Abstract
Background. Noninvasive electrical stimulation of the brain (ESB) is being investigated as a valued intervention to enhance motor performance. Objective. To ascertain the safety and ability of transcranial pulsed current stimulation (tPCS) to modulate variables of protective stepping and gait of individuals with Parkinson’s disease. Method. Ten patients participated in a pilot study. During the first session, a tPCS delivered current for 20 minutes via a positive electrode placed over the primary motor area (M1). In week 2, participants walked for 20 minutes on a treadmill. In week 3, tPCS and treadmill for 20 minutes were combined. Pre-testing and post-testing of gait and protective stepping were administered, comparing post-intervention (tPCS alone, treadmill alone, tPCS + treadmill) with pre-intervention data. The 3 interventions were compared by calculating the difference between post-intervention and pre-intervention data. A significance level of P < .05 was adopted. Results. Stride length increased from 102.1 ± 24.4 to 111.2 ± 22.1 cm, and gait velocity increased from 0.90 ± 0.23 to 0.985 ± 0.19 m/s after tPCS. Treadmill or treadmill + tPCS did not result in changes in the studied gait parameters. The tPCS session increased gait velocity and stride length significantly compared with treadmill or tPCS + treadmill. Overall, the number of steps needed to recover balance decreased after tPCS and tPCS + treadmill. Conclusion. Noninvasive tPCS over the primary motor cortex had no adverse effects on those with long-standing Parkinson’s disease and may lead to acute improvement of gait and balance recovery.
Introduction
People with Parkinson disease (PD) have altered sensorimotor processing and concomitant deficits in motor control of posture and gait. Disordered neural control contributes to the pathophysiology of the disease and its clinical manifestations, including bradykinesia, hypometria, rigidity, postural instability, and altered locomotion ability.1-6 Transcranial magnetic stimulation data have implicated cortical loci as not receiving, or receiving inadequate, proprioceptive input, which leads to erroneous processing and abnormal motor control. 7 Enduring deficits in standing balance and mobility are likewise documented. These deficits include diminished ability to appropriately merge the sequential component of tasks and longer delays between task initiation and execution. Typically, patients with PD show delay in step initiation and altered protective stepping responses.4,6,8-11 While walking, PD patients tend to progress at slower velocities, with shorter strides, and lower cadence. Additionally, they have increasing difficulty transitioning between sitting and standing, responding to suddenly applied external force, or walking through a constrained space, all of which contribute to increased likelihood of falling. 12
Electrical stimulation of deep brain loci, known as deep brain stimulation (DBS), has been the technological intervention of choice for improving motor control in PD with severe clinical presentation. DBS is indicated when very specific criteria are met, leaving the majority of patients with PD ineligible for DBS. 13 In contrast, advances in noninvasive electrical stimulation of the brain (ESB), in the form of transcranial direct current stimulation (tDCS) and transcranial pulsed current stimulation (tPCS), have provided novel and low-risk options minimizing impairment of the central nervous system in processing afferent input.14-16
Previous studies have addressed basic electrophysiological questions, leading to a general consensus that the effects of ESB depend on the direction of the electric current flow. Specifically, ESB may enhance cortical excitability through the positive electrode (anodal stimulation) and diminish activation under the negative electrode (cathodal stimulation). 17 The position of the electrodes over the cortical target of stimulation appears to be critical. 16 Likewise, the stimulation dosage, typically calculated as the product of stimulation intensity (current amplitude) and treatment time, is an important determinant of clinical outcome. 18 From a precautionary safety perspective, most investigators prefer to limit the current intensity of tDCS and tPCS to 2 mA (4 mA peak for tPCS).15,19 Using these current intensities and limiting stimulation sessions to 20 minutes, ESB has been demonstrated to be a very safe intervention, and only anecdotal, minor, and transient adverse reactions have been reported.15,19,20
Recent clinical investigations provide preliminary evidence that some patients with PD may benefit from ESB. In a placebo-controlled study, Fregni et al 21 tested 9 patients and used a number of electrode and polarity configurations to measure the effects of single-session tDCS on the Unified Parkinson’s Disease Rating Scale, simple reaction time, Purdue Pegboard Test, and working memory. The authors concluded that all primary outcomes improved significantly immediately after the 20-minute session of anodal stimulation over the primary motor area (M1) compared with placebo or other electrode positions. Boggio and colleagues 22 tested 18 individuals with PD who performed the 3-back letter test, a reflection of working memory, during anodal tDCS at 2 mA that targeted the left dorsolateral prefrontal cortex. During the last 5 minutes of one 20-minute session, working memory as indexed by task accuracy improved significantly compared with 1-mA amplitude or sham stimulation. In the most recent randomized, double-blind, sham-controlled clinical trial, Benninger et al 23 tested the effect of anodal tDCS, delivered via a large (97.5 cm2) electrode placed over the premotor and motor area, on gait and hand-arm movement times. Testing was carried out 24 hours after the completion of 8 sessions of tDCS, each lasting 20 minutes. Testing was repeated 1 and 3 months after the intervention. Results were mixed, favoring tDCS more when patients were “off” than “on” medication. Upper-extremity bradykinesia improved more than walking time. The authors concluded that better stimulation parameters should be investigated and established to make the intervention clinically viable.
