Abstract
Introduction. Reactive movements in response to a loss of balance are altered in people with Parkinson’s disease (PD) and are critical for fall prevention. Further, falls are more common while attention is divided. Although divided attention has been shown to impact postural responses in healthy older adults, the impact of dividing attention on reactive balance, and the natural prioritization across postural and cognitive tasks in people with PD is largely unknown. Objectives. To characterize (1) the impact of a secondary cognitive task on reactive postural control and (2) the prioritization across stepping and cognitive tasks in people with PD. Methods. Sixteen people with PD and 14 age-matched controls underwent step-inducing, support-surface perturbations from stance, with and without an auditory Stroop secondary cognitive task. Cognitive, neuromuscular, and protective stepping performance were calculated for single and dual task scenarios. Results. In PD and control participants, cognitive reaction times (P = .001) and muscle onset latency (P = .007), but not protective step outcomes (P > .12 for all) were worse during dual tasking compared with single-task scenarios. Both PD and control groups prioritized the protective stepping task over the cognitive task. Overall, people with PD exhibited worse first-step margin of stability (a measure of protective step performance) than controls (P = .044). Conclusion. This study provides preliminary evidence that people with PD, like age-matched controls, exhibit cognitive and neuromuscular, but not protective step, dual-task interference. The lack of dual-task interference on step performance indicates a postural prioritization for PD and healthy older adults during dual-task protective stepping.
Introduction
Balance impairments in people with Parkinson’s disease (PD) can increase risk of falls and disability, and reduce quality of life.1,2 In particular, compared with non-Parkinsonian peers, people with PD exhibit poorer automatic or “reactive” postural responses, defined as the quick movements in response to a balance challenge. 3 These reactive postural responses, which include protective stepping, are essential in preventing falls 4 and are smaller and less effective in people with PD compared with age-matched controls.3,5
While reactive postural responses occur quickly and rely heavily on brainstem regions,6,7 recent work demonstrates the important role of the cortex in this movement.8,9 For example, when young and older adults are exposed to a large postural perturbation while undergoing a cognitive secondary task, the neuromuscular response to the perturbation (ie, time from perturbation to muscular activity) and cognitive performance are worse when completing both tasks together.10-12
Data also suggest that when healthy adults are exposed to perturbations, they quickly redirect attention to the postural task, effectively prioritizing posture over the cognitive task. 12 However, whether people with PD prioritize posture or cognitive tasks under dual-task scenarios is less understood, with literature showing inconsistent results. For example, two reports13,14 suggested that during dual-task walking, people with PD disproportionately focus on the cognitive task, a strategy described as “posture second.” Contrasting these results, Smulders et al 15 did not observe this posture-second phenomenon during dual-task walking in a large sample of people with PD. Notably, these studies were all conducted while walking. Given the unique nature of reactive postural responses, dual-task prioritization dynamics may or may not behave similarly to continuous postural tasks such as walking in those with PD.
Together, previous work demonstrates the impact of secondary tasks on gait in people with PD, and on protective stepping in healthy adults. However, the impact of a secondary task on protective stepping in people with PD is largely unknown. To our knowledge, only one study examined this topic. Jacobs and colleagues 16 exposed people with PD and freezing of gait to quick movements of the support surface while completing a secondary cognitive test. Results showed that while adding a secondary cognitive task did not delay protective steps, dual tasking resulted in more frequent losses of balance. While this provides initial insights regarding dual-task protective stepping in people with PD, it did not address several key issues, including (1) how dual tasking affected protective step or center of mass kinematics, (2) performance on the secondary cognitive task, or (3) prioritization across cognitive and postural tasks. Given the ecological relevance of protective stepping for falls, understanding how PD affects protective steps and prioritization would provide important insights into functionally relevant deficits and potential rehabilitative targets.
Therefore, the purpose of this study was to (1) determine the effect of dual tasking on postural and cognitive performance and (2) characterize the natural prioritization across tasks in people with PD and age-matched controls. We hypothesize that adding a secondary task will result in worse postural and cognitive performance in both groups; however, postural control will be more affected in people with PD. Furthermore, given the possibility of a “posture second” approach toward dual tasking, 14 we hypothesize that people with PD, but not controls, will prioritize the cognitive task over the reactive postural response task.
