Abstract
Background
Parkinson’s disease (PD) leads to impaired mobility and limited independence.
Objective
We investigated the effects of acupuncture on gait disturbance and analyzed hemodynamic changes caused by acupuncture in the cerebral cortex of patients with PD.
Methods
Participants (n = 26) with gait disturbance due to PD were randomly assigned to the intervention (acupuncture twice a week for 4 weeks + conventional therapy) or control (conventional therapy) groups. We analyzed gait parameters using the GAITRite system and hemodynamic responses in the cerebral cortices using functional near-infrared spectroscopy, Unified Parkinson’s Disease Rating Scale (UPDRS) scores, neurotransmitter levels, as well as the immediate effects of acupuncture in patients with PD.
Results
The participants tended to walk with hypometric gait (high cadence, short steps) overground. After acupuncture treatment, those in the intervention group showed a significant reduction in cadence and the UPDRS scores involving “walking and balance” compared with those in the control group (P = .004 and P = .020, respectively); the stride, swing, and single support times were significantly increased (P = .006, P = .001, and P = .001, respectively). Oxyhemoglobin levels in the intervention group while walking on a treadmill were significantly increased in the prefrontal and supplementary motor areas. The oxyhemoglobin levels in the prefrontal cortex and swing time revealed significant positive correlations.
Conclusions
Our findings indicated that acupuncture tended to improve hypometric gait and rearranged activation of the cerebral cortex. Thus, acupuncture may be a useful complementary treatment for gait disturbance, including hypometric gait, in patients with PD. Trial Registration Number. Clinical Research Information Service (KCT0002603), https://cris.nih.go.kr/cris/index.jsp
Introduction
Patients with Parkinson’s disease (PD), striatal dopamine deficiency by neuronal loss in substantia nigra, have complex motor and nonmotor symptoms.1,2 The motor symptoms in particular decrease the quality of life due to impaired mobility and limited independence.1 -5 Among motor symptoms, postural instability and gait worsen with PD progression and treatment resistance. 6 A previous study indicated that gait disorder in PD is characterized by an increased cadence and decreased swing and stride times; these are compensatory mechanisms to maintain velocity under a dual-task condition. 4 These findings are consistent with those of another study that reported the presence of festinating gait—defined as an increase in speed during a repetitive motor task, high-cadence, short steps, and a center of gravity in front of the feet—in patients with PD. 7 Early-stage motor symptoms, such as bradykinesia and rigidity, respond best to levodopa, which is the gold standard for symptomatic therapy in patients with PD. 8 Unfortunately, levodopa has little effect on late-stage symptoms such as gait and balance dysfunction. 8 Moreover, several limitations have been reported, such as levodopa-induced complications of motor fluctuations and dyskinesia and wearing-off of levodopa effects with long-term administration.8,9 Accordingly, prompt treatment via the most suitable intervention are necessary; this would help to prevent or delay the risk of falling and freezing of gait in advanced-stage PD.4,10,11
Acupuncture has been most commonly used as a complementary treatment for patients with PD; in several clinical studies, it has demonstrated significant positive effects, such as relieving PD symptoms 12 and reducing the required levodopa dosage and its side effects. 13 Regarding the effect of acupuncture on gait disturbance in PD, several clinical trials have demonstrated its ability to improve gait speed.14 -17 Electroacupuncture reportedly improves gait parameters, such as speed, stride length, and balance performance, in patients with PD.14,15 Cho et al 16 demonstrated improvements in the Unified Parkinson’s Disease Rating Scale (UPDRS) II (activities of daily living) and III (motor symptoms) scores after 8 weeks of acupuncture and bee venom acupuncture, compared with no treatment. Doo et al 17 found improvements in gait speed, quality of life, and UPDRS II and III scores after 12 weeks of combined treatment with acupuncture and bee venom, compared with pretreatment.
To generate locomotion movements, including gait, motor commands are projected from the cerebral cortex to the subcortical structures (basal ganglia and spinal cord); this involves automatic movement processes, such as the adjustment of balance and postural muscle tone.18,19 It is well known that motor dysfunction in patients with PD involves the cortico–basal ganglia–thalamocortical circuit and 2 major projection systems (direct and indirect pathways). 20 Another pathway, the cortico–subthalamic nucleus–pallidal hyperdirect pathway, has been acknowledged: the subthalamic nucleus receives direct glutamatergic projection from the motor cortex. 21 The motor circuits are connected via neurotransmitter pathways such as dopaminergic, glutamatergic, adenosinergic, noradrenergic, serotonergic, γ-aminobutyric acid (GABA) ergic, cholinergic, and histaminergic pathways. 1 Acupuncture or electroacupuncture stimulation may facilitate basal ganglion activity normalization and ameliorate motor disorder by changing the neurotransmitter levels in various PD animal models. 22 Electroacupuncture ameliorates motor impairment through enhancing GABAergic inhibition in the basal ganglion in the medial forebrain bundle–injured rat 23 and reducing group II metabotropic glutamate receptor (mGluR2/3) protein and mGluR3 mRNA expression in the striatum in 6-hydroxydopamine damages. 24 The dysfunction of subcortical structures in PD is accompanied by cerebral cortex rearrangement as a compensation strategy for the cortical basal ganglia circuit.18,25 Many studies have demonstrated, using functional near-infrared spectroscopy (fNIRS), that the oxyhemoglobin levels within the prefrontal cortex are increased in patients with PD and may be sensitive to compensation for restoration of gait disturbance.18,26 fNIRS is an important tool for the evaluation of neurofunctional activity during movements, such as walking. 26 However, fNIRS has never been used to assess the effects of therapies including acupuncture for improving gait disturbance in patients with PD. 26 Regarding the effect of acupuncture on the molecular mechanism in patients with PD, we hypothesized that acupuncture treatment may normalize motor circuits by changing the neurotransmitter levels in the subcortical area and rearrange cerebral cortex activity and improve gait disturbance.
Based on several clinical trials, acupuncture is known to improve gait performance in patients with PD14 -17; however, information on the brain regions and projection systems involved in the clinical effects in PD is limited. Our aims were as follows: (1) to evaluate the clinical effect of acupuncture on gait disorder in patients with PD using the GAITRite system (CIR Systems Inc), which can objectively assess gait parameters; (2) to examine the acupuncture effect on cerebral cortex rearrangement by identifying hemodynamic changes that occur in the motor and prefrontal cortices using the fNIRS technique; and (3) to elucidate the possible hypothesis for the effect of acupuncture on gait disturbance by changing cerebral cortex activity via projection system normalization and the neurotransmitter levels following acupuncture.
