Abstract
Background and Aim:
Individuals with Parkinson’s disease (PD) with and without freezing of Gait (FoG) may respond differently to exercise interventions for several reasons, including disease duration. This study aimed to determine whether both people with and without FoG benefit from the Agility Boot Camp with Cognitive Challenges (ABC-C) program.
Methods:
This secondary analysis of our ABC-C trial included 86 PD subjects: 44 without FoG (PD−FoG) and 42 with FoG (PD + FoG). We collected measures of standing sway balance, anticipatory postural adjustments, postural responses, and a 2-minute walk with and without a cognitive task. Two-way repeated analysis of variance, with disease duration as covariate, was used to investigate the effects of ABC-C program. Effect sizes were calculated using standardized response mean (SRM) for PD−FoG and PD + FoG, separately.
Results:
The ABC-C program was effective in improving gait performance in both PD−FoG and PD + FoG, even after controlling for disease duration. Specifically, dual-task gait speed (P < .0001), dual-cost stride length (P = .012), and these single-task measures: arm range of motion (P < .0001), toe-off angle (P = .005), gait cycle duration variability (P = .019), trunk coronal range of motion (P = .042), and stance time (P = .046) improved in both PD−FoG and PD + FoG. There was no interaction effect between time (before and after exercise) and group (PD−FoG/PD + FoG) in all 24 objective measures of balance and gait. Dual-task gait speed improved the most in PD + FoG (SRM = 1.01), whereas single-task arm range of motion improved the most in PD−FoG (SRM = 1.01).
Conclusion:
The ABC-C program was similarly effective in improving gait (and not balance) performance in both PD−FoG and PD + FoG.
Introduction
Parkinson’s disease (PD) is a progressive, neurodegenerative movement disorder characterized by motor and non-motor impairments. 1 Balance and gait impairments are among the most debilitating motor symptoms that may become increasingly problematic as the disease progresses leading to postural instability, gait disturbance, immobility and falls, and ultimately affecting activities of daily living and quality of life.2,3 Although certain aspects of gait can be improved by pharmacological and surgical interventions, aspects of balance often worsen with pharmacological treatments. 4 Nonpharmacological interventions, such as exercise, on the other hand, could serve as an important element in the management of balance and gait impairments in people with PD, and thus may improve the quality of life.5,6
Exercise is advocated as a supplement to pharmacological therapy to improve balance and gait impairments in PD. 6 Several studies have shown that mobility and cognition, particularly executive function and attention, are highly interconnected and affect each other in PD. 7 Given the evidence of cognitive network involvement in mobility tasks and the overlap between cognitive and motor function, integrated motor, and cognitive training may increase functional improvement and quality of life in people with PD. 8 Due to the deficits in balance and gait, we originally proposed the Agility Boot Camp (ABC) program for people with PD and results showed an improvement in mobility such as gait speed and balance in people with PD. 9 However, the original ABC program did not address freezing of gait (FoG).
The majority of patients with PD eventually develop FoG, a brief episode of inability to step that typically occur on initiating gait or on turning while walking. 10 Up to 26% of people with mild PD and 80% of those with severe PD are affected by FoG, and it is one of the most common reasons for falls and lack of independence. 11 FoG leads to overall loss of mobility, increased social isolation, and decreased quality of life. 10 In addition, cognitive problems are more apparent in people with PD with FoG (PD + FoG) than in those without FoG (PD−FoG). 12 Specifically, executive function, such as response inhibition, set-switching, and dual-task function are more impaired in those with FoG. 13
Furthermore, there is currently no consensus on the brain mechanism underlying FoG and no comprehensive treatment for this troubling feature of PD. 10 Hence, with a motivation to design an intervention that may be effective for those with, as well as without, FoG, we designed the Agility Boot Camp–Cognitive (ABC-C) that is an adaption of the original ABC program with additional executive-function tasks during mobility exercises known to be challenging for people with PD.8,13,14
There is no consensus about whether PD + FoG responds similarly to a rehabilitation intervention compared to PD−FoG. For example, one study showed that PD + FoG, but not PD−FoG, showed improved performance with closed-loop cueing strategies compared with open-loop strategies. 15 However, PD + FOG showed compromised retention of motor learning and compromised implicit sequence learning compared to PD−FoG.16,17 Furthermore, PD + FoG showed less improvement and less retention of improvement in postural responses through 1 day of repeated perturbation exposure compared to PD−FoG. 18 In contrast, PD + FoG and PD−FoG both showed improvement in postural instability with a treadmill intervention 19 and with use of rhythmic auditory stimuli for balance training. 20 Similarly, PD + FoG benefited similarly to PD−FoG in improving gait velocity after dual-task gait training 21 and in improving turning and freezing after cued-gait training.22,23 Most studies comparing effects of exercise in PD−FoG versus PD + FoG focused on clinical measures of balance/gait and freezing. Previous studies did not investigate the effects of an exercise intervention on a comprehensive set of objective measures including automatic postural responses (APR), anticipatory postural adjustments (APA), postural sway as well as single-task and dual-task gait.