Methods
Participants
A convenience sample of 16 people with neurologist-confirmed idiopathic Parkinson’s disease and 14 healthy older adults was recruited to participate in this study. Five of the individuals with PD self-reported experiencing freezing of gait in the previous month. 17 Exclusion criteria included the following: <55 years of age, presence of another neurological condition apart from Parkinson’s disease, and orthopedic injuries that could impede balance performance. People with PD were recruited via local support groups and through physician referral. Age-matched controls were recruited via local retirement communities. The institutional review board of Arizona State University approved the methods used in this study and all participants provided consent prior to participation.
Tasks
Participants experienced 3 types of trials: cognitive task alone (ie, single-task cognitive), protective stepping alone (ie, single-task step), and stepping with cognitive task (ie, dual tasking).
The cognitive task was an auditory Stroop task. Participants listened to the words “high” and “low” vocalized in high and low tones and were asked to ignore the word and verbally identify the stimulus pitch as quickly and accurately as possible. Stimuli were delivered via headphones every 3 seconds. Order of stimuli were pseudo-random, and the same sequence was provided to all participants.
For the protective stepping task, participants underwent a quick movement of the support surface. Support-surface movements were delivered via an instrumented treadmill (CAREN system, Motecforce Link) via a ramp, hold, and deceleration protocol (constant acceleration for 300 ms, constant velocity for 500 ms, constant deceleration for 300 ms to stop). 3 Prior to perturbations, participants were instructed to stand naturally but still, looking forward without anticipating the timing or direction of the upcoming perturbation. For all trials, participants were instructed to take as few steps as possible after the perturbation. A minimum of 20 seconds passed between each trial, and this time was varied to prevent temporal anticipation of the perturbation. Direction of perturbation was pseudo-randomly ordered to prevent spatial anticipation of the perturbation (see “Protocol” for additional details).
For dual-task perturbations, participants were instructed to stand relaxed while responding as quickly and accurately as they could to the auditory stimuli. They were told that a perturbation may occur and, if it does, react naturally. No instructions were given regarding how or where to focus attention across tasks. For each trial, participants were exposed to 8 consecutive auditory Stroop stimuli. One support surface perturbation was released during Stroop stimuli 5, 6, 7, or 8 (pseudo-randomly distributed across trials). Support surface perturbations occurred just after delivery of the relevant auditory Stroop stimulus. On average, the support surface translation occurred 0.33 seconds (range −0.098 to 0.885 seconds) after the onset of the auditory stimulus. Although precise synchrony between perturbations and stimuli was not achieved due to technical challenges, no differences in the degree of synchrony were observed across groups (P = 0.52). Furthermore, there were no significant correlations between stimulus synchrony and outcomes of interest (P > .15; r2 < 0.03 for all; data not shown). Perturbations were delivered and synchronized to cognitive stimuli via D-Flow software (Motecforce Link).
Protocol
Throughout the article, we will describe perturbation direction with respect to the direction of the resulting lean and step (ie, backward perturbation = forward slide of the support surface and a backward step, forward perturbation = backward slide of the support surface and a forward step). The protocol is outlined in Table 1. Participants were first exposed to 4 single-task, backward stepping perturbation trials to orient them to the task and reduce potential “first trial” effects. This was followed by 3 single-task cognitive trials (11 stimuli each) to practice the cognitive task. Data from these initial stepping and cognitive trials were stored but not included in the current analysis.
Study Protocol.

Data collected but not analyzed.
Participants then completed a single-task standing cognitive trial (11 auditory Stroop stimuli), followed by seven single-task protective-step trials. During the single-task stepping block, there were 5 backward perturbations and 2 forward perturbations in randomized order to prevent participants from predicting the direction. Only backward stepping trials were included in the current analyses. This study focused on backward perturbation trials since people with PD have more difficulty with a backward loss of balance than forward.18,19 Finally, participants completed one block of dual task stepping (7 trials, 5 backward, 2 forward), in which they completed both stepping and cognitive tasks simultaneously. As noted above, for each of these dual-task stepping trials, 8 auditory stimuli were presented. Critically, the percentage of incongruent auditory Stroop stimuli across single and dual task blocks were similar (73% and 75%, respectively).
After perturbation trials, participants completed clinical assessments, including the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS Part III), New Freezing of Gait Questionnaire, 17 Mini-Balance Evaluations Systems Test (MiniBESTest), Activities of Balance Confidence Scale (ABC), and Montreal Cognitive Assessment (MoCA). People with PD were examined “On” medication, with data collections beginning approximately 1 hour after their most recent medication dose.
