Abstract
Introduction
Persons with Parkinson’s disease (PD) report prevalence rates of 35% up to 90% of yearly falls. 1 Postural instability, loss of postural reflexes, and freezing of gait (FOG) were found to be the main risk factors for falling, and these jointly explain approximately 80% of all falls.2,3 FOG is defined as “a brief, episodic absence or marked reduction of forward progression of the feet, despite the intention to walk.” 4 Prevalence rates vary from about 27% in the very early stage up to 86% in more advanced disease.5,6 A meta-analysis of clinical balance scale outcomes, showed more severe postural instability in PD patients with FOG (FOG+) compared with those without (FOG−), irrespective of medication use. 7 Also, balance deterioration was more apparent in those who converted to developing FOG. 8 However, the precise nature of postural instability in FOG+ is still unclear. Consequently, it remains difficult to design specific balance training programs for this subgroup of patients.
Single task studies investigating differences in quiet stance under normal and challenging postural conditions between FOG+ and FOG− reported variable results. Manipulating proprioceptive information resulted in more postural instability in FOG+ compared with FOG−,9,10 but the opposite was also found. 11 Restricting visual information during upright stance increased postural instability more in FOG+ than in FOG−9,12 but others found contradictory effects.10,11 Overall, the inconsistent results hamper a robust interpretation of the underlying deficits of postural instability in FOG+.
In older people, postural control is affected by dual-tasking, suggesting that the processing of postural control becomes more cognitively and less automatically controlled due to age-related degenerative processes. 13 As a result of basal ganglia dysfunction, PD patients may compensate even more by using attentional mechanisms to keep a stable posture. 14 Similarly, FOG was associated with an exaggerated loss of motor automaticity. 4 A recent study from our lab showed that dual-task (DT) gait deterioration in FOG+ was associated with resting state neural network changes indicative of an imbalance between motor and cognitive control networks.15,16 So far, only 3 studies investigated the effects of dual-tasking during stance in relation to FOG.12,17,18 One study perturbed balance in FOG+ and healthy controls (HCs), but a comparison with FOG- was lacking. 17 In this study, dual-tasking increased the incidence of falls without affecting the occurrence of freezing episodes. 17 A second study investigated DT-effects in quite stance during visual restrictions only, showing more abnormal sway patterns in FOG+ compared with HCs as indicated by center of pressure outcomes. 12 However, DT interference was not measured. The most recent study used body-worn sensors to investigate postural sway during 30-second stance as well as in other mobility tasks in PD subgroups without including a healthy reference group. No differences in DT cost between FOG+ and FOG− were found in stance unlike in the walking tasks. 18
To address the incomplete picture on freezing-related DT effects on balance, we will investigate whether postural stability is differentially compromised by additional cognitive load in PD subgroups and HCs. We will analyze by both center of pressure and center of mass metrics, the latter to better capture whole body motion. We hypothesized that due to reduced motor automaticity, FOG+ would exhibit greater postural instability during DT compared with ST (single task) and this more so than in FOG- and controls. Additionally, we expected higher cognitive DT cost in FOG+, particularly during unstable surface conditions.
Methods
Subjects
Thirty-three subjects with PD and 28 HCs participated. Patients were categorized as FOG+ (n = 19) if they scored 1 on item 1 of the new Freezing of Gait Questionnaire (NFOG-Q) or FOG− (n = 14) if they scored 0 on this item. All participants met the inclusion criteria of a score >23/30 on the Mini Mental State Examination (MMSE) and were able to stand upright for at least 15 minutes. Inclusion criteria for PD were: diagnosis of PD based on the UK Brain Bank Criteria, Hoehn and Yahr (H&Y) stage II or III (in ON-medication) and on stable medication for the past month. Exclusion criteria comprised having a neurological disorder other than PD, vestibular disorders, musculoskeletal disorders, or any other disease that could interfere with the experimental task. PD patients with a deep brain stimulator were also excluded. Patients were tested during the ON-medication state. Participants were recruited via the Movement Disorders Clinic of the University Hospital Leuven and the PD database of the Department of Rehabilitation Sciences. Screen fails were not recorded and recruitment was based on a pragmatic strategy (nonconsecutive). All participants gave their written consent according to the Declaration of Helsinki. The study was approved by the local ethics committee.