We have recently identified a set of objective measures of balance and gait, calculated with wearable inertial measurement units (IMUs) 24 sensors, that optimally discriminate people with PD from healthy control subjects. For such purpose, we used the baseline assessment prior to the ABC-C program. Subsequently, we investigated the effect of the ABC-C program on the same objective (from IMUs) and clinical measures and showed that objective measures had a higher effect size compared to the clinical measures.24,25 These results are promising, but to reach the full potential of nonpharmacological interventions, the clinicians need to know which specific people with PD (eg, those with or without FoG) would benefit most from this rehabilitation intervention.
The purpose of this secondary analysis of our clinical trial was to investigate whether the ABC-C program is similarly effective in both PD + FoG and PD−FoG in improving balance and gait with a comprehensive set of objective measures incorporating different aspects of balance and gait. We hypothesized that both PD + FoG and PD−FoG would show similar improvement after the ABC-C program as our program was tailored to challenge each participant’s motor and cognitive abilities.
Methods
Study Design and Participants
The design and outcomes of the ABC-C program have been published elsewhere.8,13,14 In summary, we performed a randomized, single-blinded, cross-over exercise trial at Oregon Health & Science University (OHSU) and the Veterans Affairs Portland Health Care System (VAPORHCS). The OHSU and VAPORHCS ethics committees approved all aspects of the study (OHSU/VAPORHCS IRB protocols 4131 and 8979, respectively). All research was in compliance with the Helsinki Declarations. This trial was registered at clinical trials.gov (NCT02231073) and the trial protocol has been published. 14
The ABC-C program was described in detail in previous publications from our group.8,13,14 In summary, people with PD were randomized into 1 of 2 intervention groups, exercise first (ABC-C) or education first, by a computerized block randomization. Exercise intervention consisted of 90-minute group exercise sessions, 3 days per week for 6 weeks, led by a certified exercise trainer. The group exercise classes were targeted to be between 3 and 6 people per class.24,25 Education intervention consisted of a 90-minute group session, once a week for 6 weeks in which participants were taught how to live better with their chronic conditions. Inclusion criteria were: (1) diagnosis of PD by movement disorders specialists according to the United Kingdom Brain Bank criteria; (2) 50 to 90 years of age; (3) mild to moderate PD; (4) on stable antiparkinsonian medication; (5) without major musculoskeletal, peripheral, or central nervous system disorders (other than PD) that could significantly affect their balance and gait; (6) without excessive use of alcohol or recreational drugs; (7) without history of structural brain disease, active epilepsy, stroke, or dementia that would interfere with consent or ability to follow testing procedures; (8) able to stand or walk for 2 minutes without an assistive device. Exclusion criteria were any other neurological disorders or musculoskeletal impairments that interfere with gait or balance and cognitive deficits, such as dementia, limiting an ability to follow procedures. The presence of FoG was assessed using the New Freezing of Gait Questionnaire (NFoGQ), 26 or the presence of FoG during the clinical and balance and gait assessment (video reviewed by MM and JGN). In case of video assessment discrepancy, MM and JGN consulted and come to a consensus about a presence of the FoG. We categorized patients as PD + FoG based on either NFoG-Q >0 or an episode of freezing was observed during a laboratory assessment.
Clinical Measures
Motor severity was assessed using Movement Disorders Society Unified Parkinson’s Disease Rating Scale motor subscale scores (MDS-UPDRS-III). 27 The Montreal Cognitive Assessment (MoCA), 28 and Scales for Outcomes in Parkinson’s Disease-Cognition (SCOPA-COG) were used to measure global cognition. The mini-BESTest and its 4 subscores (APA, APR, Sway, and Gait) were assessed as a clinical measure of dynamic balance. People with PD were assessed in the practical Off levodopa state. “Practical Off state” refers to at least 12-hours overnight withdrawal of dopaminergic medication. Participants were assessed in their practical OFF state to measure mobility disability due to PD, with less influence of their medications, which can improve some aspects of gait but impair balance. 4
Digital Measures From Wearable Sensors
Objective measures of balance and gait were obtained using 8 wearable sensors (Opals by APDM Wearable Technologies, a Clario company); each sensor has triaxial accelerometers, triaxial gyroscopes, and magnetometers. The Opals were placed on both feet, shins, and wrists, sternum and lumbar (L5 region) area of participants, who performed a total of 8 different motor tasks, summarized in detail. 24 In summary, the 8 motor tasks included Sway tasks consisting of standing still for 30 seconds on a firm surface with eyes open or closed (EOFirm and ECFirm), and on a foam surface with eyes open (EOFoam); APRs task consisted of the push and release test in the backward direction; an Instrumented Stand and Walk test and a 2-minute walk test were used to obtain measures of APA and Gait, respectively. In addition, both APA and Gait task were performed with and without a concurrent cognitive task (dual and single task). The dual-task condition consisted of serial subtraction by threes from a 3-digit number, during both quiet stance and during the gait initiation (APA task), and reciting every other letter of the alphabet while walking for the Gait task. As outcome measures, we used the 24 objective measures that we found to be most sensitive in discriminating between people with PD and healthy controls. 24 The FoG ratio was calculated from the sensors on the shins according to methods described in Mancini et al. 29 Motor and cognitive dual task (DTC) were calculated as in our previous paper. 25 Specifically, when a dual task was added to walking, the dual cost (DC) was calculated as DC (%) = 100 × (dual-task measure–single-task measure)/single-task measure. In addition, the DTC was calculated using the same equation considering the percentage of correct answers in reciting every other letter of the alphabet while the subjects were seated for 2 minutes (ST) versus while the subjects performed the 2-minute walk. In a dual-task condition, no instructions were given on which task to prioritize.
