Abstract
Background. While prior preliminary studies have broadened our understanding of how repetitive transcranial magnetic stimulation (rTMS) improves language outcomes in stroke patients with nonfluent aphasia, the evidence base of the effectiveness of this method remains inadequate. Objective. In this study, we aimed to strengthen the evidence that this approach improves language performance and to identify characteristics of patients predisposed to benefit most from this treatment. Methods. Fifty-six stroke patients with nonfluent aphasia were randomly allocated to a real or a sham stimulation group: Group A (n = 33), who underwent 10 sessions of 1-Hz rTMS over the contralesional pars triangularis (PTr), and Group B (n = 23), who received sham 1-Hz stimulation. We performed the Picture Naming Test and the Concise Chinese Aphasia Test (CCAT) at the baseline, post-rTMS intervention, and at 3-month follow-up. Results. Group A showed significantly greater improvement than Group B in CCAT scoring (P < .001), object-naming accuracy (P = .01), and naming reaction time (P = .004). The CCAT scoring and naming testing changes for Group A were persistent at 3 months following intervention (P = .008). Patients who had a lower contralesional rest motor threshold (rMT) were predisposed to a favorable therapeutic outcome (P = .006), independent of aphasia type, severity, and duration. Conclusions. The results of this study provide evidence that inhibitory rTMS, through downregulating the circuitry of the right pars triangularis (PTr), achieves a persistent and broadly modulating effect, irrespective of aphasia severity and subtype. Patients who show lower rMT in the right motor system would seem to benefit the most from inhibitory rTMS.
Keywords
Introduction
Aphasia is a devastating and intractable consequence of stroke that has tremendous impact on daily activities. Where spontaneous aphasia recovery occurs, this largely takes place in the first 2 to 3 months after a stroke, taking a slower rate and longer progress time compared with spontaneous motor recovery.1,2 Current aphasia therapies largely rely on compensatory, repetitive, or augmentative training, which are unsatisfactory for the majority of patients, who are left with communication deficit despite vigorous treatment intervention. 3
Language recovery after a stroke depends significantly on the degree of neuroplastic change, which is usually associated with reorganization and reconnection of the lesioned and perilesional dominant hemisphere regions, acquisition or unmasking of the homologous language area in the nondominant hemisphere, or activation of the nondominant cortical region. 4 The recruitment patterns that aphasic patients activate vary, depending on the location of the lesion, which part of the brain circuitry has been disrupted, the time course of injury, and premorbid differences in language lateralization. 5 Recently, noninvasive neuromodulation techniques such as repetitive transcranial magnetic stimulation (rTMS) used to facilitate neuroplasticity have shown explorative potential, based on the mechanisms of long-term potentiation and long-term depression. However, the role of the contralesional cortex in neuroplastic change remains at the center of continuing debate over viable approaches to language recovery. Heiss and Thiel have postulated that the homologous area in the nondominant hemisphere of aphasia patients may take over the role of the affected language area of the left hemisphere after a stroke, particularly among patients with extensive left hemisphere injury. 6 In other studies, neuroimaging has revealed robust activity taking place in the homotopic right hemisphere (RH) region of aphasia patients, coupled with left language-area activity.7-9 However, recent findings have indicated that an upsurge in right hemispheric activity following a stroke may not represent entirely beneficial neuroplasticity. Instead, when considered according to interhemispheric imbalance modeling, it may reflect insufficient or maladaptive change. 5 Comparisons of correct and incorrect naming responses have indicated that an increase in transcallosal inhibition from the RH on the left perisylvian areas is associated with worse language performance. 10 A number of exploratory studies in which small groups of chronic and subacute aphasia subjects received low-frequency inhibitory rTMS to the right pars triangularis (PTr; the anterior portion of the RH homologous to the Broca’s area) have shown promise for language recovery.11,12 However, the reliability of such approaches has not yet been convincingly demonstrated. Additional investigations and a wider analysis using larger cohorts of patients are needed to pinpoint relevant patient-specific factors that have an impact on language outcomes. If these factors are better defined, it may provide evidence underpinning the contributory role of the contralesional PTr.
In this study, our team further tested the concept that downregulating the intact contralesional cortical areas may help engage more efficient language processes by diminishing the impact of transcallosal imbalance. In this randomized, sham-controlled study, our aims were, first, to assess the short-term and ongoing efficacy of this protocol for achieving language recovery in chronic aphasic patients with a 3-month follow-up and, second, to identify the specific characteristics of patients who are predisposed to benefit most from this treatment.