Methods
Study Design
The study protocol was approved by the institutional review board of the Clinical Trial Center at Dunsan Korean Medicine Hospital, Daejeon University as previously published 27 and is shown in Supplementary Methods in more detail. Computer-generated block randomization was used to allocate all participants to the control (n = 13) or intervention (n = 13) groups and was performed by an independent statistical researcher who was not involved in the study. The outcomes were evaluated by 2 assessors: one assessor was blinded (assessor A), whereas the other was not (assessor B). Assessor A evaluated the outcomes in the control and intervention groups at 0, 4, and 8 weeks. Assessor B evaluated outcomes in the intervention group, which could not be blinded. All participants received all interventions and assessments in the dopaminergic “on” state. This 8-week study included an intervention phase (4 weeks) and a follow-up phase (4 weeks).
Participant Recruitment
The participants were recruited from January 2018 to June 2018, and the trial was conducted from July 2018 to April 2019. A total of 26 participants who provided written informed consent were included in the current study (Figure 1).
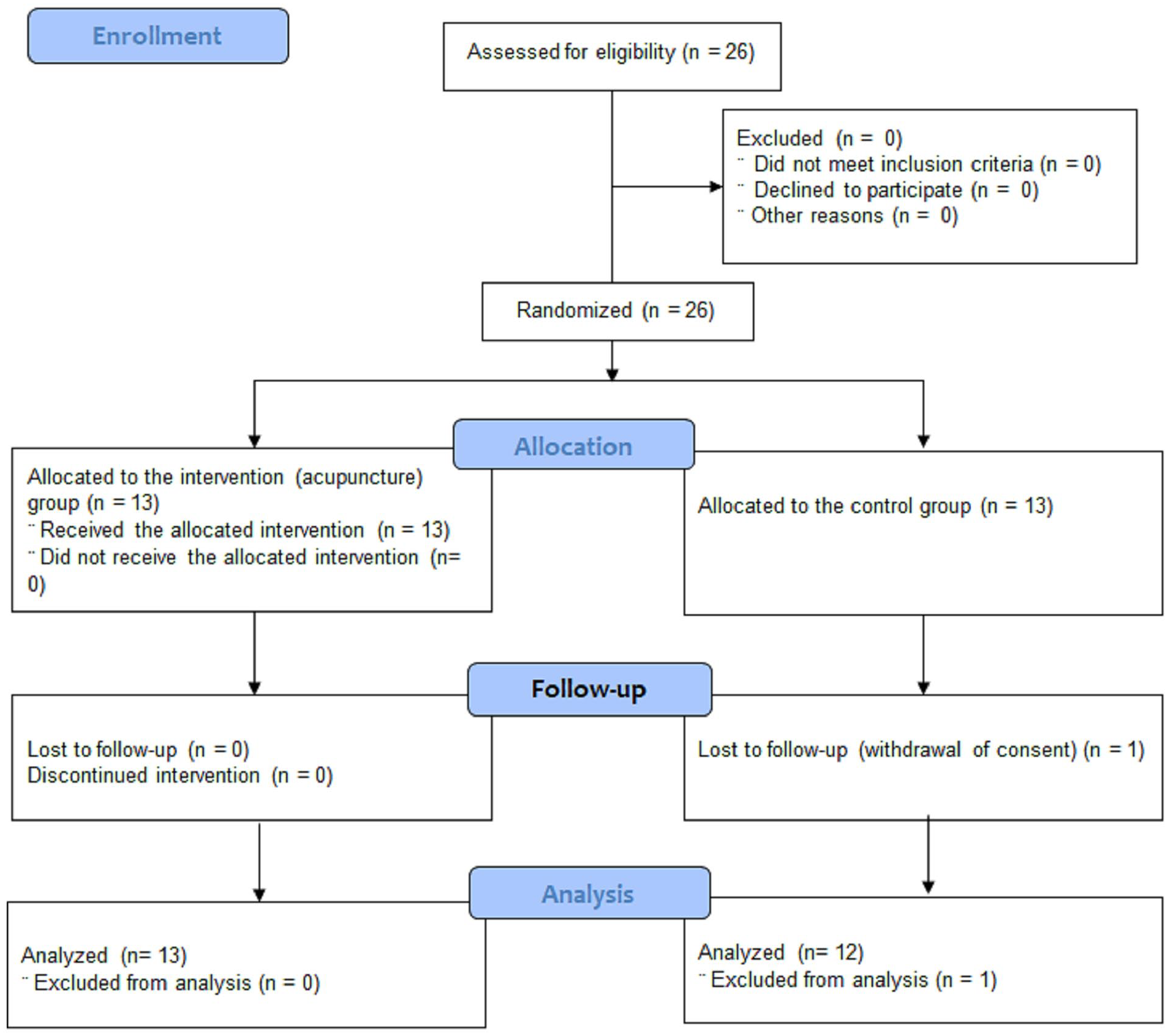
CONSORT (Consolidated Standards of Reporting Trials) flow diagram.
Intervention
The participants in the intervention group received acupuncture 2 days per week for 4 weeks. Acupuncture was performed on the dorsal side. All acupuncture treatments performed in the intervention group complied with the STRICTA (Standards for Reporting Interventions in Clinical Trials of Acupuncture; 2010) guidelines. The control group received conventional treatment only.
Measurements
Assessment of Gait Performance
Gait parameters were measured using the GAITRite system. The MDS-UPDRS (Movement Disorder Society UPDRS) scores were partially assessed and the UPDRSA (a subsection of UPDRS that includes the “walking and balance” and “freezing” parts of the UPDRS II assessment for activities of daily living) was used to evaluate the “walking and balance” and “freezing” parts of the UPDRS II and the UPDRSM (a subsection of UPDRS that includes the “gait,” “postural stability,” “posture,” and “body bradykinesia” parts of the UPDRS III motor assessment), and the “gait,” “postural stability,” “posture,” and “body bradykinesia” parts of the UPDRS III. Because this study evaluated the effect of acupuncture by limiting gait disturbance among PD motor symptoms, the measurement was based on the postural instability/gait difficulty items 28 among the UPDRS.