Procedure
Clinical Assessment
Preceding the different tests, demographic data of participants were collected. Cognitive examination consisted of the MMSE and the Montreal Cognitive Assessment (MoCA). The Mini Balance Evaluation System Test (Mini-BEST) was used as a clinical test to assess postural control. 19 PD severity was determined using the Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part III (motor examination) and the H&Y staging criteria.
Posturography
Three-dimensional marker positions were recorded using 7 infrared cameras (Vicon, Oxford Metrics, Yarnton, UK, 100 Hz). Reflective markers were attached to the body on specific anatomical landmarks, using an adapted version of the Liverpool John Moores University model 20 (Supplementary Material 1, available in the online version of the article). Simultaneously, ground reaction forces were recorded using two force plates embedded in a movable Computer Assisted Rehabilitation Environment (CAREN) (Motek Medical BV, Amsterdam, the Netherlands, AMTI, Watertown, USA, 1500 Hz). All subjects wore a safety harness to prevent falling. Also, at least 2 testers were present during the entire assessment. One tester performed the recordings, while the other stayed near the participant during the whole experiment.
Single Task Postural Paradigm
Subjects were asked to maintain a quiet standing position on a stable surface, with eyes open (EO) and eyes closed (EC) and on an unstable foam surface with EO and EC. Each condition lasted for 40 seconds, 20 seconds with EO and 20 seconds with EC after a visual sign was given. Data from second 1 till 19 and second 22 till 40 were used for further analysis. Sufficient rest in between trials was provided, according to the need to diminish the effect of fatigue.
Dual-Task Paradigm
Each postural condition was also executed while performing a cognitive DT. The DT consisted of the forward digit span. Participants were asked to repeat sequences of seven numbers, heard through a headphone while maintaining stability in standing. This task was chosen as a pilot study revealed cognitive loading in a continuous fashion throughout the trial. First, the forward digit span was performed while sitting, to assess baseline cognitive capacity in single-task (ST). Surface and ST/DT conditions were executed in random order. Each condition was performed 3 times. Patients were given the instruction to divide their attention equally over both tasks to ensure optimal performance in each.
Data Analysis
Postural Data
Data were processed using Matlab to calculate the center of pressure (CoP) and center of mass (CoM). Force data were first filtered using a low-pass fourth-order Butterworth filter at a cutoff frequency of 50 Hz before the combined CoP was calculated. To evaluate postural stability, the following variables were calculated from CoP and CoM data, averaged over the trials: (1) sway velocity, (2) 95% confidence ellipse area of the sway path length, (3) maximal excursion, and (4) root mean square (RMS). Excursion was defined as the maximal amount of postural sway in the anterior-posterior (AP) or medial-lateral (ML) direction. Sway velocity described the participant’s movement speed of the total sway path length. The RMS was calculated to define variability of performance. The confidence ellipse area resembled the size of the ellipse containing 95% of the movement path. We considered sway velocity and the ellipse area as general parameters, describing more postural instability when larger. 21 Excursion and RMS were extracted to indicate direction-specific differences.
Dual-Task: Forward Digit Span
Successful performance on the forward digit span was expressed as a percentage of the correctly repeated numbers.
Dual-task cost (DTC) was calculated using the following formula:

Depending on the outcome measure, a positive (cognitive DTC) or negative multiplier (postural DTC) was added. A higher DTC indicated deterioration of performance in the DT condition.