Statistical Analysis
The distribution for each demographic and clinical measure of the 2 groups (PD + FoG and PD−FoG) was examined by the Shapiro–Wilk test at baseline. For data that were not normally distributed, the Mann–Whitney U-test was used to determine a difference between groups at baseline. Otherwise, independent samples t-test and chi-squared tests were used to examine possible group differences at baseline.
The 94 participants were randomly assigned into 2 groups (Exercise First: n = 46, Education First: n = 45; see cohort diagram in Jung et al. 25 ) Analysis for a linear mixed-model, repeated ANOVA, and standardized response mean (SRM) was performed on the 86 participants (PD + FoG = 44, PD−FoG = 42) who had at least 2 data points. As this study was a crossover design, the treatment effect represents whether the change (delta) during the Exercise intervention differs from the change during Education intervention. A linear mixed-effects model was used including an indicator of treatment effect (Exercise vs Education), order effect (Exercise-first vs Education-first), and period effect (sequence of assessments) to determine whether the “the change” differed between Exercise and Education. In this paper, we focus only on the exercise effects on PD + FoG/PD−FoG and not education.
To investigate whether the PD + FoG improved similarly to that of PD−FoG, we used a repeated measures ANOVA to compare differences before and after exercise in both groups (PD + FoG/PD−FoG). In addition, we also used ANCOVA to adjust for baseline differences between PD−FoG and PD + FoG in disease duration (as covariate), while avoiding collinearity. As disease duration and disease severity are correlated and between group differences were larger for disease duration than differences in MDS-UPDRS-III total scores between groups (see Table 1), here we decided to use the disease duration as a covariate. The significance level for this secondary analysis was set at P < .05.
Demographic and Clinical Characteristics Between PD−FoG and PD + FoG at Baseline.
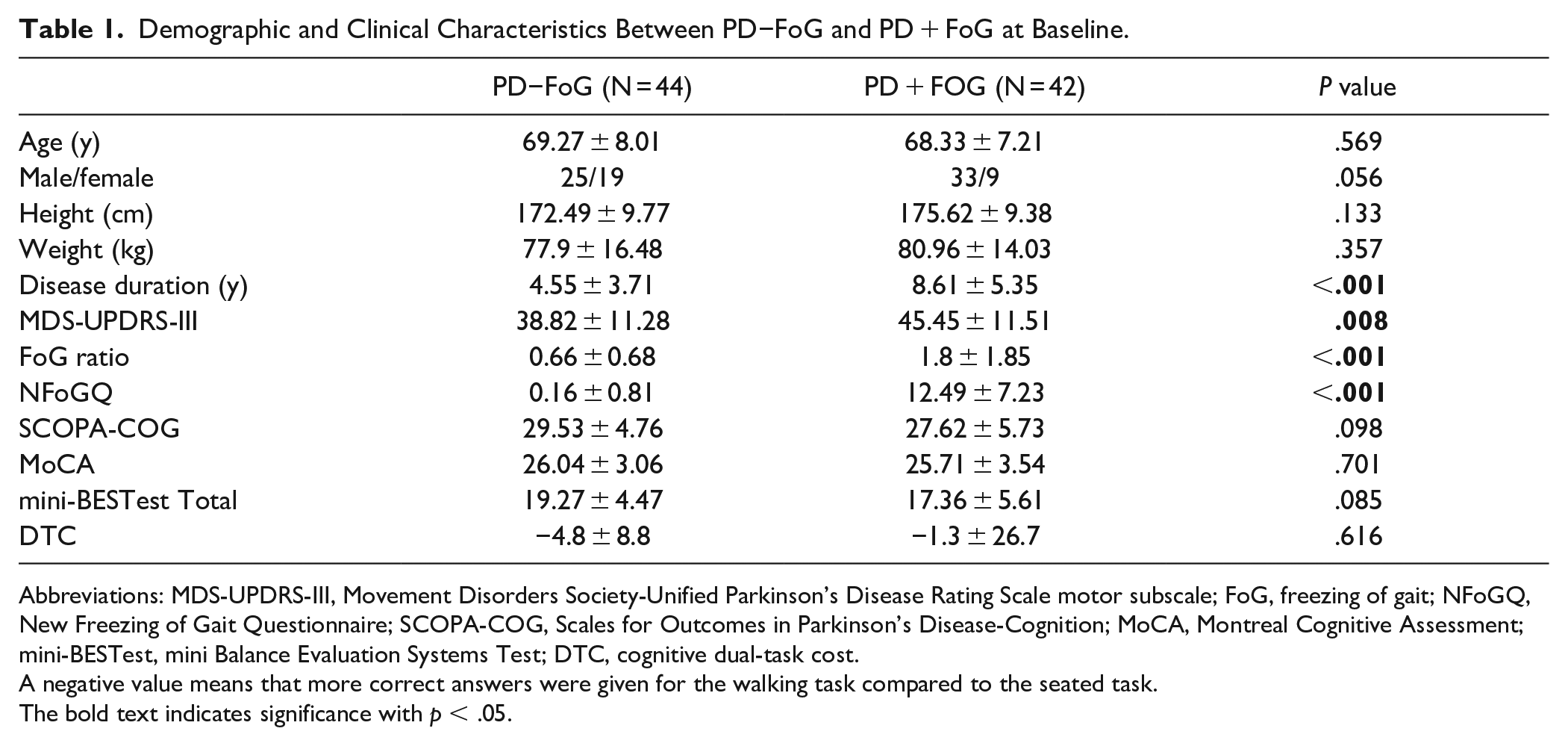
Abbreviations: MDS-UPDRS-III, Movement Disorders Society-Unified Parkinson’s Disease Rating Scale motor subscale; FoG, freezing of gait; NFoGQ, New Freezing of Gait Questionnaire; SCOPA-COG, Scales for Outcomes in Parkinson’s Disease-Cognition; MoCA, Montreal Cognitive Assessment; mini-BESTest, mini Balance Evaluation Systems Test; DTC, cognitive dual-task cost.
A negative value means that more correct answers were given for the walking task compared to the seated task.The bold text indicates significance with p < .05.
