Abstract
Keywords
Introduction
Stroke remains one of the leading causes of disability worldwide. 1 Because of an aging population, the global, absolute number of affected individuals is increasing despite falling incidence. 2
Arm paresis is one of the most frequent impairments poststroke. Paresis is defined as a deficit in strength and motor control (the ability to perform coordinated, accurate and goal-directed movements). 3 It has been reported that 48% to 77% of patients have arm paresis in the acute stroke phase4-6; of these, only 12% to 34% achieve full functional recovery at 6 months poststroke.7,8 Arm paresis is a major obstacle in regaining independence in activities of daily living (ADL), 9 especially in bimanual activities, and an almost complete functional recovery is required to routinely involve the paretic arm in ADL performance. 10 Moreover, studies show that even the ability to perform ADL with the nonaffected arm is lower in adults with chronic stroke compared with adults without stroke. 11
The largest improvement in arm functioning occurs in the first 4 to 10 weeks poststroke12-16; this is assumed to be predominantly based on spontaneous neurologic recovery. The recovery of hand dexterity (motor skills as reaching, grasping, moving, and releasing objects) appears to be determined already in the first 4 weeks poststroke, 7 implying that this is a critical period for arm recovery. However, there is still limited knowledge about the effect of therapeutic interventions in the early period poststroke when the rate of spontaneous neurologic recovery is greatest and most rehabilitation services are offered.12,17 The arm recovery continues for at least 3 to 6 months poststroke4,14,16; subsequently, only 5% to 10% of the patients demonstrate further improvements. 14
The potential for electrical somatosensory stimulation (ESS) to facilitate the arm recovery has been addressed in recent studies. In healthy persons, low-intensity electrical stimulation with no or minimal motor response applied to the peripheral hand nerves,18-20 forearm muscles, 21 and the whole hand 22 increases the cortical excitability of the brain areas that control the stimulated body parts, and this effect outlasts the stimulation period.18,21,22 A single 2-hour ESS session applied to the peripheral hand nerves transiently improves pinch force, movement kinematics, and motor skills in acute, subacute, and mostly chronic stroke patients.23-28 Of these studies, only 1 used ESS in conjunction with arm training. 26 Seemingly, the use of multiple ESS sessions followed by arm training facilitates motor skills in subacute 29 and chronic patients, 30 and the improvements appear to be long lasting in chronic patients, 30 but the evidence is conflicting.31,32 To our knowledge, the effect of multiple ESS sessions in addition to arm training and initiated in the acute stroke phase was not yet investigated in a randomized controlled trial (RCT) design. 33 Therefore, it was justified to test the effectiveness of multiple ESS sessions in addition to arm training on the recovery of the paretic arm when the intervention was delivered during the first 4 weeks poststroke. ESS has no known side effects, can easily be incorporated into clinical practice, and the electrical device is relatively inexpensive.
This trial was designed to investigate the hypothesis that the application of ESS to the affected arm immediately prior to arm training during early hospitalization after stroke is superior to arm training alone regarding the recovery of hand dexterity as measured by the Box and Block Test (BBT, primary outcome measure) at 6 months poststroke. Secondarily, we expected that improvements in dexterity would be accompanied by reductions in global disability, and motor and sensory impairments. Thus, we planned to address the following research questions: (
Methods
A detailed description of the trial protocol has been published elsewhere. 34
Trial Design
The trial was conducted as an RCT with blinded outcome adjudication and 2 parallel arms: an intervention and a control group. The trial was notified at ClinicalTrials.gov (NCT02250365).
Trial Setting and Participants
The trial was conducted in the stroke rehabilitation unit of Bispebjerg Hospital, Copenhagen, Denmark. After a few days in the acute stroke unit the patients were transferred to the stroke rehabilitation unit where they received early inpatient rehabilitation while they still needed constant medical care. Subsequently, the patients were discharged to their homes with/without referral to outpatient rehabilitation services in the community, inpatient rehabilitation centers in the community, or nursing homes, depending on their recovery potential and needed ADL assistance. All these rehabilitation services are covered by the Danish national health insurance.
The eligibility criteria were: (
Data on following potential demographic and clinical confounders were collected from medical records: (
Participants provided written informed consent before enrolment. Ethical approval was obtained from the Capital Region of Denmark’s Committee on Health Research Ethics (H-4-2014-012) and by the Danish Data Protection Agency (2012-58-0004).
Intervention
The ESS was initiated within the first 7 days poststroke and administered by trained health care personnel according to the published protocol, Additional file 1.