Hemodynamic Changes in the Cerebral Cortex
The fNIRS system (NIRScout 1624, NIRx Medical Technology) was used to assess the hemodynamic changes during gaiting on a treadmill. This system performed near-infrared topographic measurements at 2 different wavelengths (760 and 850 nm) at a sampling rate of 4.17 Hz. The protocol involved 3 rounds of standing (30 seconds) alternated with 2 rounds of walking (30 seconds) at 0, 4, and 8 weeks, to observe hemodynamic changes in the motor cortices and the prefrontal cortex (Supplementary Figure 1). The optodes were positioned based on the 10-20 international electrode system. The system consisted of 30 optodes (15 transmitters and 15 receivers), which were placed on the motor (premotor cortex, supplementary motor area, primary motor cortex, primary somatosensory cortex) and prefrontal cortices of each subject, and NirsLAB version 201601 was used for data collection and analysis. Optode position registration was conducted using a 3D magnetic digitizer stylus (PATRIOT, Polhemus). The analyses were performed using the open-source software package NIRS-SPM implemented in MATLAB (MathWorks, Inc). The hemodynamic response function was applied for correction of motion artifacts and noise from the system, and the wavelet-MDL (wavelet–minimum description length) based detrending algorithm was used for correction of low-frequency drift. 29 General linear model analysis was performed to test for significant cortical activation during the task. 30 The changes in oxyhemoglobin concentration were estimated from each channel of the regions of interest (ROIs) during treadmill walking. The grand average of each hemodynamic response was calculated for each ROI (Figure 3). In addition, t-statistic maps computed for group analyses were plotted onto a conventional brain template. Differences in oxyhemoglobin concentrations were considered significant at an uncorrected threshold of P < .05 (Figure 4).
Plasma and Serum Neurotransmitters
The analysis of neurotransmitters was performed at the Seoul clinical laboratories in South Korea.
Statistical Analysis
One participant in the control group, who dropped out at the beginning of the study without any measurement, was excluded from the statistical analysis. The continuous and categorical variables are presented as mean ± SD and percentage, respectively. To compare baseline characteristics between the 2 groups, independent t tests were performed for continuous variables and chi-square tests for categorical variables. The gait parameters, 10-m walk test, Timed Up and Go test, and UPDRS scores were measured at baseline (week 0), week 4, and week 8. The effects of the intervention on these variables were evaluated with linear models in which response variables were the observed values at week 4 and week 8; the covariates were the group indicator, observed time, and its baseline value; and the covariance structure of the error term was assumed to be a compound symmetric matrix. The effect size (ES) of the covariate (the observed time and the baseline value) adjusted mean difference between the 2 groups was calculated as ES = (the mean difference between the 2 groups)/SE, in which the parameters were estimated from the linear mixed model. The normality of each response variable was checked using a Shapiro-Wilk test. All response variables, except for the UPDRS, at week 4 did not reject normality; however, each result is not reported here. The effect of the intervention on each response was calculated from the results of the linear mixed models. The mean oxyhemoglobin values were analyzed using the linear mixed models. The changes of the neurotransmitter levels were estimated through evaluation of time points using a repeated-measures analysis of variance. Pearson’s correlation coefficients between gait parameters and oxyhemoglobin values were estimated to evaluate their relationships. A P value of less than .05 was considered statistically significant. All statistical analyses were performed using SAS for Windows, release 9.4 (SAS Institute, Inc, 2016).
Data Management and Monitoring