As defined, tDCS delivers a continuous, nonmodulated constant direct current. Direct current is known to alter the electric field, particularly immediately under the electrodes. In tDCS, only 2 parameters—current amplitude (intensity) and treatment time—are amenable to change. Other stimulation parameters can be tested using tPCS,15,24 which delivers the current in the form of unidirectional or bidirectional pulses having very short duration (typically lasting a few microseconds or milliseconds) and various frequencies (typically ranging from a few to several thousand pulses per second). tPCS can both alter the electrical field and concurrently excite peripheral nerves located under the electrode. 25 Specific to the cranium, the trigeminal nerve (cranial nerve-V) can be excited. Based on determination of thresholds from peripheral nerves of the extremity, the trigeminal nerve may be depolarized if the pulse charge approximates or exceeds 0.5 to 1 µC. 26 Furthermore, because of the inverse relation between frequency and impedance, opposition to current flow during tPCS application is considerably less than during tDCS application. 14 Alon et al 14 reported recently that the effect of the tPCS on bilateral M1 connectivity may be more pronounced and less variable than tDCS. However, the effects of tPCS on people with PD have not been reported previously.
The primary purpose of this exploratory pilot investigation was to test the safety and the ability of tPCS to modulate, within 1 session, selected variables of gait performance and protective stepping for balance recovery in individuals with PD. A secondary purpose was to compare the effects of 1 session of tPCS with 1 session of treadmill walking and 1 session of combining tPCS + treadmill walking on gait and protective stepping measures. The study has been listed in ClinicalTrials.gov Record HP-00040670.
Methods
Participants
A total of 10 patients met the following inclusion criteria: diagnosis of adult onset PD, a history of freezing of gait as evidenced by clinical assessment, 27 a stable regimen of anti-Parkinsonian medications, stage 3 of the Hoehn and Yahr disability scale, ability to walk at least 10 m without assistance, ability to walk on a treadmill for 20 minutes, personal weight of less than 500 lb, and a score >24 on the Mini-Mental State Examination. Patients did not qualify if they had evidence of any clinically significant functional impairment related to cardiovascular, pulmonary, metabolic, other neurological, or musculoskeletal disease criteria that would preclude participation in training, a history of brain surgery or placement of a deep brain stimulator, any medical condition that might require other medical or surgical treatment during the study period, dyskinesia greater than grade 2 on the Unified Parkinson’s Disease Rating Scale, or any uncorrected vision or hearing problems that may limit daily activities or communication. Study-relevant personal data of enrolled participants are summarized in Table 1, and each participant signed a consent form approved by the institutional review board.
