Abstract
Background. The role of the right hemisphere in poststroke aphasia recovery is still controversial and the effects of repetitive transcranial magnetic stimulation (rTMS) over the right homologous Broca’s region have been seldom investigated. Objective. This study aimed to compare the effect of excitatory, inhibitory, and sham rTMS delivered with H-coil over the right inferior frontal gyrus in chronic aphasic patients. Methods. Five right-handed poststroke aphasic patients underwent a picture naming task before and immediately after each of 3 sessions of rTMS: excitatory (10 Hz), inhibitory (1 Hz), and sham rTMS, in random sequence and separated by at least 1 week. Results. Only the excitatory 10-Hz stimulation was associated with a significant improvement in naming performance (P = .043) and was significantly more effective than 1-Hz rTMS (P = .043). Conclusions. A single session of excitatory deep brain rTMS over the right inferior frontal gyrus with H-coil significantly improves naming in right-handed chronic poststroke aphasic patients. This result is in line with the hypothesis of a positive, rather than detrimental, role of the right hemisphere in chronic aphasia due to a left-hemispheric stroke.
Introduction
Aphasia is a frequent consequence of stroke to the language-dominant hemisphere, associated with high mortality and reduced global functional recovery. 1 Although lesion size, lesion site, and the initial level of impairment are all identified factors that would determine recovery from aphasia,2,3 there is evidence that rehabilitation is also effective for improving language deficits.4-6 Both spontaneous and therapy-facilitated recovery from aphasia occurs in the subacute phases after stroke (within 6 months), 7 but significant improvements in language functions have also been described even several years after stroke. 8 The traditional, clinically based hypothesis of a crucial role of the contralateral hemisphere (typically the right hemisphere in right-handers) in aphasia recovery was initially supported by several positron emission tomography and functional magnetic resonance imaging (MRI) studies showing that language function is regained when right hemisphere regions compensate for the loss,9-14 especially in case of large contralateral lesions. 15 Further studies, however, provided conflicting results. While there is a general consensus that the recruitment of lesioned and perilesional left hemisphere regions could promote improvement in language performance,12,15-18 the role of the nondominant right hemisphere remains controversial. Increased activation of the right hemisphere shown with functional imaging is not always associated with improved recovery in aphasic patients.18,19 Actually, some studies have suggested that the recruitment of right hemisphere language regions may reflect an inefficient or maladaptive plastic changes in neural activity. 20
These conflicting results are relevant for neuromodulation studies of aphasia. With repetitive transcranial magnetic stimulation (rTMS) techniques, it is possible to noninvasively activate and modulate brain excitability.21,22 High-frequency rTMS promotes increase in cortical excitability while low-frequency rTMS produces a decrease in cortex excitability. To date, rTMS has been applied in poststroke nonfluent aphasic patients with the aim to excite the left Broca’s region or to inhibit the right homologous areas.23-25 In particular, improvements in naming have been observed after inhibitory rTMS performed with figure-of-eight coil targeting the pars triangularis, a specific portion of the right homologue language region.26-30 On the contrary, in a recent study, Vines et al 31 observed a significant improvement in speech fluency in a group of chronic nonfluent aphasic patients by applying excitatory anodal transcranial direct current stimulation (tDCS) to the posterior inferior frontal gyrus (IFG) of the right hemisphere. One of the main differences between rTMS and tDCS consists in the focality of stimulation. tDCS commonly uses an electrode montage delivering diffuse brain stimulation, while rTMS delivered with figure-of-eight coils is usually more focal. 32 Therefore, conflicting results could be related to the selectivity of Broca’s region stimulation.
In this study, we evaluated the effect of excitatory, inhibitory, and sham rTMS delivered over the right IFG on naming in a group of chronic poststroke aphasic patients. To obtain an extensive and not selective stimulation of the right homologous language areas, rTMS was applied with the H-coil. The H-coil is a novel tool designed to target deeper and larger brain regions as compared with the standard figure-of-eight coil because it is based on the principle of electric field summation that exhibits a significantly slower decay of electric field with depth.33,34 TMS is thought to activate axons in the cortex and subcortical white matter. 35 H-coil could therefore be more effective that the focal coil in stimulating a larger number of networks involved in naming.