The effect of exercise was calculated as SRM for each group separately (PD + FoG and PD−FoG). The SRM was calculated as the mean change between before and after exercise divided by the standard deviation (SD) of the change. An SRM value of 0.20 represents a small, 0.50 a moderate, and 0.80 a large effect of the intervention. 30
The statistical analysis was performed in MATLAB R2018b (The Mathworks Inc., Natick, MA, USA) with the Statistics and Machine Learning Toolbox, and R Studio IDE Version 1.2.5019.
Results
Table 1 summarizes the clinical characteristics in the PD−FoG (n = 44) and PD + FoG (n = 42) groups. PD + FoG had more severe motor symptoms based on MDS-UPDRS-III score and longer disease duration. In addition, PD + FoG had higher freezing of gait index (FoG ratio) and higher NFoG score compared to PD−FoG. Lastly, PD−FoG and PD + FoG had similar cognitive DTC.
The linear mixed-model results showed that none of the 24 objective measures had a significant period effect. Hence, we combined the data for both groups (Education first and Exercise first) such that we have only 2 time points (before and after exercise) to perform repeated AN(C)OVA.
The repeated ANOVA showed that there is no interaction between time (before and after exercise) and group (PD + FoG and PD−FoG). The results of interaction remain the same after using disease duration as covariate. Seven of the 24 objective measures showed a significant effect of exercise irrespective of group. The repeated ANCOVA with disease duration as covariate showed a significant time effect in: reduced dual-task gait speed (P < .0001), increased single-task arm range of motion (P < .0001), increased single-task toe off angle (P = .005), reduced dual-cost of stride length (P = .012), decreased single-task gait cycle duration variability (P = .019), increased single-task trunk coronal range of motion (P = .042), and increased single-task stance time of gait cycle (P = .046) in both groups. Table 2 summarizes the measures where we observed a significant time effect either in ANOVA or ANCOVA. Figure 1 summarizes the measures where results of repeated ANOVA and ANCOVA both showed a significant time effect. The results of ANOVA and ANCOVA for all of the measures are given in the Supplemental Material S1.
The Results of Two-Way Repeated (Time: Before and After Exercise, and Group: PD−FoG/PD + FoG) AN(C)OVA. The Color-Coding Scheme Represents Different Conditions (Single-Task, and Dual-Task).