The research nurse completed case report forms, which included information required by the protocol. Then, 2 independent research assistants entered the data separately into Excel (Microsoft Corp), and the results were compared. An independent monitoring committee that was not associated with the research team of the trial regularly monitored the patients’ safety, investigated adverse events, and reviewed the data quality control.
Results
Demographic and Clinical Characteristics
Participants’ demographic characteristics and clinical findings (including gait parameters at baseline) are shown in Table 1. The baseline characteristics did not differ significantly between the groups. One patient in the control group dropped out due to withdrawal of consent; the other participants completed the assessments without occurrence of significant adverse events (Figure 1).
Baseline Characteristics of the Intervention and Control Groups. a
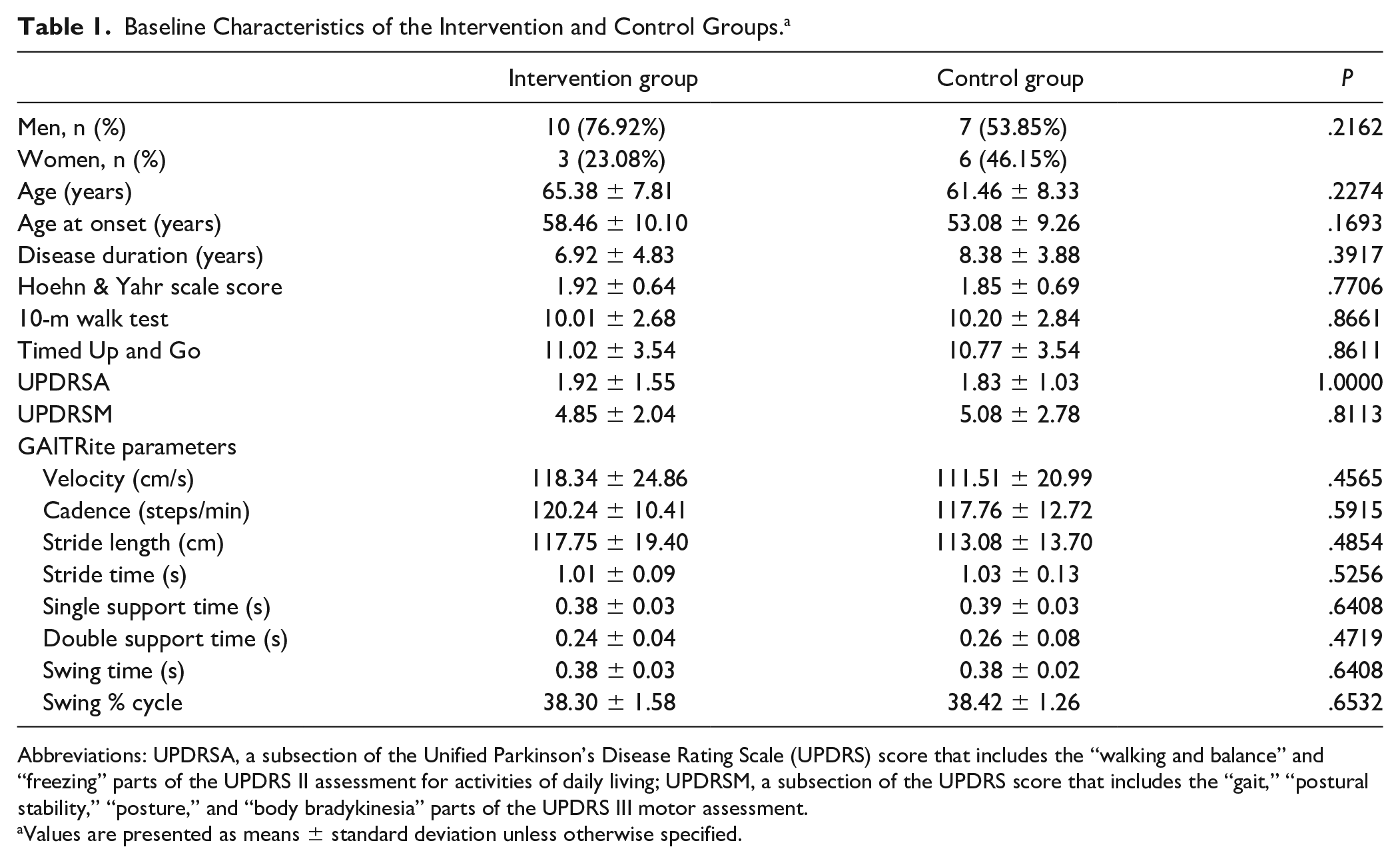
Abbreviations: UPDRSA, a subsection of the Unified Parkinson’s Disease Rating Scale (UPDRS) score that includes the “walking and balance” and “freezing” parts of the UPDRS II assessment for activities of daily living; UPDRSM, a subsection of the UPDRS score that includes the “gait,” “postural stability,” “posture,” and “body bradykinesia” parts of the UPDRS III motor assessment.
Values are presented as means ± standard deviation unless otherwise specified.
Gait Performance
The comparison of gait parameters and indexes between the intervention and control groups are shown in Table 2 and Figure 2. Results generated using the GAITRite system indicated that cadence was significantly decreased by acupuncture treatment during walking when compared with the control group (P = .004, ES = −3.163). In contrast, the stride, swing, and single support times were significantly increased in the intervention (P = .006, ES = 3.016; P = .001, ES = 3.068; P = .001, ES = 3.068, respectively) compared with the control group. The parameters evaluated using the GAITRite system included velocity, stride length, double support time, and swing % of cycle; these parameters did not differ significantly between the groups (P = .333, ES = −0.996; P = .549, ES = 0.608; P = .238, ES = 1.211; and P = .968, ES = −0.040, respectively) (Table 2). Supplementary Figure 2 shows the relationships among velocity versus cadence, velocity versus stride length, velocity versus stride time, and velocity versus swing time in both groups using scattergrams of individual gait parameters for any given velocity of walking. The slope and intercept parameters were determined using a linear model. The regression of velocity versus cadence, stride length, stride time, and swing time in the intervention group showed a similar slope as in the control group, but the intercept of cadence was lower, and the intercept of stride length, stride time, and swing time were higher in the intervention group than in the control group. These analyses indicated that for any given velocity, the cadence was lower; and stride length, stride time, and swing time were higher in the intervention group than in the control group. Only the difference in the intercept of cadence versus velocity at 8 weeks was found to be statistically significant between the intervention and control groups (P = .038). Especially, after acupuncture, the individual cadence was reduced, and the individual stride length, stride time, and swing time were increased at the same velocity in the intervention and control groups. The results in Supplementary Figure 2 support the results shown in Figure 1. In addition, the immediate and cumulative effects after acupuncture did not differ from that before acupuncture in the intervention group. The 10-m walk and Timed Up and Go test results did not differ between the intervention and control groups. The UPDRSA scores during walking were significantly decreased by acupuncture when compared with those of the control group (P = .020). The UPDRSM scores did not differ between the groups.
Gait Performance in the Intervention and Control Groups. a
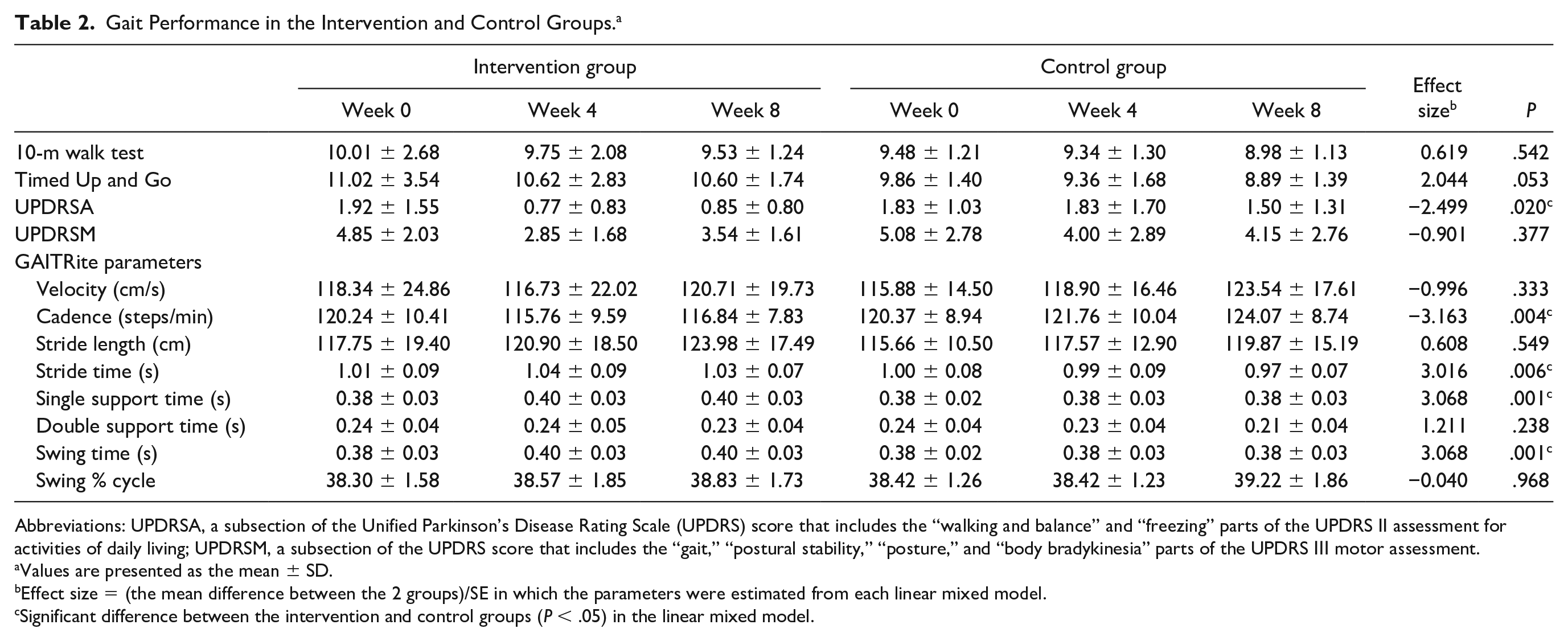
Abbreviations: UPDRSA, a subsection of the Unified Parkinson’s Disease Rating Scale (UPDRS) score that includes the “walking and balance” and “freezing” parts of the UPDRS II assessment for activities of daily living; UPDRSM, a subsection of the UPDRS score that includes the “gait,” “postural stability,” “posture,” and “body bradykinesia” parts of the UPDRS Ⅲ motor assessment.
