main standard scales for the ICF body structure/body function domain (eg, Fugl-Meyer Assessment Scale [FMA], Ashworth or modified Ashworth Scale, force and range of motion) or activity level (eg, Wolf Motor Function Test [WMFT], Action Research Arm Test [ARAT], Motor Activity Log [MAL], Box and Blocks Test [BBT], Jebsen-Taylor Hand Function Test, Nine Hole Peg Test, and Motor Assessment Scale). 17
Data Extraction
Data were extracted from the records to a standard form by 2 reviewers. Disagreements were resolved by consensus. If information was missing/unclear in articles, their authors were contacted. In case of crossover studies, only data of the first intervention were extracted for metanalyses.
If more than 1 outcome was assessed to measure body function/structure, the priority for analysis was the Fugl-Meyer according to recommendations of the measurement working group of the Stroke Recovery and Rehabilitation Roundtable. 18 To measure activity limitation, priority of analysis was given to the ARAT according to recommendations of the same panel. 18 If these scales were not present in the study, the choice of outcome was based on the most frequent outcomes across the selected studies.
Methodological Quality
Risks of bias were assessed independently by 2 reviewers. Disagreements were settled by consensus, and no consultation with a third reviewer was necessary. If there was a lack of, or unclear, information, authors were asked for clarification, or requests were made for the provision of the missing information.
The following domains were assessed for each study: randomization, 19 concealment allocation, 19 blinding of outcome assessment, 19 blinding of participants and personnel, 19 description or implicit intention-to-treat analysis, extent of loss, 20 sample homogeneity (similarity between characteristics data of active and control groups), sample representativeness (absence or presence of exclusion criteria other than those usually present on RT or NIBS trials in general), presence of description of sample calculation, information regarding early cessation of trials, and selective reporting. 19 For each domain, the study was classified as having high, low, or unclear risk of bias.
The classification of bias was not used as a criterion to exclude studies from a possible meta-analysis but was used for analysis of quality of the evidence according to GRADEpro GDT (Grading of Recommendations Assessment, Development and Evaluation), 16 by a GRADE Evidence Profile across the domain study design, risk of bias, inconsistency, indirectness, and imprecision. The domain risk of bias was classified prioritizing randomization, allocation, blinding, and extent of loss. Other domains contributed with the percentage of high or unclear risk of bias (ie, <50% not serious; 50%-75%, serious; >75%, very serious). The domain inconsistency was classified according to I2 of metanalysis (<50% not serious; 50%-75%, serious; >75%, very serious). The domain imprecision was classified according to the Z test for overall effect (<50% not serious; 50%- 75%, serious; >75%, very serious).
The quality was stated as high (further research is very unlikely to change our confidence in the estimate of the effect), moderate (further research is likely to have an important impact on our confidence in the estimate of the effect and may change the estimate), low (further research is very likely to have an important impact on the confidence in the estimate of effect and is likely to change the estimate), or very low (very uncertain about the estimate of the effect). 16
Quantitative Data Analysis
For studies in which the same scale was used to evaluate the outcome (ie, Fugl-Meyer for body structure/function), the number of participants in each group, mean scores, and SDs after interventions in the active and control groups were analyzed in RevMan 5.3. (Review Manager 5.3). For the Fugl-Meyer, a higher score was regarded as positive. The mean difference (MD) was established in individual studies by calculating the difference between the means of the active and control groups.
For types of outcomes assessed with different scales (ie, Activity Limitation), measures of postintervention and preintervention of each subject were assessed after contacting the authors and requesting individual data. The individual relative difference between postintervention and preintervention (Postintervention − Preintervention/Preintervention) was calculated for each participant. The means and SDs of relative differences in active and control groups were analyzed in RevMan 5.3. In individuals who presented a baseline score of 0, the relative difference was also considered to be 0. We opted for this approach because none of the individuals who scored 0 at baseline presented a postintervention to preintervention difference above the minimal detectable changes of the respective scale.
For scales in which a lower score was regarded as positive compared with a higher score, the mean was multiplied by −1. The standard mean difference (SMD) was established in individual studies by calculating the difference between the means of the active and control groups.
The summary effect size (SES) was calculated by the average of the MD or SMD of individual studies, with the corresponding 95% CI. Heterogeneity among studies was assessed by I2. In case of statistical heterogeneity, defined as an I2 ≥50%, a random-effect model was applied, whereas a fixed-effect model was applied for I2 <50%. 21 All analyses were performed using RevMan 5.3, and P values ≤0.05 were considered statistically significant.