Methods
Five participants, aged from 49 to 66 years, were selected from a consecutive series of chronic aphasic subjects. To be included, patients had to be right-handed and should have suffered for a first-ever stroke involving the left hemisphere as confirmed by computed tomography or magnetic resonance imaging. Patient data and lesion localization are reported in Table 1. For each patient, speech pathology was classified using the Aachener Aphasia Test. Moreover, the Snodgrass naming test 36 was administered twice separated by 1 week to ensure a stable baseline of patients aphasic deficits and to obtain the information necessary to structure the groups of images used for the speech task in the rTMS sessions (see section Naming Task: Stimuli Selection).
Patient Data and Lesion Localization.
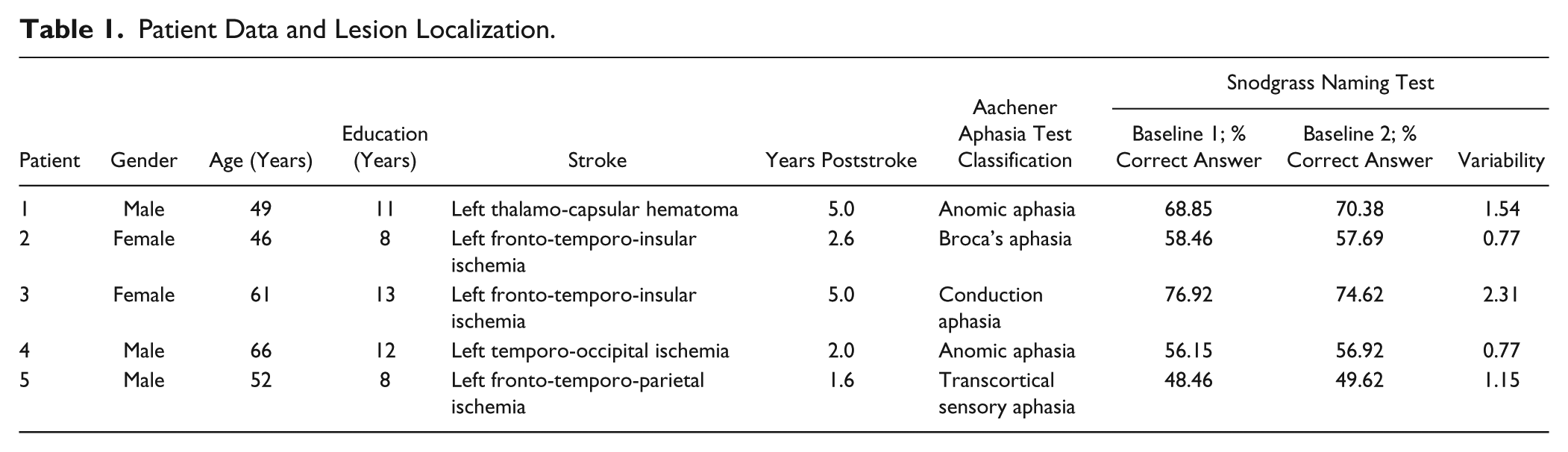
Exclusion criteria were as follows: history of other neurological disorders, use of drugs acting on central nervous system, presence of contraindications to undergo TMS, severe comprehension deficits, and instability of the naming deficit, defined as a variability >5% between the total scores of the naming test administered twice at baseline. A total of 6 consecutive patients were screened. One patient was excluded because of high variability in naming between the 2 baseline evaluations (10% of variability: 46.15% and 56.15% correct answers at the first and second baseline, respectively).
All patients gave their informed consent to participate in the study that was approved by our local ethics committee.