34
Two sets of external electrodes were positioned at the wrist, elbow, and shoulder level. The electrode at the wrist level covered all three peripheral hand nerves; the stimulation at the elbow and shoulder level was of cutaneous art. The intervention group received suprasensory ESS delivered in continuous mode (pulse width = 250 µs, frequency = 10 Hz). Suprasensory ESS was defined as the highest current amplitude that elicits paresthesia without any discomfort, pain, or visible muscle twitches. Because suprasensory ESS can be perceived and the participants could communicate to each other during hospitalization, using a completely inactive placebo ESS gave rise to concerns about a possible high drop-out in the control group. Consequently, we designed a placebo ESS with a low treatment dose that we considered unlikely to induce any training effects. The placebo ESS consisted of suprasensory ESS delivered in intermittent mode (active stimulation intervals of 3 seconds delivered in loops of 2.5 minutes, pulse width = 250 µs, frequency = 10 Hz), and the control group received a total dose corresponding to 2% of the amount of active ESS delivered to the intervention group per ESS session (see Supplementary Material, available in the online version of the article). The groups received 1 hour of daily high-dose/low-dose ESS from Monday to Sunday throughout the hospital stay, but no longer than 4 weeks poststroke. The stimulation level was determined individually for each participant at the beginning of each ESS session. To avoid adaptation to ESS in the high-dose group, the stimulation level was adjusted, if necessary, after the first 30 minutes, whereas the low-dose group got a short visit from the ESS staff to ensure the same amount of attention. Patients were neither requested to focus on the stimulation nor allowed to participate in other training sessions (eg, speech and language therapy, gait training) during ESS sessions. The high-dose/low-dose ESS was followed by minimum 15-minute arm training; this intervention component was considered a part of the usual inpatient rehabilitation. The arm training consisted of active, repetitive, task-oriented practice and was provided within 30 minutes after cessation of ESS when we expected the brain excitability to be increased.18,20 If the participants were unable to perform volitional arm movements, the treating therapist decided which training methods to be employed. Except from an inspirational task-oriented exercise bank,
34
the arm training was not further standardized. The high-dose/low-dose ESS was provided in addition to (
Outcomes
Our primary outcome measure was BBT at 6 months poststroke. Normative data are available for the healthy adult population 42 and the instrument has been validated for use in stroke patients. 43 To our knowledge, minimal clinically important difference (MCID) for BBT has not been established yet; the minimal detectable change (MDC) is 5.5 blocks/min. 43
The secondary outcomes measures were: (
The outcome measures were assessed at 3 time points by EG: (
Sample Size
Our pretrial sample size analysis showed that 37 patients were required for detecting a within-group improvement corresponding to the reported MDC = 5.5 blocks/min 43 on BBT with a 2-sided significance level = 5% and power = 80%. After adjusting for a case fatality rate = 8% and an estimated drop-out = 20% for other reasons, 51 participants were needed in each group, which resulted in a total sample size of 102 participants.
Randomization
Participants were allocated sequentially to the low-dose or the high-dose group using a randomization list constructed by block randomization with variable block size. Randomization was stratified by: (
Blinding
Complete blinding of participants was not feasible as the participants could perceive the stimulation and figure out their group allocation. However, the participants were kept unaware of which stimulation mode was the intervention and which one functioned as control. The ESS staff was not blinded to the group allocation. The therapists providing usual rehabilitation, the staff involved in medical care, the outcome assessor (EG) and data analysts (VS, EG) were kept blinded to group allocation until all analyses were completed.
Statistical Methods
The demographic and clinical characteristics at baseline were compared between the groups with Fisher’s exact test for categorical variables and with Wilcoxon test for continuous variables as they did not follow a normal distribution. The development of primary and secondary outcome variables was analyzed in longitudinal models over the stroke recovery trajectory (baseline, postintervention, and follow-up), and the differences of the outcomes between the 2 groups at each of the trial time points were analyzed in multivariable linear regression models. Analyses were adjusted for the stratification variables (sex and the ability to perform voluntary finger extension at baseline). An informal inspection of the summary statistics of the missing participants showed some general trends: they were mostly women, had the dominant hand affected and a prestroke mRS >0. To adjust for possible differential dropout, the available data at each of the trial time points was weighted with the inverse probability of being observed; these probabilities were estimated from logistic regression models with sex, affected dominant hand, prestroke mRS and outcomes at previous trial points as covariates. To account for this weighting and for repeated observations on the same participant, generalized estimating equations (GEE) methods were used to adjust the variance of the parameter estimates. For the hand grip strength and pinch strength, the maximum value of the 3 to 5 measurements done per assessment was entered in the data set. For the PTT, 3 to 5 measurements per assessment were undertaken, and the value entered in the data set was the mean of the remaining measurements after the lowest and the highest values were discarded. BBT was performed once per assessment. Because baseline data on mRS were not collected, we assigned a baseline mRS of (4 + 5)/2 = 4.5 points to all the trial participants. This estimation is supported by the assumption that hospitalized stroke patients have a mRS of 4 or 5 because of their need for constant medical care. Analyses were performed with SAS version 9.4. The statistical significance level was set to 1% to guard against false detection of effects because of multiple comparisons. For differences in our primary outcome, the significance level was of 5%.
Results
Figure 1 details the participant flow through the trial. All patients consecutively admitted to the stroke rehabilitation unit and diagnosed with IS or HS from October 2014 to March 2017 were screened for a newly developed arm paresis, except a total of 6 months (holidays, recruitment/training of new trial staff) (n = 1214). More than 537 stroke patients were identified (the screening log for the initial 5 months was incomplete). Of these, 102 patients were randomized; 49 to the low-dose group and 53 to the high-dose group. Two patients were assigned to the low-dose group although it was not possible to initiate the ESS within 7 days poststroke; 1 patient was assigned to the high-dose group despite a prestroke mRS = 5 and inability to initiate the ESS within 7 days poststroke. All randomized participants received the assigned intervention. All baseline assessments were conducted during the first 7 days poststroke. Postintervention data were collected approximately 1 day after the last ESS session and 19 to 20 days after stroke onset from 45 participants in the low-dose group and from all 53 participants in the high-dose group. Follow-up assessments were collected approximately 3.5 to 5 days after the actual date for 6 months poststroke from 42 participants in the low-dose group, and 46 participants in the high-dose group (Table 1). Data from all randomized participants were entered in the intention-to-treat analysis.