Statistical Analysis
Statistical analysis was performed using IBM SPSS software (version 22). Demographic characteristics between patient groups were compared using one-way and Friedman analysis of varaince, independent
Results
Demographics
Subject characteristics are shown in Table 1. FOG+ and FOG− were well-matched for all demographic and disease-related parameters. However, HCs were significantly older compared with FOG+ (Δ7.5 years,
Participants’ Demographics. a
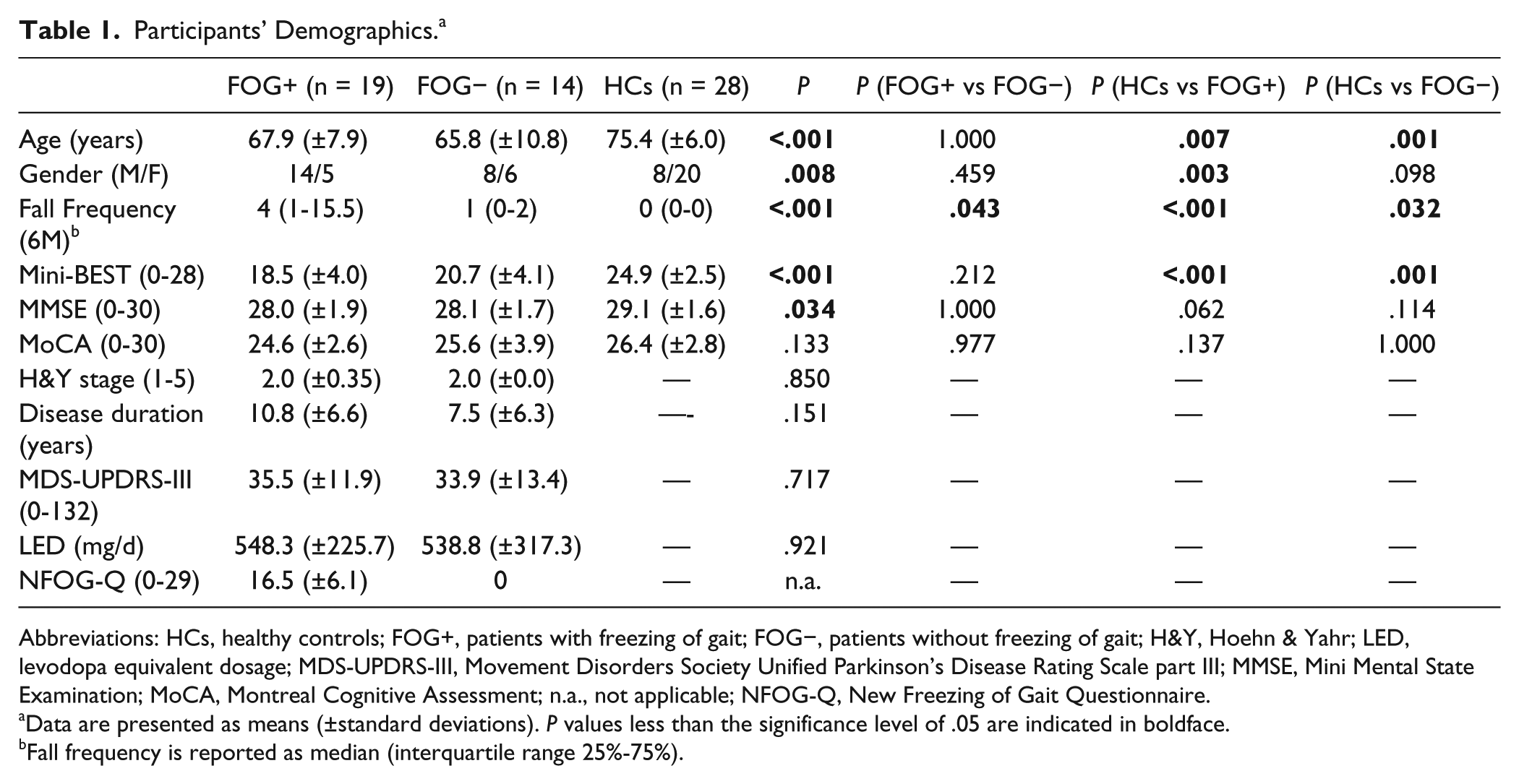
Abbreviations: HCs, healthy controls; FOG+, patients with freezing of gait; FOG−, patients without freezing of gait; H&Y, Hoehn & Yahr; LED, levodopa equivalent dosage; MDS-UPDRS-III, Movement Disorders Society Unified Parkinson’s Disease Rating Scale part III; MMSE, Mini Mental State Examination; MoCA, Montreal Cognitive Assessment; n.a., not applicable; NFOG-Q, New Freezing of Gait Questionnaire.