Experimental Design
We performed a double-blind placebo-controlled crossover study. Each patient received inhibitory, excitatory, and sham rTMS treatments over the right IFG in a random sequence. The 3 rTMS sessions were performed at the same time of the day (3-5
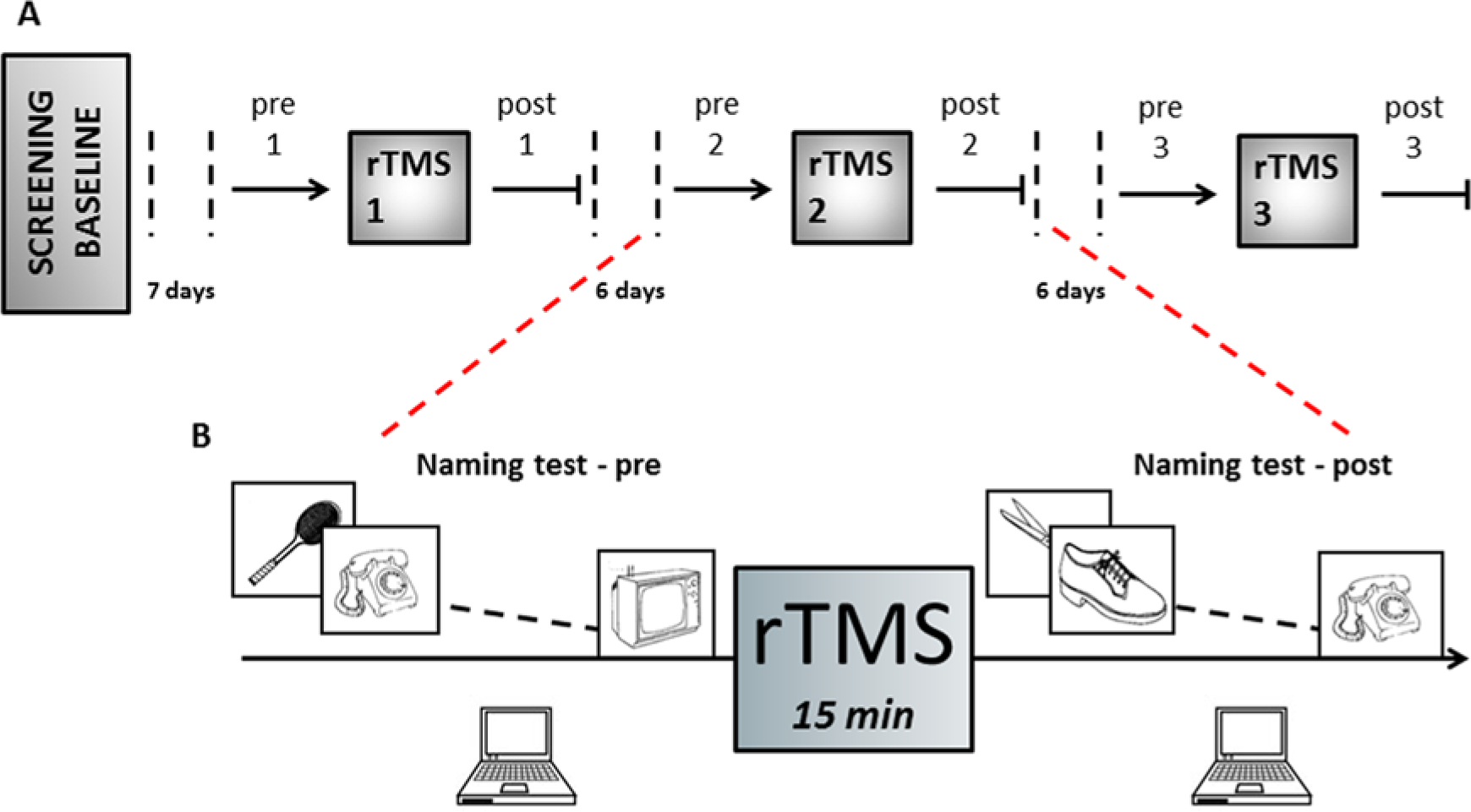
Experimental design. (A) After 1 week from basal evaluations, 3 repetitive transcranial magnetic stimulation (rTMS) sessions were performed separated by 6 days wash-out period. Patients underwent sham, 10 Hz, and 1 Hz stimulation in random sequence. (B) The picture-naming task was tested immediately before and after each rTMS.