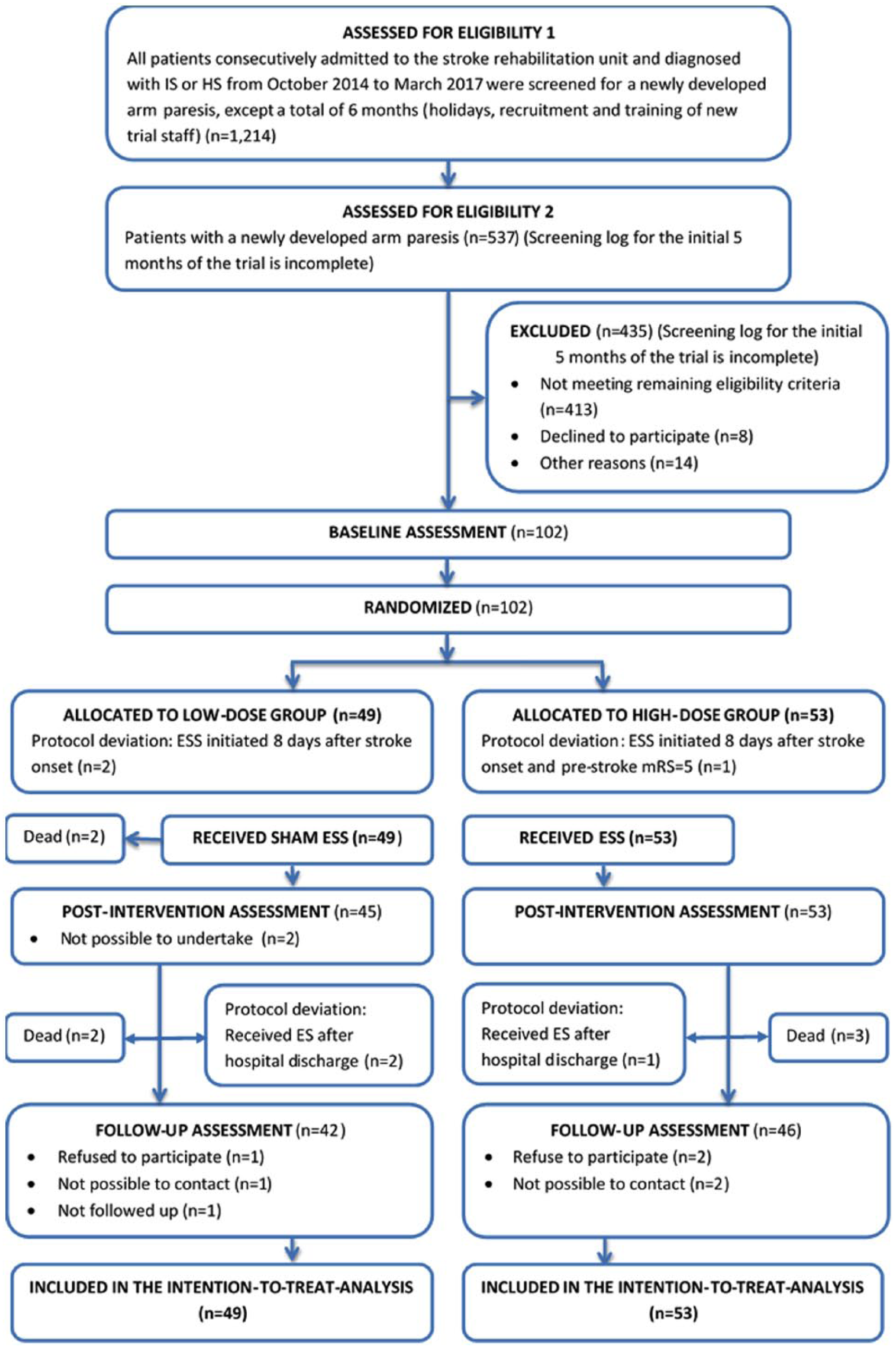
The flow of participants through the trial.
Description of the Achieved ESS Intervention, Usual Rehabilitation (Including Arm Training), and Actual Time Points for Postintervention and Follow-up Assessments.
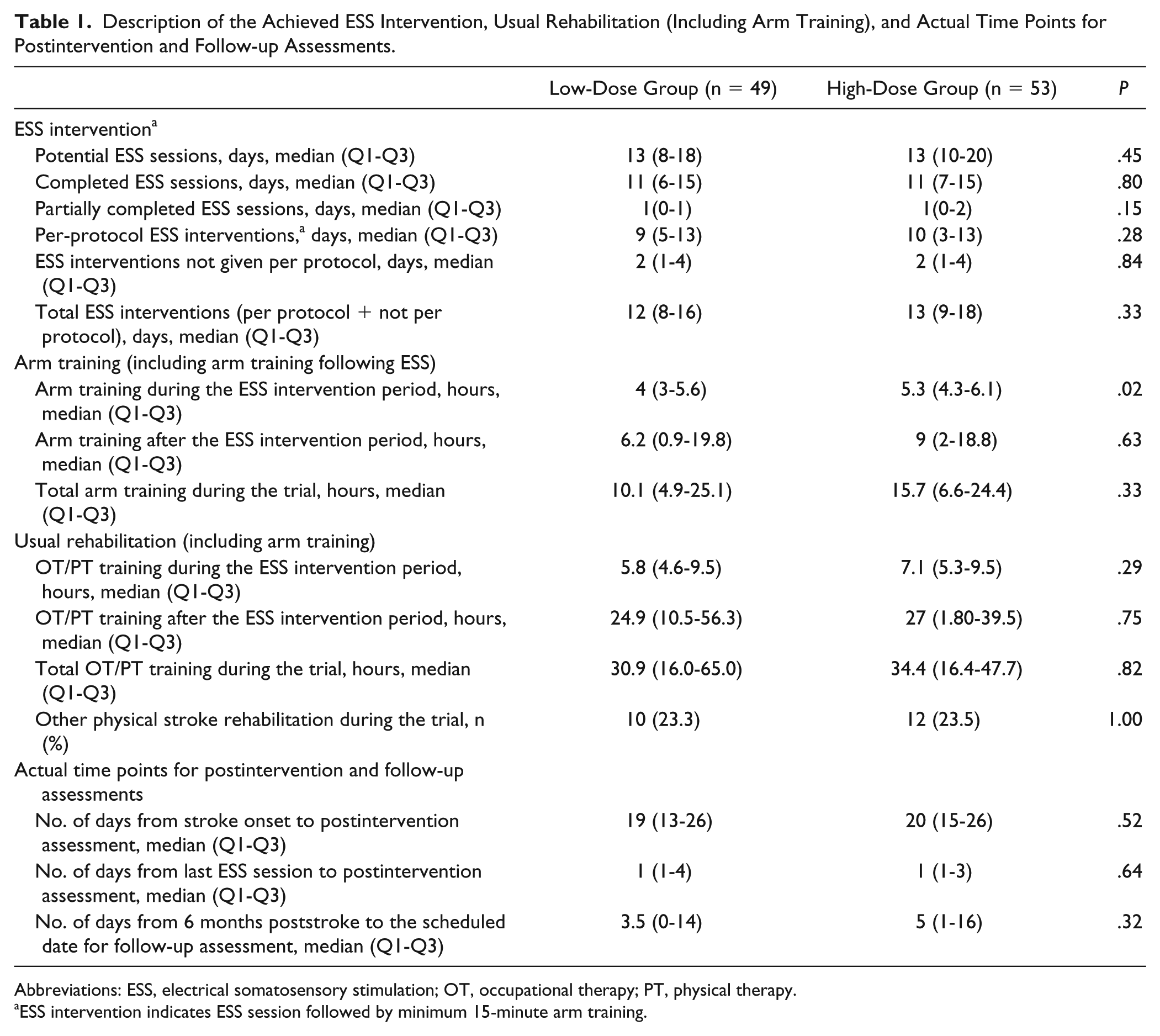
Abbreviations: ESS, electrical somatosensory stimulation; OT, occupational therapy; PT, physical therapy.
ESS intervention indicates ESS session followed by minimum 15-minute arm training.
The 2 groups were comparable with regard to demographic and clinical characteristics at