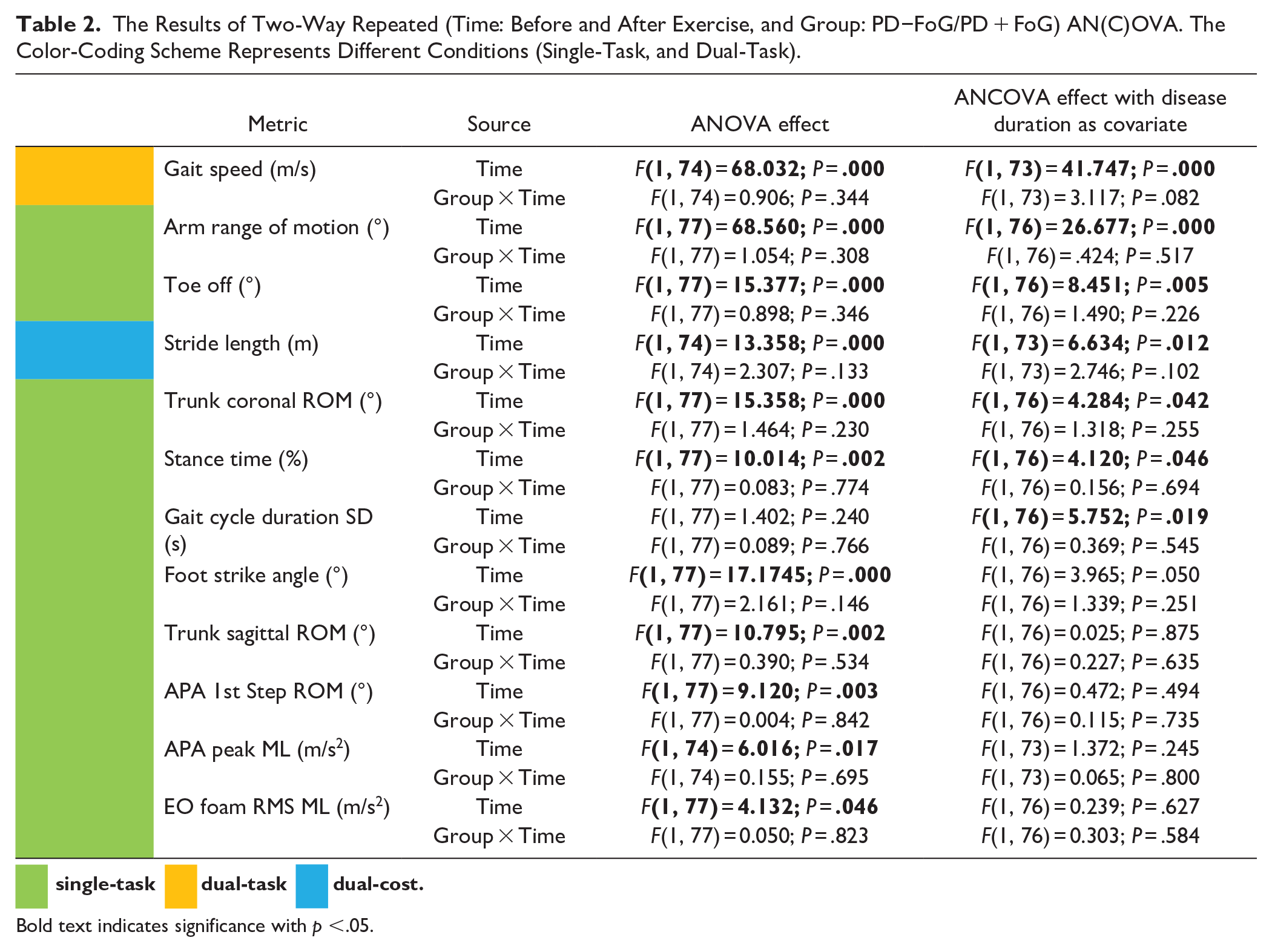



Bold text indicates significance with p <.05.
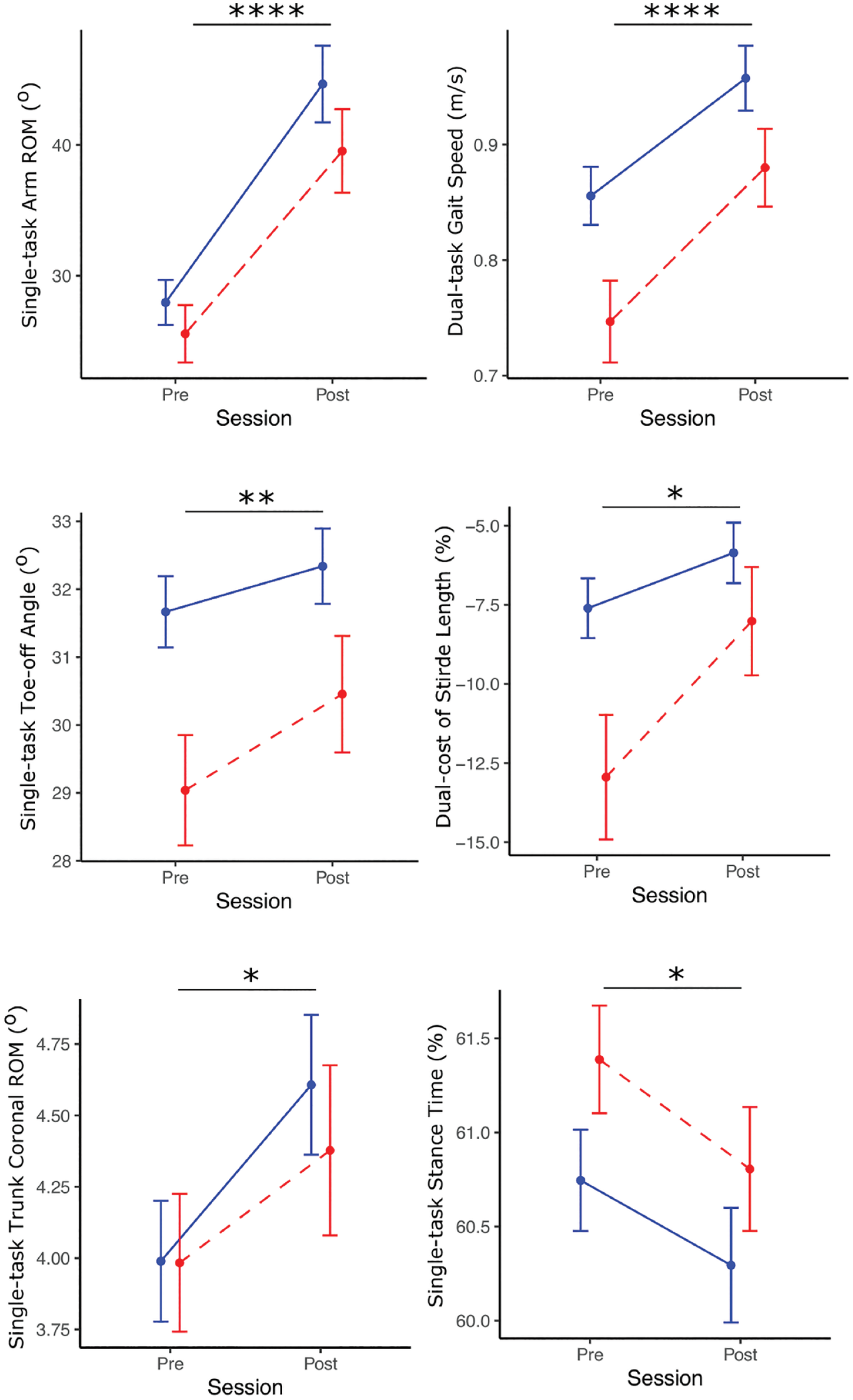
Gait measures before and after exercise program (mean ± SE) between PD−FoG (blue; n = 44) and PD + FoG (red; n = 42). Asterisks indicate a significant time effect (*P < .05, **P < .01, ****P < .0001) of ANCOVA analysis with disease duration as a covariate.
A large effect of exercise was observed in both dual-task gait speed and arm range of motion in PD + FoG and PD−FoG. Further, a moderate to small effect size was observed in 4 of the following measures in both PD + FoG and PD−FoG: trunk coronal range of motion; toe-off angle; stance time; and dual-cost of stride length. The top 3 objective measures with the largest effects from the ABC-C program in PD−FoG were: arm swing ROM (SRM = −1.01), dual-task gait speed (SRM = −0.88), and trunk coronal ROM (SRM = −0.55). Similarly, the top 3 objective measures with the largest effects from the ABC-C program in PD + FoG were: dual-task gait speed (SRM = −1.01), arm swing ROM (SRM = −0.86), and foot strike angle (SRM = −0.55). Table 3 summarizes effects of exercise on objective measures (for measures in Table 2) in both PD−FoG and PD + FoG. The results of SRM for all of the measures are given in the Supplemental Material S2.
Effect Size (in Descending Order of Absolute Standardized Response Mean (SRM) Value for PD−FoG) of the Exercise for Each Mobility Measure in PD−FoG and PD + FoG. The Color-Coding Scheme Represents Different Conditions (Single-Task, and Dual-Task).
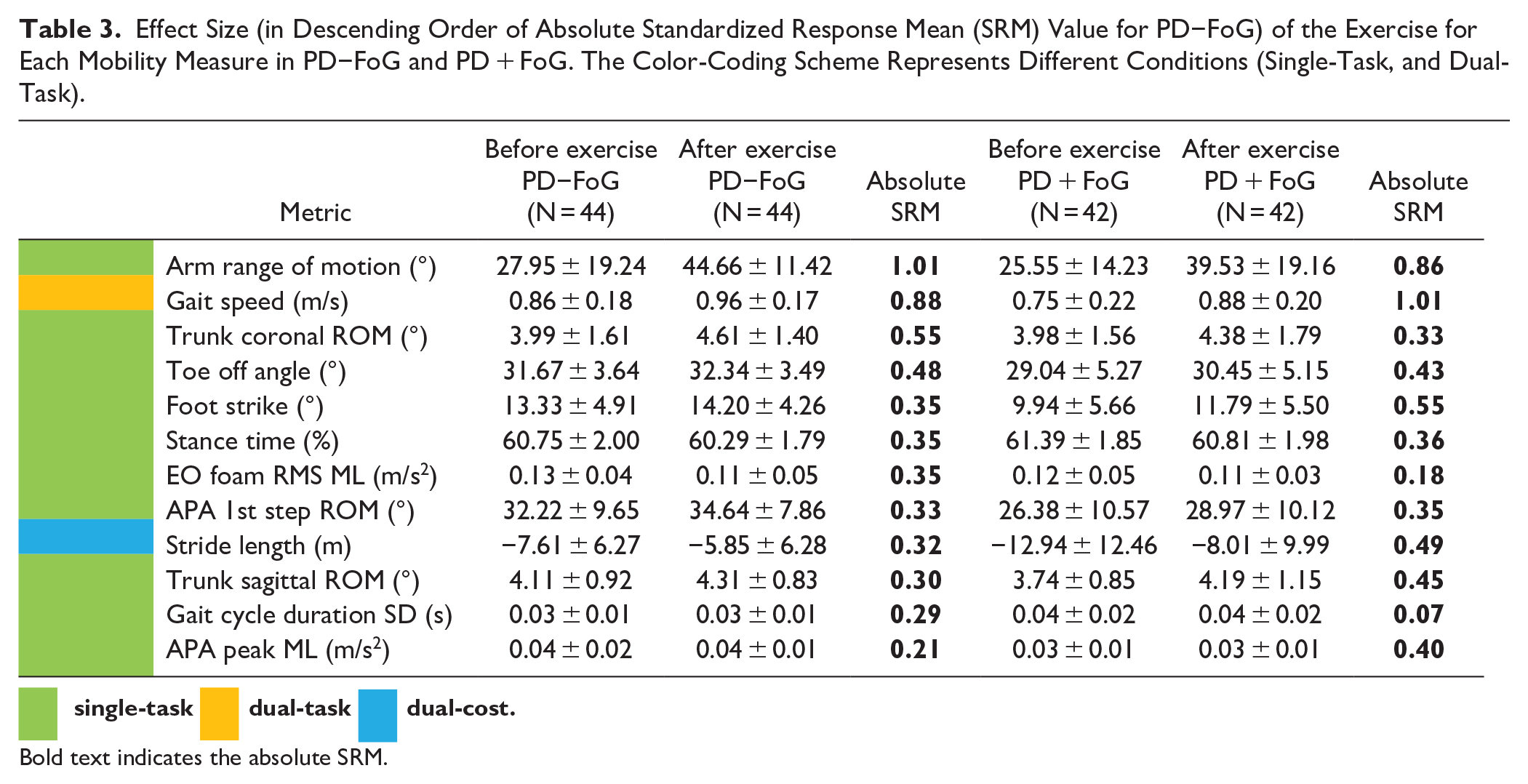



Bold text indicates the absolute SRM.
Discussion
Our findings showed that the small group exercise, the ABC-C program, was effective in improving gait performance (and not balance) in both PD + FoG and PD−FoG, even after controlling for disease duration. Specifically, dual-task gait speed, arm range of motion, toe-off angle, stance time, and dual-cost stride length improved in both the PD + FoG and PD−FoG groups. These findings indicate that both PD + FoG and PD−FoG respond similarly compared to the overall group reported earlier in our previous study. 24 These findings suggest that clinicians should consider adding exercise that simultaneously focuses on both motor and cognitive function when aiming to improve the gait performance in persons with PD, irrespective of whether an individual has FoG or not.