Methods
Subjects
Seventy-five patients admitted to the stroke unit of a tertiary medical center were evaluated consecutively for participation in this study. Two patients declined to participate, and 56 right-handed patients met the inclusion criteria (Figure 1): (a) diagnosis of nonfluent aphasia secondary to a first-ever left middle cerebral artery infarction with cortical involvement, confirmed by magnetic resonance imaging; (b) at least 3 months after stroke and in a stable medical and cognitive condition; (c) no history of dementia, cognitive impairment as evaluated by Mini-Mental State Examination, 13 or other confirmed neurodegenerative diseases; (d) absence of severe global aphasia or fluent aphasia, spatial neglect, visual field deficit, or emotional problems, as evaluated by the Aphasic Depression Rating Scale 14 ; and (e) absence of TMS contraindication. Handedness was evaluated using the Edinburgh Handedness Inventory. 15
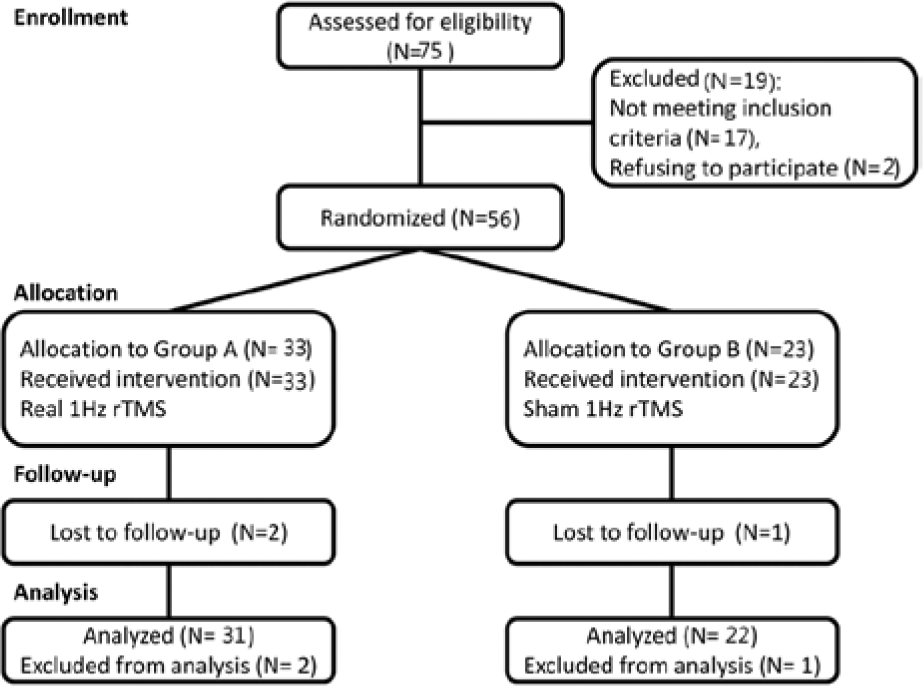
Flowchart for recruitment, group allocation, allocation of treatment, follow-up, and analysis.
The 56 patients recruited to the study (15 women and 41 men aged 39-80 years) gave their written informed consent prior to participating, in accordance with the 2008 Declaration of Helsinki and with the approval of the local institutional review board.
Experimental Design
We conducted a sham-controlled, double-blinded parallel study design. The randomization order was computer generated and concealed in sequentially numbered opaque envelopes by an independent statistician. All subjects were allocated to Group A or Group B by a researcher blinded to the assessment, and matched for aphasia severity, aphasia type, and time since stroke. The experimental group (Group A) comprised 33 subjects, each of whom underwent 10 minutes of 1-Hz rTMS treatment for 10 consecutive days. Twenty-three subjects were assigned to the sham group (Group B), which proceeded along the same time frame as Group A. All the subjects continued their conventional speech rehabilitation program and other medical treatments regardless of which group they were assigned to. On every day of the procedure and within 30 minutes after undergoing real or sham rTMS intervention, all the participants undertook the same amount of 1-hour speech therapy, conducted by a therapist blinded to group allocation. The speech therapy emphasized on the expressive production including semantic training, phonemic training, repetition, naming, conversation, picture-description tasks, and phrase generation tasks, based on the learning principle of constraint-induced therapy. The training difficulty level was adjusted according to individual communicative capacity with the aim of reaching adequate training intensity, which was evaluated weekly. Other substitutive modalities, such as using drawing, gesturing, or melody intonation, were not encouraged during the training course and self-practice activity. The training exercises at home involved mainly picture-naming activity and were assisted by the patient’s family members; these sessions lasted 30 minutes per day.