In addition, the following subgroup analyses were conducted7,12,22,23:
According to participants’ characteristics: Phase poststroke (acute, 1-7 days; early subacute, >7 days to 3 months; late subacute, 3-6 months; chronic, >6 months) 24
According to robotic device characteristics: Arm support (end-effector or exoskeleton), 25 joints involved (shoulder, elbow, wrist, hand), and unimanual or bimanual training
According to NIBS characteristics: NIBS paradigm (aiming to increase or decrease cortical excitability), timing of stimulation (preintervention, postintervention, or during RT)
According to general characteristics: Number of sessions (1-6, 7-12, 13-24, 25-36) 26
Results
Search Results
At the last search in July 2019, a total of 1266 records were identified from databases, gray literature, and hand search. After removing duplicates, 1176 articles were identified, and 14 abstracts were selected for full-text reading. The study selection process is shown in Supplementary Material, Figure SM1. At the end of the selection, 6 trials were excluded for the following reasons: 3 did not attempt criteria for control groups,22,27,28 and 4 did not use standard clinical scales as outcome measures.23,28-30
Characteristics of the Trials
The characteristics of participants of trials selected for the systematic review and meta-analysis are shown in Table 1, and the characteristics of interventions are shown in Table 2. One study 31 applied 3 different interventions (sham tDCS, anodal tDCS [atDCS], cathodal tDCS [ctDCS]), and in this review, was divided into 2 (Hesse et al, 31 A: anodal stimulation; and Hesse et al, 31 C: cathodal stimulation). Thus, considering Hesse et al, 2011 as 2 different trials for statistical analysis, the review included 9 trials, with a total of 324 participants (161, active; 163, control).
Characteristics of the Participants of the Studies Included in the Meta-analysis.
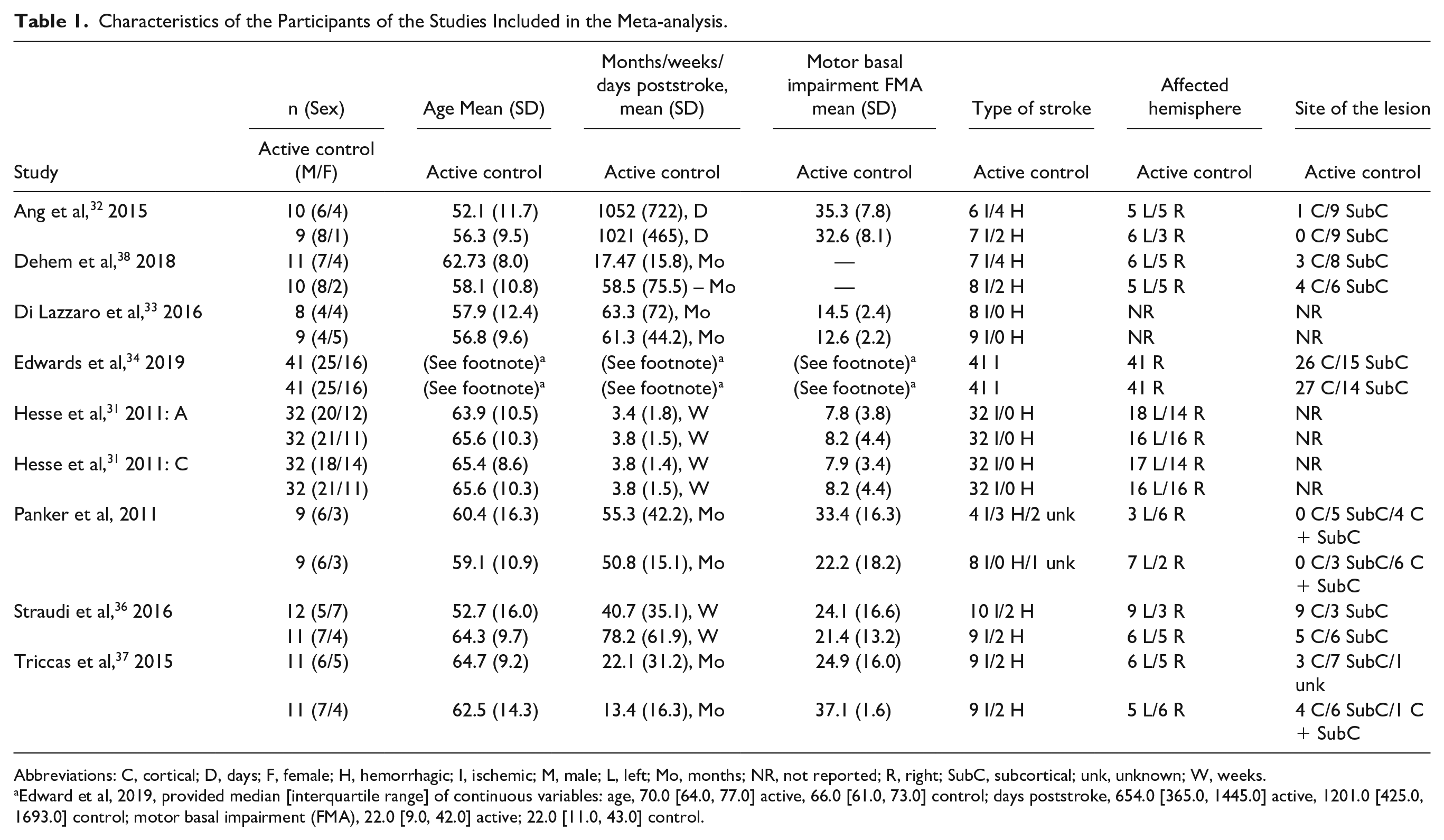
Abbreviations: C, cortical; D, days; F, female; H, hemorrhagic; I, ischemic; M, male; L, left; Mo, months; NR, not reported; R, right; SubC, subcortical; unk, unknown; W, weeks.