The largest effects of exercise were observed in both dual-task gait speed and single-task arm range of motion in PD + FoG and PD−FoG. For the dual-task gait speed, we were surprised to find that PD + FoG group not only improved in the ABC-C program but also showed a similar improvement to that of PD−FoG participants. This finding is consistent with Strouwen et al 21 where they found that PD + FoG and PD−FoG both showed a similar improvement in dual-task gait speed after a dual-task training. This finding is in contrast with other findings.17,31 Specifically, PD + FoG showed a less effective motor-learning capacity, 17 less effective implicit sequence learning capacity, 16 and an exaggerated loss of automaticity 31 compared to PD−FoG. Thus, similar improvement in dual-task gait speed suggests that PD + FoG can show even larger improvements of automaticity of gait compared to PD−FoG after the ABC-C program. In this study, the cognitive DTC while walking was similar between PD + FoG and PD−FoG and did not change after the ABC-C program, meaning that both groups were able to improve their motor DT after ABC-C at no expense of the cognitive performance. For the arm range of motion, both groups (PD + FoG and PD−FoG) benefited similarly from the ABC-C program. The increase in arm range of motion after an exercise intervention may indicate reduced burden on executive resources to control gait as we recently showed that this outcome measure best identified PD-specific dual-cost to gait. 32 It may also be due to a reduction in rigidity in both groups (PD + FoG and PD−FoG) as rigidity is known to be associated with a reduction in arm range of motion during gait. 33
Freezers showed improvements in dual-task gait speed and several gait quality measures despite the fact that the ABC-C program did not use external cues, such as music, metronome, visual cues on the ground, etc. External auditory, visual and proprioceptive cues have been shown to improve gait in freezers with PD.22,23 In fact, the ABC-C program, like the ABC program, was designed specifically to encourage the damaged basal ganglia to self-initiate larger, faster movements for mobility rather than substitute externally-initiated movements via alternative, cerebellar pathways. 9
When considering freezers and non-freezers as a separate group, the ABC-C program did not show significant changes in balance (measured by postural sway) in either group. Similarly, in our previous work by Hasegawa et al, 24 we observed smaller effect size in postural sway measures when considering the 2 subgroups as 1 group, after the ABC-C program. In addition, with the ABC program, we also observed smaller effect size in postural sway measures. 34 In contrast, other studies reported significant improvements in balance in both freezers and non-freezers with either digital sway measures, or clinical scales.35 -37 This discrepancy may be due to the medication status when assessing balance, in fact, it has been found that levodopa may worsen sway while standing. 4