Data are presented as means (±standard deviations).
Fall frequency is reported as median (interquartile range 25%-75%).
Postural Control in Single Task and Dual-Task
Interaction Effects
Results per condition are presented in Supplementary Materials 2 and 3 (available in the online version of the article). As CoP results largely reiterated CoM findings, only the latter are reported here. Details of CoP results are provided in Supplementary Material 3 (available in the online version of the article). Three-way interaction effects were demonstrated for group × surface × task in ML direction for excursion (
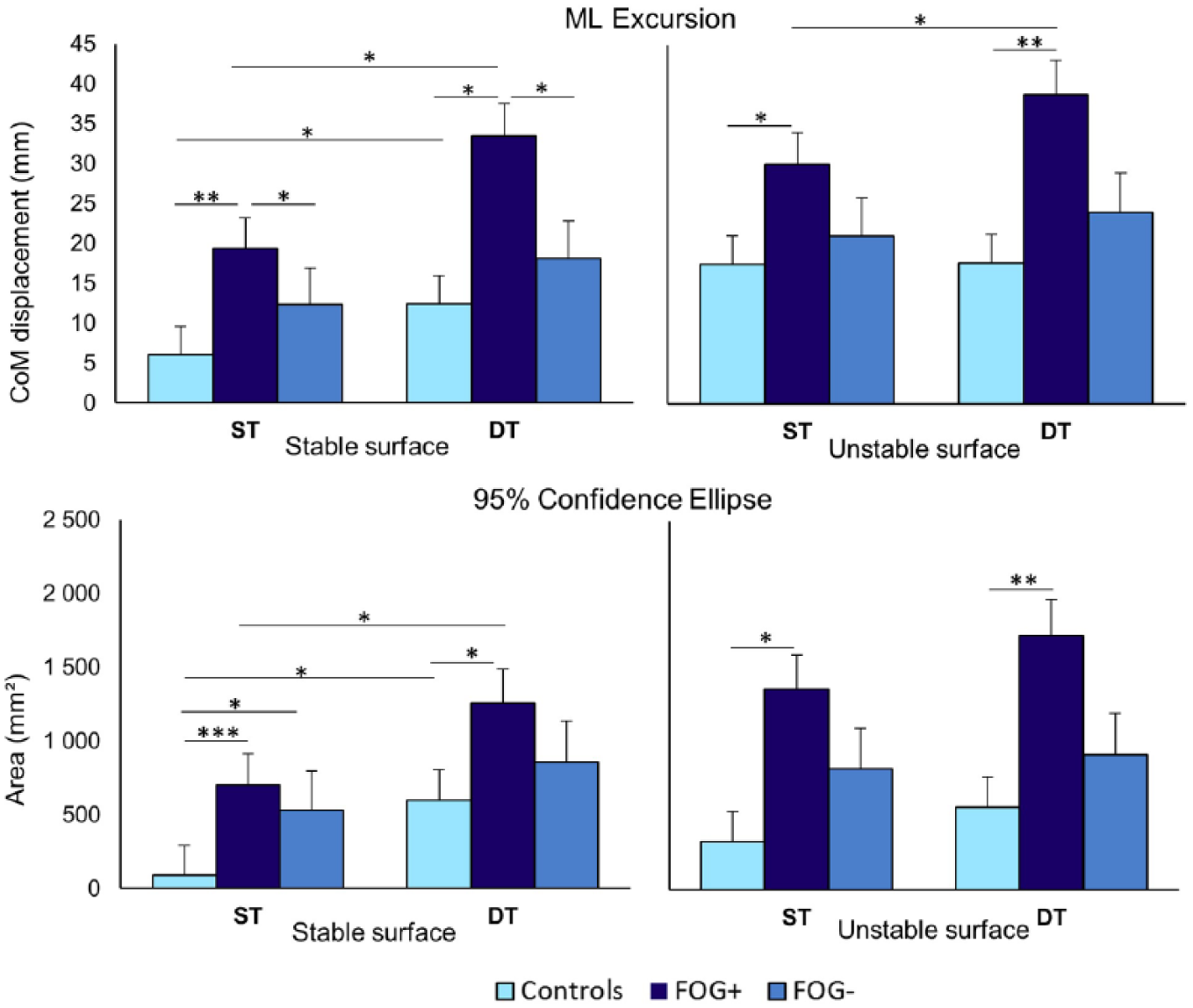
Effects of group × surface × task for center of mass (CoM) parameters. Means and standard errors are presented. Significant interaction effects were found for peak excursion in ML direction (
A significant group × task effect was found for CoM velocity (
Group × surface effects were also found for velocity (
No group × vision interaction effects were found.