Relevant Personal Data
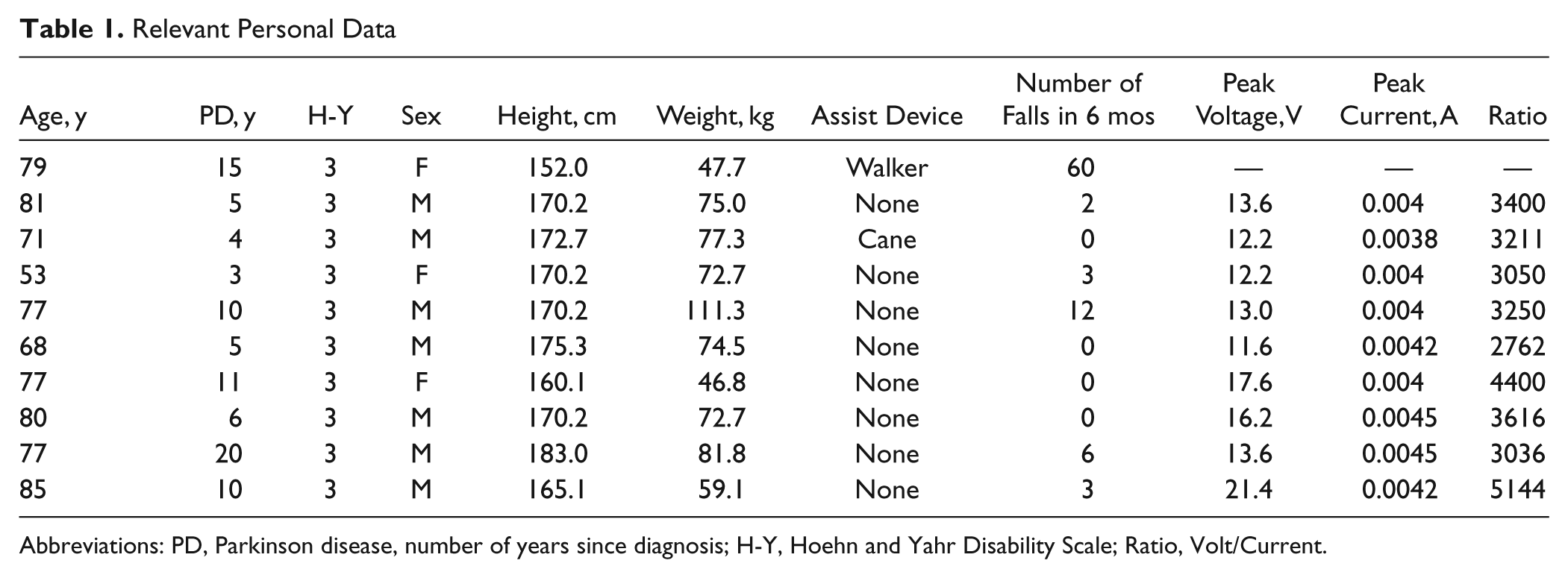
Abbreviations: PD, Parkinson disease, number of years since diagnosis; H-Y, Hoehn and Yahr Disability Scale; Ratio, Volt/Current.
Testing Procedures
Two tests, 1 to quantify gait performance and 1 to quantify protective stepping, were performed 1 to 2 minutes before and 1 to 2 minutes after the intervention during each of 3 sessions, with 1 week between sessions. To test gait performance, patients stood at 1 end of an instrumented walkway (GAITRite), and on verbal command, walked at their natural self-selected speed until they traversed the entire walkway. Data collection began automatically with the first step by means of 27 648 sensors embedded in the 10-m walkway.
Protective stepping is normally a fundamental means for recovering dynamic balance following external perturbations and has been shown to be impaired in PD. 9 To test protective stepping, the patients stood with their feet positioned comfortably using a self-selected stance width. Stepping kinematics were recorded using a motion capture system (Vicon, Los Angeles, California) that recorded the 3-dimensional motions of 2 markers each attached bilaterally over the lateral malleolus. Induced forward or backward stepping was evoked using a method described previously. 10 Briefly, a motor-driven waist-pull system controlled by custom-developed software was attached to rigid connections aligned at the level of the umbilicus, 1 in front and 1 on the back of a waist belt. A single magnitude combination of pulling displacement (13.5 cm), velocity (27 cm/s), and acceleration (540 cm/s2) was applied.
The waist-pull magnitude was selected to ensure that steps would be consistently induced and is based on our previous studies indicating that this perturbation level always triggers stepping responses. A safety harness prevented accidental falling but otherwise did not restrict movement. Participants were instructed to “react naturally to prevent themselves from falling” in response to the pulls. A “fall incident” was recorded when patients were unable to recover their balance and were “saved” by the harness. Following 1 practice trial in each direction, 10 pulling trials were performed—5 in the anterior direction and 5 in the posterior. The directions of the pulling trials were pairwise randomized with no more than 2 successive trials using the same direction (Figure 1). All stepping data were collected for 5 s. Data were sampled at 120 Hz. The beginning and end of the first step (right or left) were defined from the ankle marker of the stepping foot, providing measurement of step duration and step displacement.