Data Collected
Kinematic data were collected via optical tracking of a modified full-body Helen Hayes marker set 20 at 200 Hz (Vicon Corp). Specifically, markers were placed on the head (3; top, front, back), trunk (3; bilateral shoulders and offset), pelvis (5; sacrum and bilateral anterior superior iliac spine/posterior superior iliac spine [ASIS/PSIS]), and bilateral thigh (4; greater trochanter, front thigh, medial/lateral condyles), shank (3; front shank, medial/lateral malleoli), foot (2; toe and heel), and arms (2; elbow and wrist). Marker data were low-pass filtered at 15 Hz prior to outcome calculation. Electromyographic (EMG) electrodes were placed on the tibialis anterior and gastrocnemius of both legs. EMG data were and collected at 2000 Hz, and high-pass filtered at 20 Hz, de-meaned, and full-wave rectified. A microphone was utilized to record participants’ vocal responses. Microphone data and auditory stimulus data were also recorded at 2000 Hz. Microphone data were de-meaned, full-wave rectified, and low-pass filtered at 30 Hz. All data were collected in and synchronized using Vicon Nexus 2.2. Finally, oral responses during the auditory Stroop test were also hand recorded to measure accuracy of responses.
Outcomes
One primary outcome was selected for each of 3 domains: cognitive, neuromuscular, and protective stepping. The primary outcome variable for the cognitive task is reaction time: time between initiation of the auditory stimulus and when the participant vocally responded. The primary neuromuscular outcome was the time between movement of the support surface (perturbation onset) and onset of the tibialis anterior. For each trial, the fastest value from the left or right tibialis was recorded. Tibialis anterior was chosen as it is the agonist muscle for re-stabilizing balance during a backward fall. For both cognitive and EMG reactions, “onset” was identified as the moment that the signal increased above 2 standard deviations of the mean, where signal variability was determined over the first 2 seconds of data collection. Both outcomes were calculated by custom Matlab software. The primary protective stepping outcome was first step length, described as the distance between the left and right toe at the moment of first foot contact. Secondary outcome measures for protective stepping included: step latency, step width, and anterior-posterior margin of stability (MOS) at first foot contact. MOS is defined as the distance between the base of support and the extrapolated center of mass (COM), where the extrapolated COM is the sum of the position and velocity of the COM. 21 For the current study, COM was approximated as the 3-dimentional position of the trunk, utilizing bilateral ASIS and PSIS, and shoulder markers (for details, see 22 ). Finally, the secondary outcome variable for cognitive performance is the number of correct responses. Although not a primary outcome for this analysis, the percentage of correct responses were calculated across group and task to confirm reliable and interpretable data from cognitive response times.
For all outcomes, dual task interference was calculated via the following equation to represent the percent change in performance from single to dual task: Dual task interference (DTI): 100 × (Single Task − Dual Task) / Single Task. 23 Prioritization was also assessed to determine whether DTI was more prominent for the cognitive or postural task. This outcome was calculated as cognitive task DTI minus postural task DTI. As such, one prioritization score was calculated for each postural task. Negative values reflect postural prioritization, and positive values reflect cognitive prioritization.
Statistical Analysis
The primary analysis for this study utilized the averaged outcomes from (1) the standing single-task cognitive trial, (2) the 5 backward single-task step trials, and (3) the 5 backward dual-task step trials. To determine how cognitive and postural performance changes when moving from single to dual-task scenarios, a mixed model repeated-measures analysis of variance (ANOVA) was utilized with task (single, dual) and group (PD, control), as the 2 factors.
To statistically assess task prioritization, a single-sample t test was run on each prioritization score to determine if means were statistically different from zero. Alpha was set to P = .05 for all analyses.
Shapiro-Wilk tests assessed normality of data. In the case of nonnormally distributed data, log transformations were run to reduce skewness, and parametric statistical assessments run. MOS prioritization data were observed to be skewed due to the value of one outlier (a PD participant). The prioritization value for this participant was >3 times that of the next largest value. Prioritization data were run with and without this participant and no changes in statistical significance were observed. Therefore, this value was excluded from the analyses presented below.
Results
Participant demographics are presented in Table 2. Only the ABC was observed to be statistically different across groups, indicating that this PD cohort was relatively mild.