baseline (Table 2). Participants exhibited a mean FMA-UE-AD of 33.2 to 33.6 points at baseline, indicating a moderate arm impairment (Table 3). 52
Baseline Demographic and Clinical Characteristics.
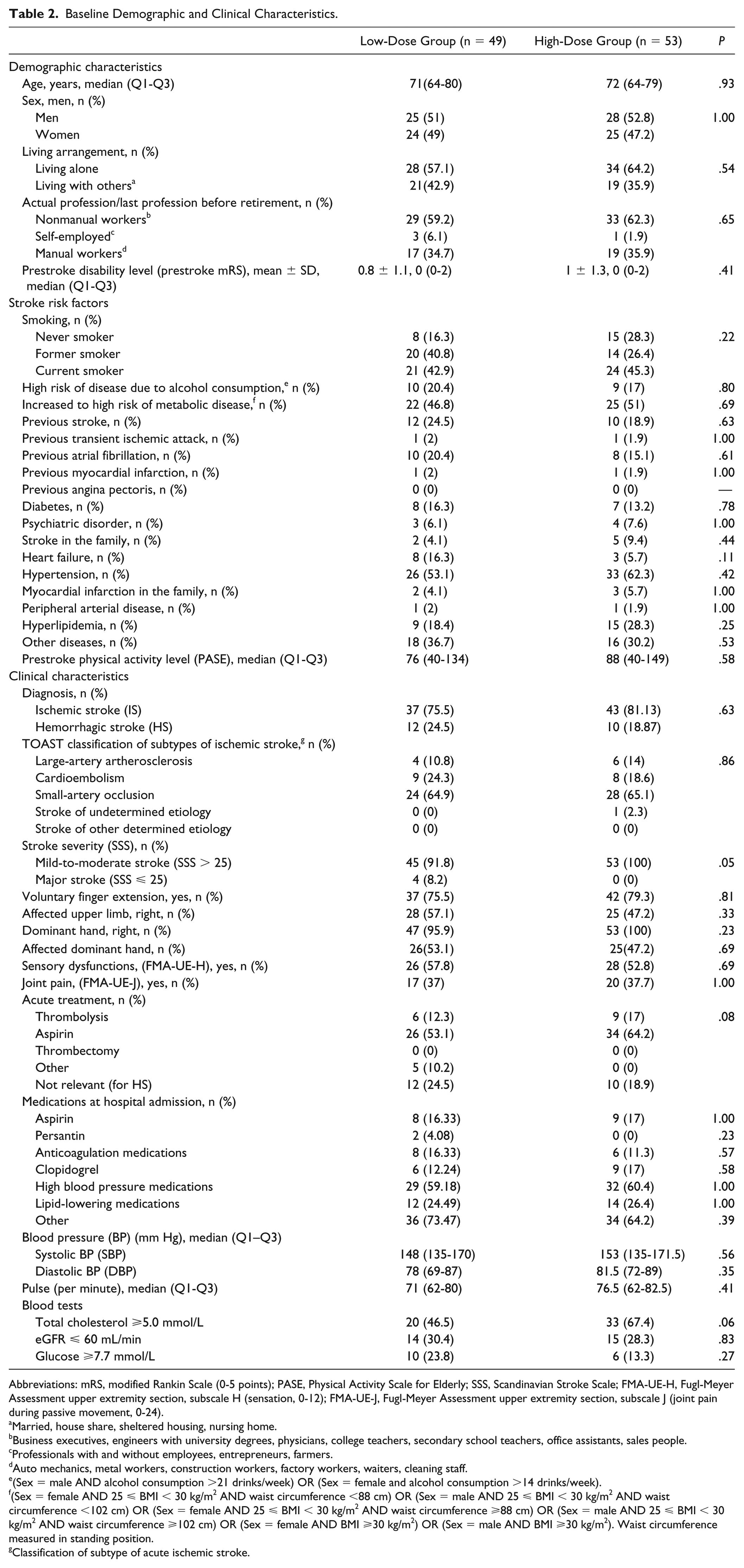
Abbreviations: mRS, modified Rankin Scale (0-5 points); PASE, Physical Activity Scale for Elderly; SSS, Scandinavian Stroke Scale; FMA-UE-H, Fugl-Meyer Assessment upper extremity section, subscale H (sensation, 0-12); FMA-UE-J, Fugl-Meyer Assessment upper extremity section, subscale J (joint pain during passive movement, 0-24).