Setup for testing protective stepping: custom built electromechanical robot, programmable pulling system, a 3D motion capture and analysis system (not seen), and a work station controlling, capturing, and analyzing the data are included.
Interventions
In week 1, the intervention consisted of a 20-minute session of tPCS. Patients were fitted with a thin, cushioned, thermoplastic molded cup that included two 7 × 4.5-cm (area 31.5 cm2) carbon-silicon flexible electrodes positioning the positive (anodal) electrode over the right primary motor area of the cortex (M1) and the negative (cathodal) electrode over the supraorbital area on the left side of the head, duplicating the template used in previous studies of PD21,22 (Figure 2). The electrodes and hair over the M1 were hydrated profusely with tap water to ensure good conductivity. A commercially available tPCS device (Fisher Wallace model FW 100-C, New York, New York) delivered a monophasic (unidirectional) waveform with a pulse duration of 33.3 µs and an interpulse interval of 33.3 µs. The stimulator’s carrier frequency was 15 kHz. However, having 1 ms interburst intervals decreased the number of pulses per second to 7500 pulses. These pulses were on for 50 ms, yielding 375 pulses/50 ms. Furthermore, in 1 s, there were 15 bursts of pulses, yielding an effective frequency of 5625 pulses per second (15 × 375). Peak current amplitude was set at maximum output, and the actual peak voltage and peak current delivered to each participant are included in Table 1.
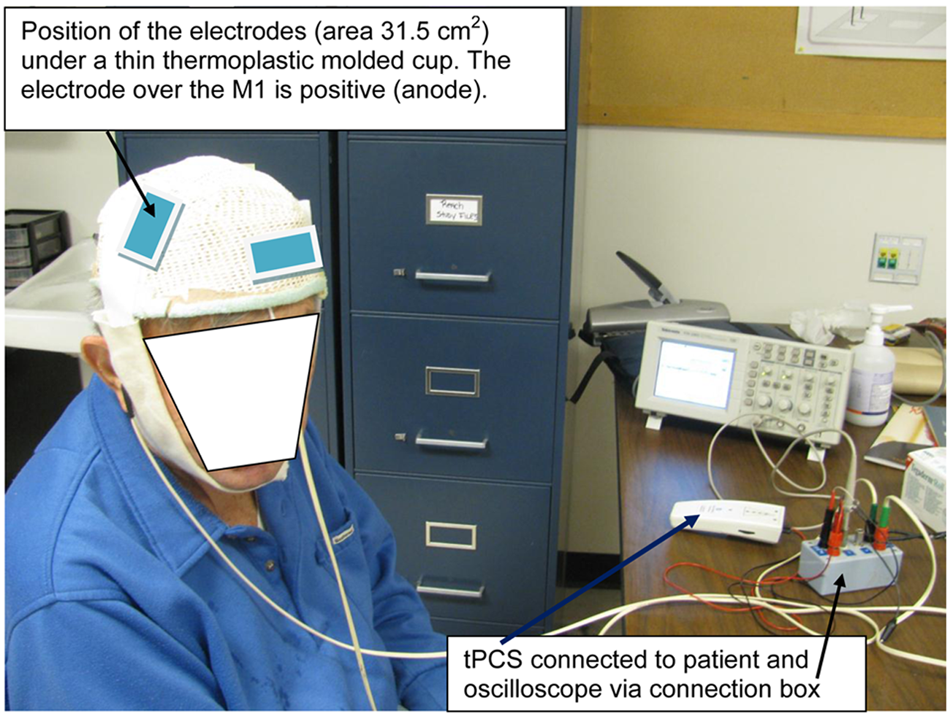
Patient setup for 20-minute session of stimulation using transcranial pulsed current stimulation (tPCS).
In week 2, the intervention was a 20-minute session walking on a treadmill. A safety harness was attached to the participant, and the treadmill speed was set at a walking velocity value determined for each patient from the overground walking speed data recorded in pre-tPCS gait testing in week 1. The investigator stood next to the treadmill ensuring the safety and the willingness of the participant to complete the 20-minute walk. Intervention in week 3 was the application of tPCS while walking on the treadmill for 20 minutes. Identical pretesting and posttesting of gait and protective stepping were administered during each of the 3 sessions.