Edward et al, 2019, provided median [interquartile range] of continuous variables: age, 70.0 [64.0, 77.0] active, 66.0 [61.0, 73.0] control; days poststroke, 654.0 [365.0, 1445.0] active, 1201.0 [425.0, 1693.0] control; motor basal impairment (FMA), 22.0 [9.0, 42.0] active; 22.0 [11.0, 43.0] control.
Interventions Reported in the Meta-analysis.
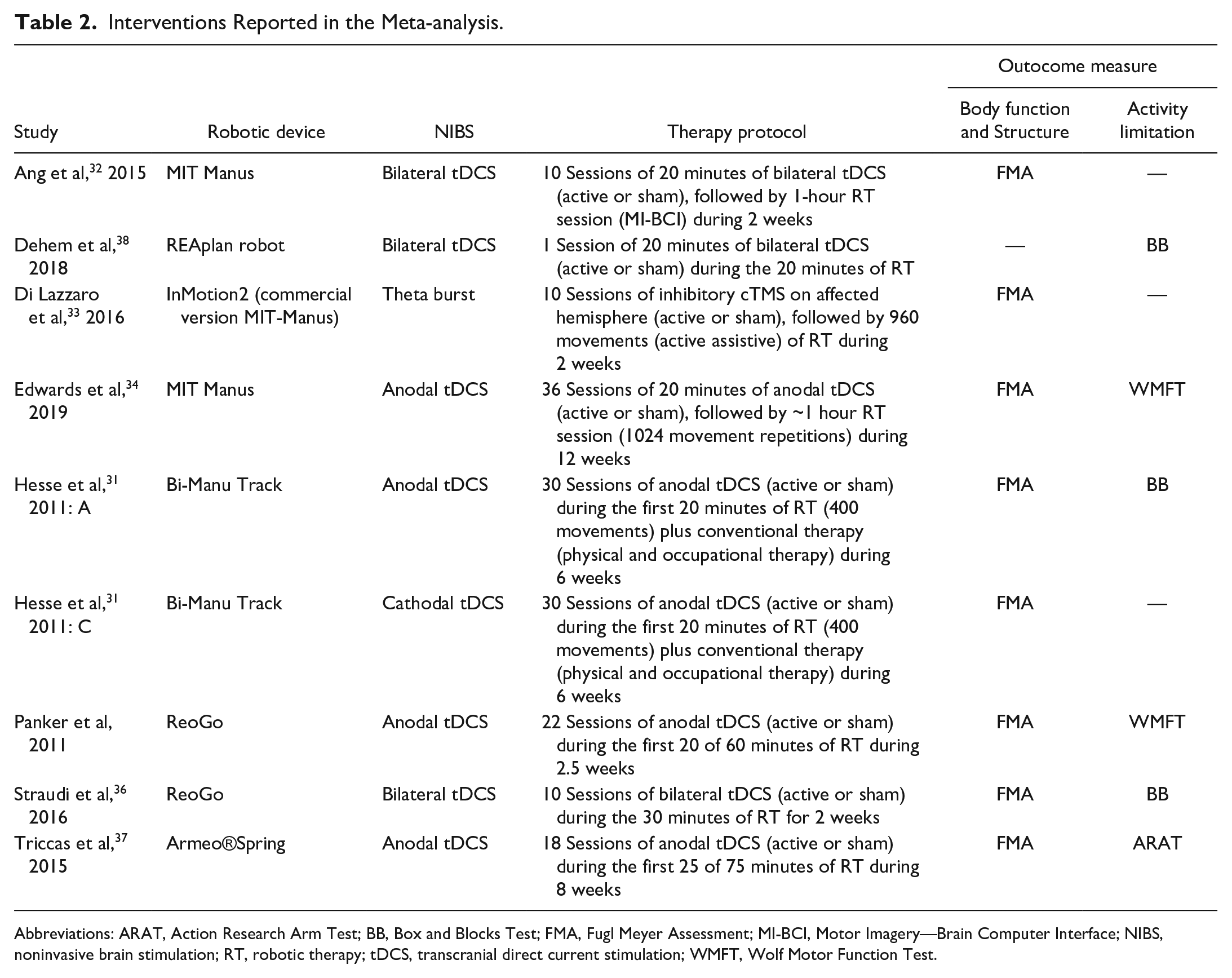
Abbreviations: ARAT, Action Research Arm Test; BB, Box and Blocks Test; FMA, Fugl Meyer Assessment; MI-BCI, Motor Imagery—Brain Computer Interface; NIBS, noninvasive brain stimulation; RT, robotic therapy; tDCS, transcranial direct current stimulation; WMFT, Wolf Motor Function Test.