Naming Task: Stimuli Selection
For the Snodgrass naming test, a standardized set of 260 images of objects were randomly displayed on a laptop screen using Presentation software v. 12.0 (http://www.neurobs.com). Each figure was presented for a period of 5 seconds, between 2 successive images a black screen was projected for 6 seconds. Patients were asked to name the picture just during the 5 seconds in which the image appeared on the screen. If naming occurred after this time limit the answer would be considered as wrong. Wrong responses included anomias, phonological and lexical errors, and neologisms. Since this test was administered twice at baseline (separated by 1 week), the 260 presented images were classified as follows for each patient:
Correct (C): Pictures correctly named at both evaluations
Wrong (W): Pictures incorrectly named at both evaluations
Inconsistent (I): Pictures named correctly only one time (C-W or W-C)
To reduce the possibility of learning effects, for each patient we created 3 different groups of images to be used in the 3 rTMS session. Each set was obtained selecting different pictures from the 3 categories (“correct,” “wrong,” and inconsistent”) and was balanced for a number of variables including the percentage of C, W, and I picture naming across assessment sessions (the 2 baselines), target word frequency in the Italian lexicon, and word length. 37 Each of the 3 sessions was thus based on different stimuli (with a different total number of pictures for each patient; Table 2), but the sessions were balanced in term of difficulty, of word frequency and length, and of the likelihood of a correct/wrong performance, based on the data from the 2 baseline sessions. We adopted a similar procedure for a functional MRI study of anomia training. 38 The concept of response consistency was introduced by Elizabeth Warrington and is frequently considered in anomia training experiments.39,40
Composition of Naming Task for Each Patient. a
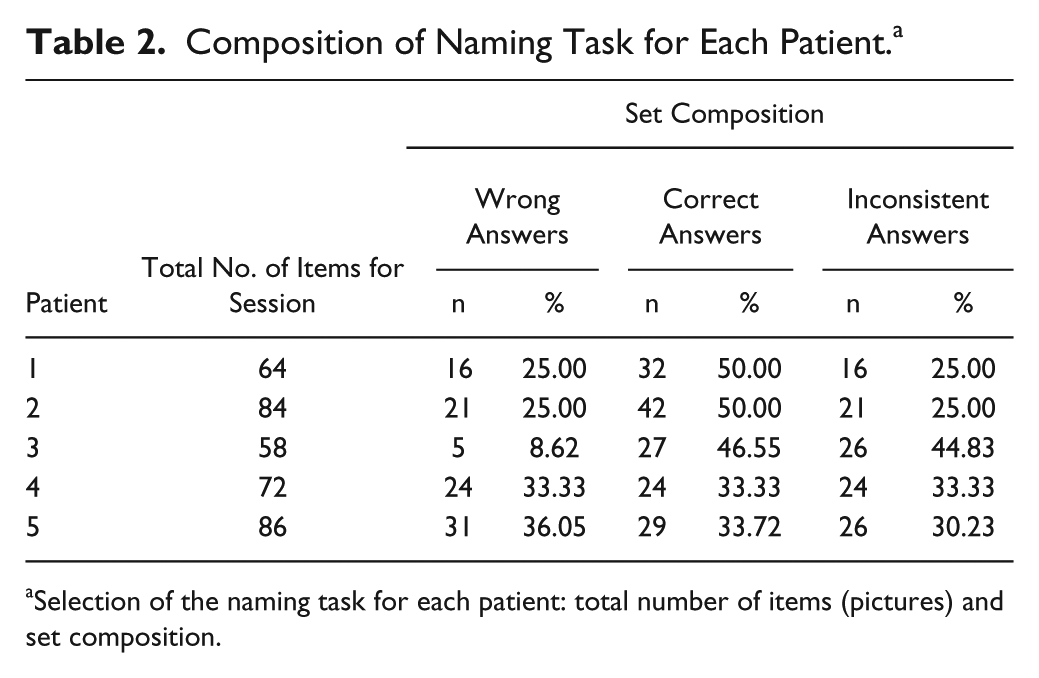
Selection of the naming task for each patient: total number of items (pictures) and set composition.
Repetitive Transcranial Magnetic Stimulation
To deliver rTMS, a Magstim Rapid 2 stimulator (Magstim Company Ltd, Whitland, Dyfed, UK) was coupled with a H-coil (Brainsway Ltd, Jerusalem, Israel). The right IFG was stimulated by moving the H-coil 5 cm anterior and 1.5 cm lateral to the right hand motor area. This displacement was obtained from calculations using Brain Voyager software (Brain Innovation BV, Maastricht, NL). Stimulation intensity was set at 100% of resting motor threshold. Resting motor threshold was defined as the minimal intensity evoking a visible twitch of left hand muscles or a motor-evoked potentials on the left abductor pollicis brevis with amplitude of 50 µV or higher in 5 out of 10 stimuli, using 1% increments of stimulator output. High-frequency stimulation consisted in 40 consecutive trains with a waiting time of 20 seconds each other; each train consisted in 20 stimulus delivered at a frequency of 10 Hz (total of 800 pulses in a 15-minute session). Low-frequency stimulation was obtained by delivering a total of 900 pulses at a frequency of 1 Hz. Sham stimulation was performed with a sham coil placed in the same Brainsway helmet designed to mimic a similar acoustic artifact and some scalp sensation evoked by the real H-coil but without inducing an effective field inside the brain. The sham stimulation was randomly delivered at high or low frequencies, reproducing in 2 patients the frequency of 10 Hz (patients 1 and 5) and in 3 patients the frequency of 1 Hz (patients 2, 3, and 4). This procedure ensured patient blindness. Examiner blindness was achieved by the involvement of different personnel in the stimulation sessions.