Interventions
Determination of Stimulation Target
The TMS conditioning took place after 3-T structure magnetic resonance imaging (MRI) acquisition. A computerized high-resolution whole-brain T1-weighted image was acquired for each patient (TR = 8.20 ms, TE = 3.24 ms, number of slices = 180, slice thickness = 4 mm, field of view = 23 × 23 cm, magic angle turning = 256 × 256) on a General Electric (Milwaukee WI) scanner. We used a frameless stereotaxic system (Brainsight, Rogue Research, Montreal, Canada) in real time to coregister the stimulation target area in relation to the location of the subject and coil in the intact hemisphere, which is defined ventrally by the horizontal ramus of the sylvian fissure and caudally by the vertical ramus of the sylvian fissure (homologous to the left dorsal anterior pars triangularis). 16 Within the right inferior frontal gyrus, the dorsal anterior pars triangularis (DA PTr, Brodmann Area [BA] 45) effectively incorporates the target area for inhibitory rTMS, but not the nearby anterior pars opercularis (BA44) or the ventral posterior PTr, which reversely depress naming ability, according to study findings of Naeser et al. 17 Brain lesion, location, and the presence of periventricular, subcortical hyperintensity indicating white matter lesions were all registered during the MRI examination.
Determination of Resting Motor Threshold
We established the resting motor threshold (rMT) for MEP as the lowest intensity at which motor evoked potential (MEPs) of at least 50 µV amplitudes could be elicited in half of 10 consecutive stimuli. 18 Each patient sat in a comfortable reclining armchair with both hands relaxed and keeping their eyes open. We recorded MEPs bilaterally from the first dorsal interosseous (FDI) hand muscles using surface Ag/AgCl electrodes. A Dantec Keypoint electromyograph (Dantec, Skovlunde, Denmark) was connected to the stimulator to record the MEP signals.
rTMS Protocol
We performed transcranial magnetic stimulation using Magstim Rapid 2 (Magstim Company, Withland, Dyfed, UK) through a 70-mm figure-of-eight coil. One-hertz rTMS trains consisting of 600 pulses were applied for 10 minutes at 90% of rMT over the target area. The stimulation protocol for Group B was identical to Group A, but in this case a placebo coil (Magstim) for the sham stimulation was used; this delivered less than 5% of the magnetic output with audible click-on discharge. Since none of the patients had ever undergone rTMS, they could not identify whether the stimulation was real or sham. Intervention for both groups consisted of 10 daily sessions (5 days per week for 2 weeks).
Assessments
An independent assessor, blinded to treatment allocation, performed the baseline measurements shortly before the first session of intervention (baseline), on the day after the 10th session (post 1), and at 3 months after the last intervention session (post 2). All the participants underwent detailed clinical and neuropsychological examinations to ascertain (a) the severity of the stroke according to the National Institute of Health Stroke Scale (NIHSS) 19 ; (b) their physical and cognitive disability measured with Functional Independence Measure (FIM) 20 ; (c) object and action naming ability, using the Picture Naming Test; and (d) language performance using the Concise Chinese Aphasia Test (CCAT), the only standardized, linguistically, and culturally neutral assessment for native Mandarin Chinese speakers that tests all language modalities. 21 The CCAT consists of 9 subtests, and responses for each subtest are scored on a 12-point scale. The scoring of CCAT, calculated by averaging the scores of each subtest, ranges from 0 (minimum) to 12 (maximum). Severity of aphasia, stratified from slight to extremely severe, is composed of 7 grades: slight deficit (<12 points), mild (10-11 points), mildly moderate (9-10 points), moderate (7-9 points), moderately severe (5-7 points), severe (3-5 points), and extremely severe (0-3 points). 21 For this study, 4 subcategories of CCAT related to language production were evaluated during follow-up: (a) conversation, (b) family picnic picture description, (c) object naming and expression, and (d) repetition.
To measure changes in picture naming and category analyses, we created 2 sets of 20 object and action items selected from 373 black-and-white line drawings of objects and actions from the International Picture Naming Database. 22 We selected different action items executed by hands (such as writing, washing) to demonstrate somatotopic brain connection,23,24 and matched the items in each set for familiarity, frequency, visual complexity, and agreement measures. Each illustration was presented to each subject in order, with 20 seconds allowed for the item to be identified. Total reaction time (RT) was recorded and averaged for comparison. If the patients succeed in completing the word correctly after receiving a phonemic cue, we recorded the response as “failed.” Items named correctly with no phonemic cue were recorded as “accurate.”
Statistics
At baseline assessments, the mean values of the clinical and aphasia assessments between the groups were compared using one-way analysis of variance (ANOVA) for independent samples for continuous data or χ2 tests for categorical data.
We completed the intergroup comparisons of linguistic performance using repeated-measures ANOVA with time (levels: 1, baseline; 2, Post1; 3, Post2) as the within-patient factor and the groups (levels: 1, Group A; 2, Group B) as the between-patient factor. Analysis of covariance (ANCOVA) was employed to determine the influence of time poststroke, aphasia types, baseline aphasia severity, and comorbidities of diabetes mellitus (DM), hypertension, and hyperlipidemia on the improvement of CCAT score within the real group A and on intergroup comparisons. We used the Spearman test to perform correlation analysis of performance change and baseline clinical features. The level of significance was set at P < .05.