Participant Characteristics for Parkinsonian (PD) and Control (HC) Particiants. a
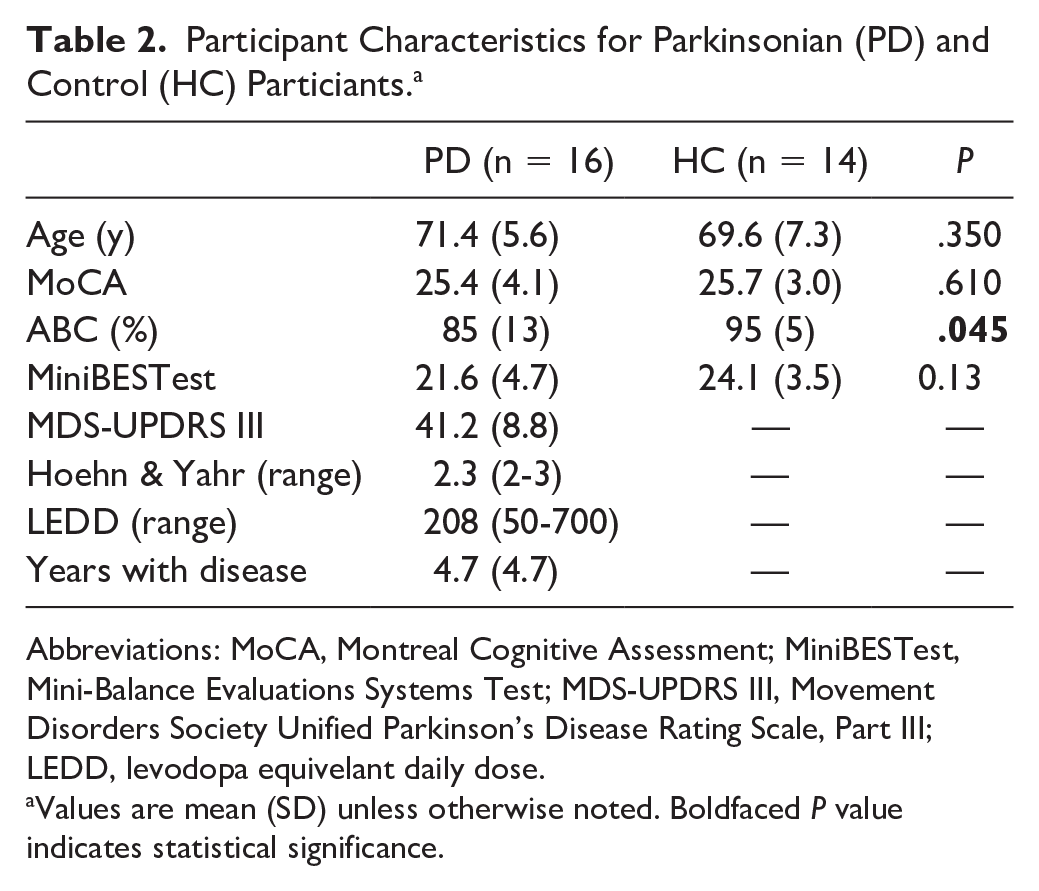
Abbreviations: MoCA, Montreal Cognitive Assessment; MiniBESTest, Mini-Balance Evaluations Systems Test; MDS-UPDRS III, Movement Disorders Society Unified Parkinson’s Disease Rating Scale, Part III; LEDD, levodopa equivelant daily dose.
Values are mean (SD) unless otherwise noted. Boldfaced P value indicates statistical significance.
Impact of Dual Tasking on Task Performance
Results of all ANOVAs are reported in Table 3. EMG data for 2 control participants were corrupted. Therefore, 12 controls were included in EMG analyses. All other analyses included the full dataset of 16 PD and 14 HC. Compared with performance during the single-task scenario, verbal (cognitive) reaction times and muscle onset latency were significantly worse (ie, later) while dual tasking (P = .001 and P = .007, respectively, Figure 1a and b). No significant dual-task effects were observed in any of the stepping performance outcomes (P > .12 for all). There was a main effect of group on MOS, such that people with PD exhibited smaller (worse) MOS across both tasks on average (P = .044; Figure 1d). Finally, there was a group by task interaction for step width, such that controls took more narrow steps during dual tasking compared with single task, but people with PD did not (P = .03; Figure 1f). Although not a primary outcome, accuracy on cognitive Stroop performance was observed to not be statistically different across groups during single-task (P = .43) or dual-task (P = .967) trials. Accuracy was however reduced from single to dual tasking for PD (P = .003) and HC (P = .021) groups. Mean (SD) accuracy across all subjects during single- and dual-task trials was 92.2% (13.5%) and 77.0% (26.0%), respectively.