Statistical Analysis
The total number of items presented during the naming task differed, for each patient, between the basal and rTMS sessions, and differed also among patients during the rTMS sessions. To obtain a normalized parameter for statistical analyses, we calculated the percentage of correct answers at each evaluation ([number of correct answers/total number of items] ×100).
Because of the small sample size, nonparametric tests were used. Differences between baseline (average of the 2 measurements) and the pre-rTMS scores (obtained at each stimulation session) and, separately, between baseline and post-rTMS scores were investigated with nonparametric repeated-measures analysis of variance (ANOVA; Friedman test). If a significant main effect was found, Wilcoxon tests were performed for post hoc comparisons. To evaluate a possible daily variability, the pre-rTMS versus post-rTMS scores (percentage of correct answers) obtained at each stimulation session were compared using a Wilcoxon test.
Data were analyzed with SPSS 13.0 (SPSS, Chicago, IL). Significance level was set at P≤ .05.
Results
A significant carry-over effect between rTMS sessions was excluded by the ANOVA analysis performed between baseline and pre-rTMS sessions scores (P = .8). Interestingly, the Friedman ANOVA analysis demonstrated a significant difference in naming performance between baseline and post-rTMS evaluations (P = .041). The post hoc analysis showed that only the real 10-Hz rTMS was associated with a significant improvement in the percentage