Results
No subjects reported any adverse effects during the course of the study or after the 3-month follow-up. Table 1 shows the averaged group values of the biographic data and clinical features of all the subjects. There were no differences in the baseline features, such as the period poststroke, aphasia types and grading, status in terms of cognition and functional independence (functional independence measurement) for intergroup comparison (P > .05, Table 1). The majority of our subjects were graded as having either moderate (n = 24) or severe aphasia (n = 22), followed by extremely severe (n = 6), mildly moderate (n = 2), and mild aphasia (n = 2).
Biographic Data and Clinical Features of All Subjects.
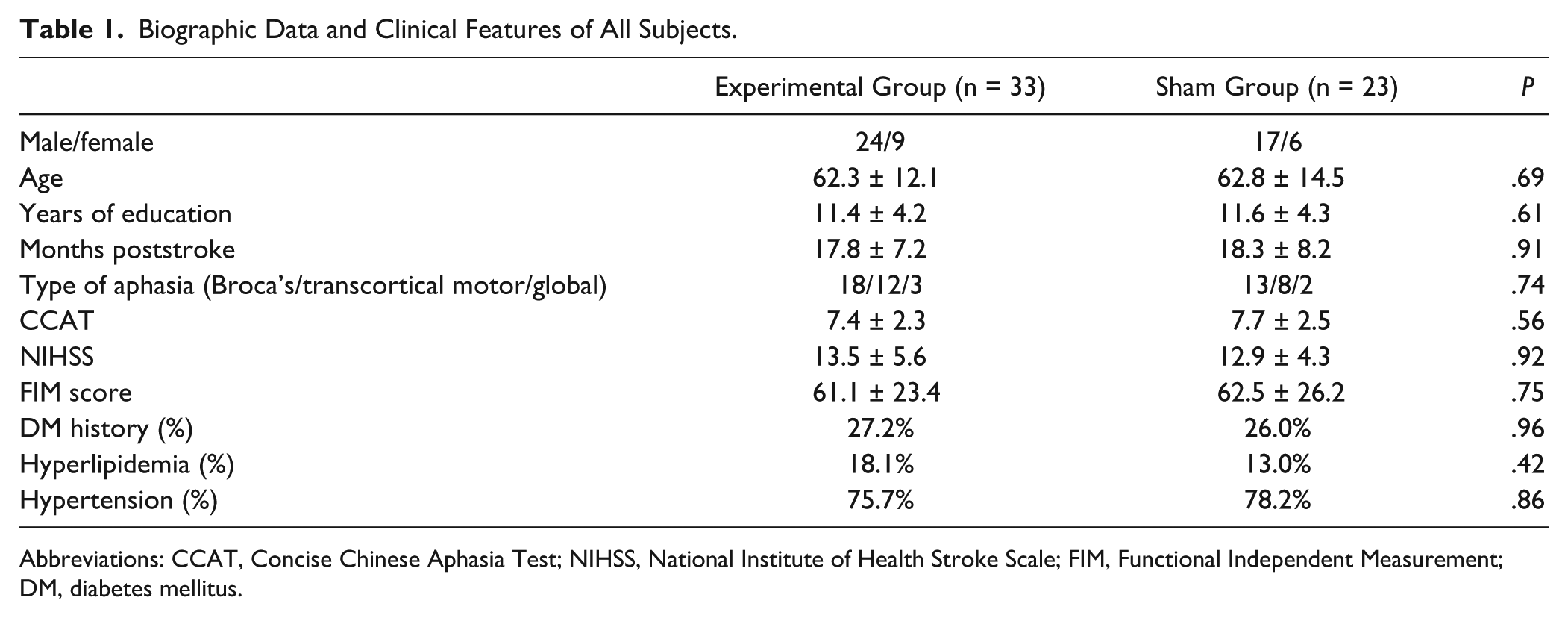
Abbreviations: CCAT, Concise Chinese Aphasia Test; NIHSS, National Institute of Health Stroke Scale; FIM, Functional Independent Measurement; DM, diabetes mellitus.
Real Versus Sham Group Comparison
Following rTMS intervention, Group A showed significant improvement over Group B in overall CCAT scoring (F[1,54] = 13.98, P < .001) and in CCAT subcategories: conversation (F[1,54] = 4.81, P = .032), description (F[1,54] = 6.32, P = .024), expression (F[1,54] = 9.68, P = .002), and repetition (F[1,54] = 6.01, P = .023) (see Table 2). Within Group A, significant differences were found in overall CCAT scoring (P = .001), conversation (P = .002), expression (P = .002), and repetition (P = .012), relative to pretreatment measurements.
Mean Group Data (±SD) of CCAT Scores and Naming Tests for the experimental Group A and the sham Group B After rTMS Interventions.