Means, Standard Deviations, and Statistical Outputs (F, P, and partial eta-square [
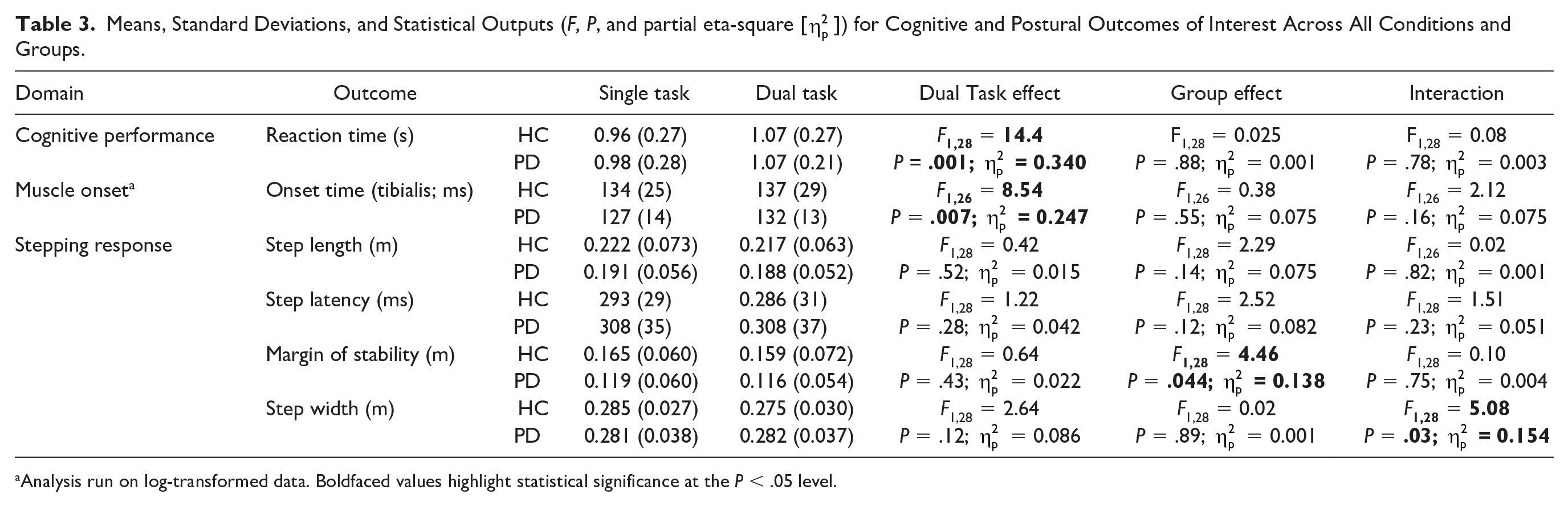
Analysis run on log-transformed data. Boldfaced values highlight statistical significance at the P < .05 level.
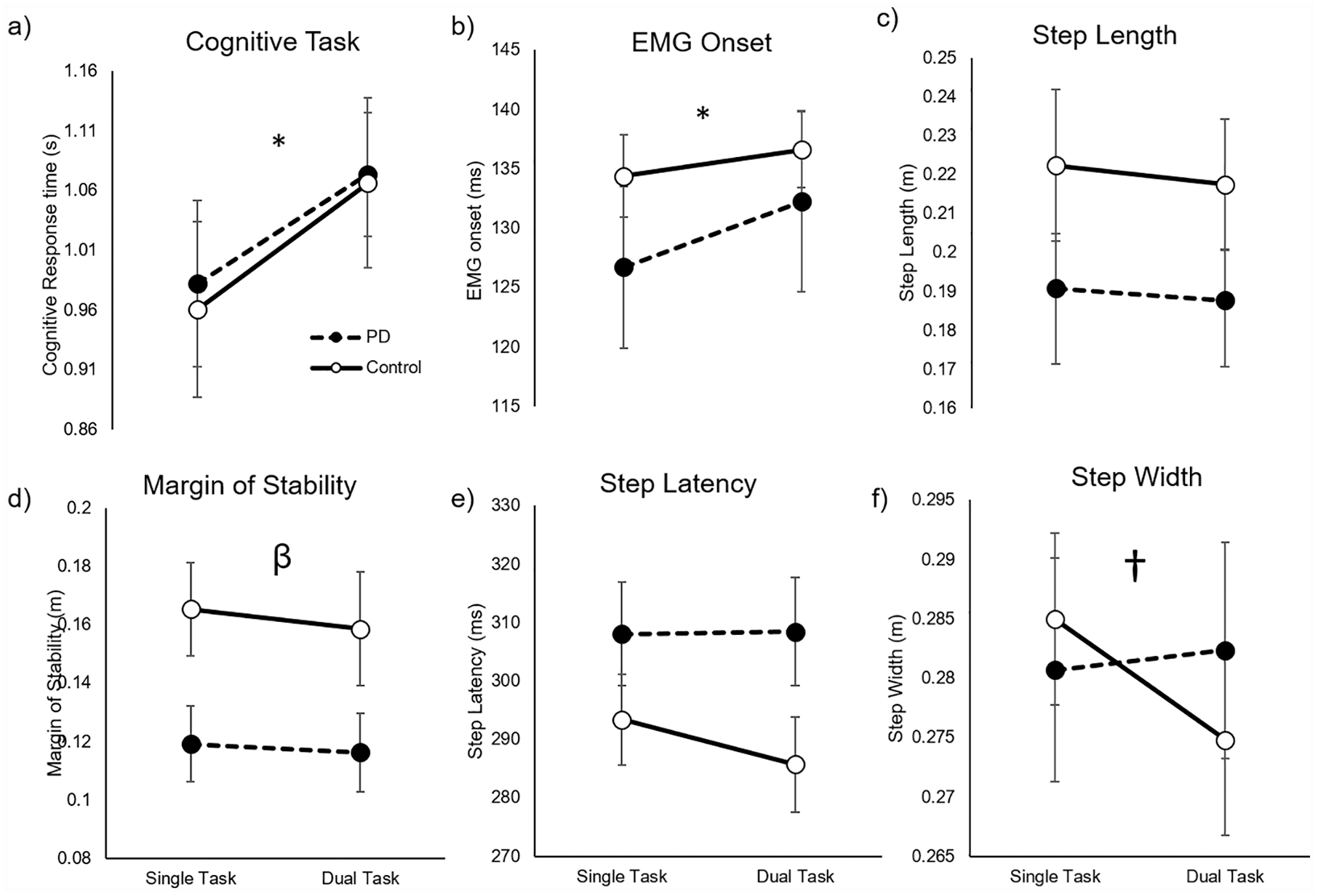
Effects of dual tasking on the 3 primary outcomes—cognitive performance (a), muscle onset time (b), and protective step length (c), as well as secondary protective stepping outcomes (d-f). Significant task (*), group (β), and interaction effects (†) are indicated.
Prioritization
Prioritization was characterized as the difference in cognitive DTI and DTI of each postural outcome (muscle onset and step outcomes). For both groups, postural prioritization was observed for all outcomes. Specifically, there was a statistically significant postural prioritization when cognitive DTI outcomes were compared with muscle onset (P = .003), step length (P = .011), latency (P < .001), and step width (P = .001). For MOS, there was a nonsignificant postural prioritization (P = .083, Figure 2a). The relative postural prioritization can be qualitatively observed when cognitive DTI and postural (EMG) DTI data are plotted together for all participants (Figure 2b).
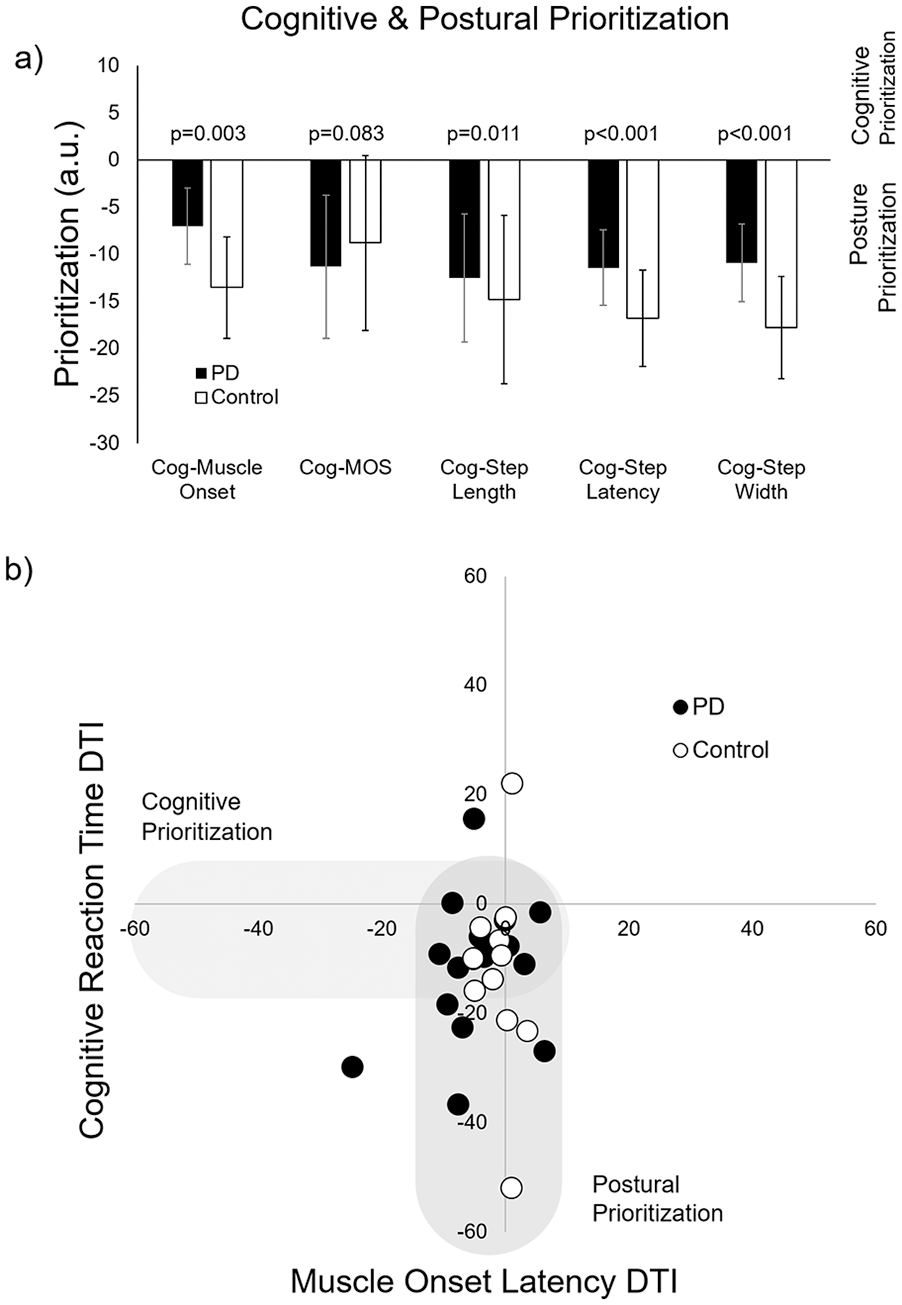