Data Management and Analyses
All gait data were processed by the GAITRite software, generating and tabulating the study’s variables of interest: gait velocity, stride length, and cadence. For the protective stepping test, custom motion analysis software extracted the anteroposterior (A-P) length, duration, and velocity of the first step from the motion capture records. The number of steps needed to recover standing balance were counted by the investigators and confirmed using the motion capture system. For forward or backward steps, the first step distances were measured from the starting position to the maximum displacement at foot contact; step duration was the time taken to complete the first step; forward or backward displacement divided by duration determined step velocity.
Statistical Analyses
Both descriptive and inferential statistical procedures were used to characterize gait and protective stepping variables. Primary outcome measures representing gait performance included the following: (1) gait velocity, (2) stride length, and (3) cadence. Protective stepping included the following: (1) first step length, (2) first step velocity, and (3) number of steps needed to recover stable standing. Each variable was analyzed comparing postintervention with preintervention data (tPCS alone, treadmill alone, and tPCS + treadmill). Comparison of the 3 interventions was accomplished by calculating the differences between postintervention and preintervention data and applying the nonparametric Wilcoxon test to evaluate group mean differences. A significance level of P < .05 was adopted.
Results
A total of 10 patients completed all 3 sessions without detectable adverse reactions to the tPCS, treadmill, or tPCS + treadmill, with the exception that 2 patients with pretest walking speeds of 1.24 and 1.32 m/s requested a lowering of the speed during the treadmill sessions and completed the sessions successfully at 1.0 m/s. One patient tolerated only 7 minutes of treadmill walking.
Stride length increased from 102.1 ± 24.4 to 111.2 ± 22.1 cm (t = 3.87; P = .013), and gait velocity increased from 0.90 ± 0.23 to 0.985 ± 0.19 m/s (t = 3.07; P = .013) after 20 minutes of tPCS. Stimulation with tPCS did not change cadence (steps per minute) from prestimulation (106.2 ± 11.6) to poststimulation (107 ± 9.8). Walking for 20 minutes on the treadmill or combining 20 minutes of treadmill with tPCS did not result in statistically significant changes in the 3 studied gait parameters.
Comparison of the 3 interventions was done by averaging the differences in postdata minus predata and applying nonparametric Wilcoxon tests. The tPCS session significantly increased gait velocity compared with treadmill (z = −2.49; P = .012) or tPCS + treadmill (z = −2.49; P = .012). Stride length also increased significantly after tPCS compared with treadmill (z = −2.70; P = .007) or tPCS + treadmill (z = −2.60; P = .009).
The results for protective stepping are summarized in Table 2. The only significant increase, a 2.0 cm in forward step length, was found following the treadmill alone session (z = −2.29; P = .022). tPCS or tPCS + treadmill pos intervention data did not reach statistically significant changes in forward step length compared with preintervention data. The group’s mean number of steps taken to recover standing balance are shown in Figure 3. Significantly fewer steps backward were recorded only after tPCS and forward only after tPCS + treadmill. The marked interparticipant variability in response to pulling forward or backward is of interest (Table 3).