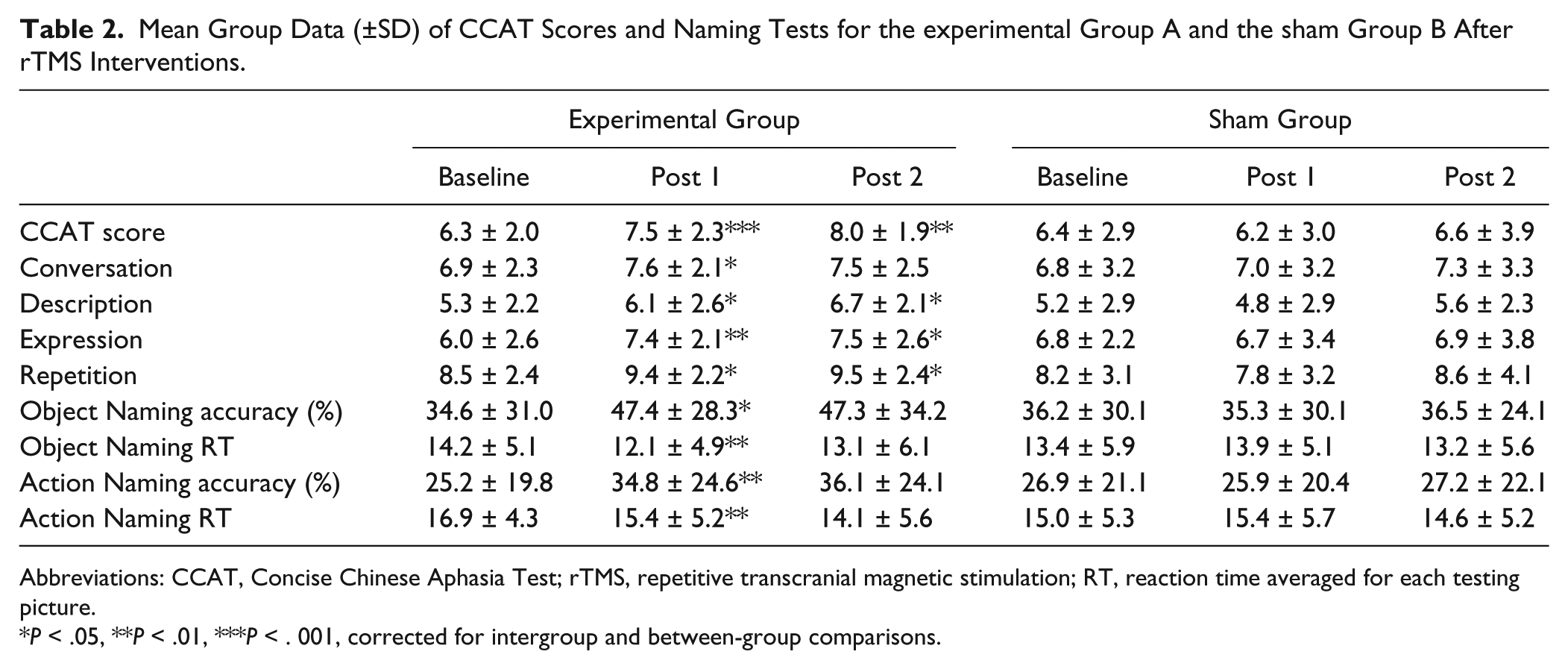
Abbreviations: CCAT, Concise Chinese Aphasia Test; rTMS, repetitive transcranial magnetic stimulation; RT, reaction time averaged for each testing picture.
P < .05, **P < .01, ***P < . 001, corrected for intergroup and between-group comparisons.
Group A also manifested higher object naming accuracy (F[1,54] = 7.24, P = .01), action naming accuracy (F[1,54] = 9.16, P = .003), shorter object naming RT (F[1,54] = 8.38, P = .004), and action naming RT (F[1,54] = 9.53, P = .005) than Group B and in comparison with its own baseline levels (P = .01, .001, .002, and .003, respectively). At 3 months postintervention, Group A continued to show statistical differences in overall CCAT scoring (F[1,51] = 8.69, P = .008), description (F[1,51] = 4.32, P = .031), expression (F[1,51] = 5.12, P = .026), and repetition (F[1,51] = 7.48, P = .013) subscales relative to their baseline data.
When the potential mediating factors such as aphasia type, severity, duration, and the presence of comorbidities were included as covariates, the intergroup comparisons continued to yield significant differences in CCAT scoring (F[1,43] = 10.49, P = .004), conversation (F[1,43] = 4.66, P = .041), expression (F[1,43] = 6.86, P = .015), and repetition (F[1,43] = 6.53, P = .018). Among the variables we recruited for predicting therapeutic efficacy, only the presence of a history of DM indicated a significant influence on the improvement of CCAT scoring for Group A (F[7,25] = 4.84, P = .036). DM comorbidity yielded a negative impact on CCAT improvement in comparison with the patients without a DM history (P = .024, Figure 2A).