Main Effects
Significant main effects of group, task, and surface were found for most postural parameters. Differences were only found in comparison with HCs, but not between FOG+ and FOG−. Compared with HCs, FOG+ exhibited higher measures for CoM confidence ellipse (
Cognitive Dual-Task Performance
The scores from the forward digit span during ST (digit span while seated) did not differ between the 3 groups (
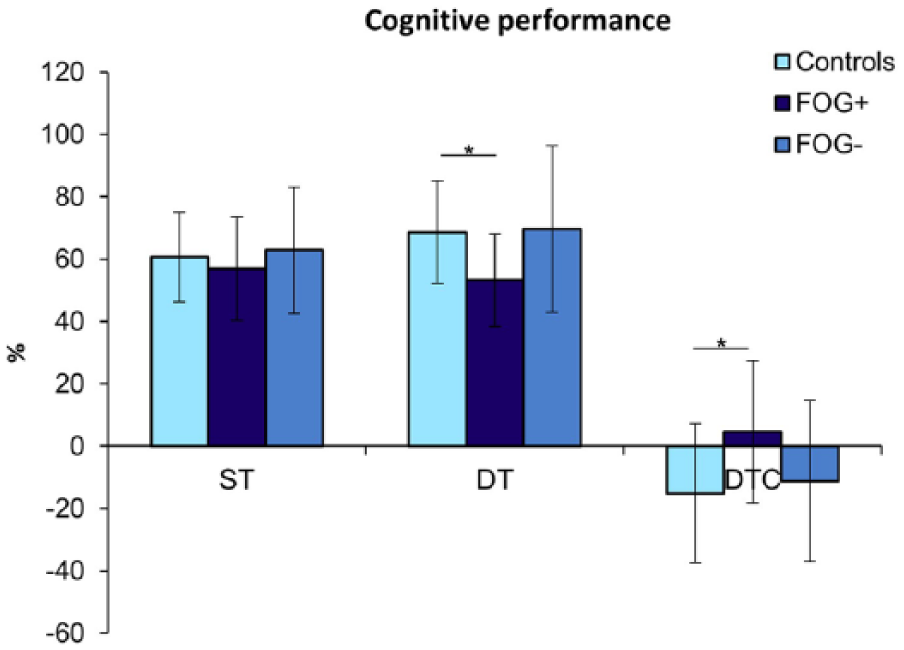
Performance on the forward digit span. Scores are expressed as an average percentage of correct scores (±standard deviation). *
Dual-Task Cost on Postural Control
The effect of dual-tasking expressed as DTC (% interference) were more prone to missing values and generated more variable results (Supplementary Material 4, available in the online version of the article). It revealed significant group differences on the 95% ellipse for CoM (
Dual-Task Cost on Digit Span Performance
The effect of dual-tasking was also expressed in the cognitive DTC (
Exploratory Correlation Analysis With Dual-Task Costs
Correlation analysis revealed that DTC were unrelated to LED in all PD patients. In contrast, MoCA scores were significantly correlated with 7 postural DTC outcomes for CoP in patients with FOG+. The influence of cognitive scores on CoP DTC was most pronounced during stable conditions (−0.5 < ρ < −0.688), signifying that lower MoCA scores were related to more DTC on postural stability. For CoP velocity (
Discussion
This study investigated whether postural control was compromised more in patients with FOG compared with those without FOG and healthy controls, when imposing additional cognitive load during several quiet stance conditions. We found that postural stability deteriorated during dual-tasking in all 3 groups, but FOG+ showed significantly more deterioration than FOG−, albeit for specific ML outcomes in stable conditions only. In addition, DTC (%) was significantly higher in FOG+ compared with FOG− in unstable conditions with visual restrictions. Overall, most differences in postural stability were found between FOG+ and HCs. As for cognitive DT performance, HCs were able to improve digit span scores during standing compared with sitting, unlike the PD subjects and particularly the FOG+ group.