Protective Stepping First Step Length (cm) and Velocity (cm/s) a
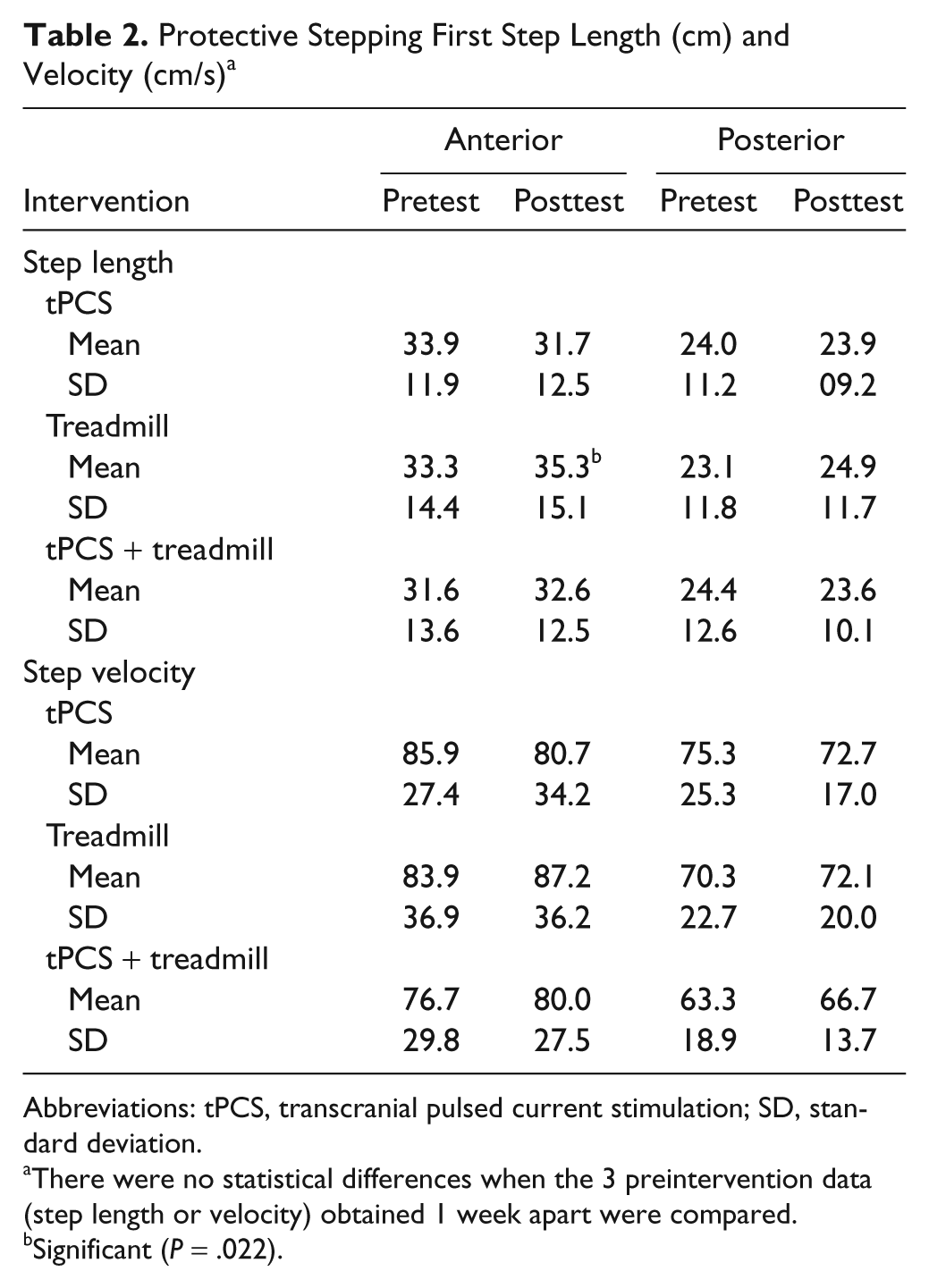
Abbreviations: tPCS, transcranial pulsed current stimulation; SD, standard deviation.
There were no statistical differences when the 3 preintervention data (step length or velocity) obtained 1 week apart were compared.
Significant (P = .022).
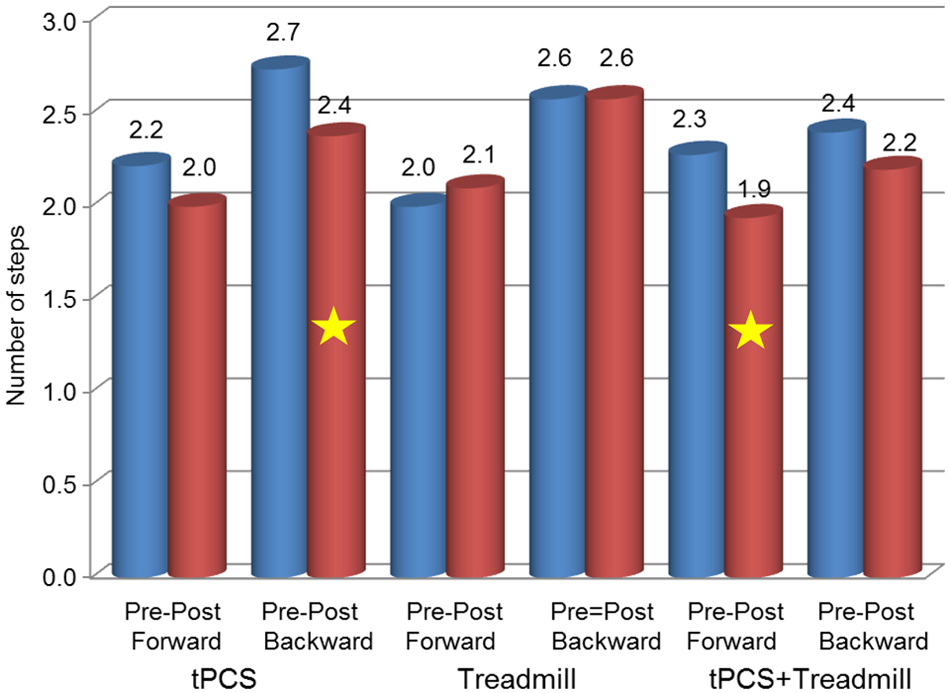
Group data of number of steps taken in response to perturbation. *Significantly fewer steps backward following transcranial pulsed current stimulation (tPCS; P = .046) and fewer steps forward following tPCS + treadmill (P = .035) were recorded. All other pre-post comparisons were not statistically significant.