Married, house share, sheltered housing, nursing home.
Business executives, engineers with university degrees, physicians, college teachers, secondary school teachers, office assistants, sales people.
Professionals with and without employees, entrepreneurs, farmers.
Auto mechanics, metal workers, construction workers, factory workers, waiters, cleaning staff.
(Sex = male AND alcohol consumption >21 drinks/week) OR (Sex = female and alcohol consumption >14 drinks/week).
(Sex = female AND 25 ⩽ BMI < 30 kg/m2 AND waist circumference <88 cm) OR (Sex = male AND 25 ⩽ BMI < 30 kg/m2 AND waist circumference <102 cm) OR (Sex = female AND 25 ⩽ BMI < 30 kg/m2 AND waist circumference ⩾88 cm) OR (Sex = male AND 25 ⩽ BMI < 30 kg/m2 AND waist circumference ⩾102 cm) OR (Sex = female AND BMI ⩾30 kg/m2) OR (Sex = male AND BMI ⩾30 kg/m2). Waist circumference measured in standing position.
Classification of subtype of acute ischemic stroke.
Between-Group Differences.
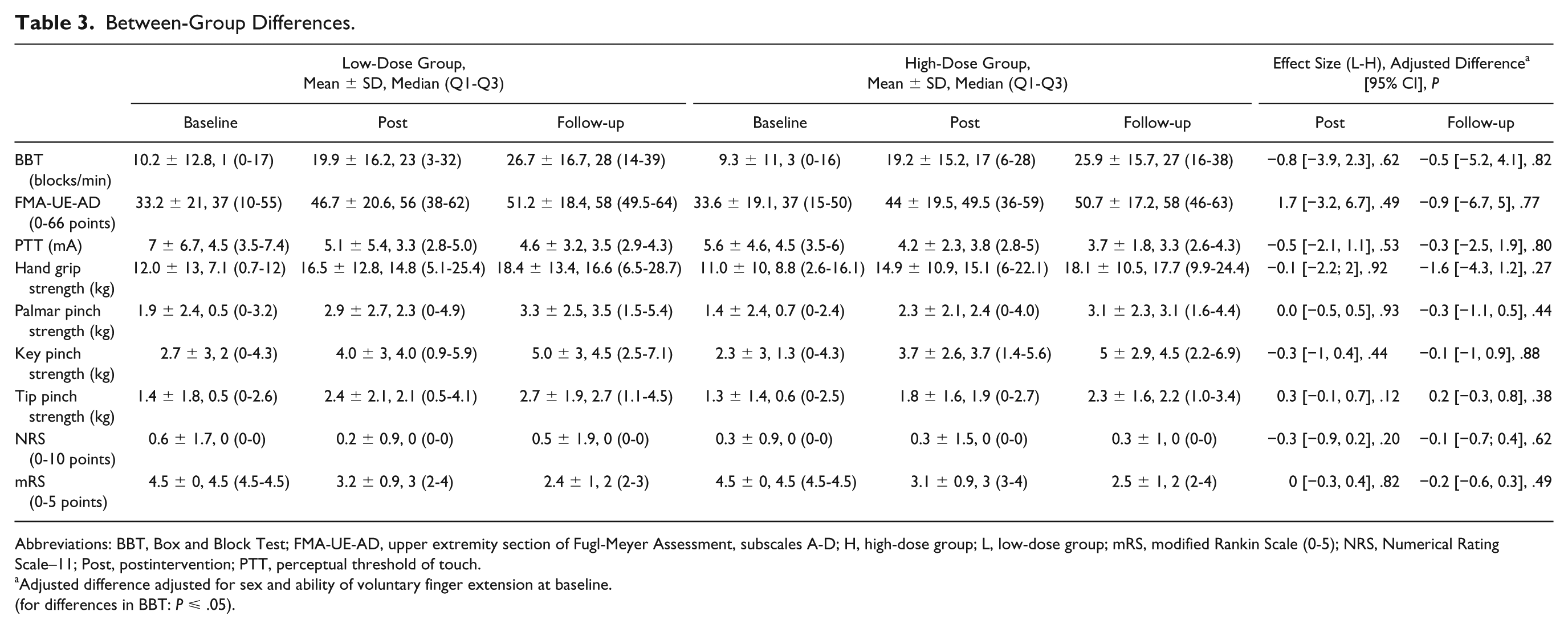
Abbreviations: BBT, Box and Block Test; FMA-UE-AD, upper extremity section of Fugl-Meyer Assessment, subscales A-D; H, high-dose group; L, low-dose group; mRS, modified Rankin Scale (0-5); NRS, Numerical Rating Scale–11; Post, postintervention; PTT, perceptual threshold of touch.