Values are presented as the mean ± SD.
Effect size = (the mean difference between the 2 groups)/SE in which the parameters were estimated from each linear mixed model.
Significant difference between the intervention and control groups (P < .05) in the linear mixed model.
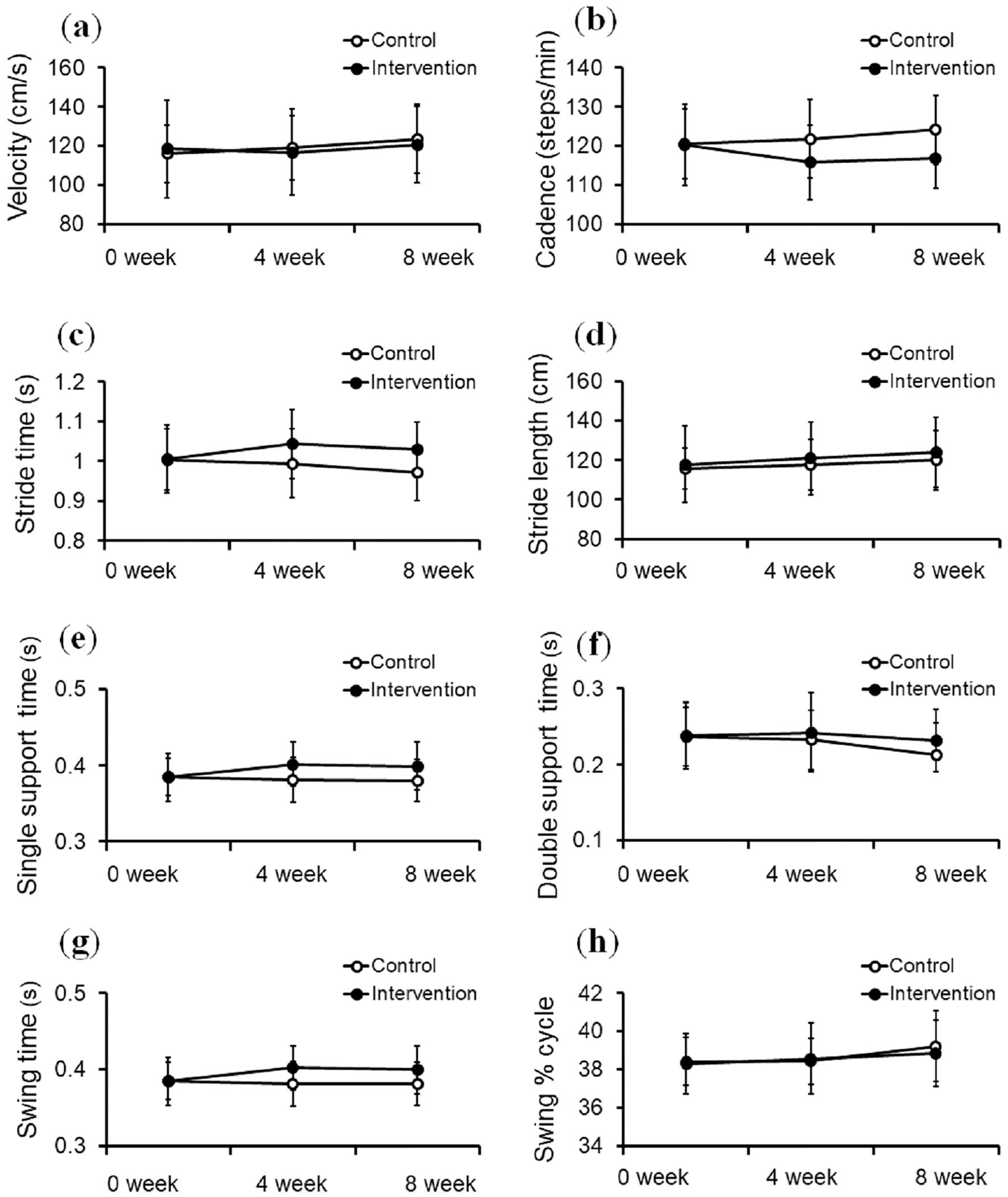
Changes in gait parameters in the intervention and control groups: (a) velocity, (b) cadence, (c) stride time, (d) stride length, (e) single support time, (f) double support time, (g) swing time, and (h) swing % of cycle.
Hemodynamic Change in the Cerebral Cortex While Walking on a Treadmill
We demonstrated that the hemodynamic response of oxyhemoglobin indicated an increase in cerebral cortex activity after acupuncture, compared to the control group (Figures 3 and 4). The time response and mapping image of oxyhemoglobin are shown in Figures 3 and 4, respectively. In particular, channels 11 and 15 corresponding to the supplementary motor area showed significantly greater increases during walking on the treadmill in the intervention than in the control group (P = .037 and P = .022, respectively) (Supplementary Table 1). Additionally, channels 35, 39, and 40 corresponding to the prefrontal cortex showed significantly greater increases during walking on the treadmill in the intervention than in the control group (P = .035, P = .023, and P = .006, respectively) (Supplementary Table 1). The anatomical location of each channel is shown in Supplementary Table 2.