The robot devices used in the studies were the following: InMotion2, Mit Manus, BiManu Track, ReoGo, Armeo®Spring, and REAplan robot. Their characteristics are shown in Supplementary Material, Table SM2.
Methodological Quality
Risk-of-bias assessment can be found in Supplementary Material, Figure SM2.
Main Analysis
Body structure/body function: 7 of the included studies31-37 chose the FMA as an outcome measure of body structure/function. Overall, a nonsignificant homogeneous SES (MD = 0.15; 95% CI = −3.10 to 3.40; P = .93; I2 = 0%) was found (Figure 1).
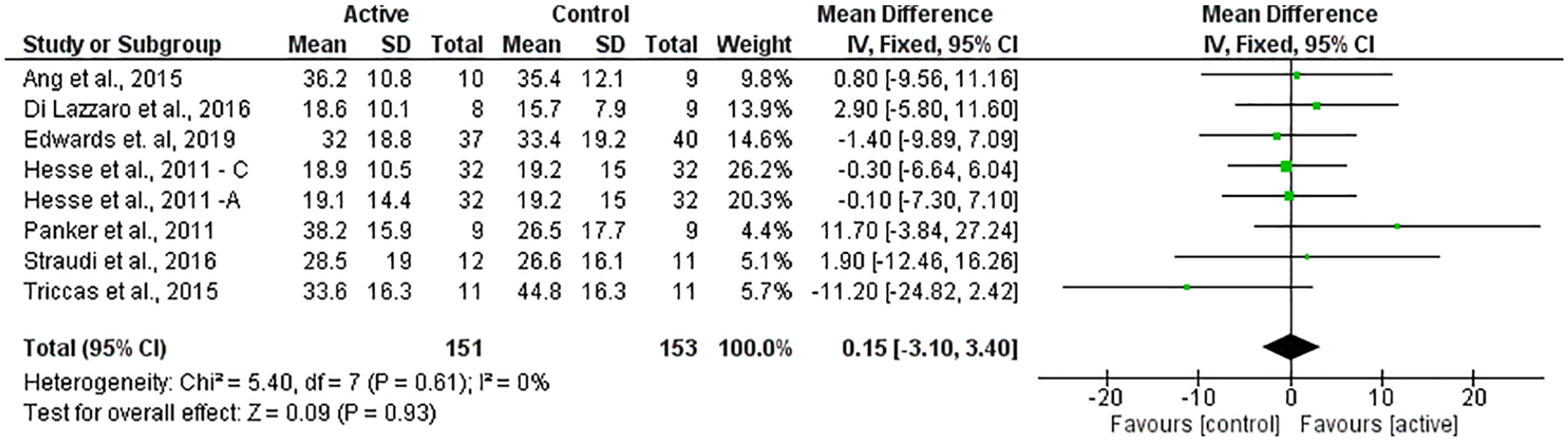
Forest plot of all trials comparing RT associated with NIBS (active) versus RT associated with sham NIBS (control) for body structure/function outcomes.
Activity Limitation
Six of the included studies31,34-38 evaluated eligible scales of activity limitation: ARAT, 37 BBT,31,36,38 MAL, 36 and WMFT.34,35 Of the 2 studies that assessed the WMFT, one reported the WMFT-Time score (time required to complete the tasks: a lower score was regarded as positive compared with a higher score). 35 The other study described the WMFT Functional Ability Scale score (a higher score was regarded as positive compared with a lower score). 34 Data from Hesse et al 31 were not available. Therefore, results of 5 studies34-38 were analyzed. Overall, a nonsignificant homogeneous SES (SMD = 0.03; 95% CI = −0.28 to 0.33; P = .87; I2 = 0%) was found (Figure 2).