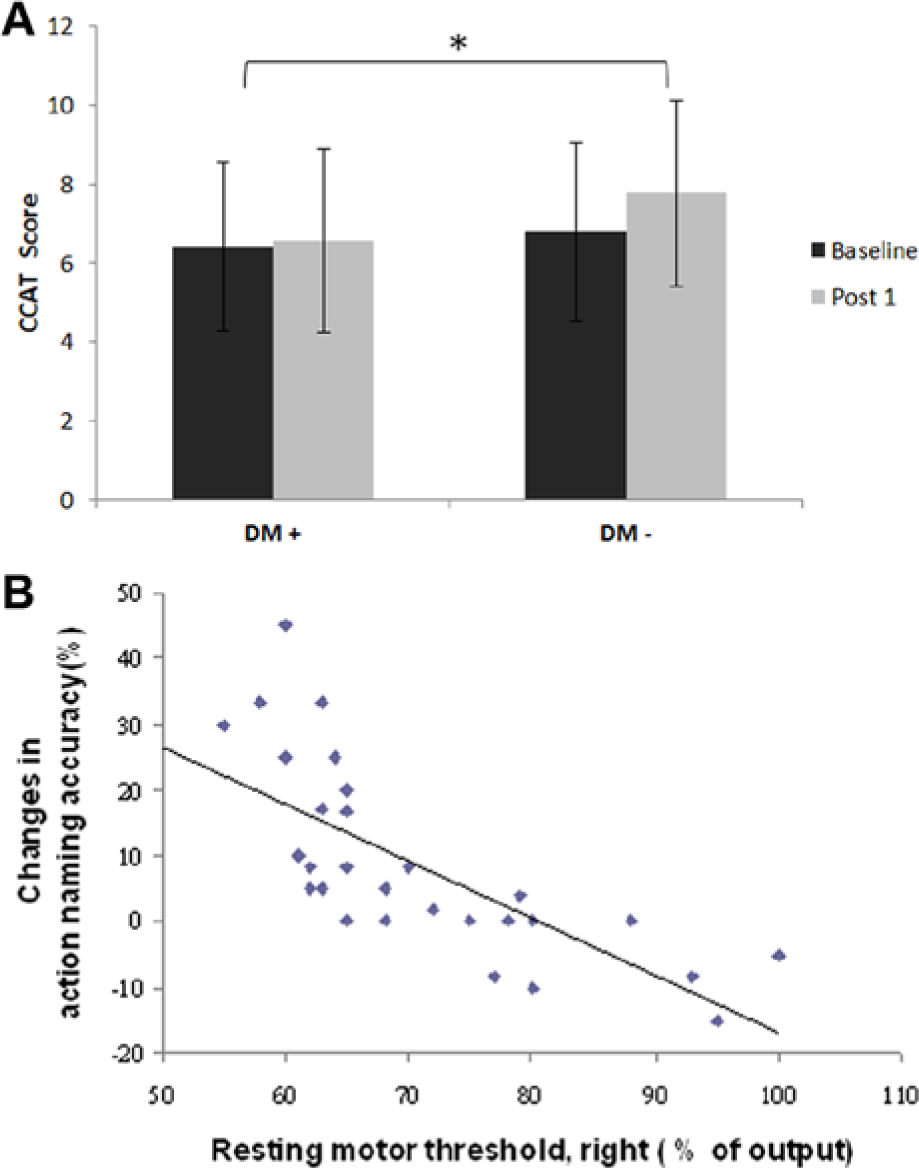
(A) Subgroup analyses revealed that the patients with DM comorbidity (DM+) yielded lower CCAT scoring improvement (P = .024) compared with patients with no DM history (DM−). (B) Correlation between corticomotor excitability and gain in action-naming performance. This scatter plot shows the negative correlation (r = −.67; P = .006) between right rMTS recorded in FDI and improvement in action naming accuracy after rTMS intervention (post 1).
Correlation Analyses
The effect of rTMS intervention on all Group A subjects, as measured by CCAT scoring and naming testing, did not correlate with either the demographic data (subjects’ age, education status, NIHSS, aphasia type and severity, and time poststroke) or the baseline FIM scores. Notable relationships were found between the contralesional rMT and improvement to action naming accuracy (r = −.67, P = .006; Figure 2B). There was no further significant relationship found in other demographic variables or clinical features relative to treatment outcomes.