Cognitive and postural prioritization: (a) Prioritization between cognitive (verbal reaction time) and each postural response outcomes. Negative values reflect posture/step prioritization, and positive values reflect cognitive prioritization. (b) Scatter plot for all participants showing dual task interference (DTI) for the cognitive task (y-axis) and muscle onset latency (x-axis). Participants largely cluster along the negative y-axis, suggesting more prominent step prioritization. Shaded regions represent areas that likely reflect postural or cognitive prioritization (see also: Plummer and Eskes 23 ).
Discussion
The main objective of this study was to provide a preliminary characterization of dual-task protective stepping in people with PD. Specifically, we sought to understand (1) the effects of dual tasking on postural and cognitive performance and (2) prioritization of the cognitive and postural tasks. Three findings address these objectives. First, we observed that, similarly to controls, people with PD exhibit significant delays in both cognitive reaction time and muscle onset time when completing dual-task protective stepping, reflecting DTI of both the cognitive and postural tasks. However, second, protective stepping outcomes (step length, latency, width, and MOS at first foot contact) were not consistently affected when a secondary cognitive task was added. Third, similarly to controls, people with PD prioritize postural control over cognition during protective-stepping dual tasking scenarios. These findings extend our knowledge regarding how secondary tasks impact protective stepping in people with mild PD.
Previous work in young and older adults suggests that when a secondary cognitive task is added to a large, protective-step inducing postural perturbation, the earliest aspects of the automatic postural response, measured as the latency of the muscular onset after perturbation, and performance on the cognitive task are delayed.10,12 Notably, although the amount of delay in muscle onset is consistent and statistically significant, this change is small in magnitude, typically <10 ms. 12 ]. Furthermore, evidence suggests that this automatic postural response may occur prior to a decrease in attentional focus on the secondary cognitive task.24-26 This suggests that although a secondary cognitive task likely does have an impact on the initial aspects of reactive postural control, the impact is small in magnitude. Our results are consistent with these findings, extending previous work to show that people with PD also exhibit DTI in cognitive performance and muscle onset latency, although, as with healthy controls, the magnitude of this neuromuscular response delay is small (see Figure 1b).