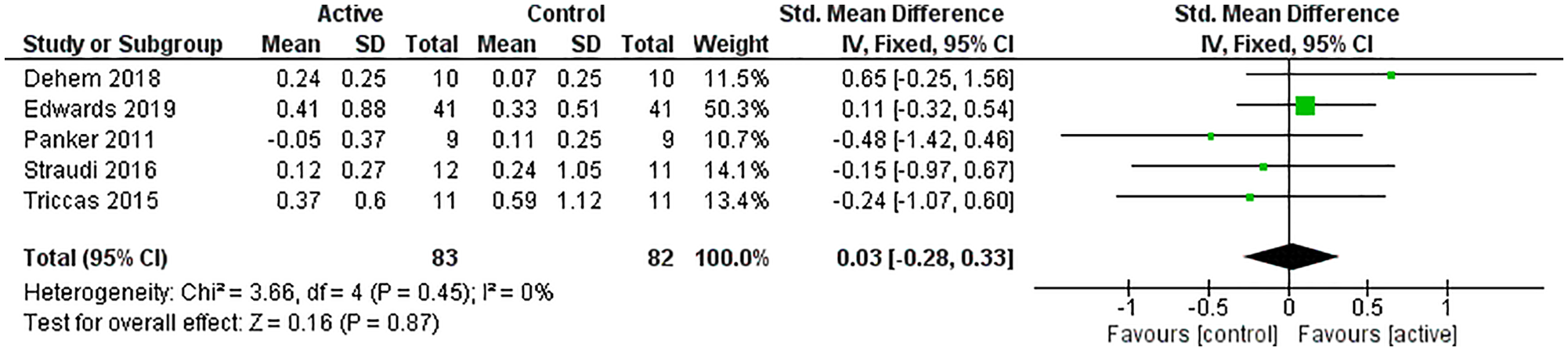
Forest plot of trials comparing RT associated with NIBS (active) versus RT associated with sham NIBS (control) for activity limitation outcomes.
Subgroup Analyses
Subgroup analyses are shown in Table 3.
Paradigms of Studies Included in Subgroup Analyses.
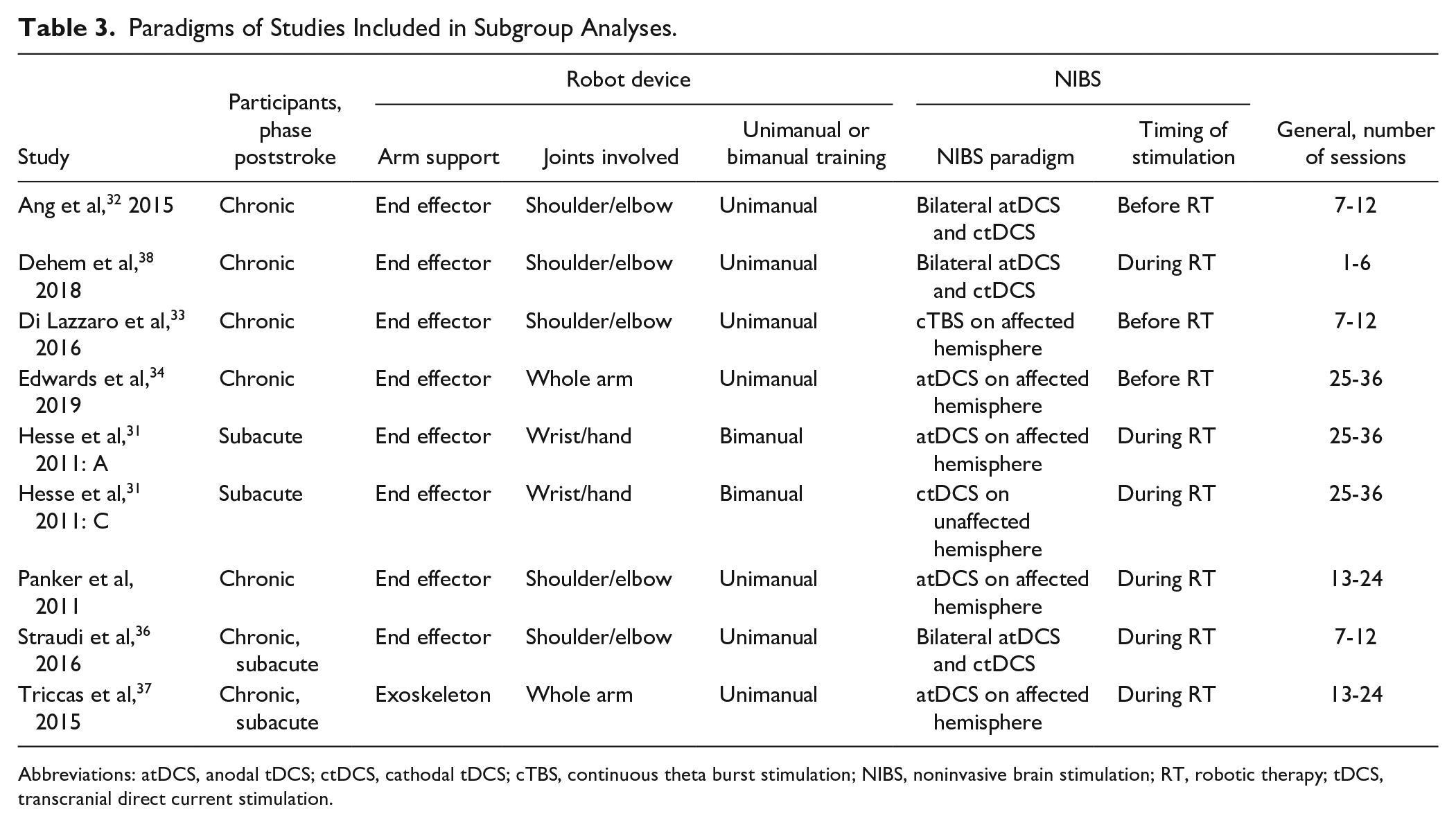
Abbreviations: atDCS, anodal tDCS; ctDCS, cathodal tDCS; cTBS, continuous theta burst stimulation; NIBS, noninvasive brain stimulation; RT, robotic therapy; tDCS, transcranial direct current stimulation.