Adjusted difference adjusted for sex and ability of voluntary finger extension at baseline.
(for differences in BBT:
Our analysis showed no statistically significant between-group differences for any outcome measures at any time points (Table 3). However, there were positive, statistically significant within-group effects in both trial groups on all outcome measures, except for pain; improvements in FMA-UE-AD and mRS were moreover ⩾MCID at all time points, and for the hand grip strength at follow-up (Table 4).
Within-Group Differences.
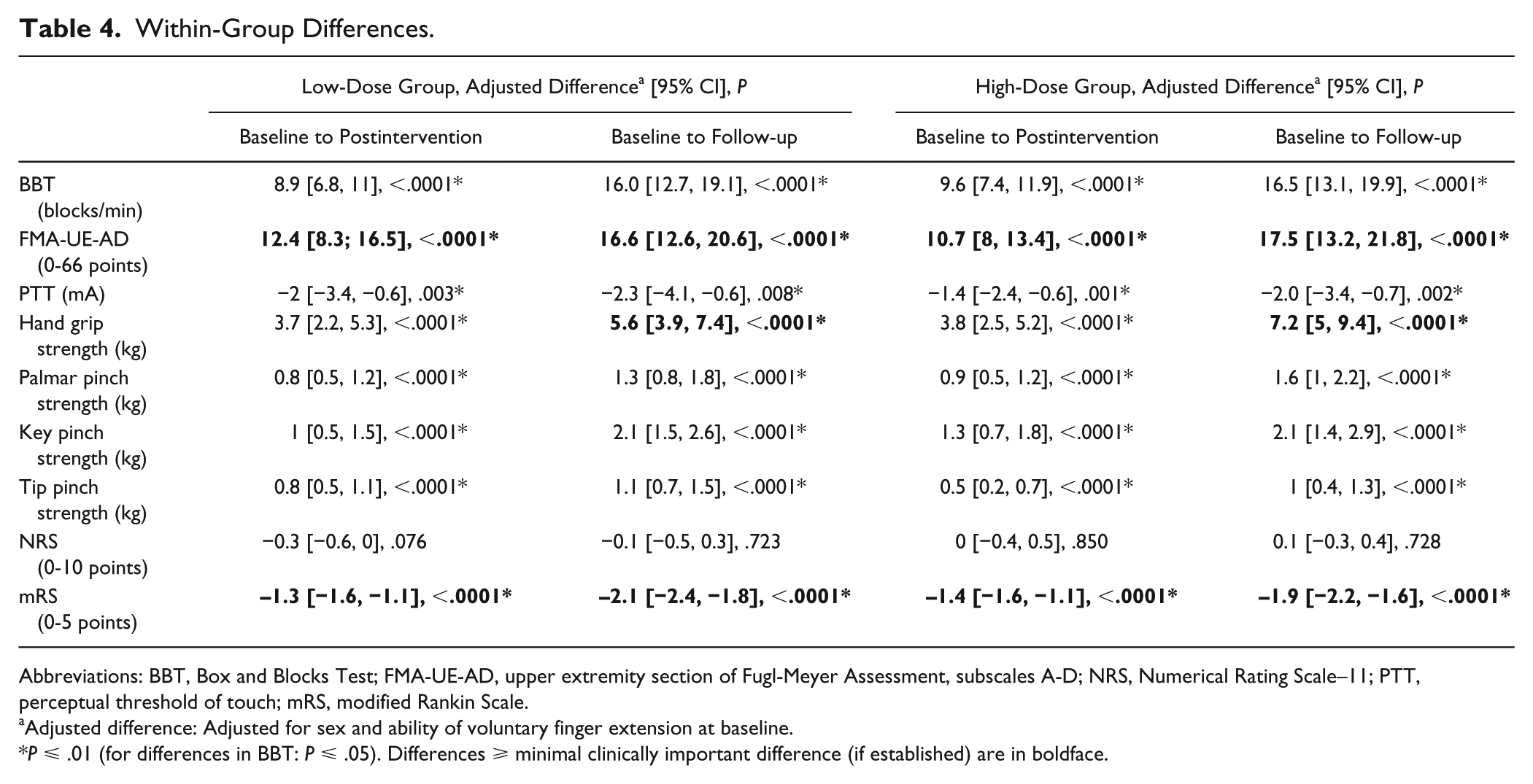
Abbreviations: BBT, Box and Blocks Test; FMA-UE-AD, upper extremity section of Fugl-Meyer Assessment, subscales A-D; NRS, Numerical Rating Scale–11; PTT, perceptual threshold of touch; mRS, modified Rankin Scale.
Adjusted difference: Adjusted for sex and ability of voluntary finger extension at baseline.
During their hospital stay (17-18 days) (Table 5), the participants received 12 to 13 ESS interventions; of these, 9 to 10 were given per protocol. The amount of arm training during the ESS intervention period was of 4 to 5.3 hours and the total amount of arm training during the whole trial was 10.1 to 15.7 hours. During the ESS intervention period, the amount of additional OT/PT training was 5.8 to 7.1 hours and the total amount of additional OT/PT training during the whole trial was 30.9 to 34.4 hours. Around 23% of the participants in both groups also received other stroke rehabilitation, mostly consisting of individually purchased PT services. Table 1 presents detailed information on the delivered ESS intervention and usual rehabilitation (including arm training), showing similarity between the groups with regard to these parameters.
Additional Results.