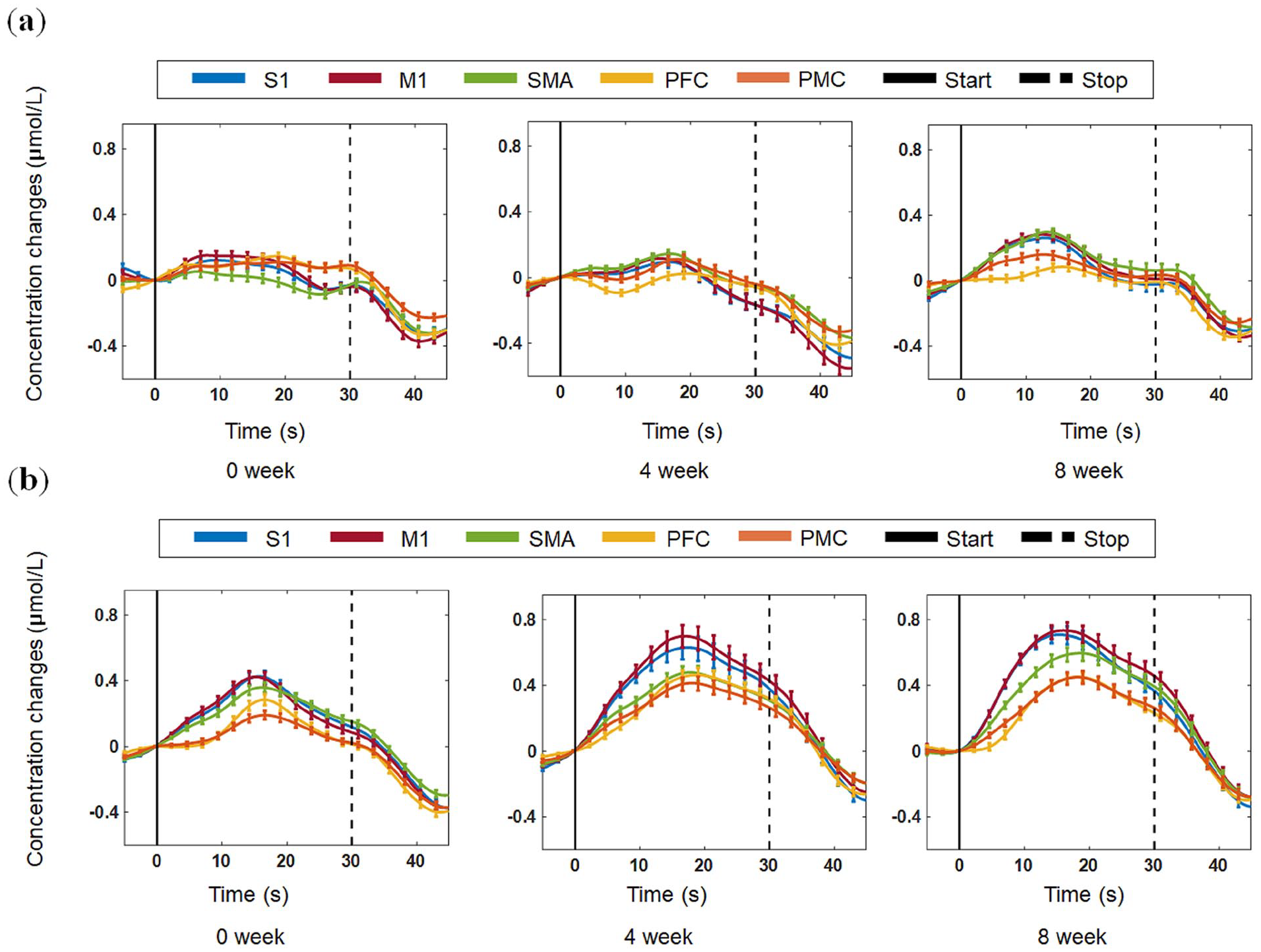
Hemodynamic changes shown using functional near-infrared spectroscopy. (a) Time response of the cerebral cortex in the control group. (b) Time response of the cerebral cortex in the intervention group. S1, primary somatosensory cortex; M1, primary motor cortex; SMA, supplementary motor area; PFC, prefrontal cortex; PMC, premotor cortex.