Phase Poststroke
Body structure/body function: Nonsignificant homogeneous SESs were found for the chronic (MD = 1.18; 95% CI = −3.13 to 5.50; P = .59; I2 = 0%) and subacute (MD = −1.60; 95% CI = −6.14 to 2.94; P = .49; I2 = 36%) phases (Supplementary Material, Figure SM3).
Activity limitation: Nonsignificant homogeneous SESs were found for participants in the chronic (SMD = 0.10; 95% CI = −0.23 to 0.43; P = .56; I2 = 30%) and subacute phases (SMD = −0.28, 95% CI = −1.14 to 0.59, P = .53, I2 = 0%; Supplementary Material, Figure SM4).
Arm Support
Body structure/body function: Nonsignificant homogeneous SESs were found for end-effector (MD = 0.83, 95% CI = −2.51 to 4.18; P = .62; I2 = 0%) and exoskeleton (MD = −11.20; 95% CI = −24.82 to 2.42; P = .11) devices (Supplementary Material, Figure SM5).
Activity limitation: Nonsignificant homogeneous SESs were found for end-effector (SMD 0.07, 95% CI -0.26 to 0.40; P = .69, I2 = 7%) and exoskeleton (SMD = −0.24; 95% CI = −1.07 to 0.60; P = .58) devices (Supplementary Material, Figure SM6).
Joints Involved
Body structure/body function: Nonsignificant homogeneous SESs were found for the comparisons involving robots with only shoulder/elbow movements (MD = 3.28; 95% CI = −2.35 to 8.91; P = .25; I2 = 0%), robots with wrist movements only (MD = −0.21; 95% CI = −4.97 to 4.55; P = .93; I2 = 0%), and robots with whole arm movements (MD = −4.14, 95% CI = −11.35 to 3.06, P = .26, I2 = 30%; Supplementary Material, Figure SM7).
Activity limitation: Nonsignificant homogeneous SESs were found for the comparison involving robots with only shoulder/elbow movements (SMD = 0.01; 95% CI = −0.50 to 0.52; P = .98; I2 = 36) and for the comparison involving robots with whole arm movements (SMD = −0.04, 95% CI = −0.35 to 0.42, P = .85, I2 = 0%; Supplementary Material, Figure SM8).
Unimanual or Bimanual Training
Only 1 trial 31 used a robotic device with bimanual training. The results of this trial were split according to the intervention performed: Hesse et al, 31 A (atDCS) and Hesse et al, C (ctDCS).
Body structure/body function: For unimanual (MD = 0.47; 95% CI = −3.97 to 4.90; P = .84; I2 = 7%) and bimanual (MD = −0.21; 95% CI = −4.97 to 4.55; P = .93; I2 = 0%) training, nonsignificant homogeneous SESs were found.
Activity limitation: A nonsignificant homogeneous SES (SMD = 0.03; 95% CI = −0.28 to 0.33; P = .87; I2 = 0%) was found.
NIBS Paradigm
Three studies used atDCS to improve cortical excitability of the affected hemisphere34,35,37; 3 studies placed the tDCS anode over the affected hemisphere and the cathode over the unaffected hemisphere32,36,38; and 1 study used atDCS and ctDCS in different groups. 31 Just 1 study designed a proof-of-principle trial, aiming to induce long-term depression-like changes on the affected hemisphere by continuous theta burst stimulation (cTBS), a specific paradigm of repetitive TMS. 33
Body structure/body function: For atDCS, a nonsignificant heterogeneous SES (MD = −0.78; 95% CI = −5.62 to 4.06; P = .75; I2 = 37%) was found. For ctDCS, a nonsignificant SES (MD = −0.30; 95% CI = −6.64 to 6.04; P = .93) was found. For bilateral atDCS and ctDCS, a nonsignificant homogeneous SES (MD = 1.18; 95% CI = −7.22 to 9.58; P = .78; I2 = 0%) was found. For cTBS of the affected hemisphere, a nonsignificant SES (MD = 2.90; 95% CI = −5.80 to 11.60; P = .51) was found (Supplementary Material, Figure SM9).