The ABC-C program also did not improve in turning, and APR in either the PD + FoG nor PD−FoG groups. The non-significant effect of exercise on turning aspects may be due to the ABC-C program not specifically focusing on practicing turning, due to difficulty in maintaining safety with 3 to 6 subjects in the group exercise program, as previously discussed. 24 Similarly, the non-significant effect of exercise on APR may be due to relatively mild perturbations induced by boxing with a contact of gloved fist onto a padded hand during the ABC-C program, as previously discussed. 24
Perhaps not entirely surprisingly, when correcting the groups for disease duration, the following measures of gait, upper body, APA, and sway were no longer statistically significant: 1 gait (foot strike angle), 1 upper body (trunk sagittal range of motion), 2 APA (APA 1st Step ROM; APA Peak ML), and 1 sway (EO Foam RMS ML) measures. This result is consistent with longer disease duration reducing the effectiveness of exercise.16 -18
It is important to consider that our intervention was not designed specifically to improve FoG, but rather to improve automaticity of gait and balance performance using cognitive and motor tasks together. A recent systematic review 36 looked at studies that were either specifically targeted to FoG or aimed at the underlying correlates to FoG and found moderate improvements in FoG, while non-specific exercise programs had minimal to no effect on reducing FoG. Another recent systematic review reported that dual task training improved several measures of gait and balance but did not report on whether those people with FoG responded differently compared to people without FoG. 38 However, in a small non randomized feasibility study on dual task training, 39 it was noted that improvements in dual task gait performance were seen in all participants, regardless of the FoG severity.
There are several limitations to this study. First, although subjects underwent the exercise program in their “on” state, all outcomes were measured in their “off” state to exclude the effects of dopaminergic medication. This may have dampened the effects of the ABC-C program on daily life functional performance. Second, there was no washout period between the first and second intervention in this cross-over design, so we could not rule out possible carry-over effects from the first intervention. However, sequence and period effects were not significant. Third, we only included patients without any severe cognitive impairments that could interfere with their ability to follow testing procedures and who were in mild to moderate disease stages so results cannot be generalized to a more severe disease stage. Forth, we have used Cohen’s cutoff values for interpreting small, moderate, and