of correct answers in comparison with baseline (P = .042) and in comparison with performance after the inhibitory 1-Hz stimulation (P = .043). The within-session pre-rTMS versus post-rTMS comparisons confirmed that the percentage of correct naming significantly improved only with high-frequency stimulation (P = .043; Figure 2). We also examined the ability to name the same individual stimuli and we observed that picture correctly named before the high-frequency rTMS were always correctly renamed after the stimulation for all patients.
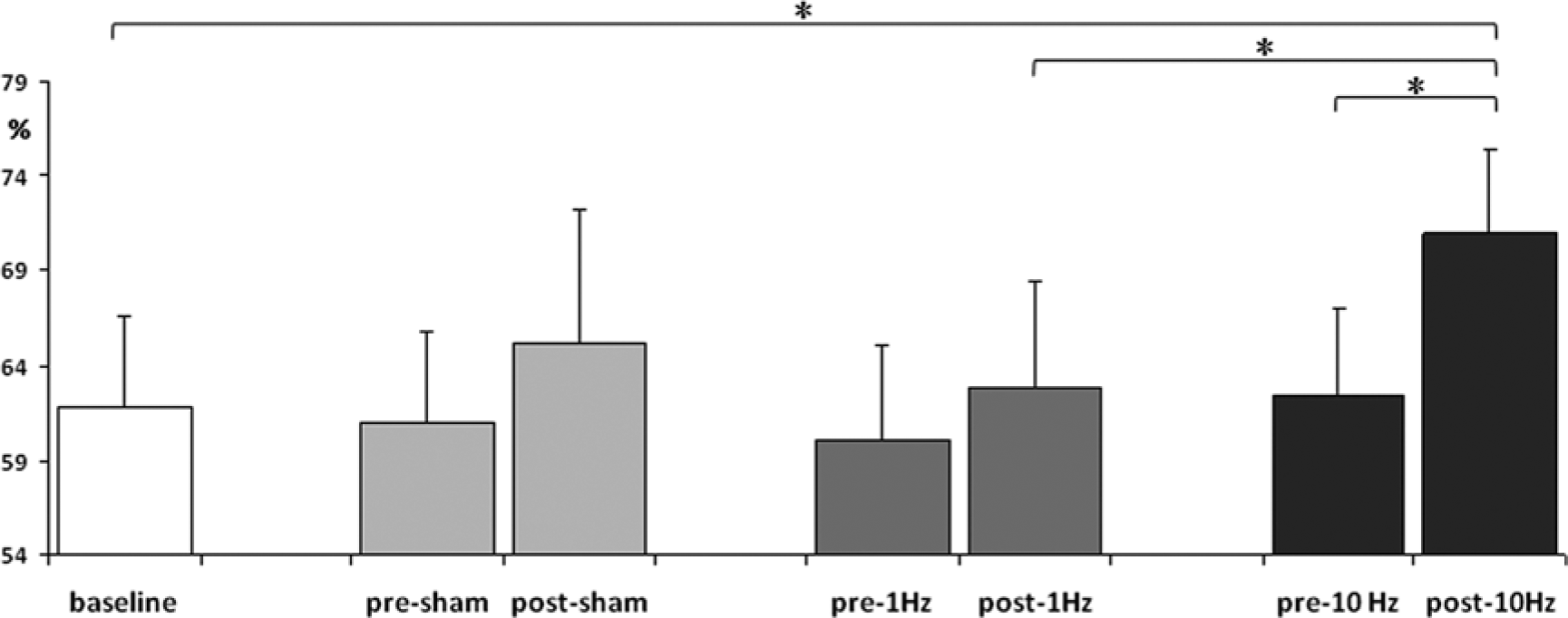
Effect on naming performance (percentage of correct number) of sham, excitatory (10 Hz), and inhibitory (1 Hz) repetitive transcranial magnetic stimulation (rTMS) delivered over the right inferior frontal gyrus (IFG). After 10-Hz stimulation, significant improvement in naming performance was observed in comparison with both baseline and pre–10-Hz rTMS. Moreover, 10-Hz rTMS was significantly more effective than the 1-Hz rTMS (*P< .05).
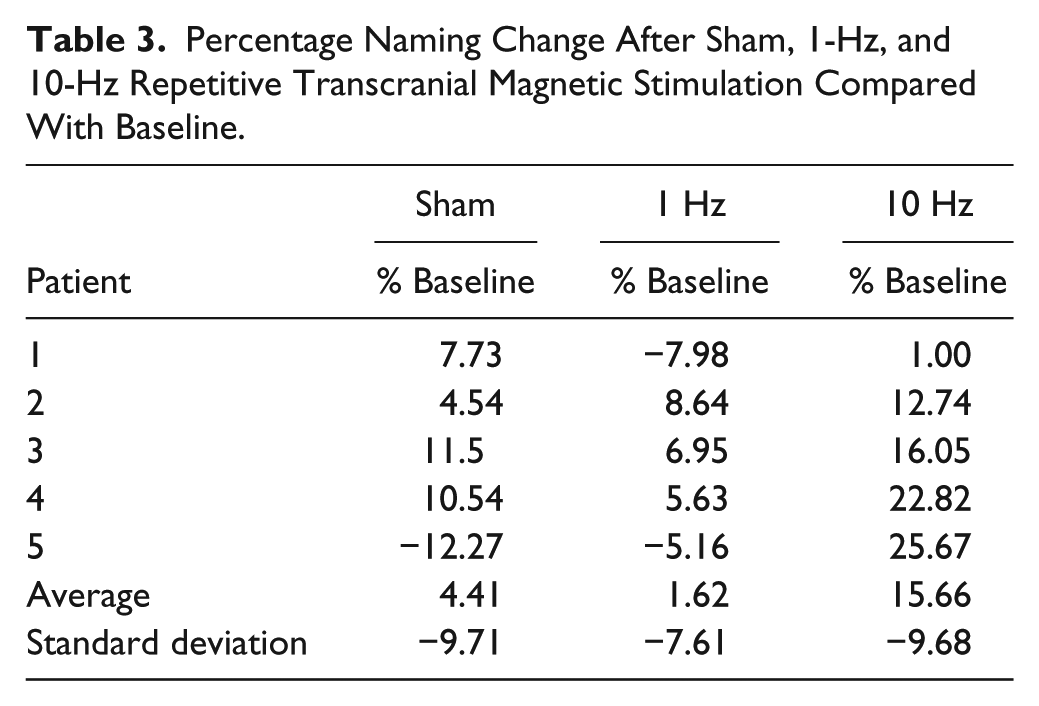
Table 3 shows the variations from baseline of the naming scores after the 3 rTMS sessions (percentage of improvement or worsening compared with baseline). We can observe that the “best responder” to the excitatory stimulation (patient 5) was the patient with a large lesion involving the cortical frontal regions and more severe naming deficits. In contrast, patient 1, suffering from a subcortical hematoma milder deficit, did not show a meaningful improvement after the 10-Hz rTMS.