Interparticipant Variability of First-Step Length and Step Velocity in Response to Forward or Backward Perturbation Following Each Intervention a
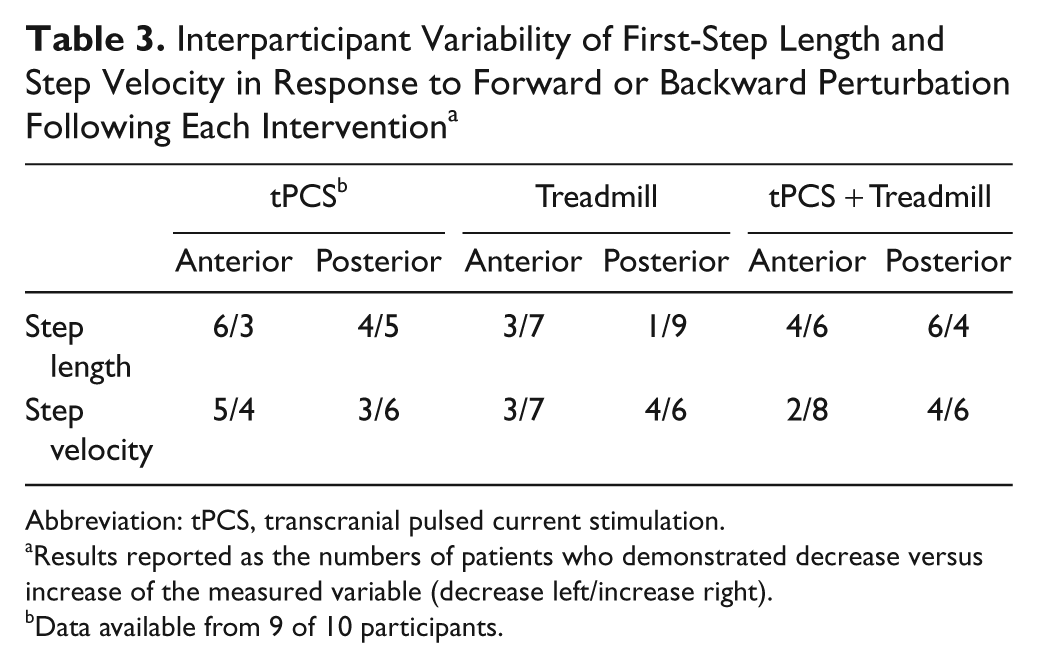
Abbreviation: tPCS, transcranial pulsed current stimulation.
Results reported as the numbers of patients who demonstrated decrease versus increase of the measured variable (decrease left/increase right).
Data available from 9 of 10 participants.
Discussion
Three prior studies reported favorable outcomes of selected measures of motor performance following noninvasive ESB of people with PD.21-23 Common to all the studies was the use of at least 1 session of tDCS, applying the positive electrode over the primary motor cortex (M1) and using 2 mA of direct current, a safe intensity, typically perceived by the patients as a mild itching/tingling sensation.21-23 To our knowledge, the present study is possibly the first to apply monophasic pulsed current (tPCS) over M1 of patients with long-standing PD. Therefore, establishing the protocol’s safety was a critical prerequisite for future investigation. The fact that all 10 participants tolerated the maximum stimulation intensity by reporting only minimal tingling without noticeable adverse responses during and between sessions is a positive finding. Furthermore, testing the effects of a 20-minute session of tPCS or treadmill walking on objective, parametric gait variables and protective stepping of people with PD is also apparently a new idea. Accordingly, comparisons with similar previous studies are difficult to make.