Literature surrounding the impact of a secondary cognitive task on later aspects of reactive postural control (eg, COM control and protective step characteristics) is mixed. When secondary cognitive tasks are added to small to medium perturbations, eliciting in-place (ie, non-stepping) responses, center of mass displacement is often negatively affected.24-27 Interestingly, however, when larger-magnitude perturbations are used, the resulting protective step is not consistently impacted by a secondary dual task.12,28,29 For example, 2 studies,12,28 showed little to no impact of a secondary task on step outcomes in stroke survivors and age-matched controls, respectively. Zettel et al 29 showed that in older adults, only mediolateral anticipatory postural responses prior to the protective step was affected by dual tasking. Our findings are consistent with these previous results, as protective-step outcomes were largely unaffected by during dual tasking. Of note, Patel et al 10 observed contradicting findings, as in this study a secondary task did affect protective step outcomes (step length, latency and margin of stability) in healthy young adults after a large external perturbation. 10 The authors suggested that negative findings of previous studies may have been related to relatively small perturbation size.
A second goal of this study was to understand prioritization across multiple tasks in people with PD during protective stepping. Previous studies investigating dual task walking in people with PD suggested that this population may prioritize cognition over gait, known as a “posture second” strategy. 14 This approach, if true, would be problematic in fall-risk populations, particularly during reactive postural response situations. However, previous literature surrounding the posture-second approach in people with PD has focused on dual tasking during walking, and even then, support for a “posture second” strategy is mixed. 15 As noted above, our data suggest that during a protective stepping dual-task scenario, people with PD do not exhibit a robust posture second strategy. Instead, people with PD and controls seemed to prioritize the protective stepping task over the cognitive task. This is consistent with previous results demonstrating that healthy older adults quickly redirect attention to the protective stepping task, delaying responses to the cognitive task rather than the postural control task. 12 Notably, severity of PD participants in the current study was relatively mild. Additional research should be conducted to confirm the current finding in people with more severe PD. In particular, studies with larger samples and participants across a breadth of cognitive capacity should be conducted, as cognition may contribute to prioritization changes across control and Parkinsonian populations.
The lack of an impact of PD on cognitive or postural DTI is intriguing, and contrary to our hypothesis. Previous work indicates that PD has a pronounced effect on the ability walk while completing a secondary task, 30 likely due to reduced automaticity of gait. However, although protective steps may be generally worse in people with PD compared to controls 3 (a finding that was partially supported in the current study, see Figure 1d), the ability to quickly release a reactive postural control movement seems to be relatively intact. Said differently, PD did not seem to significantly affect the ability to appropriately prioritize and quickly redirect attention toward the postural perturbation via release of a protective step. Although these findings are preliminary, they beg the question of why, despite the lack of impact of PD on these outcomes, this population experiences increased falls compared with age-matched controls. In addition to the highly complex nature of falls, the altered ability to produce quick steps generally, regardless of presence of a secondary task, may increase fall risk in people with PD. Furthermore, changes in the ability to release protective postural movements after internal perturbations (ie, inappropriate weight shifts) may also contribute to increased fall risk. 31 Additional work will be necessary to clarify the impacts of dual tasking on protective stepping, particularly in neurological populations.
Several limitations should be noted. First, a small number of participants were included in this preliminary study, and additional work with larger samples should be conducted. Second, we included a relatively small number of trials in our single- and dual-task assessments. This approach was chosen to reduce the chance of learning effects (as this question was out of the scope of the current study) and fatigue. However, it could reduce the robustness of our conclusions. Finally, our PD cohort was relatively mild, potentially reducing generalization to a more severe cohort.
Conclusions
This work provides preliminary evidence that people with mild PD respond similarly to dual-task scenarios to controls, demonstrating delayed cognitive and muscle onset, but not worse stepping, during dual task protective stepping compared with single-task protective stepping. In addition to improving our knowledge regarding dual tasking in people with PD, this work may have clinical implications. For example, we observed dual task interference in cognitive performance in all participants. Although the effects of dual task gait and balance training are mixed in people with PD, 32 future studies could target this outcome via dual-task protective step training. Of note, recent pilot studies have demonstrated that single-task protective stepping can be improved through practice in people with PD. 33
Footnotes
Acknowledgements
We would like to thank the participants for donating their time to facilitate this study. We also thank Anandita Nadkarni, Joseph King, Nicole Elms, Xi Zong, and Jenna Martinez for their assistance with data collection and processing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Arizona State University and Mayo Medical Center Pilot Seed Grant