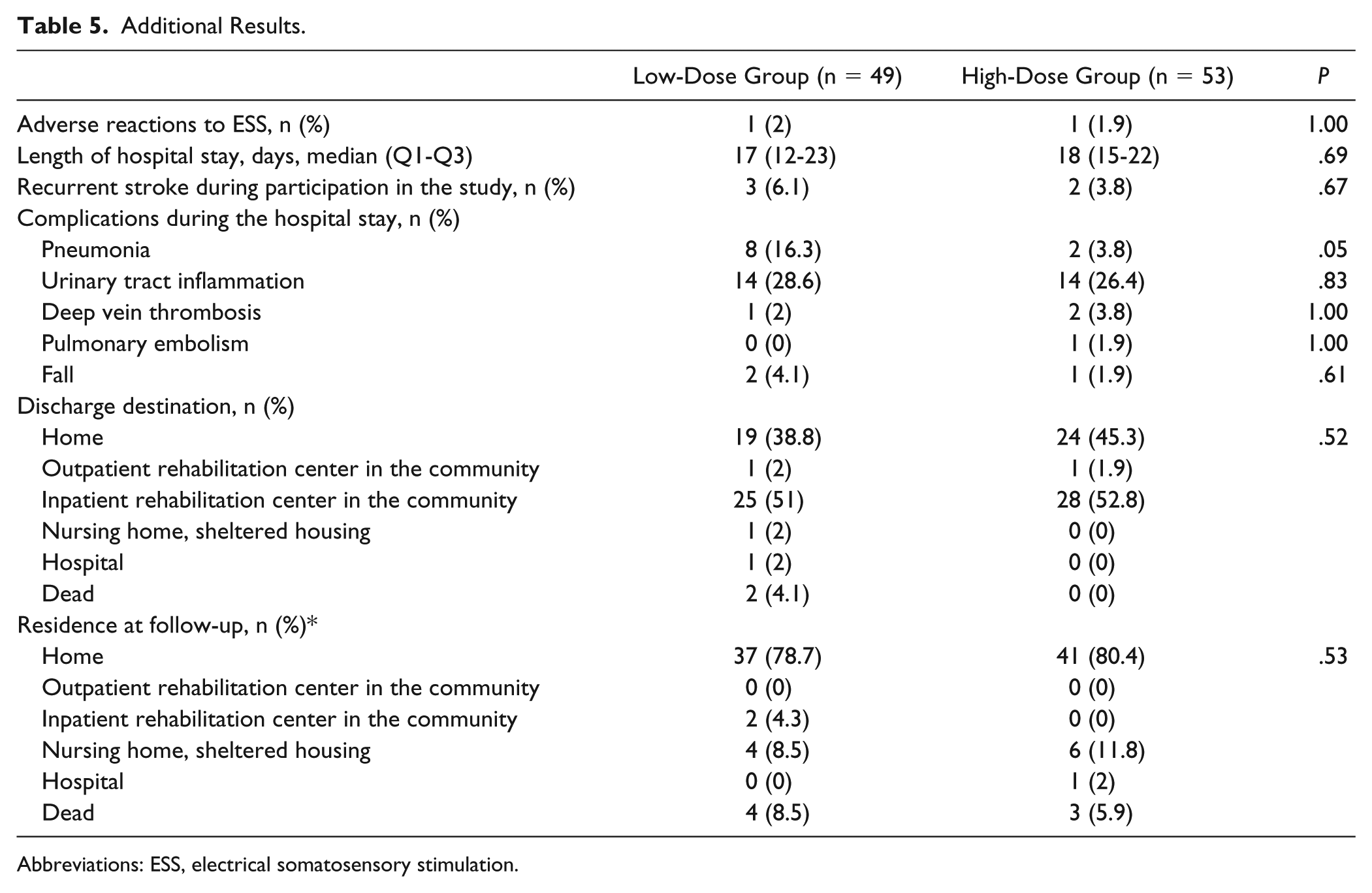
Abbreviations: ESS, electrical somatosensory stimulation.
Adverse reactions (itch) to ESS were reported in 1 patient in each group and alleviated by using allergy friendly electrodes. The percentage of complications during the hospitalization was similar in both groups. Likewise, the percentage of recurrent stroke was comparable; 6% in the low-dose group and 4% in the high-dose group. The ESS intervention had no effect on the discharge destination or the participants’ residence at follow-up (Table 5).
Discussion
This RCT investigated the effect of ESS on arm recovery and demonstrated that adding the present ESS protocol to arm training in the first 4 weeks poststroke is equally effective as arm training alone in improving hand dexterity, motor and sensory functions, and global disability. Both trial groups showed a positive, statistically significant effect of time for the most outcome measures; moreover, improvements in motor function, hand grip strength and global disability were ⩾MCID.