Percentage Naming Change After Sham, 1-Hz, and 10-Hz Repetitive Transcranial Magnetic Stimulation Compared With Baseline.
Discussion
The role of the right hemisphere (RH) in aphasia recovery, and more specifically in the reorganization of language neural networks after stroke, is unclear. In almost all the previous studies in poststroke aphasic patients, low-frequency focal rTMS was delivered over the right PTr (the anterior portion of the homologue Broca’s area) inducing improvements in pictures naming.26-29,41,42 The application of inhibitory rTMS over the contralesional hemisphere is based on the theory that the RH activation after left hemisphere (LH) stroke conditioning aphasia would represent an inefficient or maladaptive plastic reorganization of language circuits. 20 TMS studies performed after unilateral stroke involving the motor system demonstrated an acute hyperexcitability of the contralesional motor cortex.43,44 In accordance with the theory of interhemispheric competition, this overactivation of the unlesioned hemisphere was associated with poor motor function recovery.43,45 It has thus been hypothesized that in aphasic patients a hyperactivation of the RH may result in increased interhemispheric inhibitory influences on the LH perisylvian areas. 24 Low-frequency rTMS over the right homologue language areas has been therefore finalized to suppress maladaptive RH activations and promote the activation of LH perilesional and perisylvian areas. 46 The latter hypothesis stands in contrast with evidences suggesting that the RH could play a beneficial role in language recovery in aphasic patients. Turkeltaub et al 47 in 2011 demonstrated that the pattern of RH activation in aphasic patients is largely consistent across studies and mirrors that of the LH. These data suggest that RH might possess the functional architecture needed to assure language operations after LH injury. 47 Moreover, positron emission tomography and functional MRI studies indicate that language function can be restored when right hemisphere regions compensate for the loss.9-14 It is possible that the relative contribution of the 2 hemispheres in language recovery may be modulated by factors such as lesion size 48 and time post-onset of aphasia. 49 Also, in physiological conditions, the mutual inhibition between homologous areas is not constant. For example, during movement preparation of the nondominant hand, the dominant hemisphere is facilitated. 50
To our knowledge, this study is the first to report the effect of both low- and high-frequency deep rTMS applied over the right IFG in patients affected by chronic poststroke aphasia. We found that only the excitatory rTMS induced significant improvement in naming in comparison with baseline and with pre-rTMS evaluations, and that this effect was significantly greater than that observed with inhibitory rTMS. As expected with single session stimulation, in our patients this effect was transitory, but likely not mediated by day to day variability. Indeed, no differences between the baseline versus the pre-rTMS (at each stimulation session) performance measurements were observed. The naming task was performed before each stimulation session not only to assess the effects of rTMS treatments but also to preactivate the linguistic circuits involved in naming. Indeed, there is evidence that the efficacy of brain stimulation can be increased if combined to cognitive rehabilitation.51-56 In our patients, the consistent improvement in naming could be related to a combined effect of 10-Hz rTMS over the right IFG with the language task. Likewise, the improvement observed with sham stimulation, although not significant, might represent a placebo effect, possibly reflecting learning or training effect mediated by the preceding naming. Our results underline the importance to include a control group in this kind of studies in order to evaluate the effective contribution of the real stimulation when rTMS treatment is associated with language therapy. Most of rTMS studies performed with poststroke aphasic patients were not placebo controlled.26,28,29,55,57
An interesting point to discuss regarding the different effect of excitatory rTMS observed among our patients is possibly in relation to lesion localization and the severity of aphasic deficit. Indeed, the only patient who did not improve with 10-Hz rTMS over the right IFG (patient 1) had a subcortical stroke sparing the cortical regions. On the opposite, the best responder to the excitatory stimulation (patient 5) was the patient presenting with the most severe naming deficit at baseline and with a large lesion involving the cortical frontal regions. Functional MRI studies show that patients with left inferior frontal cortex lesions recruit functionally homologous right IFG areas (mostly in the pars opercularis and pars orbitalis) more reliably than those without.38,47 Other studies provide evidence for a bihemispheric role or even a role for the RH in language recovery only following insults involving large parts of the LH.18,19,58 It is well known that the effects of rTMS are stronger and longer lasting if several sessions are applied. 59 A single session of rTMS was probably just sufficient to transiently modulate the activity of RH language networks already functional in patients with an extended damage of the contralateral hemisphere. Moreover, as the effect of rTMS is not limited to directly targeted brain region, 60 it is possible that the excitatory stimulation delivered over the right IFG was able to enhance the activity of bihemispheric functional networks in patients with bilateral compensatory reorganization of speech circuits.