Activity limitation: For atDCS of the affected hemisphere, a nonsignificant homogeneous SES was found (SMD = −0.04; 95% CI = −0.39 to 0.32; P = .84; I2 = 0%). For bilateral atDCS and ctDCS, a nonsignificant homogeneous SES was found (SMD = −0.21, 95% CI = −0.40 to 0.82, P = .50, I2 = 40%; Supplementary Material, Figure SM10).
Timing of Stimulation
Body structure/body function: Nonsignificant homogeneous SESs were found for NIBS applied during RT (MD = −0.21; 95% CI = −4.34 to 3.93; P = .92; I2 = 17%) and for NIBS applied before RT (MD = 0.72, 95% CI = −4.52 to 5.97, P = .79, I2 = 0%; Supplementary Material, Figure SM11).
Activity limitation: Nonsignificant homogeneous SESs were found for both NIBS applied during RT (SMD = −0.06, 95% CI = −0.50 to 0.38, P = .79, I2 = 11%) and before RT (SMD = 0.11, 95% CI = −0.32 to 0.54, P = .062; Supplementary Material, Figure SM12).
Number of Sessions
In 1 crossover trial with 1 active and 1 sham session of treatment, the results of the first session were included in the analysis. 38 The other trials had a parallel design.
Body structure/body function: Nonsignificant homogeneous SESs were found for trials which had 7 to 12 sessions (MD = 2.01; 95% CI = −4.03 to 8.05; P = .51; I2 = 0%) and from 25 to 36 sessions (MD = −0.50; 95% CI = −4.65 to 3.66; P = .81; I2 = 0%). A nonsignificant heterogeneous SES was found for trials with 13 to 24 sessions (MD = −0.07, 95% CI = −22.50 to 22.36, P = 1.00, I2 = 79%; Supplementary Material, Figure SM13).
Activity limitation: Nonsignificant homogeneous SESs were found for the first session of the crossover trial (SMD = 0.65; 95% CI = −0.25 to 1.56; P = .16) and for trials with 7 to 12 sessions (SMD = −0.15; 95% CI = −0.97 to 0.67; P = .71), 13 to 24 sessions (SMD = −0.35; 95% CI = −0.97 to 0.28; P = .28; I2 = 0%), and 25 to 36 sessions (SMD = 0.11, 95% CI = −0.32 to 0.54, P = .62; Supplementary Material, Figure SM14).
Quality of the Evidence
An evidence profile was performed on GRADEpro GDT. Quality was considered high for both body structure/function and activity limitation outcome measures, suggesting that further research is very unlikely to change the confidence in the estimate of the effect with the paradigms used in these studies 16 (Supplementary Material, Table SM3). However, quality of the evidence ranges from very low to high scores according to subgroup analysis (Supplementary Material, Table SM4).
Discussion
A total of 324 individuals were included in this meta-analysis, and there was no statistical heterogeneity in the results. Overall, the quality of the evidence was high. Despite the increase in the number of clinical trials that assessed NIBS as an add-on intervention of RT over the past years, there is no evidence to support the hypothesis that effects of RT on outcomes of body structure and function or on outcomes of activity limitation can be enhanced by NIBS. This conclusion is in line with the conclusion of a previous narrative review. 12 Despite the lack of statistical heterogeneity, however, the NIBS paradigms and motor training protocols were very diverse across studies.
Four different NIBS paradigms were used in the included studies, and all, except one, were based on the hypothesis of the interhemispheric competition model. This hypothesis seems not to apply to patients with stroke with different levels of impairment39,40 or lesion locations. 41 Other variables that raise concerns about the potential of NIBS as a therapeutic tool are intersubject and intrasubject variability that can be caused by individual brain anatomy, level of ongoing cortical activity, muscle precontraction, participant attentional focus, and even menstrual cycle and circadian rhythms. 42 Neuronavigated NIBS and electroencephalogram monitoring of stimulation effects in real time may be useful to decrease variability of effects. 42 To select patients for clinical trials or proof-of-principle studies involving NIBS and motor training, understanding of mechanisms and biomarkers of responsiveness may be key to enhance the probability of success. 43
The need to tailor NIBS interventions to particular characteristics has been underscored by the negative results of the NICHE study, the largest clinical trial to date in which low-frequency rTMS was administered to the unaffected hemisphere of patients in the chronic phase after stroke with the goal of downregulating excessive inhibition of the affected hemisphere, prior to sessions of motor training. 44 Another plausible explanation of the negative results of the NICHE trial as well as those of this meta-analysis is a ceiling effect: the impact of high-intensity motor practice on improvement of performance may exceed the magnitude of NIBS effects.