large effect sizes from SRM which may lead to over- or underestimation of the magnitude, 40 therefore, we recommend caution when applying our findings until they are replicated. Finally, the intervention was limited to only 6-weeks. Hence, only the immediate effects of exercise were evaluated and we did not collect fall data in our subjects or have a follow-up period to investigate the long-term effect of the ABC-C program. Future studies should investigate the effects of ABC-C program on long-term benefits.
Conclusion
Our findings provide evidence that exercise simultaneously focused on both motor and cognitive function (the ABC-C program) can improve objective measures of gait (and not balance) and hence, be beneficial to both PD + FoG and PD−FoG. The ABC-C program shows a good potential for a wider clinical implementation in PD.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683221119757 – Supplemental material for Effects of a Cognitively Challenging Agility Boot Camp Program on Balance and Gait in People With Parkinson’s Disease: Does Freezing of Gait Status Matter?
Supplemental material, sj-docx-1-nnr-10.1177_15459683221119757 for Effects of a Cognitively Challenging Agility Boot Camp Program on Balance and Gait in People With Parkinson’s Disease: Does Freezing of Gait Status Matter? by Vrutangkumar V. Shah, Rodrigo Vitorio, Naoya Hasegawa, Patricia Carlson-Kuhta, John G. Nutt, Laurie A. King, Martina Mancini and Fay B. Horak in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors thank all participants for generously donating their time to participate, Peter Fino, Carolin Curtze, Mike Fleming, Heather Schlueter, Peter Martin, and Graham Harker for helping with data collection, Daniel Peterson and Katrijn Smulders for data collection and help with study procedures, and Edward King for helping with data collection and management.
Financial Disclosure/Conflict of Interest
OHSU and Dr. Horak have a significant financial interest in APDM Wearable Technologies, a Clario company, that may have a commercial interest in the results of this research and technology. This potential conflict has been reviewed and managed by OHSU.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: OHSU and Dr. Horak have a significant financial interest in APDM Wearable Technologies, a Clario company, that may have a commercial interest in the results of this research and technology. This potential conflict has been reviewed and managed by OHSU. All the other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Institutes of Health under award number NIA R01AG006457 (Horak), VA 5I01RX001075 (Horak), and Japan Society for the Promotion of Science 20K19371 (Hasegawa).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