In contrast with the results obtained with the 10-Hz stimulation, the improvement observed after 1-Hz rTMS was similar if not smaller than that observed with sham stimulation (Figure 2). In a recent study 10 sessions of 1-Hz rTMS over the right pars triangularis in combination with speech therapy significantly improved naming in subacute poststroke aphasic patients. 30 The latter study, however, cannot be directly compared with the present findings, given the differences in number of sessions, stroke stage, and above all, in the target and the paradigm of stimulation. More specifically, in the present study the application of the H-coil, a particular type of coil designed to target deeper and larger brains regions,33,34,61,62 may have induced specific effects. In fact, the standard figure-of-eight coil delivers a relatively focal and superficial stimulation, mainly confined to the cortical regions. In the majority of studies reporting speech improvement after inhibitory rTMS of the right Broca’s homologous in subacute and in chronic stroke patients, the stimulation was performed on the pars triangularis, a specific portion of the right homologue speech region.26-29,30 Converging evidence from neurophysiological and MRI suggests that different portions of the right perisylvian region may induce different effects on residual speech in relation to their respective interactions. Naeser et al 63 reported that a session of 1-Hz inhibitory rTMS applied on the right PTr induces transient improvement in accuracy and reduction in reaction in a picture naming. Conversely, 1-Hz rTMS to the right POp (posterior portion of the right homologue of Broca’s area) was associated with transient decrease in picture-naming accuracy and increase in reaction time. The authors hypothesized that the PTr could modulate the activity of the POp through inhibitory u-fiber connections. The suppression of PTr activity could therefore allow a better modulation of the POp on other right and left temporo-parietal regions important for naming. In agreement, MRI studies showed differences in white matter pathways between the PTr, the Pop, and the posterior language zone in the LH as well as in the RH indicating a different integration of the 2 Broca’s areas in neuronal speech networks involving both hemispheres. 64 In this study, the use of the H-coil allowed us to deliver a diffuse and nonselective stimulation of the right homologue Broca’s areas and of its connections. We observed that the overall facilitatory stimulation of this region was associated with naming improvement. Our results are in agreement with data obtained by Vines et al 31 with anodal excitatory tDCS applied to the right posterior IFG in a group of chronic nonfluent aphasic patients. It is possible that a nonfocal stimulation, independently from the technique, may induce facilitatory effects. However, contrary to tDCS, which delivers a wider but superficial electric field, H-coil has been designed to stimulate larger and also deeper brain regions. It is therefore possible that excitatory rTMS delivered with H-coil induced facilitation of intra- and interhemispheric language networks by activating axons in the cortex and subcortical white matter tracts as well. 35
In conclusion, this study provides evidence that high-frequency rTMS delivered with the H-coil over the right IFG enhances naming in patients with chronic poststroke aphasic deficits. The main limitations of our study are the small sample size and the lack of neurophysiological or neuroimaging outcome measures. Moreover, the naming test and the matching procedure we adopted did not allow to look for semantic category effects (eg, living vs nonliving) and the response time was not evaluated. Further studies are needed to evaluate the application of the H-coil rTMS with therapeutic purposes in order to induce long-lasting effects following multiple stimulation sessions.
Footnotes
Authors’ Note
R. Chieffo participated in this study as partial fulfillment of her PhD in Molecular Medicine, Program in Experimental Neurology, San Raffaele University, Milan, Italy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A. Zangen is a key inventor of deep TMS H-coils and has financial interest in Brainsway Ltd. The other authors declare no conflicts of interest related to the present study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Joint Italian-Israeli Laboratory San Raffaele-Weizmann (Italian Ministry of Foreign Affairs).