Discussion
Our study supports a growing body of evidence that inhibition of the contralesional pars triangularis enhances the language recovery of individuals with chronic nonfluent aphasia following a stroke. In this instance, aphasic patients who underwent inhibitory rTMS conditioning continued to demonstrate persistent language improvement 3 months after the intervention, with DM-related aftereffects and invariance in the representation of aphasia type, severity, and duration. The significant correlation between naming performance and contralesional motor threshold provides further evidence that right PTr suppression brings about greater benefit to a subset of patients with stronger rightward asymmetries.
Functional imaging experiments have identified that aphasic patients recruit different networks of brain regions in both hemispheres in attempting language tasks. Upsurges to left perilesional activity are primarily identified as having a crucial role in facilitating residual language function.25,26 The left orbitalis and anterior insula sites likely represent cortical activity, which has become augmented to compensate for lesioning of the inferior frontal language center. 5 Administering inhibitory rTMS to right PTr has known to exert bihemispheric modulating effect and concurrently promote ipsilesional reorganization mediated by recruitment of the above-mentioned potential areas. 4
We preferred using low-frequency rTMS as our first attempt for this adjuvant treatment because neuroimaging has indicated that there is robust homotopic activation in the right PTr regions after an aphasic stroke, which may compromise language network recovery. 5 In a recent study in which the authors performed an Activation Likelihood Estimation meta-analysis from functional imaging studies, they found that the right pars opercularis (POp) plays a causal role in phonologic processing (even in healthy subjects) whereas the right PTr is truly harmful to the operation of the language circuits. 5 Increased activity in this area, when malfunction of its normal role results in response inhibition, may hinder language production.5,27 The results of this study indicate that inhibitory rTMS applied to the right PTr could release its abnormal suppression of and negative influence on the dominant, lesioned language center, which is subsequently facilitated to engage more efficient lexical processing. An alternative pathway for the beneficial effect could be through the direct U-shape fiber connecting the right PTr and the POp. 28 The hyperactivity of right PTr may be able to suppress the facilitating effect of right POp, which seems to play a compensatory role in phonological process after damage to dominant Broca’s area. 29 Right POp extensively connects with temporo-parietal cortex and premotor area via superior longitudinal fasciculus, and this supports the residual bilateral linguistic pathway.30,31 Suppressing the right PTr with rTMS could directly improve the functional execution of the right POp and hence benefit language recovery. Despite a variety of proposed pathways, up to the present, the exact mechanism underlying the success of this strategy remains unknown. However, in this study we have demonstrated the profound influence of the PTr suppression strategy on language processing and confirmed the applicability of this protocol to different language systems.
The significant correlation our team noted between right cortical excitability and linguistic gain following intervention further supports the possible existence of transcallosal imbalance or intracortical dysinhibition. The more intense is the right corticomotor hyperactivity, the more effective is the inhibitory rTMS treatment. It should be noted that the right corticomotor excitability cannot be simply interpreted as the neural activity of right PTr; this is because it may merely be a reflection of the extent of left motor cortex implication.5,32 However, there could be an another explanation for the revealed relationship. As is known, Broca’s area in the inferior frontal gyrus has a major function in the processing of meaning, sounds, and syntax, as well as many other nonlinguistic functions.33,34 There is a functional link between the meaning of words and specific motor centers that would generate corresponding actions. 23 Buccino’s research, which used transcranial magnetic stimulation (TMS) and a behavioral paradigm to assess whether listening to action-related sentences modulated motor system activity, found that the size of the MEP recorded in each effector muscle was only affected when participants listened to sentences or verbs containing actions referring to the specifically named effector. Action related to hand muscles did not affect the MEP recorded on the foot muscles. The study by Pulvermüller et al study found a further connection between word meaning and specific motor centers that would generate corresponding actions. This study found that the RTs on lexical decisions for action executed by the arms (eg, “washing”) and the legs (eg, “kick”) could be facilitated by single-pulse TMS over the arm or leg cortex, respectively. 35 Moreover, the left dorsolateral prefrontal cortex was preferentially affected by verbs in comparison to nouns.36,37 These results support our earlier findings whereby improvements in hand-related action naming have been shown to be tightly correlated with the rMT recorded in FDI (Figure 2B); this was observed during action naming and not during object naming. An important inference can be drawn here, namely, that patients who yield lower rMT in the right motor system may benefit the most from inhibitory rTMS.