Confirming our hypothesis, FOG+ showed worse postural control in DT conditions and had higher cognitive and postural DT costs compared with HCs, whereas these differences were absent between FOG− and HCs. This indicates that the FOG+ group needed more attention to maintain balance and perform a cognitive task at the same time. As our groups were well-matched for disease severity, we can ascribe these results to an association between postural control disturbances and the freezing phenomenon. Paradoxically, another study manipulating cognitive load during postural perturbations showed that freezing episodes were not affected by DT, whereas falling was. 17 Accordingly, the effect of a start-react paradigm, controlled by brainstem circuitry, was attenuated in FOG+ and not in patients who presented postural instability, suggesting that postural control and freezing are at least partially controlled by separate neural routes. 22
Between-group postural differences were mainly found in directional CoM parameters and in both CoP and CoM sway velocity and area, all of which increased to a larger extent in DT conditions in FOG+ compared with the other groups. Moreover, only in FOG+, dual-tasking affected postural stability during the more challenging unstable conditions and affected importantly on the secondary cognitive task performance. Interestingly, in FOG− and HCs an improvement of performance on the cognitive task was found in the DT relative to the ST condition. We interpret this finding as due to a learning effect, which was absent in FOG+. Although ST and DT postural conditions were randomized, the digit span task in sitting was always performed first, constituting a limitation of this study. The fact that FOG+ did not learn to remember the digits, as the other subjects did, but instead showed cognitive interference, could reflect either an implicit learning deficit and/or a reduced cognitive capacity, both of which were demonstrated in FOG+ in earlier studies.23-27 However, cognitive abilities were comparable between groups in this study, as were baseline forward digit span outcomes. Furthermore, all group analyses were statistically corrected for MoCA. Predominantly within FOG+ and to a lesser extent in FOG−, correlation analysis demonstrated that lower cognitive scores were associated with higher DT cost. Therefore, our results most likely indicate that the compensatory mechanisms to cope with integrating both tasks are insufficient in FOG+. Reduced DT capacity in stance may also point toward reduced automaticity of postural control as the underlying problem in FOG+. FOG-related exaggerated de-automaticity was also observed in studies investigating turning and walking.15,26-28 Although to a lesser extent, both FOG− and HCs also showed increased instability on DT, a result which can be attributed to basal ganglia deterioration in the first group and age-related degeneration in the latter. 13 These results were corrected for age differences, as HCs were significantly older in our population.
While we argued earlier that neural circuitry of FOG and postural control may at least be partially separate, the current findings underscore the globally altered neural profiles of FOG+ compared to FOG- found in earlier work. Altered functional and structural connectivity patterns were associated with cognitive frontal dysfunction and DT deficits during gait in FOG+ relative to FOG−.15,16,29,30 Changes in the structural connectivity of the pedunculopontine nucleus (PPN)27,30 and impaired connectivity in the frontoparietal network 16 were previously put forward to explain why gait disturbances occurred in FOG+. It is still contentious whether these differences also explain postural deficits and whether they are specific for disease phenotype or express overall disease progression.8,15
FOG+ showed greater postural instability during disturbances of sensory input9,10,31 compared to the other cohorts. Standing on foam compromises exteroceptive information from the foot soles, while proprioceptive input from ankle muscle spindles is likely to be increased. Disturbed processing of proprioceptive information was shown to be enhanced in FOG+ compared with FOG−9,32-34 and might be associated with greater dopamine depletion in the basal ganglia, 35 greater postural control deficits, and greater freezing episodes. 36 Unfortunately, proprioceptive function was not directly measured in the present study and therefore a direct link cannot be established. This lacuna needs to be addressed in future work. Interestingly and in contrast to earlier work, 12 visual restrictions did not consistently show alterations of postural instability in FOG+ versus FOG−.