Alternatively, the association of NIBS and RT may have effects on retention of improved motor performance rather than on immediate effects of motor training. Results of a trial that randomized 164 patients to active or sham atDCS of the motor cortex of the affected hemisphere prior to RT, 34 included in this review, contradict this hypothesis. After 36 sessions of treatment, the benefits of RT were unchanged by add-on atDCS. However, at 6 months posttreatment, the percentage of “responders” (improvement in FM >5 points) was significantly greater in the control group than in the active group. Finally, it is necessary to consider that NIBS objectives are not only to enhance effects of training, but also to yield comparable results of a training intervention when administered for a shorter period of time. No studies addressing this issue were identified.
Another point that deserves attention is that an optimal training RT paradigm to be associated with NIBS has not yet been determined. Giacobbe et al 23 demonstrated that a single session of tDCS can enhance effects of motor training when applied immediately prior to RT but not during or after this intervention. Also, alternating sessions between proximal and distal joints seem to be a more efficient strategy to improve upper-limb motor performance than isolated block sessions of proximal and distal joints.11,34 It remains to be determined if this result also applies to NIBS + robotic training because no head-to-head comparisons of different paradigms of training associated with the same NIBS intervention were found. Finally, the role of robotic bimanual training remains to be clarified. This intervention may enhance the output to the paretic limb in some individuals with stroke, 45 but we only included in this review 1 study that applied this approach. 31
Clinical trials that test a large number of combinations of NIBS and training paradigms may be challenging as a result of resource and time constraints. Kinematic measurements are sensitive and correlate well with clinical assessment in patients with stroke. 46 Therefore, proof-of-principle studies that assess responsiveness to different paradigms may inform the design of larger clinical trials.
None of the studies included in this meta-analysis chose outcomes of activity limitation as primary end points, and only 1 study assessed the ARAT, 37 an outcome recommended by the Stroke Recovery and Rehabilitation Roundtable. 18 Future studies may include this outcome measure as performed in the RATULS (Robot Assisted Training for the Upper Limb after Stroke) study. 11 No significant changes were reported for the ARAT in RATULS, 11 a trial in which RT was not associated with functional training. On the other hand, a smaller trial compared RT training (n = 22) with RT associated with therapist-assisted functional training (n = 23) and found significant improvement on activity limitation favoring the experimental group. 47 Overall, these results suggest that changes in measures of body structure and function or activity limitation may vary according to the specific model of motor training. It is possible that translation-to-task interventions as add-on therapies to RT are required to ameliorate activity limitation. In trials that plan to address the possibility of boosting the effect of RT by adding NIBS to enhance activity, inclusion of translation-to-task interventions should be considered.
Limitations
The number of included studies in this systematic review was relatively low, but the sample size (n = 324) was greater than those reported in other meta-analyses about effects of specific rehabilitation interventions in stroke. For instance, a meta-analysis 48 that evaluated the effectiveness of a combination of NIBS and virtual reality included 213 individuals. Nevertheless, one needs to interpret our result with the appropriate caveats: the small pooled sample size, the large number of combinations of NIBS and RT, and the lack of details about baseline motor impairment and intensity of robot training protocols. Yet one could argue that the fact that our approach led to an inconclusive result might be an indication that the benefit of NIBS added on top of RT might be small, if any, and represent the natural limitation of the remaining neuronal substrate. This systematic review and meta-analysis points to directions of future research: to define whether one or more types of NIBS can be useful as add-on therapies to RT, we might track the profile and speed of the improvements rather than simply register the changes from admission to completion of the protocols.
Conclusion
According to this systematic review and meta-analysis, at the moment, there are not enough data about the benefits of NIBS as an add-on intervention to RT on upper-limb motor function or activity in individuals with stroke. There is a need for homogeneous clinical trials regarding participants’ characteristics, kind of robotic device, NIBS paradigm, duration of treatment, and primary end points.
Supplemental Material
sj-docx-1-nnr-10.1177_1545968321989353 – Supplemental material for Effects of Robotic Therapy Associated With Noninvasive Brain Stimulation on Upper-Limb Rehabilitation After Stroke: Systematic Review and Meta-analysis of Randomized Clinical Trials
Supplemental material, sj-docx-1-nnr-10.1177_1545968321989353 for Effects of Robotic Therapy Associated With Noninvasive Brain Stimulation on Upper-Limb Rehabilitation After Stroke: Systematic Review and Meta-analysis of Randomized Clinical Trials by Suzana Bleckmann Reis, Wanderley Marques Bernardo, Carlos Andre Oshiro, Hermano Igo Krebs and Adriana Bastos Conforto in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SBR, WMB, CAO, and ABC have no conflicts of interest. HIK has a patent Bionik Laboratories with royalties paid to Massachusetts Institute of Technology.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the MIT International Science and Technology Initiatives (MISTI Brazil TVLM), under grant number 2160714.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