Several studies have demonstrated a positive, albeit transient, effect of a single 2-hour ESS session on different aspects of upper limb functioning in all stroke phases.23-28 A systematic review with meta-analyses, 53 including trials with repetitive ESS sessions could not demonstrate any convincing, beneficial effect in chronic stroke. Importantly, this conclusion rests on a very limited amount of data. Recently, an RCT 54 in subacute/chronic stroke patients with some active finger and wrist extension demonstrated positive, long-term effects in favor of ESS on functional capacity, but not on motor functions and the Stroke Impact Scale. Interestingly, the Stroke Impact Scale was positively influenced by the inactive placebo ESS, but the effect was temporary. This trial stimulated the radial and ulnar nerves and applied a high intervention dose (18 sessions × 2-hour ESS followed by 4-hour task-oriented arm training). To our knowledge, only 1 other trial 55 except the present one has been conducted in the acute stroke phase. Contrary to our results, it showed a positive long-term effect of ESS on hand grip strength and tip pinch strength in early rehabilitation of patients with mild-to-moderate stroke and severe arm impairments. It should be noted that the intervention dose was higher (20 sessions × 60-minute ESS) and the stimulation protocol was different compared with ours (200 µs pulses at 20Hz with maximum tolerated intensity to acupuncture points); ESS was given in addition to usual rehabilitation, but not specifically followed by arm training. In subacute stroke, 1 study 29 reported beneficial short-term effect of 12 sessions × 20-minute task-oriented practice preceded by 2-hour subsensory ESS to the median nerve. Furthermore, a postintervention positive effect was also shown after 10 sessions × 45-minute ESS delivered by a stimulation glove supplementary to usual rehabilitation. 56 Therefore, it appears that the current evidence for ESS during the acute stroke phase is conflicting. It also remains unknown if ESS may play a role in later stroke phases, and if so under which conditions. Interestingly, a recent systematic review with meta-analyses 57 demonstrated an advantageous effect of ESS when applied on to the lower limb, especially in the acute/subacute stroke.
In the current trial, 1-hour ESS was provided in conjunction with minimum 15-minute task-oriented arm training, which has previously shown to be an effective intervention during the first 4 weeks poststroke. 58 When used early after stroke, it is possible that ESS is not sufficiently potent to induce improvements beyond spontaneous neurologic recovery and the effect of task-oriented arm training. Note that the aforementioned trial in acute stroke 55 did not combined ESS with task-oriented arm training. A second possible explanation for the lack of effect in our trial is the relatively low number of ESS-sessions (12-13) compared with higher number (18-28) in other studies.30,54,55 Unfortunately, financial and logistical reasons prevented an ESS intervention beyond 4 weeks poststroke in this trial, and we cannot rule out that a longer intervention period might have altered the results. Likewise, we cannot rule out that using a standardized arm training protocol in which interventions such as neuromuscular electrical stimulation or robot-assisted therapy 59 prescribed to patients with severe impairments could have influenced the outcomes. Thirdly, we cannot exclude the possibility of a placebo effect, which has also been suggested by other, 55 or of our placebo intervention having a real effect. The influence of electrode location and stimulation parameters is difficult to assess as prior stimulation protocols vary considerably.33,53 Although we demonstrated within-group improvements ⩾MCID, it is important to emphasize that it is unknown whether they translated into an increased use of the affected arm in real-life situations. We would recommend that future studies employ, for example, accelerometers to capture the impact of arm interventions on real-life ADL performance, as well as participation measures that provide information on whether the intervention affects the patient’s fulfilment of life roles.
Stroke recovery can be defined as improved performance without distinguishing between the degree of compensation and pure neurologic recovery caused by physiological changes. In this trial, we applied outcome measures that capture both aspects of recovery: pinch strength, hand grip strength, PTT and FMA-UE for pure neurologic recovery, and BBT and mRS for recovery of functional capacity that may partially be obtained through compensatory strategies. However, we acknowledge that clinical measures as our selected primary outcome (BBT) cannot distinguish between pure neurologic recovery and compensation. To address this limitation, we support the recently published consensus-based recommendations for measurement in stroke recovery and rehabilitation trials 60 in urging the use of kinematic and kinetic measures in future intervention trials for the upper limb after stroke.
There was a trend that the low-dose group received a lower dose of arm training during the ESS intervention period (4 vs 5.3 hours,
Our eligibility criteria were broad. However, the final sample was homogenous, comprising mostly of patients with mild-to-moderate stroke, moderate arm impairments, and some voluntary finger extension. Consequently, the generalizability of our results is limited to stroke patients with these characteristics. Because the characteristics of the patients excluded from the trial (Figure 1) were not recorded, we cannot assess whether these patients were significantly different from those enrolled in the trial. Finally, the trial was not powered to perform subgroup analyses. Therefore, we were not able to detect possible effects on subgroups based on, for example, severity of arm paresis or potential for recovery based on transcranial magnetic stimulation and MRI. 61
Conclusion
Using the ESS protocol described herein immediately followed by arm training during the first 4 weeks poststroke does not add to arm training alone in enhancing the recovery of arm functioning. These results apply for patients with mild-to-moderate stroke, moderate arm impairments and some voluntary finger extension. Whether ESS initiated early after stroke is effective in patients with severe-to-moderate stroke and/or with potential for recovery despite initial inability of finger extension remains unknown. Likewise, whether ESS is effective in later time intervals after stroke and under which conditions (electrode placement, stimulation parameters, intervention dose, timing in relation to arm training) requires further investigation.
Supplemental Material
Supplemental_File – Supplemental material for Electrical Somatosensory Stimulation in Early Rehabilitation of Arm Paresis After Stroke: A Randomized Controlled Trial
Supplemental material, Supplemental_File for Electrical Somatosensory Stimulation in Early Rehabilitation of Arm Paresis After Stroke: A Randomized Controlled Trial by Emma Ghaziani, Christian Couppé, Volkert Siersma, Mette Søndergaard, Hanne Christensen and S. Peter Magnusson in Neurorehabilitation and Neural Repair
Footnotes
Authors’ Note
The data set is available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Capital Region of Denmark, Foundation for Health Research, Bevica Fonden, Lundbeck Foundation, the Danish Association of Occupational Therapists, Direktør Jacob Madsen & Hustru Olga Madsen’s fond, and the Department of Physical and Occupational Therapy at Bispebjerg Hospital. The funding sources had no influence on the trial design, data collection, analyses, interpretation and reporting of results.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