Whereas all participants were rated as stage 3 on the Hoehn and Yahr disability scale, their physical ability and degree of freezing varied greatly. 27 Two were very sedentary—1 for fear of falling despite having no history of falls and the other using a front wheel walker because of a very high incidence of falls; yet this patient was able to walk in the laboratory and on the treadmill independently. Two participants were physically very active prior to and during the study, and the physical ability of the remaining 6 participants ranged between these 2 extremes. As a result, interpretation of the study findings is offered with caution, particularly in recognition of the potential for a discordance between measured postural instability and absence of clinical symptoms in PD. 28 Moreover, the absence of a placebo tPCS group permits only a tentative discussion regarding the study’s findings.
The measured improvement of 9.1 cm in stride length and 0.085 m/s in gait velocity following 20 minutes of tPCS may indicate support for the proposed mechanism of upregulating the motor drive using positive (anodal) electric charge. Delivering the charge can be done using direct current29,30 or monophasic pulsed current, as was done in this study and 2 case series studies on children with cerebral palsy.15,24 One previous report has indicated improved fast walking speed in PD patients “off” medications but not “on” medications after 8 sessions of tDCS. 27 In contrast, our data showing improved gait velocity and stride length following tPCS while “on” medications, reflecting the immediate effect of 1 session. Accordingly, the question of whether tPCS and tDCS have the same or different effects on enhancing cortical motor drive during gait remains to be answered by future studies.
The findings that 20 minutes of treadmill walking or combined treadmill walking with tPCS had no measurable effects on stride length or gait velocity are somewhat difficult to interpret. Fisher et al 31 reported that a group of patients with PD who completed high-intensity treadmill training increased their gait speed and step and stride length, but improvements in gait measures were not consistently observed in low- and zero-intensity groups. It is conceivable that walking at a self-selected speed for 20 minutes, as done in our pilot study, is not sufficient to increase the motor drive, particularly in sedentary patients. 32 In addition, as indicated in Table 3, responses to sudden perturbations, an objective measure of protective stepping, varied considerably among patients. This diversity of locomotion ability and the relatively small sample size may also account for our results.
Information about the influence of noninvasive electrical stimulation on the control of balance more generally and with PD in particular was difficult to locate in the available literature. The observed reduction in the number of steps used to stabilize balance during backward postural perturbations after tPCS and in the forward direction after tPCS + treadmill may indicate that balance performance was acutely improved. These results may be indicative of the potential for tPCS to be used to prime the central nervous system as a component of balance and gait training interventions. In previous studies of protective stepping in healthy older individuals and in Parkinsonian patients, an increased number of balance recovery steps have been associated with an increased risk of falling.9,33 A reduction in the length of the first balance recovery step following perturbations of standing also distinguishes the risk of falls among older adults.9,33,34 The increase in first step length observed immediately following treadmill walking in our study would appear to indicate that this could be another way to affect the control of balance in PD, 8 possibly in conjunction with tPCS.
The limitations of this pilot study include the absence of a placebo tPCS, a necessary testing arm to delineate the contribution of placebo to behavioral improvement. Moreover, applying the stimulation to only 1 region of interest (right M1) knowing that PD involves several regions of the brain is an obvious shortcoming. The nonrandom order of intervention may have been a confounding factor, but the need to establish the safety of tPCS was a higher priority in this pilot study. Another limitation was the use of tPCS for one 20-minute session, thus restricting the likely benefit of repeated sessions and more long-lasting effects.16,21-23,35 These shortcomings can be used to direct further studies aimed at determining the critical stimulation sites, parameters of tPCS, and dosages of stimulation required to maximize treatment effectiveness. Moreover, recent technological advances permitting application of tPCS during magnetic resonance imaging 14 provide new opportunities for delineating how tPCS may help ameliorate the pathophysiological processes that underlie the deficits in gait and balance and other motor functions of patients with PD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was supported in part by a grant from Fisher Wallace Laboratories, 515 Madison Avenue, New York, NY 10022.