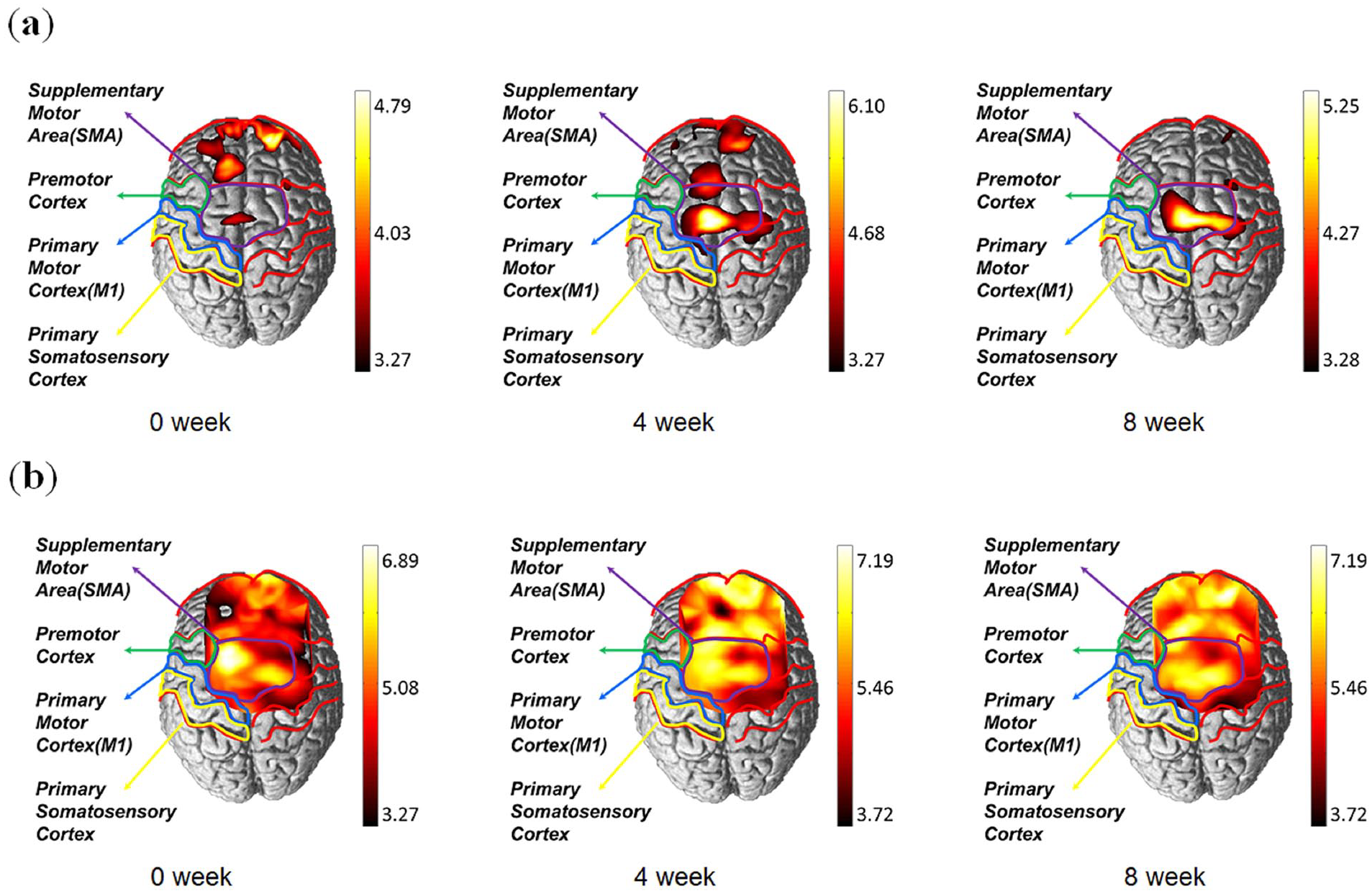
Hemodynamic changes shown using functional near-infrared spectroscopy. (a) Mapping changes in the cerebral cortex in the control group. (b) Mapping changes in the cerebral cortex in the intervention group.
Changes of Neurotransmitter Levels
The neurotransmitter levels were compared depending on the cumulative frequency of acupuncture treatment in the 11 participants of the intervention group. There was a tendency for the tyrosine levels to decrease and for the epinephrine and norepinephrine levels to increase after acupuncture, but there was no statistical significance (Supplementary Table 3). The GABA concentrations were low in blood samples and could not be detected by the instrument.
Discussion
To the best of our knowledge, this study is the first to investigate the effect of acupuncture on gait performance and brain activity in patients with PD. We found that acupuncture treatment in such patients decreased the cadence and increased the stride and swing times. Moreover, in the present study, hemodynamic responses in the prefrontal and motor cortices, including the supplementary motor area, during walking on the treadmill were significantly increased by acupuncture. The supplementary motor area and prefrontal cortex are known to control balance,31,32 and we hypothesized that activation of these regions might modulate gait disturbance to correct balance in patients with PD. Our results suggested that acupuncture has the effect of improving gait disturbance in patients with PD, which may be related to a neural mechanism, such as cerebral cortical reorganization.
The slowness that accompanies gait disturbance in patients with PD is typically characterized as reduced stride length and cadence.33,34 Accordingly, in this study, we expected that acupuncture in patients with PD with bradykinesia improved walking velocity by increasing stride length and cadence. However, the results of this study did not support our expectations. Acupuncture treatment did not improve walking velocity in the intervention group; it reduced the cadence and increased the swing and stride times compared with the control group (Figure 2). In addition, cadence against velocity was lower, and the stride length against velocity was higher for any given velocity in the intervention than in the control group (Supplementary Figure 2). In particular, when the patients walked at the same velocity, our results indicated that acupuncture reduced the frequency and increased the amplitude of the gait. Based on these findings, we speculate that acupuncture improved the tendency toward hypometric steps, such as the festination-like gait in patients with PD. In a previous study, a higher cadence and shorter stride length were reported in patients with PD than in elderly healthy controls. 35 This gait pattern can be regarded as gait festination, making hypometric steps to correct balance; this is one of the locomotion disturbances in PD characterized by walking in a sped-up fashion with a high cadence, short steps, and a forward center of gravity while the trunk leans forward involuntarily.7,36 In this study, we also identified that patients in the control group showed a tendency toward hypometric gait with a significantly higher cadence and lower stride length than those in the intervention group, as time passed from 0 to 8 weeks (Figure 2). It was presumed that the gait characteristics of the recruited participants were more prone to hypometric gait. We assume that the relative increase in the cadence was caused by a compensatory mechanism to maintain velocity, thereby regulating stride length and preventing a fall due to the forward center of gravity. These findings suggested that acupuncture treatment may improve hypometric steps by modulating cadence and stride length of gait disturbances in patients with PD.
In previous studies, deficits in cerebral cortex activation were shown to be associated with PD 37 ; this may have been due to the decreased positive efferent feedback arising from the cortico-basal ganglia-thalamocortical circuit. 38 The subcortical structure, including the basal ganglia, is involved in balance adjustment and muscle-tone regulation for walking. 19 PD affects the subcortical pathway, requiring increased cortical activity as compensation to perform walking and balance tasks. 25 Several studies have been conducted on cortical motor reorganization in patients with PD. Frontal cortical activity in such patients tends to increase with walking and balance tasks. 18 Maidan et al 39 reported higher prefrontal activation in patients with PD compared with healthy controls during usual walking and obstacle negotiation using fNIRS. These findings were consistent, as they suggested complementary approaches, including increasing the efficiency of neural control, that is, increasing cerebral cortex activation, and extending capacity by cortical recruitment for improving movement in patients with PD.39,40 In addition to the prefrontal cortex, high-frequency repetitive transcranial magnetic stimulation, a noninvasive method used to stimulate the human brain, over the supplementary motor area in patients with PD, improves the total UPDRS scores. Hupfeld et al 32 reported that transcranial direct current stimulation transiently modulated excitability in cortical regions; applied to the supplementary motor area, it may improve motor tasks, including balance tasks. 32 Therefore, the supplementary motor area has been suggested as a potential stimulation region for PD treatment. 38 To determine the effect of acupuncture on cerebral cortex activation in patients with PD, we also investigated changes in prefrontal and motor cortex activity using an fNIRS system, while the patients walked on a treadmill. According to our findings, the oxyhemoglobin, indicating brain activity, increased significantly more following acupuncture in the intervention than in the control group, especially in some channels of the prefrontal cortex and supplementary motor area. The supplementary motor area and prefrontal cortex have been known to play important roles in balance control.31,32 Based on these findings and the results of our study, it may be suggested that acupuncture treatment improves balance control by reorganizing the cerebral cortex, that is, activation of the supplementary motor area and prefrontal cortex, which might improve hypometric gait to correct balance in patients with PD.