With approximately half of our subjects experiencing moderate aphasia and the other half having severe or extremely severe aphasia, our analyses found that the possible covariates of aphasia severity did not drive the intergroup effect of rTMS intervention. Patients presenting with a variety of aphasia severity might have substantially improved as a result of inhibitory rTMS treatment independent of initial linguistic impairment. Jung et al, investigating the factors associated with good responses to transcranial direct current stimulation (tDCS) for treatment of aphasia, found that lower initial severity was associated with good responses. 38 However, a number of factors point to the need for caution in the interpretation of these results. The lack of a sham-controlled group in the study by Jung et al may have biased this finding. 38 Spontaneous recovery, which may be greater for mild aphasia patients, was indicated as a possible contributing factor to the significantly greater improvement. Moreover, the study subjects were heterogeneous in aphasia types and those who had fluent aphasia scored higher during assessment and responded well to treatment. A further possible reason for the divergent results between Jung et al and the current study could be the different intervention methods used, that is, the tDCS in their research and rTMS in this study. It is plausible that effective reorganization could be achieved by contralesional inhibitory rTMS, even when applied to patients with severely compromised aphasia in whom the substitutional cortex for efficient language generation could be inducted in the left orbitalis and anterior insula cortex. 5 The linguistic gain, irrespective of the severity of the aphasia secondary to the variation in brain insult, as registered in this study, further demonstrates the broad utility of this novel approach undertaken by our team.
In this study, time poststroke was proven not to be a contributing factor for the therapeutic outcome to rTMS treatment. Some of our participants who had suffered aphasia for a couple of years still showed significant linguistic enhancement post-rTMS modulation. These findings again indicate that this approach is able to achieve type-invariant and duration-invariant language modulation. In contrast, however, we were able to identify the presence of DM medical comorbidity was a predictor with respect to the efficacy of the treatment. Previous studies have demonstrated that an association exists between preemptive vascular disease in DM and ischemic brain injury, mediated by modifying adaptive neovascularization and postischemic neural remodeling.39,40 When people with DM experience progressive depletion of microvascular neuroprotection and their levels of cerebrovascular brain-derived neurotrophic factor (BDNF) become reduced, they have an elevated risk of neural injury and of maladaptation if they subsequently experience brain insult. 41 Cheeran et al found that subjects with different polymorphisms of the BDNF gene had different levels of susceptibility to rTMS modulation for long-term neuroplasticity. 42 From the neuroimaging viewpoint, in the subjects experiencing stroke, the ratio of insulin resistance has been found to correlate with higher grades of subcortical and white matter lesions as registered by brain MRI. 43 Such subcortical microangiopathy and related encephalopathy may impair corticospinal tract integrity, further compromising the therapeutic efficacy to modulation. For our subjects, in addition to the main stroke lesion, the incidence of concomitant white matter lesions in the DM group was 80%, in contrast to 48% in the non-DM group. This intergroup discrepancy may meanwhile contribute to their differential susceptibility to rTMS modulation. The findings of these investigations, linking diabetic neurotrophic dysfunction with the inferior modulating effect of rTMS conditioning, point to these neurovascular mechanisms as underlying factors in the decreased response of subjects with DM to the manipulation of neuroplasticity.
Emerging research that has explored the potential effect of inhibitory rTMS on language recovery has to date been based on relative small subject populations. The strengths of this study include the size of the study cohort, the strict inclusion criteria used, utility of a standardized aphasic assessment tool that yields 7 clearly defined levels of severity granularity, and the inclusion in our study of cofactor contributions to the subjects’ responses to the intervention. Given that a hemorrhagic stroke preferentially involves the subcortical areas of the brain—including the basal ganglia and corona radiate—and generally results in relatively mild consequences and better prognoses than a cortical stroke,44,45 we did not include any hemorrhagic stroke patients in the current study, which improved the homogeneity of our subjects.
The shortcomings of this study include the lack of neuroimaging data, which may have been able to provide robust evidence both on cortical reorganization secondary to inhibitory rTMS conditioning and on the nonidentification of the mechanisms that underlie recovery from aphasia. Likewise, the current study did not include excitatory rTMS applied to the perilesional cortical region, which has been reported as a promising strategy for poststroke aphasia. 46 However, our team has already employed this designation and incorporated advanced neuroimaging techniques in our ongoing exploration of the efficacy of this treatment. With respect to the influence on the therapeutic response of aphasic type, severity, and duration, further large-scale study is needed to address this issue for more conclusive findings, since this study, yielding as it did nonsignificant contributions from these factors, may have been of insufficient magnitude to be able to do so.
Overall, with patients’ enhanced language production and naming performance, sustained at least 3 months beyond the intervention and accompanied by neurophysiological correlation in corticomotor excitability, this study has provided evidence that inhibitory rTMS in nondominant PTr exerts a significant, unifying aftereffect on language recovery across various nonfluent aphasia types with differentially compromised ability. Our study has provided the first body of evidence that contralesional hyperexcitability and absence of DM comorbidity predispose aphasic patients to a better response to rTMS intervention. Further investigation with functional imaging evidence would more fully clarify the impact of rTMS on the structures involved in underlying neuroplasticity processes that allow recovery of language function by nonfluent aphasia stroke patients.
Footnotes
Acknowledgements
The authors thank Yu-Kuei Weng for her contribution to this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Taiwan National Science Council Grant (NSC Number 1012314B075003) and the Taipei Veterans General Hospital Grant (V102B004) .