Curiously, FOG+ demonstrated postural instability in the ML direction. Other studies found evidence for loss of control in the AP direction, which coincides with the tendency of FOG+ to have exaggerated forward propulsion during festination.18,31,37 However, ML weight-shifting problems and greater impairment of anticipatory postural adjustments were earlier demonstrated to be present in FOG+.38,39 In addition, medial CoM displacement was less pronounced in FOG+ prior to a freezing episode while turning. 40
Increased postural instability in FOG+, based on clinical assessment, is now a well-established finding both ON and OFF medication. 7 In the present study, scores on the Mini-BEST tended to be attenuated in FOG+ compared with FOG−, but fall frequency was significantly higher. Thus, the current results refine earlier work by showing that distinct posturographic abnormalities were found ON medication, which were direction-specific and not apparent on clinical testing, highlighting the importance of instrumented assessment.
In this study, postural control in FOG+ differed consistently from that of HCs, but not many specific differences between FOG+ and FOG− were found. DTC showed more FOG+/FOG− differences, especially for CoP. Here, we considered the DTC results less robust due to variability and greater impact of missing values. As well, CoM differences may better reflect the whole body motion captured via detailed motion tracking than CoP deviations. Other studies also showed no differences in postural DTC during quiet standing between subgroups. 18 We speculate that other components of postural control such as anticipatory postural adjustment and dynamic balance may be more affected in FOG+.18,41 The limited power may also explain our results and may have created false negative outcomes. However, the fact that we controlled for disease severity by using a matching procedure and included age and MoCA as covariates, strengthens the specificity of our findings.
As DT conditions are prevalent in daily life, our results may partly explain the higher fall frequency in FOG+. Studies on DT gait training and increasing sensory attention for gait already showed beneficial effects on postural stability and falls42-44 in PD, irrespective of FOG. Furthermore, training cognitive tasks together with highly challenging balance work proved to be effective in PD. 45 However, in a subanalysis, it was found that reduced retention of learning was apparent in FOG+ and not in FOG−. 42 This points to the need for an individualized rehabilitation approach, taking into account the freezing phenotype and designing strategies to address consolidation. 46
In this study, patients were recruited using a nonconsecutive strategy and tested in the ON medication state. Therefore, the current results do not generalize widely to the PD population and to OFF-medication conditions.10,47 As PD subjects were in a relatively mild H&Y stage, it is crucial for future studies to investigate subgroups in the more advanced disease stages. The significantly higher age in HCs could have underestimated the actual differences in postural stability and HCs and FOG+ were not matched for gender. Also the high number of missing values, mainly in DT conditions, constitutes a limitation especially for the DTC analysis. Detailed analysis showed that PD patients with missing values had a higher FOG severity and a longer disease duration. Hence, this indicates that our results may have underestimated the DTC in FOG+, warranting cautious interpretation.
Conclusion
We showed that postural control was more affected by DT interference in FOG+ than in FOG− and HCs, particularly in the ML direction and during unstable surface conditions. In addition, cognitive DT performance was positively influenced by standing upright in HCs, a benefit that was absent in FOG+. Together, we interpret these findings as that the compensatory mechanisms to cope with the demands of a simultaneously executed task during challenging standing positions are insufficient in FOG+, which may have implications for fall risks in daily life. These results support the implementation of complex motor-cognitive training to target postural instability.
Supplemental Material
Supplementary_Materials
Supplemental material, Supplemental_Materials for The Impact of Dual-Tasking on Postural Stability in People With Parkinson’s Disease With and Without Freezing of Gait by Esther M. J. Bekkers, Kim Dockx, Surendar Devan, Sam Van Rossom, Sabine M. P. Verschueren, Bastiaan R. Bloem and Alice Nieuwboer in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to thank Lova Hulst and Diede Vingerhoets for their help with data processing. We thank Mr Jean Heymans for his generous contribution and support of this study.
Supplementary material for this article is available on the
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support of this study was provided through a grant from the KU Leuven Special Research Fund and Research Foundation Flanders (FWO) (grant number G.0867.15).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