To identify the potential mechanism at work in acupuncture treatment for PD gait disturbance, we investigated the changes in neurotransmitter levels involving the cortico–basal ganglia–thalamocortical circuit. In previous studies, acupuncture ameliorated motor disorder by changing GABA and glutamate contents in the basal ganglia of animal models.23,24 In this study, the change of the neurotransmitter levels in the blood was not statistically significant and GABA content was not detected. We expected a change in the blood neurotransmitter levels of patients with PD following the acupuncture treatment, but we could not confirm the change. We assumed that the improvement of hypometric gait in our patients may be due to the improvement of neurotransmitter availability and not due to a change in the neurotransmitter levels. A previous study showed that acupuncture might improve motor dysfunction with accompanying dopaminergic neuron protection in PD mouse models. Dopamine availability might be enhanced by increasing dopamine release in the synaptic cleft, but might not restore the depletion of striatal dopamine levels. 41 Based on these findings, we speculated that increased neurotransmitter availability in the subcortical area might affect the cortico–basal ganglia–thalamocortical circuit, leading to cerebral cortical rearrangement, and resulting in improved hypometric gait. Other possibilities for different neurotransmitter levels changes in PD animal model studies and patients with PD in this study may be part of the accuracy of neurotransmitter analysis. Generally, in animal testing, dopamine is measured in the dopaminergic fibers (terminal) of the striatum. However, as the neurotransmitter was measured in the patients’ blood in this study, the accuracy of the ease of analysis may be somewhat reduced. Therefore, further research will be necessary to supplement the protocols that overcome these limitations or to replace them with nonclinical experiments.
There were some limitations to this study. First, the control group in this study consisted of patients with PD who received non-acupuncture treatment. Therefore, the participants and the practitioner were not blinded to the intervention allocations because nontreatment, rather than sham treatment, was used in the control group. Second, it is difficult to judge the effect of each acupoint when several acupoints are used in a session. In this study, acupoints were selected according to clinically validated points. Therefore, further studies are needed to clarify the effects of specific acupoints. Third, the study scale was small; the intervention duration was short (only 4 weeks), and the sample size was small. Fourth, the assessments of gait performance and hemodynamic response were performed during different tasks on overground and treadmill walking, respectively. We intended for the conditions to be identical, but it was impossible to walk overground during the fNIRS assessment because our fNIRS systems are not portable. In a previous study, walking on the treadmill in patients with PD caused different hemodynamic changes in the cerebral cortex and improved gait rhythmicity, including cadence decrease, compared to overground walking.42,43 Therefore, it was difficult to confirm a direct correlation between gait improvement and hemodynamic changes in this study. Nevertheless, this is the first study to provide clues regarding the possibility of correlation between hypometric gait improvement and cerebral cortex activation by acupuncture in patients with PD. As a result of the correlation analysis (using Pearson’s correlation coefficient) between gait performance and hemodynamic change, the delta (week 8 – week 0) activities of the prefrontal cortex (channels 35 and 39) were positively correlated with the delta swing and single support times in the intervention group (r = 0.640, P = .019 and r = 0.652, P = .016, respectively; data not shown). It is thought that further research is needed to confirm such a direct correlation. Fifth, neurotransmitter levels were measured in the blood; as a result, the changes in neurotransmitter levels in the brains of patients with PD were not evaluated directly. Recently, using the living brain of patients with PD has been reported to more accurately reflect disease-relevant altered gene expression than the use of peripheral tissues. 44 To confirm the neural changes by acupuncture in patients with PD in this study, further research with molecular analyses of PD brain tissue is essential. Despite these limitations, our results provide a basis for further large-scale clinical trials to confirm the effect of acupuncture in PD and identify possible concurrent therapies.
Conclusions
This is the first study to suggest acupuncture’s benefit as a complementary treatment for the improvement of hypometric gait in patients with PD, and that it could improve balance, including activation of the cerebral cortex (prefrontal cortex and supplementary motor area). Particularly, acupuncture improved the cadence that could not be improved by levodopa treatment,35,45 making it valuable as a concurrent therapy.
Supplemental Material
supplementary_fig_2-resubmit – Supplemental material for Gait Disturbance Improvement and Cerebral Cortex Rearrangement by Acupuncture in Parkinson’s Disease: A Pilot Assessor-Blinded, Randomized, Controlled, Parallel-Group Trial
Supplemental material, supplementary_fig_2-resubmit for Gait Disturbance Improvement and Cerebral Cortex Rearrangement by Acupuncture in Parkinson’s Disease: A Pilot Assessor-Blinded, Randomized, Controlled, Parallel-Group Trial by Jung-Hee Jang, Sangsoo Park, Jinung An, Jong-duk Choi, In chan Seol, Gunhyuk Park, Seung Hyun Lee, Young Moon, Weechang Kang, Eun-Sun Jung, Ji-yun Cha, Chan-young Kim, Siyeon Kim, In Chul Jung and Horyong Yoo in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank nurse Misun An (Dunsan Korean Medicine Hospital) for her help.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant of the Traditional Korean Medicine R&D Project, Ministry of Health & Welfare, Republic of Korea. (HI15C0006).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
Data Availability Statement
The source document data used to support the findings of the present clinical trial have been deposited in the Clinical Trial Center of Dunsan Korean Medicine Hospital, Daejeon University repository (IRB number DJDSKH-17-BM-20).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
